Preliminary Assessment of Thermal and Mechanical Properties of a Graphene-Rich Carbon Coating on 3003-H14 Aluminum Alloy for Potential Anti-Icing Applications
Abstract
1. Introduction
2. Materials of Study
3. Chemically Exfoliated Graphene-Rich Carbon Platelets: Synthesis and Coating Application Procedure
4. Characterization Techniques
5. Results and Discussion
5.1. Surface Morphology Analysis
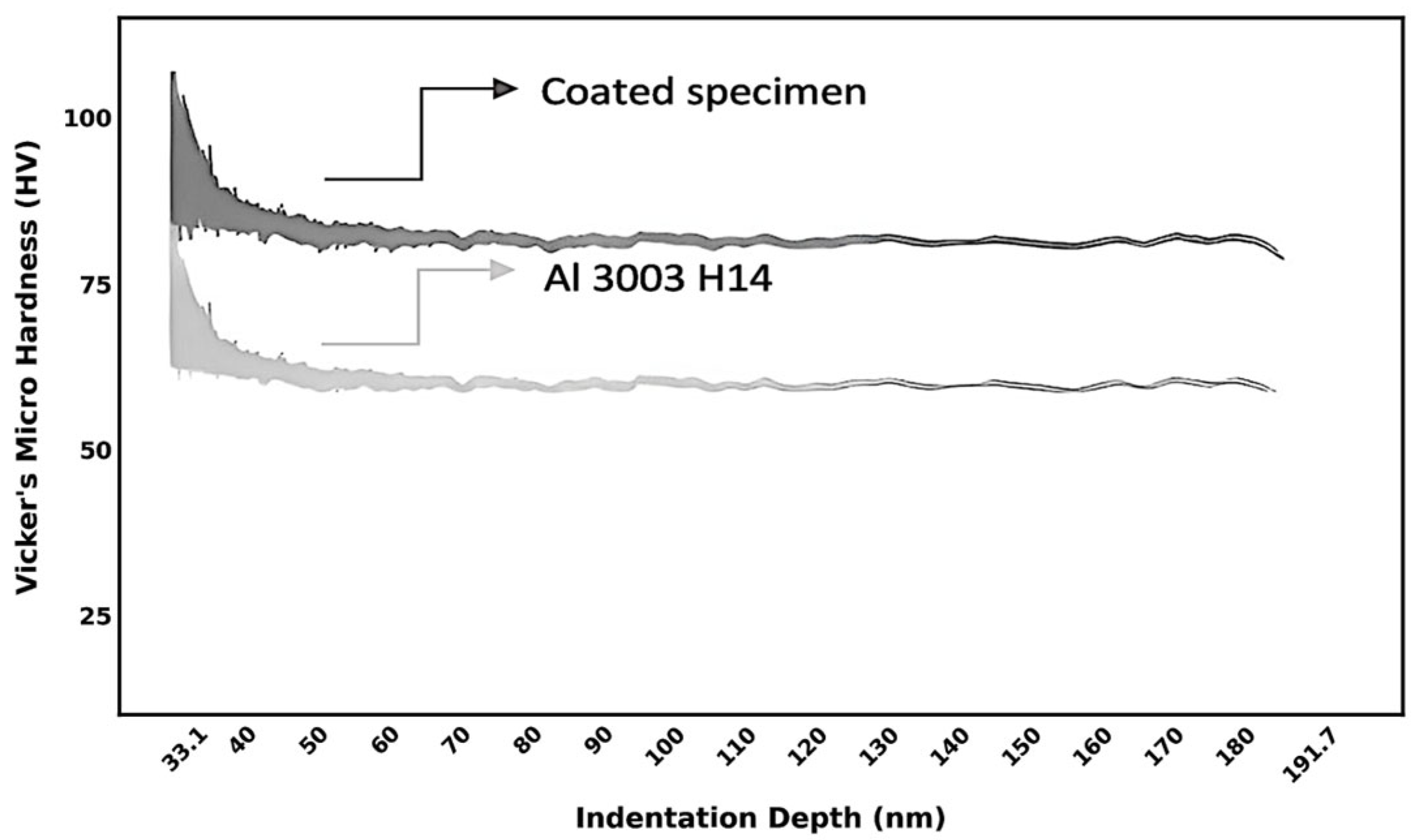
5.2. Microhardness Evaluation
5.3. Observed Surface-Cooling Behavior
6. Literature Context for Potential Anti-Icing Functionality
6.1. Hydrophobicity, Icephobicity, and Their Role in Aircraft Anti-Icing
6.2. Enhanced Corrosion Resistance
6.3. Environmental Protection and Surface Integrity
6.4. Aerospace-Engine Relevance (Cowls/Baffles)
6.5. Comparative Performance of Graphene-Rich Epoxy Coatings with CNT Epoxy Coatings
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Goraj, Z. An overview of the deicing and anti-icing technologies with prospects for the future. In Proceedings of the 24th International Congress of the Aeronautical Sciences, Yokohama, Japan, 29 August–3 September 2004. [Google Scholar]
- Cao, Y.; Tan, W.; Wu, Z. Aircraft icing: An ongoing threat to aviation safety. Aerosp. Sci. Technol. 2018, 75, 353–385. [Google Scholar] [CrossRef]
- Rekuviene, R.; Saeidiharzand, S.; Mažeika, L.; Samaitis, V.; Jankauskas, A.; Sadaghiani, A.K.; Gharib, G.; Muganlı, Z.; Koşar, A. A review on passive and active anti-icing and de-icing technologies. Appl. Therm. Eng. 2024, 250, 123474. [Google Scholar] [CrossRef]
- Piscitelli, F. Special Issue “Superhydrophobic and Icephobic Coatings as Passive Ice Protection Systems for Aeronautical Applications”. Coatings 2024, 14, 1288. [Google Scholar] [CrossRef]
- Zhu, Y. Structural Tailoring and Actuation Studies for Low Power Ultrasonic De-Icing of Aluminum and Composite Plates. Ph.D. Thesis, University of Pittsburgh, Pittsburgh, PA, USA, 2010. Available online: https://etda.libraries.psu.edu/catalog/10648 (accessed on 12 March 2026).
- Loughborough, D.L.; Green, H.E.; Roush, P.A. A Study of Wing De-Icer Performance on Mount Washington. Aero. Eng. Rev. 1948, 7, 41–50. [Google Scholar]
