Solubilization of Hydrophobic Dyes in Surfactant Solutions
Abstract
:1. Introduction
2. Dyes
| Purpose | Chemicals |
|---|---|
| Dispersants/Surfactants | lignin sulfonates, naphthalene sulfonate, formaldehyde condensates, ethylene oxide-propylene oxide block copolymers |
| Salts | sodium sulfate, sodium chloride |
| Dust-bonding agents | mineral oils, paraffin oils (+additives) |
| Antifoams | ditertiary acetylene glycols |
| Antifreezes | glycerol, glycols |
| Thickeners | carboxymethylcellulose, polyacrylates |
| Buffer systems | phosphate, acetate |
3. Coloration with Hydrophobic Dyes
3.1. Coloration with Disperse and Solvent Dyes
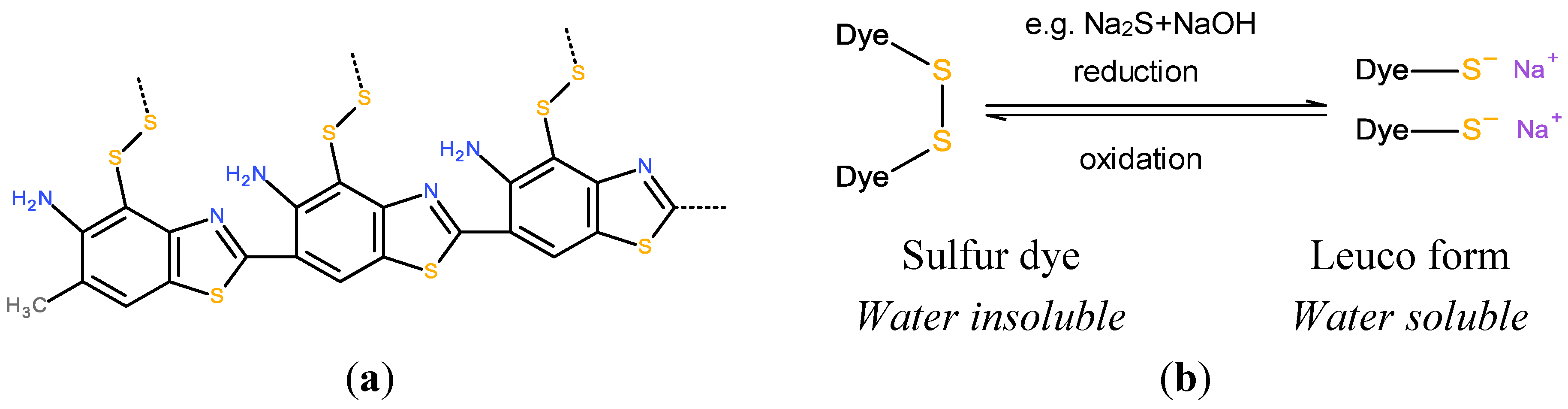
3.2. Coloration with Vat and Sulfur Dyes
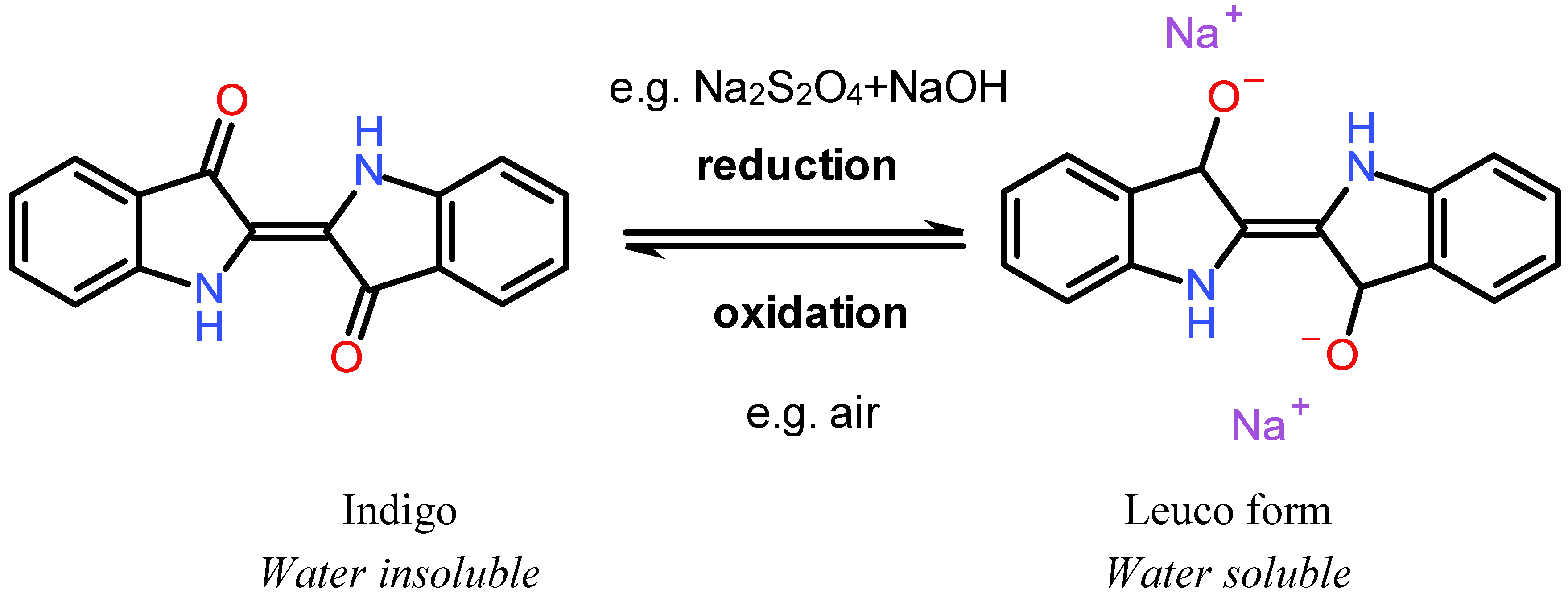
4. Surfactants
5. Standard Test Method for Dye Solubilization
6. Important Factors in Dye Solubilization
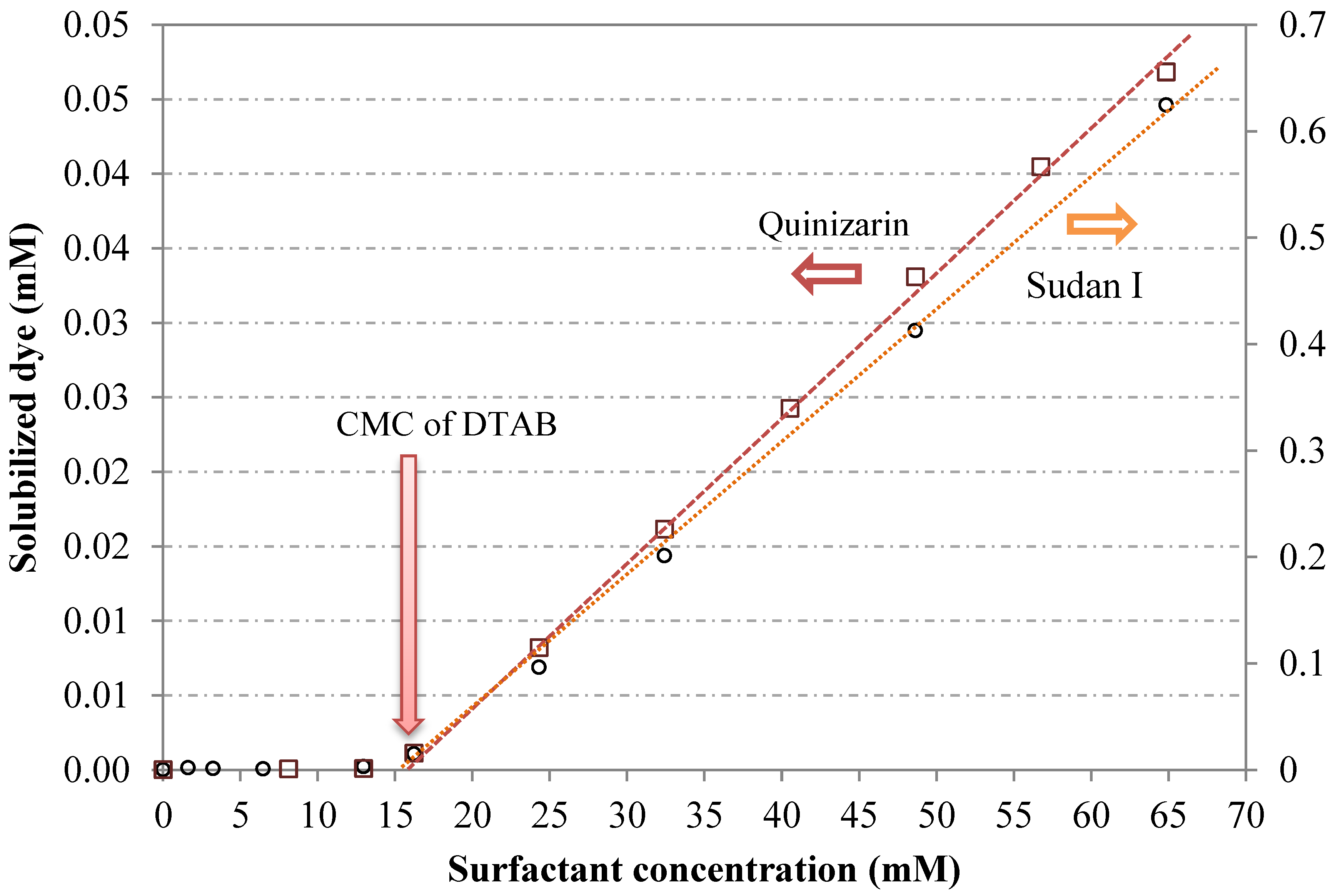
6.1. Surfactant Concentration
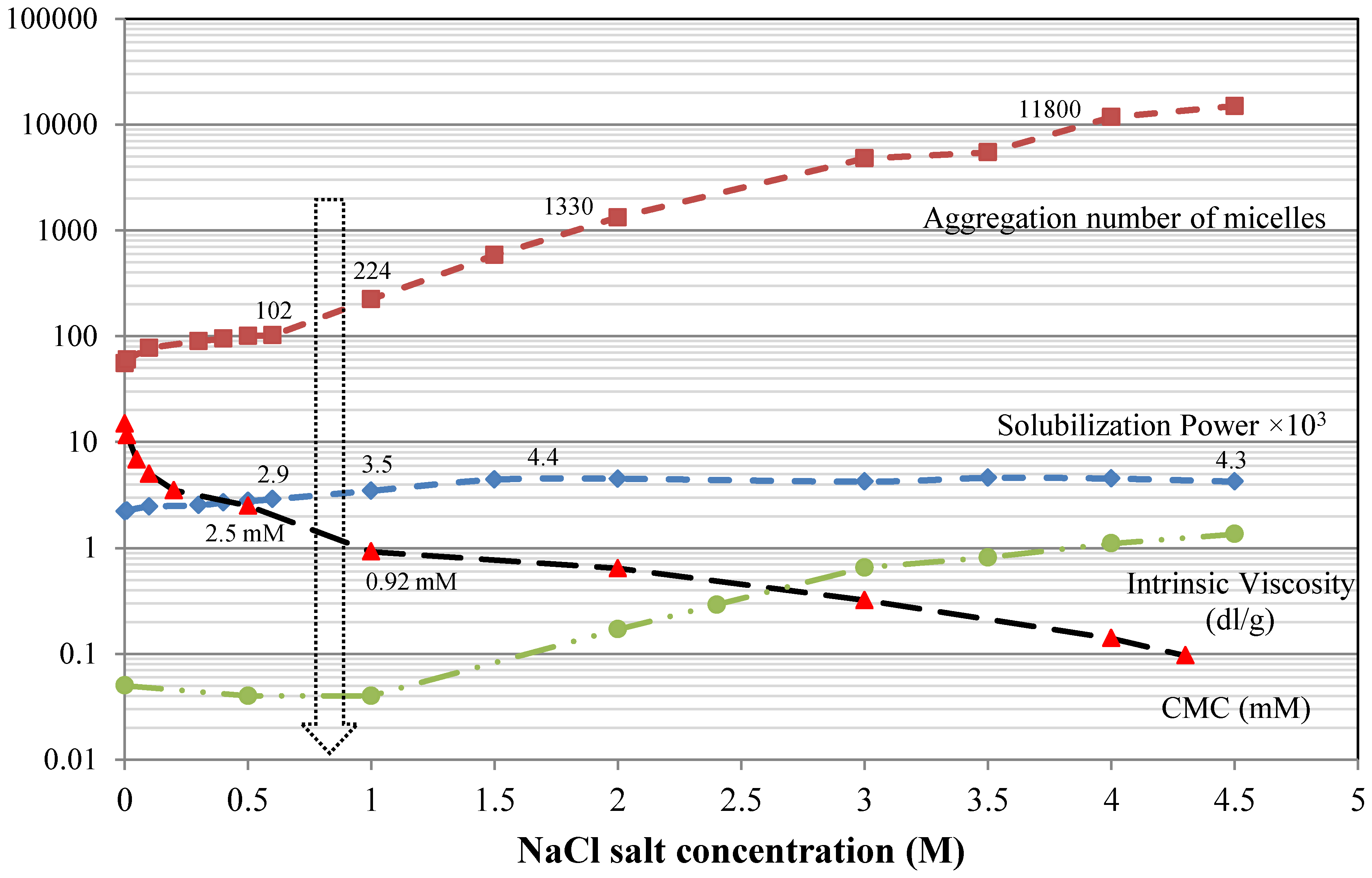
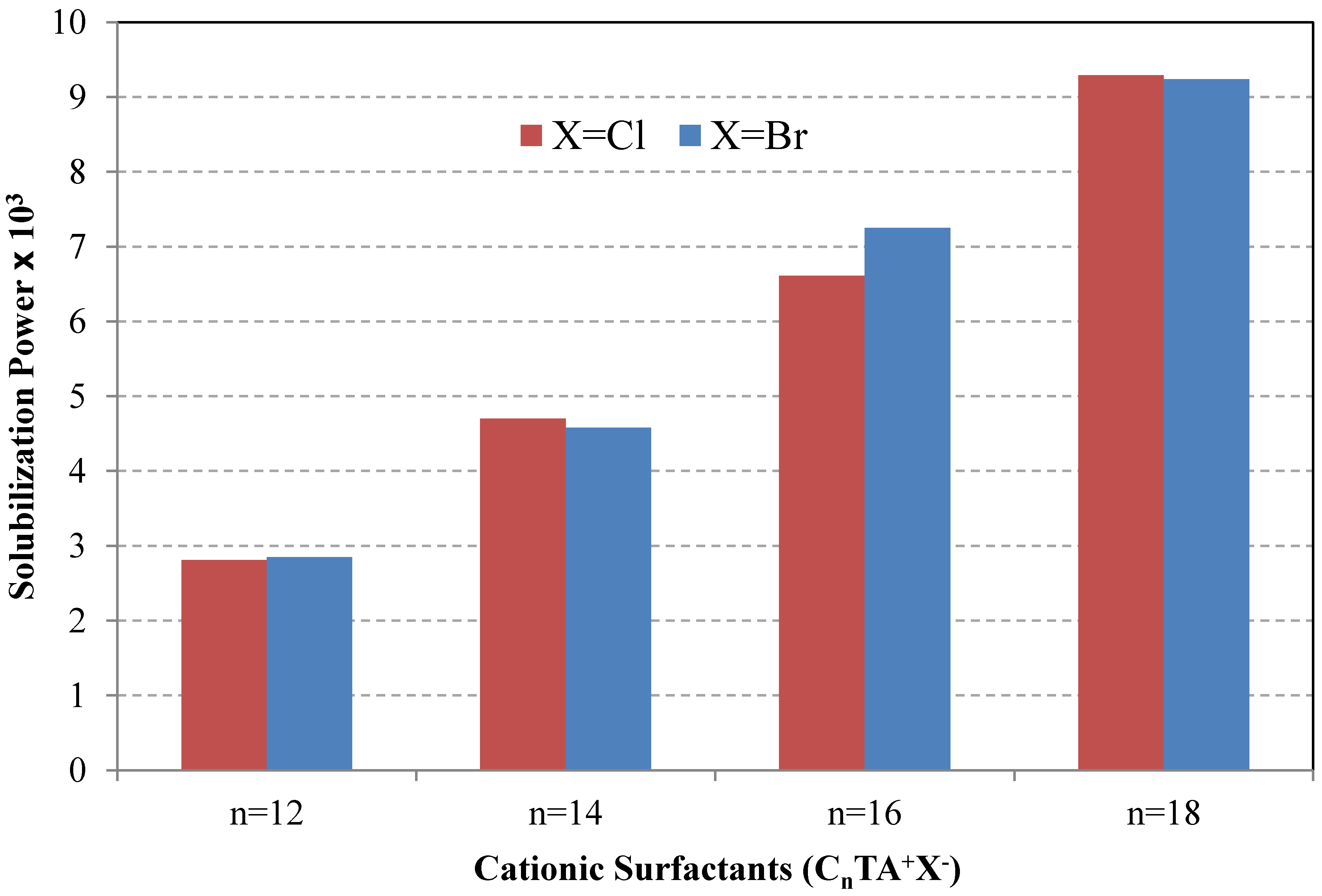
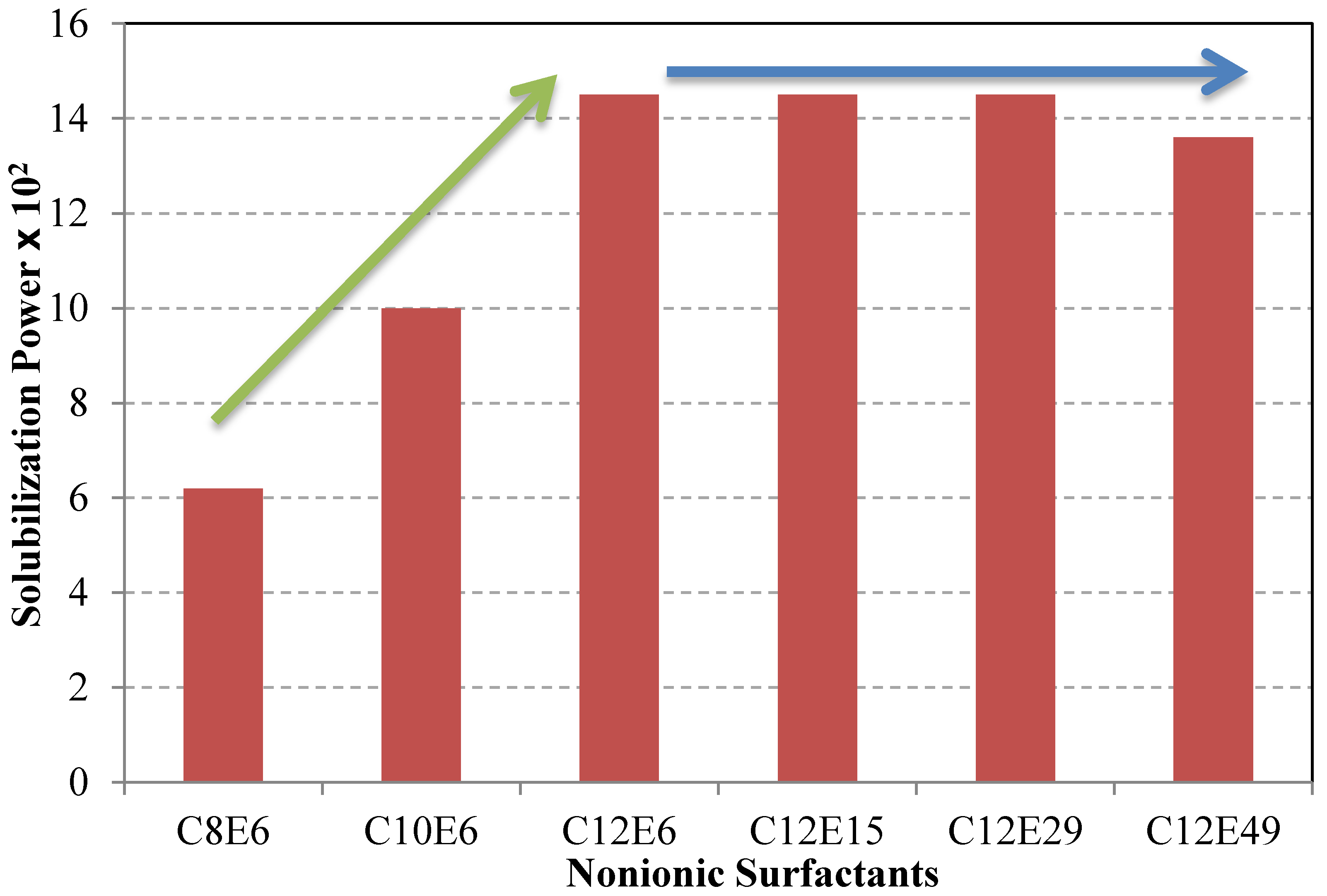
