(Li0.4Co0.2Ni0.2Cu0.2Zn0.2)WO4: A Novel High-Entropy Wolframite Ceramic with Tailored Microwave Dielectric Properties
Abstract
1. Introduction
2. Experimental Procedure
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Yin, C.Z.; Yin, Y.H.; Du, K.; Song, X.Q.; Guo, H.H.; Xiong, Y.; Cheng, M.F.; Yang, J.Q.; Luo, W.; Lu, W.Z.; et al. Fabrication of high-efficiency dielectric patch antennas from temperature-stable Sr3-xCaxV2O8 microwave dielectric ceramic. J. Eur. Ceram. Soc. 2023, 43, 1492–1499. [Google Scholar] [CrossRef]
- Bao, J.; Zhang, Y.P.; Kimura, H.D.; Wu, H.T.; Yue, Z.X. Crystal structure, chemical bond characteristics, infrared reflection spectrum, and microwave dielectric properties of Nd2(Zr1-xTix)3(MoO4)9 ceramics. J. Adv. Ceram. 2023, 12, 82–92. [Google Scholar] [CrossRef]
- Xiang, H.C.; Kilpijärvi, J.; Myllymäki, S.; Yang, H.T.; Fang, L.; Jantunen, H. Spinel-olivine microwave dielectric ceramics with low sintering temperature and high quality factor for 5 GHz wi-fi antennas. Appl. Mater. Today 2020, 21, 100826. [Google Scholar] [CrossRef]
- Du, Q.; Chen, Z.; Wei, K.; Liu, J.; Ma, L.; Li, H. Effect of phase composition on the crystal structure and microwave dielectric properties of ZnMg2TiO5 ceramics. J. Eur. Ceram. Soc. 2025, 45, 116944. [Google Scholar] [CrossRef]
- Jiang, J.; Fang, D.; Lu, C.; Dou, Z.; Wang, G.; Zhang, F.; Zhang, T. Solid-state reaction mechanism and microwave dielectric properties of CaTiO3–LaAlO3 ceramics. J. Alloys Compd. 2015, 638, 443–447. [Google Scholar] [CrossRef]