- Matthias, M.; Becker, D.; Koch, T.; Davies, C.; Ameduri, S. Development of a vibration-based deicing system for aircraft wings. In Active and Passive Smart Structures and Integrated Systems XIX; SPIE: Bellingham, WA, USA, 2025; Volume 13432, pp. 190–202. [Google Scholar] [CrossRef]
- Barbarino, S.; Saavedra Flores, E.I.; Ajaj, R.M.; Dayyani, I.; Friswell, M.I. A review on shape memory alloys with applications to morphing aircraft. Smart Mater. Struct. 2014, 23, 063001. [Google Scholar] [CrossRef]
- Myose, R.Y.; Horn, W.J.; Hwang, Y.; Herrero, J.; Huynh, C.; Boudraa, T. Application of Shape Memory Alloys for Leading Edge Deicing; SAE Technical Paper; SAE International: Warrendale, PA, USA, 1999. [Google Scholar] [CrossRef]
- Sommerwerk, H.; Horst, P.; Bansmer, S. Studies on electro impulse de-icing of a leading edge structure in an icing wind tunnel. In Proceedings of the 8th AIAA Atmospheric and Space Environments Conference, Washington, DC, USA, 13–17 June 2016. [Google Scholar] [CrossRef]
- Abdesselam, F.; Wijekoon, T.; Castellazzi, A.; Johnson, M.; Wheeler, P. Design of an expulsive de-icing system for avionic applications: Preliminary study and functional tests. In 2010 IEEE International Conference on Industrial Technology; IEEE: New York, NY, USA, 2010. [Google Scholar] [CrossRef]
- Zilio, C.; Patricelli, L. Aircraft anti-ice system: Evaluation of system performance with a new time dependent mathematical model. Appl. Therm. Eng. 2014, 63, 40–51. [Google Scholar] [CrossRef]
- Perron, E.; Louchez, P.; Laforte, J. Study of the shearing of the ground de-icing and anti-icing fluids. In Proceedings of the 33rd Aerospace Sciences Meeting and Exhibit, Reno, Nevada, 9–12 January 1995. [Google Scholar] [CrossRef]
- Chen, B.; Wang, L.W. Simulation and research of aircraft deicing fluids deicing process. Adv. Mater. Res. 2012, 121, 4695–4699. [Google Scholar] [CrossRef]
- Yao, X.; Falzon, B.G.; Hawkins, S.C.; Tsantzalis, S. Aligned carbon nanotube webs embedded in a composite laminate: A route towards a highly tunable electro-thermal system. Carbon 2018, 129, 486–494. [Google Scholar] [CrossRef]
- Yao, X.; Hawkins, S.C.; Falzon, B.G. An advanced anti-icing/de-icing system utilizing highly aligned carbon nanotube webs. Carbon 2018, 136, 130–138. [Google Scholar] [CrossRef]
