Effect of Mouthwashes on the Mechanical Properties and Color Stability of Composite Material
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Sample Preparation
2.3. Exposure to Liquids
2.4. Test Methods
2.4.1. Diametral Tensile Strength Test
2.4.2. Compressive Strength Test
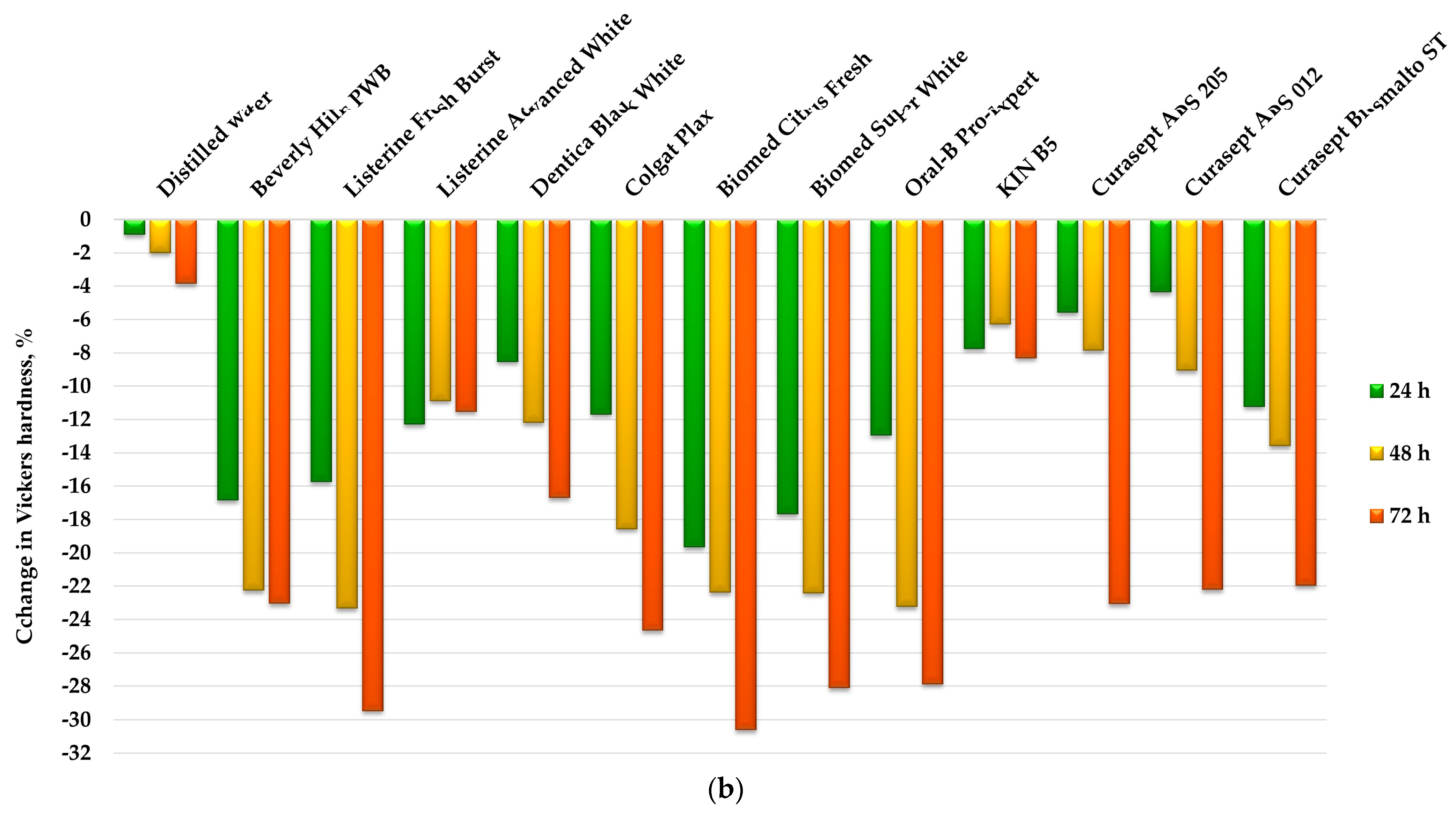
2.4.3. Vickers Hardness Test
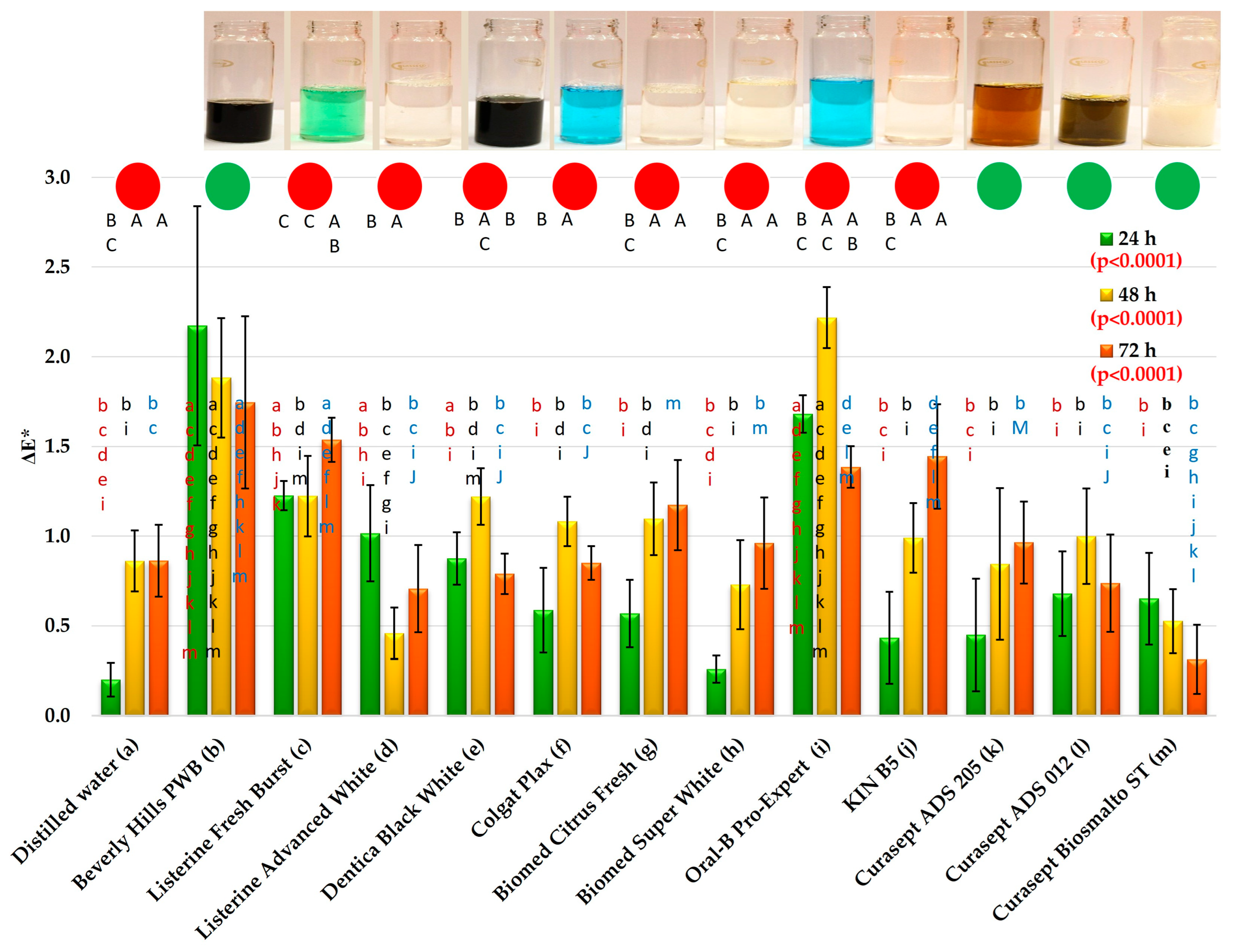
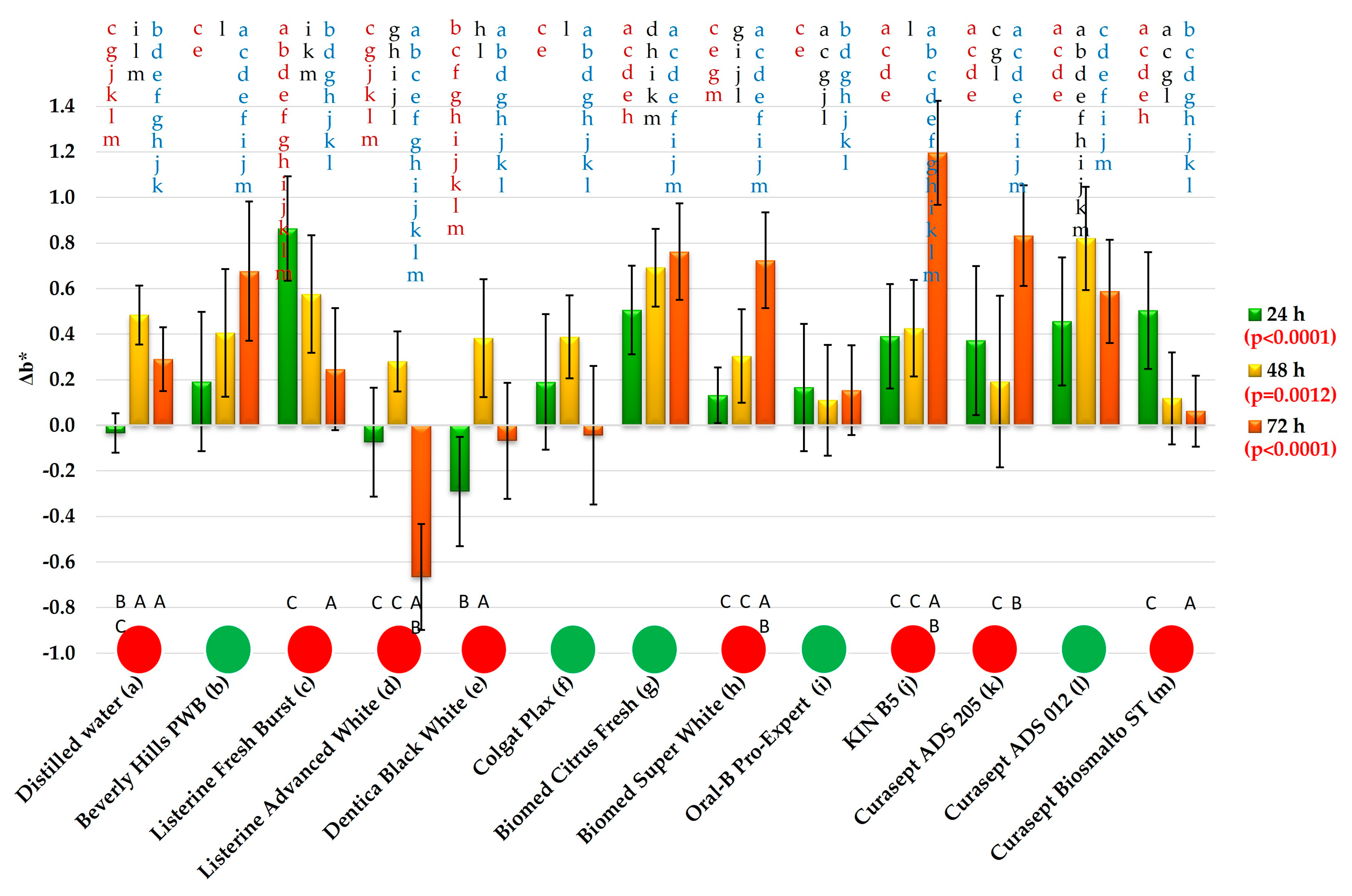
2.4.4. Color Stability
2.4.5. Sample Size Calculation and Statistical Analysis
3. Results
3.1. Mechanical Properties
3.2. Color Changes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CS | Compressive Strength |
| DTS | Diametral Tensile Strength |
| HV | Vickers Hardness |
| ADS | Anti-Discoloration System |
| ΔE* | Color difference |
| L* | Lightness coordinate |
| a* | coordinate red–green |
| b* | coordinate yellow–blue |
| AT | acceptability threshold |
| PT | perceptibility threshold |
| ANOVA | one-way analysis of variance |
References
- Towle, I.; Irish, J.D.; Groote, I.D. Dental Pathology, Wear and Developmental Defects in South African Hominins. In Proceedings of the 86th Annual Meeting of the American Association of Physical Anthropologists, New Orleans, LA, USA, 19–22 April 2017. [Google Scholar]
- Forshaw, R. Dentistry and Dental Care in Antiquity: Part 1—Prehistory, Mesopotamia, Israel, Etruria and the Far East. Br. Dent. J. 2025, 239, 851–856. [Google Scholar] [CrossRef] [PubMed]
- Coppa, A.; Bondioli, L.; Cucina, A.; Frayer, D.W.; Jarrige, C.; Jarrige, J.-F.; Quivron, G.; Rossi, M.; Vidale, M.; Macchiarelli, R. Palaeontology: Early Neolithic Tradition of Dentistry. Nature 2006, 440, 755–756. [Google Scholar] [CrossRef]
- Akhter, H.; Kha, F.A. History of Dentistry. Arch. Med. Health Sci. 2014, 1, 106–110. [Google Scholar] [CrossRef]
- Grimoud, A.-M.; Lucas, S.; Sevin, A.; Georges, P.; Passarrius, O.; Duranthon, F. Frequency of Dental Caries in Four Historical Populations from the Chalcolithic to the Middle Ages. Int. J. Dent. 2011, 2011, 519691. [Google Scholar] [CrossRef]
- Mandel, I.D. Chemotherapeutic Agents for Controlling Plaque and Gingivitis. J. Clin. Periodontol. 1988, 15, 488–498. [Google Scholar] [CrossRef]
- Manipal, S.; Hussain, S.; Wadgave, U.; Duraiswamy, P.; Ravi, K. The Mouthwash War—Chlorhexidine vs. Herbal Mouth Rinses: A Meta-Analysis. J. Clin. Diagn. Res. 2016, 10, ZC81–ZC83. [Google Scholar] [CrossRef] [PubMed]
- Ferracane, J.L. A Historical Perspective on Dental Composite Restorative Materials. J. Funct. Biomater. 2024, 15, 173. [Google Scholar] [CrossRef]
- AL-Ibrahim, I.; Shono, N.; Al-Saud, L.; Al-Nahedh, H. Five Years of Restorative Resin-Based Composite Advancements: A Narrative Review. BMC Oral Health 2025, 25, 1061. [Google Scholar] [CrossRef]
- Alomran, W.K.; Nizami, M.Z.I.; Xu, H.H.K.; Sun, J. Evolution of Dental Resin Adhesives-A Comprehensive Review. J. Funct. Biomater. 2025, 16, 104. [Google Scholar] [CrossRef]
- Bompolaki, D.; Lubisich, E.B.; Fugolin, A.P. Resin-Based Composites for Direct and Indirect Restorations: Clinical Applications, Recent Advances, and Future Trends. Dent. Clin. N. Am. 2022, 66, 517–536. [Google Scholar] [CrossRef] [PubMed]
