Effect of Waste Glass Incorporation Methods on the Physical, Mechanical and Microstructural Properties of Cementitious Binders
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Test Methods and Preparation of Samples
3. Results and Discussion
3.1. Influence of Different Mixtures Preparation Methods and Waste Glass Types on Binder Paste Workability
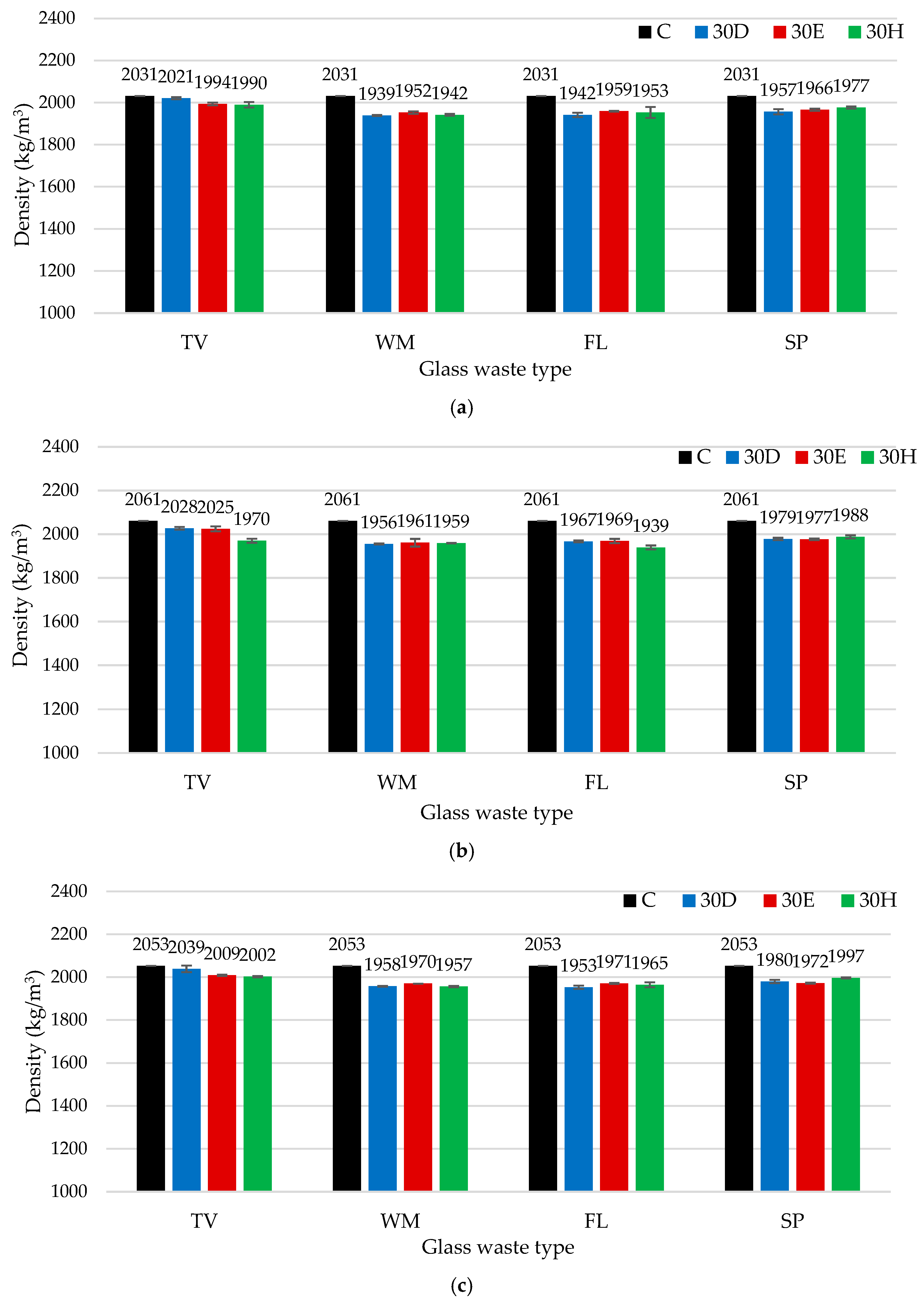
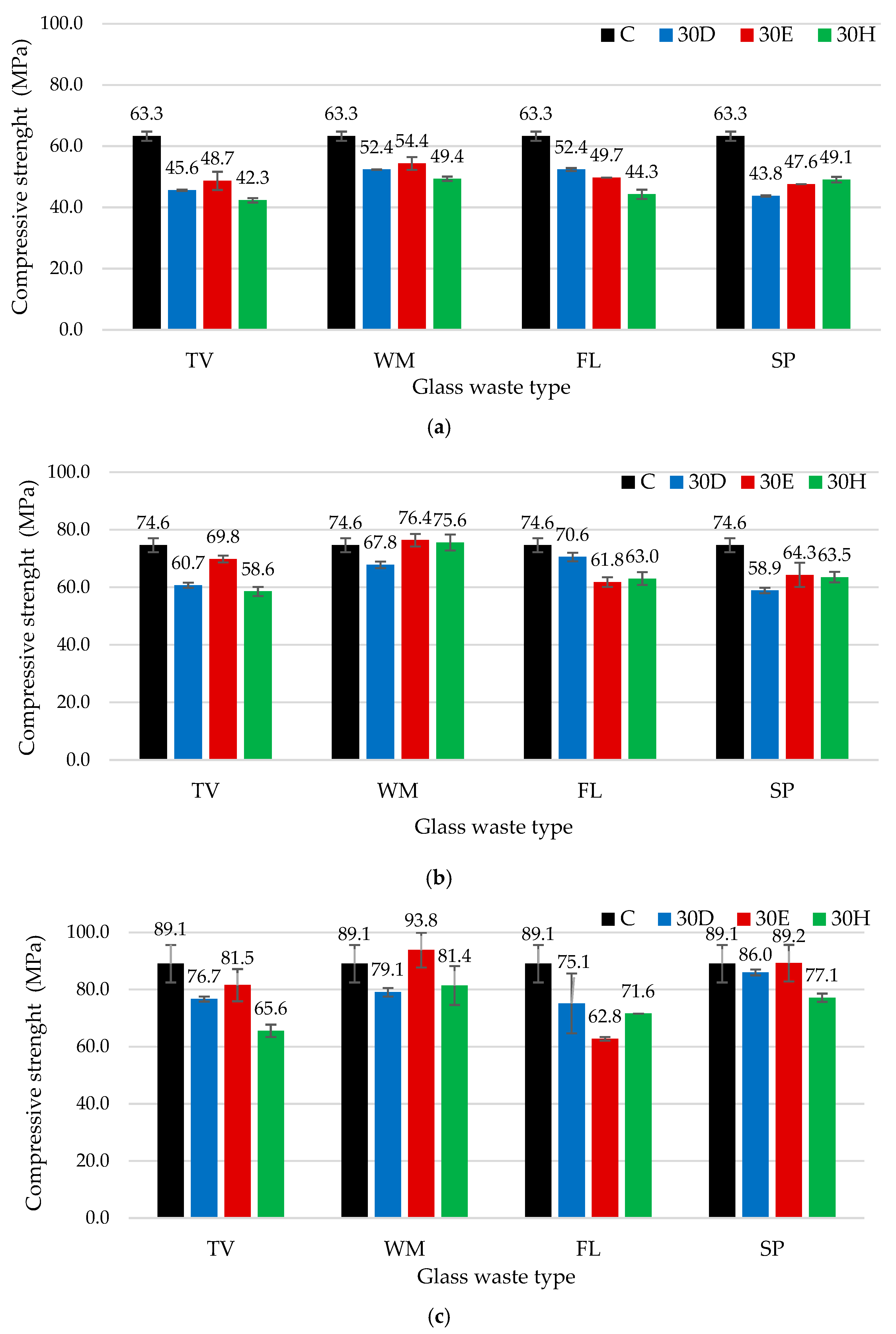
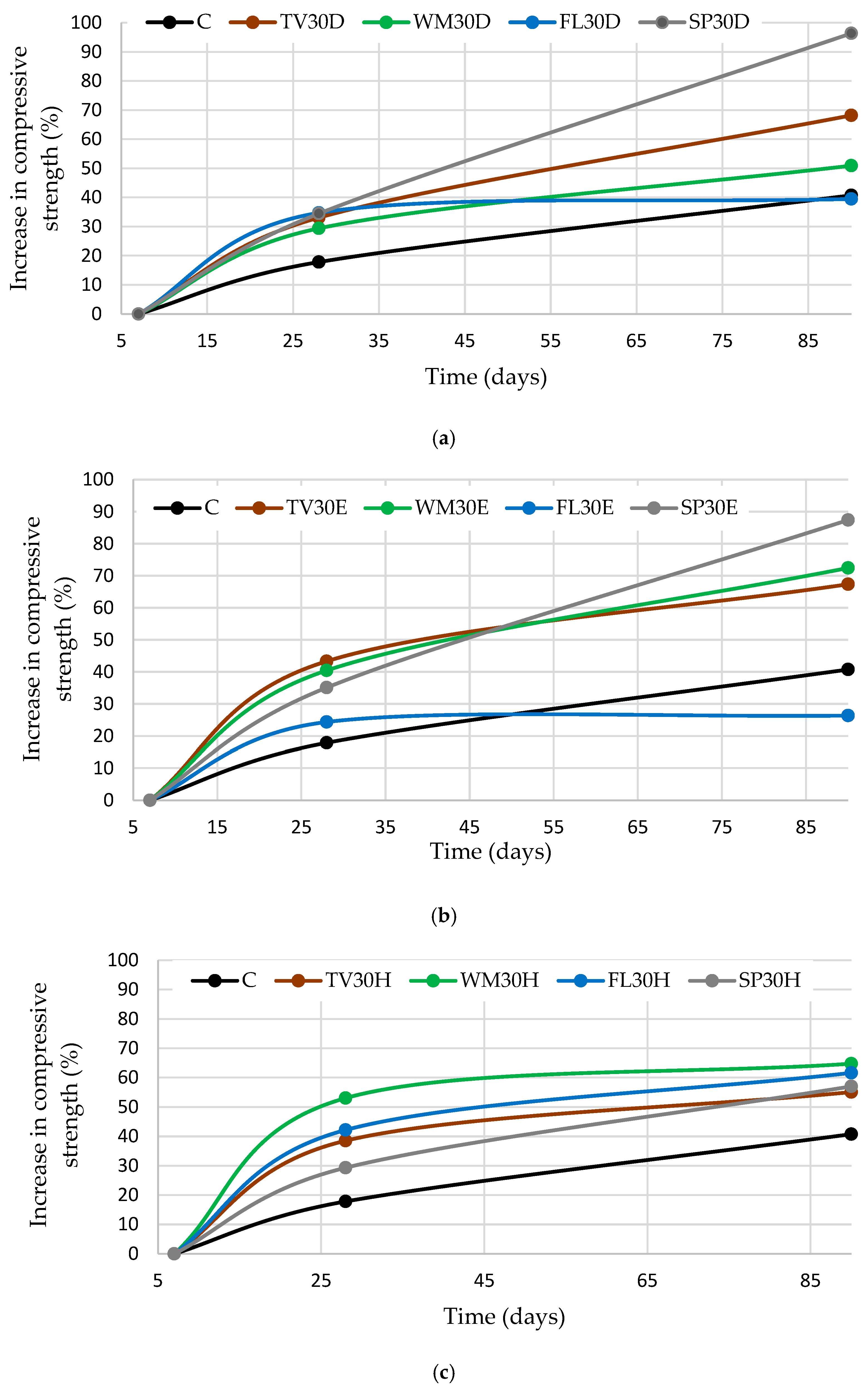
3.2. Influence of Different Mixtures Preparation Methods and Waste Glass Types on the Binder Density and Compressive Strength
3.3. Influence of Mixtures Preparation Methods and Waste Glass Types on Binder Mineral Composition and Microstructure
4. Conclusions
- The method of mixing raw materials was determined to have a significant effect on the rheological properties of cement-based binders with ground waste glass, depending on the type of glass. The ultrasonic dispersion technique was found to be most effective for low-soluble TV glass, yielding spread values that were comparable to those of the reference mixture due to enhanced particle dispersion. The findings of the study suggest that while WM and FL glass powders exhibit comparable particle sizes to those of TV, their responses to dispersion methods differ. The application of intensive mixing did not result in an enhancement of paste spread; indeed, it led to a reduction in spread, particularly in the case of WM. Conversely, ultrasonic dispersion yielded only a marginal increase in spread. This behaviour is attributed to the higher solubility of these glasses. It has been established that dissolution processes at the glass–cement interface are conducive to the formation of a hydrated amorphous gel layer. Consequently, glass solubility plays a pivotal role in the control of dispersion efficiency and paste rheology.
