Characterization of Nanocomposites by Thermal Analysis
Abstract
:1. Introduction
2. Results and Discussion
2.1. Differential Scanning Calorimetry (DSC)
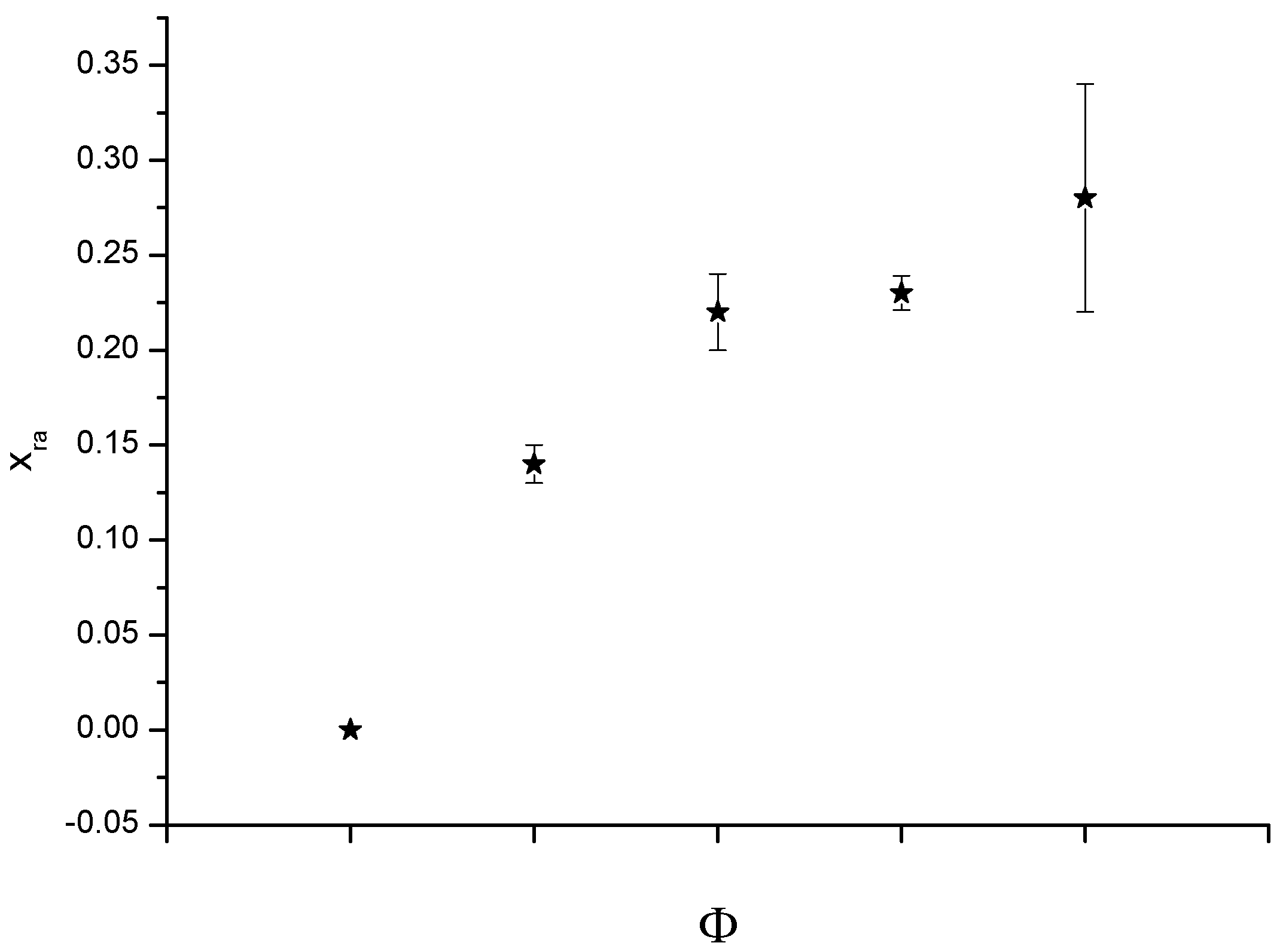
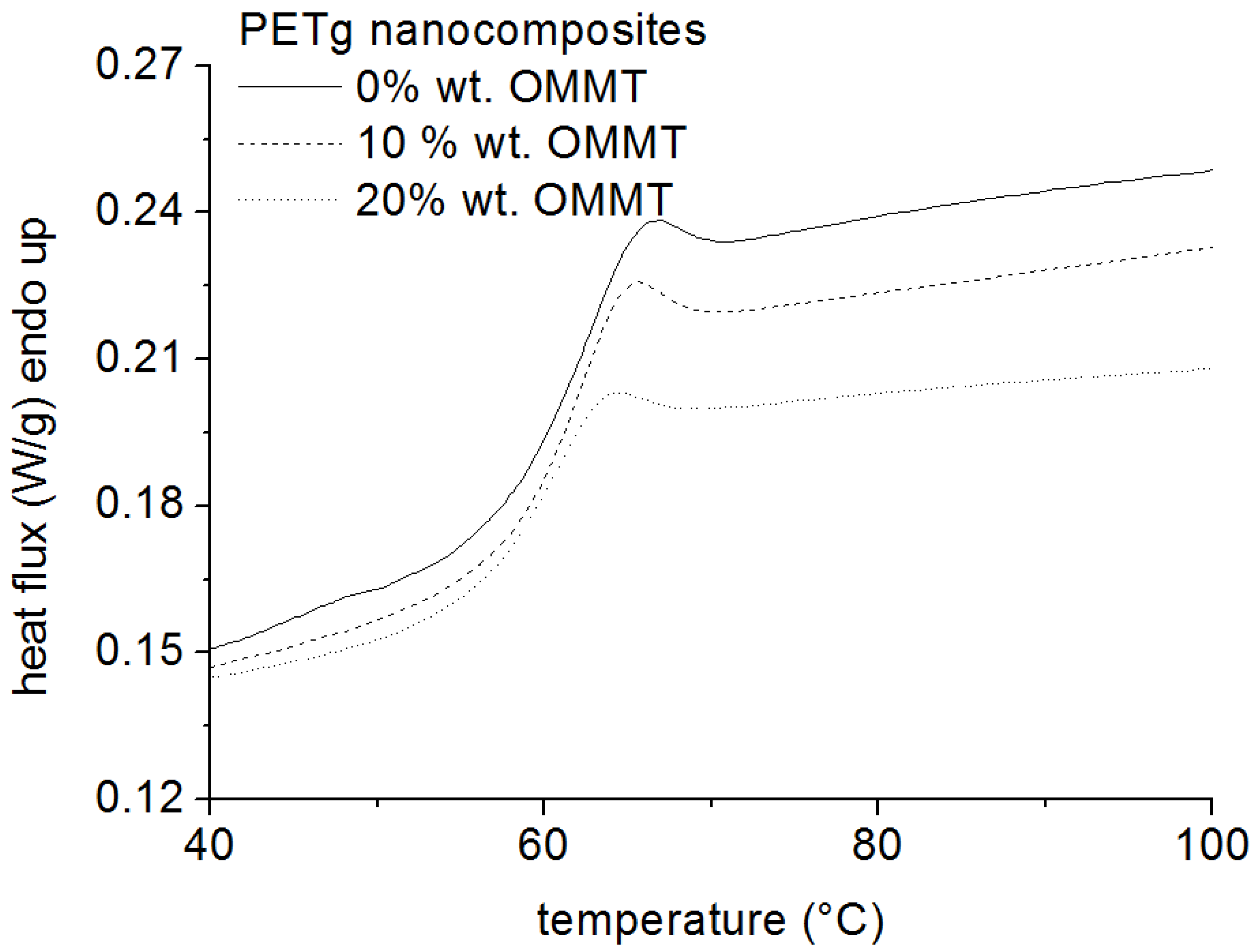
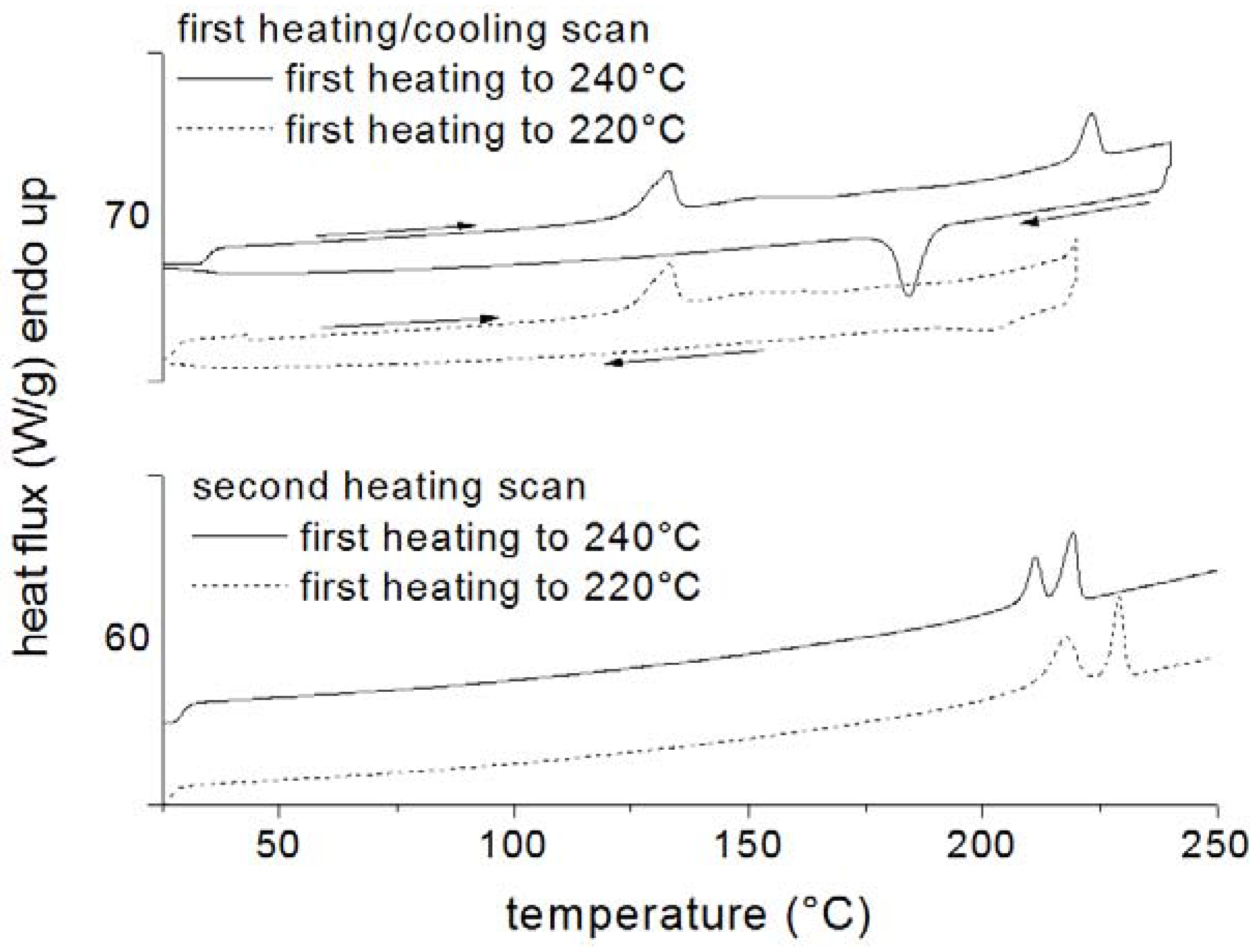
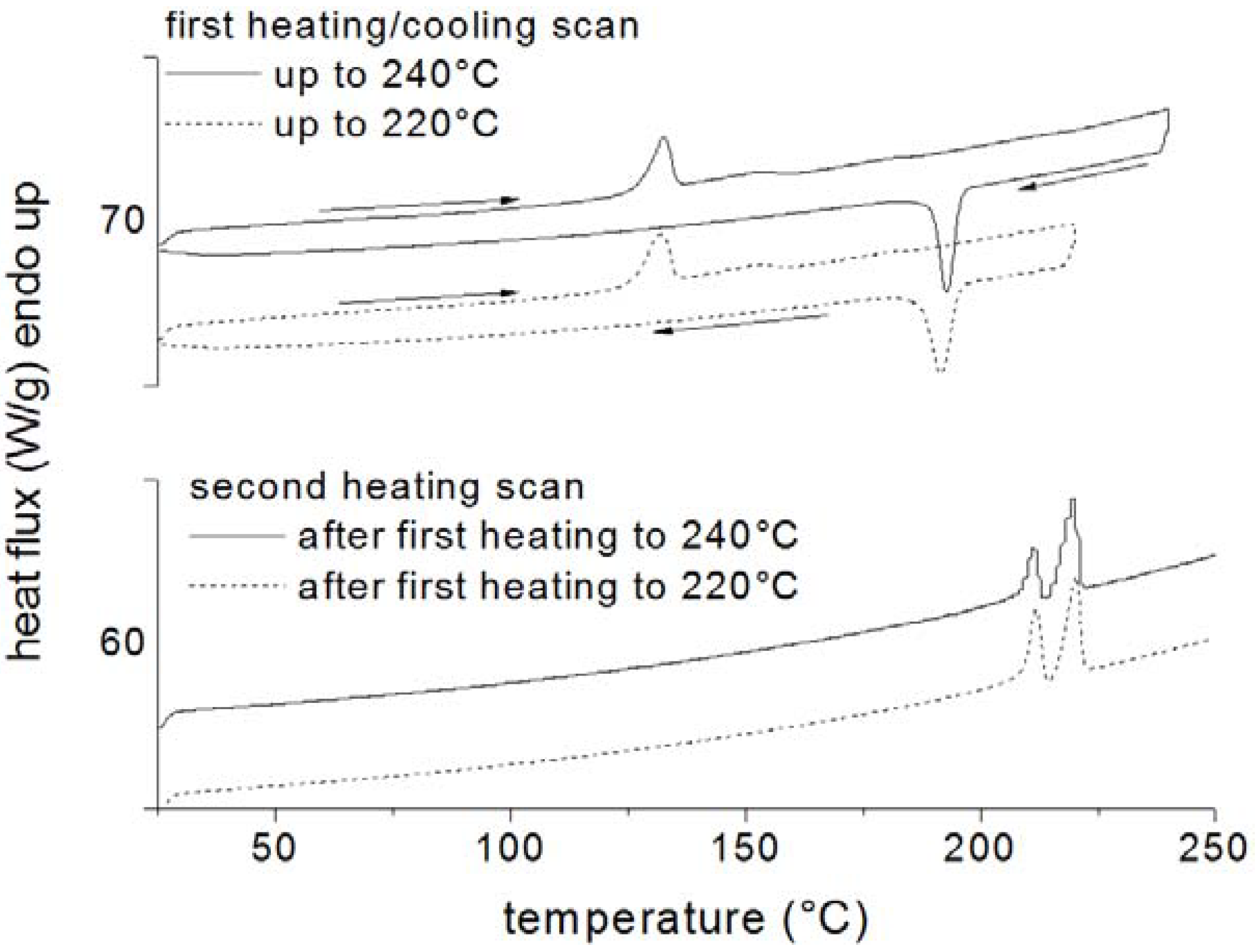
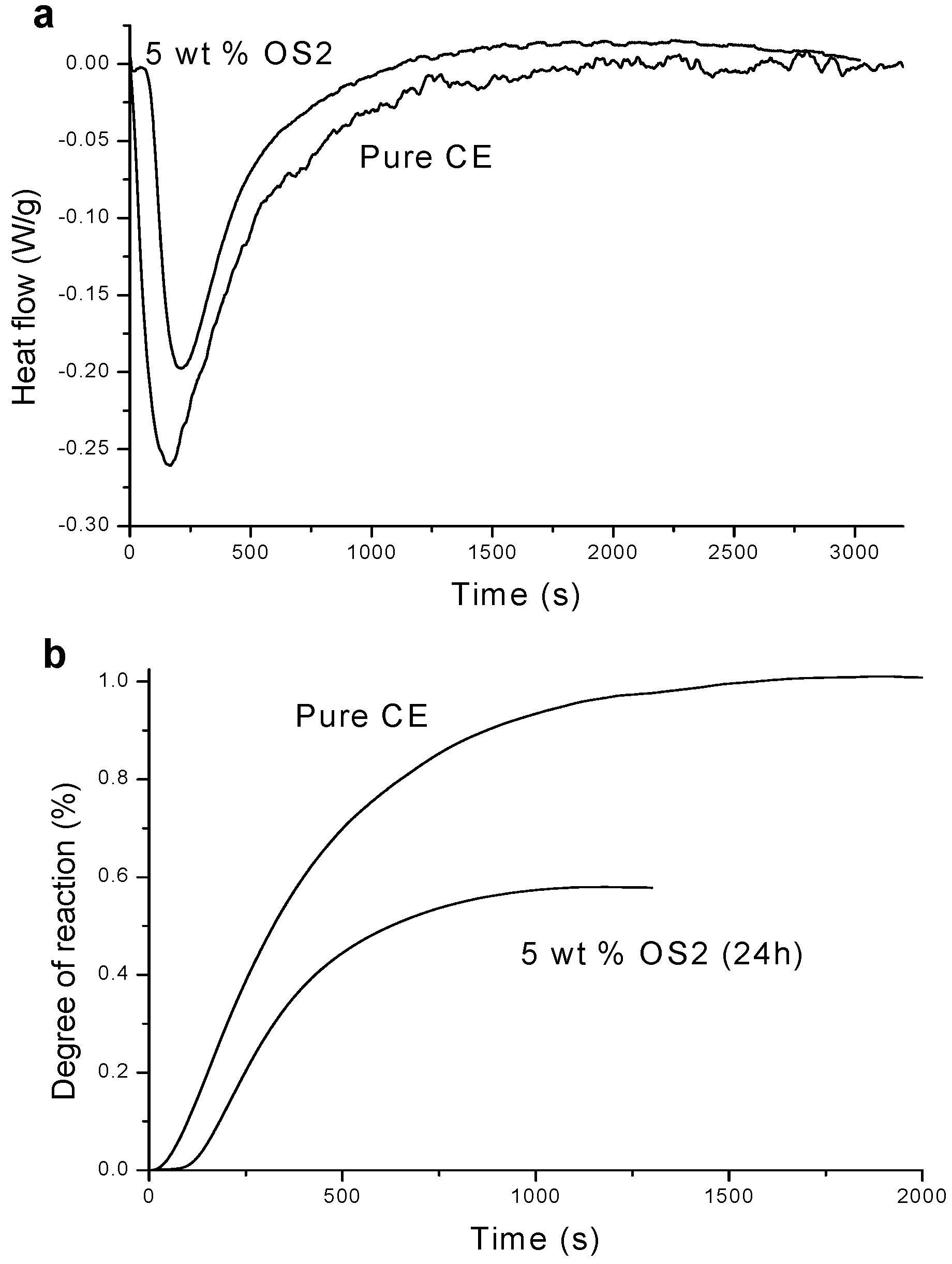
2.2. Thermogravimeric Analysis (TGA)
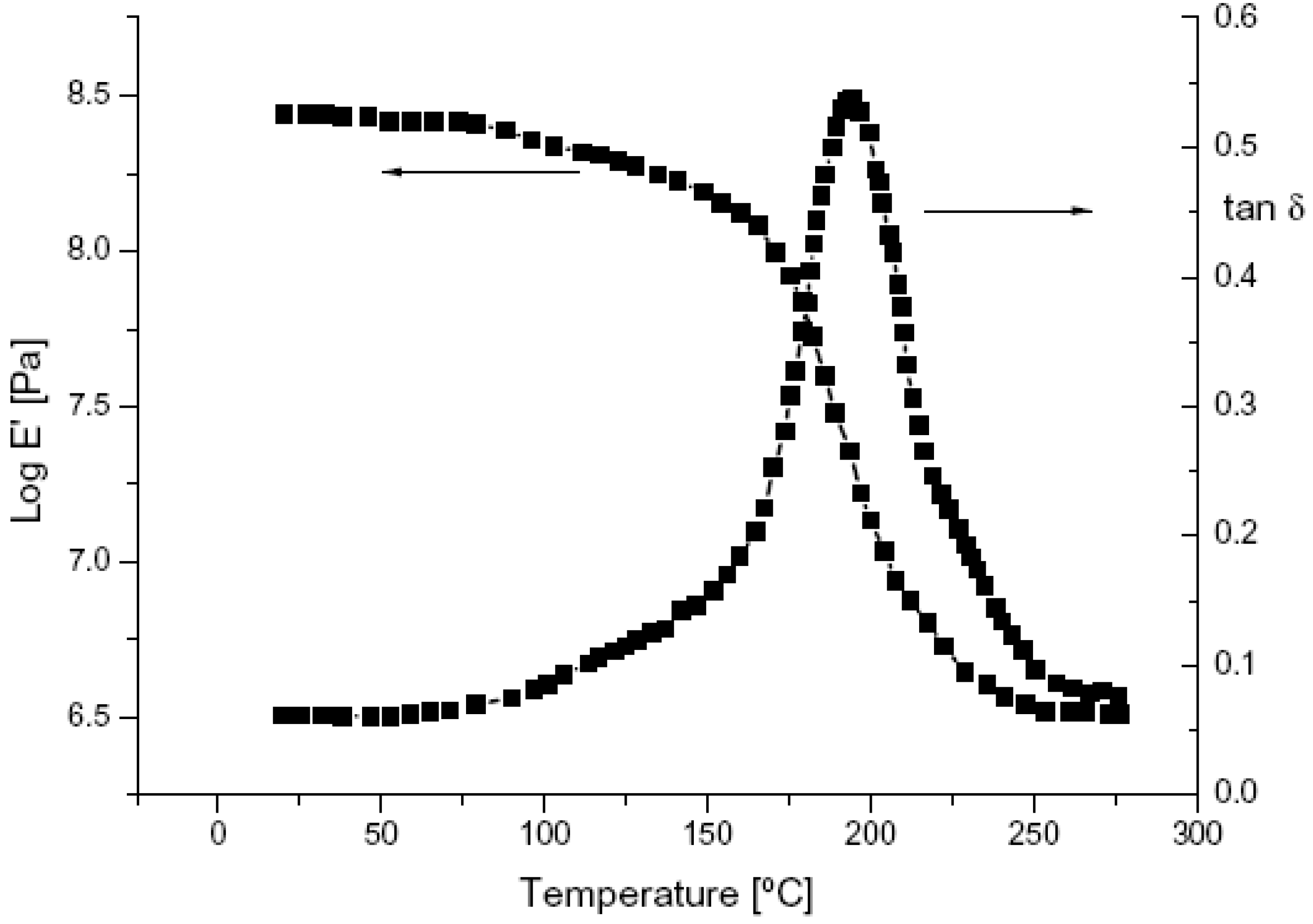
2.3. Dynamic Mechanical Thermal Analysis (DMTA)
2.4. Thermal Mechanical Analysis (TMA)
3. Conclusions
References
- Di Lorenzo, M.L.; Frigione, M. Compatibilization criteria and procedures for binary blends: A review. J. Polym. Eng. 1997, 17, 429–459. [Google Scholar]
- Lionetto, F.; Frigione, M. Mechanical and natural durability properties of wood treated with a novel organic preservative/consolidant product. Mater Des. 2009, 30, 3303–3307. [Google Scholar] [CrossRef]
- Frigione, M.; Calò, E. Influence of an hyperbranched aliphatic polyester on the cure kinetic of a trifunctional epoxy resin. J. Appl. Polym. Sci. 2008, 107, 1744–1758. [Google Scholar] [CrossRef]
- Esposito Corcione, C.; Frigione, M. A novel procedure able to predict the rheological behavior of Trifunctional epoxy resin/hyperbranched aliphatic polyester misture. Polym. Test. 2009, 28, 830–835. [Google Scholar]
- Komarneni, S.J. Nanocomposites. Mater. Chem. 1992, 2, 12–19. [Google Scholar] [CrossRef]
- Gleiter, H. Nanostructured materials. Adv. Mater. 1992, 4, 474–479. [Google Scholar] [CrossRef]
- Ziolo, R.F.; Giannelis, E.P.; Weinstein, B.A.; O’Horo, M.P.; Granguly, B.N.; Mehrota, V.; Russel, M.W.; Huffman, D.R. Matrix-mediated synthesis of nanocrystalline ggr-Fe2O3: A new optically transparent magnetic material. Science 1992, 257, 219–223. [Google Scholar] [CrossRef] [PubMed]
- Bauera, F.; Glasela, H.-J.; Hartmanna, E.; Langgutha, H.; Hinterwaldner, R. Functionalized inorganic/organic nanocomposites as new basic raw materials for adhesives and sealants. Int. J. Adhes. Adhes. 2004, 24, 519–522. [Google Scholar] [CrossRef]
- Indennidate, L.; Cannoletta, D.; Lionetto, F.; Greco, A.; Maffezzoli, A. Nanofilled polyols for viscoelastic polyurethane foams. Polym. Int. 2010, 59, 486–491. [Google Scholar] [CrossRef]
- Esposito Corcione, C.; Frigione, M.; Acierno, D. Rheological characterization of UV—Curable epoxy systems: Effects of o-Boehmite nanofillers and a hyperbranched polymeric modifier. J. Appl. Polym. Sci. 2009, 112, 1302–1310. [Google Scholar] [CrossRef]
- Esposito Corcione, C.; Frigione, M. UV—cured polymer—Boehmite nanocomposite as protective coating for wood elements. Prog. Org. Coat. 2012, 74, 781–787. [Google Scholar] [CrossRef]
- Esposito Corcione, C.; Manera, M.G.; Maffezzoli, A.; Rella, R. Synthesis and characterization of optically transparent epoxy matrix nanocomposites. Mater. Sci. Eng. C 2009, 29, 1798–1802. [Google Scholar] [CrossRef]
