Kinetic Study on the Formation of Bimetallic Core-Shell Nanoparticles via Microemulsions
Abstract
:1. Introduction
2. Simulation Model
2.1. Reaction Media Description
2.2. Microemulsion Composition
2.3. Initial Concentration inside Droplets
2.4. Time Unit Base
2.5. Intermicellar Exchange Criterium of Isolated Species (Reactants and Metal Atoms): Intermicellar Exchange Rate
2.6. Chemical Reduction Rates
2.7. Nucleation
2.8. Nanoparticle Growth
2.9. Intermicellar Exchange Protocol of Growing Particles
2.10. Droplet Size
2.11. Describing Metal Distribution in Bimetallic Nanoparticles
3. Results and Discussion
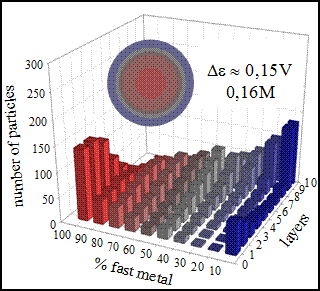
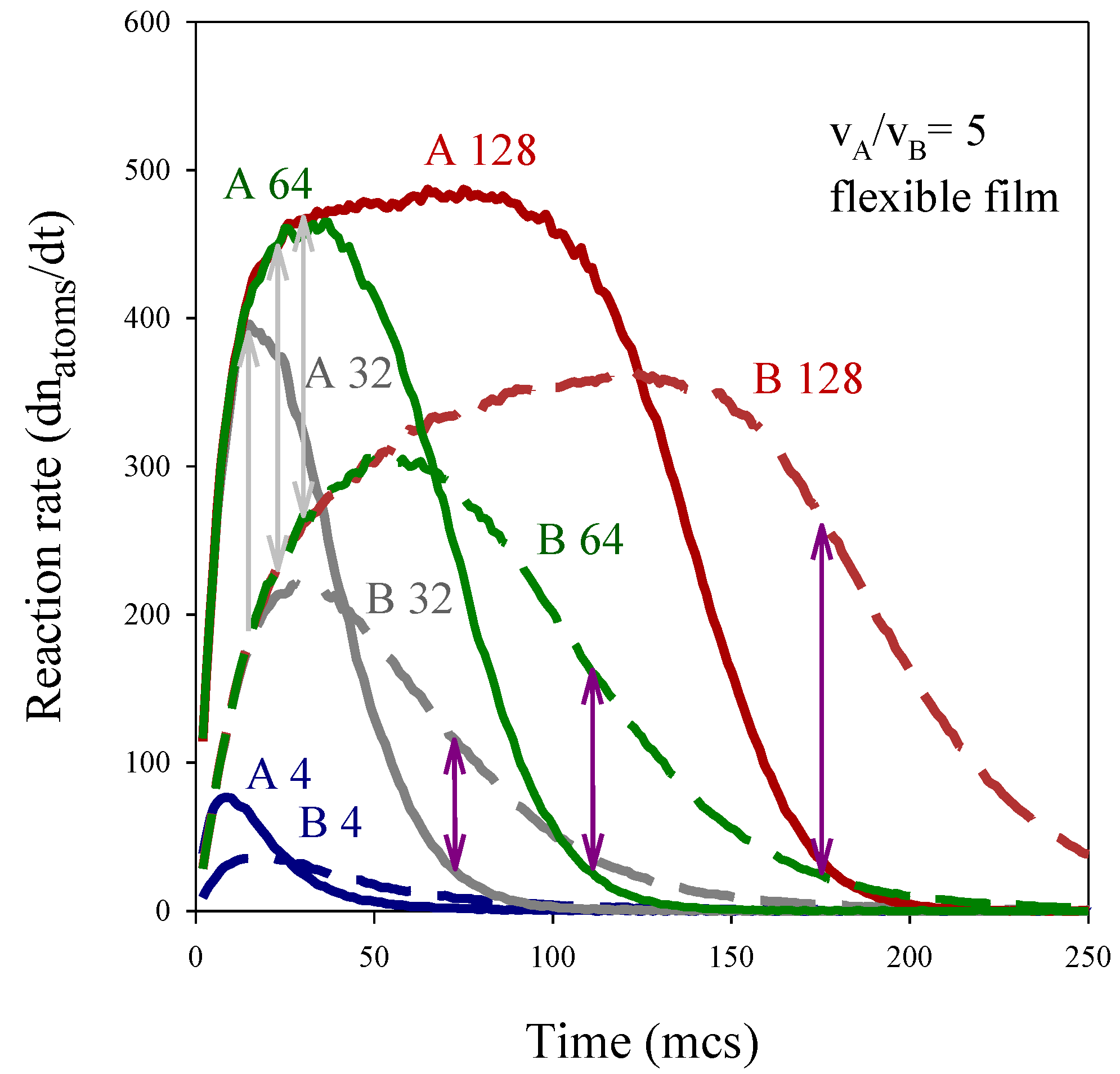
3.1. Chemical Reduction Rate Ratio
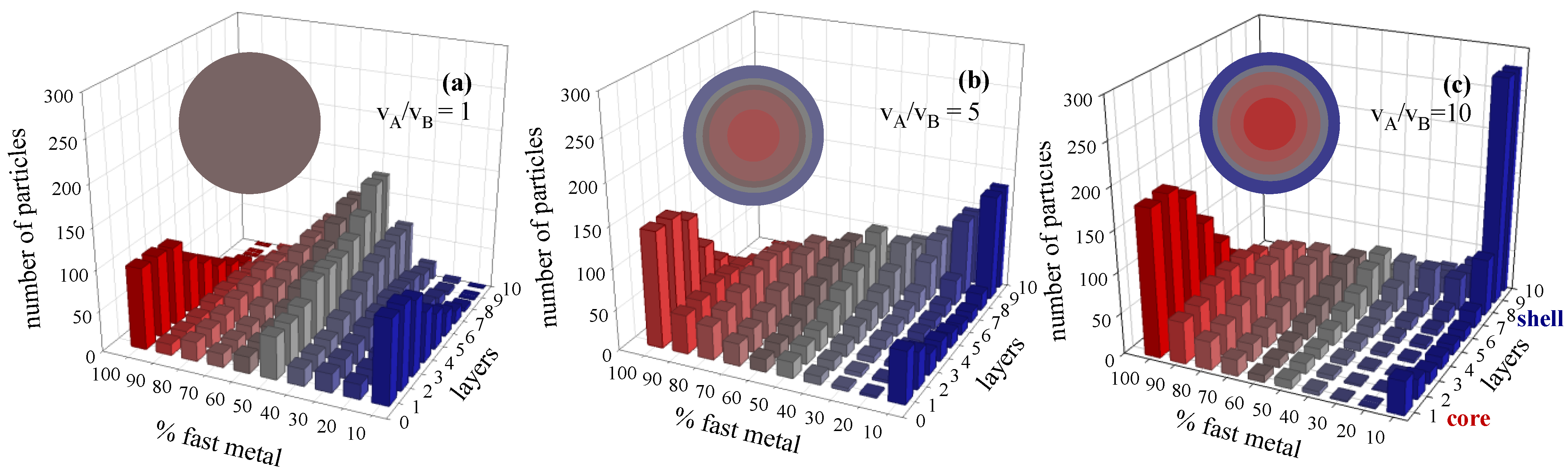
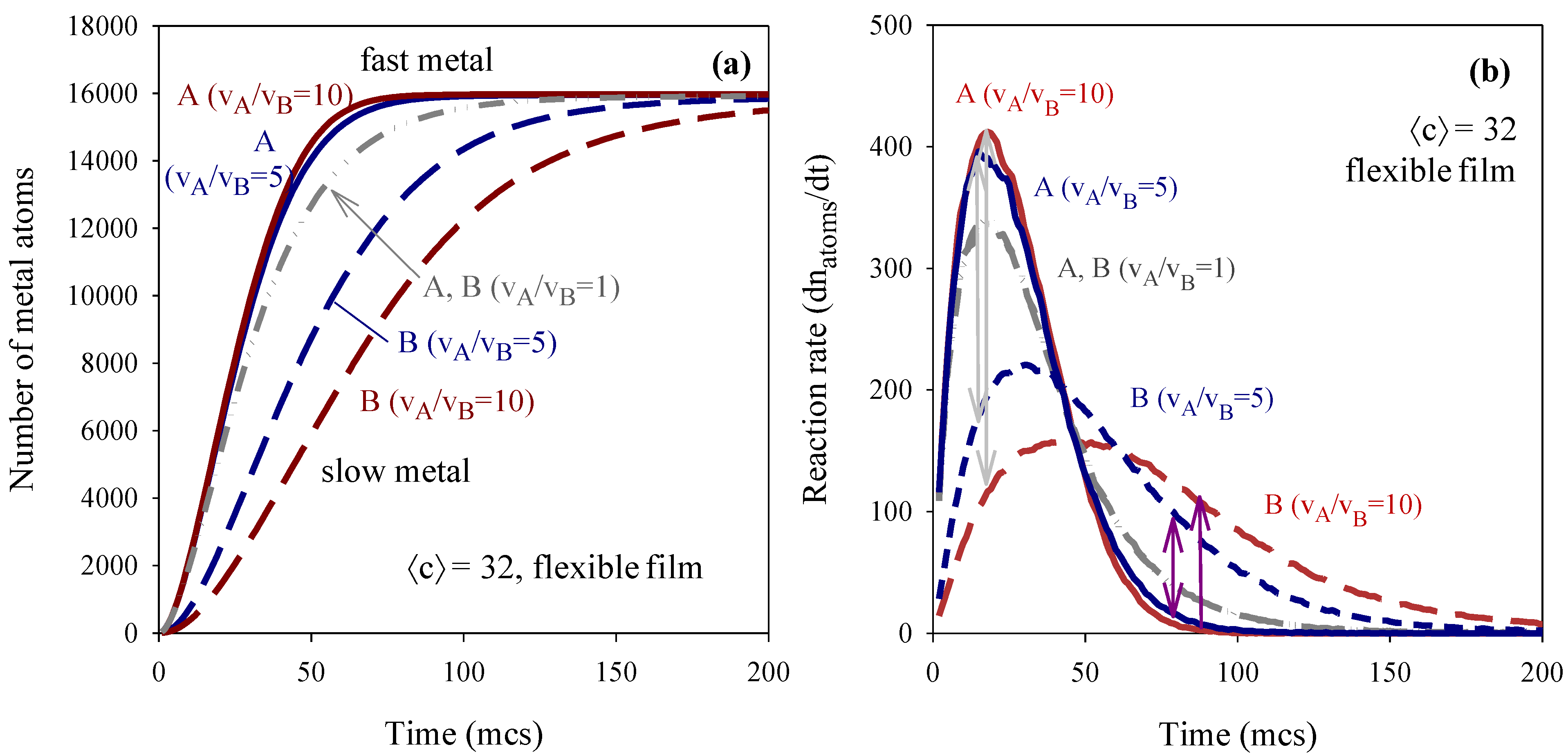
3.2. Metal Salt Concentration
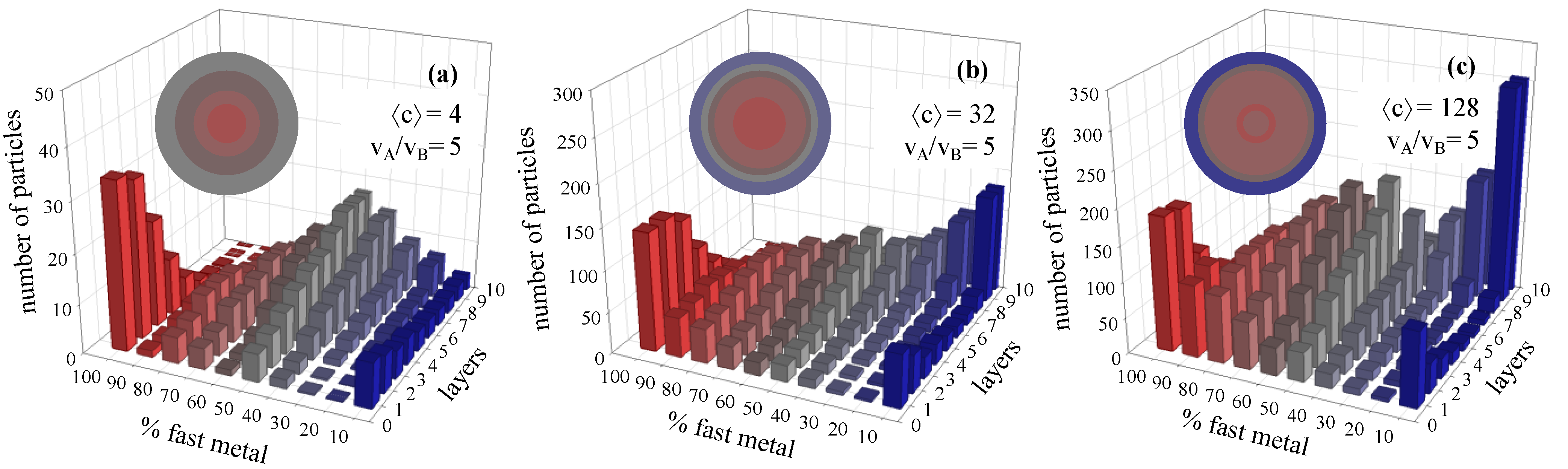
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Sinfelt, J.H. Structure of bimetallic clusters. Acc. Chem. Res. 1987, 20, 134–139. [Google Scholar] [CrossRef]
- Han, S.W.; Kim, Y.; Kim, K. Dodecanethiol-derivatized Au/Ag bimetallic nanoparticles: TEM, UV/Vis, XPS, and FTIR analysis. J. Coll. Interface Sci. 1998, 208, 272–278. [Google Scholar] [CrossRef]
- Tokonami, S.; Morita, N.; Takasaki, K.; Toshima, N. Novel synthesis, structure, and oxidation catalysis of Ag/Au bimetallic nanoparticles. J. Phys. Chem. C 2010, 114, 10336–10341. [Google Scholar] [CrossRef]
- Shibata, T.; Bunker, B.A.; Zhang, Z.; Meisel, D.; Vardeman, C.F.; Gezelter, J.D. Size-dependent spontaneous alloying of Au-Ag nanoparticles. J. Am. Chem. Soc. 2002, 124, 11989–11996. [Google Scholar] [CrossRef] [PubMed]
- Feng, L.; Gao, G.; Huang, P.; Wang, K.; Wang, X.; Luo, T.; Zhang, C. Optical properties and catalytic activity of bimetallic gold-silver nanoparticles. Nano Biomed. Eng. 2010, 2, 258–267. [Google Scholar] [CrossRef]