- The findings indicate that the density and compressive strength of cement-based binders are contingent on both the type of glass and the method of mixing employed. The highest density for TV glass was achieved by dispersion in water, while intensive mixing was most effective for WM and FL glass, likely due to enhanced hydration and microstructural densification. The effect was negligible for SP glass. Intensive mixing was found to be optimal for TV and WM glass, while ultrasonic dispersion yielded the best results for FL glass due to the highest solubility, highlighting the need to tailor mixing methods to glass properties. The results of XRD and SEM analyses demonstrated that the mixing methods employed had a significant impact on both hydration and microstructure. Intensive mixing generally produced a denser matrix, whereas ultrasonic dispersion resulted in a more porous structure, with the exception of FL glass, which exhibited the highest solubility.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| GW | Glass waste |
| TV | Discarded television screens |
| WM | Washing machines |
| FL | Fluorescent lamps |
| SP | Photovoltaic solar panels |
| C-S-H | Calcium silicate hydrates |
References
- Macaskie, L.E.; Sapsford, D.J.; Mayes, W.M. (Eds.) Resource Recovery from Wastes: Towards a Circular Economy (ISSN), 1st ed.; Royal Society of Chemistry: London, UK, 2019; ISBN 978-1788013819. [Google Scholar]
- Wang, B.; Ren, C.; Dong, X.; Zhang, B.; Wang, Z. Determinants Shaping Willingness towards On-Line Recycling Behaviour: An Empirical Study of Household e-Waste Recycling in China. Resour. Conserv. Recycl. 2019, 143, 218–225. [Google Scholar] [CrossRef]
- Zhang, Z.; Malik, M.Z.; Khan, A.; Ali, N.; Malik, S.; Bilal, M. Environmental Impacts of Hazardous Waste, and Management Strategies to Reconcile Circular Economy and Eco-Sustainability. Sci. Total Environ. 2022, 807, 150856. [Google Scholar] [CrossRef]
- Premathilaka, K.K.W.; Liyanapathirana, D.S.; Leo, C.J.; Hu, P. Application of Recycled Waste Glass to Replace Traditional Quarried Aggregates: A Comprehensive Review. J. Build. Eng. 2024, 86, 108846. [Google Scholar] [CrossRef]
- Mohajerani, A.; Vajna, J.; Cheung, T.H.H.; Kurmus, H.; Arulrajah, A.; Horpibulsuk, S. Practical Recycling Applications of Crushed Waste Glass in Construction Materials: A Review. Constr. Build. Mater. 2017, 156, 443–467. [Google Scholar] [CrossRef]
- Sobolev, K.; Türker, P.; Soboleva, S.; Iscioglu, G. Utilization of Waste Glass in ECO-Cement: Strength Properties and Mi-crostructural Observations. Waste Manag. 2007, 27, 971–976. [Google Scholar] [CrossRef]
- Hamada, H.; Alattar, A.; Tayeh, B.; Yahaya, F.; Thomas, B. Effect of Recycled Waste Glass on the Properties of High-Performance Concrete: A Critical Review. Case Stud. Constr. Mater. 2022, 17, e01149. [Google Scholar] [CrossRef]
- Dong, W.; Li, W.; Tao, Z. A Comprehensive Review on Performance of Cementitious and Geopolymeric Concretes with Recycled Waste Glass as Powder, Sand or Cullet. Resour. Conserv. Recycl. 2021, 172, 105664. [Google Scholar] [CrossRef]
