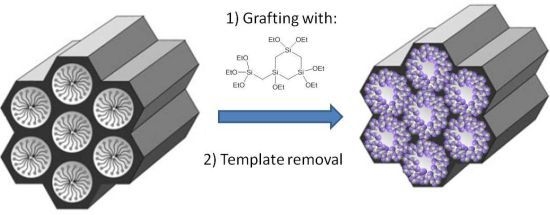
Pore Narrowing of Mesoporous Silica Materials
Abstract
:1. Introduction
2. Experimental Section
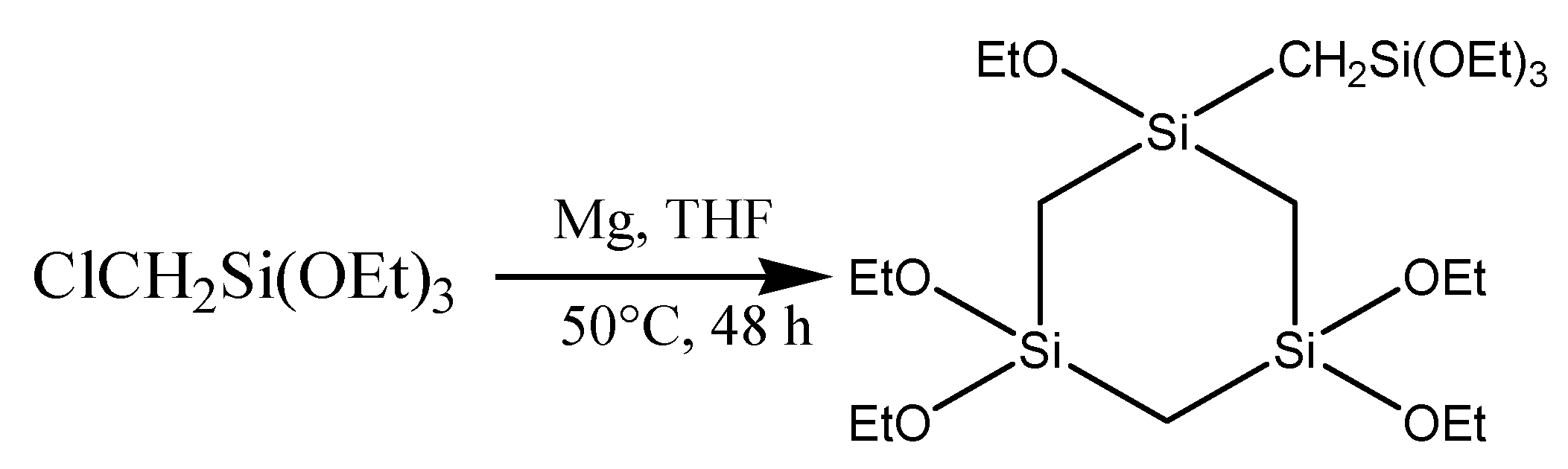
2.1. Chemicals
2.2. Synthesis
2.2.1. Synthesis of MCM-41
2.2.2. Synthesis of Organosilica Films
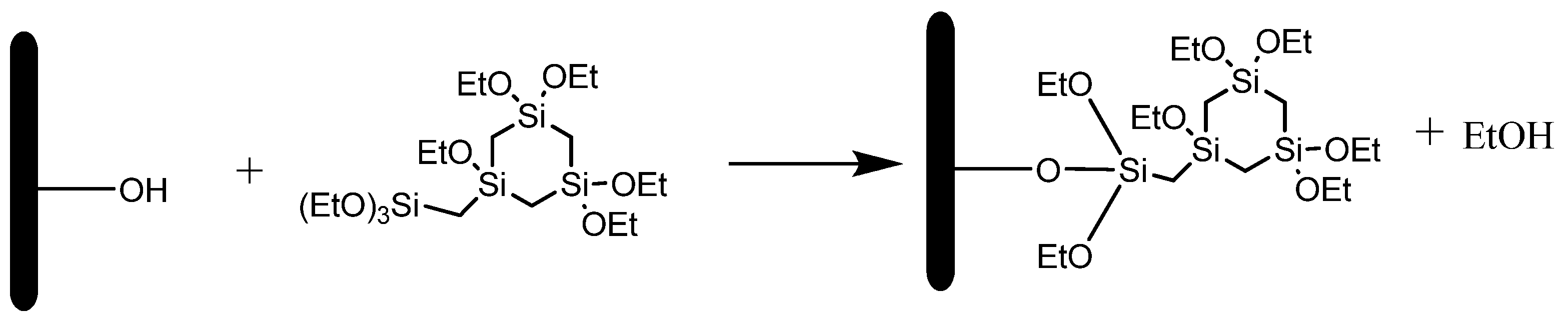
2.2.3. Grafting on MCM-41
2.2.4. Grafting on the Organosilica Films
2.3. Characterization
3. Results and Discussion
3.1. Grafting on MCM-41 without Presence of Surfactant
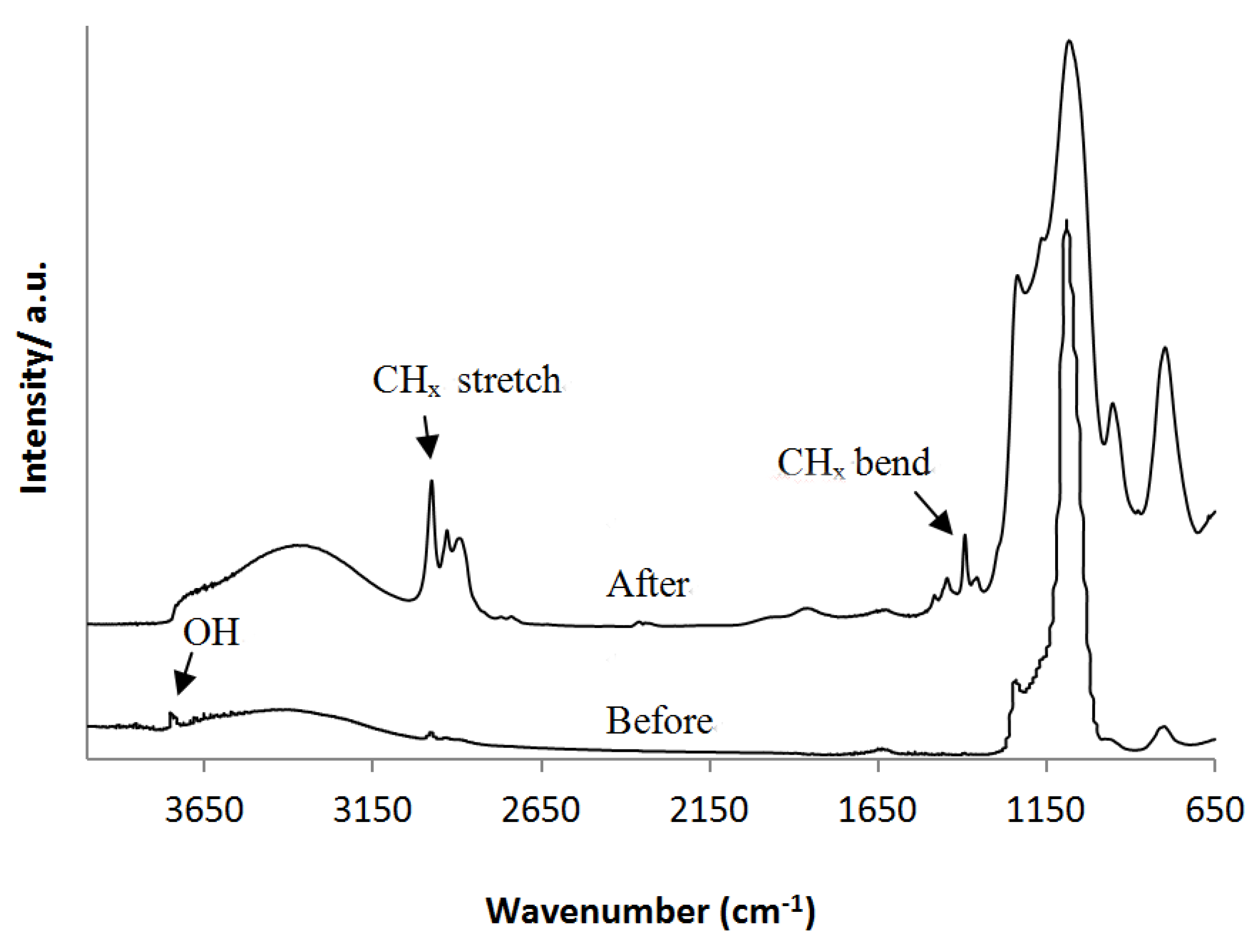
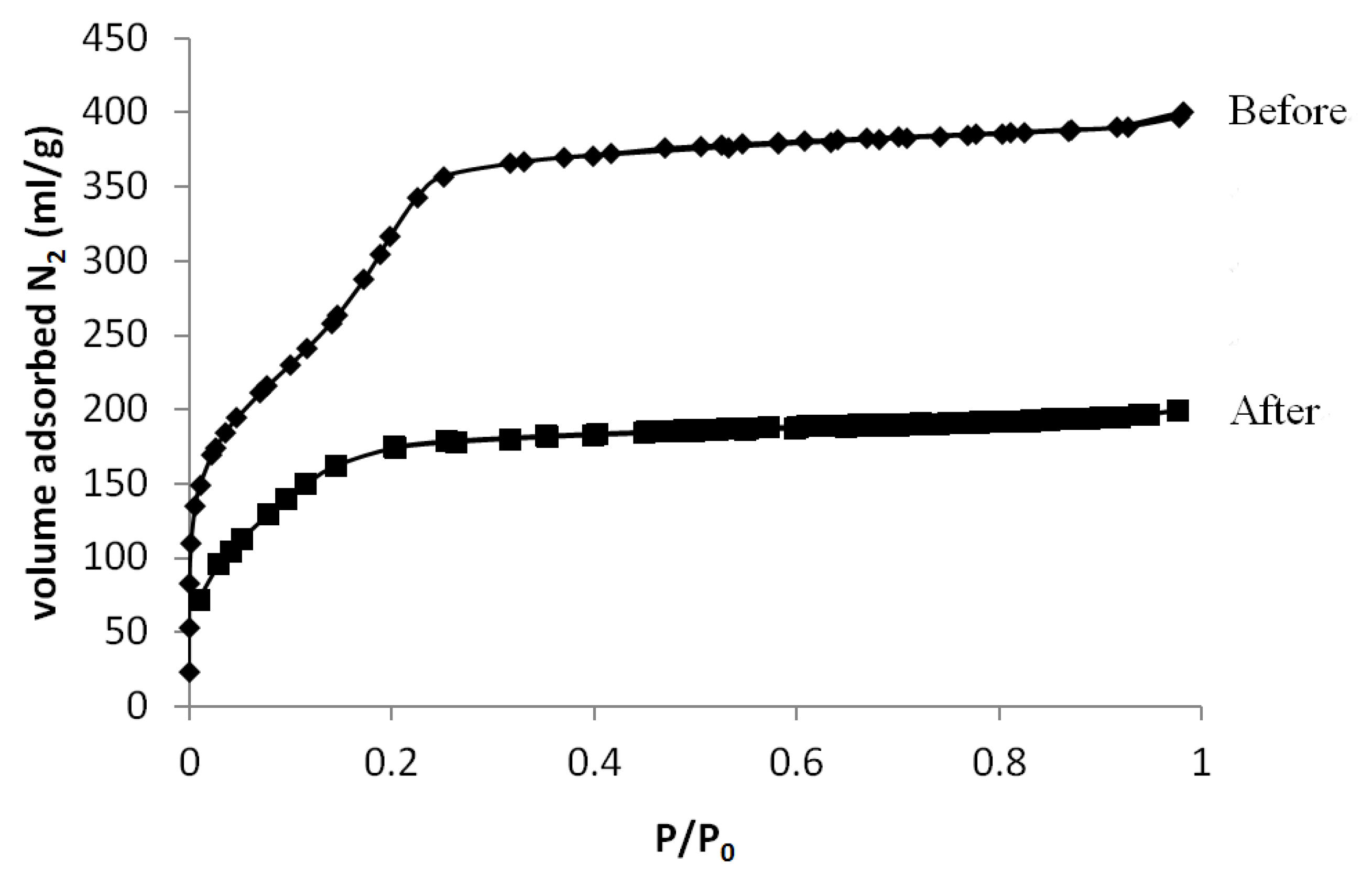
3.2. Grafting on MCM-41 Where the Surfactant was Initially Left Inside
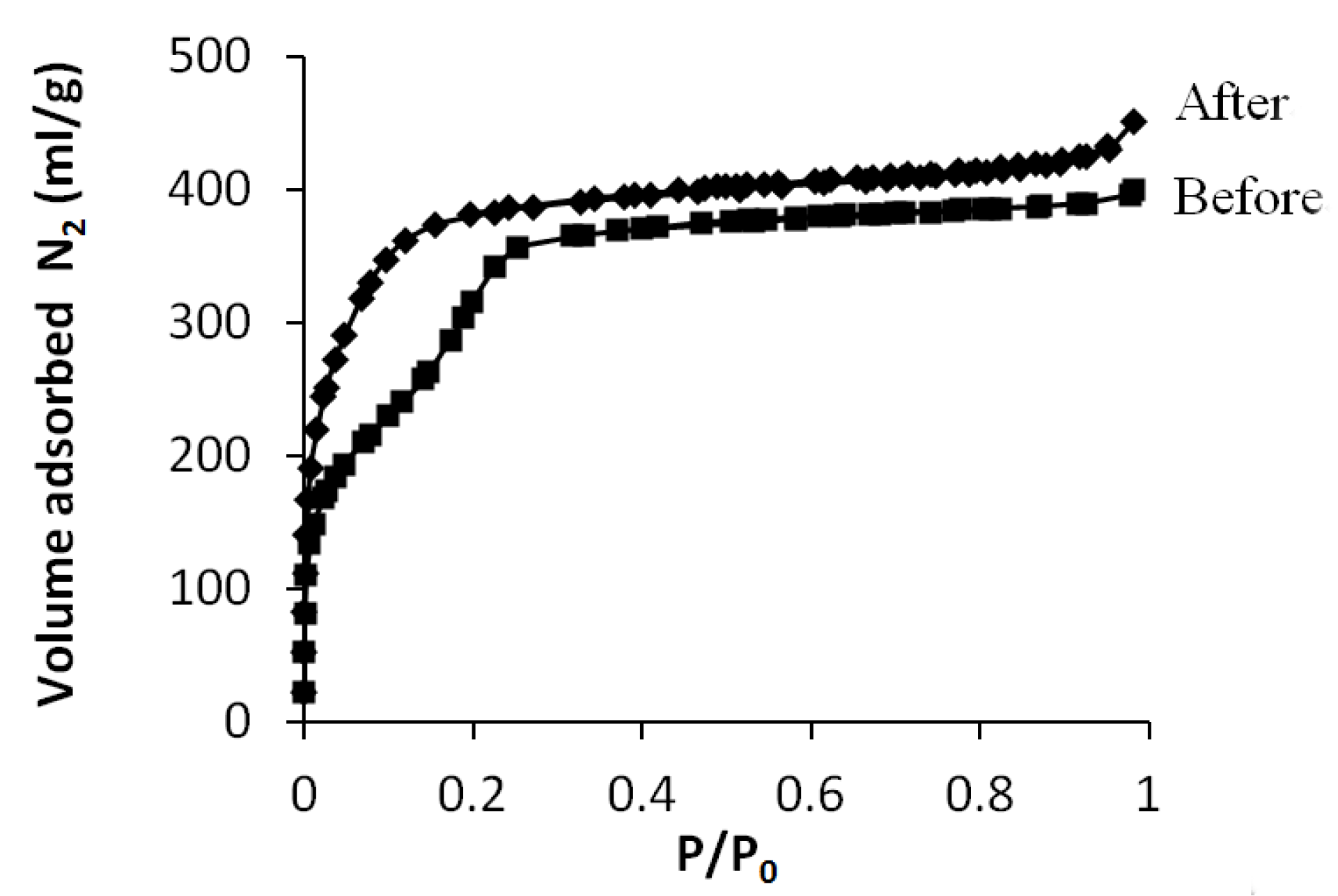
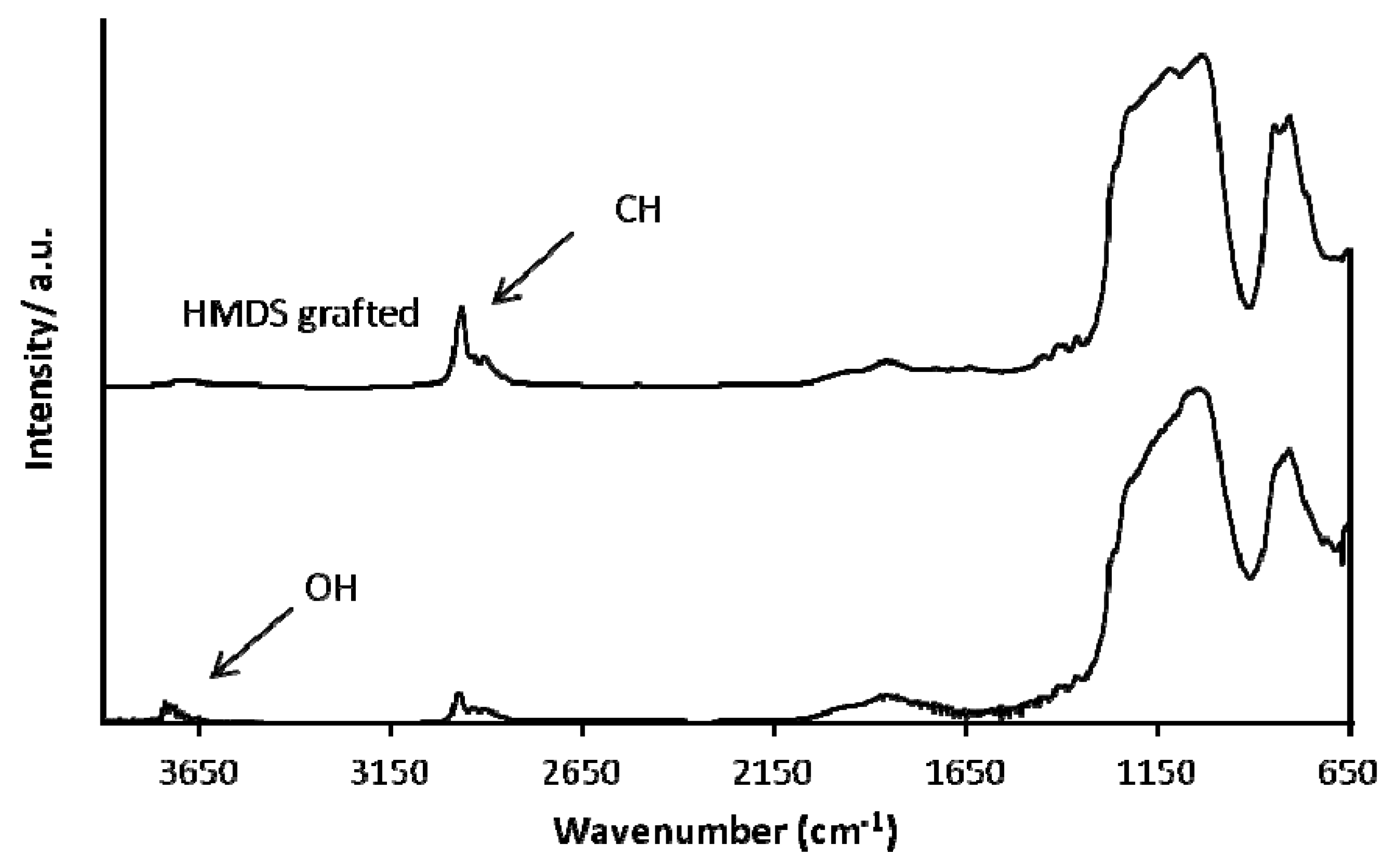
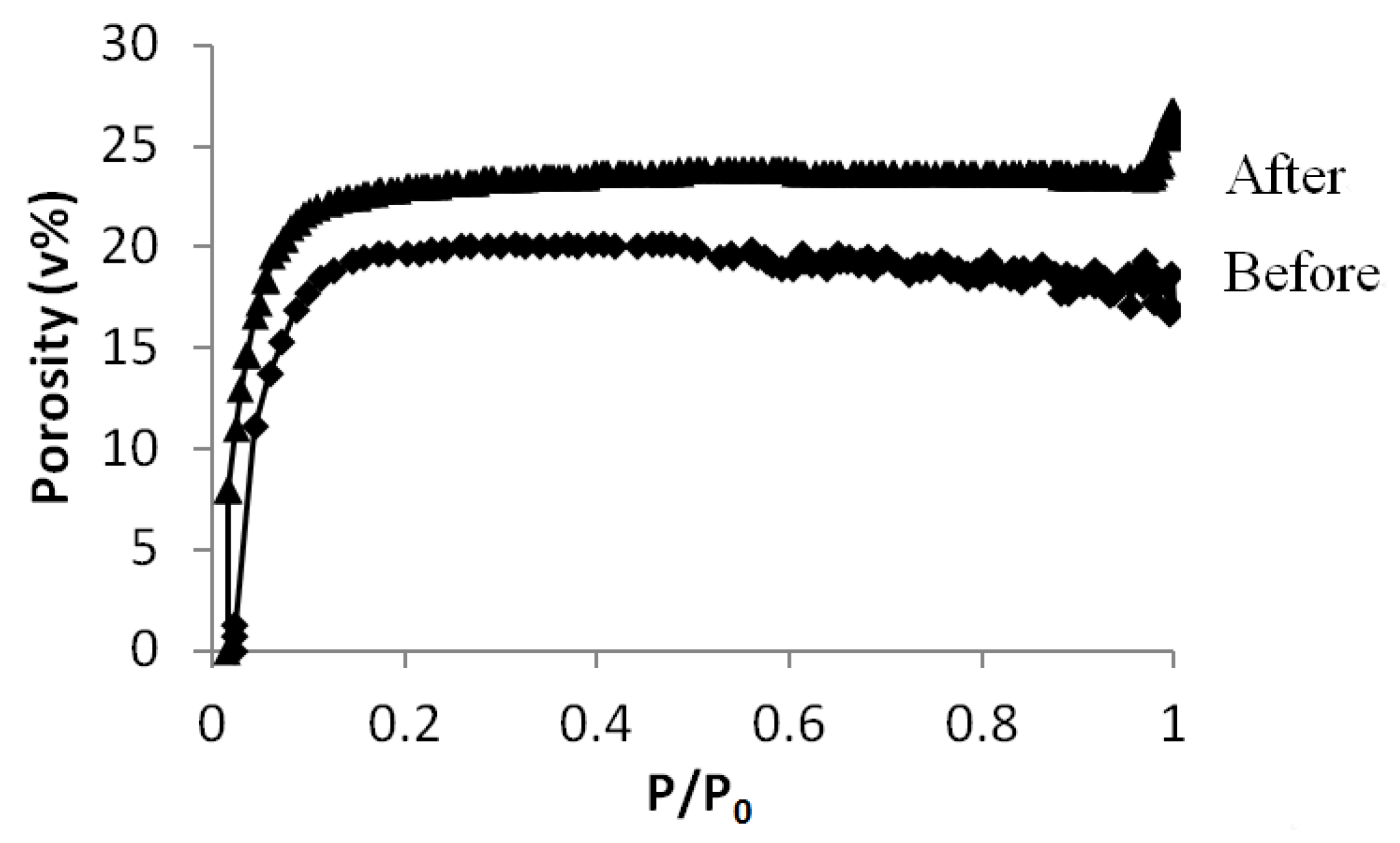
3.3. Grafting on Organosilica Films
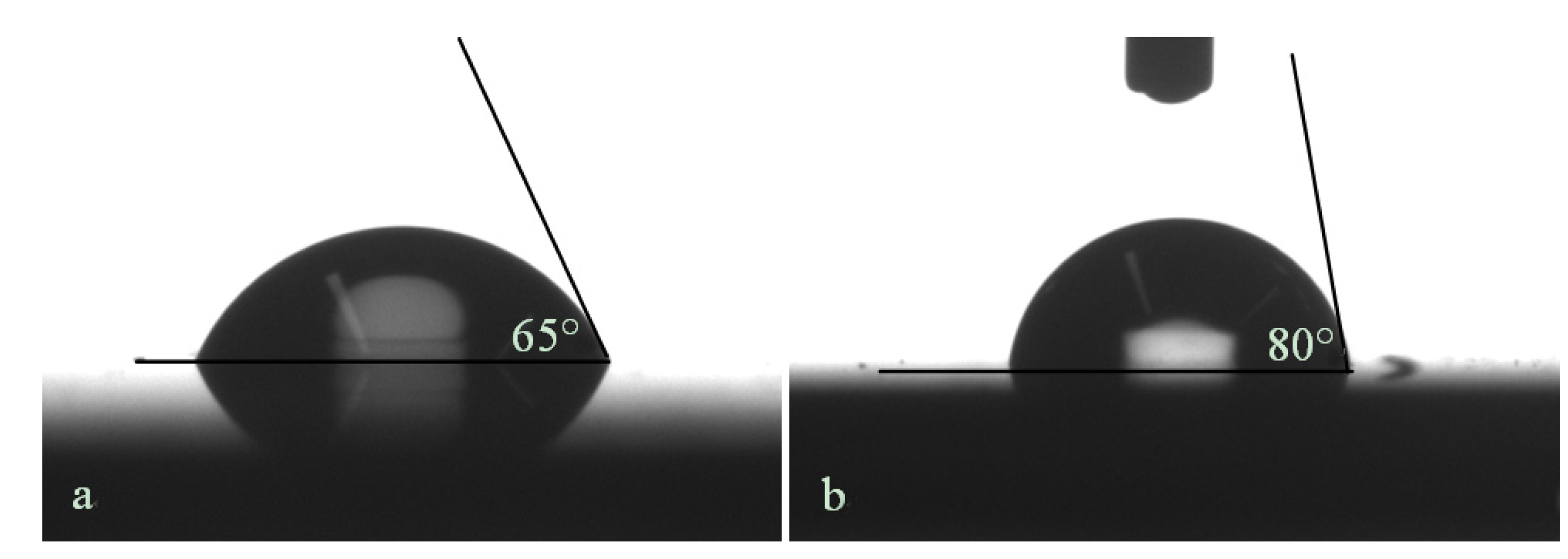
4. Conclusions
References
