Study on the Performance Evolution Law and Microscopic Mechanism of Cement–Sodium Silicate Grout Prepared by Seawater
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation Method and Performance Testing Methods of CSG
2.3. Experimental Design
3. Test Results and Analyses
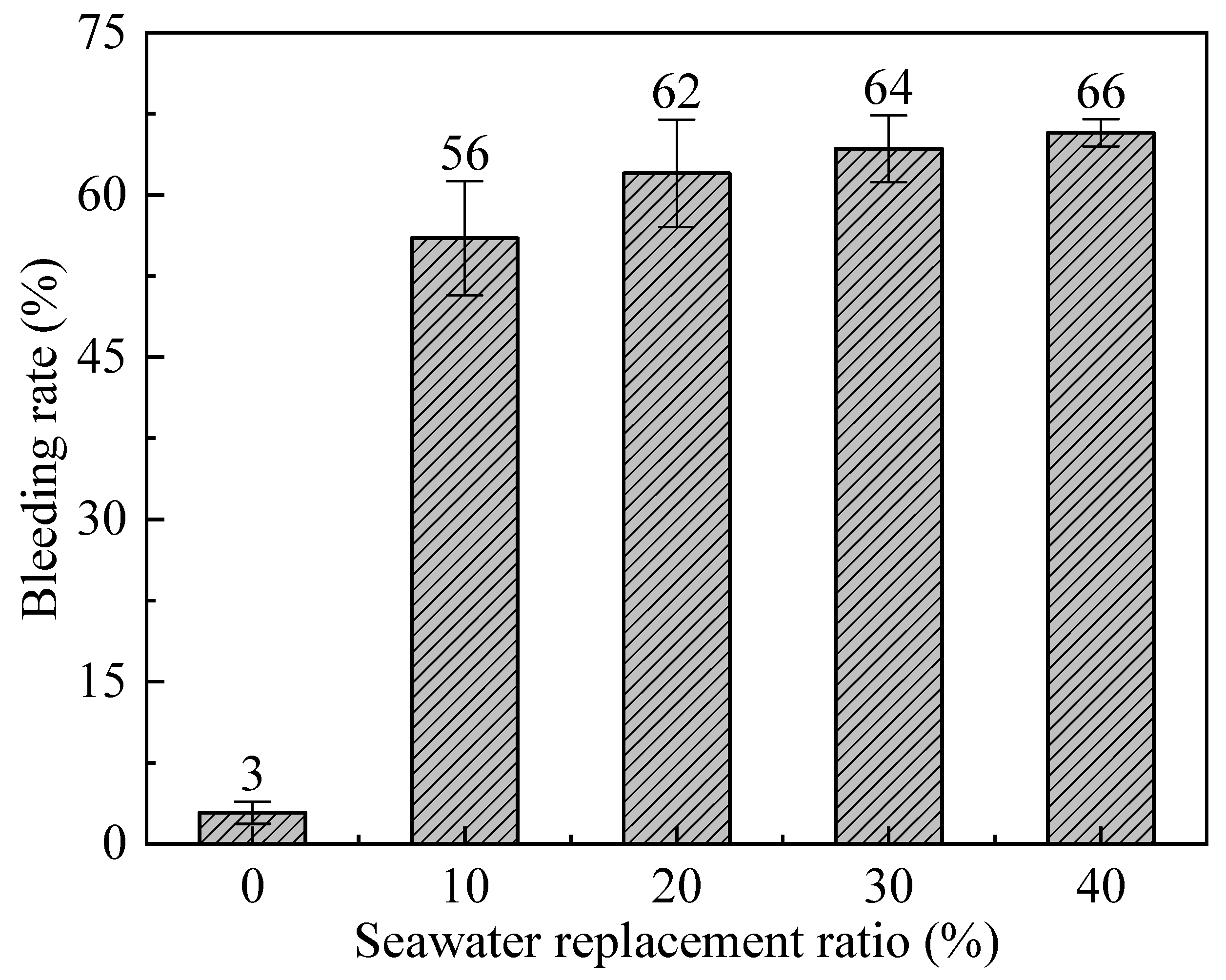
3.1. Bleeding Rate and Viscosity
3.2. Gel Time
3.3. Compressive Strength
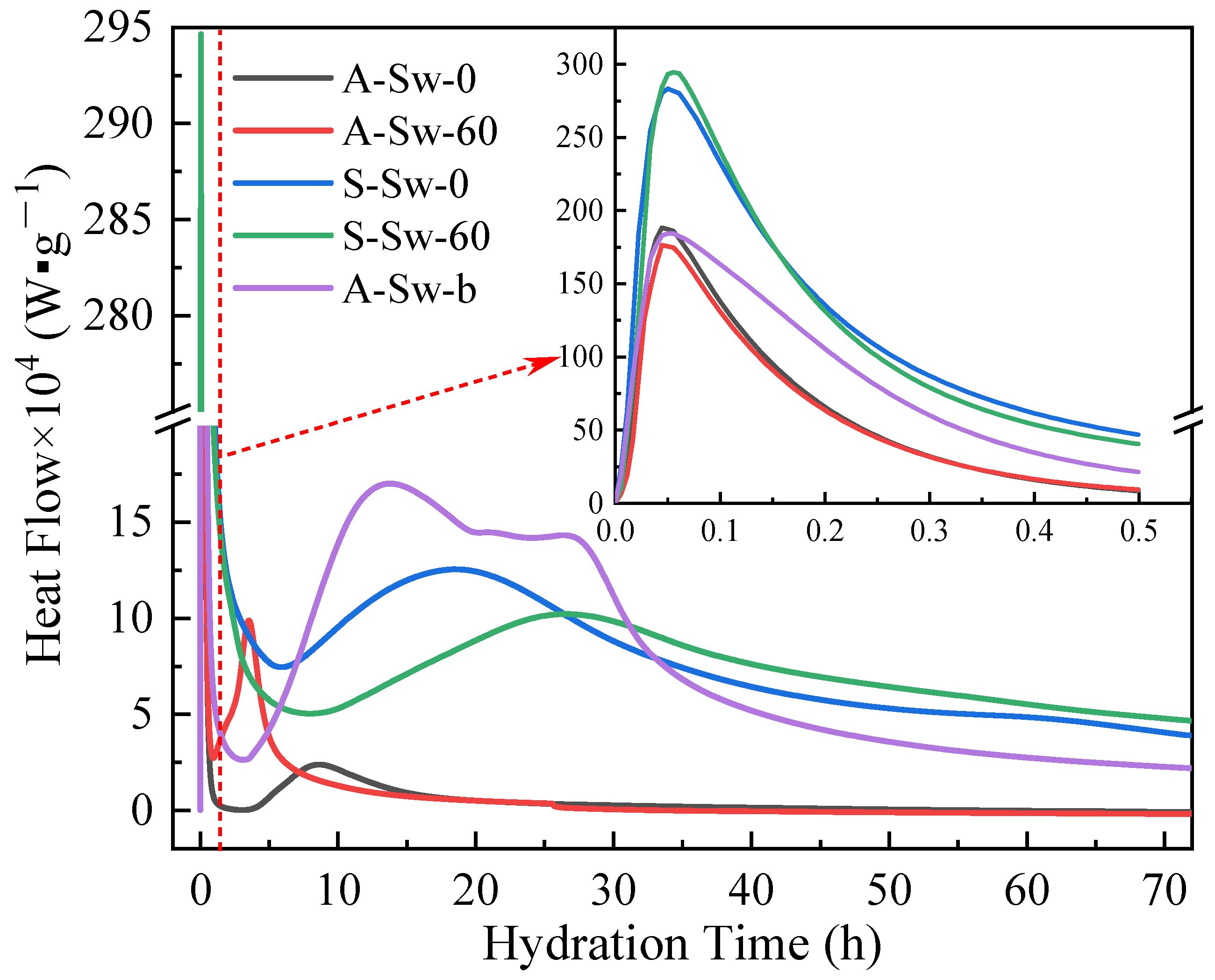
3.4. Hydration Heat Analysis
3.5. XRD Analysis
3.6. SEM Analysis
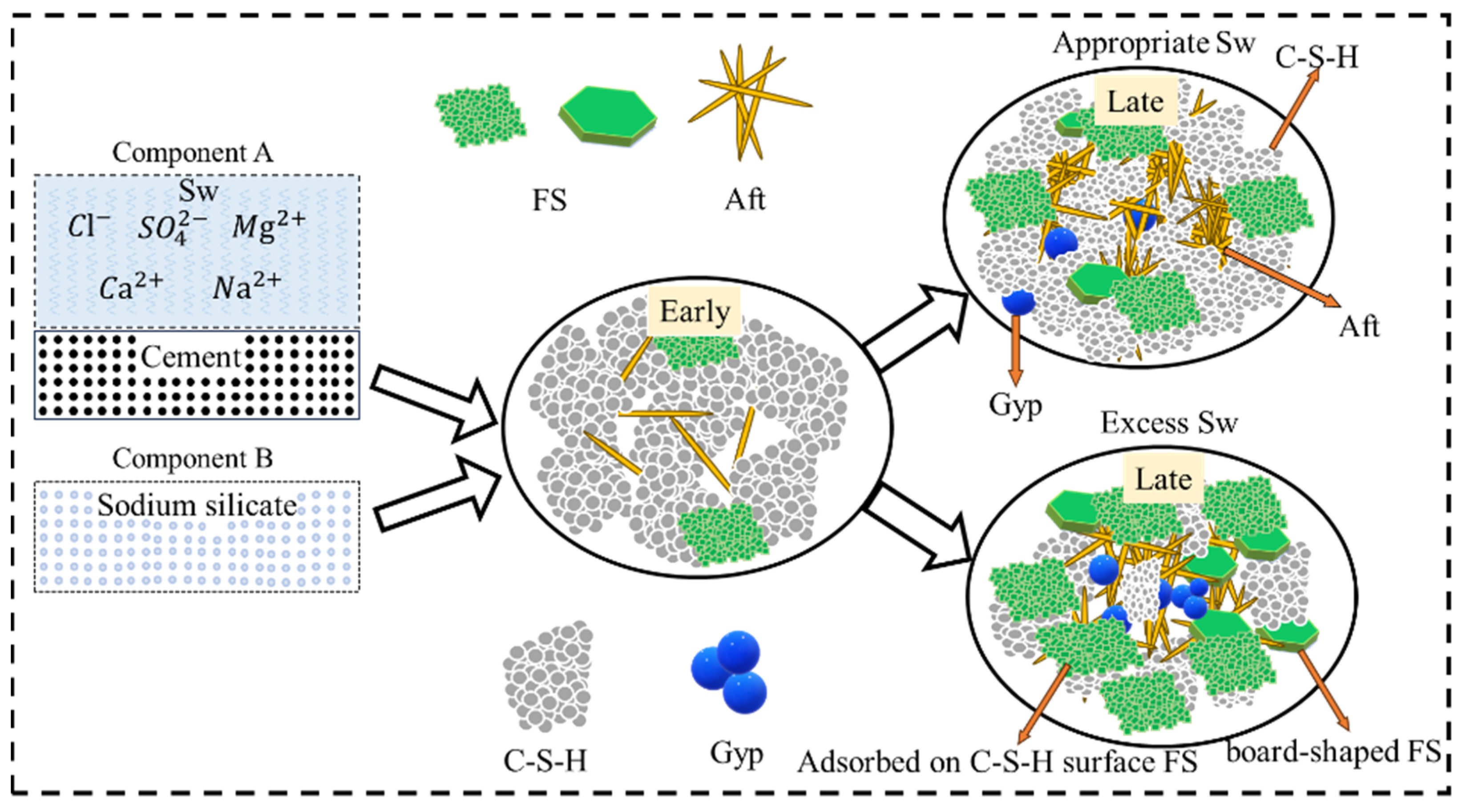
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Feng, K.; He, C.; Qiu, Y.; Zhang, L.; Wang, W.; Xie, H.; Zhang, Y.; Cao, S. Full-scale tests on bending behavior of segmental joints for large underwater shield tunnels. Tunn. Undergr. Space Technol. 2018, 75, 100–116. [Google Scholar] [CrossRef]
- Chen, Y. Investigation into the Characteristics and Strength Simulation Test of the Complex Two-Component in Synchronous Grouting of Shield Tunneling; Beijing Jiaotong University: Beijing, China, 2017. [Google Scholar]
- Wang, D.; Min, F.; Li, Z. Investigation into the properties of two-component backfilling grouts prepared by substituting bentonite with slurry from slurry shield tunnelling treated by a hydrocyclone. Tunn. Undergr. Space Technol. 2024, 154, 106142. [Google Scholar] [CrossRef]
- Todaro, C.; Carigi, A.; Martinelli, D.; Peila, D. Study of the shear strength evolution over time of two-component backfilling grout in shield tunnelling. Case Stud. Constr. Mater. 2021, 15, 00689. [Google Scholar] [CrossRef]
- Sharghi, M.; Chakeri, H.; Ozcelik, Y. Investigation into the effects of two component grout properties on surface settlements. Tunn. Undergr. Space Technol. 2017, 63, 205–216. [Google Scholar] [CrossRef]
