Mn(II) Complex with Rutin—Spectral Characteristic, Quantum-Chemical Calculations, Antioxidant and α-Amylase Inhibitory Activity
Abstract
1. Introduction
2. Materials and Methods
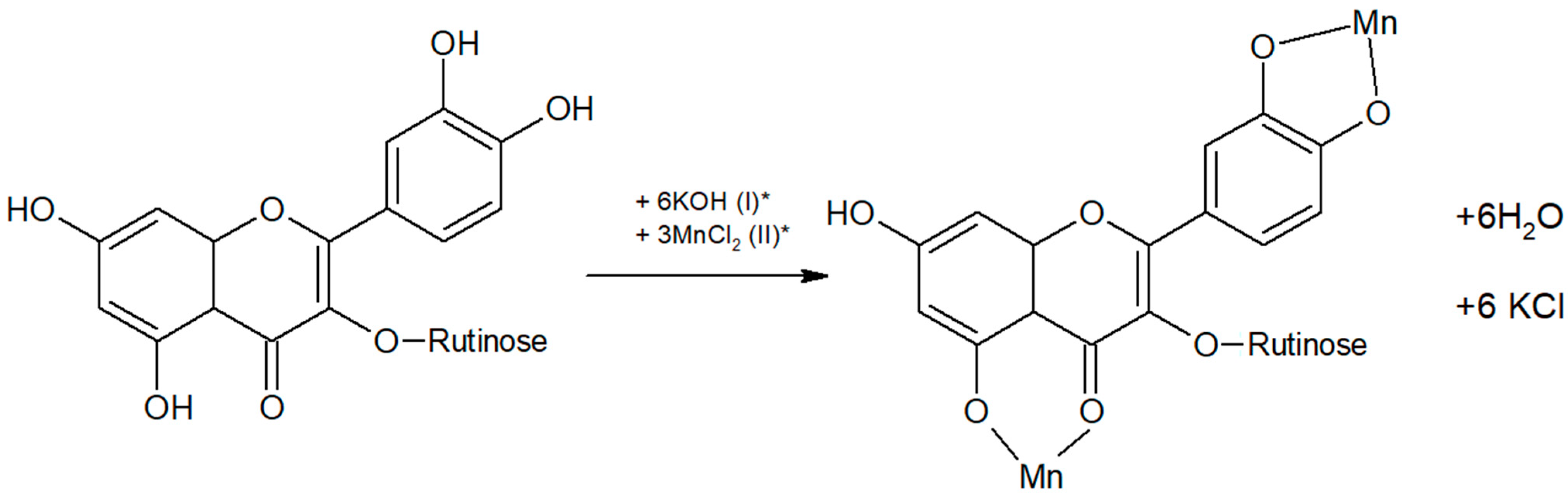
2.1. Synthesis
2.2. FT-IR Spectra
2.3. Raman Spectra
2.4. NMR Spectra
2.5. UV-Vis Spectra
2.6. Mass Spectrometry
2.7. Thermal Analysis (TG-DCS)
2.8. ABTS Assay
2.9. DPPH Assay
2.10. CUPRAC Assay
2.11. FRAP Assay
2.12. Lipid Peroxidation Inhibition Capacity
2.13. α-Amylase Inhibitory Assay
2.14. Theoretical Calculations
2.15. Statistical Analysis
3. Results and Discussion
3.1. Studies for Solid Samples
3.1.1. Elemental and Thermogravimetric Analysis
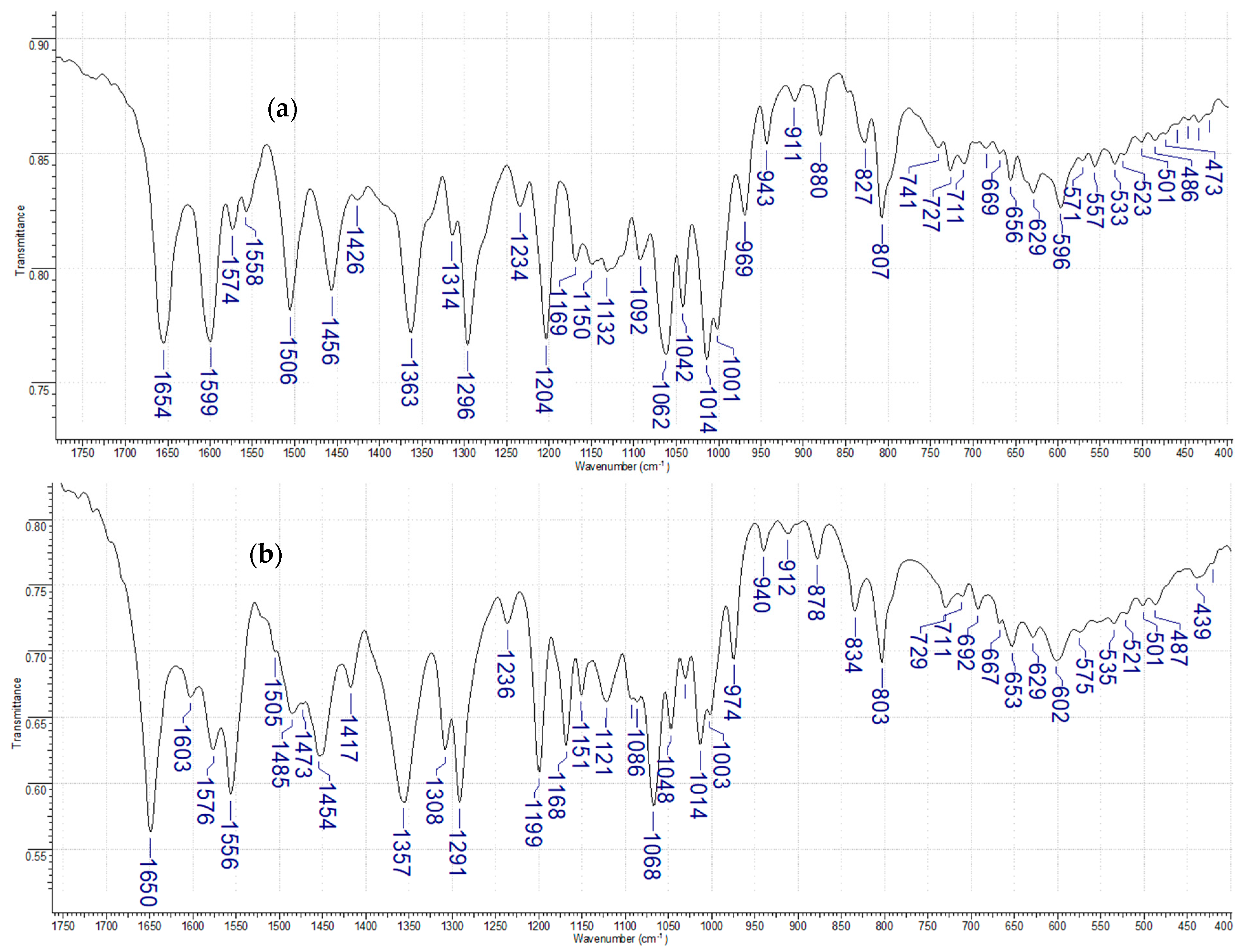
3.1.2. FT-IR and Raman Analysis
3.2. NMR Spectroscopy
3.3. Studies of Methanolic and Aqueous Solutions
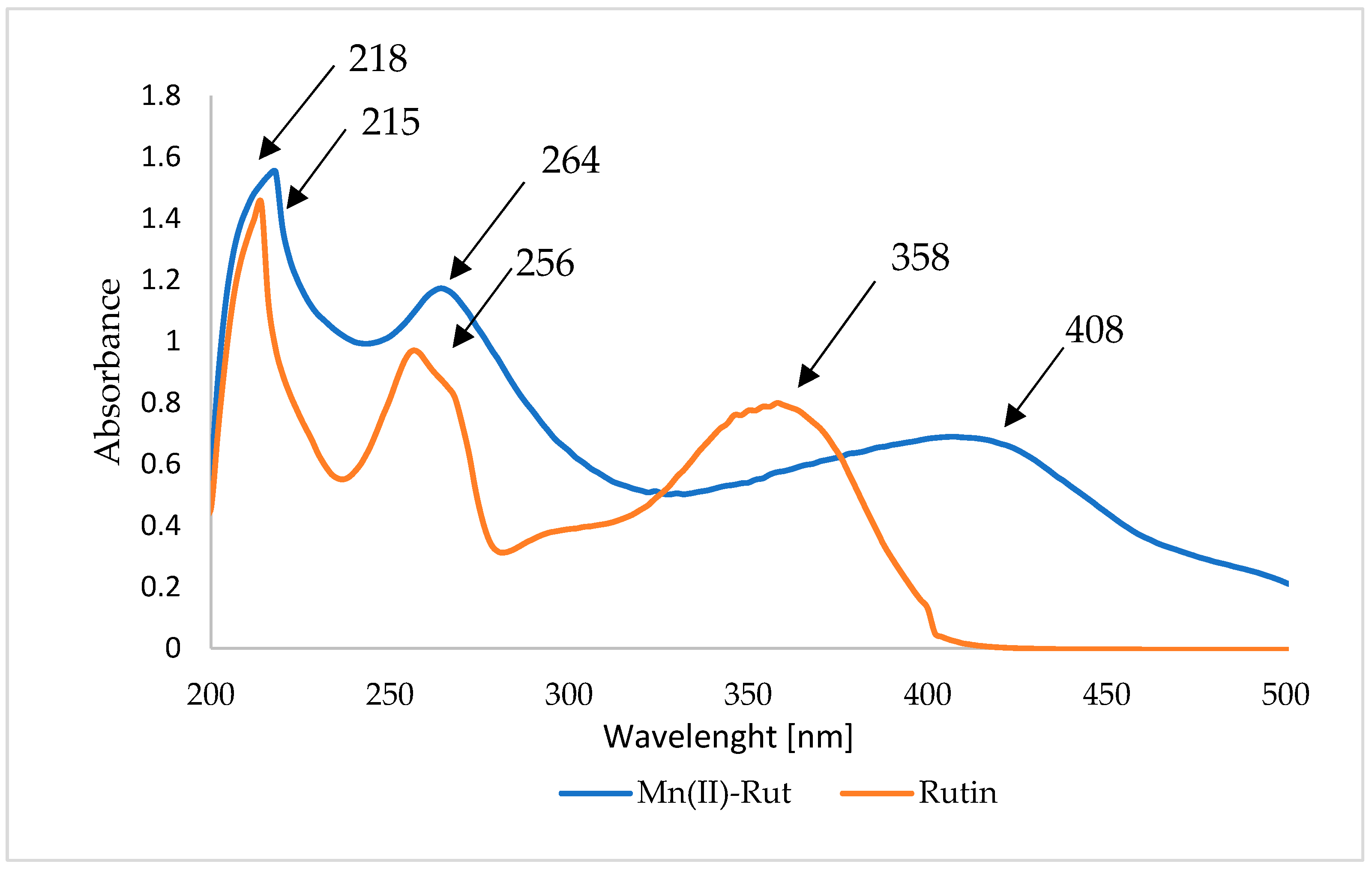
3.3.1. UV-Vis Studies
3.3.2. Mass Spectrometry
3.3.3. Antioxidant Activity
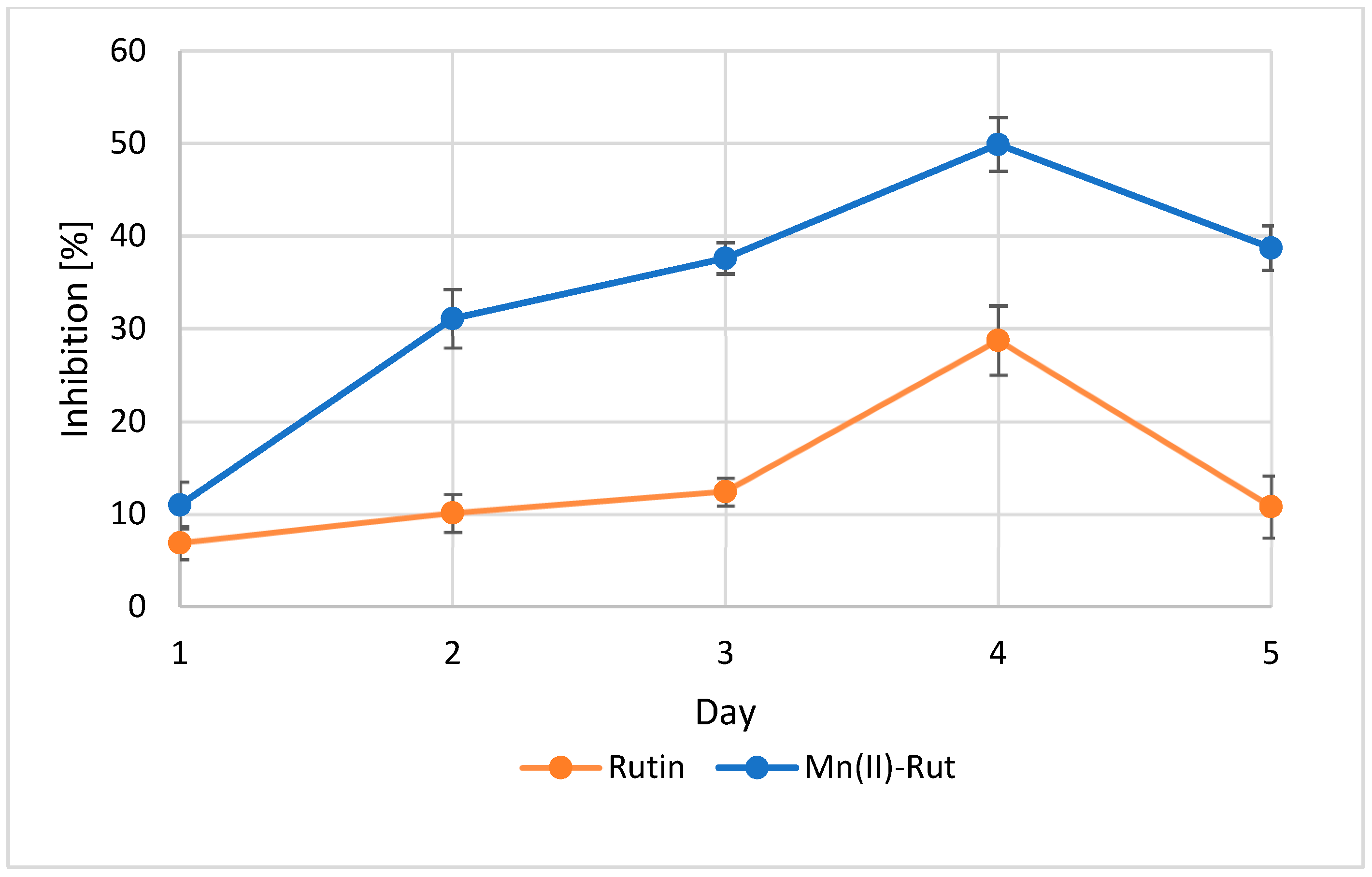
3.3.4. Enzymatic Studies α-Amylase Inhibitory Assay
3.4. Theoretical Calculations for Molecules in a Gas Phase
3.4.1. Theoretical Structure
3.4.2. Molecular Electrostatic Potential
3.4.3. HOMO–LUMO Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Harborne, J.B.; Williams, C.A. Advances in Flavonoid Research since 1992. Phytochemistry 2000, 55, 481–504. [Google Scholar] [CrossRef]
- Motoki, S.; Kitazawa, H.; Maeda, T.; Suzuki, T.; Chiji, H.; Nishihara, E.; Shinohara, Y. Effects of Various Asparagus Production Methods on Rutin and Protodioscin Contents in Spears and Cladophylls. Biosci. Biotechnol. Biochem. 2012, 76, 1047–1050. [Google Scholar] [CrossRef] [PubMed]