- Wang, X.; Zhu, X.L.; Li, L.; Chen, X.M. Structure evolution and adjustment of (Ba, Sr)HfO3 and (Sr, Ca)HfO3 microwave dielectric ceramics. J. Am. Ceram. Soc. 2024, 107, 285–299. [Google Scholar] [CrossRef]
- Mohanty, P.; Keshri, S.; Sinha, M.K.; Gupta, V.R. Study on microwave dielectric properties of corundum type (Mg1−xCox)4Ta2O9 (x = 0–0.6) ceramics for designing a microwave low pass filter. Ceram. Int. 2016, 42, 5911–5920. [Google Scholar] [CrossRef]
- Tang, Y.; Zhang, Z.W.; Li, J.; Xu, M.Y.; Zhai, Y.F.; Duan, L.; Su, C.X.; Liu, L.J.; Sun, Y.H.; Fang, L. A3Y2Ge3O12 (A = Ca, Mg): Two novel microwave dielectric ceramics with contrasting τf and Q × f. J. Eur. Ceram. Soc. 2020, 40, 3989–3995. [Google Scholar] [CrossRef]
- Zhang, N.; Chen, J.Q.; Zhang, Y.H.; Xiang, H.C.; Tang, Y.; Fang, L. Crystal structure, bond characterization and lattice energy of B-site 1:3 ordering inverse spinel Li1.25Ga0.25Ti1.5O4 ceramic. Ceram. Int. 2024, 50, 126–133. [Google Scholar] [CrossRef]
- Liu, K.; Zhang, H.W.; Liu, C.; Li, J.; Shi, L.; Wang, X.Y.; Zhang, D.N. Crystal structure and microwave dielectric properties of (Mg0.2Ni0.2Zn0.2Co0.2Mn0.2)2SiO4 A novel high-entropy ceramic. Ceram. Int. 2022, 48, 23307–23313. [Google Scholar] [CrossRef]
- Yao, Y.; Wang, Y.Y.; Bafrooei, H.B.; Mao, M.M.; Liu, B.; Lu, Z.L.; Lin, H.X.; Spreitzer, M.; Wang, D.W.; Zheng, X.H.; et al. Ni-Co Complex Ionic Synergistically Modifying Octahedron Lattice of Cordierite Ceramics for the Application of 5G Microwave-Millimeter-wave Antennas. Inorg. Chem. 2024, 63, 10022–10030. [Google Scholar] [CrossRef]