- Volksen, W.; Miller, R.D.; Dubois, G. Low dielectric constant materials. Chem. Rev. 2010, 110, 56–110. [Google Scholar] [CrossRef] [PubMed]
- Shamiryan, D.; Abell, T.; Iacopi, F.; Maex, K. Low-k dielectric materials. Mater. Today 2004, 7, 34–39. [Google Scholar] [CrossRef]
- Maex, K.; Baklanov, M.R.; Shamiryan, D.; Iacopi, F.; Brongersma, S.H.; Yanovitskaya, Z.S. Low dielectric constant materials for microelectronics. J. Appl. Phys. 2003, 93, 8793–8841. [Google Scholar] [CrossRef]
- Hatton, B.D.; Landskron, K.; Hunks, W.J.; Bennett, M.R.; Shukaris, D.; Perovic, D.D.; Ozin, G.A. Materials chemistry for low-k materials. Mater. Today 2006, 9, 22–31. [Google Scholar] [CrossRef]
- Seo, T.; Yoshino, T.; Ohnuki, N.; Seino, Y.; Cho, Y.; Hata, N.; Kikkawa, T. Effect of silylation hardening on the electrical characteristics of mesoporous pure silica zeolite film. J. Electrochem. Soc. 2009, 156, H98–H105. [Google Scholar] [CrossRef]