- Rozema, J.; Björn, L.O.; Bornman, J.F.; Gaberščik, A.; Häder, D.-P.; Trošt, T.; Germ, M.; Klisch, M.; Gröniger, A.; Sinha, R.P.; et al. The Role of UV-B Radiation in Aquatic and Terrestrial Ecosystems—An Experimental and Functional Analysis of the Evolution of UV-Absorbing Compounds. J. Photochem. Photobiol. B 2002, 66, 2–12. [Google Scholar] [CrossRef]
- Babazadeh, T.; Dianatinasab, M.; Daemi, A.; Nikbakht, H.A.; Moradi, F.; Ghaffari-fam, S. Association of Self-Care Behaviors and Quality of Life among Patients with Type 2 Diabetes Mellitus: Chaldoran County, Iran. Diabetes Metab. J. 2017, 41, 449. [Google Scholar] [CrossRef]
- Kreft, I.; Fabjan, N.; Yasumoto, K. Rutin Content in Buckwheat (Fagopyrum esculentum Moench) Food Materials and Products. Food Chem. 2006, 98, 508–512. [Google Scholar] [CrossRef]
- Musallam, I.; Duwayri, M.; Shibli, R.; Alali, F. Investigation of Rutin Content in Different Plant Parts of Wild Caper (Capparis spinosa L.) Populations from Jordan. Res. J. Med. Plants 2012, 6, 27–36. [Google Scholar] [CrossRef][Green Version]
- Kalinova, J.; Dadakova, E. Rutin and Total Quercetin Content in Amaranth (Amaranthus spp.). Plant Foods Human. Nutr. 2009, 64, 68–74. [Google Scholar] [CrossRef]
- Goyeneche, R.; Roura, S.; Ponce, A.; Vega-Gálvez, A.; Quispe-Fuentes, I.; Uribe, E.; Di Scala, K. Chemical Characterization and Antioxidant Capacity of Red Radish (Raphanus sativus L.) Leaves and Roots. J. Funct. Foods 2015, 16, 256–264. [Google Scholar] [CrossRef]
