Potential of Wollastonite-Based Brushite Cement for the Conditioning of Radioactive Waste Contaminated by 90Sr
Abstract
1. Introduction
2. Experimental
2.1. Material and Sample Preparation
2.2. Irradiation Experiments
2.3. Leaching Experiments
2.4. Solid Phase Characterization
2.4.1. Hydration Stoppage
2.4.2. X-Ray Diffraction
2.4.3. Raman Spectroscopy
2.4.4. Thermogravimetry
2.4.5. X-Ray Fluorescence
2.4.6. Scanning Electron Microscopy
2.4.7. Porosity
3. Results and Discussion
3.1. Characterization of Pristine WBC Paste Samples
3.2. Gamma Irradiation of WBC-C Paste
3.2.1. Hydrogen Gas Production
| Cement Hydrate | Dose | G(H2)material × 10−7 (mol·J−1) | G(H2)water × 10−7 (mol·J−1) | Ref. |
|---|---|---|---|---|
| Brushite CaHPO4·2H2O | 250 kGy up to 5 MGy | 0.070 ± 0.007 | 0.26 ± 0.02 | This work |
| Gibbsite Al(OH)3 | 200 kGy | 0.009 ± 0.005 | 0.027 ± 0.003 | [30] |
| Katoite Ca3Al2(OH)12 | 200 kGy | 0.003 ± 0.002 | 0.011 ± 0.001 | |
| Calcium monocarboaluminate hydrate Ca4Al2(CO3)(OH)12·5H2O | 200 kGy | 0.12 ± 0.01 | 0.38 ± 0.04 | |
| Portlandite Ca(OH)2 | 0.21 | [56] | ||
| 200 kGy | 0.042 ± 0.004 | 0.19 ± 0.03 | [57] | |
| 150 kGy | 0.081 ± 0.005 | 0.33 ± 0.02 | [35] | |
| Brucite Mg(OH)2 | 200 kGy | 0.055 ± 0.006 | 0.18 ± 0.03 | [57] |
| 0.053 | [56] | |||
| Calcium silicate hydrate (C-S-H) with CaO/SiO2 of: | 100 and 200 kGy | [55] | ||
| 0.80 | 0.61 ± 0.06 | 3.23 ± 0.32 | ||
| 0.97 | 0.58 ± 0.06 | 3.11 ± 0.31 | ||
| 1.14 | 0.49 ± 0.05 | 2.85 ± 0.29 | ||
| 1.30 | 0.42 ± 0.04 | 2.44 ± 0.24 | ||
| 1.40 | 0.36 ± 0.04 | 2.13 ± 0.21 | ||
| Bulk water (pH 13) | 0.44 ± 0.04 | [54] |
3.2.2. Mineralogy of Irradiated Materials
3.3. Leaching of Sr-Doped WBC Pastes
3.3.1. Characterization of the Leachates
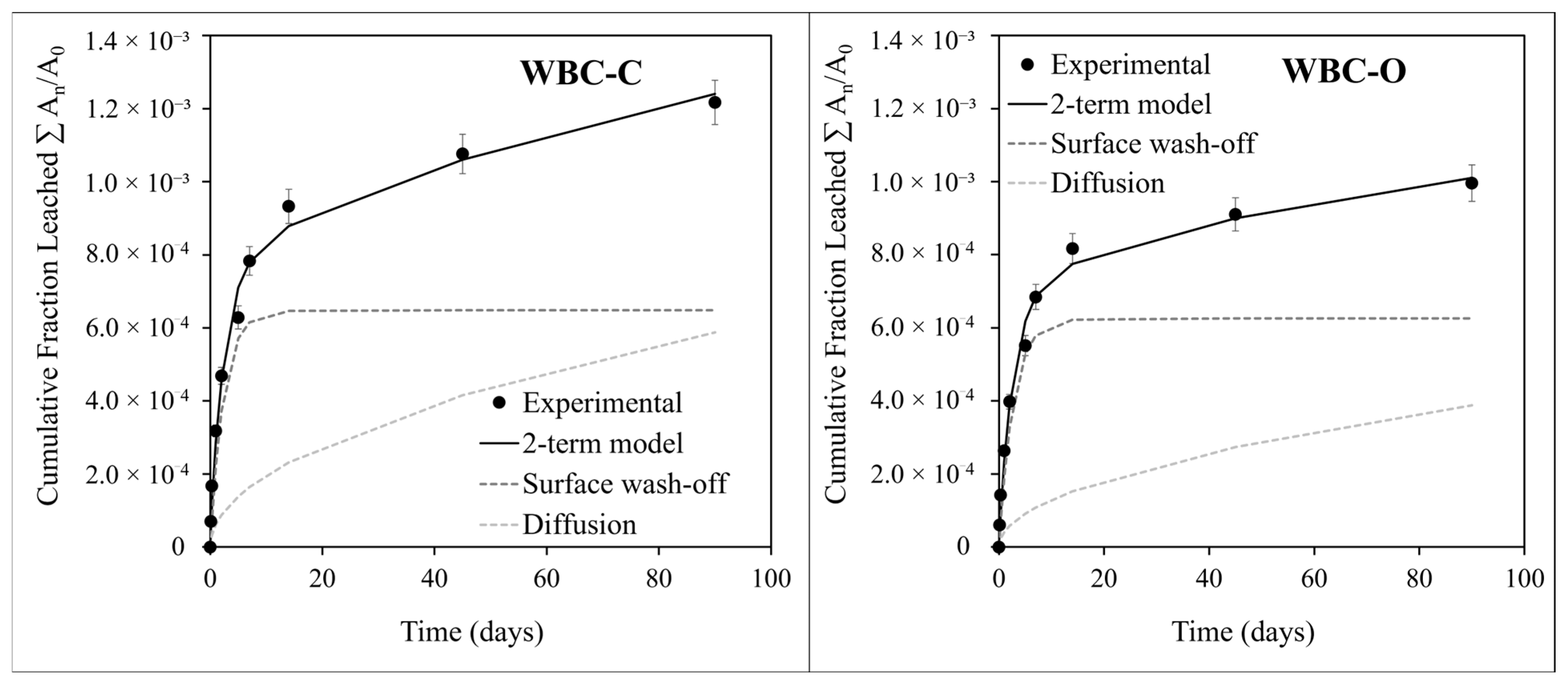
3.3.2. Modeling the Leaching of Strontium from WBC Pastes: Evaluation of the Diffusion Coefficient and Leachability Index
- Time-invariant chemical environment: The leaching solution was periodically renewed to prevent accumulation of released species (the Ca, P, and Si concentrations remained below 1 mmol·L−1 (Ca) and 2 mmol·L−1 (P, Si), whereas the Sr concentration did not exceed 0.3 µmol/L between two renewals), and the pH remained relatively stable throughout the test (comprised between 6.5 and 7.5).