- Frigione, M.; Esposito Corcione, C. Rheological and kinetic characterization of UV photopolymerizable formulations as a function of the boehmite nanoparticle content. Open Mater. Sci. J. 2012, 6, 68–76. [Google Scholar] [CrossRef]
- Moniruzzaman, M.; Karen, I.W. Polymer nanocomposites containing carbon nanotubes. Macromolecules 2006, 39, 5194–5205. [Google Scholar] [CrossRef]
- Bredeau, S.; Peeterbroeck, S.; Bonduel, D.; Alexandre, M.; Dubois, P. From carbon nanotube coatings to high-performance polymer nanocomposites. Polym. Int. 2008, 57, 547–553. [Google Scholar] [CrossRef]
- Breuer, O.; Sundararaj, U. Big returns from small fibers: A review of polymer/carbon nanotube composites. Polym. Compos. 2004, 25, 641–647. [Google Scholar] [CrossRef]
- Martone, A.; Grassia, L.; Zarrelli, M.; Giordano, M.; D’Amore, A.A. Enthalpy relaxation of an epoxy matrix/carbon nanotubes. AIP Conf. Proc. 2012, 1459, 347–349. [Google Scholar]
- Grassia, L.; D’Amore, A. Calculation of the shrinkage-induced residual stress in a viscoelastic dental restorative material AIP. AIP Conf. Proc. 2012, 1459, 312–315. [Google Scholar]
- Esposito Corcione, C.; Prinari, P.; Cannoletta, D.; Mensitieri, G.; Maffezzoli, A. Synthesis and characterization of clay-nanocomposite solvent-based polyurethane adhesives. Int. J. Adhes. Adhes. 2008, 28, 91–100. [Google Scholar] [CrossRef]
- Calò, E.; Massaro, C.; Terzi, R.; Cancellara, A.; Pesce, E.; Rea, M.; Greco, A.; Maffezzoli, A.; Gonzalez-Chi, P.I.; Salomi, A. Rotational molding of polyamide-6 nanocomposites with improved flame retardancy. Int. Polym. Porcess. 2012, 27, 370–377. [Google Scholar] [CrossRef]
- Greco, A.; Maffezzoli, A.; Calò, E.; Massaro, C.; Terzi, R. An investigation into sintering of PA6 nanocomposite powders for rotational molding. J. Therm. Anal. Calorim. 2012, 109, 1493–1502. [Google Scholar] [CrossRef]
- Iijima, S. Helical microtubules of graphitic carbon. Nature 1991, 354, 56–58. [Google Scholar] [CrossRef]
- Leszczynska, A.; Pielichowski, K. Application of thermal analysis methods for characterization of polymer/montmorillonite nanocomposites. J. Therm. Anal. Calorim. 2008, 93, 677–687. [Google Scholar] [CrossRef]
- Reinhard, W.; Thomas, J.G.; Reisinger, A. rheological method to compare the degree of exfoliation of nanocomposites. Polymer 2003, 44, 7513–7518. [Google Scholar] [CrossRef]
- Serge, B.; VanderHart, D.L.; Gilman, J.W.; Awad, W.H.; Davis, R.D.; Morgan, A.B.; Wilkie, C.A. Investigation of nanodispersion in polystyrene–montmorillonite nanocomposites by solid-state NMR. J. Polym. Sci. Part B Polym. Phys. 2003, 41, 3188–3213. [Google Scholar]
- Zammarano, M.; Maupin, P.H.; Li, P.; Gilman, J.W.; McCarthy, E.D.; Kim, Y.S.; Fox, D.M. Revealing the Interface in polymer nanocomposites. ACS Nano 2011, 5, 3391–3399. [Google Scholar] [CrossRef] [PubMed]
