1. Introduction
In biomedical engineering, tissue-mimicking phantoms are not merely experimental surrogates but engineered functional materials whose performance is governed by well-defined structure–property relationships. By reproducing the electromagnetic and thermal behavior of real biological tissues, these materials enable safe, controlled, and repeatable studies for dosimetric assessment, such as the evaluation of the specific absorption rate (SAR) in microwave hyperthermia, as well as the establishment of exposure limits for near-field and therapeutic devices [
1,
2]. From a materials-science perspective, however, the fidelity of such studies depends critically on rigorous characterization of phantom composition and multiphysical properties, ensuring that dielectric permittivity, electrical conductivity, thermal conductivity, and specific heat capacity remain consistent with those of the target tissue under realistic operating conditions, as reported in optical phantom studies [
2,
3], microwave, and thermal hyperthermia analysis [
4,
5,
6]. Only through such validation can phantoms be regarded as reliable material platforms capable of predicting in vivo behavior.
Over the past two decades, phantom design has advanced in deliberate and meaningful steps to meet the growing demands of biomedical research. Early efforts focused on reconstructing internal temperature changes using capacitance-based measurements in brain-mimicking media [
7]. Building on that foundation, just a year later, Ito and colleagues developed a solid, muscle-equivalent phantom for microwave dosimetry, laying out the first recipes to match tissue permittivity and conductivity [
8]. Subsequent developments expanded phantom functionality, including reusable heat-sensitive materials for focused ultrasound hyperthermia that enabled spatially resolved thermal mapping and improved understanding of treatment dynamics [
5]. Gelatin-based models with adaptable Bloom resistance were a significant advancement in phantom fabrication because they allowed for the adjustment of gel stiffness and network structure to mimic multiple soft tissues while systematically characterizing the resulting acoustic, magnetic resonance, and thermal responses within a single material platform [
9]. Collectively, these studies illustrate a gradual transition from empirical phantom recipes toward more rational material design, where composition and microstructure are tailored to achieve reproducible multiphysical responses.
By the early 2020s, phantom research had focused on the evaluation of a diverse tapestry of specialized materials and measurement techniques. Researchers started with liquid optical phantoms designed to cover the visible-near-infrared spectrum (500–1000 nm). They used an enhanced single-integrating sphere setup, meticulously prepared dozens of absorption, scattering, and mixed component samples to benchmark the absorption coefficient (
) and reduced scattering coefficient (
), demonstrating the stability and repeatability of the system [
10]. At the same time, both Zerdine (a commercial material) and agar phantoms were illuminated at 635 nm to extract their core optical parameters, such as transmittance, absorbance, reflectance, refractive index, and attenuation, via direct power comparisons in a homogeneous setup and with a single integrating sphere system. The integrating sphere observations most nearly matched expected tissue values, confirming its role as the gold standard for optical phantom characterization [
11]. Complementary efforts extended dielectric characterization to higher microwave and submillimeter-wave frequencies; for example, water–oil–agar blends were employed to investigate D-band (150 GHz) permittivity in breast tissue mimics [
12], while heterogeneous solid skin-equivalent phantoms were validated for diagnostic microwave applications across 0.5–26.5 GHz using coaxial probe measurements and vector network analyzers [
8,
13]. Agar–SiO
2 tumor phantoms tailored for MRI-guided focused ultrasound demonstrated realistic lesion formation and thermal dose deposition by combining intermittent thermometry with multimodal imaging contrast [
14].
These characterization advances established the basis for optimizing hyperthermia therapies [
15]. For example, Nizam-Uddin et al. paired a flexible monopole applicator with a patient-mimicking breast phantom and fiber-optic thermometry to demonstrate precise, localized heating of multiple lesions [
16]. In parallel, Rahpeima and Lin used anatomically realistic breast phantoms loaded with magnetic nanofluids to simulate nanoparticle diffusion, heat generation via Pennes’ bioheat equation, and resulting tissue necrosis [
17]. Meanwhile, Fakhradini et al. optimized microwave antenna power in liver-cancer phantoms loaded with superparamagnetic nanoparticles, achieving maximal tumor heating with minimal collateral damage [
18]. On the monitoring front, Gaffoglio et al. introduced a real-time 3D temperature reconstruction method combining sparse fiber-optic readings with a simulation library, delivering full-field maps with sub-degree accuracy [
19], while Dietrich et al. validated 3D MRI thermometry in agar phantoms during microwave heating, reporting RMS errors below 0.7 °C without RF interference [
20]. Complementary reviews and parametric studies have further clarified the influence of applicator design, frequency selection, and antenna configuration on penetration depth, heating uniformity, and hotspot control in hyperthermia treatments [
21,
22], while microwave radiometry has been demonstrated as a non-invasive technique for detecting sub-degree internal temperature gradients in anatomically realistic breast phantoms [
23].