- Song, Z.J.; Song, K.X.; Liu, B.; Zheng, P.; Bafrooei, H.B.; Su, W.T.; Lin, H.X.; Shi, F.; Wang, D.W.; Reaney, I.M. Temperature-dependent dielectric and Raman spectra and microwave dielectric properties of gehlenite-type Ca2Al2SiO7 ceramics. Int. J. Appl. Ceram. Technol. 2020, 17, 771–777. [Google Scholar] [CrossRef]
- Ramarao, S.D.; Kiran, S.R.; Murthy, V.R.K. Structural, morphological and microwave dielectric studies on microwave sintered ZnWO4 ceramic compounds. Ceram. Int. 2023, 49, 23075–23081. [Google Scholar] [CrossRef]
- Zhang, Q.; Su, H.; Huang, F.; Wu, X.; Li, Y.; Tang, X. Effect of B-site ion substitution on chemical bond characteristics and microwave dielectric properties of ZnWO4 ceramics. J. Eur. Ceram. Soc. 2021, 41, 6502–6507. [Google Scholar] [CrossRef]
- Tian, H.; Zheng, J.; Liu, L.; Wu, H.; Kimura, H.; Lu, Y.; Yue, Z. Structure characteristics and microwave dielectric properties of Pr2(Zr1−Ti )3(MoO4)9 solid solution ceramic with a stable temperature coefficient. J. Mater. Sci. Technol. 2022, 116, 121–129. [Google Scholar] [CrossRef]
- An, Z.; Lv, J.; Wang, X.; Xu, Y.; Zhang, L.; Shi, F.; Guo, H.; Zhou, D.; Liu, B.; Song, K. Effects of LiF additive on crystal structures, lattice vibrational characteristics and dielectric properties of CaWO4 microwave dielectric ceramics for LTCC applications. Ceram. Int. 2022, 48, 29929–29937. [Google Scholar] [CrossRef]