- Ray, S.S.; Okamoto, M. Polymer/layered silicate nanocomposites: A review from preparation to processing. Prog. Polym. Sci 2003, 28, 1539–1641. [Google Scholar] [CrossRef]
- Vyazovkin, S. Thermal analysis. Anal. Chem. 2004, 76, 3299–3312. [Google Scholar] [CrossRef] [PubMed]
- Vyazovkin, S. Thermal analysis. Anal. Chem. 2006, 78, 3875–3886. [Google Scholar] [CrossRef] [PubMed]
- Vyazovkin, S. Thermal analysis. Anal. Chem. 2008, 80, 4301–4316. [Google Scholar] [CrossRef] [PubMed]
- Matusinovic, Z.; Lu, H.; Wilkie, C.A. The role of dispersion of LDH in fire retardancy: The effect of dispersion on fire retardant properties of polystyrene/Ca-Al layered double hydroxide nanocomposites. Polym. Degrad. Stab. 2012, 97, 1563–1568. [Google Scholar] [CrossRef]
- Wang, L.; He, X.; Lu, H.; Feng, J.; Xie, X.; Su, S.; Wilkie, C.A. Flame retardancy of polypropylene nanocomposites containing LDH and zinc borate. Polym. Adv. Technol. 2011, 22, 1131–1138. [Google Scholar] [CrossRef]
- Lionetto, F.; Maffezzoli, A. Polymer characterization by ultrasonic wave propagation. Adv. Polym. Technol. 2008, 27, 63–73. [Google Scholar] [CrossRef]
- Sreedhar, B.; Chattopadhyay, D.K.; Swapna, V. Thermal stability and flame retardancy of polyurethanes. Prog. Polym. Sci. 2006, 34, 1068–1133. [Google Scholar]
- Esposito Corcione, C.; Maffezzoli, A.; Mensitieri, G. Analysis of the structure and mass transport properties of nanocomposite polyurethane. Polym. Eng. Sci. 2009, 49, 1708–1718. [Google Scholar] [CrossRef]
- Esposito Corcione, C.; Maffezzoli, A. Glass transition in thermosetting clay-nanocomposite polyurethanes. Thermochim. Acta 2009, 485, 43–48. [Google Scholar] [CrossRef]
- Xia, H.; Song, M. Characteristic length of dynamic glass transition based on polymer/clay intercalated nanocomposites. Thermochim. Acta 2005, 429, 1–5. [Google Scholar] [CrossRef]
- Tian, H.; Tagaya, H. Preparation, characterization and mechanical properties of the polylactite/perlite and the polylactite/montmorillonite composites. J. Mater. Sci. 2007, 42, 3244–3250. [Google Scholar] [CrossRef]
- Greco, A.; Esposito Corcione, C.; Strafella, A.; Maffezzoli, A. Analysis of the structure and mass transport properties of clay nanocomposites based on amorphous PET. J. Appl. Polym. Sci. 2010, 118, 3666–3672. [Google Scholar] [CrossRef]
- Greco, A.; Gennaro, R.; Rizzo, M. Glass transition and cooperative rearranging regions in amorphous thermoplastic nanocomposites. Polym. Int. 2012, 61, 1326–1333. [Google Scholar] [CrossRef]
- Greco, A.; Rizzo, M.; Maffezzoli, A. Effect of the addition of organically modified nanofiller on the relaxation behavior of a thermoplastic amorphous matrix. Thermochim. Acta 2012, 534, 226–231. [Google Scholar] [CrossRef]