- Arun, D.; Adikari Mudiyanselage, D.; Gulam Mohamed, R.; Liddell, M.; Monsur Hassan, N.M.; Sharma, D. Does the Addition of Zinc Oxide Nanoparticles Improve the Antibacterial Properties of Direct Dental Composite Resins? A Systematic Review. Materials 2020, 14, 40. [Google Scholar] [CrossRef]
- Aminoroaya, A.; Neisiany, R.E.; Khorasani, S.N.; Panahi, P.; Das, O.; Madry, H.; Cucchiarini, M.; Ramakrishna, S. A Review of Dental Composites: Challenges, Chemistry Aspects, Filler Influences, and Future Insights. Compos. Part B Eng. 2021, 216, 108852. [Google Scholar] [CrossRef]
- de Araujo, D.B.; Campos, E.d.J.; Bastos, I.H.d.A.; de Paula, D.M.; Tenório Junior, E.R.; de Araujo, R.P.C. Mouthrinses: Active Ingredients, Pharmacological Properties and Indications. RGO Rev. Gaúcha Odontol. Online 2012, 60, 349–357. [Google Scholar]
- Charugundla, B.R.; Anjum, S.; Mocherla, M. Comparative Effect of Fluoride, Essential Oil and Chlorhexidine Mouth Rinses on Dental Plaque and Gingivitis in Patients with and without Dental Caries: A Randomized Controlled Trial. Int. J. Dent. Hyg. 2015, 13, 104–109. [Google Scholar] [CrossRef]
- Atabek, D.; Alaçam, A.; Çakılcı, B.; Berkkan, A. A Comparative Study of Fluoride Containing Chlorhexidine and Non-Chlorhexidine Mouthrinses in a Teenage Group. Int. Dent. Res. 2012, 2, 1–7. [Google Scholar] [CrossRef][Green Version]
- Ustrell-Borràs, M.; Traboulsi-Garet, B.; Gay-Escoda, C. Alcohol-Based Mouthwash as a Risk Factor of Oral Cancer: A Systematic Review. Med. Oral Patol. Oral Cir. Bucal 2020, 25, e1–e12. [Google Scholar] [CrossRef] [PubMed]
- Craig, R.G.; Powers, J.M. (Eds.) Restorative Dental Materials, 11th ed.; Mosby: St. Louis, MO, USA, 2002; ISBN 978-0-323-01442-7. [Google Scholar]
- Filipović, G.; Stevanović, M.D.; Stojanović-Radić, Z.; Obradović, R.; Randjelović, P.J.; Radulović, N.S. Choosing the Right Essential Oil for a Mouthwash: Chemical, Antimicrobial and Cytotoxic Studies. Chem. Biodivers. 2020, 17, e2000748. [Google Scholar] [CrossRef] [PubMed]
- Maltz, M. Over-the-Counter Preventive and Therapeutic Oral Products. Braz. Oral Res. 2009, 23, 4–7. [Google Scholar] [CrossRef]
- Radzki, D.; Wilhelm-Węglarz, M.; Pruska, K.; Kusiak, A.; Ordyniec-Kwaśnica, I. A Fresh Look at Mouthwashes—What Is Inside and What Is It For? Int. J. Environ. Res. Public Health 2022, 19, 3926. [Google Scholar] [CrossRef] [PubMed]
- Marinho, V.C.; Chong, L.Y.; Worthington, H.V.; Walsh, T. Fluoride Mouthrinses for Preventing Dental Caries in Children and Adolescents. Cochrane Database Syst. Rev. 2016, 2016, CD002284. [Google Scholar] [CrossRef]
- Ardizzoni, A.; Pericolini, E.; Paulone, S.; Orsi, C.F.; Castagnoli, A.; Oliva, I.; Strozzi, E.; Blasi, E. In Vitro Effects of Commercial Mouthwashes on Several Virulence Traits of Candida Albicans, Viridans Streptococci and Enterococcus Faecalis Colonizing the Oral Cavity. PLoS ONE 2018, 13, e0207262. [Google Scholar] [CrossRef] [PubMed]
