Synthesis of New Volatile Derivatives of Biogenic Amines, Carbamates for Analytical Applications
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Procedures
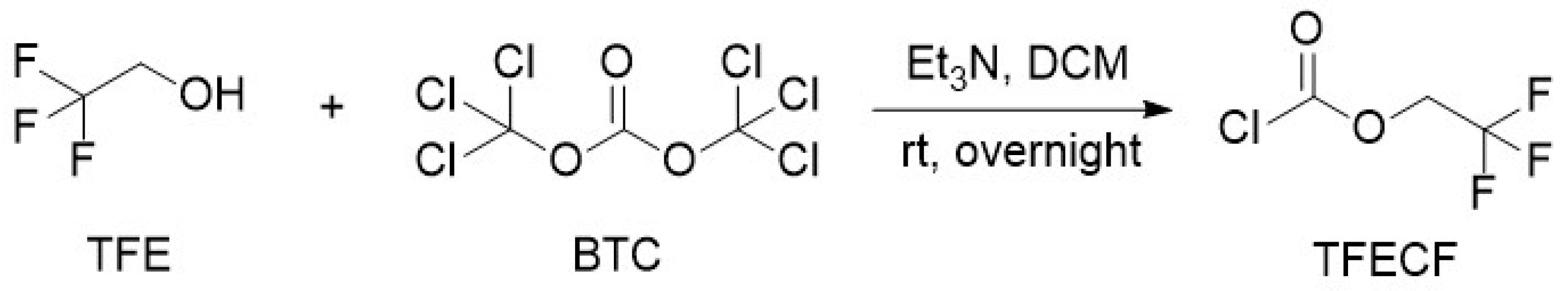
2.2.1. 2,2,2-Trifluoroethyl Chloroformate (TFECF)
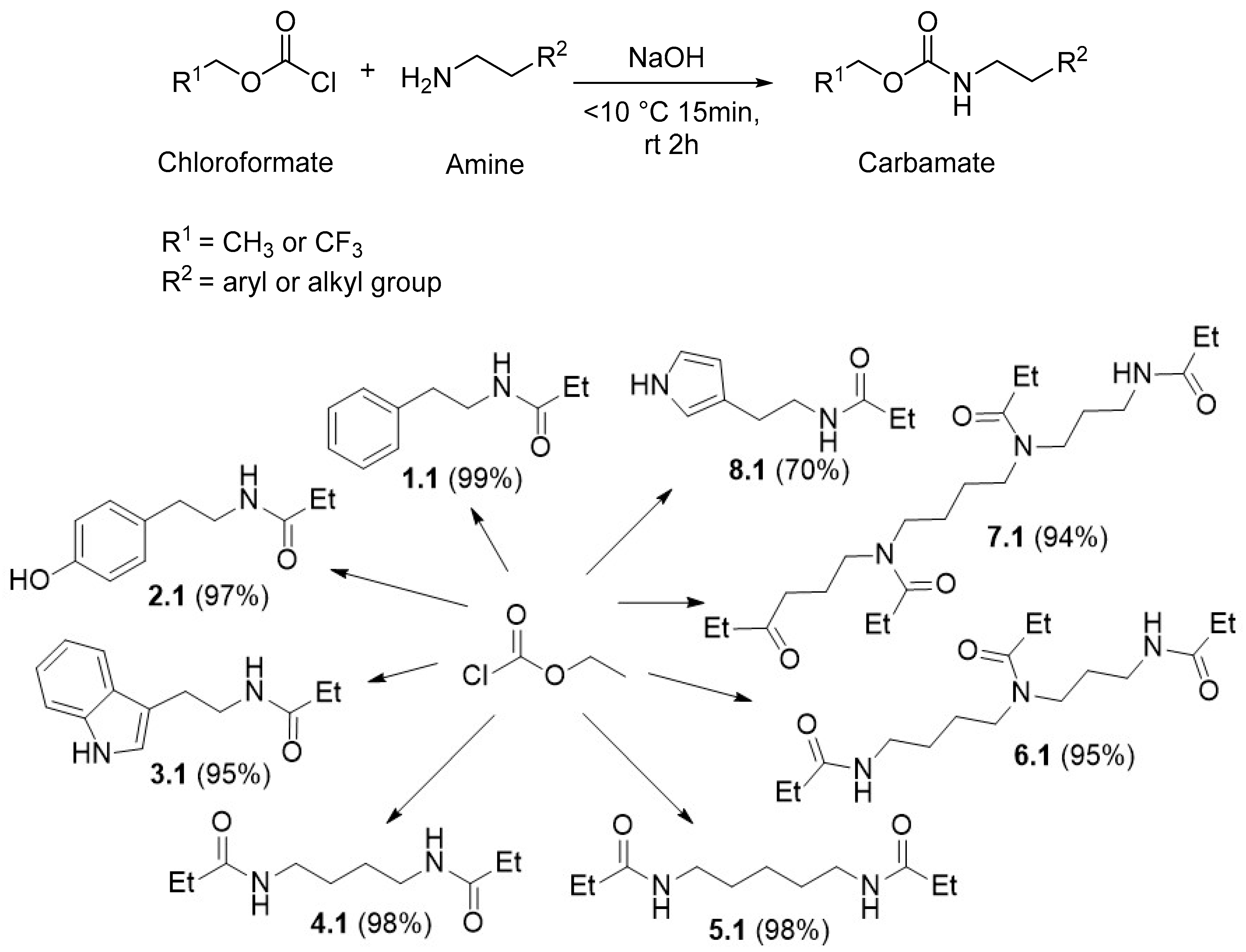
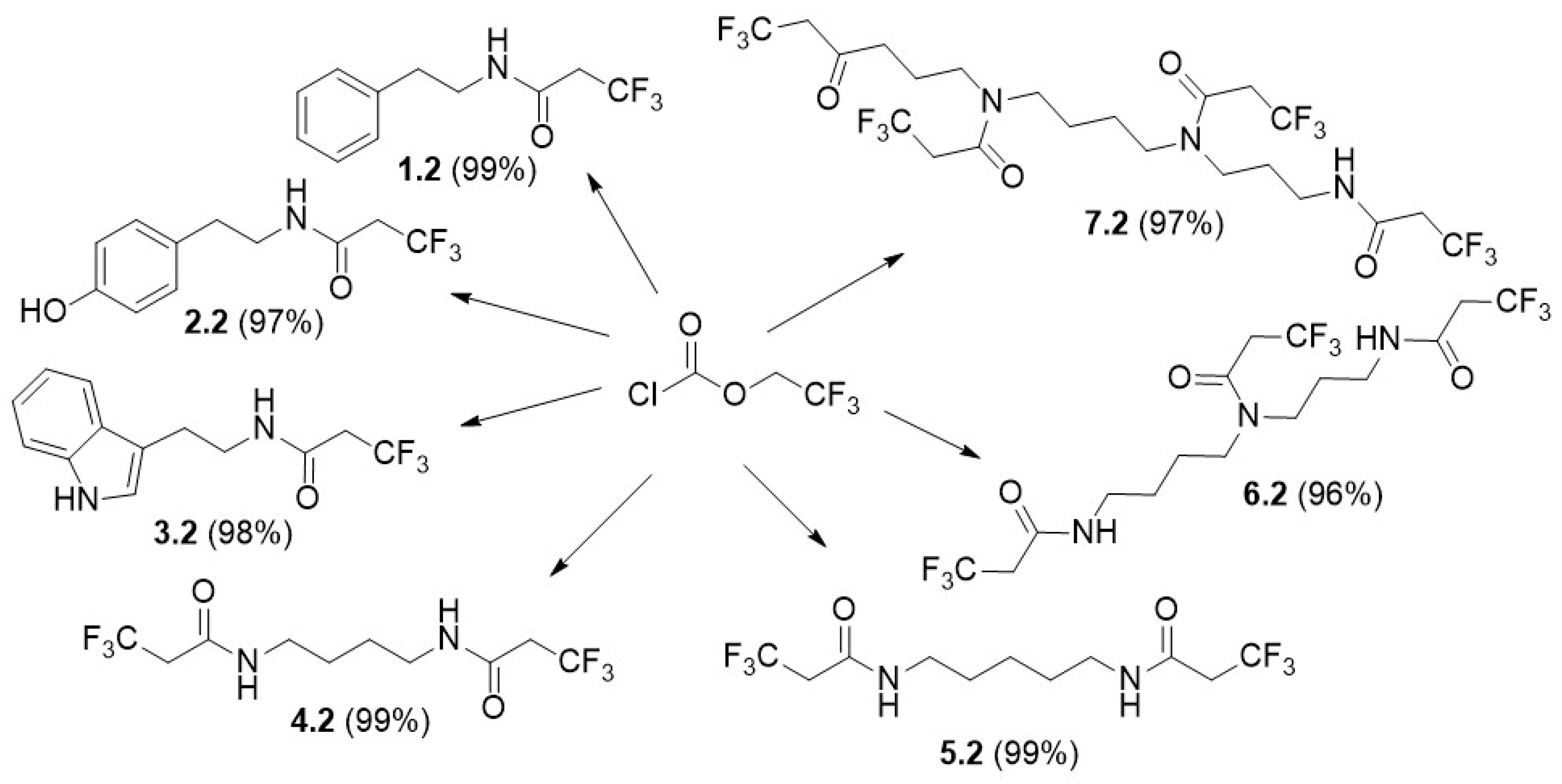
2.2.2. General Procedure for the Synthesis of the BA Carbamates
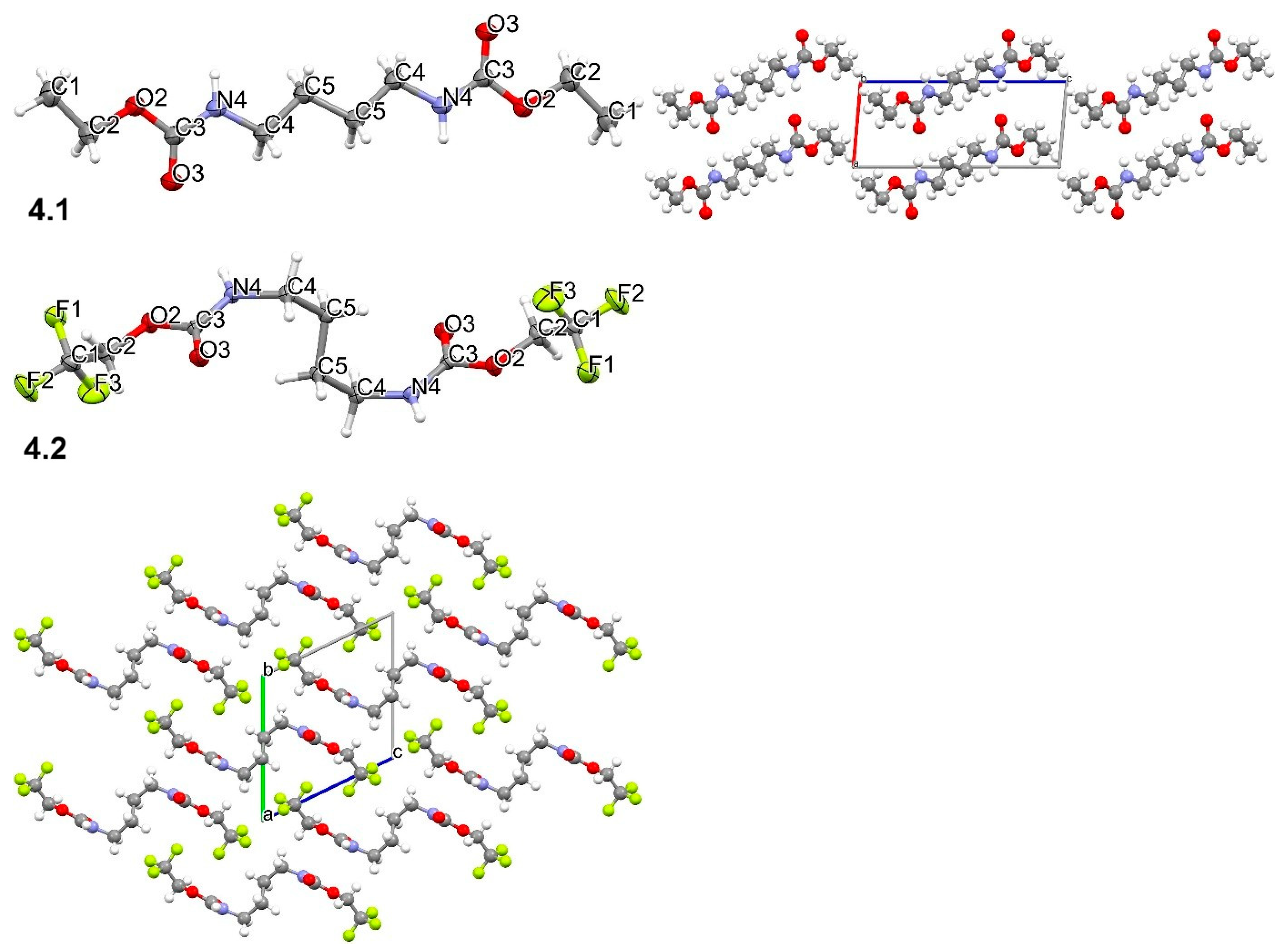
2.3. Analysis Data—1H, 13C, 19F NMR, and MS Studies and Crystal Structure Determination
Spectral Characteristics of the Obtained Derivatives
2.4. GC Analysis
2.5. Sample Preparation
2.6. Statistical Evaluation of the GC-MS Procedure
3. Results and Discussion
Application Aspect of the Obtained Derivatives
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- del Rio, B.; Fernande, M.; Redruell, B.; Ladero, V.; Alvarez, M.A. New insights into the toxicological effects of dietary biogenic amines. Food Chem. 2024, 435, 137558. [Google Scholar] [CrossRef] [PubMed]
- Xia, T.; Fu, S.; Wang, Q.; Wen, Y.; Chan, S.; Zhu, S.; Gao, S.; Tao, X.; Zhang, F.; Chen, W. Targeted metabolomic analysis of 33 amino acids and biogenic amines in human urine by ion-pairing HPLC-MS/MS: Biomarkers for tacrolimus nephrotoxicity after renal transplantation. Biomed. Chromatogr. 2018, 32, 4198. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Wang, L.; Zheng, J.; Jiang, X.; Zhang, Q.; Zhang, Y.; Bi, K.; Li, D.; Li, Q. Nanoconfinement effect based in-fiber extraction and derivatization method for ultrafast analysis of twenty amines in human urine by GC-MS: Application to cancer diagnosis biomarkers’ screening. Anal. Chim. Acta 2022, 1217, 339985. [Google Scholar] [CrossRef]