- Wang, S.; He, C.; Nie, L.; Zhang, G. Study on the long-term performance of cement-sodium silicate grout and its impact on segment lining structure in synchronous backfill grouting of shield tunnels. Tunn. Undergr. Space Technol. 2019, 92, 103015. [Google Scholar] [CrossRef]
- Shah, R.; A. Lavasan, A.; Peila, D.; Todaro, C.; Luciani, A.; Schanz, T. Numerical study on backfilling the tail void using a two-component grout. J. Mater. Civil. Eng. 2018, 30, 04018003. [Google Scholar] [CrossRef]
- Chen, F.; Huang, P.; Ba, M.; Bao, X. Experimental study on dual-fluid-grout composite materials with slag steel modified portland cement and sodium silicate. Acta Mater. Compos. Sin. 2013, 30, 139. [Google Scholar]
- Dou, H. Investigation on the Performance and Microscopic Characteristics of Complex Cementitious-Sodium Silicate Two-Shot Grouting Materials; China University of Geosciences: Wuhan, China, 2014. [Google Scholar]
- Sharghi, M.; Chakeri, H.; Afshin, H.; Ozcelik, Y. An experimental study of the performance of two-component backfilling grout used behind the segmental lining of a tunnel-boring machine. J. Test. Eval. 2018, 46, 2083–2099. [Google Scholar] [CrossRef]
- Peila, D.; Chieregato, A.; Martinelli, D.; Onate Salazar, C.G.; Shah, R.; Boscaro, A.; Dal Negro, E.; Picchio, A. Long term behavior of two component back-fill grout mix used in full face mechanized tunneling. Geoing. Ambient. 2015, 144, 57–63. [Google Scholar]
- He, X.; Ma, S.; Chen, J.; Li, M.; Yu, L.; He, J. Field test on construction influence on large-diameter shield tail segment floating of tunnel. Tunnel. Constr. 2023, 43, 136. [Google Scholar]