- George, R.; Kavyashree, G. Effect of Four Mouth Rinses on Microhardness of Esthetic Restorative Material: An In Vitro Study. J. Int. Oral Health 2017, 9, 55. [Google Scholar] [CrossRef]
- de Moraes Porto, I.C.; das Neves, L.E.; de Souza, C.K.; Parolia, A.; Barbosa dos Santos, N. A Comparative Effect of Mouthwashes with Different Alcohol Concentrations on Surface Hardness, Sorption and Solubility of Composite Resins. Oral Health Dent. Manag. 2014, 13, 502–506. [Google Scholar]
- Al-Samadani, K.H. Surface Hardness of Dental Composite Resin Restorations in Response to Preventive Agents. J. Contemp. Dent. Pract. 2016, 17, 978–984. [Google Scholar] [CrossRef]
- Nowak, M.; Kalamarz, I.; Chladek, G. Mechanical Properties of Easy Fill Composites after Storage in Mouthwashes. J. Achiev. Mater. Manuf. Eng. 2018, 88, 25–34. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, J. Effect of Immersion in Various Media on the Sorption, Solubility, Elution of Unreacted Monomers, and Flexural Properties of Two Model Dental Composite Compositions. J. Mater. Sci. Mater. Med. 2008, 19, 2477–2483. [Google Scholar] [CrossRef]
- de Oliveira, A.L.B.M.; Botta, A.C.; Campos, J.Á.D.B.; Garcia, P.P.N.S. Effects of Immersion Media and Repolishing on Color Stability and Superficial Morphology of Nanofilled Composite Resin. Microsc. Microanal. 2014, 20, 1234–1239. [Google Scholar] [CrossRef]
- Álvarez-Horna, J.; Aliaga-Mariñas, A.; Castro-Ramirez, L.; López-Gurreonero, C.; Cornejo-Pinto, A.; Scipión-Castro, R.; Cayo-Rojas, C. Color Stability of Resin Composites Immersed for Different Durations in Alcohol-Based and Alcohol-Free Mouthwashes: An In Vitro Study. J. Clin. Exp. Dent. 2025, 17, e1189–e1196. [Google Scholar] [CrossRef] [PubMed]
- de Angelis, F.; Mandatori, D.; Schiavone, V.; Melito, F.P.; Valentinuzzi, S.; Vadini, M.; Di Tomo, P.; Vanini, L.; Pelusi, L.; Pipino, C.; et al. Cytotoxic and Genotoxic Effects of Composite Resins on Cultured Human Gingival Fibroblasts. Materials 2021, 14, 5225. [Google Scholar] [CrossRef]
- Dantagnan, C.-A.; Babajko, S.; Nassif, A.; Houari, S.; Jedeon, K.; François, P.; Dursun, E.; Attal, J.-P.; Bosco, J. Analysis of Resin-Based Dental Materials’ Composition Depending on Their Clinical Applications. Polymers 2024, 16, 1022. [Google Scholar] [CrossRef] [PubMed]
- Stencel, R.; Kasperski, J.; Pakieła, W.; Mertas, A.; Bobela, E.; Barszczewska-Rybarek, I.; Chladek, G. Properties of Experimental Dental Composites Containing Antibacterial Silver-Releasing Filler. Materials 2018, 11, 1031. [Google Scholar] [CrossRef] [PubMed]
- Penn, R.W.; Craig, R.G.; Tesk, J.A. Diametral Tensile Strength and Dental Composites. Dent. Mater. 1987, 3, 46–48. [Google Scholar] [CrossRef] [PubMed]
- Galvão, M.R.; Caldas, S.G.F.R.; Calabrez-Filho, S.; A Campos, E.; Bagnato, V.S.; Rastelli, A.N.S.; Andrade, M.F. Compressive Strength of Dental Composites Photo-Activated with Different Light Tips. Laser Phys. 2013, 23, 045604. [Google Scholar] [CrossRef]
- EN ISO 4049:2019; Dentistry—Polymer-Based Restorative Materials. ISO International Organization for Standardization: Geneva, Switzerland, 2019.