- Puyrenier, W.; Rouessac, V.; Broussous, L.; Rebiscoul, D.; Ayral, A. Effect of plasma treatments on a porous low-k material—Study of pore sealing. Microporous Mesoporous Mater. 2007, 106, 40–48. [Google Scholar] [CrossRef]
- Yuan, J.; Ye, C.; Xing, Z.Y.; Xu, Y.J.; Ning, Z.Y. Reducing Cu diffusion in SiCOH low-k films by O-2 plasma treatment. Microelectron. Eng. 2009, 86, 2119–2122. [Google Scholar] [CrossRef]
- Juneja, J.S.; Wang, P.I.; Karabacak, T.; Lu, T.M. Dielectric barriers, pore sealing, and metallization. Thin Solid Films 2006, 504, 239–242. [Google Scholar] [CrossRef]
- Jiang, Y.B.; Liu, N.G.; Gerung, H.; Cecchi, J.L.; Brinker, C.J. Nanometer-thick conformal pore sealing of self-assembled mesoporous silica by plasma-assisted atomic layer deposition. J. Am. Chem. Soc. 2006, 128, 11018–11019. [Google Scholar] [CrossRef] [PubMed]
- Whelan, C.M.; Le, Q.T.; Cecchet, F.; Satta, A.; Pireaux, J.J.; Rudolf, P.; Maex, K. Sealing of porous low-k dielectrics—Ellipsometric porosimetry study of UV-O-3 oxidized SiOxCy films. Electrochem. Solid State Lett. 2004, 7, F8–F10. [Google Scholar] [CrossRef]
- Fayolle, M.; Passemard, G.; Louveau, O.; Fusalba, F.; Cluzel, J. Challenges of back end of the line for sub 65 nm generation. Microelectron. Eng. 2003, 70, 255–266. [Google Scholar] [CrossRef]
- Bonitz, J.; Schulz, S.E.; Gessner, T. Ultra thin CVD TiN layers as diffusion barrier films on porous low-k materials. Microelectron. Eng. 2004, 76, 82–88. [Google Scholar] [CrossRef]
- Nakahira, J.; Nagano, S.; Gawase, A.; Ohashi, Y.; Shimizu, H.; Chikaki, S.; Oda, N.; Kondo, S.; Hasaka, S.; Saito, S. High-etching-selectivity barrier SiC (k < 3.5) Film for 32-nm-node copper/low-k interconnects. Jpn. J. Appl. Phys. 2010, 49, 05FD04:1–05FD04:8. [Google Scholar] [CrossRef]