- Semi-infinite solid geometry: As defined by the ANS/ANSI 16.1 specification, the semi-infinite assumption is valid if the cumulative fraction leached is less than 20%. In this study, the cumulative fraction of strontium leached remained well below this threshold (~0.12% for WBC-C and 0.10% for WBC-O at the end of the test).
- Zero surface concentration of strontium: The Sr concentrations in the leachates were consistently low (<20 ppb), and the frequency of leachant renewal was high enough to support the boundary condition of zero surface concentration.
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Barré, Y.; Schrive, L.; Lepeytre, C.; Hertz, A.; Grandjean, A.; Blet, V. Decontamination of aqueous effluents. In Clean-Up and Dismantling of Nuclear Facilities; Lecomte, M., Ed.; Editions du Moniteur: Paris, France, 2018; pp. 99–106. [Google Scholar]
- Ojovan, M.I.; Lee, W.E.; Kalmykov, S.N. Immobilisation of radioactive waste in cement. In An Introduction to Nuclear Waste Immobilization, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 271–303. [Google Scholar] [CrossRef]
- Atkins, M.; Glasser, F.P. Application of Portland cement-based materials to radioactive waste immobilization. Waste Manag. 1992, 12, 105–131. [Google Scholar] [CrossRef]
- Milestone, N.B. Reactions in cement encapsulated nuclear wastes: Need for toolbox of different cement types. Adv. Appl. Ceram. 2006, 105, 13–20. [Google Scholar] [CrossRef]
- Evans, N.D.M. Binding mechanisms of radionuclides to cement. Cem. Concr. Res. 2008, 38, 543–553. [Google Scholar] [CrossRef]
- Tits, J.; Wieland, E.; Müller, C.J.; Landesman, C.; Bradbury, M.H. Strontium binding by calcium silicate hydrates. J. Colloid Interface Sci. 2006, 300, 78–87. [Google Scholar] [CrossRef]
- Wang, L.; Ochs, M.; Mallants, D.; Vielle-Petit, L.; Martens, E.; Jacques, D.; De Cannière, P.; Berry, J.A.; Leterme, B. A new radionuclide sorption database for benchmark cement accounting for geochemical evolution of cement. In Cement-Based Materials for Nuclear Waste Storage; Bart, F., Cau Dit Coumes, C., Frizon, F., Lorente, S., Eds.; Springer: Berlin/Heidelberg, Germany, 2013. [Google Scholar] [CrossRef]
- Stamoulis, K.C.; Assimakopoulos, P.A.; Ioannides, K.G.; Johnson, E.; Soucacos, P.N. Strontium-90 concentration measurements in human bones and teeth in Greece. Sci. Total Environ. 1999, 229, 165–182. [Google Scholar] [CrossRef]
- Nielsen, S.P. The biological role of strontium. Bone 2004, 35, 583–588. [Google Scholar] [CrossRef] [PubMed]
- Ohura, K.; Bohner, M.; Hardouin, P.; Lemaître, J.; Pasquier, G.; Flautre, B. Resorption of, and bone formation from, new beta-tricalcium phosphate-monocalcium phosphate cements: An in vivo study. J. Biomed. Mater. Res. 1996, 30, 193–200. [Google Scholar] [CrossRef]
- Constantz, B.R.; Barr, B.M.; Ison, I.C.; Fulmer, M.T.; Baker, J.; McKinney, L.A.; Goodman, S.B.; Gunasekaren, S.; Delaney, D.C.; Ross, J.; et al. Histological, chemical, and crystallographic analysis of four calcium phosphate cements in different rabbit osseous sites. J. Biomed. Mater. Res. 1998, 43, 451–461. [Google Scholar] [CrossRef]
- Grover, L.M.; Knowles, J.C.; Fleming, G.J.P.; Barralet, J.E. In vitro ageing of brushite calcium phosphate cement. Biomaterials 2003, 24, 4133–4141. [Google Scholar] [CrossRef]
- Grover, L.M.; Gbureck, U.; Wright, A.J.; Tremayne, M.; Barralet, J.E. Biologically mediated resorption of brushite cement in vitro. Biomaterials 2006, 27, 2178–2185. [Google Scholar] [CrossRef]
- Gorst, N.J.S.; Perrie, Y.; Gbureck, U.; Hutton, A.L.; Hofmann, M.P.; Grover, L.M.; Barralet, J.E. Effects of fibre reinforcement on the mechanical properties of brushite cement. Acta Biomater. 2006, 2, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Huan, Z.; Chang, J. Novel bioactive composite bone cements based on the β-tricalcium phosphate-monocalcium phosphate monohydrate composite cement system. Acta Biomater. 2009, 5, 1253–1264. [Google Scholar] [CrossRef]
- Aberg, J.; Brisby, H.; Henriksson, H.B.; Lindahl, A.; Thomsen, P.; Engqvist, H. Premixed acidic calcium phosphate cement: Characterization of strength and microstructure. J. Biomed. Mater. Res.—Part B Appl. Biomater. 2010, 93, 436–441. [Google Scholar] [CrossRef]
- Laniesse, P.; Cau Dit Coumes, C.; Poulesquen, A.; Bourchy, A.; Mesbah, A.; Le Saout, G.; Gaveau, P. Setting and hardening process of a wollastonite-based brushite cement. Cem. Concr. Res. 2018, 106, 65–76. [Google Scholar] [CrossRef]
- Laniesse, P.; Cau Dit Coumes, C.; Le Saout, G.; Mesbah, A. Understanding the setting and hardening process of wollastonite-based brushite cement. Part 2: Influence of the boron and aluminum concentrations in the mixing solution. Cem. Concr. Res. 2021, 140, 106288. [Google Scholar] [CrossRef]
- Semler, C.E. Quick-setting wollastonite phosphate cement. Am. Ceram. Soc. Bull. 1976, 55, 983–988. [Google Scholar]
- Alkhraisat, M.H.; Rueda, C.; López Cabarcos, E. Strontium ions substitution in brushite crystals: The role of strontium chloride. J. Funct. Biomater. 2011, 2, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Pina, S.; Vieira, S.I.; Rego, P.; Torres, P.M.C.; da Cruz e Silva, O.A.B.; da Cruz e Silva, E.F.; Ferreira, J.M.F. Biological responses of brushite-forming Zn-and ZnSr-substituted beta-tricalcium phosphate bone cements. Eur. Cells Mater. 2010, 20, 162–177. [Google Scholar] [CrossRef]
- Boanini, E.; Gazzano, M.; Bigi, A. Ionic substitutions in calcium phosphates synthesized at low temperature. Acta Biomater. 2010, 6, 1882–1894. [Google Scholar] [CrossRef]
- Hurle, K.; Oliveira, J.M.; Reis, R.L.; Pina, S.; Goetz-Neunhoeffer, F. Ion-doped brushite cements for bone regeneration. Acta Biomater. 2021, 123, 51–71. [Google Scholar] [CrossRef]
- Jdaini, J.; Cau Dit Coumes, C.; Barré, Y.; de Noirfontaine, M.-N.; Courtial, M.; Garcia-Caurel, E.; Dunstetter, F.; Gorse-Pomonti, D. Strontium release from wollastonite-based brushite cement paste under semi-dynamic leaching conditions. In Proceedings of the NUWCEM 2022 (International Symposium on Cement-Based Materials for Nuclear Wastes), Avignon, France, 4–6 May 2022. [Google Scholar]
- ANSI/ANS-16. 1-2003 (R2008); Measurement of the Leachability of Solidified Low-Level Radioactive Wastes by a Short-Term Test Procedure. American Nuclear Society: Westmont, IL, USA, 2003.
