Advances in Nanotechnology for Restorative Dentistry
Abstract
:1. Introduction
2. Why Need Nanomaterials in Dentistry?
| Option | Description |
|---|---|
| Material Synthesis | Producing synthetic materials matching morphology and properties similar to natural dental tissues. |
| Biomimetic Approaches | To replace lost dental tissues follow the nature’s principles and producing biomaterials resembling their properties very closely to the replacing tissues. |
| Tissue Engineering | Use of regenerative medicine and tissue engineering approaches for replacing the lost dental tissues by regenerations. |
3. Approaches in Nanodentistry
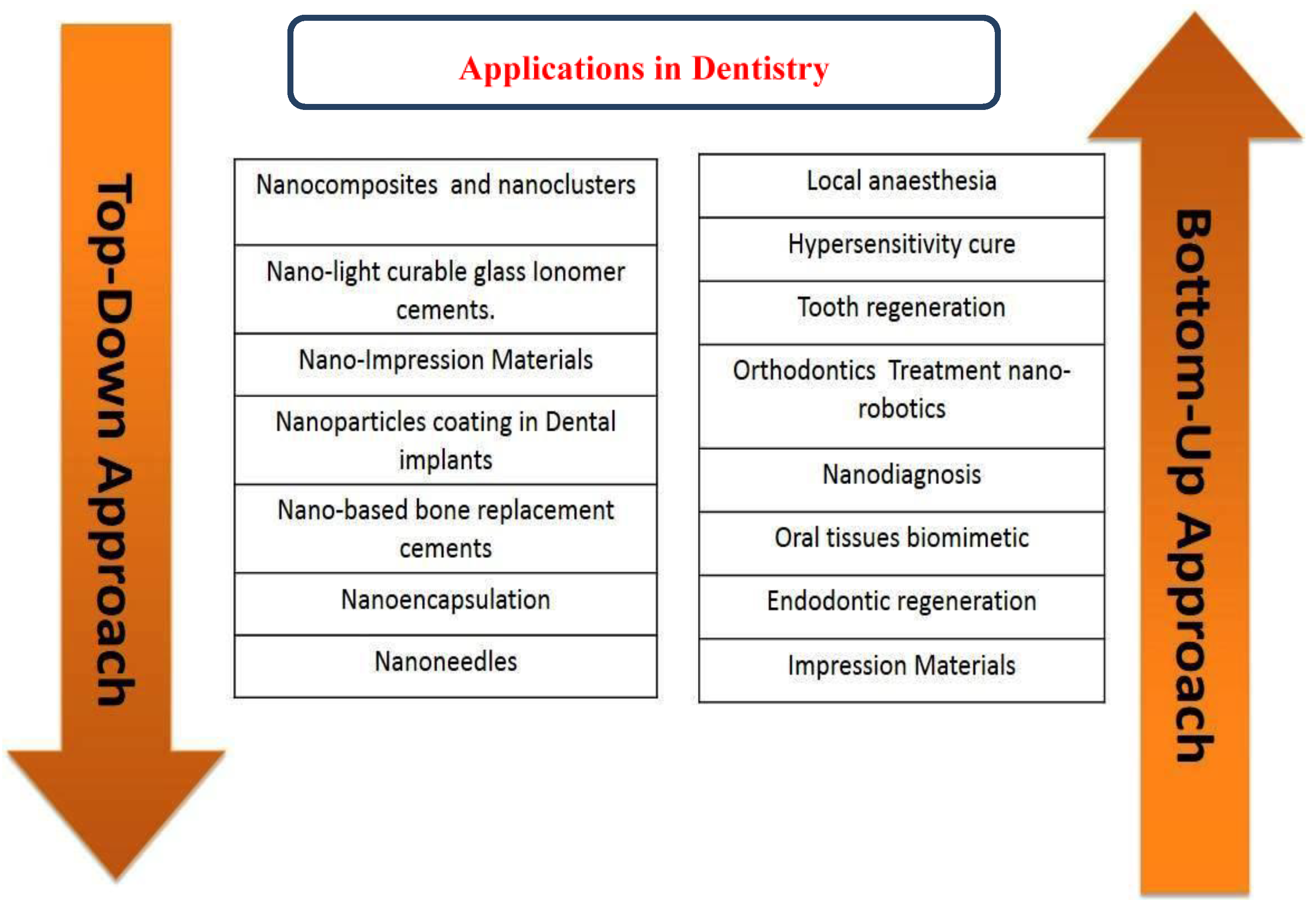
4. Applications in Dentistry
| Discipline | Available Materials |
|---|---|
| Restorative Dentistry | Ketac™ (3M ESPE, St. Paul, MN, USA), Ketac N100; Nano-ionomers (3M ESPE), Filtek Supreme XT (3M ESPE), Fuji IX GP (GC, Leuven, Belgium), Nano-primer, Premise™ (Kerr/Sybron, Orange, CA, USA), Adper™ Single bond plus Adhesive (3M ESPE), Ceram X™ (DENTSPLY International, Milford, CT, USA). |
| Regenerative Dentistry and Tissue Engineering | Ostim® (Osartis GmbH, Elsenfeld, Germany), VITOSSO™ (Orthovita-Inc, Malvern, PA, USA), Nano-Bone® (ARTOSS, Rostock, Germany). |
| Periodontics | Arestin® (Valeant, Bridgewater, MA, USA), Nanogen® (Orthogen, Springfield, IL, USA). |
| Preventive Dentistry | NanoCare® Gold (Nano-Care, Saarwellingen, Germany). |
| Orthodontics | Ketac™ N100 Light Curing Nano-Ionomers (3M ESPE), Filtek Supreme Plus Universal (3M ESPE). |
| Prosthodontics | Nanotech elite H-D plus (Zhermack, Badia Polesine, Italy), GC OPTIGLAZE color® (GC). |
| Oral Implantology | Nanotite™ Nano-coated implant (BIOMET 3i, Palm Beach Gardens, FL, USA). |
| Endodontic | AH plus™ (DENTSPLY International), Epiphany (Pentron Clinical Technologies, Wallingford, CT, USA), Guttaflow® (Coltène, Altstätten, Switzerland). |
5. Nanomaterials for Restorative Dentistry
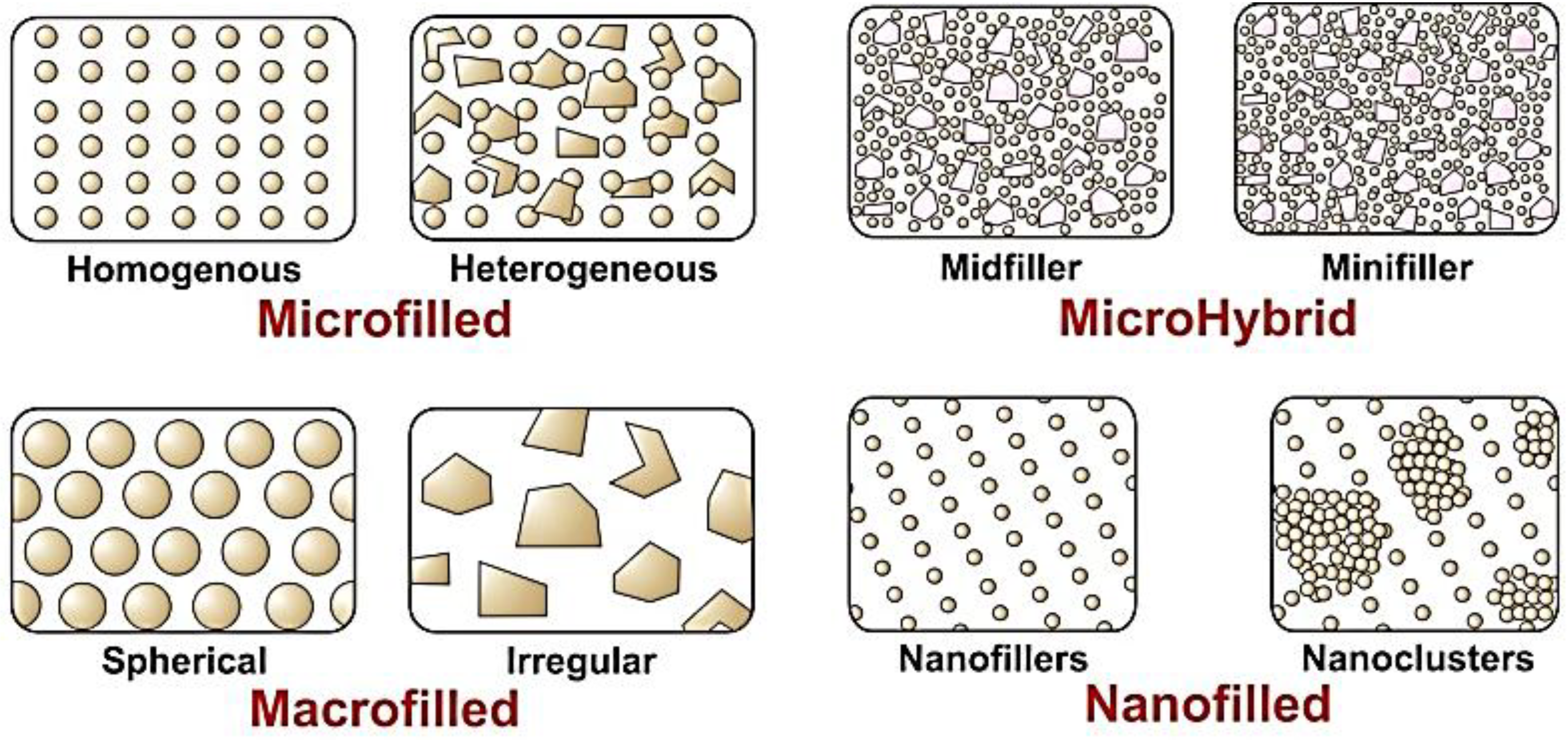
5.1. Nanocomposites
5.1.1. Composition
5.1.2. Evolution of Organic Resin Matrix
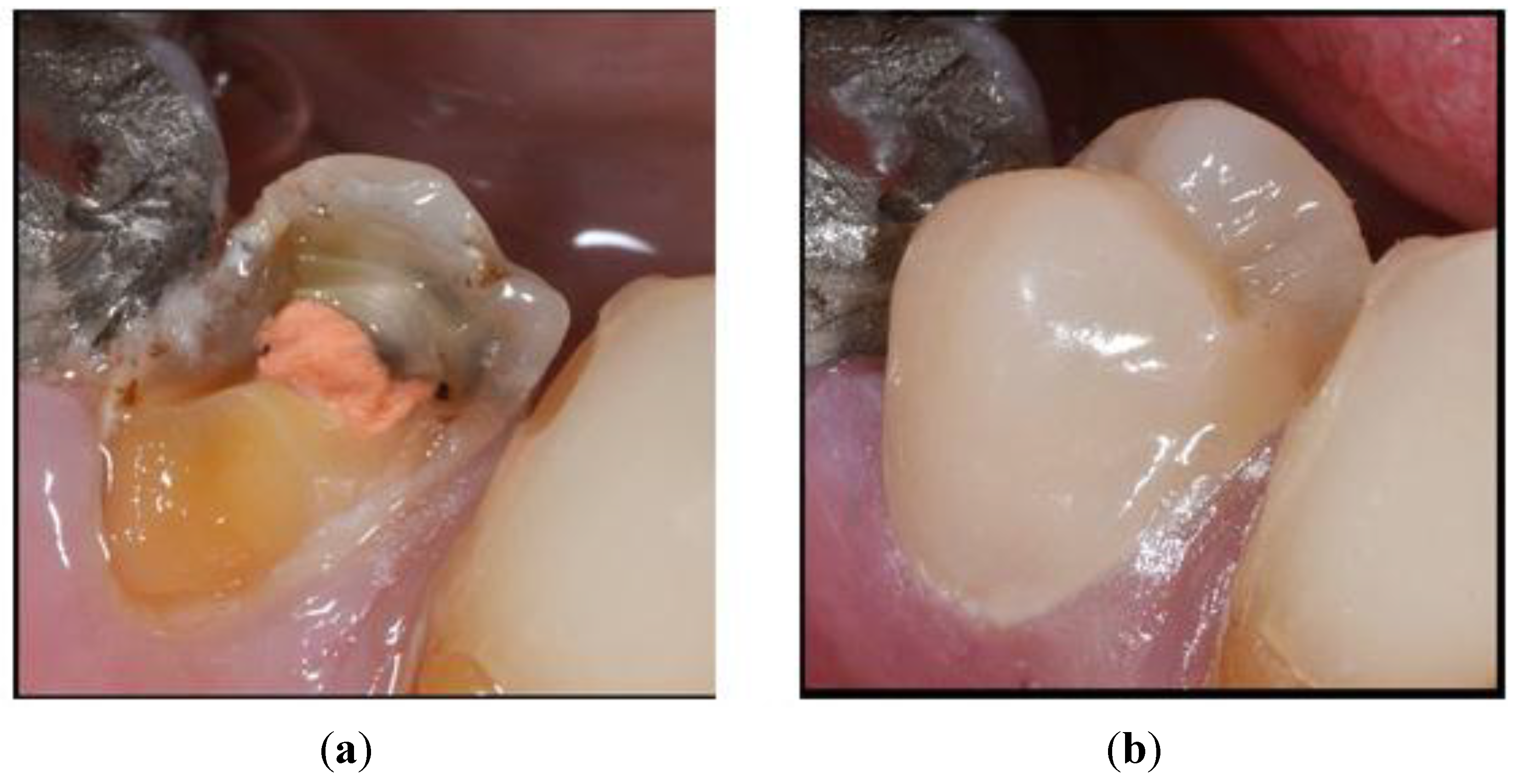