- Esposito Corcione, C.; Cavallo, A.; Pesce, E.; Greco, A.; Maffezzoli, A. Evaluation of the degree of dispersion of nanofillers by mechanical, rheological and permeability analysis. Polym. Eng. Sci. 2011, 51, 1280–1285. [Google Scholar] [CrossRef]
- Kattan, M.; Dargent, E.; Ledru, J.; Grenet, J. Strain-induced crystallization in uniaxially drawn PETG plates. J. Appl. Polym. Sci. 2001, 81, 3405–3412. [Google Scholar] [CrossRef]
- Lewis, S.L. Interface Control in Polymer Nanocomposites. Ph.D Dissertation, Rensselaer Polytechnic Institute, Troy, NY, USA, 2007. [Google Scholar]
- Torre, L.; Lelli, G.; Kenny, J.M. Synthesis and characterization of sPS/montmorillonite nanocomposites. J. Appl. Polym. Sci. 2006, 100, 4957–4963. [Google Scholar] [CrossRef]
- Verdonck, E.; Schaap, K.; Thomas, L.C. A discussion of the principles and applications of Modulated Temperature DSC (MTDSC). Int. J. Pharm. 1999, 192, 3–25. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Lau, K.T.; Xu, J.; Li, H. Electroless plating of copper through successive pretreatment with silane and colloidal silver. Colloid. Surface. A 2005, 257–258, 283–286. [Google Scholar]
- Drzal, L.T. The interphase in epoxy composites. Adv. Polym. Sci. 1986, 75, 1–32. [Google Scholar]
- Lanciano, G.; Greco, A.; Maffezzoli, A.; Mascia, L. Effects of thermal history in the ring opening polymerization of CBT and its mixtures with montmorillonite on the crystallization of the resulting poly(butylene terephthalate). Thermochim. Acta 2009, 493, 61–67. [Google Scholar] [CrossRef]
- Esposito Corcione, C.; Frigione, M.; Maffezzoli, A.; Malucelli, G. Photo—DSC and real time FT—IR kinetic study on a UV curable epoxy resin containing O-boehmites. Eur. Polym. J. 2008, 44, 2010–2023. [Google Scholar] [CrossRef]
- Choi, D.; Kader, M.A.; Cho, B.-H.; Huh, Y. Vulcanization kinetics of nitrile rubber/layered clay nanocomposites. J. Appl. Polym. Sci. 2005, 98, 1688–1696. [Google Scholar] [CrossRef]
- López-Manchado, M.A; Arroyo, M.; Herrero, B.; Biagiotti, J. Vulcanization kinetics of natural rubber—Organoclay nanocomposites. J. Appl. Polym. Sci. 2003, 89, 1–15. [Google Scholar] [CrossRef]
- Kader, M.A.; Nah, C. Influence of clay on the vulcanization kinetics of fluoroelastomer nanocomposites. Polymer 2004, 45, 2237–2247. [Google Scholar] [CrossRef]
- Lan, T.; Kaviratna, P.D.; Pinnavaia, T.J.J. Epoxy self-polymerization in smectite clays. Phys. Chem. Sol. 1996, 57, 1005–1010. [Google Scholar] [CrossRef]
- Ton-That, M.-T.; Ngo, T.-D.; Ding, P.; Fang, G.; Coleand, K.C.; Hoa, S.V. Epoxy nanocomposites: Analysis and kinetics of cure. Polym. Eng. Sci. 2004, 44, 1132–1141. [Google Scholar] [CrossRef]
- Seo, K.S.; Dae, S.K. Curing behavior and structure of an epoxy/clay nanocomposite system. Polym. Eng. Sci. 2006, 46, 1318–1325. [Google Scholar] [CrossRef]