- Bibler, N.E. Radiolytic Gas Production From Concrete Containing Savannah River Plant Waste; Report DP-1464; Savanah River Laboratory: Aiken, SC, USA, 1978. [Google Scholar] [CrossRef][Green Version]
- Bouniol, P.; Bjergbakke, E. A comprehensive model to describe radiolytic processes in cement medium. J. Nucl. Mater. 2008, 372, 1–15. [Google Scholar] [CrossRef]
- Rosseel, T.M.; Maruyama, I.; Le Pape, Y.; Kontani, O.; Giorla, A.B.; Remec, I.; Wall, J.J.; Sircar, M.; Andrade, C.; Ordonez, M. Review of the current state of knowledge on the effects of radiation on concrete. J. Adv. Concr. Technol. 2016, 14, 368–383. [Google Scholar] [CrossRef]
- Chartier, D.; Sanchez-Canet, J.; Bessette, L.; Esnouf, S.; Renault, J.-P. Influence of formulation parameters of cement based materials towards gas production under gamma irradiation. J. Nucl. Mater. 2018, 511, 183–190. [Google Scholar] [CrossRef]
- Acher, L.; de Noirfontaine, M.-N.; Chartier, D.; Gorse-Pomonti, D.; Courtial, M.; Tusseau-Nenez, S.; Cavani, O.; Haas, J.; Dannoux-Papin, A.; Dunstetter, F. H2 production under gamma irradiation of a calcium aluminate cement: An experimental study on both cement pastes and its stable hydrates. Radiat. Phys. Chem. 2021, 189, 109689. [Google Scholar] [CrossRef]
- Kertesz, C.J.; Chenavas, P.R.; Auffret, L. Conditionnement de Cendres D’incinérateur Alpha et Bêta Gamma, Obtenues par Incinération des Déchets Radioactifs, par Enrobage, dans Différentes Matrices; Report EUR 14365 FR; Sciences et Techniques Nucléaires; Commission Européenne: Luxembourg, 1993. (In French) [Google Scholar]
- Madic, C.; Koehly, G. Comportement à Long Terme Des Matrices D’enrobage Contaminées en Émetteurs Alpha; Etude du phénomène de rupture d’éprouvettes constituées de liants hydrauliques contaminés en plutonium 238 et curium 244 au cours de leur lixiviation, DGR, Report C.E.A; Central Electricity Authority: New Delhi, India, 1986; Volume 134. (In French) [Google Scholar]
- de Noirfontaine, M.-N.; Garcia-Caurel, E.; Funes-Hernando, D.; Courtial, M.; Tusseau-Nenez, S.; Cavani, O.; Jdaini, J.; Cau Dit Coumes, C.; Dunstetter, F.; Gorse-Pomonti, D. Amorphization of a proposed sorbent of strontium, brushite, CaHPO4·2H2O, studied by X-ray diffraction and Raman spectroscopy. J. Nucl. Mater. 2021, 545, 152751. [Google Scholar] [CrossRef]
- Chartier, D.; Sanchez-Canet, J.; Antonucci, P.; Esnouf, S.; Renault, J.-P.; Farcy, O.; Lambertin, D.; Parraud, S.; Lamotte, H.; Cau Dit Coumes, C. Behaviour of magnesium phosphate cement-based materials under gamma and alpha irradiation. J. Nucl. Mater. 2020, 541, 152411. [Google Scholar] [CrossRef]
- Herin, T.; Charpentier, T.; Bouniol, P.; Le Caër, S. Behavior of portlandite upon exposure to ionizing radiation: Evidence of delayed H2 production. J. Phys. Chem. C 2023, 127, 20245–20254. [Google Scholar] [CrossRef]
- Frizon, F.; Galle, C. Experimental investigations of diffusive and convective transport of inert gas through cement pastes. J. Porous Media 2009, 12, 221–237. [Google Scholar] [CrossRef]
- Snellings, R.; Chwast, J.; Cizer, Ö.; De Belie, N.; Dhandapani, Y.; Durdzinski, P.; Elsen, J.; Haufe, J.; Hooton, D.; Patapy, C.; et al. RILEM TC-238 SCM recommendation on hydration stoppage by solvent exchange for the study of hydrate assemblages. Mater. Struct. 2018, 51, 172. [Google Scholar] [CrossRef]
- Frost, R.L.; Palmer, S.J. Thermal stability of the ‘cave’ mineral brushite CaHPO4·2H2O—Mechanism of formation and decomposition. Thermochim. Acta 2011, 521, 14–17. [Google Scholar] [CrossRef]
- Angel, R.J. Structural variation in wollastonite and bustamite. Mineral. Mag. 1985, 49, 37–48. [Google Scholar] [CrossRef]
- Frontera, C.; Rodríguez-Carvajal, J. FullProf as a new tool for flipping ratio analysis. Phys. B Condens. Matter 2003, 335, 219–222. [Google Scholar] [CrossRef]
- Schofield, P.F.; Knight, K.S.; van der Houwen, J.A.M.; Valsami-Jones, E. The role of hydrogen bonding in the thermal expansion and dehydration of brushite, di-calcium phosphate dihydrate. Phys. Chem. Miner. 2004, 31, 606–624. [Google Scholar] [CrossRef]
- Ohashi, Y. Polysynthetically-twinned structures of enstatite and wollastonite. Phys. Chem. Miner. 1984, 10, 217–229. [Google Scholar] [CrossRef]
- Antao, S.M.; Hassan, I.; Wang, J.; Lee, P.L.; Toby, B.H. State-of-the-art high-resolution powder X-ray diffraction (HRPXRD) illustrated with Rietveld structure refinement of quartz, sodalite, tremolite, and meionite. Can. Mineral. 2008, 46, 1501–1509. [Google Scholar] [CrossRef]
- Dollase, W.A. Correction of intensities of preferred orientation in powder diffractometry: Application of the March model. J. Appl. Crystallogr. 1986, 19, 267–272. [Google Scholar] [CrossRef]
- Landín, M.; Rowe, R.C.; York, P. Structural changes during the dehydration of dicalcium phosphate dihydrate. Eur. J. Pharm. Sci. 1994, 2, 245–252. [Google Scholar] [CrossRef]
- ISO 15901-1:2005; Pore Size Distribution and Porosity of Solid Materials by Mercury Porosimetry and Gas Adsorption—Part 1: Mercury Porosimetry. International Organization for Standardization: Geneva, Switzerland, 2005.