- Pandey, M.M.; Vijayakumar, M.; Rastogi, S.; Rawat, A.K.S. Phenolic Content and Antioxidant Properties of Selected Indian Spices of Apiaceae. J. Herbs Spices Med. Plants 2012, 18, 246–256. [Google Scholar] [CrossRef]
- Tao, H.; Cui, B.; Zhang, H.; Bekhit, A.E.-D.; Lu, F. Identification and Characterization of Flavonoids Compounds in Cassava Leaves (Manihot esculenta Crantz) by HPLC/FTICR-MS. Int. J. Food Prop. 2019, 22, 1134–1145. [Google Scholar] [CrossRef]
- Brazaitytė, A.; Vaštakaitė-Kairienė, V.; Sutulienė, R.; Rasiukevičiūtė, N.; Viršilė, A.; Miliauskienė, J.; Laužikė, K.; Valiuškaitė, A.; Dėnė, L.; Chrapačienė, S.; et al. Phenolic Compounds Content Evaluation of Lettuce Grown under Short-Term Preharvest Daytime or Nighttime Supplemental LEDs. Plants 2022, 11, 1123. [Google Scholar] [CrossRef]
- Slimestad, R.; Fossen, T.; Brede, C. Flavonoids and Other Phenolics in Herbs Commonly Used in Norwegian Commercial Kitchens. Food Chem. 2020, 309, 125678. [Google Scholar] [CrossRef] [PubMed]
- Wach, A.; Pyrzyńska, K.; Biesaga, M. Quercetin Content in Some Food and Herbal Samples. Food Chem. 2007, 100, 699–704. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Structure-Antioxidant Activity Relationships of Flavonoids and Phenolic Acids. Free Radic. Biol. Med. 1996, 20, 933–956. [Google Scholar] [CrossRef] [PubMed]