- Habrioux, A.; Vogel, W.; Guinel, M.; Guetaz, L.; Servat, K.; Kokoh, B.; Alonso-Vante, N. Structural and electrochemical studies of Au-Pt nanoalloys. Phys. Chem. Chem. Phys. 2009, 11, 3573–3579. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.D.; Stefano, D.; Nechache, R.; Rosei, R.; Rosei, F.; Ma, D.L. Bifunctional catalytic/magnetic Ni@Ru core-shell nanoparticles. Chem. Commun. 2011, 47, 6308–6310. [Google Scholar] [CrossRef]
- Shi, L.; Wang, A.; Zhang, T.; Zhang, B.; Su, D.; Li, H.; Song, Y. One-step synthesis of Au-Pd alloy nanodendrites and their catalytic activity. J. Phys. Chem. C 2013, 117, 12526–12536. [Google Scholar] [CrossRef]
- Fan, F.R.; Liu, D.Y.; Wu, Y.F.; Duan, S.; Xie, Z.X.; Jiang, Z.Y.; Tian, Z.Q. Epitaxial growth of heterogeneous metal nanocrystals: From gold nano-octahedra to palladium and silver nanocubes. J. Am. Chem. Soc. 2008, 130, 6949–6951. [Google Scholar] [CrossRef] [PubMed]
- Camargo, P.H.C.; Xiong, Y.; Ji, L.; Zuo, J.M.; Xia, Y. Facile synthesis of tadpole-like nanostructures consisting of Au heads and Pd tails. J. Am. Chem. Soc. 2007, 129, 15452–15453. [Google Scholar] [CrossRef]
- Rodríguez-González, B.; Burrows, A.; Watanabe, M.; Kiely, C.J.; Liz-Marzán, L.M. Multishell bimetallic AuAg nanoparticles: Synthesis, structure and optical properties. J. Mater. Chem. 2005, 15, 1755–1759. [Google Scholar] [CrossRef]
- Chen, D.H.; Chen, C.J. Formation and characterization of Au-Ag bimetallic nanoparticles in water-in-oil microemulsions. J. Mater. Chem. 2002, 12, 1557–1562. [Google Scholar] [CrossRef]
- Yashima, M.; Falk, L.K.L.; Palmqvist, A.E.C.; Holmberg, K. Structure and catalytic properties of nanosized alumina supported platinum and palladium particles synthesized by reaction in microemulsion. J. Coll. Interface Sci. 2003, 268, 348–356. [Google Scholar] [CrossRef]
- Boutonnet, M.; Lögdberg, S.; Svensson, E.E. Recent developments in the aplication of nanoparticles prepared from W/O microemulsions in heterogeneous catalysis. Curr. Opin. Coll. Interface Sci. 2008, 13, 270–286. [Google Scholar] [CrossRef]