- Zhang, Y.; Xiang, H.; Wu, X.; Zhou, Y.; Tang, Y.; Fang, L. Low dielectric loss in vanadium-based zircon ceramics via high-entropy strategy. J. Adv. Ceram. 2025, 14, 9221012. [Google Scholar] [CrossRef]
- Tian, H.; Zhang, X.; Du, W.; Feng, Z.; Wang, L.; Wu, H.; Xia, W. Structure characteristics and microwave/terahertz dielectric response of low-permittivity (La0.2Nd0.2Sm0.2Eu0.2Gd0.2)2Zr3(MoO4)9 high-entropy ceramics. Ceram. Int. 2024, 50, 6403–6411. [Google Scholar] [CrossRef]
- Song, X.Q.; Du, K.; Li, J.; Lan, X.K.; Lu, W.Z.; Wang, X.H.; Lei, W. Low-fired fluoride microwave dielectric ceramics with low dielectric loss. Ceram. Int. 2019, 45, 279–286. [Google Scholar] [CrossRef]
- Lai, Y.M.; Su, H.; Wang, G.; Tang, X.L.; Huang, X.; Liang, X.F.; Zhang, H.W.; Li, Y.X.; Huang, K.; Wang, X.R. Low-temperature sintering of microwave ceramics with high Qf values through LiF addition. J. Am. Ceram. Soc. 2019, 102, 1893–1903. [Google Scholar] [CrossRef]
- Vaidya, M.; Muralikrishna, G.M.; Murty, B.S. High-entropy alloys by mechanical alloying: A review. J. Mater. Res. 2019, 34, 664–686. [Google Scholar] [CrossRef]
- Emil, E.; Gürmen, S. Estimation of yttrium oxide microstructural parameters using the Williamson-Hall analysis. Mater. Sci. Technol. 2018, 34, 1549–1557. [Google Scholar] [CrossRef]