- Tian, H.; Tagaya, H. Preparation, characterization and mechanical properties of the polylactide/montmo-rillonite composites. J. Mater. Sci. 2007, 42, 3244–3250. [Google Scholar] [CrossRef]
- Hikosaka, M.Y.; Pulcinelli, S.H.; Santilli, C.V.; Dahmouche, K.; Craievich, A.F. Montmorillonite (MMT) effect on the structure of poly(oxyethylene) (PEO)-MMT nanocomposites and silica-PEO-MMT hybrid materials. J. Non-Cryst. Solids 2006, 352, 3705–3710. [Google Scholar] [CrossRef]
- Gopakumar, T.G.; Lee, J.A.; Kontopoulou, M.; Parent, J.S. Influence of clay exfoliation on the physical properties of montmorillonite/polyethylene composites. Polymer 2002, 43, 5483–5491. [Google Scholar] [CrossRef]
- Morawiec, A.; Pawlak, J.; Slouf, M.; Galeski, A.; Piorkowska, E.; Krasnikowa, N. Preparation and properties of compatibilized LDPE/organo-modified montmorillonite nanocomposites. Eur. Polym. J. 2005, 41, 1115–1122. [Google Scholar] [CrossRef]
- Priya, L.; Jog, J.P. Poly(vinylidene fluoride)/clay nanocomposites prepared by melt intercalation: Crystallization and dynamic mechanical behavior studies. J. Polym. Sci. Part B Polym. Phys. 2002, 40, 1682–1689. [Google Scholar] [CrossRef]
- Tjong, S.C.; Bao, S.P. Preparation and nonisothermal crystallization behavior of polyamide 6/montmorillonite nanocomposites. J. Polym. Sci. Part B Polym. Phys. 2004, 42, 2878–2891. [Google Scholar] [CrossRef]
- Wu, J.-Y.; Wu, T.-M.; Chen, W.-Y.; Tsai, S.-J.; Kuo, W.-F.; Chang, G.-Y. Preparation and characterization of PP/clay nanocomposites based on modified polypropylene and clay. J. Polym. Sci. Part B Polym. Phys. 2005, 43, 3242–3254. [Google Scholar] [CrossRef]
- Xu, W.B.; Zhai, H.B.; Guo, H.Y.; Zhou, Z.F.; Whitely, N.; Pan, W.-P. Application of thermal analysis methods for characterization of polymer/montmorillonite nanocomposites. J. Therm. Anal. Calorim. 2004, 78, 101–107. [Google Scholar] [CrossRef]
- Wang, Y.; Shen, C.; Li, H.; Li, Q.; Chen, J. Nonisothermal melt crystallization kinetics of poly(ethylene terephthalate)/clay nanocomposites. J. Appl. Polym. Sci. 2004, 91, 308–314. [Google Scholar] [CrossRef]
- Xu, J.-T.; Wang, Q.; Fan, Z.-Q. Non-isothermal crystallization kinetics of exfoliated and intercalated polyethylene/montmorillonite nanocomposites prepared by in situ polymerization. Eur. Polym. J. 2005, 41, 3011–3017. [Google Scholar] [CrossRef]
- Chiu, F.-C.; Lai, S.-M.; Chen, Y.-L.; Lee, T.-H. Investigation on the polyamide 6/organoclay nanocomposites with or without a maleated polyolefin elastomer as a toughener. Polymer 2005, 46, 11600–11609. [Google Scholar] [CrossRef]
- Wu, T.M.; Chen, E.C.; Liao, C.S. Polymorphic behavior of nylon 6/saponite and nylon 6/montmorillonite nanocomposites. Polym. Eng. Sci. 2002, 42, 1141–1150. [Google Scholar] [CrossRef]
- González, I.; Eguiazábal, J.I.; Nazábal, J. Rubber-toughened polyamide 6/clay nanocomposites. Compos. Sci. Technol. 2006, 66, 1833–1843. [Google Scholar] [CrossRef]