5.1.3. A Paradigm Shift of Nano Fillers to Clusters to Hybrids
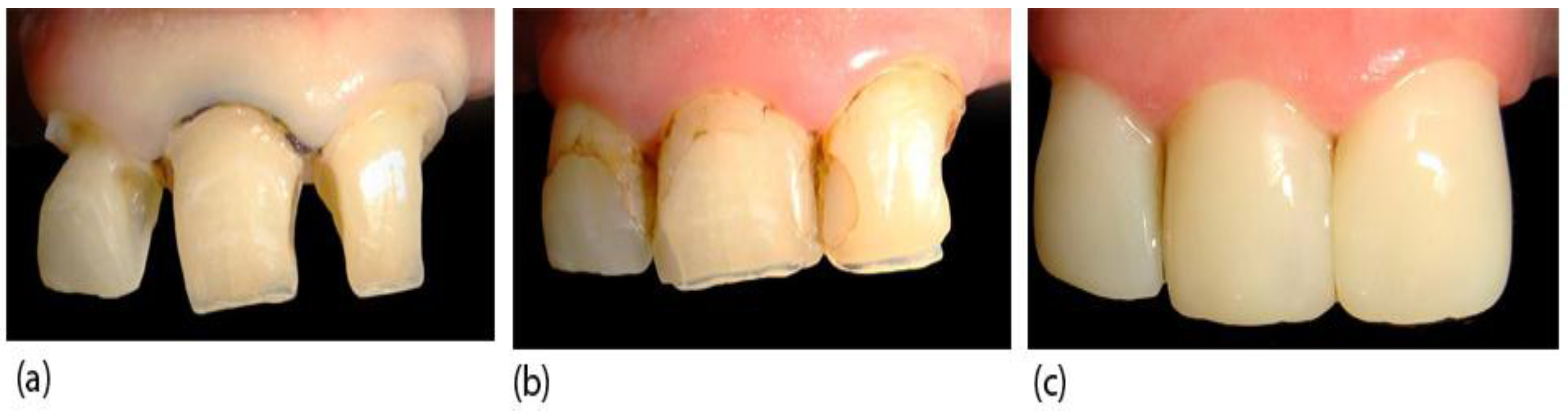
5.1.4. Coupling Agents
5.2. Nano Glass Ionomers (Nano-Ionomers)
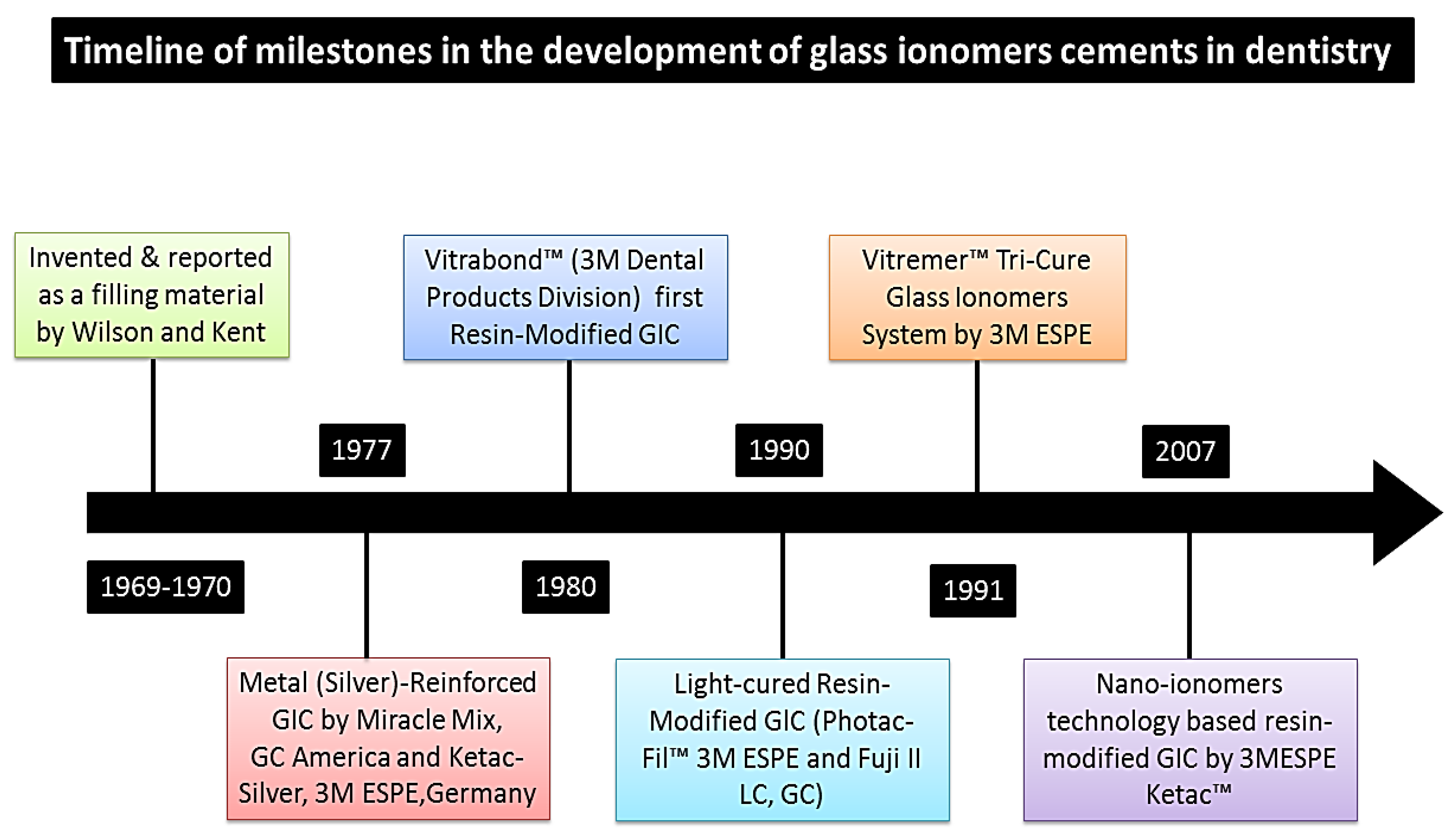
5.3. Endodontic Sealer
5.4. Nanomaterials for Dental Tissue Regeneration
6. Conclusions
Acknowledgments
Conflicts of Interest
References
- Mnyusiwalla, A.; Daar, A.S.; Singer, P.A. “Mind the gap”: Science and ethics in nanotechnology. Nanotechnology 2003, 14, R9–R13. [Google Scholar] [CrossRef]
- Roco, M.C. Nanotechnology: Convergence with modern biology and medicine. Curr. Opin. Biotechnol. 2003, 14, 337–346. [Google Scholar] [CrossRef] [PubMed]
- Silva, G.A. Introduction to nanotechnology and its applications to medicine. Surg. Neurol. 2004, 61, 216–220. [Google Scholar] [CrossRef] [PubMed]
- Koruga, D.; Hameroff, S.; Withers, J.; Loutfy, R.; Sundareshan, M. Fullerene C60: History, Physics, Nanobiology, Nanotechnology; Elsevier: Amsterdam, The Netherlands, 1993. [Google Scholar]
- Baker, J.R., Jr. Nanotechnology and medicine. J. Oral Maxillofac. Surg. 2007, 65, 27. [Google Scholar] [CrossRef]
- Ferrari, M. Cancer nanotechnology: Opportunities and challenges. Nat. Rev. Cancer 2005, 5, 161–171. [Google Scholar] [CrossRef] [PubMed]
- Mitra, S.B.; Wu, D.; Holmes, B.N. An application of nanotechnology in advanced dental materials. JADA 2003, 134, 1382–1390. [Google Scholar] [PubMed]
- Eley, B.M. The future of dental amalgam: A review of the literature. 2. Mercury exposure in dental practice. Br. Dent. J. 1997, 182, 293–297. [Google Scholar] [CrossRef] [PubMed]
- Eley, B.M. The future of dental amalgam: A review of the literature. 4. Mercury exposure hazards and risk assessment. Br. Dent. J. 1997, 182, 373–381. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.W. A Canadian perspective on the dental amalgam issue. Br. Dent. J. 1998, 184, 581–586. [Google Scholar] [CrossRef] [PubMed]
- Warfvinge, K. Mercury exposure of a female dentist before pregnancy. Br. Dent. J. 1995, 178, 149–152. [Google Scholar] [CrossRef] [PubMed]