- Gullón, B.; Lú-Chau, T.A.; Moreira, M.T.; Lema, J.M.; Eibes, G. Rutin: A Review on Extraction, Identification and Purification Methods, Biological Activities and Approaches to Enhance Its Bioavailability. Trends Food Sci. Technol. 2017, 67, 220–235. [Google Scholar] [CrossRef]
- Yang, J.; Guo, J.; Yuan, J. In Vitro Antioxidant Properties of Rutin. LWT-Food Sci. Technol. 2008, 41, 1060–1066. [Google Scholar] [CrossRef]
- Kamalakkannan, N.; Prince, P.S.M. Rutin Improves the Antioxidant Status in Streptozotocin-Induced Diabetic Rat Tissues. Mol. Cell. Biochem. 2006, 293, 211–219. [Google Scholar] [CrossRef]
- Frutos, M.J.; Rincón-Frutos, L.; Valero-Cases, E. Rutin. In Nonvitamin and Nonmineral Nutritional Supplements; Elsevier: Amsterdam, The Netherlands, 2019; pp. 111–117. [Google Scholar]
- Samsonowicz, M.; Kamińska, I.; Kalinowska, M.; Lewandowski, W. Alkali Metal Salts of Rutin—Synthesis, Spectroscopic (FT-IR, FT-Raman, UV–VIS), Antioxidant and Antimicrobial Studies. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2015, 151, 926–938. [Google Scholar] [CrossRef]
- Roy, S.; Majumdar, S.; Singh, A.K.; Ghosh, B.; Ghosh, N.; Manna, S.; Chakraborty, T.; Mallick, S. Synthesis, Characterization, Antioxidant Status, and Toxicity Study of Vanadium–Rutin Complex in Balb/c Mice. Biol. Trace Elem. Res. 2015, 166, 183–200. [Google Scholar] [CrossRef]
- Panhwar, Q.K.; Memon, S. Synthesis, Characterization and Antioxidant Study of Tin(II)–Rutin Complex: Exploration of Tin Packaging Hazards. Inorganica Chim. Acta 2013, 407, 252–260. [Google Scholar] [CrossRef]