- Shi, Y.; Ruan, J.; Lin, S. Floating Analysis and Control of Subsea Shield Tunnel Segment. Chin. J. Undergr. Space Eng. 2022, 18, 1665–1677. [Google Scholar]
- Dal, E.; Boscaro, A.; Plescia, E. Two-component backfill grout system in TBM. The experience of the tunnel “Sparvo” in Italy. In Proceedings of the Tunnelling in a Resource Driven World Conference, TAC, Vancouver, BC, Canada, 26–28 October 2014. [Google Scholar]
- Todaro, C.; Saltarin, S.; Cardu, M. Bentonite in two-component grout applications. Case Stud. Constr. Mater. 2022, 16, 00901. [Google Scholar] [CrossRef]
- Youn, B.; Shulte-Schrep, C.; Breitenbucher, R. Properties and Requirements of Two-Component Grouts in Mechanized Tunneling. In Proceedings of the ITA WTC World Tunnel Congress 2016, San Francisco, CA, USA, 22–28 April 2016. [Google Scholar]
- Jia, Y.; Li, F.; Wu, D.; Wang, Y. Experimental Study on Mix Proportions of Synchronous Two-component Grouting for Shield Tunnels. Cogent Eng. 2019, 56, 143–151,157. [Google Scholar]
- Mesboua, N.; Benyounes, K.; Benmounah, A. Study of the impact of bentonite on the physico-mechanical and flow properties of cement grout. Cogent Eng. 2018, 5, 1446252. [Google Scholar] [CrossRef]
- Todaro, C.; Zanti, D.; Carigi, A.; Peila, D. The role of bentonite in two-component grout: A comparative study. Tunn. Undergr. Space Technol. 2023, 142, 105412. [Google Scholar] [CrossRef]
- Todaro, C.; Pace, F. Elastic properties of two-component grouts at short curing times: The role of bentonite. Tunn. Undergr. Space Technol. 2022, 130, 104756. [Google Scholar] [CrossRef]
- Liao, W.; Wu, P.; Huang, J.; Chen, G.; Lin, J.; Guo, Y.; Chen, R. Cost-Effective Engineered Cementitious Composites with Hybrid PVA and Basalt/PP Fiber: A Study on Compressive, Tensile and Impact Performance. Materials 2023, 16, 5172. [Google Scholar] [CrossRef]