- Smart, E.R.; Macleod, R.I.; Lawrence, C.M. Resolution of lichen-planus following removal of amalgam restorations in patients with proven allergy to mercury salts—a pilot-study. Br. Dent. J. 1995, 178, 108–112. [Google Scholar] [CrossRef] [PubMed]
- Eley, B.M. The future of dental amalgam: A review of the literature. 7. Possible alternative materials to amalgam for the restoration of posterior teeth. Br. Dent. J. 1997, 183, 11–14. [Google Scholar] [CrossRef] [PubMed]
- Mclean, J.W. Alternatives to Amalgam Alloys: 1. Br. Dent. J. 1984, 157, 432–433. [Google Scholar] [CrossRef] [PubMed]
- Yardley, R.M. Alternatives to Amalgam Alloys: 2. Br. Dent. J. 1984, 157, 434–435. [Google Scholar] [CrossRef] [PubMed]
- Saunders, S.A. Current practicality of nanotechnology in dentistry. Part 1: Focus on nanocomposite restoratives and biomimetics. Clin. Cosmet. Investig. Dent. 2009, 1, 47–61. [Google Scholar] [CrossRef] [PubMed]
- Kanaparthy, R.; Kanaparthy, A. The changing face of dentistry: Nanotechnology. Int. J. Nanomed. 2011, 6, 2799–2804. [Google Scholar] [CrossRef]
- Shekaran, A.; Garcia, A.J. Nanoscale engineering of extracellular matrix-mimetic bioadhesive surfaces and implants for tissue engineering. Biochim. Biophys. Acta BBA Gen. Subj. 2011, 1810, 350–360. [Google Scholar] [CrossRef]
- Chandki, R.; Kala, M.; Kumar, K.N.; Brigit, B.; Banthia, P.; Banthia, R. “Nanodentistry”: Exploring the beauty of miniature. J. Clin. Exp. Dent. 2012, 4, e119. [Google Scholar] [CrossRef] [PubMed]
- Gaiser, S.; Deyhle, H.; Bunk, O.; White, S.N.; Müller, B. Understanding nano-anatomy of healthy and carious human teeth: A prerequisite for nanodentistry. Biointerphases 2012, 7, 4. [Google Scholar] [CrossRef] [PubMed]
- Armentano, I.; Arciola, C.R.; Fortunati, E.; Ferrari, D.; Mattioli, S.; Amoroso, C.F.; Kenny, J.M.; Imbriani, M.; Visai, L. The interaction of bacteria with engineered nanostructured polymeric materials: A review. Sci. World J. 2014, 2014, 1–18. [Google Scholar] [CrossRef]
- Seo, S.; Mahapatra, C.; Singh, R.K.; Knowles, J.C.; Kim, H. Strategies for osteochondral repair: Focus on scaffolds. J. Tissue Eng. 2014, 5, 1–14. [Google Scholar] [CrossRef]