- Hirthna; Sendhilnathan, S. Enhancement in dielectric and magnetic properties of Mg2+ substituted highly porous super paramagnetic nickel ferrite nanoparticles with Williamson-Hall plots mechanistic view. Ceram. Int. 2017, 43, 15447–15453. [Google Scholar] [CrossRef]
- Zhang, P.; Wu, S.X.; Xiao, M. The microwave dielectric properties and crystal structure of low temperature sintering LiNiPO4 ceramics. J. Eur. Ceram. Soc. 2018, 38, 4433–4439. [Google Scholar] [CrossRef]
- Xiao, K.; Tang, Y.; Tian, Y.F.; Li, C.C.; Duan, L.; Fang, L. Enhancement of the cation order and the microwave dielectric properties of Li2ZnTi3O8 through composition modulation. J. Eur. Ceram. Soc. 2019, 39, 3064–3069. [Google Scholar] [CrossRef]
- Tian, Y.F.; Tang, Y.; Xiao, K.; Li, C.C.; Duan, L.; Fang, L. Crystal structure, Raman spectroscopy and microwave dielectric properties of Li1+xZnNbO4 (0 ≤ x ≤ 0.05) ceramics. J. Alloys Compd. 2019, 777, 1–7. [Google Scholar] [CrossRef]
- Li, M.T.; Meng, Q.H.; Li, S.Y.; Li, F.; Zhu, Q.; Kim, B.N.; Li, J.G. Photoluminescent and photocatalytic ZnWO4 nanorods via controlled hydrothermal reaction. Ceram. Int. 2019, 45, 10746–10755. [Google Scholar] [CrossRef]
- Pereira, P.F.S.; Gouveia, A.F.; Assis, M.; de Oliveira, R.C.; Pinatti, I.M.; Penha, M.; Gonçalves, R.F.; Gracia, L.; Andrès, J.; Longo, E. ZnWO4 nanocrystals: Synthesis, morphology, photoluminescence and photocatalytic properties. Phys. Chem. Chem. Phys. 2018, 20, 1923–1937. [Google Scholar] [CrossRef]