- Yi, J.; Wang, L.; Ma, L.; Zhang, Q.; Zhang, J.; Chi, J. Experimental Study on Basic Mechanical Properties of PVA Fiber-Reinforced Coral Cement-Based Composites. Materials 2023, 16, 2914. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Zhu, Z.; Pu, S.; Wan, Y.; Huo, W.; Peng, Y. Preparation and engineering properties of alkali-activated filling grouts for shield tunnel. Constr. Build. Mater. 2022, 314, 125620. [Google Scholar] [CrossRef]
- Schulte-Schrep, C.; Ov, D.; Breitenbücher, R. Solidification of two-component grouts by the use of superabsorbent polymers as activator. In 3rd International Conference on the Application of Superabsorbent Polymers (SAP) and Other New Admixtures Towards Smart Concrete; Springer International Publishing: Cham, Switzerland, 2020; Volume 3, pp. 230–238. [Google Scholar]
- Sun, Z.; Li, X.; Liu, Q.; Tang, Q.; Lin, X.; Fan, X.; Huang, X.; Gan, M.; Chen, X.; Ji, Z. Recent Advances in Alkali-Activated Materials with Seawater and Sea Sand. Materials 2023, 16, 3571. [Google Scholar] [CrossRef]
- Xiao, J.; Qiang, C.; Nanni, A.; Zhang, K. Use of sea-sand and seawater in concrete construction: Current status and future opportunities. Constr. Build. Mater. 2017, 155, 1101–1111. [Google Scholar] [CrossRef]
- Ge, L.; Feng, Z.; Sayed, U.; Li, H. Research on the performance of seawater sea-sand concrete: A review. Constr. Build. Mater. 2023, 409, 133921. [Google Scholar] [CrossRef]
- Chen, J.; Jia, J.; Zhu, M. Development of admixtures on seawater sea sand concrete: A critical review on Concrete hardening, chloride ion penetration and steel corrosion. Constr. Build. Mater. 2024, 411, 134219. [Google Scholar] [CrossRef]