- Nanci, A. Ten Cate’s Oral Histology: Development, Structure, and Function; Mosby: St. Louis, MO, USA; London, UK, 2012. [Google Scholar]
- Zafar, M.S.; Ahmed, N. Nano-mechanical evaluation of dental hard tissues using indentation technique. World Appl. Sci. J. 2013, 28, 1393–1399. [Google Scholar]
- Zafar, M.S.; Ahmed, N. Nanomechanical characterization of exfoliated and retained deciduous incisors. Technol. Health Care 2014, 22, 785–793. [Google Scholar] [PubMed]
- Moszner, N.; Salz, U. Recent developments of new components for dental adhesives and composites. Macromol. Mater. Eng. 2007, 292, 245–271. [Google Scholar] [CrossRef]
- Subramani, K.; Ahmed, W. Emerging Nanotechnologies in Dentistry: Processes, Materials and Applications; William Andrew: Amsterdam, The Netherlands, 2011. [Google Scholar]
- Mikkilineni, M.; Rao, A.; Tummala, M.; Elkanti, S. Nanodentistry: New buzz in dentistry. Eur. J. Gen. Dent. 2013, 2, 109. [Google Scholar] [CrossRef]
- Zhang, L.; Webster, T.J. Nanotechnology and nanomaterials: Promises for improved tissue regeneration. Nano Today 2009, 4, 66–80. [Google Scholar] [CrossRef]
- Roszek, B.; De Jong, W.; Geertsma, R. Nanotechnology in Medical Applications: State-of-the-Art in Materials and Devices; Rijksinstituut voor Volksgezondheid en Milieu RIVM: Bilthoven, The Netherlands, 2005. [Google Scholar]
- Wickson, F. Narratives of nature and nanotechnology. Nat. Nanotechnol. 2008, 3, 313–315. [Google Scholar] [CrossRef] [PubMed]
- Dresch, W.; Volpato, S.; Gomes, J.; Ribeiro, N.; Reis, A.; Loguercio, A. Clinical evaluation of a nanofilled composite in posterior teeth: 12-month results. Oper. Dent. 2006, 31, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Jandt, K.D.; Sigusch, B.W. Future perspectives of resin-based dental materials. Dent. Mater. 2009, 25, 1001–1006. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.S.; Aamer, S.; Chaudhry, A.A.; Wong, F.S.; Rehman, I.U. Synthesis and characterizations of a fluoride-releasing dental restorative material. Mater. Sci. Eng. C 2013, 33, 3458–3464. [Google Scholar] [CrossRef]