Beyond the microwave regime, optical and terahertz (THz) characterization has emerged as a complementary approach to probe phantom composition, microstructure, and hydration-dependent behavior from a materials-science perspective. In the optical domain, solid tissue phantoms with adjustable subdiffusive scattering parameters have demonstrated that controlled incorporation of scattering inclusions enables not only precise tuning of optical response but also long-term stability and mechanical robustness [
24]. These studies highlight that optical properties in tissue-mimicking materials are intrinsically linked to microstructural organization and constituent dispersion, rather than being merely spectral fitting parameters [
24,
25]. At higher frequencies, THz time-domain spectroscopy has gained increasing attention as a sensitive tool for interrogating soft and hydrated materials, owing to its strong interaction with water content, weak intermolecular forces, and mesoscale structural features. Recent advances in THz-based material characterization have emphasized the importance of reflection-mode approaches, which overcome the limitations of conventional transmission geometries when dealing with optically thick, opaque, or highly absorbing samples [
26]. Such developments significantly extend the applicability of THz spectroscopy to realistic tissue-mimicking materials, which can potentially enable the extraction of frequency-dependent dielectric properties.
Importantly, the application of THz techniques to hydrated phantoms has been systematically investigated in recent years. Jayasankar et al. demonstrated, using THz time-domain spectroscopy, that both the effective refractive index and absorption coefficient of hydrogel-based skin phantoms strongly depend on hydration level and water-binding mechanisms [
27]. Similarly, Saxena et al. reported that gelatin-based phantoms exhibit pronounced sensitivity of time-domain signatures and frequency-dependent attenuation to moisture redistribution and structural organization of bound water [
28]. These studies show that the THz response of hydrated phantoms cannot be accurately described using simplified effective-medium models. In particular, assuming invariant bound-water contributions leads to systematic deviations, as both the fraction and organization of bound water evolve with hydration state.
Despite the substantial progress outlined above, most existing studies still address dielectric, optical, terahertz, or thermal characterization—or hyperthermia validation—in isolation. Consequently, the relationship between phantom composition, broadband electromagnetic dispersion across multiple frequency decades, and thermal stability under sustained electromagnetic excitation remains insufficiently explored, particularly for muscle-equivalent materials intended for microwave hyperthermia and SAR validation.
In this study, we address this gap by fabricating a muscle-equivalent phantom following the formulation proposed by Ito et al. [
8] to reproduce real-tissue permittivity and conductivity. A multimodal characterization strategy was implemented to evaluate its electromagnetic and thermal behavior across multiple spectral regimes. The dielectric response was measured from 1.5 to 4 GHz using a coplanar waveguide method, optical absorption was analyzed over the 350–1200 nm range, and the THz response was investigated in reflection mode using THz-TDS to examine time- and frequency-domain signatures and their temporal stability. In parallel, thermal conductivity and volumetric heat capacity were experimentally determined. Finally, the phantom was exposed to 2.45 GHz microwave irradiation under hyperthermia conditions, and its temperature evolution was monitored using infrared thermography. By combining broadband dielectric characterization with controlled thermal validation, this work establishes a quantitative link between hydration state, dielectric dispersion, and thermal response and explicitly identifies hydration-driven dynamics as the key factor governing both electromagnetic performance and long-term stability under RF loading.
2. Materials and Methods
2.1. Materials
The muscle-equivalent phantom was prepared using deionized water, agar powder, polyethylene (PE) powder, sodium chloride (NaCl), TX-151 emulsifier, and sodium azide. The composition and suppliers are listed in
Table 1.
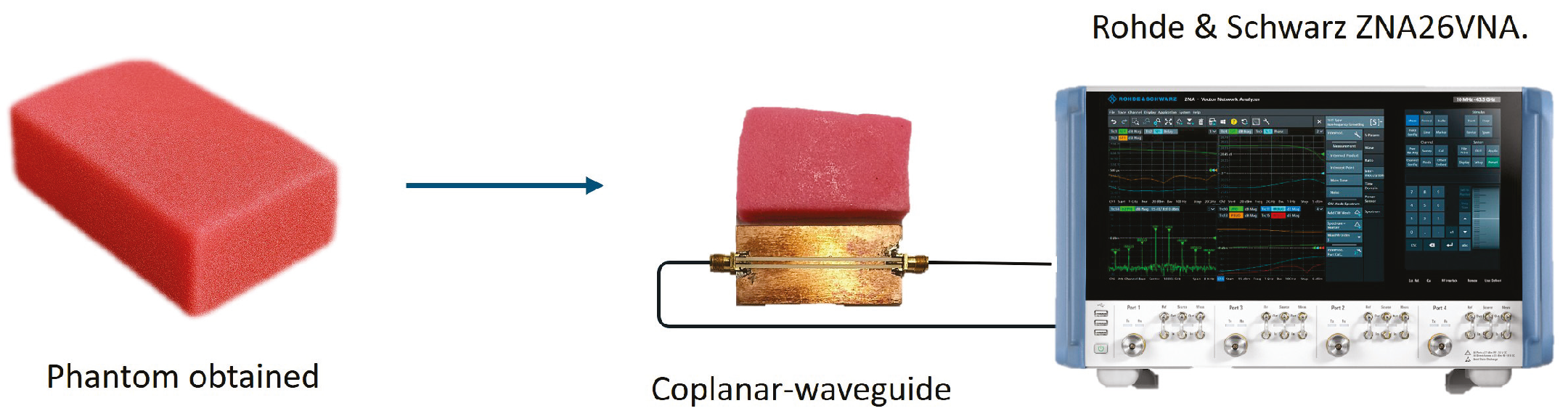
2.2. Phantom Fabrication
The sample preparation procedure was based on research from [
8], utilizing proportions that create a phantom with electrical properties similar to those of human muscle tissue. First, sodium chloride (NaCl) and sodium azide were dissolved in deionized water using a magnetic stirrer while the solution was heated. Then, the mixture was heated to 70 ºC to facilitate the incorporation of agar.