- Yoon, Y.-H.; Song, J.-W.; Kim, D.; Kim, J.; Park, J.-K.; Oh, S.-K.; Han, C.-S. Transparent film heater using single-walled carbon nanotubes. Adv. Mater. 2007, 19, 4284–4287. [Google Scholar] [CrossRef]
- Wood, D.H. Design of Cowlings for Air-Cooled Aircraft Engines; SAE International: Warrendale, PA, USA, 1937; pp. 581–595. [Google Scholar] [CrossRef]
- AAA Support. Aluminum Factoids. Available online: https://www.aaaairsupport.com/alloy-identification/ (accessed on 5 March 2026).
- Chatterjee, B.; Bhowmik, S. Chapter 9 - Evolution of material selection in commercial aviation industry—A review. Sustain. Eng. Prod. Manuf. Technol. 2019, 199–219. [Google Scholar] [CrossRef]
- Yang, L.; Li, Y.; Huan, D.; Zhu, C. An anti-icing coating with superhydrophobic and photothermal properties for aircraft icing protection system. Colloids Surf. A Physicochem. Eng. Asp. 2025, 709, 136150. [Google Scholar] [CrossRef]
- Nguyen-Tri, P.; Tran, H.N.; Ouellet-Plamondon, C.M.; Tuduri, L.; Vo, D.-V.N.; Nanda, S.; Mishra, A.; Chao, H.-P.; Bajpai, A.K. Recent progress in the preparation, properties and applications of superhydrophobic nano-based coatings and surfaces: A review. Prog. Org. Coat. 2019, 132, 235–256. [Google Scholar] [CrossRef]
- Lin, Y.; Chen, H.; Wang, G.; Liu, A. Recent progress in preparation and anti-icing applications of superhydrophobic coatings. Coatings 2018, 8, 208. [Google Scholar] [CrossRef]
- Veronesi, F.; Boveri, G.; Mora, J.; Corozzi, A.; Raimondo, M. Icephobic properties of anti-wetting coatings for aeronautical applications. Surf. Coat. Technol. 2021, 421, 127363. [Google Scholar] [CrossRef]
- Villeneuve, E.; Brassard, J.-D.; Volat, C. Effect of various surface coatings on de-icing/anti-icing fluids aerodynamic and endurance time performances. Aerospace 2019, 6, 114. [Google Scholar] [CrossRef]
- Ferrari, M.; Cirisano, F. Superhydrophobic coating solutions for deicing control in aircraft. Appl. Sci. 2023, 13, 11684. [Google Scholar] [CrossRef]
- Zeng, D.; Li, Y.; Liu, H.; Yang, Y.; Peng, L.; Zhu, C.; Zhao, N. Superhydrophobic coating induced anti-icing and deicing characteristics of an airfoil. Colloids Surf. A Physicochem. Eng. Asp. 2023, 660, 130824. [Google Scholar] [CrossRef]