- Toshima, N.; Yonezawa, T. Bimetallic nanoparticles—Novel materials for chemical and physical applications. New J. Chem. 1998, 22, 1179–1201. [Google Scholar] [CrossRef]
- Langille, M.R.; Personick, M.L.; Zhang, J.; Mirkin, C.A. Defining rules for the shape evolution of gold nanoparticles. J. Am. Chem. Soc. 2012, 134, 14542–14554. [Google Scholar] [CrossRef] [PubMed]
- Ferrando, R.; Jellinek, J.; Johnston, R.L. Nanoalloys: From theory to applications of alloy clusters and nanoparticles. Chem. Rev. 2008, 108, 845–910. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Zhang, C. Preparation of Cu-Ni alloy nanocrystallites in water-in-oil microemulsions. J. Coll. Interface Sci. 2006, 293, 414–420. [Google Scholar] [CrossRef]
- Wu, M.; Chen, D.; Huang, T. Synthesis of Au/Pd bimetallic nanoaprticles in reverse micelles. Langmuir 2001, 17, 3877–3883. [Google Scholar] [CrossRef]
- Wu, M.; Chen, D.; Huang, T. Preparation of Au/Pt bimetallic nanoaprticles in water-in-oil microemulsions. Chem. Mater. 2001, 13, 599–606. [Google Scholar] [CrossRef]
- Meyer, T.E.; Przysiecki, C.T.; Watkins, J.A.; Bhattacharyya, A.; Simondsen, R.P.; Cusanovich, M.A.; Tollin, G. Correlation between rate constant for reduction and redox potential as a basis for systematic investigation of reaction mechanisms of electron transfer proteins. Proc. Natl. Acad. Sci. USA 1983, 80, 6740–6744. [Google Scholar] [CrossRef] [PubMed]
- Pal, A.; Shah, S.; Devi, S. Synthesis of Au, Ag and Au-Ag alloy nanoparticles in aqueous polymer solution. Coll. Surf. A Physicochem. Eng. Asp. 2007, 302, 51–57. [Google Scholar] [CrossRef]
- Hernández-Fernández, P.; Rojas, S.; Ocón, P.; Gómez de la Fuente, J.L.; San Fabián, J.; Sanza, J.; Peña, M.A.; García-García, F.J.; Terreros, P.; Fierro, J.L.G. Influence of the preparation route of bimetallic Pt-Au nanoparticle electrocatalysts for the oxygen reduction reaction. J. Phys. Chem. C 2007, 111, 2913–2923. [Google Scholar] [CrossRef]
- Fletcher, P.D.I.; Howe, A.M.; Robinson, B.H. The kinetics of solubilisate exchange between water droplets of a water-in-oil microemulsion. J. Chem. Soc. Faraday Trans. 1987, 83, 985–1006. [Google Scholar] [CrossRef]