- Shi, D.; Yao, Y.; Ye, J.; Zhang, W. Effects of seawater on mechanical properties, mineralogy and microstructure of calcium silicate slag-based alkali-activated materials. Constr. Build. Mater. 2019, 212, 569–577. [Google Scholar] [CrossRef]
- Lao, J.; Huang, B.; Xu, L.; Khan, M.; Fang, Y.; Dai, J. Seawater sea-sand Engineered Geopolymer Composites (EGC) with high strength and high ductility. Cement Concrete Comp. 2023, 138, 104998. [Google Scholar] [CrossRef]
- Cao, Y.Z.; Guo, L.P.; Xue, X.L. Effects of sodium chloride and sodium sulfate on hydration process. J. Southeast Univ. (Nat. Sci. Ed.) 2019, 49, 1001-0505. [Google Scholar]
- Sikora, P.; Cendrowski, K.; Abd Elrahman, M.; Chung, S.Y.; Mijowska, E.; Stephan, D. The effects of seawater on the hydration, microstructure and strength development of Portland cement pastes incorporating colloidal silica. Appl. Nanosci. 2020, 10, 2627–2638. [Google Scholar] [CrossRef]
- Younis, A.; Ebead, U.; Suraneni, P.; Nanni, A. Fresh and hardened properties of seawater-mixed concrete. Constr. Build. Mater. 2018, 190, 276–286. [Google Scholar] [CrossRef]
- Xiao, S.; Zhang, M.; Zou, D.; Liu, T.; Zhou, A.; Li, Y. Influence of seawater and sea sand on the performance of Anti-washout underwater concrete: The overlooked significance of Mg2+. Constr. Build. Mater. 2023, 374, 130932. [Google Scholar] [CrossRef]
- Wang, X.; Shi, C.; He, F.; Yuan, Q.; Wang, D.; Huang, Y.; Li, Q. Chloride binding and its effects on microstructure of cement-based materials. J. Chin. Ceram. Soc. 2013, 41, 187–198. [Google Scholar]
- De Weerdt, K.; Colombo, A.; Coppola, L.; Justnes, H.; Geiker, M.R. Impact of the associated cation on chloride binding of Portland cement paste. Cement Concrete Res. 2015, 68, 196–202. [Google Scholar] [CrossRef]