6.2. Surfactant Structure
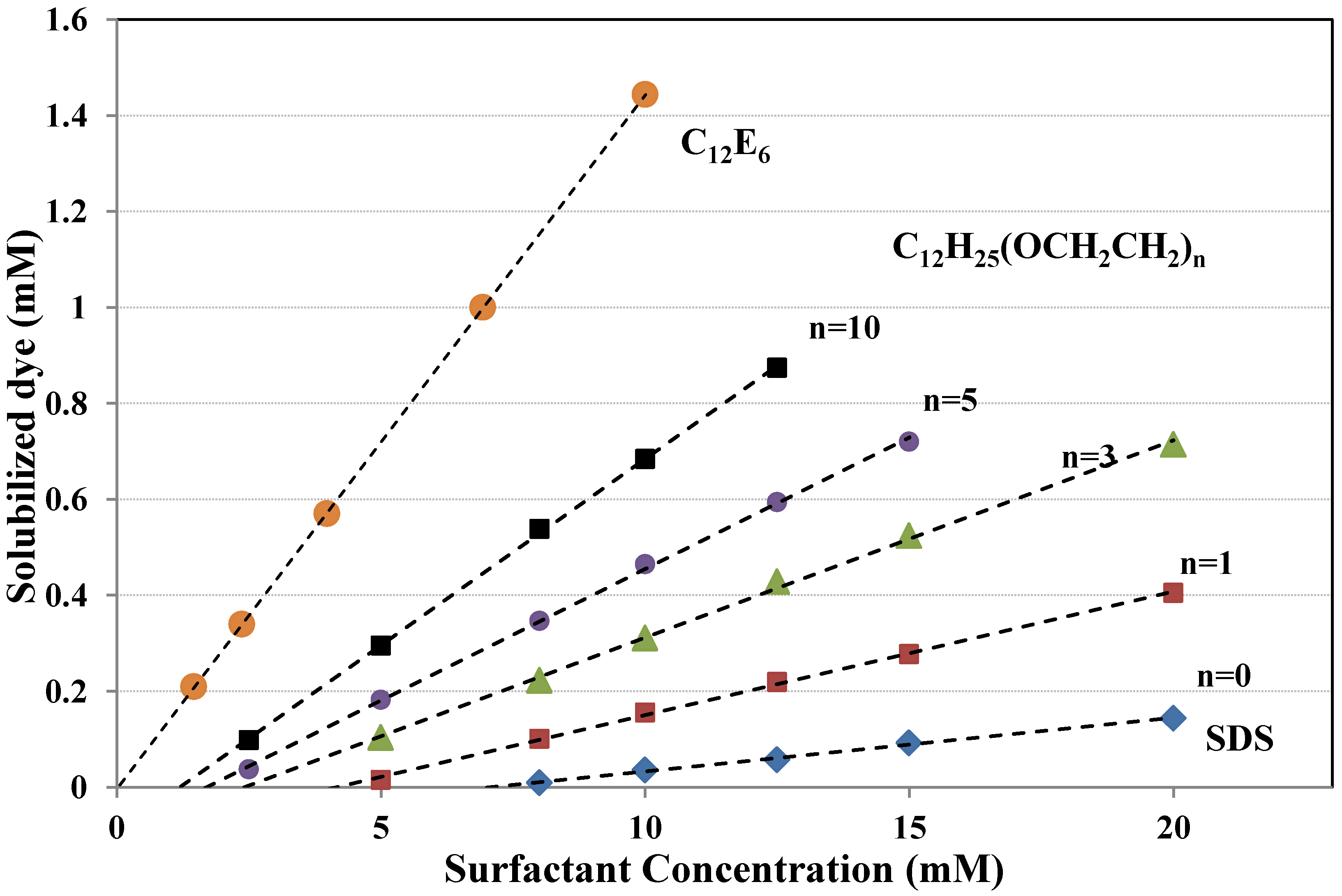

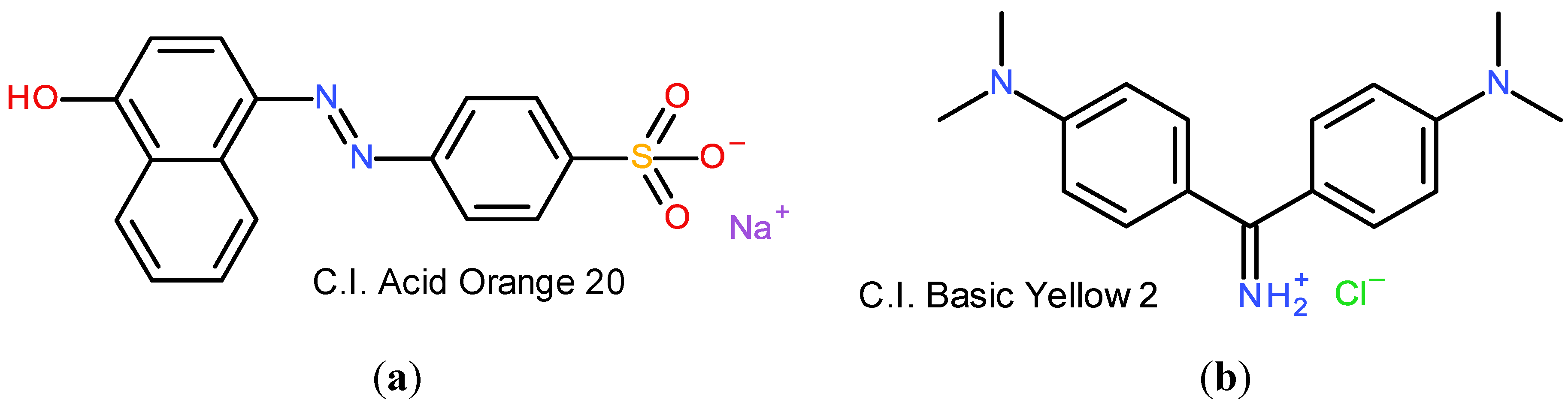
6.3. Dye Structure
| Dye | Dye molecular weight (g/mol) | Solubilization power (103 mol/mol) | Reference |
|---|---|---|---|
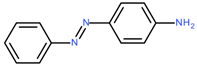 C.I. Solvent Yellow 1 | 197.2 | 256 | [43] |
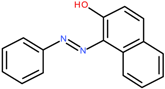 Sudan I/C.I. Solvent Yellow 14 | 248.3 | 13.5 | [44,45] |
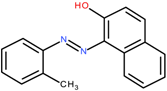 Orange OT/C.I. Solvent Orange 2 | 262.3 | 11.5 | [74] |
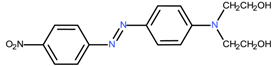 C.I. Disperse Red 19 | 330.3 | 2.7 | [43] |
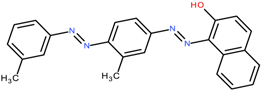 Sudan IV / C.I. Solvent Red 25 | 380.4 | 2.2 and 2.8 | [54,75,76] |
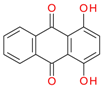 Quinizarin / C.I. Solvent Orange 86 | 240.2 | 0.9 | [44,45] |
 C.I. Disperse Violet 1 | 238.2 | 4 | [43] |
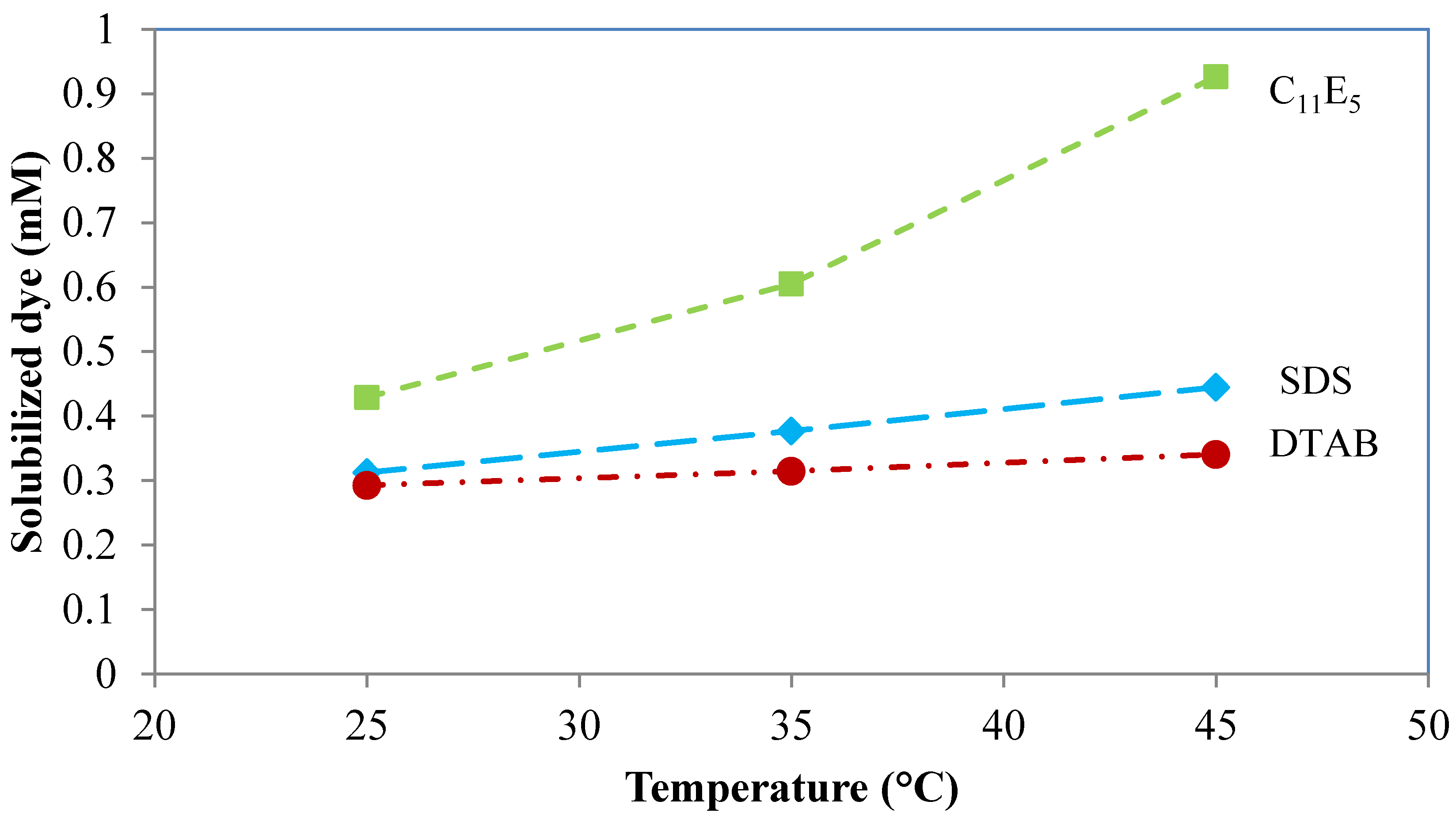
6.4. Temperature
6.5. Addition of Salt
6.6. Addition of Polyelectrolytes

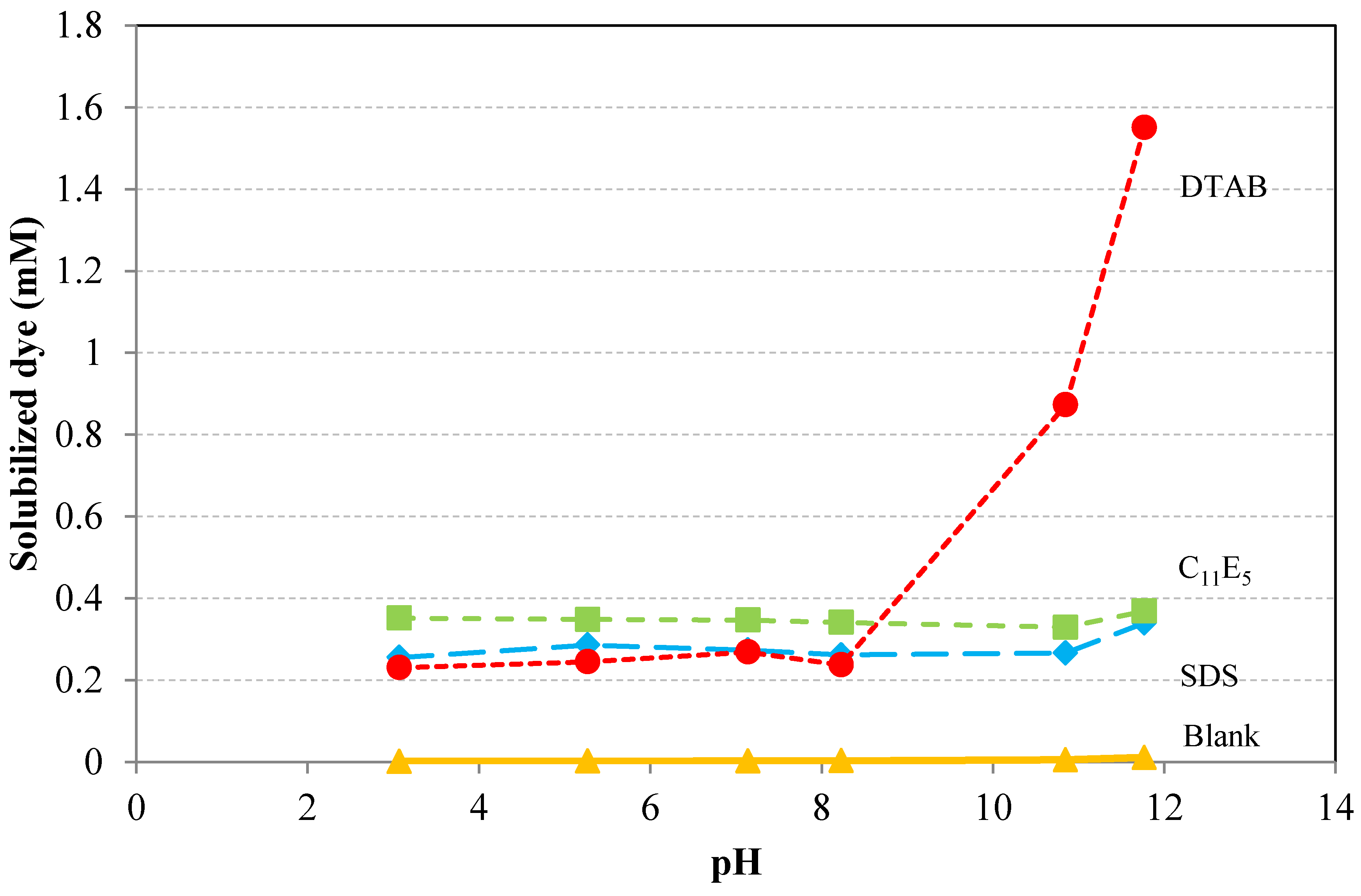
6.7. Influence of pH
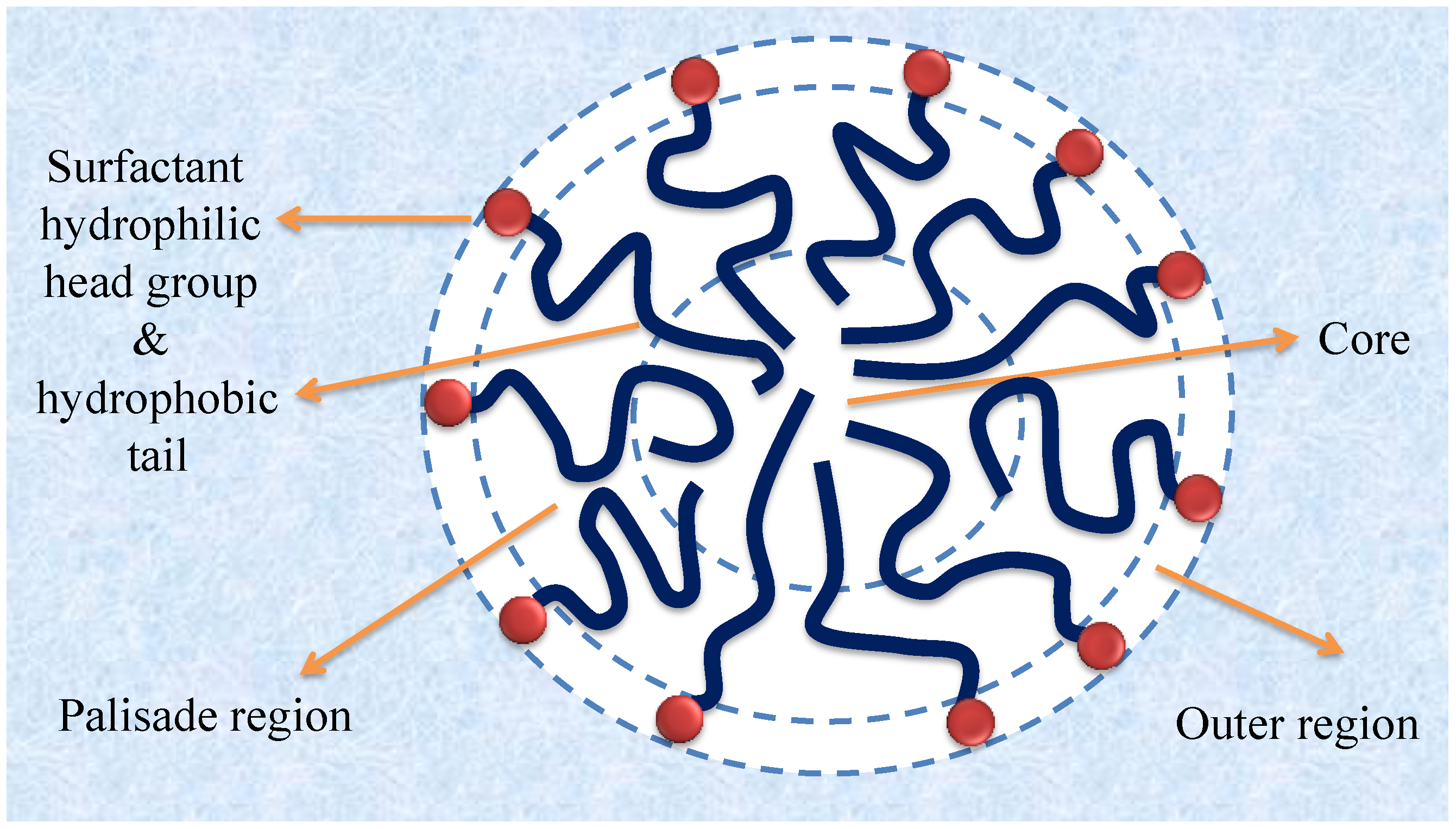