- Banicod, R.J.S.; Ntege, W.; Njiru, M.N.; Abubakar, W.H.; Kanthenga, H.T.; Javaid, A.; Khan, F. Production and transformation of biogenic amines in different food products by the metabolic activity of the lactic acid bacteria. Int. J. Food Microbiol. 2025, 428, 110996. [Google Scholar] [CrossRef]
- Mansour, S.T.; Ibrahim, H.; Zhang, J.; Farag, M.A. Extraction and analytical approaches for the determination of post-food processing major carcinogens: A comprehensive review towards healthier processed food. Food Chem. 2025, 46, 141736. [Google Scholar] [CrossRef]
- Cai, Q.; Li, X.; Ran, Z.; Li, S.; Shi, Q.; Zhang, J.; Gao, L. Simultaneous determination of 13 biogenic amines and their metabolites in liquid fermented food by HPLC-QTRAP-MS without derivatization. Microchem. J. 2024, 206, 111659. [Google Scholar] [CrossRef]
- Turna, N.S.; Chung, R.; McIntyre, L. A review of biogenic amines in fermented foods: Occurrence and health effects. Heliyon 2024, 10, 24501. [Google Scholar] [CrossRef]
- Luo, Q.; Shi, R.; Gong, P.; Liu, Y.; Chen, W.; Wang, C. Biogenic amines in Huangjiu (Chinese rice wine): Formation, hazard, detection, and reduction. LWT 2022, 168, 113952. [Google Scholar] [CrossRef]
- Chen, Z.; Xie, J.; Mei, J. A review on analytical techniques for quantitative detection of biogenic amines in aquatic products. Chemosensors 2024, 12, 274. [Google Scholar] [CrossRef]
- Ahmad, W.; Mohammed, G.I.; Al-Eryani, D.A.; Saigl, Z.M.; Alyoubi, A.O.; Alwael, H.; El-Shahawi, M.S. Biogenic amines formation mechanism and determination strategies: Future challenges and limitations. Crit. Rev. Anal. Chem. 2019, 50, 485–500. [Google Scholar] [CrossRef]
- Liu, Y.; He, Y.; Li, H.; Jia, D.; Fu, L.; Chen, J.; Zhang, D.; Wang, Y. Biogenic amines detection in meat and meat products: The mechanisms, applications, and future trends. J. Future Foods 2024, 4, 21–36. [Google Scholar] [CrossRef]