Once the agar was fully incorporated, the magnetic stirrer was replaced with a mixing motor to combine the polyethylene powder and the TX-151 solidifier simultaneously. Finally, the mixture was allowed to rest in a sealed container covered with plastic wrap for 24 h. It was then fully wrapped in the same film for storage at room temperature, ensuring it was not exposed to direct sunlight. The phantom was stored for two months before characterization. Any variations observed in the following measurements correspond to intra-sample changes associated with gradual moisture loss within the same specimen, and not to differences between independently fabricated samples.
Figure 1 shows the summary of the main steps for the development of the phantom.
2.3. EDS-Based Chemical Characterization
The chemical composition of the phantom was analyzed by energy-dispersive X-ray spectrometry (EDS) using an X-MaxN silicon drift detector (SDD, Oxford Instruments, Abingdon, Oxfordshire, UK) coupled to a field-emission scanning electron microscope (FE-SEM, JEOL JSM-7100F, JEOL Ltd., Tokyo, Japan). The microscope was operated in high-vacuum mode at an accelerating voltage of 20 kV and a working distance of approximately 10 mm. The analysis was performed at multiple locations across the phantom surface to assess compositional homogeneity. A total of ten independent EDS measurements were acquired under identical acquisition parameters, and the reported elemental composition corresponds to the averaged values obtained from these measurements.
2.4. Thermogravimetric and Thermal Properties Measurements
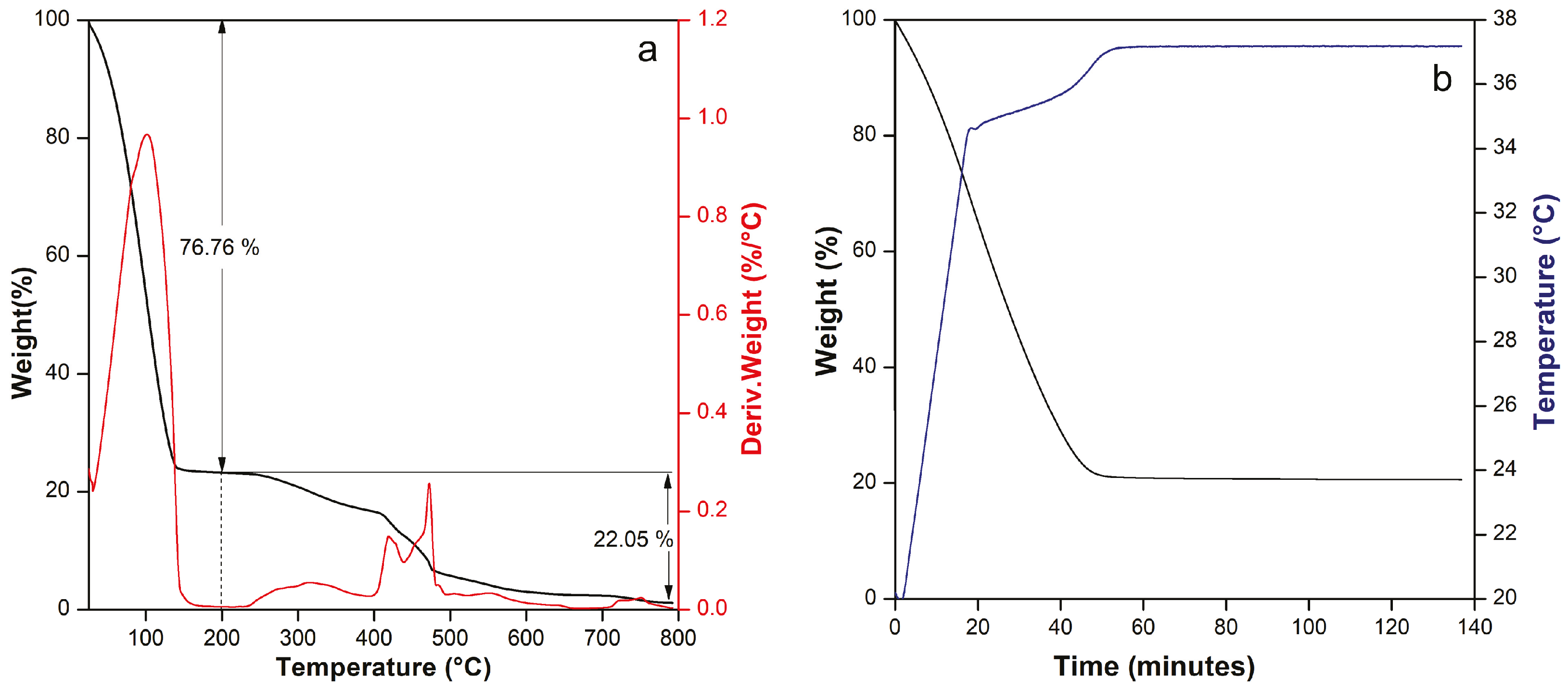
Thermogravimetric analysis (TGA) was conducted on a Discovery 550 Analyzer (TA Instruments, New Castle, DE, USA) to assess the thermal stability of the muscle-equivalent phantom under hyperthermia-relevant conditions. Samples were heated in ambient air from room temperature up to 800 °C at a constant 10 °C/min, allowing us to pinpoint any mass-loss transitions and confirm that the phantom matrix remains structurally intact well above therapeutic temperature ranges [
1]. In addition, to analyze the thermal behavior of the phantom at body temperature (37 °C), it was heated at a rate of 1 °C/min up to 37 °C. Finally, an isothermal process was conducted for 70 min.
Furthermore, the phantom’s capacity to conduct and store heat was evaluated using a KD2 Pro Thermal Properties Analyzer (Decagon Devices, Inc., Pullman, WA, USA). The KD2 Pro features a dual-needle sensor (30 mm active length, 6 mm needle spacing) that captures both thermal conductivity and volumetric heat capacity simultaneously. To reduce ambient fluctuations, the sample was placed inside a climatic chamber (Memmert, model HPP 110). The samples were kept at temperatures of 25 °C, 35 °C, and 45 °C, with a constant relative humidity of 65%. The sensor was placed 3 cm into the phantom and allowed to equilibrate for 15 min. This time was enough to ensure that the temperature was uniform across the surrounding sample before taking a reading. In addition, each measurement was performed four times to verify accuracy and reproducibility. The experimental setup for these measurements is shown in