- Panhwar, Q.; Memon, S. Synthesis, Characterisation, and Antioxidant Study of Cr(III)-Rutin Complex. Chem. Pap. 2014, 68, 614–623. [Google Scholar] [CrossRef]
- Weinhold, F.; Landis, C.R. Natural Bond Orbitals and Extensions of Localized Bonding Concepts. Chem. Educ. Res. Pract. 2001, 2, 91–104. [Google Scholar] [CrossRef]
- Dong, J.-W.; Cai, L.; Xing, Y.; Yu, J.; Ding, Z.-T. Re-Evaluation of ABTS•+ Assay for Total Antioxidant Capacity of Natural Products. Nat. Prod. Commun. 2015, 10, 2169–2172. [Google Scholar] [CrossRef] [PubMed]
- Kedare, S.B.; Singh, R.P. Genesis and Development of DPPH Method of Antioxidant Assay. J. Food Sci. Technol. 2011, 48, 412–422. [Google Scholar] [CrossRef] [PubMed]
- Apak, R.; Güçlü, K.; Özyürek, M.; Karademir, S.E. Novel Total Antioxidant Capacity Index for Dietary Polyphenols and Vitamins C and E, Using Their Cupric Ion Reducing Capability in the Presence of Neocuproine: CUPRAC Method. J. Agric. Food Chem. 2004, 52, 7970–7981. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Diplock, A.T.; Symons, M.C.R. Techniques in Free Radical Research; Laboratory Techniques in Biochemistry and Molecular Biology; Elsevier: Amsterdam, The Netherlands, 1991; Volume 22, pp. 1–278. [Google Scholar]
- Kikuzaki, H.; Nakatani, N. Antioxidant Effects of Some Ginger Constituents. J. Food Sci. 1993, 58, 1407–1410. [Google Scholar] [CrossRef]
- Dennington Roy, Keith Todd A., and Millam John M., GaussView 6; Semichem Inc.: Shawnee, Kansas, 2016.