- Jain, A.; Verma, K.K. Strategies in liquid chromatographic methods for the analysis of biogenic amines without and with derivatization. TrAC Trends Anal. Chem. 2018, 109, 62–82. [Google Scholar] [CrossRef]
- Munir, M.A.; Badri, K.H. The importance of derivatizing reagent in chromatography applications for biogenic amine detection in food and beverages. J. Anal. Methods Chem. 2020, 2020, 5814389. [Google Scholar] [CrossRef] [PubMed]
- Płotka-Wasylka, J.M.; Morrison, C.; Biziuk, M.; Namieśnik, J. Chemical derivatization processes applied to amine determination in samples of different matrix composition. Chem. Rev. 2015, 115, 4693–4718. [Google Scholar] [CrossRef] [PubMed]
- Abe, I.; Fujimoto, N.; Nakahara, T. 2,2,2-Trifluoroethyl Chloroformate as a Rapid Derivatizing Reagent of Amino Acids for Fast Enantiomer Separation by Gas Chromatography. Chem. Lett. 1995, 24, 113–114. [Google Scholar] [CrossRef]
- Dalene, M.; Skarping, G.; Tinnerberg, H. Biological monitoring of hexamethylene diisocyanate by determination of 1,6-hexamethylene diamine as the trifluoroethyl chloroformate derivative using capillary gas chromatography with thermoionic and selective-ionmonitoring. J. Chromatogr. B 1994, 656, 319–328. [Google Scholar] [CrossRef]
- CrysAlis Red and CrysAlis CCD, version 1.171.35.11; Oxford Diffraction Ltd.: Oxford, UK, 2000.
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Crystallogr. Sect. C Struct. Chem. 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Macrae, C.F.; Sovago, I.; Cottrell, S.J.; Galek, P.T.A.; McCabe, P.; Pidcock, E.; Platings, M.; Shields, G.P.; Stevens, J.S.; Towler, M.; et al. Mercury 4.0: From visualization to analysis, design and prediction. J. Appl. Cryst. 2020, 53, 226–235. [Google Scholar] [CrossRef]