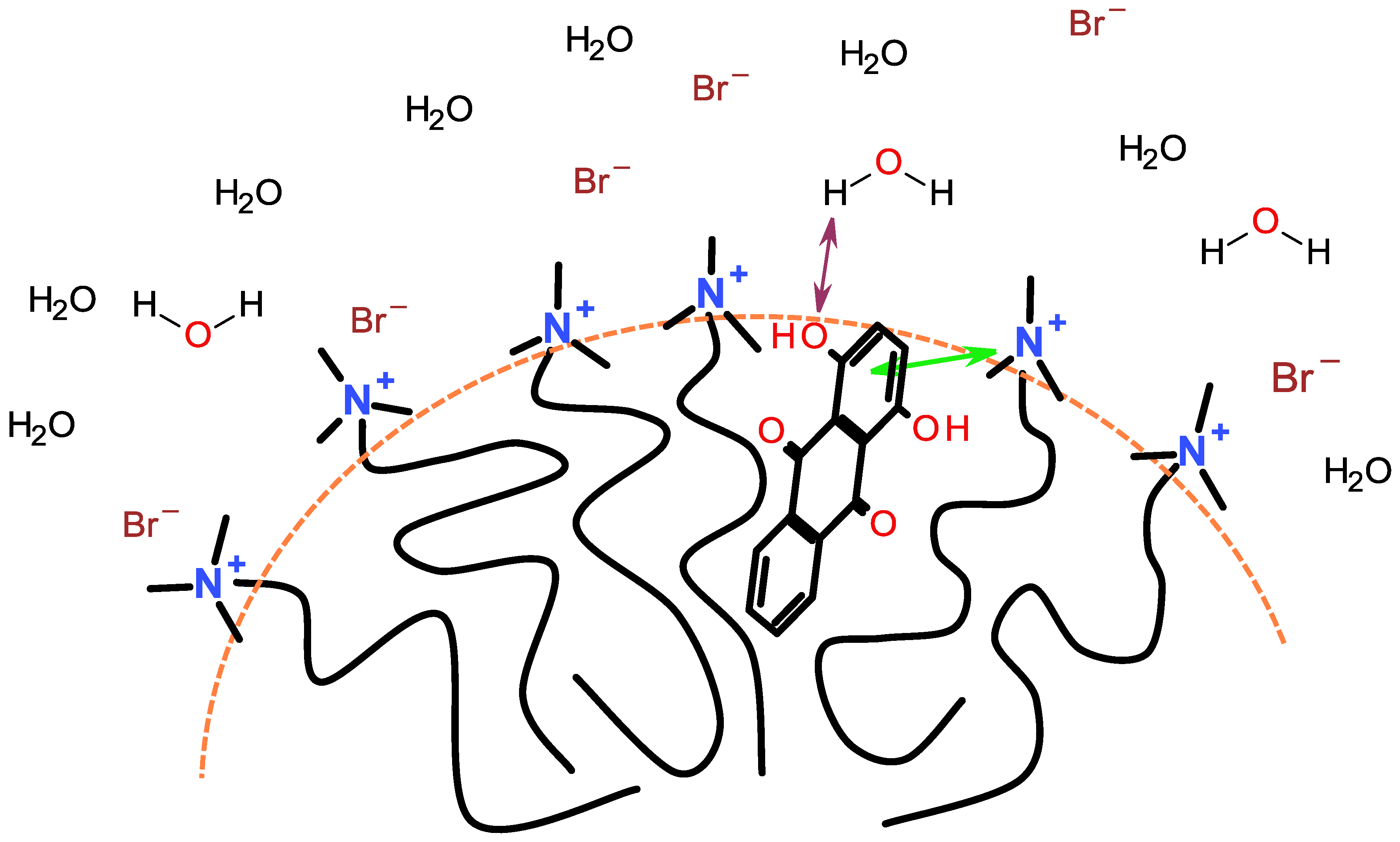
7. Location of Solubilized Dye in the Micelle
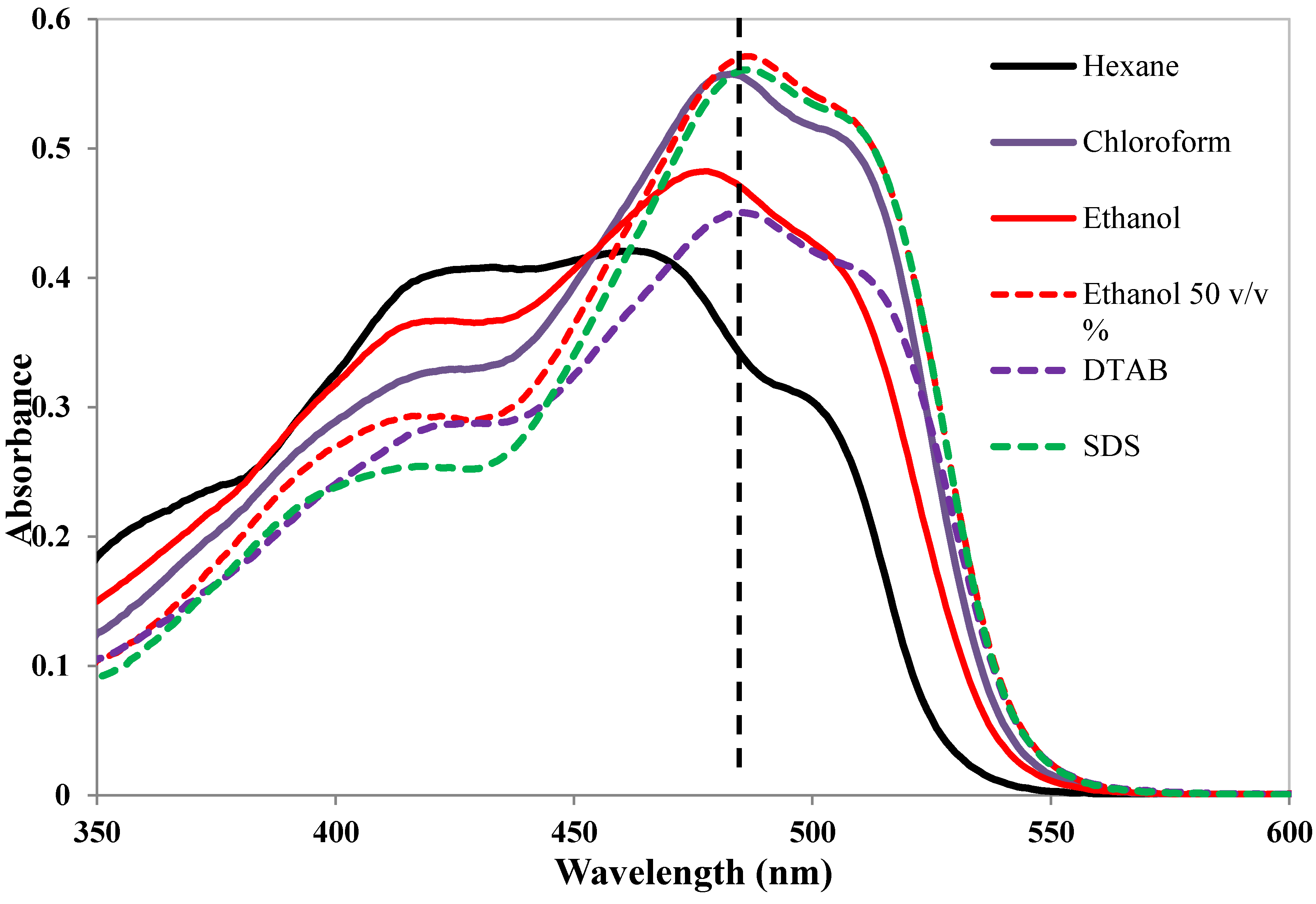
8. Solubilization in Mixed Micelles
9. Conclusions
References
- Mcbain, J.W.; Johnson, K.E. Solubilization and the colloidal micelles in soap solution. J. Am. Chem. Soc. 1944, 66, 9–13. [Google Scholar] [CrossRef]
- McBain, J.W.; Merrill, R.C.; Vinograd, J.R. The solubilization of water-insoluble dye in dilute solutions of aqueous detergents. J. Am. Chem. Soc. 1941, 63, 670–676. [Google Scholar] [CrossRef]
- Merrill, R.C.; Mcbain, J.W. Studies on solubilization. J. Phys. Chem. 1942, 46, 10–19. [Google Scholar] [CrossRef]
- Green, A.A.; Mcbain, J.W. Solubilization of water-insoluble dye by pure soaps and detergents of different types. J. Phys. Chem. 1947, 51, 286–298. [Google Scholar] [CrossRef]
- Kolthoff, I.M.; Stricks, W. Solubilization of dimethylaminoazobenzene in solutions of detergents. I: The effect of temperature on the solubilization and upon the critical concentration. J. Phys. Colloid Chem. 1948, 52, 915–941. [Google Scholar] [CrossRef] [PubMed]