- Li, T.-C.; Ma, J.; Wang, M.; Tjiu, W.C.; Liu, T.; Huang, W. Effect of clay addition on the morphology and thermal behavior of polyamide 6. J. Appl. Polym. Sci. 2007, 103, 1191–1197. [Google Scholar] [CrossRef]
- Fukushima, H.; Drzal, L.T.; Rook, B.P.; Rich, M.J. Thermal conductivity of exfoliated graphite nanocomposites. J. Therm. Anal. Calorim. 2006, 85, 235–238. [Google Scholar] [CrossRef]
- Blumstein, A. Polymerization of adsorbed monolayers. II. Thermal degradation of the inserted polymer. J. Polym. Sci. Part A 1965, 3, 2665–2672. [Google Scholar]
- Burnside, S.D.; Giannelis, E.P. Synthesis and properties of new poly(dimethylsiloxane) nanocomposites. Chem. Mater. 1995, 7, 1597–1603. [Google Scholar] [CrossRef]
- Qin, H.; Su, Q.; Zhang, S.; Zhao, B.; Yang, M. Thermal stability and flammability of polyamide 66/montmorillonite nanocomposites. Polymer 2003, 44, 7533–7538. [Google Scholar] [CrossRef]
- Ide, F.; Hasegawa, A. Studies on polymer blend of nylon 6 and polypropylene or nylon 6 and polystyrene using the reaction of polymer. J. Appl. Polym. Sci. 1974, 18, 963–974. [Google Scholar] [CrossRef]
- Zanetti, M.; Camino, G.; Peichert, P.; Mülhaupt, R. Thermal behaviour of poly(propylene) layered silicate nanocomposites. Macromol. Rapid Commun. 2001, 22, 176–180. [Google Scholar] [CrossRef]
- Leszczyńska, A.; Njuguna, J.; Pielichowski, K.; Banerjee, J.R. Polymer/montmorillonite nanocomposites with improved thermal properties: Part I. Factors influencing thermal stability and mechanisms of thermal stability improvement. Thermochim. Acta 2007, 453, 75–96. [Google Scholar] [CrossRef]
- Alexandre, M.; Dubois, P. Polymer-layered silicate nanocomposites: Preparation, propertiesand uses of a new class of materials. Mater. Sci. Eng. 2000, 28, 1–63. [Google Scholar] [CrossRef]
- Leszczy´nska, A.; Njuguna, J.; Pielichowski, K.; Banerjee, J.R. Polymer/montmorillonite nanocomposites with improved thermal properties: Part II. Thermal stability of montmorillonite nanocomposites based on different polymeric matrixes. Thermochim. Acta 2007, 454, 1–22. [Google Scholar] [CrossRef]
- Ge, J.; Hou, H.; Li, Q.; Graham Matthew, J.; Greiner, A.; Reneker Darrell, H.; Harris Frank, W.; Cheng, Z. Assembly of well-aligned multiwalled carbon nanotubes in confined polyacrylonitrile environments: Electrospun composite nanofiber sheets. J. Am.Chem. Soc. 2004, 126, 15754–15761. [Google Scholar] [CrossRef] [PubMed]
- Huxtable, S.T.; Cahill, D.G.; Shenogin, S.; Xue, L.; Ozisik, R.; Barone, P.; Usrey, M.; Strano, M.S.; Siddons, G.; Shim, M.; Keblinski, P. Interfacial heat flow in carbon nanotube suspensions. Nat. Mater. 2003, 2, 731–734. [Google Scholar] [CrossRef] [PubMed]