- Mitra, S.B.; Oxman, J.D.; Falsafi, A.; Ton, T.T. Fluoride release and recharge behavior of a nano-filled resin-modified glass ionomer compared with that of other fluoride releasing materials. Am. J. Dent. 2011, 24, 372–378. [Google Scholar] [PubMed]
- Ozak, S.T.; Ozkan, P. Nanotechnology and dentistry. Eur. J. Dent. 2013, 7, 145–151. [Google Scholar] [PubMed]
- Chen, M. Update on dental nanocomposites. J. Dent. Res. 2010, 89, 549–560. [Google Scholar] [CrossRef] [PubMed]
- Ben-Nissan, B.; Choi, A.H. Sol-gel production of bioactive nanocoatings for medical applications. Part 1: An introduction. Nanomedicine 2006, 1, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Choi, A.H.; Ben-Nissan, B.; Matinlinna, J.P.; Conway, R.C. Current perspectives: Calcium phosphate nanocoatings and nanocomposite coatings in dentistry. J. Dent. Res. 2013, 92, 853–859. [Google Scholar] [CrossRef] [PubMed]
- Choi, C.; Kim, Y. A study of the correlation between organic matrices and nanocomposite materials in oyster shell formation. Biomaterials 2000, 21, 213–222. [Google Scholar] [CrossRef] [PubMed]
- Kaur, P. Nanocomposites-A step towards improved restorative dentistry. Indian J. Dent. Sci. 2011, 3, 28. [Google Scholar]
- Cramer, N.B.; Stansbury, J.W.; Bowman, C.N. Recent advances and developments in composite dental restorative materials. J. Dent. Res. 2011, 90, 402–416. [Google Scholar] [CrossRef] [PubMed]
- Hahn, B.; Lee, J.; Park, D.; Choi, J.; Ryu, J.; Yoon, W.; Lee, B.; Shin, D.; Kim, H. Mechanical and in vitro biological performances of hydroxyapatite-carbon nanotube composite coatings deposited on Ti by aerosol deposition. Acta Biomater. 2009, 5, 3205–3214. [Google Scholar] [CrossRef] [PubMed]
- Kealley, C.; Elcombe, M.; van Riessen, A.; Ben-Nissan, B. Development of carbon nanotube-reinforced hydroxyapatite bioceramics. Phys. B Condens. Matter 2006, 385, 496–498. [Google Scholar] [CrossRef]
- Wepasnick, K.A.; Smith, B.A.; Bitter, J.L.; Fairbrother, D.H. Chemical and structural characterization of carbon nanotube surfaces. Anal. Bioanal. Chem. 2010, 396, 1003–1014. [Google Scholar] [CrossRef] [PubMed]
- Terry, D.A. Applications of nanotechnology. Pract. Proced. Aesthet. Dent. 2004, 16, 220–222. [Google Scholar] [PubMed]