- McBain, J.W.; Wilder, A.G.; Merrill, R.C. Solubilization of water-insoluble dye by colloidal electrolytes and non-ionizing detergents. J. Phys. Colloid Chem. 1948, 52, 12–22. [Google Scholar] [CrossRef] [PubMed]
- Shore, J. Colorants and Auxiliaries; SDC: Bradford, UK, 1990. [Google Scholar]
- Rosen, M.J. Surfactants and Interfacial Phenomena, 3rd ed.; Wiley: New York, NY, USA, 2004. [Google Scholar]
- Holmberg, K.; Jonsson, B.; Kronberg, B.; Lindman, B. Surfactant and Polymers in Aqueous Solution, 2nd ed.; John Wiley & Sons Ltd.: Chichester, UK, 2003. [Google Scholar]
- Oakes, J.; Dixon, S. Physical interactions of dyes in solution—Influence of dye structure on aggregation and binding to surfactants/polymers. Rev. Prog. Coloration 2004, 34, 110–128. [Google Scholar] [CrossRef]
- Oakes, J.; Gratton, P. Solubilisation of dyes by surfactant micelles. Part 2: Molecular interactions of azo dyes with cationic and zwitterionic surfactants. Color. Technol. 2003, 119, 100–107. [Google Scholar] [CrossRef]