- Esmaeili, J.; AL-Mwanes, A.O. Performance Evaluation of Eco-Friendly Ultra-High-Performance Concrete Incorporated with Waste Glass-A Review. IOP Conf. Ser. Mater. Sci. Eng. 2021, 1094, 012030. [Google Scholar] [CrossRef]
- Stefanidou, M.; Kesikidou, F.; Konopisi, S.; Vasiadis, T. Investigating the Suitability of Waste Glass as a Supplementary Binder and Aggregate for Cement and Concrete. Sustainability 2023, 15, 3796. [Google Scholar] [CrossRef]
- Surendran, H.; Akhas, P.K. Properties of High-Performance Concrete Incorporating Toughened Glass Waste Coarse Ag-gregate: An Experimental Study. Structures 2024, 60, 105897. [Google Scholar] [CrossRef]
- Ferdous, W.; Manalo, A.; Siddique, R.; Mendis, P.; Zhuge, Y.; Wong, H.S.; Lokuge, W.; Aravinthan, T.; Schubel, P. Recycling of Landfill Wastes (Tyres, Plastics and Glass) in Construction—A Review on Global Waste Generation, Performance, Ap-plication and Future Opportunities. Resour. Conserv. Recycl. 2021, 173, 105745. [Google Scholar] [CrossRef]
- Rashid, K.; Hameed, R.; Ahmad, H.A.; Razzaq, A.; Ahmad, M.; Mahmood, A. Analytical Framework for Value Added Utilization of Glass Waste in Concrete: Mechanical and Environmental Performance. Waste Manag. 2018, 79, 312–323. [Google Scholar] [CrossRef]
- Shi, C.; Zheng, K. A Review on the Use of Waste Glasses in the Production of Cement and Concrete. Resour. Conserv. Recycl. 2007, 52, 234–247. [Google Scholar] [CrossRef]
- Máčalová, K.; Václavík, V.; Dvorský, T.; Figmig, R.; Charvát, J.; Lupták, M. The Use of Glass from Photovoltaic Panels at the End of Their Life Cycle in Cement Composites. Materials 2021, 14, 6655. [Google Scholar] [CrossRef] [PubMed]
- Mariaková, D.; Mocová, K.A.; Fořtová, K.; Pavlů, T.; Hájek, P. Waste Glass Powder Reusability in High-Performance Concrete: Leaching Behavior and Ecotoxicity. Materials 2021, 14, 4476. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, K.I.M. Recycled Waste Glass Powder as a Partial Replacement of Cement in Concrete Containing Silica Fume and Fly Ash. Case Stud. Constr. Mater. 2021, 15, e00630. [Google Scholar] [CrossRef]
- Kazmi, D.; Serati, M.; Williams, D.J.; Qasim, S.; Cheng, Y.P. The Potential Use of Crushed Waste Glass as a Sustainable Alternative to Natural and Manufactured Sand in Geotechnical Applications. J. Clean. Prod. 2021, 284, 124762. [Google Scholar] [CrossRef]
- De Castro, S.; De Brito, J. Evaluation of the Durability of Concrete Made with Crushed Glass Aggregates. J. Clean. Prod. 2013, 41, 7–14. [Google Scholar] [CrossRef]
- Muhedin, D.A.; Ibrahim, R.K. Effect of Waste Glass Powder as Partial Replacement of Cement & Sand in Concrete. Case Stud. Constr. Mater. 2023, 19, e02512. [Google Scholar] [CrossRef]
- Li, Q.; Qiao, H.; Li, A.; Li, G. Performance of Waste Glass Powder as a Pozzolanic Material in Blended Cement Mortar. Constr. Build. Mater. 2022, 324, 126531. [Google Scholar] [CrossRef]
- Chen, W.; Liu, D.; Liang, Y. Influence of Ultra Fine Glass Powder on the Properties and Microstructure of Mortars. Fluid Dyn. Mater. Process. 2024, 20, 915–938. [Google Scholar] [CrossRef]
- Guo, P.; Meng, W.; Nassif, H.; Gou, H.; Bao, Y. New Perspectives on Recycling Waste Glass in Manufacturing Concrete for Sustainable Civil Infrastructure. Constr. Build. Mater. 2020, 257, 119579. [Google Scholar] [CrossRef]