- Huang, J.; Peng, Z.; Zhang, B.; Yao, Y.; Chen, S. A flexible and high-efficient anti-icing/deicing coating based on carbon nanomaterials. ACS Appl. Mater. Interfaces 2024, 16, 44210–44224. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Zhuo, Y.; He, J.; Zhang, Z. Design and preparation of sandwich-like PDMS sponges with super-low ice adhesion. Soft Matter 2018, 14, 4846–4851. [Google Scholar] [CrossRef]
- Memon, H.; Wang, J.; Hou, X. Interdependence of surface roughness on icephobic performance: A review. Materials 2023, 16, 4607. [Google Scholar] [CrossRef] [PubMed]
- Flemming, R. Considerations on the Use of Hydrophobic, Superhydrophobic or Icephobic Coatings as a Part of the Aircraft Ice Protection System (2013-01-2108); SAE Technical Paper; SAE International: Warrendale, PA, USA, 2015. [Google Scholar]
- Kumar, R.; Bhowmick, H.; Sharma, B. Recent Trends in Icephobic Polymer Coatings for Aerospace Applications in Cold Climates. Polym. Adv. Technol. 2025, 36, e70211. [Google Scholar] [CrossRef]
- Kanti, P.K.; Prashantha Kumar, H.G.; Wanatasanappan, V.V.; Kumar, A.; Regasa, M.B. Graphene’s frontier in aerospace: Current applications, challenges, and future directions for space engineering. Nanoscale Adv. 2025, 7, 3603–3618. [Google Scholar] [CrossRef] [PubMed]
- Alloys, W.A. International Alloy Designations and Chemical Composition Limits for Wrought Aluminum. 2015. Available online: https://www.aluminum.org/sites/default/files/2021-10/Teal%20Sheet.pdf (accessed on 5 March 2026).
- The Aluminum Association. Aluminum Standards and Data; Aluminum Association: Washington, DC, USA, 2003; Available online: https://f.machineryhost.com/53adaf494dc89ef7196d73636eb2451b/01596f1b8cb905a92af310e4adaac2b7/Alum%20Standards%20and%20Data-2003.pdf (accessed on 12 March 2026).
- Korzec, I.; Samborski, S.; Łusiak, T. A study on mechanical strength and failure of fabric reinforced polymer composites. Adv. Sci. Technol. Res. J. 2022, 16, 120–130. [Google Scholar] [CrossRef]
- Mucha, M.; Sterzyński, T.; Krzyżak, A. The effect of the heat treatment on the crosslinking of epoxy resin for aviation applications. Polimery 2020, 65, 776–783. [Google Scholar] [CrossRef]
- Aircraft Spruce & Specialty Co. Epoxy Laminating Systems Catalog. Available online: https://www.aircraftspruce.com/catalog/cmpages/mgsresin.php (accessed on 12 March 2026).