- Oakes, J.; Gratton, P. Solubilisation of dyes by surfactant micelles. Part 1: Molecular interactions of azo dyes with nonionic and anionic surfactants. Color. Technol. 2003, 119, 91–99. [Google Scholar] [CrossRef]
- Oakes, J.; Gratton, P.; Dixon, S. Solubilisation of dyes by surfactant micelles. Part 3: A spectroscopic study of azo dyes in surfactant solutions. Color. Technol. 2003, 119, 301–306. [Google Scholar] [CrossRef]
- Rangel-Yagui, C.O.; Pessoa-Junior, A.; Tavares, L.C. Micellar solubilization of drugs. J. Pharm. Sci. 2005, 8, 147–163. [Google Scholar]
- Kumar, M.P.; Kumar, G.Y.S.; Apte, S.; Rao, Y.M. Review of solubilization techniques for a poorly water-soluble drug: Carbamazepine. PDA J. Pharm. Sci. Technol. 2010, 64, 264–277. [Google Scholar] [PubMed]
- Bhat, P.A.; Dar, A.A.; Rather, G.M. Solubilization capabilities of some cationic, anionic, and nonionic surfactants toward the poorly water-soluble antibiotic drug erythromycin. J. Chem. Eng. Data 2008, 53, 1271–1277. [Google Scholar] [CrossRef]