- Mendoza-Wilson, A.M.; Glossman-Mitnik, D. CHIH-DFT Determination of the Molecular Structure, Infrared and Ultraviolet Spectra of the Flavonoid Quercetin. J. Mol. Struct. THEOCHEM 2004, 681, 71–76. [Google Scholar] [CrossRef]
- Niu, S.-Y.; Wu, M.-L.; Hu, L.-Z.; Mei, Z.-H.; Liu, S.-F. Nucleic Acid Biosensor for DNA Hybridization Detection Using Rutin–Cu as an Electrochemical Indicator. Electrochim. Acta 2009, 54, 1564–1569. [Google Scholar] [CrossRef]
- Uivarosi, V.; Barbuceanu, S.F.; Aldea, V.; Arama, C.-C.; Badea, M.; Olar, R.; Marinescu, D. Synthesis, Spectral and Thermal Studies of New Rutin Vanadyl Complexes. Molecules 2010, 15, 1578–1589. [Google Scholar] [CrossRef]
- Ikeda, N.E.A.; Novak, E.M.; Maria, D.A.; Velosa, A.S.; Pereira, R.M.S. Synthesis, Characterization and Biological Evaluation of Rutin–Zinc(II) Flavonoid-Metal Complex. Chem. Biol. Interact. 2015, 239, 184–191. [Google Scholar] [CrossRef]
- Trouillas, P.; Marsal, P.; Siri, D.; Lazzaroni, R.; Duroux, J.-L. A DFT Study of the Reactivity of OH Groups in Quercetin and Taxifolin Antioxidants: The Specificity of the 3-OH Site. Food Chem. 2006, 97, 679–688. [Google Scholar] [CrossRef]
- Leopoldini, M.; Russo, N.; Toscano, M. The Molecular Basis of Working Mechanism of Natural Polyphenolic Antioxidants. Food Chem. 2011, 125, 288–306. [Google Scholar] [CrossRef]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized Methods for the Determination of Antioxidant Capacity and Phenolics in Foods and Dietary Supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. The Ferric Reducing Ability of Plasma (FRAP) as a Measure of “Antioxidant Power”: The FRAP Assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef]
- Samsonowicz, M.; Kalinowska, M.; Gryko, K. Enhanced Antioxidant Activity of Ursolic Acid by Complexation with Copper (II): Experimental and Theoretical Study. Materials 2021, 14, 264. [Google Scholar] [CrossRef]
- Dong, H.; Yang, X.; He, J.; Cai, S.; Xiao, K.; Zhu, L. Enhanced Antioxidant Activity, Antibacterial Activity and Hypoglycemic Effect of Luteolin by Complexation with Manganese(II) and Its Inhibition Kinetics on Xanthine Oxidase. RSC Adv. 2017, 7, 53385–53395. [Google Scholar] [CrossRef]
- Dubey, S.; Ganeshpurkar, A.; Ganeshpurkar, A.; Bansal, D.; Dubey, N. Glycolytic Enzyme Inhibitory and Antiglycation Potential of Rutin. Futur. J. Pharm. Sci. 2017, 3, 158–162. [Google Scholar] [CrossRef]
- Oboh, G.; Akinyemi, A.; Ademiluyi, A. Inhibition of α-Amylase and α-Glucosidase Activities by Ethanolic Extract of Telfairia occidentalis (Fluted Pumpkin) Leaf. Asian Pac. J. Trop. Biomed. 2012, 2, 733–738. [Google Scholar] [CrossRef]
- Aleixandre, A.; Gil, J.V.; Sineiro, J.; Rosell, C.M. Understanding Phenolic Acids Inhibition of α-Amylase and α-Glucosidase and Influence of Reaction Conditions. Food Chem. 2022, 372, 131231. [Google Scholar] [CrossRef]
- Sun, L.; Warren, F.J.; Gidley, M.J. Natural Products for Glycaemic Control: Polyphenols as Inhibitors of Alpha-Amylase. Trends Food Sci. Technol. 2019, 91, 262–273. [Google Scholar] [CrossRef]
- Yan, J.; Zhang, G.; Hu, Y.; Ma, Y. Effect of Luteolin on Xanthine Oxidase: Inhibition Kinetics and Interaction Mechanism Merging with Docking Simulation. Food Chem. 2013, 141, 3766–3773. [Google Scholar] [CrossRef]