- Laniesse, P. Wollastonite-Based Brushite Cements: Reactivity, Properties, and Application to the Treatment Of Strontium-Contaminated Aqueous Wastestreams. Ph.D. Thesis, Montpellier University, Montpellier, France, 2019. (In French) [Google Scholar]
- Krauskopf, K.B. Dissolution and precipitation of silica at low temperatures. Geochim. Cosmochim. Acta 1956, 10, 1–26. [Google Scholar] [CrossRef]
- Jdaini, J. Potential of Brushite Cement for Treating and Conditioning Radioactive Waste Contaminated with Strontium. Ph.D. Thesis, Montpellier University, Montpellier, France, 2022. (In French) [Google Scholar]
- Hossain, S.K.S.; Mathur, L.; Bhardwaj, A.; Roy, P.K. A facile route for the preparation of silica foams using rice husk ash. Int. J. Appl. Ceram. Technol. 2019, 16, 1069–1077. [Google Scholar] [CrossRef]
- Weissbart, E.J.; Rimstidt, J.D. Wollastonite: Incongruent dissolution and leached layer formation. Geochim. Cosmochim. Acta 2000, 64, 4007–4016. [Google Scholar] [CrossRef]
- Chupin, F. Characterisation of Irradiation Effects on Geopolymers. Ph.D. Thesis, Pierre and Marie Curie University, Paris, France, 2015. (In French) [Google Scholar]
- Möckel, H.J.; Köster, R.H. Gas formation during the gamma radiolysis of cemented low- and intermediate-level waste products. Nucl. Technol. 1982, 59, 494–497. [Google Scholar] [CrossRef]
- Pastina, B.; LaVerne, J.A.; Pimblott, S.M. Dependence of molecular hydrogen formation in water on scavengers of the precursor to the hydrated electron. J. Phys. Chem. A 1999, 103, 5841–5846. [Google Scholar] [CrossRef]
- Yin, C.; Dannoux-Papin, A.; Haas, J.; Renault, J.-P. Influence of calcium to silica ratio on H2 gas production in calcium silicate hydrate. Radiat. Phys. Chem. 2019, 162, 66–71. [Google Scholar] [CrossRef]
- LaVerne, J.A.; Tandon, L. H2 and Cl2 production in the radiolysis of calcium and magnesium chlorides and hydroxides. J. Phys. Chem. A 2005, 109, 2861–2865. [Google Scholar] [CrossRef]