- Dannenfelser, R.-M.; Liu, R.; Li, S. Micellization and drug solubility enhancement. In Water-Insoluble Drug Formulation; Liu, R., Ed.; CRC Press: Boca Rotan, FL, USA, 2008. [Google Scholar]
- Torchilin, V.P. Structure and design of polymeric surfactant-based drug delivery systems. J. Control. Release 2001, 73, 137–172. [Google Scholar] [CrossRef] [PubMed]
- Banat, I.M.; Nigam, P.; Singh, D.; Marchant, R. Microbial decolorization of textile-dye-containing effluents: A review. Bioresour. Technol. 1996, 58, 217–227. [Google Scholar] [CrossRef]
- Kant, R. Textile dyeing industry and environmental hazard. Nat. Sci. 2012, 4, 22–26. [Google Scholar]
- Leube, H.; Ruttiger, W.; Kuhnel, G.; Wolff, J.; Ruppert, G.N.; Schmit, M.; Heid, C.; Huckel, M.; Flath, H.-J.; Beckmann, W.; Brossmann, R.; Soll, M.; Sewekow, U. Textile dyeing. In Ullmanns’ Encyclopedia of Industrial Chemistry, 6th ed.; Wiley-VCH: Weinheim, Germany, 2000. [Google Scholar]
- Mills, J. Polyester & Cotton: Unequal Competitors. Tecnon OrbiChem. Available online: http://www.afcot.org (accessed on 16 February 2013).