- Dhir, R.K.; Dyer, T.D.; Tang, M.C. Alkali-Silica Reaction in Concrete Containing Glass. Mater. Struct./Mater. Constr. 2009, 42, 1451–1462. [Google Scholar] [CrossRef]
- Samarakoon, M.H.; Ranjith, P.G.; De Silva, V.R.S. Effect of Soda-Lime Glass Powder on Alkali-Activated Binders: Rheology, Strength and Microstructure Characterization. Constr. Build. Mater. 2020, 241, 118013. [Google Scholar] [CrossRef]
- Utton, C.A.; Hand, R.J.; Bingham, P.A.; Hyatt, N.C.; Swanton, S.W.; Williams, S.J. Dissolution of Vitrified Wastes in a High-PH Calcium-Rich Solution. J. Nucl. Mater. 2013, 435, 112–122. [Google Scholar] [CrossRef]
- Dyer, T.D.; Dhir, R.K. Chemical Reactions of Glass Cullet Used as Cement Component. J. Mater. Civ. Eng. 2001, 13, 412–417. [Google Scholar] [CrossRef]
- Ramadan, M.; El-Gamal, S.M.A.; Selim, F.A. Mechanical Properties, Radiation Mitigation and Fire Resistance of OPC-Recycled Glass Powder Composites Containing Nanoparticles. Constr. Build. Mater. 2020, 251, 118703. [Google Scholar] [CrossRef]
- Jang, H.; Jeon, S.; So, H.; So, S. Properties of Different Particle Size of Recycled TFT-LCD Waste Glass Powder as a Cement Concrete Binder. Int. J. Precis. Eng. Manuf. 2015, 16, 2591–2597. [Google Scholar] [CrossRef]
- Guler, S.; Akbulut, Z.F. Effect of High-Temperature on the Behavior of Single and Hybrid Glass and Basalt Fiber Added Geopolymer Cement Mortars. J. Build. Eng. 2022, 57, 104809. [Google Scholar] [CrossRef]
- Li, X.; Liu, Y.M.; Li, W.G.; Li, C.Y.; Sanjayan, J.G.; Duan, W.H.; Li, Z. Effects of Graphene Oxide Agglomerates on Workability, Hydration, Microstructure and Compressive Strength of Cement Paste. Constr. Build. Mater. 2017, 145, 402–410. [Google Scholar] [CrossRef]
- Guo, S.; Yu, T.; Mao, K.; Cao, P.; Lu, J.; Zhong, H. Study on Fracture Properties and Damage of Glass Fiber Reinforced Concrete under Corrosive Environment. Constr. Build. Mater. 2025, 501, 144238. [Google Scholar] [CrossRef]
- Afshinnia, K.; Rangaraju, P.R. Impact of Combined Use of Ground Glass Powder and Crushed Glass Aggregate on Selected Properties of Portland Cement Concrete. Constr. Build. Mater. 2016, 117, 263–272. [Google Scholar] [CrossRef]
- Elaqra, H.A.; Haloub, M.A.A.; Rustom, R.N. Effect of New Mixing Method of Glass Powder as Cement Replacement on Mechanical Behavior of Concrete. Constr. Build. Mater. 2019, 203, 75–82. [Google Scholar] [CrossRef]
- Poudel, S.; Bhetuwal, U.; Kharel, P.; Khatiwada, S.; KC, D.; Dhital, S.; Lamichhane, B.; Yadav, S.K.; Suman, S. Waste Glass as Partial Cement Replacement in Sustainable Concrete: Mechanical and Fresh Properties Review. Buildings 2025, 15, 857. [Google Scholar] [CrossRef]
- Patriota, A.L.S.; Barros, F.B.M.; Carneiro, A.M.P.; Guzzo, P.L. An Investigation on the Physical Properties of Cementitious Pastes Modified with Low Dosage of Waste Glass Powder and Silica Fume. An. Acad. Bras. Cienc. 2024, 96, e20231153. [Google Scholar] [CrossRef]
- Bekerė, K.; Malaiškienė, J.; Škamat, J. An Analysis of a Cement Hydration Process Using Glass Waste from Household Appliances as a Supplementary Material. Processes 2025, 13, 840. [Google Scholar] [CrossRef]