- Kruk, M.; Hui, C.M. Thermally induced transition between open and closed spherical pores in ordered mesoporous silicas. J. Am. Chem. Soc. 2008, 130, 1528–1529. [Google Scholar] [CrossRef] [PubMed]
- Mandal, M.; Kruk, M. Large-pore ethylene-bridged periodic mesoporous organosilicas with face-centered cubic structure. J. Phys. Chem. C 2010, 114, 20091–20099. [Google Scholar] [CrossRef]
- Mandal, M.; Kruk, M. Surfactant-templated synthesis of ordered silicas with closed cylindrical mesopores. Chem. Mater. 2012, 24, 149–154. [Google Scholar] [CrossRef]
- Goethals, F.; Baklanov, M.R.; Ciofi, I.; Detavernier, C.; van der Voort, P.; van Driessche, I. A new procedure to seal the pores of mesoporous low-k films with precondensed organosilica oligomers. Chem. Commun. 2012, 48, 2797–2799. [Google Scholar] [CrossRef]
- Goethals, F.; Ciofi, I.; Madia, O.; Vanstreels, K.; Baklanov, M.R.; Detavernier, C.; van der Voort, P.; van Driessche, I. Ultra-low-k cyclic carbon-bridged PMO films with a high chemical resistance. J. Mater. Chem. 2012, 22, 8281–8286. [Google Scholar] [CrossRef]
- Beck, J.S.; Vartuli, J.C.; Roth, W.J.; Leonowicz, M.E.; Kresge, C.T.; Schmitt, K.D.; Chu, C.T.W.; Olson, D.H.; Sheppard, E.W.; Mccullen, S.B.; Higgins, J.B.; Schlenker, J.L. A new family of mesoporous molecular-sieves prepared with liquid-crystal templates. J. Am. Chem. Soc. 1992, 114, 10834–10843. [Google Scholar] [CrossRef]
- Brondani, D.J.; Corriu, R.J.P.; Elayoubi, S.; Moreau, J.J.E.; Man, M.W.C. Polyfunctional carbosilanes and organosilicon compounds—Synthesis via grignard reactions. Tetrahedron Lett. 1993, 34, 2111–2114. [Google Scholar] [CrossRef]