- Dunstan, T.S.; Fletcher, P.D.I. Compartmentalization and separation of aqueous reagents in the water droplets of water-in-oil high internal phase emulsions. Langmuir 2011, 27, 3409–3415. [Google Scholar] [CrossRef] [PubMed]
- Bagwe, R.P.; Khilar, K.C. Effects of intermicellar exchange rate on the formation of silver nanoparticles in reverse microemulsions of aot. Langmuir 2000, 16, 905–910. [Google Scholar] [CrossRef]
- López-Quintela, M.A.; Tojo, C.; Blanco, M.C.; García Río, L.; Leis, J.R. Microemulsion dynamics and reactions in microemulsions. Curr. Opin. Coll. Interface Sci. 2004, 9, 264–278. [Google Scholar] [CrossRef]
- Tojo, C.; de Dios, M.; Barroso, F. Surfactant effects on microemulsion-based nanoparticle synthesis. Materials 2011, 4, 55–72. [Google Scholar] [CrossRef]
- Tojo, C.; De Dios, M.; López-Quintela, M.A. On the structure of bimetallic nanoparticles synthesized in microemulsions. J. Phys. Chem. C 2009, 113, 19145–19154. [Google Scholar] [CrossRef]
- Magno, L.M.; Sigle, W.; Aken, P.A.V.; Angelescu, D.G.; Stubenrauch, C. Microemulsions as reaction media for the synthesis of bimetallic nanoparticles: Size and composition of particles. Chem. Mater. 2010, 22, 6263–6271. [Google Scholar]
- Buceta, D.; Tojo, C.; Vukmirovik, M.; Deepak, F.L.; López-Quintela, M.A. Sub-nanometer control in the synthesis of bimetallic nanoparticles using a simple strategy by the microemulsion method. 2014; submitted. [Google Scholar]
- Bochicchio, D.; Ferrando, R.; Novakovic, R.; Panizon, E.; Rossi, G. Chemical ordering in magic-size Ag-Pd nanoparticles. 2014, in press. [Google Scholar]
- Tong, Y.; Lu, X.; Sun, W.; Nie, G.; Yang, L.; Wang, C. Electrospun polyacrylonitrile nanofibers supported Ag/Pd nanoparticles for hydrogen generation from the hydrolysis of ammonia borane. J. Power Sour. 2014, 261, 221–226. [Google Scholar] [CrossRef]