- Bociong, K.; Szczesio, A.; Sokolowski, K.; Domarecka, M.; Sokolowski, J.; Krasowski, M.; Lukomska-Szymanska, M. The Influence of Water Sorption of Dental Light-Cured Composites on Shrinkage Stress. Materials 2017, 10, 1142. [Google Scholar] [CrossRef]
- Miranda, D.d.A.; Bertoldo, C.E.D.S.; Aguiar, F.H.B.; Lima, D.A.N.L.; Lovadino, J.R. Effects of Mouthwashes on Knoop Hardness and Surface Roughness of Dental Composites after Different Immersion Times. Braz. Oral Res. 2011, 25, 168–173. [Google Scholar] [CrossRef]
- International Standards for Colour. In Colour Design; Woodhead Publishing: Cambridge, UK, 2012; pp. 177–218. ISBN 978-0-85709-553-4.
- Santacruz, C.I.; Fajardo, J.I.; Paltán, C.A.; del Carmen Armas-Vega, A.; León, E.V. Comparative In Vitro Analysis of Mechanical Properties in Three High-Viscosity Bulk-Fill Composite Resins. J. Compos. Sci. 2025, 9, 623. [Google Scholar] [CrossRef]
- Abuzinadah, A.J.; Merdad, Y.M.A.; Aldharrab, R.S.; Almutairi, W.A.; Yeslam, H.E.; Hasanain, F.A. Microhardness and Compressive Strength of Bulk Fill Glass Hybrid Material and Other Direct Restorative Materials. J. Compos. Sci. 2024, 8, 508. [Google Scholar] [CrossRef]
- Chladek, G.; Tegnander, T. The Effect of Mouthwashes on Changes in the Aesthetic Properties of Dental Nanocomposite. J. Achiev. Mater. Manuf. Eng. 2023, 121, 131–140. [Google Scholar] [CrossRef]
- Miot, H.A. Correlation Analysis in Clinical and Experimental Studies. J. Vasc. Bras. 2018, 17, 275–279. [Google Scholar] [CrossRef] [PubMed]
- ElEmbaby, A.E.-S. The Effects of Mouth Rinses on the Color Stability of Resin-Based Restorative Materials. J. Esthet. Restor. Dent. 2014, 26, 264–271. [Google Scholar] [CrossRef]
- Fidan, M.; Tuncdemir, M.T. Effect of Whitening Mouthrinses on Color Change, Whiteness Change, Surface Roughness, and Hardness of Stained Resin Composites. Am. J. Dent. 2023, 36, 25–30. [Google Scholar]
- Manzoor, S.; Arooj, Z.; Waqas, M.A.; Irshad, N.; Saeed, A.; Malik, A.; Sarfaraz, Z.; Shaukat, M.S. Surface Microhardness Of Microhybrid And Nanocomposite After Storage In Mouth Washes. J. Ayub Med. Coll. Abbottabad JAMC 2022, 34, 540–547. [Google Scholar] [CrossRef] [PubMed]
- Derigi, L.P.; Barros, L.S.; Sugii, M.M.; Turssi, C.P.; França, F.; Basting, R.T.; Vieira, W.F. Effect of Commercial Mouth Rinses on Physical Properties of Conventional and Bulk-Fill Resin Composites. Oper. Dent. 2023, 48, 720–731. [Google Scholar] [CrossRef]
- Alessa, N.A. Effect of Mouthwashes on the Microhardness of Aesthetic Composite Restorative Materials. An. Sist. Sanit. Navar. 2023, 46, e1049. [Google Scholar] [CrossRef]
- Elamin, H.O.; Abubakr, N.H.; Ibrahim, Y.E. Identifying the Tooth Shade in Group of Patients Using Vita Easyshade. Eur. J. Dent. 2015, 9, 213–217. [Google Scholar] [CrossRef]
- Gürgan, S.; Onen, A.; Köprülü, H. In Vitro Effects of Alcohol-Containing and Alcohol-Free Mouthrinses on Microhardness of Some Restorative Materials. J. Oral Rehabil. 1997, 24, 244–246. [Google Scholar] [PubMed]
- Lu, H.; Roeder, L.B.; Lei, L.; Powers, J.M. Effect of Surface Roughness on Stain Resistance of Dental Resin Composites. J. Esthet. Restor. Dent. Off. Publ. Am. Acad. Esthet. Dent. Al 2005, 17, 102–108; discussion 109. [Google Scholar] [CrossRef]
- Abzal, M.S.; Rathakrishnan, M.; Prakash, V.; Vivekanandhan, P.; Subbiya, A.; Sukumaran, V.G. Evaluation of Surface Roughness of Three Different Composite Resins with Three Different Polishing Systems. J. Conserv. Dent. JCD 2016, 19, 171–174. [Google Scholar] [CrossRef] [PubMed]
- Tosco, V.; Monterubbianesi, R.; Orilisi, G.; Procaccini, M.; Grandini, S.; Putignano, A.; Orsini, G. Effect of Four Different Finishing and Polishing Systems on Resin Composites: Roughness Surface and Gloss Retention Evaluations. Minerva Stomatol. 2020, 69, 207–214. [Google Scholar] [CrossRef]
- Rigo, L.C.; Bordin, D.; Fardin, V.P.; Coelho, P.G.; Bromage, T.G.; Reis, A.; Hirata, R. Influence of Polishing System on the Surface Roughness of Flowable and Regular-Viscosity Bulk Fill Composites. Int. J. Periodontics Restor. Dent. 2018, 38, e79–e86. [Google Scholar] [CrossRef]
- Erturk-Avunduk, A.T.; Atılan-Yavuz, S.; Filiz, H.; Cengiz-Yanardag, E. A Comparative Study of Polishing Systems on Optical Properties and Surface Roughness of Additively Manufactured and Conventional Resin Based Composites. Sci. Rep. 2024, 14, 25658. [Google Scholar] [CrossRef] [PubMed]
- Augusto, M.G.; de Andrade, G.S.; Mathias-Santamaria, I.F.; Piva, A.M.d.O.D.; Tribst, J.P.M. Comparison of Polishing Systems on the Surface Roughness of Resin Based Composites Containing Different Monomers. J. Compos. Sci. 2022, 6, 146. [Google Scholar] [CrossRef]
- Kusakabe, Y.; Shindo, Y.; Kawai, T.; Maeda-Yamamoto, M.; Wada, Y. Relationships between the Response of the Sweet Taste Receptor, Salivation toward Sweeteners, and Sweetness Intensity. Food Sci. Nutr. 2020, 9, 2036. [Google Scholar] [CrossRef] [PubMed]
- Yazicioglu, O.; Ucuncu, M.K.; Guven, K. Ingredients in Commercially Available Mouthwashes. Int. Dent. J. 2023, 74, 223–241. [Google Scholar] [CrossRef]