- Acher, L. Investigation of the Behaviour of Cement Matrices and Their Hydrates Under γ and Electron Irradiation. Ph.D. Thesis, Paris Saclay University, Orsay, France, 2017. (In French) [Google Scholar]
- de Noirfontaine, M.-N.; Courtial, M.; Alessi, A.; Tusseau-Nenez, S.; Garcia-Caurel, E.; Cavani, O.; Coumes, C.C.D.; Gorse-Pomonti, D. Stability under electron irradiation of some layered hydrated minerals. J. Solid State Chem. 2024, 340, 125033. [Google Scholar] [CrossRef]
- Frost, R.L.; Xi, Y.; Pogson, R.E.; Millar, G.J.; Tan, K.; Palmer, S.J. Raman spectroscopy of synthetic CaHPO4·2H2O− and in comparison, with the cave mineral brushite. J. Raman Spectrosc. 2012, 43, 571–576. [Google Scholar] [CrossRef]
- Buzatu, M.-N.A.; Buzgar, N. The Raman study of single-chain silicates. Analele Științifice Ale Univ. “Al. I. Cuza” Iași. Geol. 2010, 56, 107–125. [Google Scholar]
- Penel, G.; Leroy, N.; Van Landuyt, P.; Flautre, B.; Hardouin, P.; Lemaître, J.; Leroy, G. Raman microspectrometry studies of brushite cement: In vivo evolution in a sheep model. Bone 1999, 25, 81S–84S. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Butler, I.S.; Gilson, F.R. FT-Raman and high-pressure infrared spectroscopic studies of dicalcium phosphate dihydrate (CaHPO4·2H2O) and anhydrous dicalcium phosphate (CaHPO4). Spectrochim. Acta—Part A Mol. Biomol. Spectrosc. 1999, 55, 2801–2809. [Google Scholar] [CrossRef]
- Van der Lee, J. Thermodynamic and Mathematical Concepts of CHESS; Technical Report LHM/RD/98/39; Ecole des Mines de Paris: Fontainebleau, France, 1998; 99p. [Google Scholar]
- Jang, J.G.; Park, S.M.; Lee, H.K. Physical barrier effect of geopolymeric waste form on diffusivity of cesium and strontium. J. Hazard. Mater. 2016, 318, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Côtê, P.L.; Constable, T.W.; Moreira, A. An evaluation of cement-based waste forms using the results of approximately two years of dynamic leaching. Nucl. Chem. Waste Manag. 1987, 7, 129–139. [Google Scholar] [CrossRef]
- Pescatore, C.; Machiels, A.J. Effect of surfaces on glass waste form leaching. J. Non-Cryst. Solids 1982, 49, 379–388. [Google Scholar] [CrossRef]