- Song, P.; Yu, Y.; Wu, Q.; Fu, S. Facile fabrication of HDPE-g-MA/nanodiamond nanocomposites via one-step reactive blending. Nanoscale Res. Lett. 2012, 7. [Google Scholar] [CrossRef]
- Priya, L.; Jog, J.P. Intercalated poly(vinylidene fluoride)/clay nanocomposites: Structure and properties. J. Polym. Sci. Part B Polym. Phys. 2003, 41, 31–38. [Google Scholar] [CrossRef]
- Lei, S.G.; Hoa, S.V.; Ton-That, M.-T. Effect of clay types on the processing and properties of polypropylene nanocomposites. Compos. Sci. Technol. 2006, 66, 1274–1279. [Google Scholar] [CrossRef]
- Meneghetti, P.; Qutubuddin, S. Synthesis, thermal properties and applications of polymer-clay nanocomposites. Thermochim. Acta 2006, 442, 74–77. [Google Scholar]
- Masenelli-Varlot, K.; Reynaud, E.; Vigier, G.; Varlet, J. Mechanical properties of clay-reinforced polyamide. J. Polym. Sci. Part B Polym. Phys. 2002, 40, 272–278. [Google Scholar] [CrossRef]
- Liu, X.; Wu, Q.; Berglund, L.A.; Lindberg, H.; Fan, J.; Qi, Z. Polyamide 6/clay nanocomposites using a cointercalation organophilic clay via melt compounding. J. Appl. Polym. Sci. 2003, 88, 953–958. [Google Scholar] [CrossRef]
- Lionetto, F.; Maffezzoli, A. Rheological characterization of concentrated nanoclay dispersions in an organic solvent. Appl. Rheol. 2009, 19, 23423:1–23423:8. [Google Scholar]
- Lionetto, F.; Maffezzoli, A. Relaxations during the postcure of unsaturated polyester networks by ultrasonic wave propagation, dynamic mechanical and dielectric analysis. J. Polym. Sci. Pol. Phys. 2005, 43, 596–602. [Google Scholar] [CrossRef]
- Nielsen, L.E. Mechanical Properties of Polymers and Composites; CRC: New York, NY, USA, 1994. [Google Scholar]
- Lionetto, F.; Montagna, F.; Maffezzoli, A. Ultrasonic dynamic mechanical analysis of polymers. Appl. Rheol. 2005, 15, 326–335. [Google Scholar]
- Malucelli, G.; Bongiovanni, R.; Sangermano, M.; Ronchetti, S.; Priola, A. Preparation and characterization of UV-cured epoxy nanocomposites based on o-montmorillonite modified with maleinized liquid polybutadienes. Polymer 2007, 48, 7000–7007. [Google Scholar] [CrossRef]
- Esposito Corcione, C.; Fasiello, A.; Maffezzoli, A. Synthesis and characterization of boehmite reinforced epoxy nanocomposites. J. Nanostruct. Polym. Nanocompos. 2007, 3, 82–89. [Google Scholar]
- Al Arbash, A.; Ahmad, Z.; Al Sagheer, F.; Ali, A.A.M. Microstructure and thermomechanical properties of polyimide-silica nanocomposites. J. Nanomater. 2006, 2006, 58648:1–58648:9. [Google Scholar]
- Huang, J.; Lim, P.C.; Shen, L.; Pallathadka, P.K.; Zeng, K.; He, C. Cubic silsesquioxane-polyimide nanocomposites with improved thermomechanical properties and dielectric properties. Acta Mater. 2005, 53, 2395–2404. [Google Scholar] [CrossRef]
- Bandyopadhyay, J.; Ray, S.S.; Bousmina, M.J. Thermal and thermo-mechanical properties of poly(ethylene terephthalate) nanocomposites. Ind. Eng. Chem. 2007, 13, 614–623. [Google Scholar]