- Yang, M.Q.; Pan, X.; Zhang, N.; Xu, Y.J. A facile one-step way to anchor noble metal (Au, Ag, Pd) nanoparticles on a reduced graphene oxide mat with catalytic activity for selective reduction of nitroaromatic compounds. Cryst. Eng. Comm. 2013, 15, 6819–6828. [Google Scholar] [CrossRef]
- Lan, J.L.; Wan, C.C.; Wang, Y.Y. Mechanistic study of Ag/Pd-PVP nanoparticles and their functions as catalyst for electroless copper deposition. J. Electrochem. Soc. 2008, 155, K77–K83. [Google Scholar] [CrossRef]
- Lee, C.L.; Chiou, H.P.; Chang, K.C.; Huang, C.H. Carbon nanotubes-supported colloidal Ag-Pd nanoparticles as electrocatalysts toward oxygen reduction reaction in alkaline electrolyte. Int. J. Hydrog. Energy 2011, 36, 2759–2764. [Google Scholar] [CrossRef]
- Farhadi, K.; Forough, M.; Pourhossein, A.; Molaei, R. Highly sensitive and selective colorimetric probe for determination of L-cysteine in aqueous media based on Ag/Pd bimetallic nanoparticles. Sens. Actuators B Chem. 2014, 202, 993–1001. [Google Scholar] [CrossRef]
- De Dios, M. Síntesis de Nanopartículas en Microemulsiones. Estudio por simulación. Ph.D. Thesis, University of Vigo, Vigo, Spain, 2009. [Google Scholar]
- Quintillán, S.; Tojo, C.; Blanco, M.C.; López-Quintela, M.A. Effects of the intermicellar exchange on the size control of nanoparticles synthsized in microemulsions. Langmuir 2001, 17, 7251–7254. [Google Scholar] [CrossRef]
- Tojo, C.; Blanco, M.C.; López-Quintela, M.A. Preparation of nanoparticles in microemulsions: A monte carlo study of the influence of the synthesis variables. Langmuir 1997, 13, 4527–4534. [Google Scholar] [CrossRef]
- Kitchens, C.L.; Bossev, D.P.; Roberts, C.B. Solvent effects on aot reverse micelles in liquid and compressed alkanes investigated by neutron spin-echo spectroscopy. J. Phys. Chem. B 2006, 110, 20392–20400. [Google Scholar] [CrossRef] [PubMed]
- Housaindokht, M.R.; Pour, A.N. Study the effect of hlb of surfactant on particle size distribution of hematite nanoparticles prepared via the reverse microemulsion. Solid State Sci. 2012, 14, 622–625. [Google Scholar] [CrossRef]