- Xia, W.S.; Li, L.X.; Ning, P.F.; Liao, Q.W. Relationship Between Bond Ionicity, Lattice Energy, and Microwave Dielectric Properties of Zn(Ta1-xNbx)2O6 Ceramics. J. Am. Ceram. Soc. 2012, 95, 2587–2592. [Google Scholar] [CrossRef]
- Wu, Z.J.; Zhang, S.Y. Semiempirical method for the evaluation of bond covalency in complex crystals. J. Phys. Chem. A 1999, 103, 4270–4274. [Google Scholar] [CrossRef]
- Zhang, Q.; Tang, X.L.; Zhong, M.F.; Li, Y.X.; Jing, Y.L.; Su, H. Bond characteristics and microwave dielectric properties of high-Q materials in li-doped Zn3B2O6 systems. J. Am. Ceram. Soc. 2021, 104, 2102–2115. [Google Scholar] [CrossRef]
- Batsanov, S.S. Dielectric methods of studying the chemical bond and the concept of electronegativity. Russ. Chem. Rev. 1982, 51, 684–697. [Google Scholar] [CrossRef]
- Guo, H.H.; Zhou, D.; Pang, L.X.; Qi, Z.M. Microwave dielectric properties of low firing temperature stable scheelite structured (Ca,Bi)(Mo,V)O4 solid solution ceramics for LTCC applications. J. Eur. Ceram. Soc. 2019, 39, 2365–2373. [Google Scholar] [CrossRef]
- Khobragade, N.; Sinha, E.; Rout, S.K.; Kar, M. Structural, optical and microwave dielectric properties of Sr1-xCaxWO4 ceramics prepared by the solid state reaction route. Ceram. Int. 2013, 39, 9627–9635. [Google Scholar] [CrossRef]
- Wang, S.Y.; Zhang, Y.C. Structure, bond characteristics and microwave dielectric properties of new A0.75Ti0.75Ta1.5O6 (A = Ni, Co, Zn and Mg) ceramics based on complex chemical bond theory. J. Eur. Ceram. Soc. 2020, 40, 1181–1185. [Google Scholar] [CrossRef]