- Engelhardt, A. World Fiber Consumption in 2010; Oerlikon Corporation AG: Pfäffikon, Sweden, 2010. [Google Scholar]
- Holme, I. Recent developments in colorants for textile applications. Surf. Coat. Int. B 2002, 85, 243–332. [Google Scholar] [CrossRef]
- Rouette, H.K. Encyclopedia of Textile Finishing; Springer: Berlin, Germany, 2001. [Google Scholar]
- Fourness, R.K. The disperse dyes—Their development and application. J. Soc. Dye. Colour. 1956, 72, 513–527. [Google Scholar] [CrossRef]
- Giles, C.H. The coloration of synthetic polymers: A review of the chemistry of dyeing of hydrophobic fibres. Br. Polym. J. 1971, 3, 279–290. [Google Scholar] [CrossRef]
- Koh, J. Dyeing with disperse dyes. In Textile Dyeing; InTech: Rijeka, Croatia, 2011. [Google Scholar]
- Cegarra, J.; Puente, P.; Valldeperas, J. The Dyeing of Textile Materials; Texilia: Biella, Italy, 1992. [Google Scholar]
- Burkinshaw, S.M. Chemical Principles of Synthetic Fibre Dyeing; Blackie Academic & Professional, an Imprint of Chapman & Hall: London, UK, 1995. [Google Scholar]
- Steingruber, E. Indigo and indigo colorants. In Ullmann’s Encyclopedia of Industrial Chemistry; Wiley-VCH: Weinheim, Germany, 2000. [Google Scholar]
- Muthyala, R. Chemistry and Applications of Leuco Dyes; Kluwer Academic Publishers: New York, NY, USA, 2002. [Google Scholar]
- Chao, Y.C.; Chunga, Y.L.; Lai, C.C.; Liao, S.K.; Chin, J.C. Dyeing of cotton-polyester blends with anthraquinonoid vat dyes. Dye. Pigment. 1998, 40, 59–71. [Google Scholar] [CrossRef]
- Nagl, G. Sulfur dyes. In Ullmann’s Encyclopedia of Industrial Chemistry; Wiley-VCH: Weinheim, Germany, 2000. [Google Scholar]
- Wood, W.E. Sulphur dyes 1966–1976. Rev. Prog. Coloration 1976, 7, 80–84. [Google Scholar] [CrossRef]
- Guest, R.A.; Wood, W.E. Sulphur dyes. Rev. Prog. Coloration 1989, 19, 63–71. [Google Scholar] [CrossRef]
- Senior, J.; Guest, R.A.; Wood, W.E. Kirk-Othmer Encyclopedia of Chemical Technology; John Wiley and Sons, Inc.: Hoboken, NJ, USA, 2000. [Google Scholar]
- Myers, D. Surfactant Science and Technology, 3rd ed.; John Wiley and Sons, Inc.: Hoboken, NJ, USA, 2006. [Google Scholar]
- Edser, C. Status of global surfactant markets. Focus Surfactant 2008, 2008, 1–2. [Google Scholar]
- Reznik, G.O.; Vishwanath, P.; Pynn, M.A.; Sitnik, J.M.; Todd, J.J.; Wu, J.; Jiang, Y.; Keenan, B.G.; Castle, A.B.; Haskell, R.F.; Smith, T.F.; Somasundaran, P.; Jarrell, K.A. Use of sustainable chemistry to produce an acyl amino acid surfactant. Appl. Microbiol. Biotechnol. 2010, 86, 1387–1397. [Google Scholar] [CrossRef] [PubMed]
- Zemb, T.; Testard, F. Solubilization; John Wiley & Sons, Inc.: Chichester, UK, 2002. [Google Scholar]
- Schott, H. Solubilization of a water-insoluble dye. J. Phys. Chem. 1967, 71, 3611–3617. [Google Scholar] [CrossRef]
- Choi, T.; Shimizu, Y.; Shirai, H.; Hamada, K. Solubilization of disperse dyes in cationic gemini surfactant micelles. Dye. Pigment. 2000, 45, 145–152. [Google Scholar] [CrossRef]
- Tehrani-Bagha, A.R.; Singh, R.G.; Holmberg, K. Solubilization of two organic dyes by cationic ester-containing gemini surfactants. J. Colloid Interf. Sci. 2012, 376, 112–118. [Google Scholar] [CrossRef]
- Tehrani-Bagha, A.R.; Singh, R.G.; Holmberg, K. Solubilization of two organic water-insoluble dyes by anionic, cationic and nonionic surfactants. Colloid Surf. A 2013, 417, 133–139. [Google Scholar] [CrossRef]
- Skoog, D.A.; West, D.M.; Holler, F.J. Analytical Chemistry: An Introduction, 7th ed.; Saunders College Pub.: Philadelphia, PA, USA, 2000. [Google Scholar]
- Tokiwa, F. Solubilization behavior of sodium dodecylpolyoxyethylene sulfates in relation to their polyoxyethylene chain lengths. J. Phys. Chem. 1968, 72, 1214–1217. [Google Scholar] [CrossRef]
- Schott, H. Solubilization of a water-insoluble dye as a method for determining micellar molecular weights. J. Phys. Chem. 1966, 70, 2966–2973. [Google Scholar] [CrossRef]
- Anacker, E.W. Comments on the paper “Solubilization of a water-insoluble dye as a method for determining micellar molecular weights” by hans schott. J. Phys. Chem. 1968, 72, 379–380. [Google Scholar] [CrossRef]
- Herzog, B.; Huber, K.; Rennie, A.R. Characterization of worm-like micelles containing solubilized dye-molecules by light scattering techniques. J. Colloid Interf. Sci. 1994, 164, 370–381. [Google Scholar] [CrossRef]
- Birdi, K.S.; Magnusson, T. Thermodynamics of mieellar solubilization of various water-insoluble dyes. Colloid Polym. Sci. 1976, 254, 1059–1061. [Google Scholar] [CrossRef]
- Hartley, G.S. The solvent properties of aqueous solutions of paraffin-chain salts. Part I. The solubility of trans-azobenzene in solutions of cetylpyridinium salts. J. Chem. Soc. 1938, 370, 1968–1975. [Google Scholar] [CrossRef]
- Mata, J.; Varade, D.; Bahadur, P. Aggregation behavior of quaternary salt based cationic surfactants. Thermochim. Acta 2005, 428, 147–155. [Google Scholar] [CrossRef]
- Abe, A.; Imae, T.; Ikeda, S. Solubilization properties of aqueous solutions of alkyltrimethylammonium halides toward a water-insoluble dye. Colloid Polym. Sci. 1987, 265, 637–645. [Google Scholar] [CrossRef]
- Fujio, K.; Mitsui, T.; Kurumizawa, H.; Tanaka, Y.; Uzu, Y. Solubilization of a water-insoluble dye in aqueous NaBr solutions of alkylpyridinium bromides and its relation to micellar size and shape. Colloid Polym. Sci 2004, 282, 223–229. [Google Scholar] [CrossRef]
- Jacobs, P.T.; Geer, R.D.; Anacker, E.W. A comparison of micellar aggregation numbers as determined by dye solubilization and light scattering. J. Colloid Interf. Sci. 1972, 89, 611–620. [Google Scholar] [CrossRef]
- McBain, J.W.; Green, A.A. Solubilization of water-insoluble dye in soap solutions: Effects of added salts. J. Am. Chem. Soc. 1946, 68, 1731–1736. [Google Scholar] [CrossRef]
- Gerry, H.E.; Jacobs, P.T.; Anacker, E.W. The effect of fluorination on surfactant aggregation number and dye solubilization efficiency. J. Colloid Interf. Sci. 1977, 62, 556–561. [Google Scholar] [CrossRef]
- Nishikido, N. Mixed micelles of polyoxyethylene-type nonionic and anionic surfactants in aqueous solutions. J. Colloid Interf. Sci. 1977, 60, 242–251. [Google Scholar] [CrossRef]
- Tokiwa, F. Solubilization behavior of a polyoxyethylene sulfate type of surfactant in connection with the micellar charge. J. Phys. Chem. 1968, 72, 4331–4333. [Google Scholar] [CrossRef]
- Rodriguez-Hornedo, N.; Murphy, D. Surfactant-facilitated crystallization of dihydrate carbamazepine during dissolution of anhydrous polymorph. J. Pharm. Sci. 2004, 93, 449–460. [Google Scholar] [CrossRef] [PubMed]
- Zana, R.; Xia, J. Gemini Surfactants; Marcel Dekker: New York, NY, USA, 2004. [Google Scholar]
- Menger, F.M.; Keiper, J.S. Gemini surfactants. Angew. Chem. Int. Ed. 2000, 39, 1906–1920. [Google Scholar] [CrossRef]
- Gharanjig, K.; Sadeghi-Kiakhani, M.; Tehrani-Bagha, A.R.; Khosravi, A.; Menger, F.M. Solubility of two disperse dyes derived from n-alkyl and n-carboxylic acid naphthalimides in the presence of gemini cationic surfactants. J. Surfactants Deterg. 2011, 14, 381–389. [Google Scholar] [CrossRef]
- Tehrani-Bagha, A.R.; Bahrami, H.; Movassagh, B.; Arami, M.; Amirshahi, S.H.; Menger, F.M. Dynamic adsorption of gemini and conventional cationic surfactants onto polyacrylonitrile. Colloids Surf. A. 2007, 307, 121–127. [Google Scholar] [CrossRef]
- Tehrani-Bagha, A.R.; Kärnbratt, J.; Löfroth, J.-E.; Holmberg, K. Cationic ester-containing gemini surfactants: Determination of aggregation numbers by time-resolved fluorescence quenching. J. Colloid Interf. Sci. 2012, 376, 126–132. [Google Scholar] [CrossRef]
- Tehrani-Bagha, A.R.; Holmberg, K. Cationic ester-containing gemini surfactants: Adsorption at tailor-made surfaces monitored by SPR and QCM. Langmuir 2008, 24, 6140–6145. [Google Scholar] [CrossRef] [PubMed]