- Spackman, P.R.; Turner, M.J.; McKinnon, J.J.; Wolff, S.K.; Grimwood, D.J.; Jayatilaka, D.; Spackman, M.A. CrystalExplorer: A program for Hirshfeld surface analysis, visualization and quantitative analysis of molecular crystals. J. Appl. Cryst. 2021, 54, 1006–1011. [Google Scholar] [CrossRef]
- Spackman, M.A.; McKinnon, J.J. Fingerprinting intermolecular interactions in molecular crystals. CrystEngComm 2002, 4, 378–392. [Google Scholar] [CrossRef]
- Spackman, M.A.; Jayatilaka, D. Hirshfeld surface analysis. CrystEngComm 2009, 11, 19–32. [Google Scholar] [CrossRef]
- Mackenzie, C.F.; Spackman, P.R.; Jayatilaka, D.; Spackman, M.A. CrystalExplorer model energies and energy framworks: Extension to metal coordination compounds, organic salts, solvates and open-shell systems. IUCrJ 2017, 4, 575–587. [Google Scholar] [CrossRef]
- Becke, A.D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef]
- Stephens, P.J.; Devlin, F.J.; Chabalowski, C.F.; Frisch, M.J. Ab Initio Calculation of Vibrational Absorption and Circular Dichroism Spectra Using Density Functional Force Fields. J. Phys. Chem. 1994, 98, 11623–11627. [Google Scholar] [CrossRef]
- Becke, A.D. Density-functional exchange-energy approximation with correct asymptotic behavior. Phys. Rev. A 1988, 38, 3098–3100. [Google Scholar] [CrossRef]
- Lee, C.; Yang, W.; Parr, R.G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 1988, 37, 785–789. [Google Scholar] [CrossRef]
- Stephens, M.D.; Nisakorn Yodsanit, N.; Melander, C. Evaluation of ethyl N-(2-phenethyl) carbamate analogues as biofilm inhibitors of methicillin resistant Staphylococcus aureus. Org. Biomol. Chem. 2016, 14, 6853–6856. [Google Scholar] [CrossRef]