Figure 2.
2.5. Optical Characterization
Optical characterization of the tissue-mimicking phantom across the visible and near-infrared (VIS–NIR) spectral range was carried out using the experimental setup illustrated in
Figure 3. The setup was based on a laser-driven plasma white light source (Energetiq EQ-99-FC, Wilmington, MA, USA), which provides a broadband and spectrally stable emission spanning from 190 nm to 2500 nm, and the reflected optical signal was measured using an optical spectrum analyzer (OSA) (Yokogawa AQ6373, Tokyo, Japan). Both the light source and the OSA were coupled to the measurement system through a bifurcated fiber-optic probe (Ocean Optics, QR200-7-UV-VIS). The probe was positioned at an incidence angle of 45° relative to the phantom surface, as is illustrated in
Figure 3. This angular configuration was selected to minimize specular reflections [
29].
The VIS–NIR measurements were performed on phantom slabs with dimensions of 8 × 8 × 1 cm. In this experiment, the fiber-optic probe was positioned at different locations randomly distributed across the sample surface. The objective of this analysis was to assess the macroscopic spatial homogeneity of the fabricated phantom using independent measurement points. In this way, the study aimed to identify potential localized variations associated with mixing, gelation, or solvent redistribution processes. In addition, a custom-designed mechanical holder was used to fix both the probe orientation and the probe–sample distance to ensure repeatability and spatial consistency.
Spectral measurements were conducted using a linear wavelength scale with a nominal resolution of 5 nm, covering the spectral range from 350 nm to 1200 nm with a wavelength increment of 1 nm. Before phantom measurements, the system was calibrated using a certified diffuse reflectance standard (USRS-99-010, Labsphere, Palo Alto, CA, USA). Following calibration, diffuse reflectance spectra of the tissue-mimicking phantom samples were acquired under identical experimental conditions.
2.6. Microwave Dielectric Spectroscopy
The dielectric response of the phantom was characterized using a coplanar waveguide (CPW) transmission-line approach, which provides a robust and broadband method for extracting complex permittivity from calibrated scattering parameters. The CPW was fabricated on Rogers RO3003 laminate (relative permittivity
, loss tangent
), with a substrate thickness of
, a center conductor width of
, and a slot gap of
. A uniform phantom slice with a thickness of
was carefully prepared and placed directly on the CPW as a superstrate (
Figure 4). Two-port measurements were performed using a vector network analyzer (ZNA26, Rohde & Schwarz, Munich, Germany) over the frequency range 1.5–4.0 GHz. A full two-port SOLT calibration was applied at the probe reference planes prior to data acquisition. Each measurement was repeated seven times under identical conditions to assess repeatability.