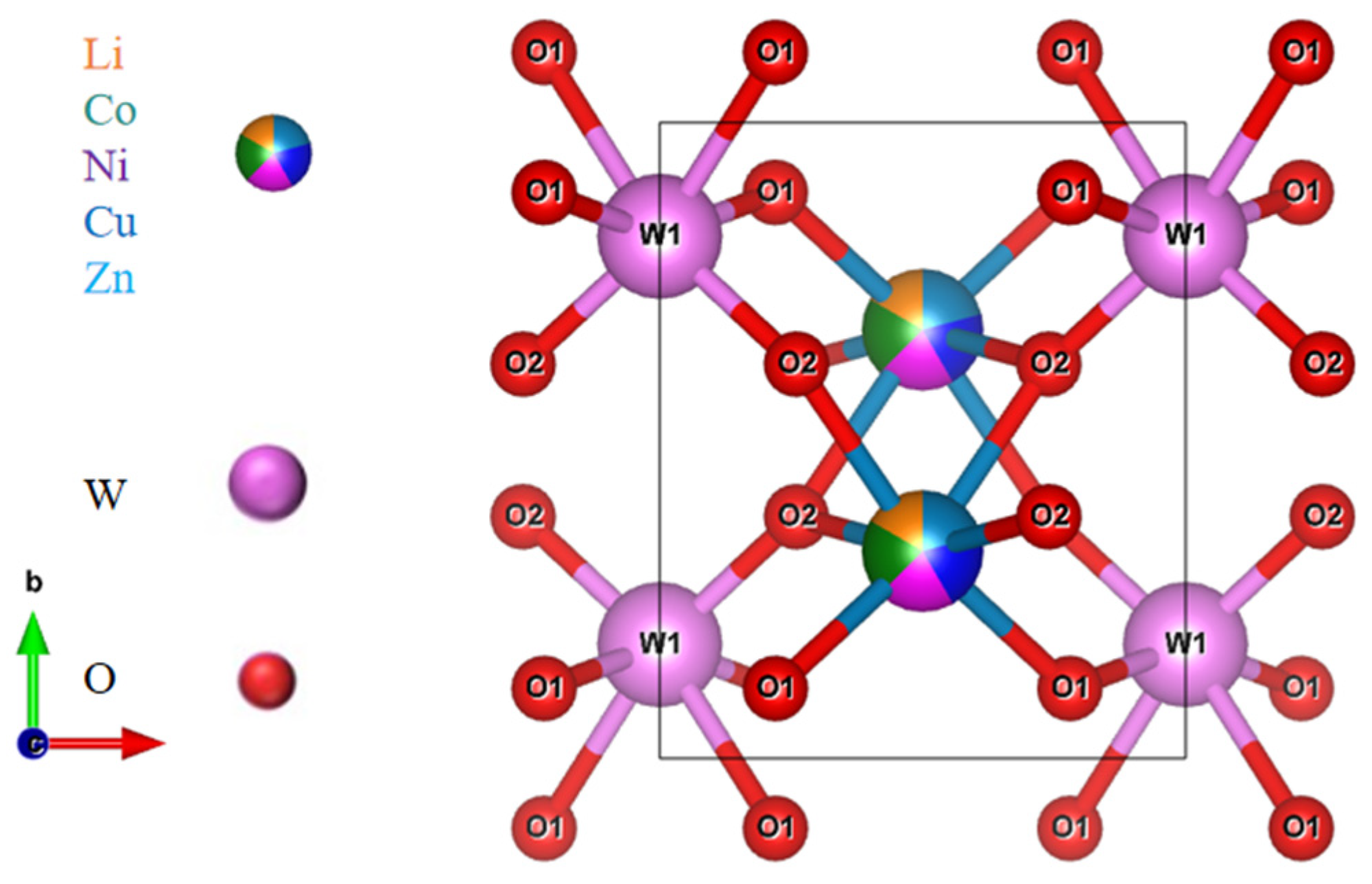



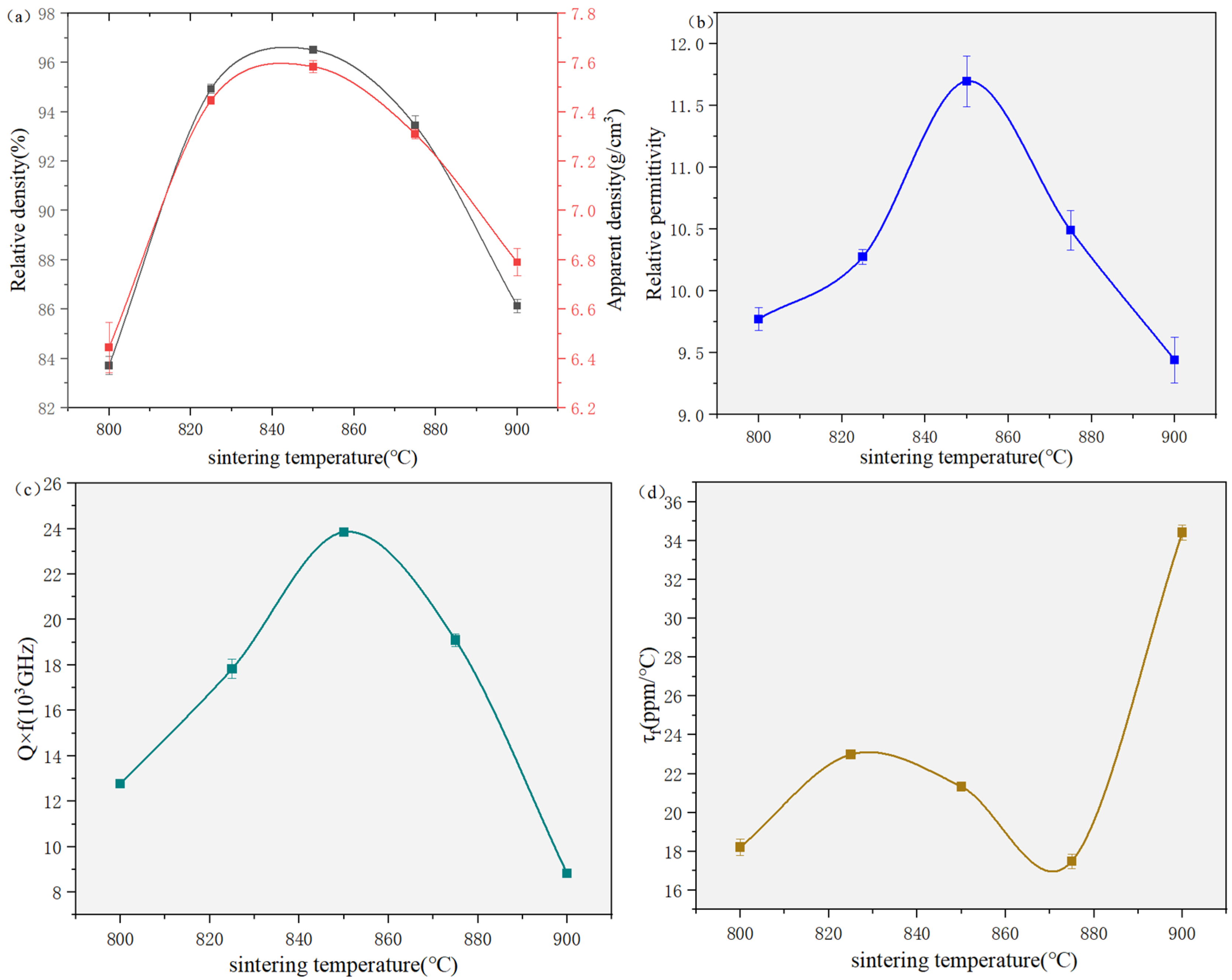
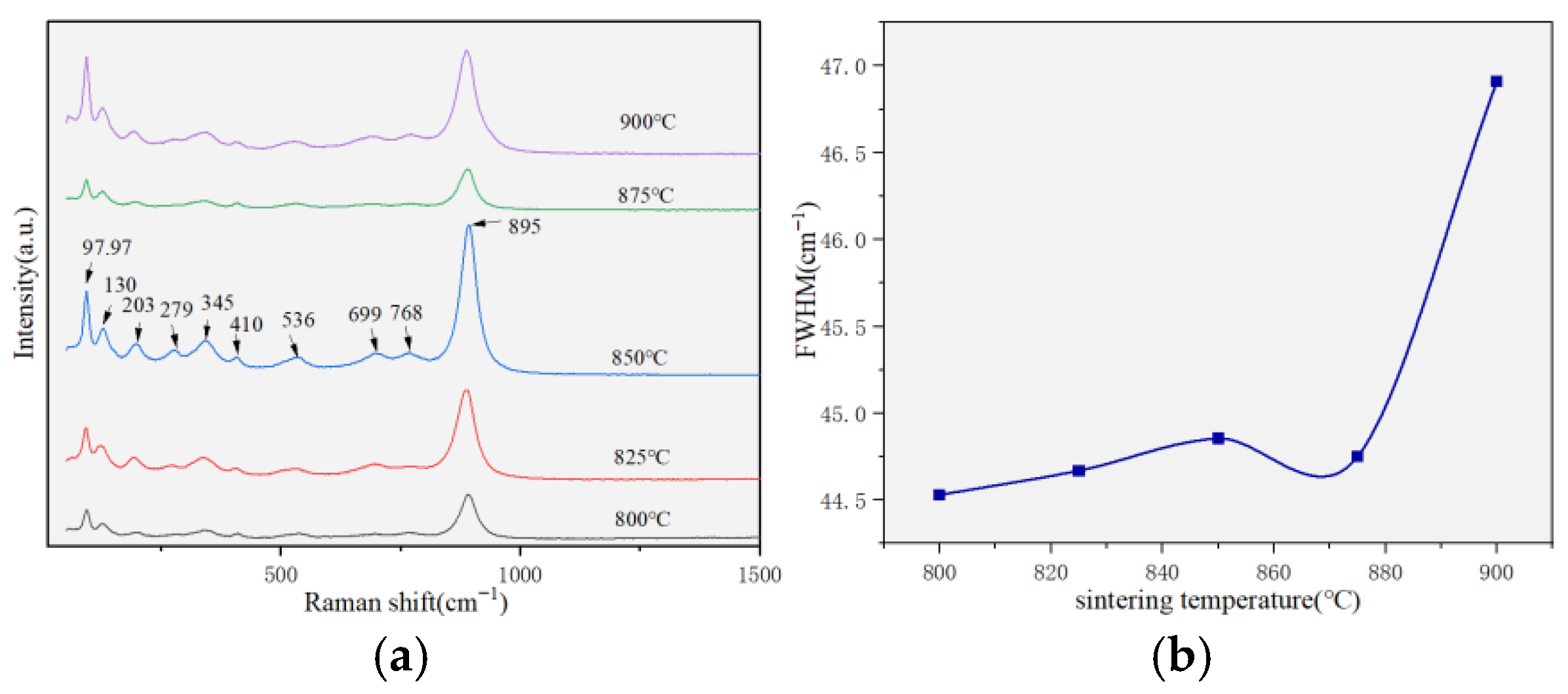
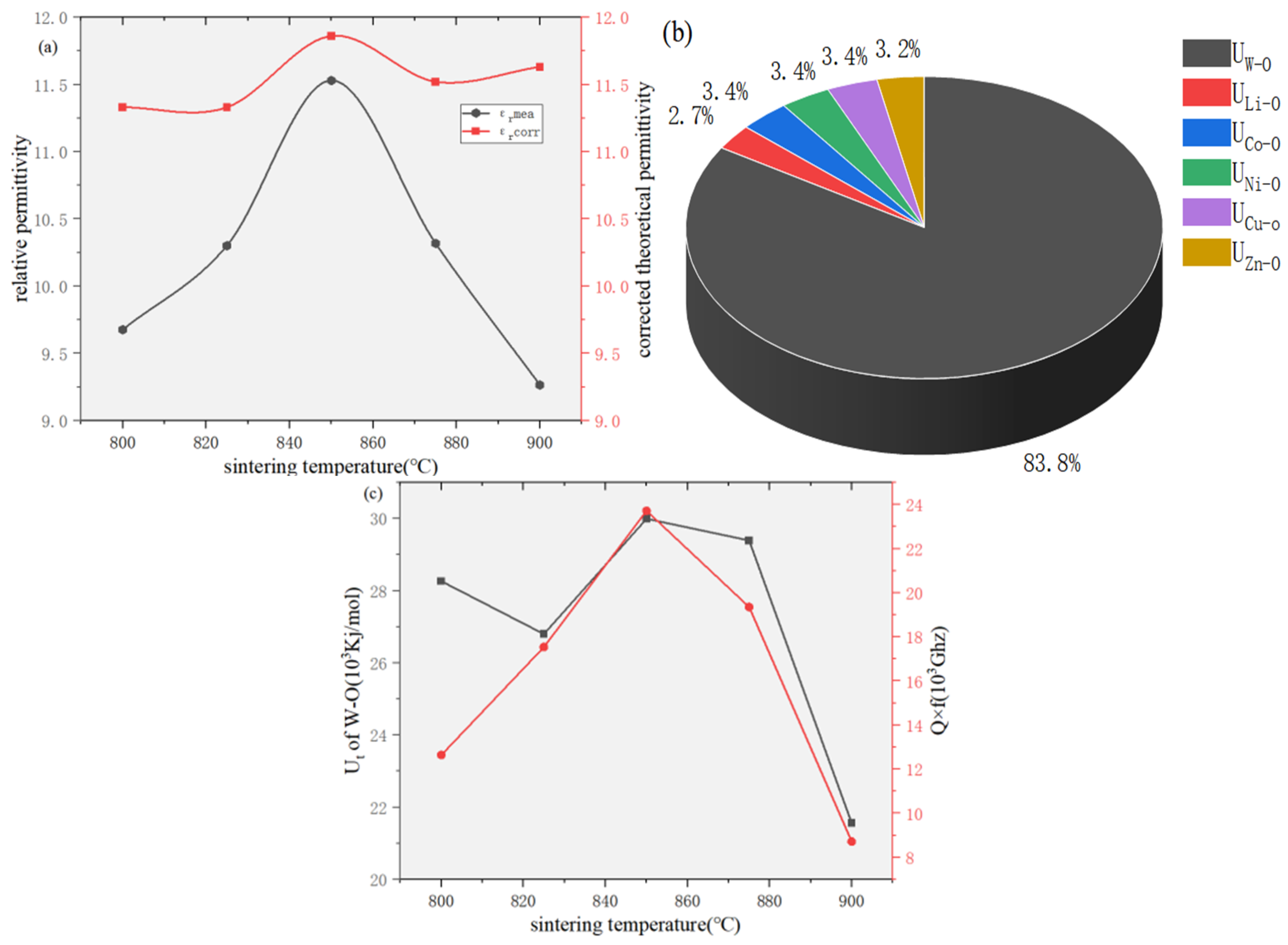
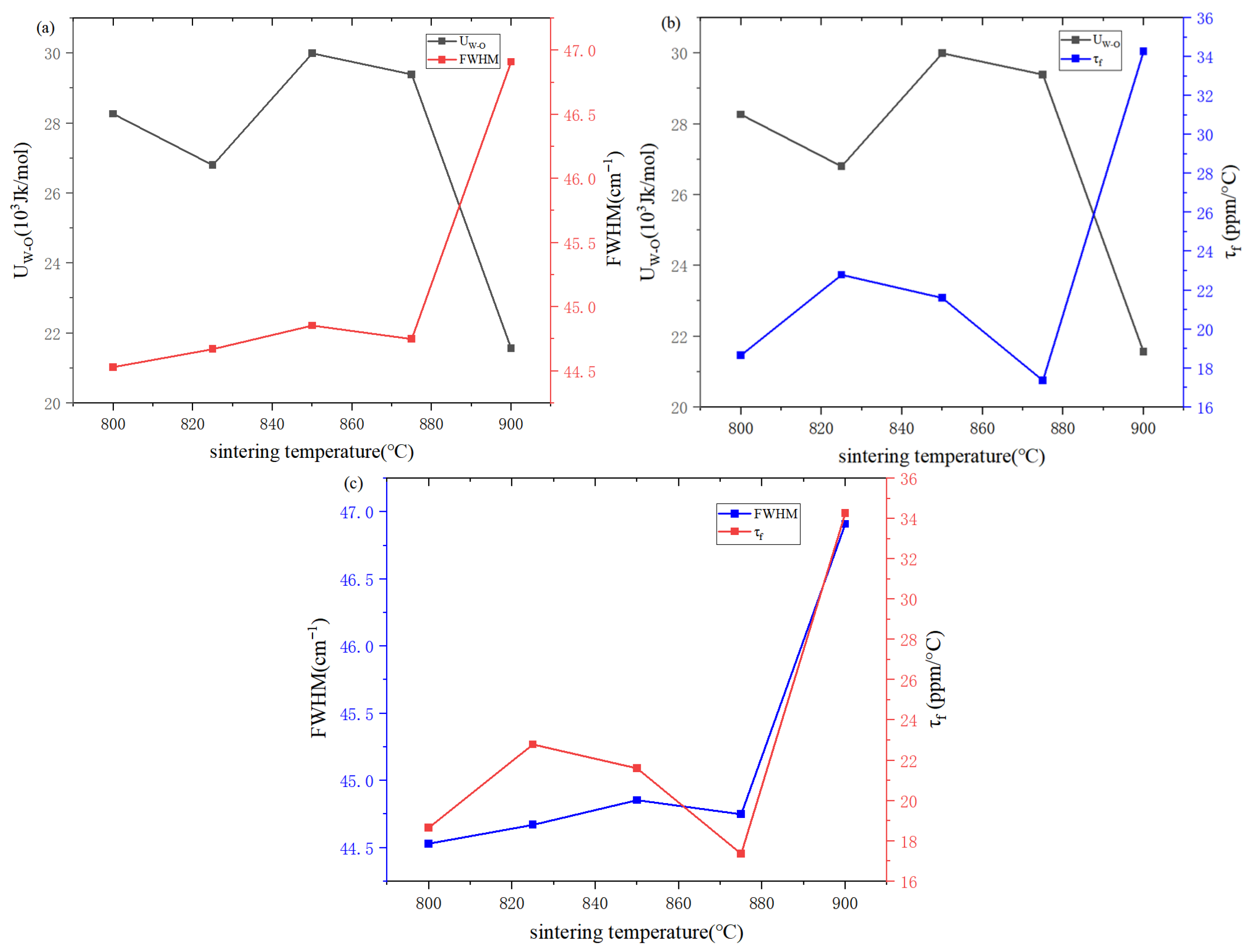
| 800 (a) | 825 (b) | 850 (c) | 875 (d) | 900 (e) | |
|---|---|---|---|---|---|
| Rwp (%) | 7.856 | 9.145 | 9.145 | 8.731 | 7.856 |
| a (Å) | 4.647 | 4.653 | 4.649 | 4.653 | 4.652 |
| b (Å) | 5.698 | 5.707 | 5.699 | 5.707 | 5.702 |
| c (Å) | 4.916 | 4.921 | 4.918 | 4.922 | 4.924 |
| GOF | 2.27 | 2.58 | 2.27 | 2.38 | 2.02 |
| Vm (Å3) | 130.193 | 130.721 | 130.339 | 130.710 | 130.656 |
| Chemical Bond | U (kJ/mol) | ||||||
|---|---|---|---|---|---|---|---|
| 800 °C | 825 °C | 850 °C | 875 °C | 900 °C | |||
| Li1-O(1) | 0.6020 | 0.3979 | 731.39 | 517.56 | 489.36 | 428.04 | 489.24 |
| Co1-O(1) | 0.3782 | 0.6217 | 774.36 | 642.11 | 583.30 | 531.05 | 606.77 |
| Ni1-O(1) | 0.3691 | 0.6308 | 575.52 | 647.23 | 587.16 | 535.26 | 611.60 |
| Cu1-O(1) | 0.3722 | 0.6277 | 773.60 | 645.54 | 585.90 | 543.43 | 610.01 |
| Zn1-O(1) | 0.4447 | 0.5552 | 760.40 | 605.39 | 555.55 | 500.62 | 572.03 |
| Li2-O(2) | 0.6020 | 0.3979 | 467.39 | 489.20 | 510.35 | 530.81 | 474.42 |
| Co2-O(2) | 0.3782 | 0.6217 | 576.63 | 606.70 | 633.09 | 658.75 | 588.36 |
| Ni2-O(2) | 0.3691 | 0.6308 | 584.24 | 611.53 | 638.14 | 672.00 | 593.03 |
| Cu2-O(2) | 0.3722 | 0.6277 | 1582.72 | 609.94 | 636.48 | 662.27 | 591.49 |
| Zn2-O(2) | 0.4447 | 0.5552 | 546.66 | 571.95 | 596.79 | 620.90 | 554.65 |