- Mortera, R.; Onida, B.; Fiorilli, S.; Cauda, V.; Brovarone, C.V.; Baino, F.; Verne, E.; Garrone, E. Synthesis and characterization of MCM-41 spheres inside bioactive glass-ceramic scaffold. Chem. Eng. J. 2008, 137, 54–61. [Google Scholar] [CrossRef]
- Armini, S.; Prado, J.L.; Swerts, J.; Sun, Y.; Krishtab, M.; Meersschaut, J.; Blauw, M.; Baklanov, M.; Verdonck, P. Pore sealing of porous ultralow-k dielectrics by self-assembled monolayers combined with atomic layer deposition. ECS Solid State Lett. 2012, 1, P42–P44. [Google Scholar] [CrossRef]
- Baklanov, M.R.; Mogilnikov, K.P. Characterization of porous dielectric films by ellipsometric porosimetry. Opt. Appl. 2000, 30, 491–496. [Google Scholar]
- Baklanov, M.R.; Mogilnikov, K.P.; Polovinkin, V.G.; Dultsev, F.N. Determination of pore size distribution in thin films by ellipsometric porosimetry. J. Vac. Sci. Technol. B 2000, 18, 1385–1391. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Goethals, F.; Levrau, E.; De Canck, E.; Baklanov, M.R.; Detavernier, C.; Van Driessche, I.; Van Der Voort, P. Pore Narrowing of Mesoporous Silica Materials. Materials 2013, 6, 570-579. https://doi.org/10.3390/ma6020570
Goethals F, Levrau E, De Canck E, Baklanov MR, Detavernier C, Van Driessche I, Van Der Voort P. Pore Narrowing of Mesoporous Silica Materials. Materials. 2013; 6(2):570-579. https://doi.org/10.3390/ma6020570
Chicago/Turabian StyleGoethals, Frederik, Elisabeth Levrau, Els De Canck, Mikhail R. Baklanov, Christophe Detavernier, Isabel Van Driessche, and Pascal Van Der Voort. 2013. "Pore Narrowing of Mesoporous Silica Materials" Materials 6, no. 2: 570-579. https://doi.org/10.3390/ma6020570
APA StyleGoethals, F., Levrau, E., De Canck, E., Baklanov, M. R., Detavernier, C., Van Driessche, I., & Van Der Voort, P. (2013). Pore Narrowing of Mesoporous Silica Materials. Materials, 6(2), 570-579. https://doi.org/10.3390/ma6020570