- Niemann, B.; Rauscher, F.; Adityawarman, D.; Voigt, A.; Sundmacher, K. Microemulsion-assisted precipitation of particles: Experimental and model-based process analysis. Chem. Eng. Proc. 2006, 45, 917–935. [Google Scholar] [CrossRef]
- Bandyopadhyaya, R.; Kumar, R.; Gandhi, K.S. Simulation of precipitation reactions in reverse micelles. Langmuir 2000, 16, 7139–7149. [Google Scholar] [CrossRef]
- Ritcey, A.; Lemyre, J.; Beaupré, A.; Lamarre, S. Mechanism of YF3 nanoparticle formation in reverse micelles. Langmuir 2011, 27, 11824–11834. [Google Scholar] [CrossRef] [PubMed]
- Tojo, C.; Barroso, F.; de Dios, M. Critical nucleus size effects on nanoparticle formation in microemulsions: A comparison study between experimental and simulation results. J. Coll. Interface Sci. 2006, 296, 591–598. [Google Scholar] [CrossRef]
- Barroso, F.; Tojo, C. Designing bimetallic nanoparticle structures prepared from microemulsions. J. Phys. Chem. C 2013, 117, 17801–17813. [Google Scholar] [CrossRef]
- Angelescu, D.G.; Magno, L.; Stubenrauch, C. Monte carlo simulation of the size and composition of bimetallic nanoparticles synthesized in water in oil microemulsions. J. Phys. Chem. C 2010, 114, 22069–22078. [Google Scholar] [CrossRef]
- Johans, C.; Pohjakallio, M.; Ijäs, M.; Ge, Y.; Kontturi, K. Control of particle size by pressure adjustment in cobalt nanoaprticles synthesis. Coll. Surf. A Physicochem. Eng. Asp. 2008, 330, 14–20. [Google Scholar] [CrossRef]
- Destrée, C.; Nagy, J.B. Mechanism of formation of inorganic and organic nanoparticles from microemulsions. Adv. Coll. Interface Sci. 2006, 123–126, 353–367. [Google Scholar] [CrossRef]
- Lianos, P.; Thomas, J.K. Cadmium sulfide of small dimensions produced in inverted micelles. Chem. Phys. Lett. 1986, 125, 299–302. [Google Scholar] [CrossRef]
- Pileni, M.P. Reverse micelles used as templates: A new understanding in nanocrystal growth. J. Exp. Nanosci. 2006, 1, 13–27. [Google Scholar] [CrossRef]