- Chappell, M.D.; Mitch, C.H.; Quimby, S.J.; Siegel, M.G. Opioid Receptor Antagonists. WO 2005/092836 A1, 6 October 2005. [Google Scholar]
- Mahale, S.; Bharate, S.B.; Manda, S.; Joshi, P.; Bharate, S.S.; Jenkins, P.R.; Vishwakarma, R.A.; Chaudhuri, B. Biphenyl-4-carboxylic acid [2-(1H-Indol-3-yl)-ethyl]-methylamide (CA224), a nonplanar analogue of fascaplysin, inhibits Cdk4 and Tubulin polymerization: Evaluation of in vitro and in vivo anticancer activity. J. Med. Chem. 2014, 57, 9658–9672. [Google Scholar] [CrossRef]
- Murato, K.; Shioiri, T.; Yamada, S.-I. Amino acids and peptides. XXVII. A novel reductive cleavage of N-C-N bonds with sodium borohydride. Chem. Pharm. Bull. 1977, 25, 1559–1565. [Google Scholar] [CrossRef][Green Version]
- Ogbu, I.M.; Lusseau, J.; Kurtay, G.; Robert, F.; Landais, Y. Urethanes synthesis from oxamic acids under electrochemical conditions. Chem. Commun. 2020, 56, 12226–12229. [Google Scholar] [CrossRef] [PubMed]
- Hušek, P. Chloroformates in gas chromatography as general purpose derivatizing agents. J. Chromatogr. B Biomed. Sci. Appl. 1998, 717, 57–91. [Google Scholar] [CrossRef]
- Cunha, S.C.; Faria, M.A.; Fernandes, J.O. Gas chromatography mass spectrometry assessment of amines in port wine and grape juice after fast chloroformate extraction/derivatization. J. Agric. Food Chem. 2011, 59, 8742–8753. [Google Scholar] [CrossRef] [PubMed]
- An, X.D.; Jiao, Y.Y.; Zhang, H.; Gao, Y.; Yu, S. Photoredox-induced radical relay toward functionalized β-amino alcohol derivatives. Org. Lett. 2018, 20, 401–404. [Google Scholar] [CrossRef] [PubMed]