- Roy, S.; Mallick, S.; Chakraborty, T.; Ghosh, N.; Singh, A.K.; Manna, S.; Majumdar, S. Synthesis, Characterisation and Antioxidant Activity of Luteolin–Vanadium(II) Complex. Food Chem. 2015, 173, 1172–1178. [Google Scholar] [CrossRef]
- Samsonowicz, M.; Regulska, E.; Kowczyk-Sadowy, M.; Butarewicz, A.; Lewandowski, W. The Study on Molecular Structure and Microbiological Activity of Alkali Metal 3-Hydroxyphenylycetates. J. Mol. Struct. 2017, 1146, 755–765. [Google Scholar] [CrossRef]
- Mahmoudi, S.; Dehkordi, M.M.; Asgarshamsi, M.H. Density Functional Theory Studies of the Antioxidants—A Review. J. Mol. Model. 2021, 27, 271. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Wang, Y.; Tan, X.; Wang, Y.; Li, W.; Li, X. Investigating the Antioxidant Efficiency of Tea Flavonoid Derivatives: A Density Functional Theory Study. Int. J. Mol. Sci. 2025, 26, 2587. [Google Scholar] [CrossRef] [PubMed]
- Tomasi, J.; Mennucci, B.; Cammi, R. Quantum Mechanical Continuum Solvation Models. Chem. Rev. 2005, 105, 2999–3094. [Google Scholar] [CrossRef]
- Kamel, E.M.; Bin-Ammar, A.; El-Bassuony, A.A.; Alanazi, M.M.; Altharawi, A.; Ahmeda, A.F.; Alanazi, A.S.; Lamsabhi, A.M.; Mahmoud, A.M. Molecular Modeling and DFT Studies on the Antioxidant Activity of Centaurea scoparia Flavonoids and Molecular Dynamics Simulation of Their Interaction with β-Lactoglobulin. RSC Adv. 2023, 13, 12361–12374. [Google Scholar] [CrossRef]
- Kalinowska, M.; Świsłocka, R.; Wołejko, E.; Jabłońska-Trypuć, A.; Wydro, U.; Kozłowski, M.; Koronkiewicz, K.; Piekut, J.; Lewandowski, W. Structural Characterization and Evaluation of Antimicrobial and Cytotoxic Activity of Six Plant Phenolic Acids. PLoS ONE 2024, 19, e0299372. [Google Scholar] [CrossRef]





| Complex Formula | Exp. C Content [%] | Theor. C Content [%] | Exp. H Content [%] | Theor. H Content [%] |
|---|---|---|---|---|
| C27H27O16Mn2·5H2O | 40.13 ± 0.23 | 40.12 | 4.82 ± 0.026 | 4.58 |
| Complex Formula | Stage | TG Trange/°C | DTG Tmax/°C | Mass Loss/% | Residue | |
|---|---|---|---|---|---|---|
| Calc | Found | |||||
| C27H27O16Mn2·5H2O | I dehydratation | 50–180 | 79.8 | 11.10% | 10.50% | C27H28O16Mn2 |
| II decomposition | 200–900 | 239, 412.8, 530.6 | - | 59.2% | MnO, Corg | |
| Rutin | Mn(II)-Rut | Assignment | ||||||
|---|---|---|---|---|---|---|---|---|
| IRKBR | Raman | IRKBR | Raman | |||||
| cm−1 | Int. | cm−1 | Int. | cm−1 | Int. | cm−1 | Int. | |
| 3425 | vs | 3431 | vs | v(OH)ar [21] | ||||
| 2938 | m | - | v(CH) [19] | |||||
| 2923 | m | 2941 | m | vas(CH2), vas(CH3) | ||||
| 1654 | m | 1678 | s | 1650 | s | - | v(C=O) [21] | |
| 1599 | m | 1637 | s | 1602 | m | 1553 | vs | v(CC) [30] |
| 1574 | m | 1576 | 1576 | m | 1522 | vs | ||
| 1558 | m | 1550 | 1556 | s | ||||
| 1506 | m | 1519 | 1485 | m | ||||
| 1456 | m | |||||||
| 1426 | w | 1441 | s | 1417 | m | 1424 | vs | vas(C–C-O) [30] |
| 1363 | m | 1383 | s | 1357 | s | 1353 | vs | v(C–O-C) [19] |
| 1314 | m | 1308 | m | v(C–OH) [19] | ||||
| 1296 | m | 1318 | s | 1291 | s | 1225 | s | v(C–O–C) [29] |
| 1234 | w | 1247 | m | 1236 | m | vas(O-C-C) [30] | ||