- Irisawa, K.; Namiki, M.; Taniguchi, T.; Garcia-Lodeiro, I.; Kinoshita, H. Solidification and stabilization of strontium and chloride ions in thermally treated calcium aluminate cement modified with or without sodium polyphosphate. Cem. Concr. Res. 2022, 156, 106758. [Google Scholar] [CrossRef]
- Goo, J.-Y.; Kim, B.-J.; Kang, M.; Jeong, J.; Jo, H.Y.; Kwon, J.-S. Leaching behavior of cesium, strontium, cobalt, and europium from immobilized cement matrix. Appl. Sci. 2021, 11, 8418. [Google Scholar] [CrossRef]
- El-Kamash, A.M.; El-Naggar, M.R.; El-Dessouky, M.I. Immobilization of cesium and strontium radionuclides in zeolite-cement blends. J. Hazard. Mater. 2006, 136, 310–316. [Google Scholar] [CrossRef] [PubMed]
- Matsuzuru, H.; Ito, A. Leaching behaviour of strontium-90 in cement composites. Ann. Nucl. Energy 1977, 4, 465–470. [Google Scholar] [CrossRef]
- Pyo, J.-Y.; Um, W.; Heo, J. Magnesium potassium phosphate cements to immobilize radioactive concrete wastes generated by decommissioning of nuclear power plants. Nucl. Eng. Technol. 2021, 53, 2261–2267. [Google Scholar] [CrossRef]

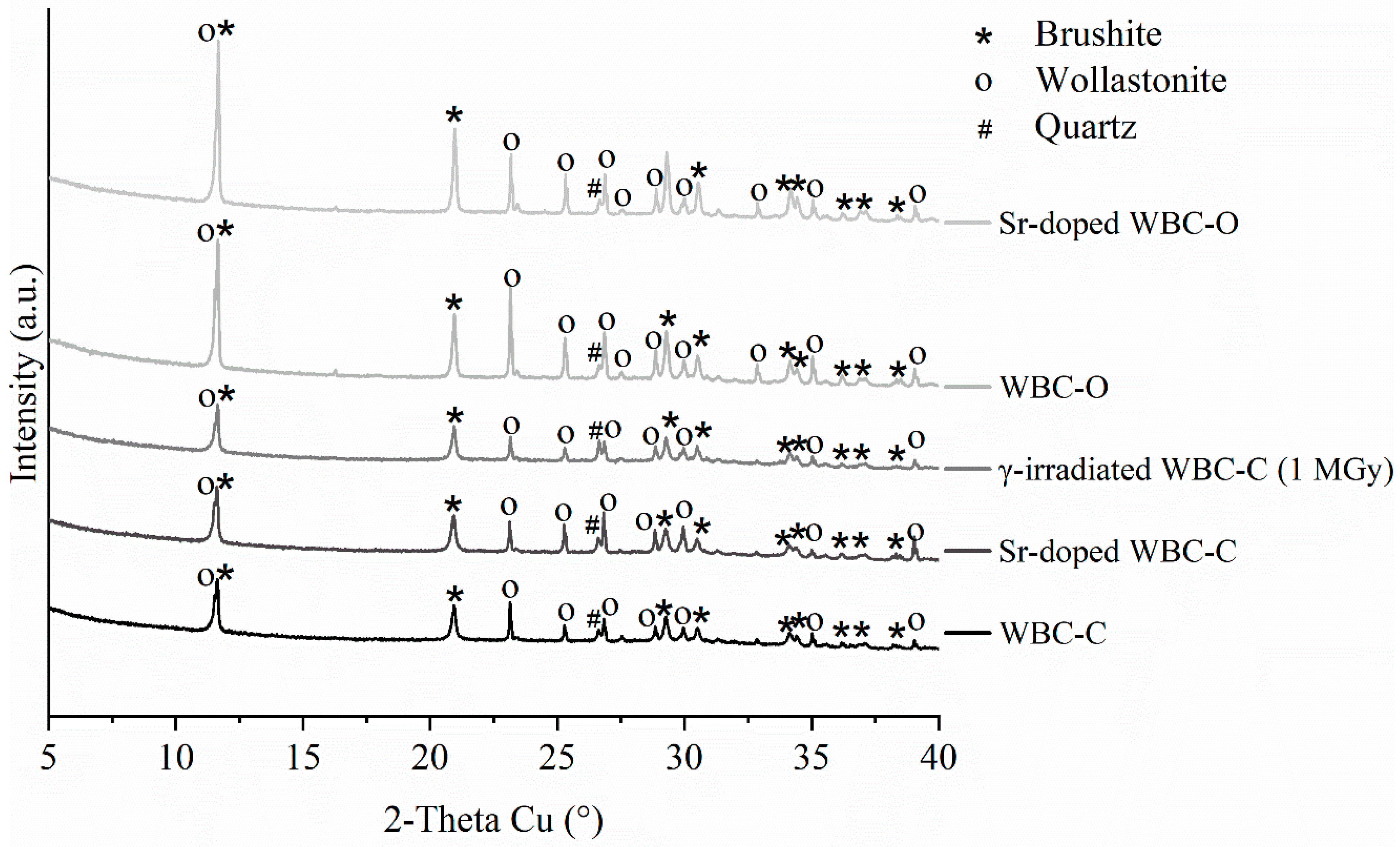
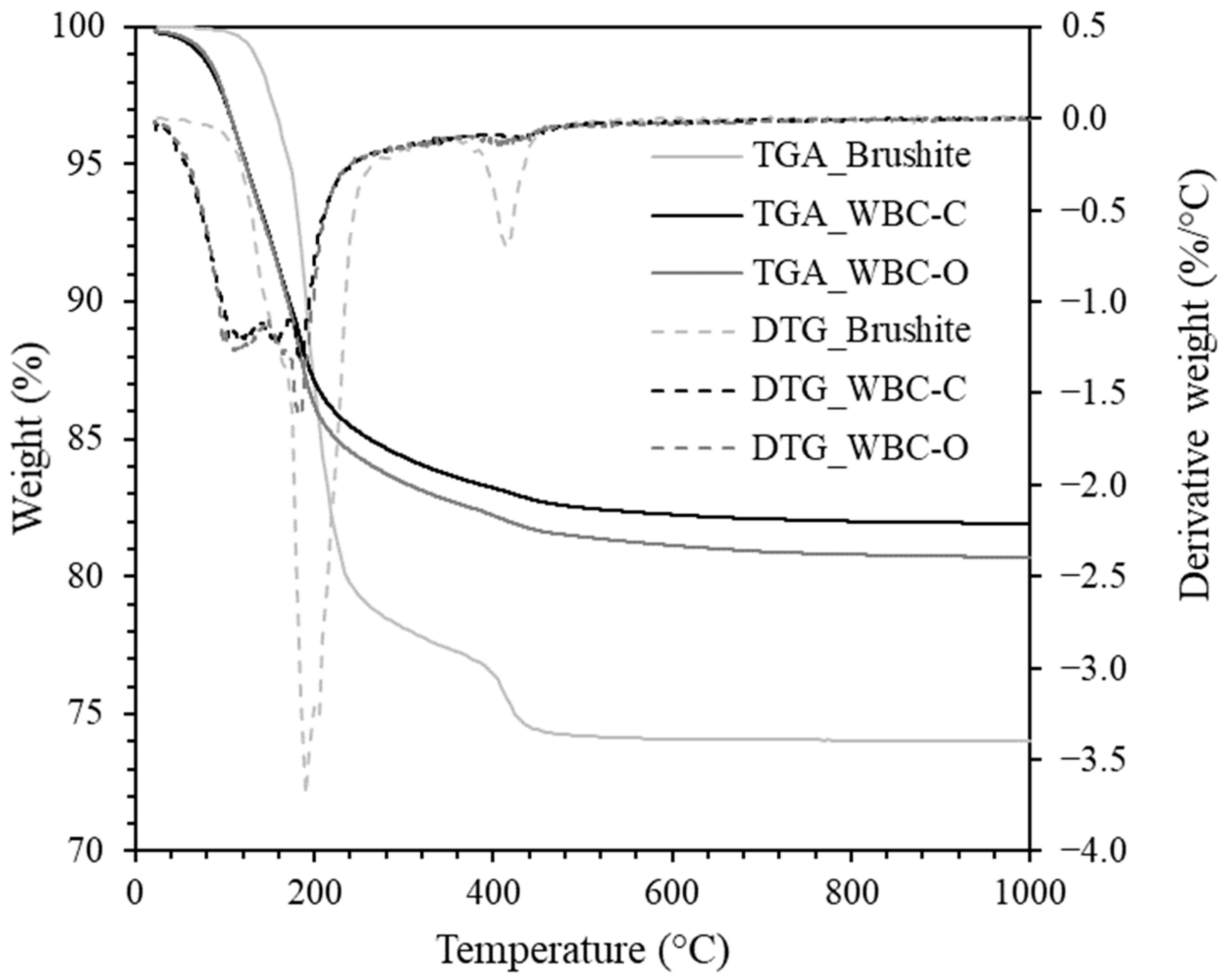
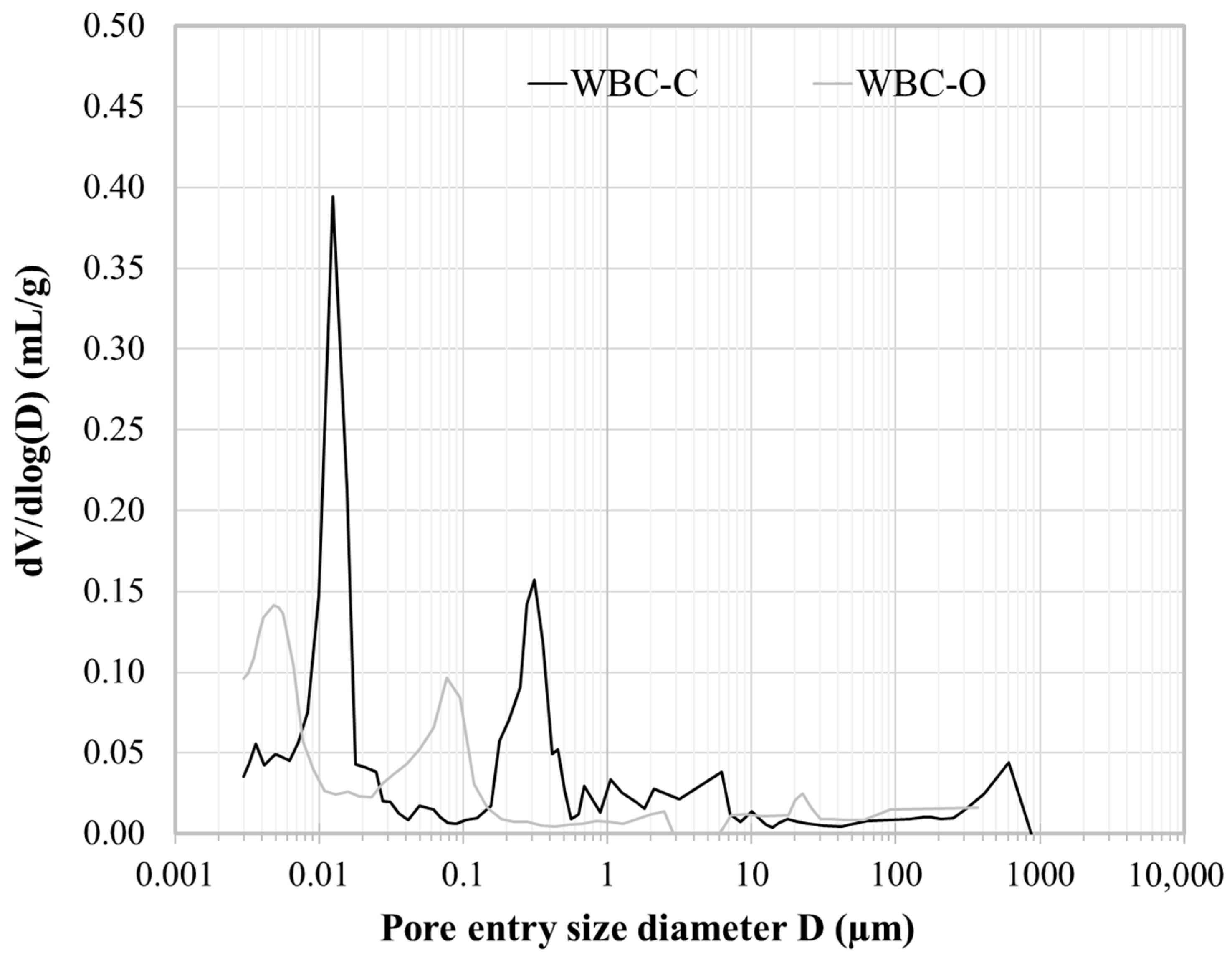






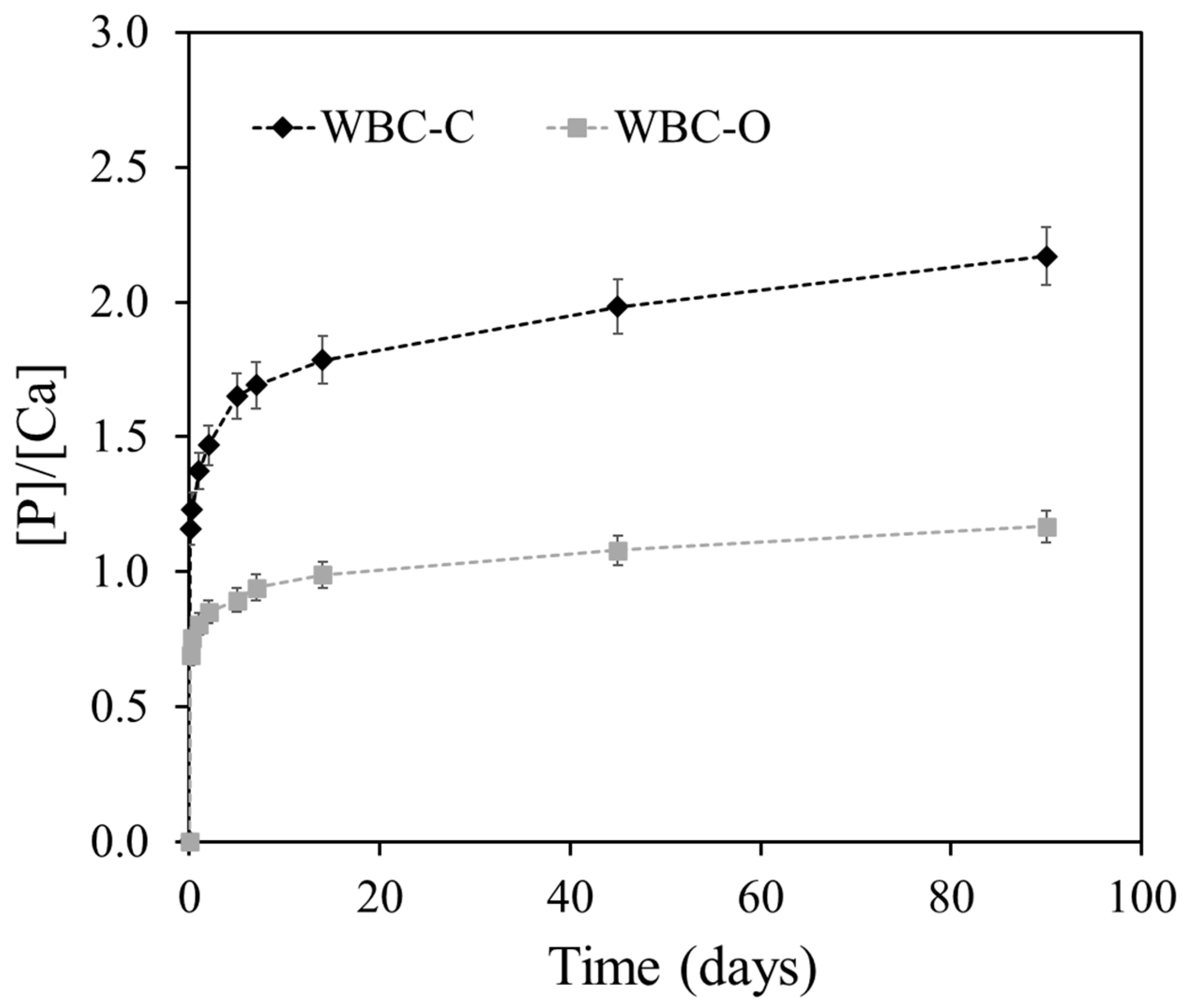

| Sample Code | Composition of the Mixing Solution | [Sr2+] in the Mixing Solution (mg·L−1) | Mixing Solution (g) | Wollastonite (g) |
|---|---|---|---|---|
| WBC-C | [H3PO4] = 9.3 mol·L−1 [Al3+] = 1.6 mol·L−1 [Zn2+] = 1.5 mol·L−1 [B] = 0.6 mol·L−1 | - | 62.5 | 50 |
| WBC-O | [H3PO4] = 9 mol·L−1 [Al3+] = 2.5 mol·L−1 [B] = 0.2 mol·L−1 | - | 62.5 | 50 |
| Sr-doped WBC-C | [H3PO4] = 9.3 mol·L−1 [Al3+] = 1.6 mol·L−1 [Zn2+] = 1.5 mol·L−1 [B] = 0.6 mol·L−1 | 1000 | 62.5 | 50 |
| Sr-doped WBC-O | [H3PO4] = 9 mol·L−1 [Al3+] = 2.5 mol·L−1 [B] = 0.2 mol·L−1 | 1000 | 62.5 | 50 |
| XRF Analysis | WBC-C (wt.%) | WBC-O (wt.%) | Phase Composition | WBC-C (g/100 g of Wollastonite) | WBC-O (g/100 g of Wollastonite) | |
|---|---|---|---|---|---|---|