- Hatrick, C.D.; Eakle, W.S.; Bird, W.F. Dental Materials: Clinical Applications for Dental Assistants and Dental Hygienists; Saunders: St. Louis, MO, USA, 2003. [Google Scholar]
- Anusavice, K.; Phillips, R. Phillips’ Science of Dental Materials; Saunders: St. Louis, MO, USA, 2003. [Google Scholar]
- Guggenberger, R.; May, R.; Stefan, K. New trends in glass-ionomer chemistry. Biomaterials 1998, 19, 479–483. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, J.W. Chemistry of glass-ionomer cements: A review. Biomaterials 1998, 19, 485–494. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, J.W.; Braybrook, J.H.; Wasson, E.A. The biocompatibility of glass-poly (alkenoate) (Glass-Ionomer) cements: A review. J. Biomater. Sci. Polym. Ed. 1991, 2, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Moshaverinia, A.; Ansari, S.; Movasaghi, Z.; Billington, R.W.; Darr, J.A.; Rehman, I.U. Modification of conventional glass-ionomer cements with vinylpyrrolidone containing polyacids, nano-hydroxy and fluoroapatite to improve mechanical properties. Dent. Mater. 2008, 24, 1381–1390. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.S.; Khan, M.; Rehman, I.U. Nanoparticles, Properties, and Applications in Glass Ionomer Cements in Nanobiomaterials in Clinical Dentistry; William Andrew Publishing: Amstrdam, The Netherlands, 2013; Chapter 5. [Google Scholar]
- Moshaverinia, A.; Ansari, S.; Moshaverinia, M.; Roohpour, N.; Darr, J.A.; Rehman, I. Effects of incorporation of hydroxyapatite and fluoroapatite nanobioceramics into conventional glass ionomer cements (GIC). Acta Biomater. 2008, 4, 432–440. [Google Scholar] [CrossRef] [PubMed]
- Uysal, T.; Yagci, A.; Uysal, B.; Akdogan, G. Are nano-composites and nano-ionomers suitable for orthodontic bracket bonding? Eur. J. Orthod. 2010, 32, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Basso, M. Teeth restoration using a high-viscosity glass ionomer cement: The Equia® system. J. Minim. Interv. Dent. 2011, 4, 74–76. [Google Scholar]