- Talero, R.; Trusilewicz, L.; Delgado, A.; Pedrajas, C.; Lannegrand, R.; Rahhal, V.; Mejía, R.; Delvasto, S.; Ramírez, F. Comparative and semi-quantitative XRD analysis of Friedel’s salt originating from pozzolan and Portland cement. Constr. Build. Mater. 2011, 25, 2370–2380. [Google Scholar] [CrossRef]
- Guo, M.; Hu, B.; Xing, F.; Zhou, X.; Sun, M.; Sui, L.; Zhou, Y. Characterization of the mechanical properties of eco-friendly concrete made with untreated sea sand and seawater based on statistical analysis. Constr. Build. Mater. 2020, 234, 117339. [Google Scholar] [CrossRef]
- Yaseen, S.; Yiseen, G.; Poon, C.; Li, Z. Influence of seawater on the morphological evolution and the microchemistry of hydration products of tricalcium silicates (C3S). ACS Sustain. Chem. Eng. 2020, 8, 15875–15887. [Google Scholar] [CrossRef]
- Sukmak, P.; De Silva, P.; Horpibulsuk, S.; Chindaprasirt, P. Sulfate resistance of clay-portland cement and clay high-calcium fly ash geopolymer. J. Mater. Civil. Eng. 2015, 27, 04014158. [Google Scholar] [CrossRef]
- Ting, M.; Yi, Y. Durability of cementitious materials in seawater environment: A review on chemical interactions, hardened-state properties and environmental factors. Constr. Build. Mater. 2023, 367, 130224. [Google Scholar] [CrossRef]
- American Society for Testing and Materials. Standard Practice for the Preparation of Substitute Ocean Water; ASTM International: West Conshohocken, PA, USA, 2013.
- T/CECS 563; Technical Specification for Simultaneous Grouting Material in Shield Projects. China Building Materials Press: Beijing, China, 2018.
- JTG/T 3650; Technical Specifications for Construction of Highway Bridges and Culverts. China Building Materials Press: Beijing, China, 2020.