- Ohsako, F.; Yoshizawa, K. Molecular theory of adhesion of metal/epoxy resin interface. Kobunshi Ronbunshu 2011, 68, 72–80. [Google Scholar] [CrossRef]
- Edwards, R.S.; Coleman, K.S. Graphene synthesis: Relationship to applications. Nanoscale 2013, 5, 38–51. [Google Scholar] [CrossRef]
- Whitener, K.E., Jr.; Sheehan, P.E. Graphene synthesis. Diam. Relat. Mater. 2014, 46, 25–34. [Google Scholar] [CrossRef]
- Shams, S.S.; Zhang, R.; Zhu, J. Graphene synthesis: A review. Mater. Sci. 2015, 33, 566–578. [Google Scholar] [CrossRef]
- Mousavi, M.V.; Khoramishad, H. Toughening mechanisms and mechanical properties of graphene oxide/epoxy nanocomposites under tensile loading. Proc. Inst. Mech. Eng. Part L J. Mater. Des. Appl. 2025. (First Published). [Google Scholar] [CrossRef]
- Song, N.; Gao, Z.; Li, X. Tailoring nanocomposite interfaces with graphene to achieve high strength and toughness. Sci. Adv. 2020, 6, eaba7016. [Google Scholar] [CrossRef]
- Ba, H.; Truong-Phuoc, L.; Romero, T.; Sutter, C.; Nhut, J.-M.; Schlatter, G.; Giambastiani, G.; Pham-Huu, C. Lightweight, few-layer graphene composites with improved electro-thermal properties as efficient heating devices for de-icing applications. Carbon 2021, 182, 655–668. [Google Scholar] [CrossRef]
- Chen, L.; Zhang, Y.; Wu, Q. Effect of graphene coating on the heat transfer performance of a composite anti-/deicing component. Coatings 2017, 7, 158. [Google Scholar] [CrossRef]
- Bhushan, B.; Jung, Y.C. Natural and biomimetic artificial surfaces for superhydrophobicity, self-cleaning, low adhesion, and drag reduction. Prog. Mater. Sci. 2011, 56, 1–108. [Google Scholar] [CrossRef]
- Quéré, D. Wetting and roughness. Annu. Rev. Mater. Res. 2008, 38, 71–99. [Google Scholar] [CrossRef]
- Piscitelli, F.; Fanciullo, M.; Sarcinella, A.; Costantini, M.; Frigione, M. Icephobic properties of superhydrophobic coatings developed for aeronautical applications. Coatings 2025, 15, 621. [Google Scholar] [CrossRef]
- Sarshar, M.A.; Swarctz, C.; Hunter, S.; Simpson, J.; Choi, C.-H. Effects of contact angle hysteresis on ice adhesion and growth on superhydrophobic surfaces under dynamic flow conditions. Colloid Polym. Sci. 2013, 291, 427–435. [Google Scholar] [CrossRef]
- Huang, X.; Tepylo, N.; Pommier-Budinger, V.; Budinger, M.; Bonaccurso, E.; Villedieu, P.; Bennani, L. A survey of icephobic coatings and their potential use in a hybrid coating/active ice protection system for aerospace applications. Prog. Aerosp. Sci. 2019, 105, 74–97. [Google Scholar] [CrossRef]
- Wang, T.; Zheng, Y.; Raji, A.-R.O.; Li, Y.; Sikkema, W.K.A.; Tour, J.M. Passive anti-icing and active deicing films. ACS Appl. Mater. Interfaces 2016, 8, 14169–14173. [Google Scholar] [CrossRef]
- Farhadi, S.; Farzaneh, M.; Kulinich, S.A. Anti-icing performance of superhydrophobic surfaces. Appl. Surf. Sci. 2011, 257, 6264–6269. [Google Scholar] [CrossRef]
- Choi, B.G.; Park, H.S. Superhydrophobic graphene/nafion nanohybrid films with hierarchical roughness. J. Phys. Chem. C 2012, 116, 3207–3211. [Google Scholar] [CrossRef]
- Liu, Q.; Yang, Y.; Huang, M.; Zhou, Y.; Liu, Y.; Liang, X. Durability of a lubricant-infused electrospray silicon rubber surface as an anti-icing coating. Appl. Surf. Sci. 2015, 346, 68–76. [Google Scholar] [CrossRef]