From the calibrated
S-parameters and the known physical length of the CPW section, the complex propagation constant
was extracted, where
and
denote the attenuation and phase constants, respectively. Under the quasi-TEM approximation, the phase constant is related to the effective permittivity
as
where
c is the speed of light in free space, and
is the efective relative permittivity of the CPW. To mitigate constant phase offsets associated with calibration and connector effects,
was evaluated from the frequency derivative of
rather than from its absolute value.
The relative permittivity of the superstrate (
) is determined based on its relationship with the effective permittivity of the CPW [
30], as expressed in Equation (
2).
where
and
are geometry-dependent filling factors. These coefficients depend on the CPW dimensions and the thicknesses of the involved layers and were computed following the formulation reported in [
30].
Dielectric losses in the phantom were quantified from the attenuation constant by isolating the dielectric contribution associated with the superstrate. Within the quasi-TEM approximation, the dielectric attenuation term can be written as:
where
is the free-space wavelength and
is the loss tangent of the phantom material. The effective conductivity of the phantom was finally obtained from
using the standard relation
.
2.7. Reflection-Mode THz-TDS Setup and Acquisition Protocol
The experimental configuration illustrated in
Figure 5 schematically represents the reflection-mode THz-TDS arrangement used in this work for the characterization of skin-mimicking phantom samples. Measurements were performed with a benchtop THz spectrometer (TDS1008, BATOP GmbH, Jena, Germany) based on internal photoconductive antennas (Tx/Rx) driven by a femtosecond laser source at 780 nm (pulse duration 100 fs, repetition rate 50–100 MHz, average power 40 mW). According to the instrument specifications, the TDS1008 provides a bandwidth of 0.05–4.5 THz, dynamic range of ≥85 dB, a maximum scan range of up to 500 ps, and a spectral resolution of 2 GHz at the maximum scan range.
All measurements were carried out in reflection mode using the SHR (Sample Holder Reflection) mount installed in the internal sample compartment. In this geometry, the THz beam emitted by the Tx antenna is redirected by an internal reflective plate toward the sample plane under oblique incidence; in the SHR configuration, the angle of incidence is 30° by default. The field reflected by the sample is redirected back into the detection path and collected by the Rx antenna, yielding the reflected waveform in the time domain.
A three-step acquisition sequence was used to ensure a consistent reference across the complete measurement campaign. First, a baseline trace was recorded by closing the spectrometer’s metallic shutter to block the THz radiation from reaching the Rx antenna; this baseline captures the system background under identical acquisition settings. Next, under the same conditions, the reference signal was acquired by opening the shutter and placing a protected silver mirror (PF30-03-P01, Thorlabs, Newton, NJ, USA) at the sample position, using the sample area of the SHR mount to reflect the THz beam. The mirror was positioned in the same plane as the phantom samples. Finally, the phantom was placed in the same plane after the mirror was removed. Two phantom slabs with lateral dimensions of 2.7 cm × 2.7 cm and thicknesses of 0.5 cm and 0.3 cm were evaluated. The reference trace acquired with the mirror was used as a fixed reference for all subsequent measurements, since no changes were made to the spectrometer configuration or acquisition parameters.
Time-domain signals were acquired by scanning the optical delay line within a restricted temporal window around the reflected pulse. For all reflection measurements, the acquisition parameters were kept constant: a stepwidth of 0.050 ps, a start position of 245 ps, an end position of 290 ps, and a time constant of 1.5 s. Here, the start and end positions define a 45-ps temporal window selected to capture the main reflected pulse and its immediate oscillations without extending the scan unnecessarily, which would increase acquisition time. The stepwidth sets the temporal sampling of the waveform, while the time constant corresponds to the effective integration time at each delay position, selected to improve measurement stability and signal-to-noise ratio for weak reflections from lossy, water-rich samples. The time constant (1.5 s) and fixed delay sampling (0.05 ps) improve short-term stability for weak reflections, so peak-to-peak and RMS metrics are used here as robust relative observables to track temporal evolution under constant measurement conditions.
To evaluate the time-dependent evolution of the phantom response associated with drying, the spectrometer was programmed to acquire one measurement every 30 min over 7.5 h, resulting in 15 consecutive measurements per sample. After completing the sequence for the first thickness, the phantom was replaced with the second slab, and the same protocol was repeated under identical acquisition settings.
The measurement data were exported using the T3DS software, which stores the baseline, reference, and sample traces in a single dataset together with the acquisition metadata. For consistency, basic preprocessing consisted of removing the baseline contribution from the reference and sample traces and subsequently comparing the phantom response with respect to the mirror reference under unchanged acquisition conditions. This methodological strategy was adopted to minimize systematic variability and to ensure that the observed changes across repeated scans predominantly reflect temporal evolution of the phantom rather than changes in the experimental settings.