- Zhang, J.; Guan, X.; Li, H.; Liu, X. Performance and hydration study of ultra-fine sulfoaluminate cement-based double liquid grouting material. Constr. Build. Mater. 2017, 132, 262–270. [Google Scholar] [CrossRef]
- Zhang, C.; Yang, J.; Fu, J.; Ou, X.; Xie, Y.; Liang, X. Performance evaluation of modified cement-sodium silicate grouting material for prereinforcing loose deposit tunnels. J. Mater. Civil. Eng. 2019, 31, 06019003. [Google Scholar] [CrossRef]
- Yu, Z.; Yang, L.; Zhou, S.; Gong, Q.; Zhu, H. Durability of cement-sodium silicate grouts with a high water to binder ratio in marine environments. Constr. Build. Mater. 2018, 189, 550–559. [Google Scholar] [CrossRef]
- GB/T 17671; Test Method of Cement Mortar Strength (ISO Method). China Standards Press: Beijing, China, 2021.
- Au, P.; Pillai, P.; Leong, Y. Ageing and collapse of bentonite gels—Effects of Mg (II), Ca (II) and Ba (II) ions. Appl. Clay Sci. 2015, 114, 141–150. [Google Scholar] [CrossRef]
- Gorakhki, M.; Bareither, C. Salinity effects on sedimentation behavior of kaolin, bentonite, and soda ash mine tailings. Appl. Clay Sci. 2015, 114, 593–602. [Google Scholar] [CrossRef]
- Ganesalingam, D.; Sivakugan, N.; Ameratunga, J. Influence of settling behavior of soil particles on the consolidation properties of dredged clay sediment. J. Waterw. Port. Coast. 2013, 139, 295–303. [Google Scholar] [CrossRef]
- Min, F.; Du, J.; Zhang, N.; Chen, X.; Lv, H.; Liu, L.; Yu, C. Experimental study of property change of slurry and filter cake of slurry shield under seawater intrusion. Tunn. Undergr. Space Technol. 2019, 88, 290–299. [Google Scholar] [CrossRef]
- Wang, D.; Min, F.; Zhou, X.; Liu, L.; Li, B.; Wang, X.; Du, J.; Zhang, J. Influence of cation on properties of slurry and filter cake used in submarine shield tunnelling. Mar. Georesour. Geotechnol. 2025, 43, 1732–1740. [Google Scholar] [CrossRef]
- Sha, F.; Liu, R.; Li, S.; Lin, C.; Li, Z.; Liu, B.; Bai, J. Application on different types of cementitious grouts for water-leakage operational tunnels. J. Cent. South. Univ. Sci. Technol. 2016, 47, 4163–4172. [Google Scholar]
- Song, B.; Min, F.; Zhang, J.; Li, M.; Zhang, Y.; Fang, Z.; Zhang, L.; Zhao, X. Influence of Triethanolamine and Rapid Hardening Sulfoaluminate Cement on Performance of Backfill Grouting in Shield Engineering. J. Chin. Ceram. Soc. 2022, 50, 2886–2896. [Google Scholar]
- Chindaprasirt, P.; De Silva, P.; Sagoe-Crentsil, K.; Hanjitsuwan, S. Effect of SiO2 and Al2O3 on the setting and hardening of high calcium fly ash-based geopolymer systems. J. Mater. Sci. 2012, 47, 4876–4883. [Google Scholar] [CrossRef]
- Chen, R.; Lai, H.; Cui, D.; Zhu, Y. Alkali-activated mortar for tunnel-lining structure repair. J. Mater. Civil. Eng. 2019, 31, 04019217. [Google Scholar] [CrossRef]
- Cai, Y.; Xuan, D.; Hou, P.; Shi, J.; Poon, C.S. Effect of seawater as mixing water on the hydration behaviour of tricalcium aluminate. Cement Concrete Res. 2021, 149, 106565. [Google Scholar] [CrossRef]
- Thomas, J.; Allen, A.; Jennings, H. Density and water content of nanoscale solid C–S–H formed in alkali-activated slag (AAS) paste and implications for chemical shrinkage. Cement Concrete Res. 2012, 42, 377–383. [Google Scholar] [CrossRef]