| Ca | 17.9 | 17.2 | Brushite CaHPO4·2H2O | 14.9 ± 2.2 | 41.2 ± 6.1 | |
| Si | 13.4 | 13.3 | Wollastonite CaSiO3 | 8.5 ± 1.2 | 22.1 ± 3.3 | |
| P | 11.6 | 11.3 | Quartz SiO2 | 2.5 ± 0.4 | 3.6 ± 0.6 | |
| Zn | 3.69 | - | Amorphous silica SiO2·3.93H2O | 97.9 ± 14.6 | 81.8 ± 12.2 | |
| Al | 1.4 | 2.65 | Amorphous aluminophosphate | 56.9 ± 8.5 Ca/P = 1.1 ± 0.1 Al/P = 0.16 ± 0.02 Zn/P = 0.17 ± 0.02 | 36.1 ± 5.4 Ca/P = 0.84 ± 0.1 Al/P = 0.42 ± 0.1 - | |
| Na | 0.902 | - | Pore solution | 44.4 ± 6.6 | 39.7 ± 5.9 | |
| Mg | 0.392 | 0.352 | ||||
| Fe | 0.214 | 0.189 | ||||
| Mineralogical composition of pristine 28°d-old WBC-C and WBC-O pastes (wt.%) | ||||||
| WBC-C | WBC-O | |||||
 |  | |||||
| Type of Paste | Mass Fraction of Water (%) | Dose | G(H2)material × 10−7 (mol·J−1) | G(H2)water × 10−7 (mol·J−1) | Ref. |
|---|---|---|---|---|---|
| WBC-C | 23.0 | 100 kGy up to 1 MGy | 0.25 ± 0.02 | 1.10 ± 0.11 | This work |
| Geopolymer Geo Na | 33.0 | 100 and 250 kGy | 0.13 ± 0.10 | 0.4 ± 0.04 | [52] |
| Geo K | 33.9 | 0.25 ± 0.02 | 0.75 ± 0.07 | ||
| Geo Cs | 26.8 | 0.48 ± 0.04 | 1.80 ± 0.18 | ||
| Portland cement | 16.7 37.5 | 1 MGy | 0.13 ± 0.10 0.33 ± 0.03 | [53] | |
| Portland cement (CEM I 52.5 N SR0 CE PM-CP2 NF) | 16.7 | 500 kGy | 0.05 ± 0.01 | 0.31 ± 0.03 | [30] |
| 23.1 | 0.07 ± 0.01 | 0.30 ± 0.04 | |||
| 28.6 | 0.10 ± 0.01 | 0.32 ± 0.03 | |||
| 33.3 | 0.11 ± 0.01 | 0.32 ± 0.03 | |||
| 37.5 | 0.11 ± 0.01 | 0.30 ± 0.03 | |||
| Calcium sulfoaluminate cement (85% clinker + 15% CaSO4) | 28.6 | 100 and 200 kGy | 0.10 ± 0.01 | 0.37 ± 0.01 | |
| Calcium aluminate cement | 16.6 | 500 kGy | 0.009 ± 0.005 | 0.055 ± 0.005 | [30] |
| 23.0 | 0.018 ± 0.002 | 0.078 ± 0.008 | |||
| 28.4 | 0.032 ± 0.003 | 0.11 ± 0.01 | |||
| 33.1 | 150 and 300 kGy | 0.057 ± 0.006 | 0.17 ± 0.01 | ||
| 37.3 | 0.083 ± 0.008 | 0.22 ± 0.02 | |||
| Magnesium phosphate cement (molar ratios) MgO/KH2PO4 = 1 H2O/MgO = 5 | 40.5 | 150 and 300 kGy | 0.084 ± 0.010 | 0.21 ± 0.03 | [34] |
| Bulk water (pH 13) | 0.44 ± 0.04 | [54] |