- Vranic, E.; Lacevic, A.; Mehmedagic, A.; Uzunovic, A. Formulation ingredients for toothpastes and mouthwashes. Bosn. J. Basic Med. Sci. 2004, 4, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, K.; Asmat, M.; Jawed, S.; Mushtaque, A.; Mohsin, F.; Hanif, S.; Sheikh, N. Role of different ingredients of tooth pastes and mouthwashes in oral health. J. Pak. Med. Assoc. 2011, 20, 8. [Google Scholar]
- Rekha Mallick, R.; Sarangi, P.; Suman, S.; Sekhar Sahoo, S.; Bajoria, A.; Sharma, G. An In Vitro Analysis of the Effects of Mouthwashes on the Surface Properties of Composite Resin Restorative Material. Cureus 2024, 16, e65021. [Google Scholar] [CrossRef]
- Al-Samadani, K.H. The Effect of Preventive Agents (Mouthwashes/Gels) on the Color Stability of Dental Resin-Based Composite Materials. Dent. J. 2017, 5, E18. [Google Scholar] [CrossRef]
- Mohamed-Tahir, M.A.; Tan, H.Y.; Woo, A.A.S.; Yap, A.U.J. Effects of pH on the Microhardness of Resin-Based Restorative Materials. Oper. Dent. 2005, 30, 661–666. [Google Scholar]
- Szalewski, L.; Wójcik, D.; Sowa, M.; Vivcharenko, V.; Pałka, K. Influence of Low pH on the Microhardness and Roughness Surface of Dental Composite-A Preliminary Study. Materials 2024, 17, 3443. [Google Scholar] [CrossRef]
- Munawar, A.H.; Febrida, R.; Nurdin, D. Influence of Alcohol-Containing Mouthwash and Alcohol-Free Mouthwash towards the Hybrid Composite Restoration Materials Surface Hardness. Padjadjaran J. Dent. 2013, 25, 15574. [Google Scholar] [CrossRef]
- Yılmaz, C.; Uygun, L.A. The Effect of Different Whitening Mouthwashes and Simulated Tooth Brushing on Surface Roughness and Microhardness of Nanohybrid Resin Composite: An in Vitro Study. BMC Oral Health 2025, 25, 631. [Google Scholar] [CrossRef]
- Hamdy, T.M.; Abdelnabi, A.; Othman, M.S.; Bayoumi, R.E.; Abdelraouf, R.M. Effect of Different Mouthwashes on the Surface Microhardness and Color Stability of Dental Nanohybrid Resin Composite. Polymers 2023, 15, 815. [Google Scholar] [CrossRef]
- Ferracane, J.L. Hygroscopic and Hydrolytic Effects in Dental Polymer Networks. Dent. Mater. 2006, 22, 211–222. [Google Scholar] [CrossRef]
- Chladek, G.; Basa, K.; Żmudzki, J.; Malara, P.; Nowak, A.J.; Kasperski, J. Influence of Aging Solutions on Wear Resistance and Hardness of Selected Resin-Based Dental Composites. Acta Bioeng. Biomech. 2016, 18, 43–52. [Google Scholar]
- Torno, V.; Soares, P. Tribological Behavior and Wear Mechanisms of Dental Resin Composites with Different Polymeric Matrices. J. Mech. Behav. Biomed. Mater. 2023, 144, 105962. [Google Scholar] [CrossRef]
- Cho, G.C.; Kaneko, L.M.; Donovan, T.E.; White, S.N. Diametral and Compressive Strength of Dental Core Materials. J. Prosthet. Dent. 1999, 82, 272–276. [Google Scholar] [CrossRef] [PubMed]
- Williams, P.D.; Smith, D.C. Measurement of the Tensile Strength of Dental Restorative Materials by Use of a Diametral Compression Test. J. Dent. Res. 1971, 50, 436–442. [Google Scholar] [CrossRef] [PubMed]
- Dejak, B.; Młotkowski, A. A Comparison of Stresses in Molar Teeth Restored with Inlays and Direct Restorations, Including Polymerization Shrinkage of Composite Resin and Tooth Loading during Mastication. Dent. Mater. 2015, 31, e77–e87. [Google Scholar] [CrossRef]
- Borkowski, K.; Kotousov, A.; Kahler, B. Effect of Material Properties of Composite Restoration on the Strength of the Restoration-Dentine Interface Due to Polymerization Shrinkage, Thermal and Occlusal Loading. Med. Eng. Phys. 2007, 29, 671–676. [Google Scholar] [CrossRef]
- van Noort, R.; Cardew, G.E.; Howard, I.C. A Study of the Interfacial Shear and Tensile Stresses in a Restored Molar Tooth. J. Dent. 1988, 16, 286–293. [Google Scholar] [CrossRef]
- Mokeem, L.S.; Garcia, I.M.; Melo, M.A. Degradation and Failure Phenomena at the Dentin Bonding Interface. Biomedicines 2023, 11, 1256. [Google Scholar] [CrossRef]
- Kopperud, S.E.; Tveit, A.B.; Gaarden, T.; Sandvik, L.; Espelid, I. Longevity of Posterior Dental Restorations and Reasons for Failure. Eur. J. Oral Sci. 2012, 120, 539–548. [Google Scholar] [CrossRef] [PubMed]
- Ausiello, P.; Ciaramella, S.; Fabianelli, A.; Gloria, A.; Martorelli, M.; Lanzotti, A.; Watts, D.C. Mechanical Behavior of Bulk Direct Composite versus Block Composite and Lithium Disilicate Indirect Class II Restorations by CAD-FEM Modeling. Dent. Mater. 2017, 33, 690–701. [Google Scholar] [CrossRef] [PubMed]
- Mitra, S.B.; Wu, D.; Holmes, B.N. An Application of Nanotechnology in Advanced Dental Materials. J. Am. Dent. Assoc. 2003, 134, 1382–1390. [Google Scholar] [CrossRef]
- Wang, Y.; Hua, H.; Li, W.; Wang, R.; Jiang, X.; Zhu, M. Strong Antibacterial Dental Resin Composites Containing Cellulose Nanocrystal/Zinc Oxide Nanohybrids. J. Dent. 2019, 80, 23–29. [Google Scholar] [CrossRef]
- Rohr, N.; Fischer, J. Effect of Aging and Curing Mode on the Compressive and Indirect Tensile Strength of Resin Composite Cements. Head Face Med. 2017, 13, 22. [Google Scholar] [CrossRef]
- Warangkulkasemkit, S.; Pumpaluk, P. Comparison of Physical Properties of Three Commercial Composite Core Build-up Materials. Dent. Mater. J. 2019, 38, 177–181. [Google Scholar] [CrossRef] [PubMed]