- Tehrani-Bagha, A.R.; Holmberg, K. Cationic ester-containing gemini surfactants: Physical—Chemical properties. Langmuir 2010, 26, 9276–9282. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, P.T.; Anacker, E.W. The role of polar head structure in dye solubilization by cationic surfactants. J. Colloid Interf. Sci. 1973, 43, 105–112. [Google Scholar] [CrossRef]
- Jacobs, P.T.; Anacker, E.W. The effect of hydroxyl functionality in the polar head on surfactant aggregation number and dyesolubilization efficiency. J. Colloid Interf. Sci. 1976, 56, 255–261. [Google Scholar] [CrossRef]
- Anacker, E.W.; Gerry, H.E. Group VA effects on surfactant aggregation number and dye solubilization efficiency. J. Colloid Interf. Sci. 1977, 60, 514–518. [Google Scholar] [CrossRef]
- Anton, P.; Laschewsky, A. Solubilization by polysoaps. Colloid Polym. Sci. 1994, 272, 1118–1128. [Google Scholar] [CrossRef]
- Hinchliffe, A.; Nikolaidi, B.; Machado, H.J.S. Density functional studies of the dipole polarizabilities of substituted stilbene, azoarene and related push-pull molecules. Int. J. Mol. Sci. 2004, 5, 224–238. [Google Scholar] [CrossRef]
- Ikeda, K.; Yasuda, M.; Ishikawa, M.; Esumi, K.; Meguro, K.; Binana-Limbele, W.; Zana, R. Physicochemical properties of α, ω-type bolaform surfactant in aqueous solution. Eicosane-1,20-bis(triethylammonium bromide). Colloid Polym. Sci. 1989, 267, 825–830. [Google Scholar] [CrossRef]
- Imae, T.; Abe, A.; Taguchi, Y.; Ikeda, S. Solubilization of a water-insoluble dye in aqueous solutions of dodecyltrimethylammonium halides, and its relation to micelle size and shape. J. Colloid Interf. Sci. 1986, 109, 567–575. [Google Scholar] [CrossRef]
- Ozeki, S.; Ikeda, S. The difference in solubilization power between spherical and rodlike micelles of dodecyldimethylammonlum chloride in aqueous solutions. J. Phys. Chem. 1985, 89, 5088–5093. [Google Scholar] [CrossRef]
- Datyner, A. The solubilization of disperse dyes by dispersing agents at 127 °C. J. Soc. Dye. Colour. 1978, 94, 256–260. [Google Scholar] [CrossRef]
- Swe, M.M.; Yu, L.E.; Hung, K.-C.; Chen, B.-H. Solubilization of selected polycyclic aromatic compounds by nonionic surfactants. J. Surfactant Deterg. 2006, 9, 237–244. [Google Scholar] [CrossRef]
- Ikeda, S.; Ozeki, S.; Tsunoda, M.-A. Micelle molecular weight of dodecyldimethylammonium chloride in aqueous solutions, and the transition of micelle shape in concentrated NaCl solutions. J. Colloid Interf. Sci. 1980, 73, 27–37. [Google Scholar] [CrossRef]
- Ozeki, S.; Ikeda, S. The viscosity behavior of aqueous NaCl solutions of dodecyldimethylammonium chloride and the flexibility of its rod-like micelle. J. Colloid Interf. Sci. 1980, 77, 219–231. [Google Scholar] [CrossRef]
- Goddard, E.D. Polymer/surfactant interaction: Interfacial aspects. J. Colloid Interf. Sci. 2002, 256, 228–235. [Google Scholar] [CrossRef]
- Goddard, E.D. Polymer/Surfactant Interaction: Manifestations, Methods, and Mechanisms, Informa Healthcare: London, UK, 1999.
- Hansson, P.; Lindman, B. Surfactant-polymer interactions. Curr. Opin. Colloid Interf. Sci. 1996, 1, 604–613. [Google Scholar] [CrossRef]
- Nizri, G.; Magdassi, S. Solubilization of hydrophobic molecules in nanoparticles formed by polymer—Surfactant interactions. J. Colloid Interf. Sci. 2005, 291, 169–174. [Google Scholar] [CrossRef]
- Ridell, A.; Evertsson, H.; Nilsson, S. Influence of counterion on the interaction of dodecyl sulfates and cellulose ethers. J. Colloid Interf. Sci. 2002, 247, 381–388. [Google Scholar] [CrossRef]
- Bahadur, P.; Sastry, N.V.; Rao, Y.K.; Riess, G. Interaction studies of styrene-ethylene oxide block copolymers with ionic surfactants in aqueous solution. Colloid Surf. 1988, 29, 343–358. [Google Scholar] [CrossRef]
- Ananthapadmanabhan, K.P.; Leung, P.S.; Goddard, E.D. Fluorescence and solubilization studies of polymer-surfactant systems. Colloid Surf. 1985, 13, 63–72. [Google Scholar] [CrossRef]
- Sudbeck, E.A.; Dubin, P.L.; Curran, I.M.E.; Skelton, J. Dye solubilization in polyelectrolyte-micelle complexes. J. Colloid Interf. Sci. 1991, 142, 512–517. [Google Scholar] [CrossRef]
- Anghel, D.F.; Saito, S.; Bǎran, A.; Iovescu, A.; Corniţescu, M. The aggregation of nonionic surfactants in the presence of poly(methacrylic acid). Colloid Polym. Sci. 2007, 285, 771–779. [Google Scholar] [CrossRef]
- Leung, P.S.; Goddard, E.D. A study of polycation-anionic surfactant systems. Colloid Surf. 1985, 13, 47–62. [Google Scholar] [CrossRef]
- Saito, S.; Taniguchi, T.; Kitamura, K. Interactions of anionic surfactants with nonionic polymers. comparison of guanidinium, tetraalkylammonium, and alkali metal ions as counterions. J. Colloid Interf. Sci. 1971, 37, 154–164. [Google Scholar] [CrossRef]
- Holmberg, C. Phase conditions, hydrodynamic features and salt influence in the polymer-amphiphile interaction for the EHEC/SDS/water system. Colloid Polym. Sci. 1996, 274, 836–847. [Google Scholar] [CrossRef]
- Persson, B.; Nilsson, S.; Sundelöf, L.-O. On the characterization principles of some technically important water-soluble nonionic cellulose derivatives. Part II: Surface tension and interaction with a surfactant. Carbohydr. Polym. 1996, 29, 119–127. [Google Scholar] [CrossRef]
- Winnik, F.M.; Regismond, S.T.A. Fluorescence methods in the study of the interactions of surfactants with polymers. Colloid Surf. A 1996, 118, 1–39. [Google Scholar] [CrossRef]
- Barbero, N.; Quagliotto, P.; Barolo, C.; Artuso, E.; Buscaino, R.; Viscardi, G. Characterization of monomeric and gemini cationic amphiphilic molecules by fluorescence intensity and anisotropy. Dye. Pigment. 2009, 83, 396–402. [Google Scholar] [CrossRef]
- Edwards, D.A.; Luthy, R.G.; Liu, Z. Solubilization of polycyclic aromatic hydrocarbons in micellar nonionic surfactant solutions. Environ. Sci. Technol. 1991, 25, 127–133. [Google Scholar] [CrossRef]
- Dar, A.A.; Rather, G.M.; Das, A.R. Mixed micelle formation and solubilization behavior toward polycyclic aromatic hydrocarbons of binary and ternary cationic-nonionic surfactant mixtures. J. Phys. Chem. B 2007, 111, 3122–3132. [Google Scholar] [CrossRef] [PubMed]
- Barry, B.W.; El-Eini, D.I.D. Solubilization of hydrocortisone, dexamethasone, testosterone and progesterone by long-chain polyoxyethylene surfactants. J. Pharm. Pharmaceut. 1976, 28, 210–218. [Google Scholar] [CrossRef]
- Tornblom, M.; Henriksson, U. Effect of solubilization of aliphatic hydrocarbons on size and shape of rodlike C16TABr micelles studied by 2H NMR relaxation. J. Phys. Chem. B 1997, 101, 6028–6035. [Google Scholar] [CrossRef]
- Karlström, G.; Halle, B. The state of water in nonionic surfactant solutions and lyotropic phases. J. Chem. Soc. Faraday Trans. 1 1989, 85, 1049–1063. [Google Scholar] [CrossRef]
- Muto, Y.; Asada, M.; Takasawa, A.; Esumi, K.; Meguro, K. The efficiency of solubilization in mixed micelles of nonionic and anionic surfactants. J. Colloid Interf. Sci. 1988, 124, 632–638. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tehrani-Bagha, A.R.; Holmberg, K. Solubilization of Hydrophobic Dyes in Surfactant Solutions. Materials 2013, 6, 580-608. https://doi.org/10.3390/ma6020580
Tehrani-Bagha AR, Holmberg K. Solubilization of Hydrophobic Dyes in Surfactant Solutions. Materials. 2013; 6(2):580-608. https://doi.org/10.3390/ma6020580
Chicago/Turabian StyleTehrani-Bagha, Ali Reza, and Krister Holmberg. 2013. "Solubilization of Hydrophobic Dyes in Surfactant Solutions" Materials 6, no. 2: 580-608. https://doi.org/10.3390/ma6020580
APA StyleTehrani-Bagha, A. R., & Holmberg, K. (2013). Solubilization of Hydrophobic Dyes in Surfactant Solutions. Materials, 6(2), 580-608. https://doi.org/10.3390/ma6020580