| Model | Value | Standard Error | t-Value | Prob > |t| (%) | |
|---|---|---|---|---|---|
r2adjusted = 0.992 | k1 | 4.04 × 10−4 | 1.17 × 10−4 | 3.45 | * |
| k2 | 4.92 × 10−1 | 1.25 × 10−1 | 3.93 | ** | |
| k3 | 1.59 × 10−4 | 4.50 × 10−5 | 3.54 | * | |
| k4 | −7.94 × 10−6 | 3.67 × 10−6 | −2.16 | 7.40 | |
r2adjusted = 0.988 | k1 | 6.48 × 10−4 | 6.25 × 10−5 | 10.37 | *** |
| k2 | 4.27 × 10−1 | 8.20 × 10−2 | 5.20 | ** | |
| k3 | 6.20 × 10−5 | 9.53 × 10−6 | 6.50 | *** |
| Model | Value | Standard Error | t-Value | Prob > |t| (%) | |
|---|---|---|---|---|---|
r2adjusted = 0.991 | k1 | 3.94 × 10−4 | 1.09 × 10−4 | 3.61 | * |
| k2 | 4.04 × 10−1 | 0.91 × 10−1 | 4.41 | ** | |
| k3 | 1.31 × 10−4 | 4.01 × 10−5 | 3.27 | * | |
| k4 | −7.29 × 10−6 | 3.22 × 10−6 | −2.27 | 6.4 | |
r2adjusted = 0.987 | k1 | 6.26 × 10−4 | 6.01 × 10−5 | 10.40 | *** |
| k2 | 3.69 × 10−1 | 0.66 × 10−1 | 5.59 | *** | |
| k3 | 4.09 × 10−5 | 8.89 × 10−6 | 4.60 | ** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Jdaini, J.; Cau Dit Coumes, C.; Barré, Y.; de Noirfontaine, M.-N.; Courtial, M. Potential of Wollastonite-Based Brushite Cement for the Conditioning of Radioactive Waste Contaminated by 90Sr. Materials 2026, 19, 1136. https://doi.org/10.3390/ma19061136
Jdaini J, Cau Dit Coumes C, Barré Y, de Noirfontaine M-N, Courtial M. Potential of Wollastonite-Based Brushite Cement for the Conditioning of Radioactive Waste Contaminated by 90Sr. Materials. 2026; 19(6):1136. https://doi.org/10.3390/ma19061136
Chicago/Turabian StyleJdaini, Jihane, Céline Cau Dit Coumes, Yves Barré, Marie-Noëlle de Noirfontaine, and Mireille Courtial. 2026. "Potential of Wollastonite-Based Brushite Cement for the Conditioning of Radioactive Waste Contaminated by 90Sr" Materials 19, no. 6: 1136. https://doi.org/10.3390/ma19061136
APA StyleJdaini, J., Cau Dit Coumes, C., Barré, Y., de Noirfontaine, M.-N., & Courtial, M. (2026). Potential of Wollastonite-Based Brushite Cement for the Conditioning of Radioactive Waste Contaminated by 90Sr. Materials, 19(6), 1136. https://doi.org/10.3390/ma19061136