- Friedl, K.; Hiller, K.; Friedl, K. Clinical performance of a new glass ionomer based restoration system: A retrospective cohort study. Dent. Mater. 2011, 27, 1031–1037. [Google Scholar] [CrossRef] [PubMed]
- Sakaguchi, R.L.; Powers, J.M. Craig’s Restorative Dental Materials; Elsevier/Mosby: Philadelphia, PA, USA, 2012. [Google Scholar]
- McCabe, J.F.; Yan, Z.; Al Naimi, O.T.; Mahmoud, G.; Rolland, S.L. Smart materials in dentistry—Future prospects. Dent. Mater. J. 2009, 28, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Koch, K.; Brave, D. A new day has dawned: The increased use of bioceramics in endodontics. Dent. April 2009, 10, 39–43. [Google Scholar]
- Zoufan, K.; Jiang, J.; Komabayashi, T.; Wang, Y.; Safavi, K.E.; Zhu, Q. Cytotoxicity evaluation of Gutta flow and endo sequence BC sealers. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2011, 112, 657–661. [Google Scholar] [CrossRef]
- Abramovitz, I.; Beyth, N.; Weinberg, G.; Borenstein, A.; Polak, D.; Kesler-Shvero, D.; Houri-Haddad, Y. In Vitro biocompatibility of Endodontic Sealers Incorporating Antibacterial Nanoparticles. J. Nanomater. 2012, 2012, 1–9. [Google Scholar] [CrossRef]
- Beyth, N.; Houri-Haddad, Y.; Baraness-Hadar, L.; Yudovin-Farber, I.; Domb, A.J.; Weiss, E.I. Surface antimicrobial activity and biocompatibility of incorporated polyethylenimine nanoparticles. Biomaterials 2008, 29, 4157–4163. [Google Scholar] [CrossRef] [PubMed]
- Venugopal, J.; Ramakrishna, S. Applications of polymer nanofibers in biomedicine and biotechnology. Appl. Biochem. Biotechnol. 2005, 125, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Fukuda, J.; Khademhosseini, A.; Yeh, J.; Eng, G.; Cheng, J.; Farokhzad, O.C.; Langer, R. Micropatterned cell co-cultures using layer-by-layer deposition of extracellular matrix components. Biomaterials 2006, 27, 1479–1486. [Google Scholar] [CrossRef] [PubMed]
- Galler, K.M.; Cavender, A.; Yuwono, V.; Dong, H.; Shi, S.; Schmalz, G.; Hartgerink, J.D.; D’Souza, R.N. Self-assembling peptide amphiphile nanofibers as a scaffold for dental stem cells. Tissue Eng. Part A 2008, 14, 2051–2058. [Google Scholar] [CrossRef] [PubMed]