| W1-O(1) | 0.4374 | 0.5626 | 28,263.13 | 26,799.85 | 29,990.83 | 29,391.03 | 21,588.13 |
| Material | Sintering Temp. (°C) | εr | Q × f (GHz) | τf (ppm/°C) | Reference |
|---|---|---|---|---|---|
| ZnWO4 | >1100 | ~15 | ~60,000 | ~−60 | [14] |
| (Li0.4Co0.2Ni0.2Cu0.2Zn0.2)WO4 | 850 | 11.529 | 23,714 | 21.6 | This work |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Sun, Y.; Zhou, X.; Feng, G.; Li, B.; Yang, D.; Zhou, D.; Han, J.; Wang, Q.; Yang, Y. (Li0.4Co0.2Ni0.2Cu0.2Zn0.2)WO4: A Novel High-Entropy Wolframite Ceramic with Tailored Microwave Dielectric Properties. Materials 2026, 19, 1421. https://doi.org/10.3390/ma19071421
Sun Y, Zhou X, Feng G, Li B, Yang D, Zhou D, Han J, Wang Q, Yang Y. (Li0.4Co0.2Ni0.2Cu0.2Zn0.2)WO4: A Novel High-Entropy Wolframite Ceramic with Tailored Microwave Dielectric Properties. Materials. 2026; 19(7):1421. https://doi.org/10.3390/ma19071421
Chicago/Turabian StyleSun, Yutao, Xiong Zhou, Guangshu Feng, Bingli Li, Daode Yang, Dacheng Zhou, Jin Han, Qi Wang, and Yong Yang. 2026. "(Li0.4Co0.2Ni0.2Cu0.2Zn0.2)WO4: A Novel High-Entropy Wolframite Ceramic with Tailored Microwave Dielectric Properties" Materials 19, no. 7: 1421. https://doi.org/10.3390/ma19071421
APA StyleSun, Y., Zhou, X., Feng, G., Li, B., Yang, D., Zhou, D., Han, J., Wang, Q., & Yang, Y. (2026). (Li0.4Co0.2Ni0.2Cu0.2Zn0.2)WO4: A Novel High-Entropy Wolframite Ceramic with Tailored Microwave Dielectric Properties. Materials, 19(7), 1421. https://doi.org/10.3390/ma19071421