- Neverov, A.A.; Cimpean, L.; Chiykowski, V.; Vance, T.; Brown, R.S. Cu(II)-promoted methanolysis of n,n-bis(2-picolyl)carbamates: Rate-limiting metal ion delivery of coordinated alcoholate nucleophile followed by fast partitioning of a tetrahedral intermediate. J. Org. Chem. 2015, 80, 1357–1364. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, D. Sampling chamber with minimal wall surface for simultaneous emission testing of diisocyanates and diamines from polyurethane product. Toxicol. Ind. Health 2023, 39, 313–324. [Google Scholar] [CrossRef]
- Cremonesi, S.; Micheli, F.; Semeraro, T.; Tarsi, L. Dopamine D3 Receptor Antagonists Having a Bicyclo Moiety. WO 2017/021920 A1, 9 February 2017. [Google Scholar]
- Flynn, D.; Petillo, P.; Kaufman, M. Kinase Inhibitors Useful for the Treatment of Myleoproliferative Diseases and Other Proliferative Diseases. US 2008/0269254 A1, 30 October 2008. [Google Scholar]
- Çelik, E.E.; Canli, M.; Kocadağl, T.; Kanmaz, E.Ö.; Gökmen, V. Formation of histamine, phenylethylamine and γ-aminobutyric acid during sprouting and fermenting of selected wholegrains. Food Res. Int. 2023, 173, 113447. [Google Scholar] [CrossRef]
- Świder, O.; Roszko, M.Ł.; Wójcicki, M.; Szymczyk, K. Biogenic amines and free amino acids in traditional fermented vegetables—Dietary risk evaluation. J. Agric. Food Chem. 2020, 68, 856–868. [Google Scholar] [CrossRef]
- Jastrzębska, A.; Kmieciak, A.; Brzuzy, K.; Gralak, Z.; Krzemiński, M.P.; Szłyk, E. Determination of selected biogenic amines in fermented vegetables juices. Food Control 2023, 154, 109980. [Google Scholar] [CrossRef]



| BAs Derivatives | RT [min] | BAs Derivatives | RT [min] |
|---|---|---|---|
| 1.1 | 2.17 | 2.1 | 1.56 |
| 2.1 | 11.53 | 2.2. | 6.87 |
| 3.1 | 14.57 | 3.2 | 8.61 |
| 4.1 | 23.44 | 4.2 | 20.28 |
| 5.1 | 39.22 | 5.2 | 24.54 |
| 6.1 | 45.73 | 6.2 | 29.48 |