| 1204 | m | 1999 | s | v(C-O-C) [31] | ||||
| 1169 | m | 1194 | m | 1168 | m | 1147 | s | (C–CO–C) stretch and bending in ketone moiety [19] |
| 1132 | m | 1152 | 1151 | m | 1103 | m | ||
| 1092 | m | 1114 | m | 1086 | m | v(C–OH) [19] | ||
| 1062 | m | 1068 | s | v(C–O-C) [31] | ||||
| 1014 | m | 1014 | m | v(C–O–C) [31] | ||||
| 943 | m | 940 | w | γ(CC) | ||||
| 807 | m | 812 | m | 803 | m | 812 | m | γ(CH) [30] |
| 628 | w | 629 | m | γC–(OH) [30] | ||||
| - | 497 | w | v(Me–O) [21] | |||||
| Assay [Units] | Concentration of Tested Compound | Rutin | Mn(II)-Rut | p |
|---|---|---|---|---|
| DPPH [IC50]/[µM] | - | 0.98 ± 0.04 | 2.73 ± 0.11 | <0.001 |
| ABTS [%] | 5 [µM] | 41.49 ± 1.38 | 50.37 ± 2.64 | <0.05 |
| CUPRAC [mM Trolox] | 10 [µM] | 0.379 ± 0.004 | 0.468 ± 0.005 | <0.001 |
| FRAP [µM Fe2+] | 10 [µM] | 0.189 ± 0.001 | 0.201 ± 0.002 | <0.01 |
| 5 [µM] | 0.079 ± 0.003 | 0.096 ± 0.003 | <0.01 |
| Atoms 1 | Charge [e] | |
|---|---|---|
| Rutin | Mn(II)-Rut | |
| Ring B | ||
| C4′ | 0.327 | 0.314 |
| Ring C | ||
| C2 | 0.375 | 0.345 |
| C3 | 0.185 | 0.195 |
| C4 | 0.470 | 0.387 |
| C10 | −0.245 | −0.232 |
| O1 | −0.520 | −0.537 |
| O4 | −0.644 | −0.635 |
| Ring A | ||
| C6 | −0.341 | −0.354 |
| C7 | 0.390 | 0.374 |
| C8 | −0.309 | −0.325 |
| O5 | −0.760 | −0.772 |
| O7 | −0.714 | −0.725 |
| H5/Mn | 0.528 | 0.642 |
| H6 | 0.240 | 0.228 |
| Parameter | Rutin | Mn(II)-Rut |
|---|---|---|
| Chemical reactivity parameters | ||
| Energy (Hartree ) | −2250.18 | −2353.45 |
| Dipole moment (Debye) | 4.50 | 5.51 |
| EHOMO (eV) | −6.269 | −4.017 |
| ELUMO (eV) | −2.642 | −1.921 |
| ΔE(HOMO—LUMO) (eV) | 3.627 | 3.910 |
| Ionization potential, I = −EHOMO (eV) | 6.269 | 4.017 |
| Electron affinity, A = −A = −ELUMO (eV) | 2.642 | 1.921 |
| Electronegativity, (eV) | 4.455 | 2.969 |
| Electronic chemical potential, (eV) | −4.455 | −2.969 |
| Chemical hardness, (eV) | 1.814 | 1.048 |
| Chemical softness, S (eV) | 0.276 | 0.477 |
| Electrophilicity index, (eV) | 5.472 | 4.205 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Kozłowski, M.; Kalinowska, M.; Samsonowicz, M.; Świderski, G.; Kalska-Szostko, B. Mn(II) Complex with Rutin—Spectral Characteristic, Quantum-Chemical Calculations, Antioxidant and α-Amylase Inhibitory Activity. Materials 2026, 19, 1466. https://doi.org/10.3390/ma19071466
Kozłowski M, Kalinowska M, Samsonowicz M, Świderski G, Kalska-Szostko B. Mn(II) Complex with Rutin—Spectral Characteristic, Quantum-Chemical Calculations, Antioxidant and α-Amylase Inhibitory Activity. Materials. 2026; 19(7):1466. https://doi.org/10.3390/ma19071466
Chicago/Turabian StyleKozłowski, Maciej, Monika Kalinowska, Mariola Samsonowicz, Grzegorz Świderski, and Beata Kalska-Szostko. 2026. "Mn(II) Complex with Rutin—Spectral Characteristic, Quantum-Chemical Calculations, Antioxidant and α-Amylase Inhibitory Activity" Materials 19, no. 7: 1466. https://doi.org/10.3390/ma19071466
APA StyleKozłowski, M., Kalinowska, M., Samsonowicz, M., Świderski, G., & Kalska-Szostko, B. (2026). Mn(II) Complex with Rutin—Spectral Characteristic, Quantum-Chemical Calculations, Antioxidant and α-Amylase Inhibitory Activity. Materials, 19(7), 1466. https://doi.org/10.3390/ma19071466