2.8. Microwave Heating and Thermal Validation Setup
Microwave thermal validation was performed to evaluate the thermophysical stability of the tissue-mimicking phantom under controlled RF exposure. The experimental setup, shown in
Figure 6, combines continuous-wave excitation at 2.45 GHz and non-contact infrared thermography for surface temperature monitoring. This setup and acquisition methodology were previously implemented to study the behavior of organic and inorganic materials [
31].
In the experimental setup, the RF excitation was generated using a USRP platform (National Instruments NI USRP-2922, Austin, TX, USA) operating at 2.45 GHz in continuous-wave mode. Two excitation regimes were used. For low-power thermal stability tests, the USRP output was set to 14 dBm and routed through an external RF power amplifier operated at a nominal gain of 28 dB. For hyperthermia-level experiments, the USRP output was set to 20 dBm and amplified by 33 dB to reach therapeutic temperatures. Moreover, a three-port circulator was incorporated to protect the amplifier from reflected power, with a matched 50 Ω load connected to the isolated port. Microwave energy was delivered to the phantom using a unidirectional antenna positioned parallel to the phantom surface, as schematically illustrated in
Figure 6. The antenna–sample separation was fixed at 5 mm using a mechanical support to maintain consistent electromagnetic coupling. The phantom was placed on a flat plastic-coated holder to reduce parasitic reflections and minimize conductive heat losses.
Surface temperature was monitored using a calibrated infrared thermal camera (Optris PI 450i, Optris GmbH, Berlin, Germany) operating in radiometric mode, which enables pixel-wise absolute temperature measurements. The camera was positioned normal to the phantom surface at a fixed distance of approximately 40 cm. Before the experiments, the camera was calibrated following the manufacturer’s procedure. The surface emissivity was set to 0.98. Ambient reflections were minimized by shielding the experimental area and avoiding high-reflectivity objects within the camera field of view.
Two operating temperature conditions were evaluated. In the low-power regime, the applied excitation resulted in a stable surface temperature of approximately 26 °C, corresponding to near-ambient conditions. In the hyperthermia-level regime, the excitation power was gradually increased until the phantom reached an initial steady-state surface temperature of 44.6 °C, representative of therapeutic hyperthermia. Once thermal stabilization was reached, infrared images were acquired at predefined time intervals during continuous excitation. At each time point, multiple consecutive thermal frames were recorded under identical conditions to quantify short-term variability and compute mean surface temperature values and associated standard deviations. To avoid cumulative heating or dehydration effects, consecutive experiments were performed on previously unexposed regions of the phantom by rotating the sample between tests.
2.9. Multivariate Analysis
To evaluate the spatial reproducibility of the VIS–NIR apparent absorbance spectra, principal component analysis (PCA) was applied as a multivariate analysis tool. In this case, each spectrum can be considered as a vector containing absorbance values at different wavelengths. When spectra are measured at several positions on the phantom surface, the full dataset can be organized as a matrix in which each row corresponds to one measurement position and each column corresponds to a wavelength.
Before applying PCA, the dataset was mean-centered in order to analyze variations with respect to the average spectral response. This was performed by subtracting the mean spectrum from each individual spectrum:
where
X represents the original spectral dataset and
is the average spectrum calculated across all positions. This step ensures that the analysis focuses on differences among measurement locations rather than on absolute absorbance levels. No scaling process was implemented because all variables correspond to absorbance values expressed in the same units. PCA determines a new set of orthogonal components, called principal components (PCs), that describe the main directions of variability within the dataset. The first principal component (PC1) accounts for the largest fraction of the total variance, while subsequent components capture progressively smaller and independent contributions. The PCA analysis was carried out using Python (v3.14.0).
4. Discussion
The purpose of this work was to verify whether the fabricated phantom reproduces the coupled dielectric–thermal behavior of hydrated biological tissue across multiple spectral regimes. The characterization was intentionally multimodal to ensure that composition, dielectric response, optical absorption, and microwave thermal performance remain physically consistent. First, the thermogravimetric analysis confirms that the phantom is dominated by a water-rich phase embedded in a polymeric matrix. The main mass loss below 200 °C corresponds to water evaporation, establishing hydration as the governing factor of its electromagnetic and thermal behavior. This finding is critical to the study because the water content of the phantom influences dipole polarization at microwave frequencies and broadband absorption in the optical and THz regimes. Similarly, water increases the material’s heat storage capacity when subjected to the hyperthermia applicator.