- Serelis, E.; Vaitkevicius, V.; Hilbig, H.; Irbe, L.; Rudzionis, Z. Effect of Ultra-Sonic Dispersion Time on Hydration Process and Microstructure Development of Ultra-High Performance Glass Powder Concrete. Constr. Build. Mater. 2021, 298, 123856. [Google Scholar] [CrossRef]
- Serelis, E.; Vaitkevicius, V.; Salehi, S.; Sinka, M.; Sapata, A. Synergistic Effects of Waste Glass Powder, High-Frequency Ultrasonic Dispersion, and Liquid Glass Treatment on the Properties of Aluminum-Based Ultra-Lightweight Concrete. Materials 2024, 17, 5430. [Google Scholar] [CrossRef] [PubMed]
- Aziz, T.; Tao, Z.; Rahmani, A.; Katwal, U.; Fanna, D.J.; Wuhrer, R. Optimisation of Hybrid Alkaline Binder with a High Glass Powder Content. J. Build. Eng. 2024, 98, 111007. [Google Scholar] [CrossRef]
- Vaitkevičius, V.; Šerelis, E.; Hilbig, H. The Effect of Glass Powder on the Microstructure of Ultra High Performance Concrete. Constr. Build. Mater. 2014, 68, 102–109. [Google Scholar] [CrossRef]
- Šerelis, E.; Vaitkevičius, V.; Kerševičius, V. Mechanical Properties and Microstructural Investigation of Ultra-High Per-formance Glass Powder Concrete. J. Sustain. Archit. Civ. Eng. 2016, 14, 14478. [Google Scholar] [CrossRef]
- Vaitkevičius, V.; Šerelis, E.; Vaičiukynienė, D.; Raudonis, V.; Rudžionis, Ž. Advanced Mechanical Properties and Frost Damage Resistance of Ultra-High Performance Fibre Reinforced Concrete. Constr. Build. Mater. 2016, 126, 26–31. [Google Scholar] [CrossRef]
- Kim, H.; Hadl, P.; Nguyen, V.T. A New Mix Design Method for UHPC Based on Stepwise Optimization of Particle Packing Density. In International Interactive Symposium on Ultra-High Performance Concrete Papers; Iowa State University Digital Press: Ames, IA, USA, 2016. [Google Scholar] [CrossRef]
- Hemalatha, T.; Ram Sundar, K.R.; Murthy, A.R.; Iyer, N.R. Influence of Mixing Protocol on Fresh and Hardened Properties of Self-Compacting Concrete. Constr. Build. Mater. 2015, 98, 119–127. [Google Scholar] [CrossRef]
- Rajadurai, R.S.; Kang, S.T. Influence of Inter-Particle Distances on the Rheological Properties of Cementitious Suspensions. Materials 2021, 14, 7869. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Fan, Y.; Shah, S.P. Rheological Properties and Structural Build-Up of Cement Based Materials with Addition of Nanoparticles: A Review. Buildings 2022, 12, 2219. [Google Scholar] [CrossRef]
- Cailleteau, C.; Angeli, F.; Devreux, F.; Gin, S.; Jestin, J.; Jollivet, P.; Spalla, O. Insight into silicate-glass corrosion mechanisms. Nat. Mater. 2008, 7, 978–983. [Google Scholar] [CrossRef]
- Mehdipour, I.; Khayat, K.H. Elucidating How Particle Packing Controls Rheology and Strength Development of Dense Cementitious Suspensions. Cem. Concr. Compos. 2019, 104, 103413. [Google Scholar] [CrossRef]
- Shayan, A.; Xu, A. Performance of Glass Powder as a Pozzolanic Material in Concrete: A Field Trial on Concrete Slabs. Cem. Concr. Res. 2006, 36, 457–468. [Google Scholar] [CrossRef]
- Balasubramanian, B.; Gopala Krishna, G.V.T.; Saraswathy, V.; Srinivasan, K. Experimental Investigation on Concrete Partially Replaced with Waste Glass Powder and Waste E-Plastic. Constr. Build. Mater. 2021, 278, 122400. [Google Scholar] [CrossRef]