- Gao, Y.; Xu, J.; Luo, X.; Zhu, J.; Nie, L. Experiment research on mix design and early mechanical performance of alkali-activated slag using response surface methodology (RSM). Ceram. Int. 2016, 42, 11666–11673. [Google Scholar] [CrossRef]
- Pan, D.; Yaseen, S.; Chen, K.; Niu, D.; Leung, C.; Li, Z. Study of the influence of seawater and sea sand on the mechanical and microstructural properties of concrete. J. Build. Eng. 2021, 42, 103006. [Google Scholar] [CrossRef]
- André, L.; Bacquié, C.; Comin, G.; Ploton, R.; Achard, D.; Frouin, L. Cyr Martin Improvement of two-component grouts by the use of ground granulated blast furnace slag. Tunn. Undergr. Space Technol. 2022, 122, 104369. [Google Scholar] [CrossRef]
- Huang, Y.; Tan, Z.; Zhang, W.; Liu, X.; Song, J.; Shang, H. Incorporating supplementary cementitious materials into ultra-high-performance seawater sea-sand concrete (UHPSSC): Hydration, microstructure, and mechanical performance. Constr. Build. Mater. 2025, 472, 140824. [Google Scholar] [CrossRef]
- Zhang, W.; Ding, D.; Sun, J.; Guo, F.; Feng, H.; Ma, H.; Hong, S.; Dong, B.; Hou, D. Novel insights on the setting process, hardened properties, and durability of sustainable ultra-high-performance seawater sea sand concrete. Constr. Build. Mater. 2024, 447, 138092. [Google Scholar] [CrossRef]






| Cement | Water | Bentonite | Stabilizer | Sodium Silicate |
|---|---|---|---|---|
| 300 | 900 | 36 | 3 | 75 |
| Samples | Number | Sw Replacement Ratio (%) | Fw Replacement Ratio (%) | Test Items |
|---|---|---|---|---|
| Component A | A-Sw-b | 0 | 100 | hydration |
| A-Sw-0 | 0 | 100 | bleeding rate, viscosity, hydration heat | |
| A-Sw-15 | 15 | 85 | bleeding rate, viscosity | |
| A-Sw-30 | 30 | 70 | bleeding rate, viscosity | |
| A-Sw-45 | 45 | 55 | bleeding rate, viscosity | |
| A-Sw-60 | 60 | 40 | bleeding rate, viscosity, hydration heat | |
| CSG | S-Sw-0 | 0 | 100 | gel time, compressive strength, hydration heat, XRD, SEM |
| S-Sw-15 | 15 | 85 | gel time, compressive strength | |
| S-Sw-30 | 30 | 70 | gel time, compressive strength | |
| S-Sw-45 | 45 | 55 | gel time, compressive strength, XRD, SEM | |
| S-Sw-60 | 60 | 40 | gel time, compressive strength, hydration heat, XRD, SEM |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Wang, D.; Li, Z.; Qi, Y.; Wei, D.; Zhao, X.; Zhang, J.; Min, F. Study on the Performance Evolution Law and Microscopic Mechanism of Cement–Sodium Silicate Grout Prepared by Seawater. Materials 2026, 19, 971. https://doi.org/10.3390/ma19050971
Wang D, Li Z, Qi Y, Wei D, Zhao X, Zhang J, Min F. Study on the Performance Evolution Law and Microscopic Mechanism of Cement–Sodium Silicate Grout Prepared by Seawater. Materials. 2026; 19(5):971. https://doi.org/10.3390/ma19050971
Chicago/Turabian StyleWang, Dengfeng, Zhen Li, Yujie Qi, Daiwei Wei, Xiaopeng Zhao, Jianfeng Zhang, and Fanlu Min. 2026. "Study on the Performance Evolution Law and Microscopic Mechanism of Cement–Sodium Silicate Grout Prepared by Seawater" Materials 19, no. 5: 971. https://doi.org/10.3390/ma19050971
APA StyleWang, D., Li, Z., Qi, Y., Wei, D., Zhao, X., Zhang, J., & Min, F. (2026). Study on the Performance Evolution Law and Microscopic Mechanism of Cement–Sodium Silicate Grout Prepared by Seawater. Materials, 19(5), 971. https://doi.org/10.3390/ma19050971