- Fuxiang, S.; Qian, C.; Rui, Y.; Naiyu, S.; Bin, L. A Novel High-Strength Dental Resin Composite Based on BaSi2O2N2 for Caries Restoration. ACS Biomater. Sci. Eng. 2025, 11, 2106–2114. [Google Scholar] [CrossRef]
- Tete, S.; Mishra, L.; Jena, D.; Govind, S.; Lukomska-Szymanska, M.M. Influence of Alcoholic and Non-Alcoholic Beverages on the Compressive Strength of Core Build-up Bulk-Fill Resin Materials. Dent. Med. Probl. 2025, 62, 701–709. [Google Scholar] [CrossRef]
- Ghinea, R.; Pérez, M.M.; Herrera, L.J.; Rivas, M.J.; Yebra, A.; Paravina, R.D. Color Difference Thresholds in Dental Ceramics. J. Dent. 2010, 38, e57–e64. [Google Scholar] [CrossRef]
- Paravina, R.D.; Ghinea, R.; Herrera, L.J.; Bona, A.D.; Igiel, C.; Linninger, M.; Sakai, M.; Takahashi, H.; Tashkandi, E.; Perez, M.d.M. Color Difference Thresholds in Dentistry. J. Esthet. Restor. Dent. 2015, 27, S1–S9. [Google Scholar] [CrossRef]
- Lee, Y.-K. Comparison of CIELAB DeltaE(*) and CIEDE2000 Color-Differences after Polymerization and Thermocycling of Resin Composites. Dent. Mater. 2005, 21, 678–682. [Google Scholar] [CrossRef] [PubMed]
- Morais Sampaio, G.A.d.; Rangel Peixoto, L.; de Vasconcelos Neves, G.; Nascimento Barbosa, D. do Effect of Mouthwashes on Color Stability of Composite Resins: A Systematic Review. J. Prosthet. Dent. 2021, 126, 386–392. [Google Scholar] [CrossRef]
- Paravina, R.D.; Pérez, M.M.; Ghinea, R. Acceptability and Perceptibility Thresholds in Dentistry: A Comprehensive Review of Clinical and Research Applications. J. Esthet. Restor. Dent. 2019, 31, 103–112. [Google Scholar] [CrossRef]
- Mokrzycki, W.S.; Tatol, M. Color Difference ΔE: A Survey. Mach. Graph. Vis. 2011, 20, 383–411. [Google Scholar]
- Lindsey, D.T.; Wee, A.G. Perceptibility and Acceptability of CIELAB Color Differences in Computer-Simulated Teeth. J. Dent. 2007, 35, 593–599. [Google Scholar] [CrossRef]
- Allccahuaman-Avalos, R.; Medina-Sánchez, R.; Castro-Ramirez, L.; Ladera-Castañeda, M.; Cervantes-Ganoza, L.; Martínez-Campos, R.; Solís-Dante, F.; Aliaga-Mariñas, A.; Verástegui-Sandoval, A.; Cayo-Rojas, C. In Vitro Color Stability Evaluation of Three Polished and Unpolished Nanohybrid Resin Composites Immersed in a 0.12% Chlorhexidine-Based Mouthwash at Different Times. Polymers 2023, 15, 1339. [Google Scholar] [CrossRef] [PubMed]
- Laky, M.; Brandl, S.; Arslan, M.; Boryshchuk, D.; Cześnikiewicz-Guzik, M.; Rausch-Fan, X.; Moritz, A.; Laky, B. Color Stability of Frequently Used Aesthetic Restorative Materials Following in Vitro Exposure to Chlorhexidine- and Octenidine-Based Mouthrinses. BMC Oral Health 2025, 25, 450. [Google Scholar] [CrossRef]
- Jaganath, B.M.; Krishnegowda, S.C.; Rudranaik, S.; Beedubail, S.P. Assessment of Color Changes in Teeth and Composite Resins under the Influence of Chlorhexidine with and without Anti-Discoloration System: An in Vitro Study. J. Conserv. Dent. JCD 2023, 26, 52–55. [Google Scholar] [CrossRef] [PubMed]
- Van Swaaij, B.W.M.; van der Weijden, G.A.F.; Bakker, E.W.P.; Graziani, F.; Slot, D.E. Does Chlorhexidine Mouthwash, with an Anti-Discoloration System, Reduce Tooth Surface Discoloration without Losing Its Efficacy? A Systematic Review and Meta-Analysis. Int. J. Dent. Hyg. 2020, 18, 27–43. [Google Scholar] [CrossRef]
- Yaghobee, S.; Dorkoosh, F.A.; Kouhestani, F.; Mozafari, G.; Aslroosta, H. Comparison of 0.2% Chlorhexidine Mouthwash with and without Anti-Discoloration System in Patients with Chronic Periodontitis: A Randomized Controlled Clinical Trial. J. Adv. Periodontol. Implant Dent. 2019, 11, 63–68. [Google Scholar] [CrossRef]
- Festuccia, M.S.C.C.; Garcia, L.d.F.R.; Cruvinel, D.R.; Pires-De-Souza, F.d.C.P. Color Stability, Surface Roughness and Microhardness of Composites Submitted to Mouthrinsing Action. J. Appl. Oral Sci. Rev. FOB 2012, 20, 200–205. [Google Scholar] [CrossRef]
- Khosravi, M.; Esmaeili, B.; Nikzad, F.; Khafri, S. Color Stability of Nanofilled and Microhybrid Resin-Based Composites Following Exposure to Chlorhexidine Mouthrinses: An In Vitro Study. J. Dent. Tehran Iran 2016, 13, 116–125. [Google Scholar]
- Toz Akalin, T.; Genc, G.; Korkmaz Ceyhan, Y.; Ozturk Bozkurt, F. The Effect of Mouth Rinses on the Color Stability of Sonicfill and a Nanohybrid Composite. J. Istanb. Univ. Fac. Dent. 2016, 50, 17–23. [Google Scholar] [CrossRef]
- Celik, C.; Yuzugullu, B.; Erkut, S.; Yamanel, K. Effects of Mouth Rinses on Color Stability of Resin Composites. Eur. J. Dent. 2008, 2, 247–253. [Google Scholar] [CrossRef] [PubMed]
- da Silva, G.P.W.; Mesquita, V.U.; Pontes, D.G. Effect of Mouthwashes on the Color Stability of a Nanohybrid Composite Resin. Gen. Dent. 2024, 72, 38–41. [Google Scholar] [PubMed]
- Bazin, P.; Safarzadeh Khosroshahi, S.; Ebrahimgol, S.; Mahdisiar, F. Comparative Evaluation of Color Stability in Nanohybrid and Microhybrid Composites Exposed to Misswake Mouthwash: An In Vitro Analysis. Int. J. Dent. 2025, 2025, 7788956. [Google Scholar] [CrossRef]
- Bansal, S.; Gupta, S.; Gupta, S.; Bogra, P.; Bansal, R.; Grover, V.; Gupta, S. Comparative Evaluation of Color Stability and Surface Roughness of Bulk-Fill and Nanohybrid Composites Following Long-Term Mouthrinse Exposure: An In Vitro Study. Cureus 2025, 17, e84320. [Google Scholar] [CrossRef]