- Emelyanenko, K.A.; Emelyanenko, A.M.; Boinovich, L.B. Water and ice adhesion to solid surfaces: Impact of temperature and surface wettability. Coatings 2020, 10, 648. [Google Scholar] [CrossRef]
- Wu, X.; Silberschmidt, V.V.; Hu, Z.-T.; Chen, Z. When superhydrophobic coatings are icephobic: Role of surface topology. Surf. Coat. Technol. 2019, 358, 207–214. [Google Scholar] [CrossRef]
- Hejazi, V.; Sobolev, K.; Nosonovsky, M. From superhydrophobicity to icephobicity: Forces and interaction analysis. Sci. Rep. 2013, 3, 2194. [Google Scholar] [CrossRef] [PubMed]
- Lu, F.; Botte, G.G. Ammonia generation via a graphene-coated nickel catalyst. Coatings 2017, 7, 72. [Google Scholar] [CrossRef]
- Zhu, X.; Zhang, Z.; Ge, B.; Men, X.; Zhou, X. Fabrication of a superhydrophobic carbon nanotube coating with good reusability and easy repairability. Colloids Surf. A Physicochem. Eng. Asp. 2014, 444, 252–256. [Google Scholar] [CrossRef]
- Buschhorn, S.T.; Kessler, S.S.; Lachmann, N.; Gavin, J.; Thomas, G.; Wardle, B.L. Electrothermal icing protection of aerosurfaces using conductive polymer nanocomposites. In Proceedings of the 54th AIAA/ASME/ASCE/AHS/ASC Structures, Structural Dynamics, and Materials Conference, Boston, MA, USA, 8–11 April 2013. [Google Scholar] [CrossRef]
- Wang, Z.-J.; Kwon, D.-J.; DeVries, K.L.; Park, J.-M. Frost formation and anti-icing performance of a hydrophobic coating on aluminum. Exp. Therm. Fluid Sci. 2015, 60, 132–137. [Google Scholar] [CrossRef]
- Nagappan, S.; Park, S.S.; Ha, C.-S. Recent advances in superhydrophobic nanomaterials and nanoscale systems. J. Nanosci. Nanotechnol. 2014, 14, 1441–1462. [Google Scholar] [CrossRef]
- Kausar, A.; Ahmad, I.; Eisa, M.H.; Maaza, M. Graphene nanocomposites in Space Sector—Fundamentals and advancements. C 2023, 9, 29. [Google Scholar] [CrossRef]
- Li, N.; Zhang, Y.; Xu, Y.; Li, J. Graphene/epoxy coating with radiation heat dissipation properties for spacecraft thermal management. Chem. Eng. J. 2025, 519, 165105. [Google Scholar] [CrossRef]
- Barletta, M.; Vesco, S.; Puopolo, M.; Tagliaferri, V. High performance composite coatings on plastics: UV-curable cycloaliphatic epoxy resins reinforced by graphene or graphene derivatives. Surf. Coat. Technol. 2015, 272, 322–336. [Google Scholar] [CrossRef]
- Pat, S.; Tanışlı, M.; Bakır, M.; Uşak, A.C. Mechanical and adhesive properties of graphene-coated thermoset and thermoplastic aircraft composite materials by physical vapor deposition technology. Int. J. Adhes. Adhes. 2025, 139, 103971. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, D.; Wei, X.; Zhong, S.; Wang, J. Enhanced tribological properties of polymer composite coating containing graphene at room and elevated temperatures. Coatings 2018, 8, 91. [Google Scholar] [CrossRef]
- Du, Y.; Zhang, Z.; Wang, D.; Zhang, L.; Cui, J.; Chen, Y.; Wu, M.; Kang, R.; Lu, Y.; Yu, J.; et al. Enhanced tribological properties of aligned graphene-epoxy composites. Friction 2022, 10, 854–865. [Google Scholar] [CrossRef]
- Kim, H.; Abdala, A.A.; Macosko, C.W. Graphene/polymer nanocomposites. Macromolecules 2010, 43, 6515–6530. [Google Scholar] [CrossRef]
- Rafiee, M.A.; Rafiee, J.; Wang, Z.; Song, H.; Yu, Z.-Z.; Koratkar, N. Enhanced mechanical properties of nanocomposites at low graphene content. ACS Nano 2009, 3, 3884–3890. [Google Scholar] [CrossRef] [PubMed]