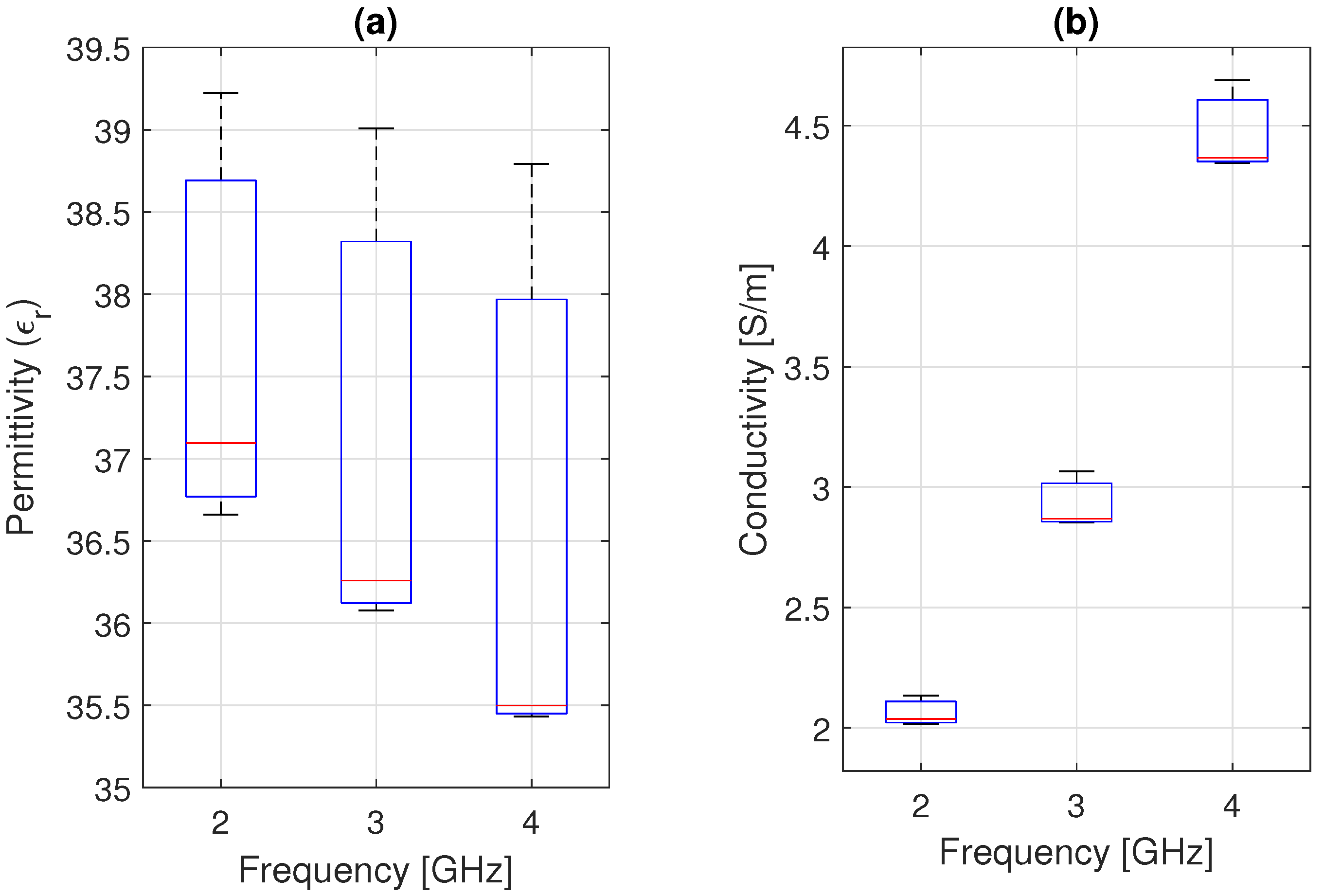
The microwave dielectric comparison presented in
Table 5 reveals a more discriminative and physically informative contrast. The close match in electrical conductivity with the reference phantom [
8] suggests that the ionic concentration and connectivity within the water-rich matrix were accurately replicated. This demonstrates that charge transport processes, dominated by free-ion mobility, are resilient to modest hydration fluctuations and can be successfully regulated by controlling the NaCl content. The relative permittivity differs significantly from both the reference phantom and database values [
38,
39]. This pattern reflects the inherent sensitivity of dipolar polarization to the fraction, condition, and spatial distribution of bound and free water, as well as polymer-water interactions within the solid matrix. As a result, tiny changes in hydration or microstructural organization have a disproportionate effect on permittivity while keeping conductivity relatively steady. This asymmetric sensitivity between permittivity and conductivity is not a limitation of the phantom but rather a physically expected feature of hydrated polymer systems. It explains the simultaneous agreement in
and deviation in
observed in
Table 5 and is fully consistent with the monotonic frequency dispersion reported in the microwave characterization. From an electromagnetic perspective, this dielectric contrast governs near-field distribution, energy confinement, and microwave power deposition, which are central to hyperthermia and SAR validation. These same mechanisms form the physical basis for a wide range of microwave sensing and characterization techniques, in which materials with dielectric properties different from those of air induce measurable field perturbations. Such near-field perturbations have been extensively exploited in resonant and non-resonant microwave structures to induce frequency shifts and characterize dielectric variations in biological tissues and tissue-mimicking materials [
38,
39,
40,
41,
42].