- Yılmaz, K.; Odabaş Hajiyev, T.; Özcan Altınsoy, G.; Özarslan, M.M. Effects of Apple Vinegar, Mouthwashes, and Bleaching on Color Stability and Surface Properties of Fiber-Reinforced and Non-Reinforced Restorative Materials. Polymers 2025, 17, 2552. [Google Scholar] [CrossRef]
- Mara da Silva, T.; Barbosa Dantas, D.C.; Franco, T.T.; Franco, L.T.; Rocha Lima Huhtala, M.F. Surface Degradation of Composite Resins under Staining and Brushing Challenges. J. Dent. Sci. 2019, 14, 87–92. [Google Scholar] [CrossRef]
- Söderholm, K.J.; Zigan, M.; Ragan, M.; Fischlschweiger, W.; Bergman, M. Hydrolytic Degradation of Dental Composites. J. Dent. Res. 1984, 63, 1248–1254. [Google Scholar] [CrossRef]
- Condò, R.; Mampieri, G.; Cioffi, A.; Pirelli, P.; Giancotti, A.; Maiolo, L.; Maita, F.; Convertino, A.; Lucarini, I.; Notargiacomo, A.; et al. Relationship between Reflectivity, Chemical Composition and Mechanical Behaviour of Orthodontic Bonding Nanofiller Resin Materials: A Proposal of an Alternative Method of Investigation. Appl. Sci. 2022, 12, 12538. [Google Scholar] [CrossRef]
- Oivanen, M.; Keulemans, F.; Garoushi, S.; Vallittu, P.K.; Lassila, L. The Effect of Refractive Index of Fillers and Polymer Matrix on Translucency and Color Matching of Dental Resin Composite. Biomater. Investig. Dent. 2021, 8, 48–53. [Google Scholar] [CrossRef]
- Pfeffer, A.; Hahnel, S.; Rauch, A.; Rosentritt, M. Effects of Translucency-Enhancing Coloring Liquids on the Mechanical Properties of 3Y- and 4Y-TZP Zirconia Ceramics. Ceramics 2025, 8, 92. [Google Scholar] [CrossRef]
- Xu, P.; Wang, H.; Cui, W.; Chen, Q.; Tu, B.; Sang, X.; Wang, W.; Fu, Z. ZnO·2.7Al2O3 Nanocomposite with High Optical Transparency. J. Am. Ceram. Soc. 2022, 105, 3735–3739. [Google Scholar] [CrossRef]
- Reise, M.; Kranz, S.; Heyder, M.; Beck, J.; Roth, C.; Guellmar, A.; von Eggeling, F.; Schubert, U.; Löffler, B.; Sigusch, B. Salivary Pellicle Formed on Dental Composites Evaluated by Mass Spectrometry—An In Situ Study. Molecules 2023, 28, 6804. [Google Scholar] [CrossRef]
- Li, F.; Weir, M.D.; Fouad, A.F.; Xu, H.H.K. Effect of Salivary Pellicle on Antibacterial Activity of Novel Antibacterial Dental Adhesives Using a Dental Plaque Microcosm Biofilm Model. Dent. Mater. 2014, 30, 182–191. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, S.; Fantozzi, G.; Bernardi, S.; Antonouli, S.; Continenza, M.A.; Macchiarelli, G. Commercial Oral Hygiene Products and Implant Collar Surfaces: Scanning Electron Microscopy Observations. Can. J. Dent. Hyg. 2020, 54, 26–31. [Google Scholar]
- Rotar, R.; Cândea, A.; Măroiu, A.; Faur, A.-B.; Cuzic, C.; Rotar, R.-E.; Jivănescu, A. Impact of Various Mouthwashes on the Color Stability of Hybrid Ceramic and Reinforced Composite CAD/CAM Restorative Materials: An In Vitro Study. Materials 2026, 19, 758. [Google Scholar] [CrossRef]
- da Silva, E.M.; de Sá Rodrigues, C.U.F.; Dias, D.A.; da Silva, S.; Amaral, C.M.; Guimarães, J.G.A. Effect of Toothbrushing-Mouthrinse-Cycling on Surface Roughness and Topography of Nanofilled, Microfilled, and Microhybrid Resin Composites. Oper. Dent. 2014, 39, 521–529. [Google Scholar] [CrossRef]
- Trauth, K.G.S.; de Godoi, A.P.T.; Colucci, V.; Corona, S.A.M.; Catirse, A.B.C.E.B. The Influence of Mouthrinses and Simulated Toothbrushing on the Surface Roughness of a Nanofilled Composite Resin. Braz. Oral Res. 2012, 26, 209–214. [Google Scholar] [CrossRef]
- Gehlot, P.M.; Sudeep, P.; Manjunath, V.; Annapoorna, B.M.; Prasada, L.K.; Nandlal, B. Influence of Various Desensitizing Mouthrinses and Simulated Toothbrushing on Surface Roughness and Microhardness of Tetric N-Ceram Bulk-Fill Resin Composite: An In Vitro Study and Scanning Electron Microscope Analysis. Eur. J. Dent. 2022, 16, 820–827. [Google Scholar] [CrossRef] [PubMed]
- Sah, A.; Kumar, A.; Telang, A.D.; Kumar, R.; Yadav, M.; Abhilash, A. Comparative Evaluation of Color Stability of Three Composite Resins in Two Mouth Rinses: An In Vitro Study. J. Pharm. Bioallied Sci. 2025, 17, S3144–S3146. [Google Scholar] [CrossRef] [PubMed]
- Szczesio-Wlodarczyk, A.; Garoushi, S.; Vallittu, P.; Bociong, K.; Lassila, L. Polymerization Shrinkage Stress of Contemporary Dental Composites: Comparison of Two Measurement Methods. Dent. Mater. J. 2024, 43, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Nocca, G.; Callà, C.; Martorana, G.E.; Cicillini, L.; Rengo, S.; Lupi, A.; Cordaro, M.; Luisa Gozzo, M.; Spagnuolo, G. Effects of Dental Methacrylates on Oxygen Consumption and Redox Status of Human Pulp Cells. BioMed. Res. Int. 2014, 2014, 956579. [Google Scholar] [CrossRef]






| Medium | Manufacturer | pH Value | Components Important for Categorization | Composition |
|---|---|---|---|---|
| Water | - | 5.8 | - | - |
| Beverly Hills perfect white black | Purity Laboratories Ltd., Dublin, Ireland | 5.5 |  | Aqua, Alcohol, Glycerin, PEG-40 Hydrogenated Castor Oil, Sodium Lauryl Sulfate, Sodium Saccharin, Aroma, Sodium Benzoate, Charcoal Powder, Sodium Fluoride, dyes (CI47005, CI44090) |