- Taha, B.; Nounu, G. Properties of Concrete Contains Mixed Colour Waste Recycled Glass as Sand and Cement Replacement. Constr. Build. Mater. 2008, 22, 713–720. [Google Scholar] [CrossRef]
- Zheng, K. Pozzolanic reaction of glass powder and its role in controlling alkali–silica reaction. Cem. Concr. Compos. 2016, 67, 30–38. [Google Scholar] [CrossRef]
- Kriptavičius, D.; Girskas, G.; Skripkiūnas, G. Use of Natural Zeolite and Glass Powder Mixture as Partial Replacement of Portland Cement: The Effect on Hydration, Properties and Porosity. Materials 2022, 15, 4219. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, J.; Martinez-Garcia, R.; Algarni, S.; de Prado-Gil, J.; Alqahtani, T.; Irshad, K. Characteristics of sustainable concrete with partial substitutions of glass waste as a binder material. Int. J. Concr. Struct. Mater. 2022, 16, 21. [Google Scholar] [CrossRef]
- Tamanna, N.; Tuladhar, R. Sustainable use of recycled glass powder as cement replacement in concrete. Open Waste Manag. J. 2020, 13, 1–13. [Google Scholar] [CrossRef]
- Maraghechi, H.; Maraghechi, M.; Rajabipour, F.; Pantano, C.G. Pozzolanic Reactivity of Recycled Glass Powder at Elevated Temperatures: Reaction Stoichiometry, Reaction Products and Effect of Alkali Activation. Cem. Concr. Compos. 2014, 53, 105–114. [Google Scholar] [CrossRef]
- Scrivener, K.L.; Juilland, P.; Monteiro, P.J.M. Advances in Understanding Hydration of Portland Cement. Cem. Concr. Res. 2015, 78, 38–56. [Google Scholar] [CrossRef]






| Chemical Component (%) | Cement | TV | WM | FL | SP |
|---|---|---|---|---|---|
| SiO2 | 20.0 | 53.1 | 81.4 | 67.9 | 69.4 |
| Al2O3 | 4.57 | 2.30 | 2.91 | 2.11 | 1.12 |
| Na2O | 0.14 | 6.37 | 6.51 | 14.2 | 12.2 |
| K2O | 1.30 | 7.85 | 1.26 | 1.75 | 0.04 |
| MgO | 3.71 | 0.31 | 0.03 | 2.54 | 3.24 |
| CaO | 63.4 | 0.93 | 3.74 | 5.45 | 9.96 |
| BaO | 0.06 | 8.62 | 0.03 | 1.38 | 0.04 |
| PbO | - | 3.98 | - | 0.10 | 0.01 |
| SrO | - | 7.70 | 0.01 | 0.06 | 0.01 |
| ZnO | 0.01 | 0.36 | - | - | 0.002 |
| Other | 6.81 | 8.48 | 4.11 | 4.51 | 3.98 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Malaiškienė, J.; Bekerė, K.; Škamat, J. Effect of Waste Glass Incorporation Methods on the Physical, Mechanical and Microstructural Properties of Cementitious Binders. Materials 2026, 19, 1346. https://doi.org/10.3390/ma19071346
Malaiškienė J, Bekerė K, Škamat J. Effect of Waste Glass Incorporation Methods on the Physical, Mechanical and Microstructural Properties of Cementitious Binders. Materials. 2026; 19(7):1346. https://doi.org/10.3390/ma19071346
Chicago/Turabian StyleMalaiškienė, Jurgita, Karolina Bekerė, and Jelena Škamat. 2026. "Effect of Waste Glass Incorporation Methods on the Physical, Mechanical and Microstructural Properties of Cementitious Binders" Materials 19, no. 7: 1346. https://doi.org/10.3390/ma19071346
APA StyleMalaiškienė, J., Bekerė, K., & Škamat, J. (2026). Effect of Waste Glass Incorporation Methods on the Physical, Mechanical and Microstructural Properties of Cementitious Binders. Materials, 19(7), 1346. https://doi.org/10.3390/ma19071346