- Misawa, H.; Kobayashi, N.; Soto-Gutierrez, A.; Chen, Y.; Yoshida, A.; Rivas-Carrillo, J.D.; Navarro-Alvarez, N.; Tanaka, K.; Miki, A.; Takei, J. PuraMatrix facilitates bone regeneration in bone defects of calvaria in mice. Cell Transplant. 2006, 15, 903–910. [Google Scholar] [CrossRef] [PubMed]
- Zafar, M.S.; Al-Samadani, K.H. Potential use of natural silk for bio-dental applications. J. Taibah Univers. Med. Sci. 2014, 9, 171–177. [Google Scholar] [CrossRef]
- Demarco, F.F.; Conde, M.; Cavalcanti, B.N.; Casagrande, L.; Sakai, V.T.; Nör, J.E. Dental pulp tissue engineering. Braz. Dent. J. 2011, 22, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Wiesmann, H.; Meyer, U.; Plate, U.; Höhling, H. Aspects of collagen mineralization in hard tissue formation. Int. Rev. Cytol. 2004, 242, 121–156. [Google Scholar]
- Kim, N.R.; Lee, D.H.; Chung, P.; Yang, H. Distinct differentiation properties of human dental pulp cells on collagen, gelatin, and chitosan scaffolds. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2009, 108, e94–e100. [Google Scholar] [CrossRef]
- Mizuno, M.; Miyamoto, T.; Wada, K.; Watatani, S.; Zhang, G.X. Type I collagen regulated dentin matrix protein-1 (Dmp-1) and osteocalcin (OCN) gene expression of rat dental pulp cells. J. Cell. Biochem. 2003, 88, 1112–1119. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khurshid, Z.; Zafar, M.; Qasim, S.; Shahab, S.; Naseem, M.; AbuReqaiba, A. Advances in Nanotechnology for Restorative Dentistry. Materials 2015, 8, 717-731. https://doi.org/10.3390/ma8020717
Khurshid Z, Zafar M, Qasim S, Shahab S, Naseem M, AbuReqaiba A. Advances in Nanotechnology for Restorative Dentistry. Materials. 2015; 8(2):717-731. https://doi.org/10.3390/ma8020717
Chicago/Turabian StyleKhurshid, Zohaib, Muhammad Zafar, Saad Qasim, Sana Shahab, Mustafa Naseem, and Ammar AbuReqaiba. 2015. "Advances in Nanotechnology for Restorative Dentistry" Materials 8, no. 2: 717-731. https://doi.org/10.3390/ma8020717
APA StyleKhurshid, Z., Zafar, M., Qasim, S., Shahab, S., Naseem, M., & AbuReqaiba, A. (2015). Advances in Nanotechnology for Restorative Dentistry. Materials, 8(2), 717-731. https://doi.org/10.3390/ma8020717