- Deng, L.; Hu, W.; Deng, H.; Xiao, S. Surface segregation and structural features of bimetallic Au-Pt nanoparticles. J. Phys. Chem. C 2010, 114, 11026–11032. [Google Scholar]
- Xiong, S.; Qi, W.; Huang, B.; Wang, M. Size-, shape- and composition-dependent alloying ability of bimetallic nanoparticles. ChemPhysChem 2011, 12, 1317–1324. [Google Scholar] [CrossRef] [PubMed]
- Ethayaraja, M.; Dutta, K.; Muthukumaran, D.; Bandyopadhyaya, R. Nanoparticle formation in water-in-oil microemulsions: Experiments, mechanism, and monte carlo simulations. Langmuir 2006, 23, 3418–3423. [Google Scholar] [CrossRef]
- Turro, N.J.; Weed, G.C. Micellar systems as supercages for reactions of geminate radical pairs. Magnetic effects. J. Am. Chem. Soc. 1983, 105, 1861–1868. [Google Scholar] [CrossRef]
- Silva, O.F.; Fernández, M.A.; Silber, J.J.; de Rossi, R.H.; Correa, N.M. Inhibited phenol ionization in reverse micelles: Confinement effect at the nanometer scale. Chem. Phys. Chem. 2012, 13, 124–130. [Google Scholar] [PubMed]
- Kleinman, M.H.; Shevchenko, T.; Bohne, C. Magnetic field effects on the dynamics of radical pairs in micelles: A new approach to undersatnding the “cage effect”. Photochem. Photobiol. 1998, 67, 198–205. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tojo, C.; Vila-Romeu, N. Kinetic Study on the Formation of Bimetallic Core-Shell Nanoparticles via Microemulsions. Materials 2014, 7, 7513-7532. https://doi.org/10.3390/ma7117513
Tojo C, Vila-Romeu N. Kinetic Study on the Formation of Bimetallic Core-Shell Nanoparticles via Microemulsions. Materials. 2014; 7(11):7513-7532. https://doi.org/10.3390/ma7117513
Chicago/Turabian StyleTojo, Concha, and Nuria Vila-Romeu. 2014. "Kinetic Study on the Formation of Bimetallic Core-Shell Nanoparticles via Microemulsions" Materials 7, no. 11: 7513-7532. https://doi.org/10.3390/ma7117513
APA StyleTojo, C., & Vila-Romeu, N. (2014). Kinetic Study on the Formation of Bimetallic Core-Shell Nanoparticles via Microemulsions. Materials, 7(11), 7513-7532. https://doi.org/10.3390/ma7117513