| Listerine Fresh Burst | Johnson & Johnson Poland Ltd., Warszawa, Poland | 4.18 |  | Water, alcohol, sorbitol, poloxamer 407, benzoic acid, sodium saccharin, eucalyptol, methyl salicylate, flavor, thymol, menthol, sodium benzoate, dyes (CI47005, CI42053). |
| Listerine Advanced White | Johnson & Johnson Poland Ltd., Warszawa, Poland | 6.13 |  | Water, sorbitol, propylene glycol, tetrapotassium pyrophosphate, pentasodium triphosphate, citric acid, poloxamer 407, sodium methyl cocoyl taurate, caprylyl glycol, eucalyptol, thymol, sodium saccharin, menthol, sodium fluoride (220 ppm F), sucralose. |
| Dentica Black White | Torf Corporation Ltd., Kąty Wrocławskie, Poland | 5.19 |  | Water, Glycerin, PEG-40, Hydrogenated Castor Oil, Charcoal Powder, Sodium Fluoride, Tetrapotassium Pyrophosphate, Tetrasodium Pyrophosphate, Aroma, Polyglyceryl-10 Myristate, Polyglycerin-10, Sodium Dehydroacetate, Sodium Citrate, Citric Acid, Sodium Benzoate, Limonene, dyes (CI42051, CI16255, CI47005). |
| Colgate Plax | Colgate-Palmolive Poland Ltd., Warszawa, Poland | 5.8 |  | Water, Glycerin, Propylene Glycol, Sorbitol, Poloxamer 407, Aroma, Cetylpyridinium Chloride, Potassium Sorbate, Sodium Fluoride (0.05%-225 ppm F), Sodium Saccharin, Menthol, dye CI 42051. |
| Biomed Citrus Fresh | Splat Global LLC, Riga, Latvia | 6.56 |  | Aqua, Hydrogenated Starch Hydrolysate, Calcium Lactate, Betula Alba Leaf Extract, Plantago Lanceolata Leaf Extract, Sodium Lauroyl Sarcosinate, Menthol, Stevia Rebaudiana Leaf Extract, Sodium Chloride, Ananas Sativus Fruit Extract, Arginine, Citrus Limon Peel Oil, Magnesium Aspartate, Zinc Gluconate, Copper Gluconate, Aroma, Maltodextrin, Glycerin, Tetrasodium Glutamate Diacetate, Polyglyceryl-4 Laurate/Sebacate, Polyglyceryl-6 Caprylate/Caprate, Benzyl Alcohol, Sodium Benzoate, Potassium Sorbate, Citric Acid, Limonene, Eugenol. |
| Biomed Super White | Splat Global LLC, Riga, Latvia | 5.76 |  | Water, Hydrogenated Starch Hydrolysate, Sodium Coco-Sulfate, Betula Alba Leaf Extract, Plantago Lanceolata Leaf Extract, Cocos Nucifera Fruit Extract, Lactobacillus, Xylitol, Stavia Rebaudiana Leaf Extract, Menthol, Papain, Polyglyceryl-4 Laurate/Sebacate, Polyglyceryl-6 Caprylate/Caprate, Aroma, Arginine, Magnesium Aspartate, Zinc Gluconate, Copper Gluconate, Glycerin, Benzyl Alcohol, Sodium Benzoate, Potassium Sorbate, Citric Acid, Tetrasodium Glutamate Diacetate. |
| ORAL-B Pro-Expert | Procter and Gamble DS Poland Ltd., Warszawa, Poland | 4.19 |  | Water, Glycerin, Aroma, Cetylpyridinium Chloride, Poloxamer 407, Methylparaben, Sodium Saccharin, Cinnamal, Propylparaben, Eugenol, dye CI 42090. |
| KIN B5 | Laboratorios KIN, Barcelona, Spain | 5.96 |  | Water, Sorbitol, Glycerin, PEG-40 Hydrogenated Castor Oil, Propylene Glycol, Xylitol, Panthenol, Zinc Lactate, Sodium Methylparaben, Citric Acid, Fragrance, Disodium EDTA, Niacinamide, Cetylpyridinium Chloride, Menthol, Sodium Fluoride, Propylparaben, d-Limonene. |
| Curasept ADS 205 | Curasept S.p.A., Saronno, Italy | 5.25 |  | Water, xylitol, propylene glycol, PEG-40 hydrogenated castor oil, ascorbic acid, chlorhexidine digluconate, flavor, sodium fluoride, poloxamer 407, sodium benzoate, sodium metabisulfite, sodium citrate, dye CI 42090. |
| Curasept ADS 012 | Curasept S.p.A., Saronno, Italy | 5.15 |  | Water, Xylitol, Propylene Glycol, PEG-40, Hydrogenated Castor Oil, Ascorbic Acid, Chlorhexidine Digluconate, Fragrance, Poloxamer 407, Sodium Methyldisulfonate, Sodium Citrate, dye CI 42090. |
| Curasept Biosmalto Sensitive Teeth | Curasept S.p.A., Saronno, Italy | 5.49 |  | Water, Xylitol, Fluorohydroxyapatite, Mg-Sr-Carbonate Hydroxyapatite conjugated with Chitosan, Strontium Acetate, Cocamidopropyl Betaine, Poloxamer 407, Potassium Acesulfame, Xanthan Gum, Ammonium Acryloyldimethyltaurate/VP Copolymer, Chlorphenesin, Ethylhexylglycerin, Phenoxyethanol, Sodium Benzoate, Sodium Monofluorophosphate, Aroma, Methylisothiazolinone, Citric Acid. |
 —ethanol;
—ethanol;  —fluorides;
—fluorides;  —charcoal;
—charcoal;  —cetylpyridinium chloride;
—cetylpyridinium chloride;  —chlorhexidine;
—chlorhexidine;  —apatites;
—apatites;  —based on natural ingredients.
—based on natural ingredients.Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Adeeb, S.; Kriger, A.; Żmudzki, J.; Kasperski, J.; Chladek, G. Effect of Mouthwashes on the Mechanical Properties and Color Stability of Composite Material. Materials 2026, 19, 1304. https://doi.org/10.3390/ma19071304
Adeeb S, Kriger A, Żmudzki J, Kasperski J, Chladek G. Effect of Mouthwashes on the Mechanical Properties and Color Stability of Composite Material. Materials. 2026; 19(7):1304. https://doi.org/10.3390/ma19071304
Chicago/Turabian StyleAdeeb, Saja, Artur Kriger, Jarosław Żmudzki, Jacek Kasperski, and Grzegorz Chladek. 2026. "Effect of Mouthwashes on the Mechanical Properties and Color Stability of Composite Material" Materials 19, no. 7: 1304. https://doi.org/10.3390/ma19071304
APA StyleAdeeb, S., Kriger, A., Żmudzki, J., Kasperski, J., & Chladek, G. (2026). Effect of Mouthwashes on the Mechanical Properties and Color Stability of Composite Material. Materials, 19(7), 1304. https://doi.org/10.3390/ma19071304