On the other hand, the VIS–NIR optical characterization shows a smooth broadband spectral response with limited spatial variability. The coefficient-of-variation analysis and PCA confirm that variability is dominated by global intensity scaling rather than spectral reshaping, indicating macroscopic homogeneity. Importantly, the integrated NIR absorbance follows the expected water-dominant baseline. While no explicit absorption model was fitted, the spectral behavior is consistent with the high water fraction identified by TGA. In hydrated media, overtone and combination bands of O–H vibrations dominate the NIR region; therefore, the broadband increase toward longer wavelengths is physically coherent with the measured water content.
In addition, the THz measurements provide a higher sensitivity probe of hydration dynamics. Strong broadband attenuation is observed, consistent with water-dominated dielectric relaxation in this frequency range. Time-lapse reflection experiments reveal a progressive temporal shift of the dominant pulse, more pronounced in thinner slabs. This behavior indicates a gradual modification of the effective propagation conditions within the phantom during repeated acquisition. Given the strong dependence of THz absorption and dispersion on water content, the magnitude of attenuation and its temporal sensitivity are consistent with the quantified water fraction obtained from TGA. In water-rich systems, even minor hydration variations produce measurable changes in THz absorption and effective time-of-flight. Therefore, the observed THz drift is attributed to gradual moisture redistribution rather than structural instability of the phantom matrix.
Microwave thermal validation further supports this interpretation. Under low-power excitation, the phantom maintains a stable surface temperature over prolonged periods, demonstrating that RF exposure at ambient conditions does not induce measurable hydration changes or structural alteration. This confirms that the dielectric and thermophysical properties remain invariant under baseline electromagnetic loading. In contrast, under hyperthermia-level excitation, a gradual decrease in surface temperature is observed during sustained exposure. Within the first 30–40 min, however, the temperature remains close to the initial steady state, indicating that the material maintains operational stability during typical short-duration hyperthermia validation experiments. Under these conditions, prolonged microwave loading in a water-dominant matrix promotes gradual moisture redistribution and partial evaporation, which in turn modifies effective microwave absorption and heat retention. The observed thermal drift, therefore, results from the coupled interaction between dielectric losses, energy deposition, and realistic environmental exchange.
For this reason, the phantom demonstrates macroscopic compositional uniformity and dielectric reproducibility under baseline conditions. However, like hydrated biological tissue, it exhibits moisture-dependent evolution under sustained thermal stress. This response does not invalidate its use; rather, it reflects the intrinsic coupling between hydration, dielectric losses, and heat diffusion that characterizes real soft tissues in hyperthermia scenarios.
5. Conclusions
In this work, a multimodal experimental framework was implemented to assess whether the fabricated phantom reproduces the coupled dielectric–thermal behavior of hydrated muscle tissue across microwave, optical, and THz regimes. Thermogravimetric analysis confirms dominant water content, which governs the dielectric dispersion, broadband absorption, and thermal storage capacity of the material.
Likewise, the fabricated phantom exhibits a relative permittivity of 37.4 and an electrical conductivity of 2.4 S/m at 2.45 GHz. While the conductivity closely matches reference formulations and remains within the range reported in tissue databases, the permittivity is lower than literature muscle values, which typically range between 47 and 52.7. This deviation reflects the known sensitivity of dipolar polarization to hydration state and polymer–water interactions, whereas ionic transport mechanisms remain accurately replicated. From an electromagnetic perspective, the phantom preserves the correct order of magnitude and dispersion behavior required for controlled hyperthermia and SAR validation in the 2.4 GHz ISM band.
Optical and THz analyses further demonstrate macroscopic compositional homogeneity. VIS–NIR measurements show smooth broadband absorption with limited spatial variability, and PCA confirms that spectral differences are governed by global intensity scaling rather than structural distortion. In the THz regime, strong broadband attenuation consistent with a water-rich matrix is observed together with measurable temporal sensitivity to hydration redistribution, highlighting the dynamic role of water in high-frequency electromagnetic response. Finally, the microwave thermal validation establishes both functional reliability and operational limits. Under low-power excitation, stable surface temperatures confirm compositional stability under baseline RF exposure. Under hyperthermia-level excitation, a significant thermal drift of −3.985 °C/h is observed during sustained irradiation. This drift arises from hydration-mediated redistribution under non-adiabatic conditions and represents the principal limitation of the material for prolonged high-power experiments. Nevertheless, this behavior remains physically coherent with the thermodynamic response of hydrated soft tissues subjected to sustained energy deposition. Thus, the proposed phantom reproduces the coupled dielectric–thermal behavior of hydrated muscle tissue at clinically relevant microwave frequencies while explicitly defining the hydration-driven constraints that govern its long-term thermal stability.