- Thostenson, E.T.; Ren, Z.; Chou, T.-W. Advances in the science and technology of carbon nanotubes and their composites: A review. Compos. Sci. Technol. 2001, 61, 1899–1912. [Google Scholar] [CrossRef]
- Hussein, S.I.; Abd-Elnaiem, A.M.; Asafa, T.B.; Jaafar, H.I. Effect of incorporation of conductive fillers on mechanical properties and thermal conductivity of epoxy resin composite. Appl. Phys. A 2018, 124, 475. [Google Scholar] [CrossRef]
- Farcas, C.; Galao, O.; Vertuccio, L.; Guadagno, L.; Romero-Sánchez, M.D.; Rodríguez-Pastor, I.; Garcés, P. Ice-prevention and de-icing capacity of epoxy resin filled with hybrid carbon-nanostructured forms: Self-heating by joule effect. Nanomaterials 2021, 11, 2427. [Google Scholar] [CrossRef] [PubMed]
- Balandin, A.A.; Ghosh, S.; Bao, W.; Calizo, I.; Teweldebrhan, D.; Miao, F.; Lau, C.N. Superior thermal conductivity of single-layer graphene. Nano Lett. 2008, 8, 902–907. [Google Scholar] [CrossRef]
- Gao, H.; Zhou, Y.; Ma, J.; Jin, H.; Bao, J.; Wen, D. Recent advancements in electro-thermal anti-/de-icing materials. RSC Adv. 2025, 15, 17102–17115. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.; Joung, D.; Zhai, L.; Das, S.; Khondaker, S.I.; Seal, S. Graphene based materials: Past, present and future. Prog. Mater. Sci. 2011, 56, 1178–1271. [Google Scholar] [CrossRef]
- de Oliveira, E.M.; Sormani, M.B.; Hurtado, L.P.; Polkowski, R.D.O. Potential use of graphene composites in epoxy resin as anticorrosive painting in automotive industry. In SAE Brasil 2023 Congress; SAE Technical Paper; SAE International: Warrendale, PA, USA, 2024. [Google Scholar] [CrossRef]



| Category | Property |
|---|---|
| Composition (wt.%) | Al (96.7–99), Si (0.6), Fe (0.7), Cu (0.05–0.20), Mn (1.0–1.5), Zn (0.10), Others (Each: 0.05, Total: 0.15) |
| Mechanical Properties | Tensile Strength: 150 MPa at 25 °C |
| Yield Strength: 0.2% Proof min: 145 MPa at 25 °C | |
| Hardness Brinell: 40 | |
| Physical Properties | Density: 2730 kg/m3 |
| Elastic Modulus: 69 GPa | |
| Coefficient of Thermal Expansion: 20–100 °C: 23.2 µm/m/°C | |
| Thermal Conductivity at 25 °C: 193 W/m·K |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Almomani, A.; Alkhasawneh, M.; Almomani, M.A.; Bani-Hani, M.A. Preliminary Assessment of Thermal and Mechanical Properties of a Graphene-Rich Carbon Coating on 3003-H14 Aluminum Alloy for Potential Anti-Icing Applications. Materials 2026, 19, 1150. https://doi.org/10.3390/ma19061150
Almomani A, Alkhasawneh M, Almomani MA, Bani-Hani MA. Preliminary Assessment of Thermal and Mechanical Properties of a Graphene-Rich Carbon Coating on 3003-H14 Aluminum Alloy for Potential Anti-Icing Applications. Materials. 2026; 19(6):1150. https://doi.org/10.3390/ma19061150
Chicago/Turabian StyleAlmomani, Abdallah, Mu’nis Alkhasawneh, Mohammed A. Almomani, and Muath A. Bani-Hani. 2026. "Preliminary Assessment of Thermal and Mechanical Properties of a Graphene-Rich Carbon Coating on 3003-H14 Aluminum Alloy for Potential Anti-Icing Applications" Materials 19, no. 6: 1150. https://doi.org/10.3390/ma19061150
APA StyleAlmomani, A., Alkhasawneh, M., Almomani, M. A., & Bani-Hani, M. A. (2026). Preliminary Assessment of Thermal and Mechanical Properties of a Graphene-Rich Carbon Coating on 3003-H14 Aluminum Alloy for Potential Anti-Icing Applications. Materials, 19(6), 1150. https://doi.org/10.3390/ma19061150