- Jihua, G. Single-walled nanotube bucky paper and nanocomposite. Polym. Int. 2006, 55, 1283–1288. [Google Scholar]
- Zhao, X.; Pramoda, K.P.; Xu, G.; Goh, S.H. Dynamic mechanical behavior of melt-processed multi-walled carbon nanotube/poly(methyl methacrylate) composites. Chem. Phys. Lett. 2001, 337, 43–47. [Google Scholar] [CrossRef]
- Krump, H.; Luyt, A.S.; Hudec, I. Effect of different modified clays on the thermal and physical properties of polypropylene-montmorillonite nanocomposites. Mater. Lett. 2006, 60, 2877–2880. [Google Scholar] [CrossRef]
- Yoon, P.J.; Fornes, T.D.; Paul, D.R. Thermal expansion behavior of nylon 6 nanocomposites. Polymer 2002, 43, 6727–6741. [Google Scholar] [CrossRef]
- Shen, L.; Lin, Y.; Du, Q.; Zhnog, W. Studies on structure-property relationship of polyamide-6/attapulgite nanocomposites. Compos. Sci. Technol. 2006, 66, 2242–2248. [Google Scholar] [CrossRef]
- Krump, H.; Luyt, A.S.; Hudec, I. Effect of different modified clays on the thermal and physical properties of polypropylene-montmorillonite nanocomposites. Mater. Lett. 2006, 60, 2877–2880. [Google Scholar]
- Liang, Z.-M.; Yin, J.; Wu, J.-H.; Qiu, Z.-X.; He, F.-F. Polyimide/montmorillonite nanocomposites with photolithographic properties. Eur. Polym. J. 2004, 40, 307–314. [Google Scholar] [CrossRef]
- Fu, H.K.; Kuo, S.W.; Yeh, D.R.; Chang, F.C. Properties enhancement of PS nanocomposites through the POSS surfactants. J. Nanomater. 2008, 2008, 739613:1–739613:7. [Google Scholar] [CrossRef]
- Asif, A.; Rao, V.L.; Saseendran, V.; Ninan, K.N. Thermoplastic toughened layered silicate epoxy ternary nanocomposites—Preparation, morphology, and thermomechanical properties. Polym. Eng. Sci. 2009, 49, 756–767. [Google Scholar] [CrossRef]
- Qiu, J.; Zhang, C.; Wang, B.; Liang, R. Carbon nanotube integrated multifunctional. Compos. Nanotechnol. 2007, 18, 275708:1–275708:12. [Google Scholar]
- Liang, G.D.; Tjong, S.C. Electrical properties of low-density polyethylene/multiwalled carbon nanotube nanocomposites. Mater. Chem. Phys. 2006, 100, 132–137. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Corcione, C.E.; Frigione, M. Characterization of Nanocomposites by Thermal Analysis. Materials 2012, 5, 2960-2980. https://doi.org/10.3390/ma5122960
Corcione CE, Frigione M. Characterization of Nanocomposites by Thermal Analysis. Materials. 2012; 5(12):2960-2980. https://doi.org/10.3390/ma5122960
Chicago/Turabian StyleCorcione, Carola Esposito, and Mariaenrica Frigione. 2012. "Characterization of Nanocomposites by Thermal Analysis" Materials 5, no. 12: 2960-2980. https://doi.org/10.3390/ma5122960
APA StyleCorcione, C. E., & Frigione, M. (2012). Characterization of Nanocomposites by Thermal Analysis. Materials, 5(12), 2960-2980. https://doi.org/10.3390/ma5122960