| T[min] ±SD | Range [mg/L] | R2 | DL [mg/L] | QL [mg/L] | Rec ±SD [%] | Intra-Day Precision [CV, %]* | ME | |
|---|---|---|---|---|---|---|---|---|
| 1.1 | 6.06 ± 0.004 | 1.0–10.0 | 0.9964 | 0.51 | 1.55 | 96 ± 9.33 * | 4.62 * | 5.22 |
| 1.2 | 4.43 ± 0.003 | 0.9964 | 0.53 | 1.61 | 97 ± 11.98 * | 3.97 * | −9.05 |
| Sample | TFECF | ECF | One-Way Anova | ||
|---|---|---|---|---|---|
| X ± SD [mg/L] | CV [%] | X ± SD [mg/L] | CV [%] | p | |
| S1 | 1.2738 ± 0.0364 | 2.85 | 1.4840 ± 0.1008 | 6.79 | 0.002329 |
| S2 | 6.7815 ± 0.1028 | 1.52 | 5.7414 ± 0.3777 | 6.58 | 0.0003456 |
| S3 | 0.5096 ± 0.0230 | 4.52 | 0.5318 ± 0.0527 | 8.03 | 0.337 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Brzuzy, K.; Jastrzębska, A.; Kmieciak, A.; Ścianowski, J.; Muzioł, T.; Gorczyca, D.; Krzemiński, M.P. Synthesis of New Volatile Derivatives of Biogenic Amines, Carbamates for Analytical Applications. Materials 2026, 19, 575. https://doi.org/10.3390/ma19030575
Brzuzy K, Jastrzębska A, Kmieciak A, Ścianowski J, Muzioł T, Gorczyca D, Krzemiński MP. Synthesis of New Volatile Derivatives of Biogenic Amines, Carbamates for Analytical Applications. Materials. 2026; 19(3):575. https://doi.org/10.3390/ma19030575
Chicago/Turabian StyleBrzuzy, Kamil, Aneta Jastrzębska, Anna Kmieciak, Jacek Ścianowski, Tadeusz Muzioł, Damian Gorczyca, and Marek P. Krzemiński. 2026. "Synthesis of New Volatile Derivatives of Biogenic Amines, Carbamates for Analytical Applications" Materials 19, no. 3: 575. https://doi.org/10.3390/ma19030575
APA StyleBrzuzy, K., Jastrzębska, A., Kmieciak, A., Ścianowski, J., Muzioł, T., Gorczyca, D., & Krzemiński, M. P. (2026). Synthesis of New Volatile Derivatives of Biogenic Amines, Carbamates for Analytical Applications. Materials, 19(3), 575. https://doi.org/10.3390/ma19030575










