One-Step Preparation of High-Purity Sodium Tungstate from Wolframite via Alkali Fusion and the Mechanism of Impurity Directional Migration
Abstract
1. Introduction
- A novel low-temperature alkali fusion synergistic phase control method specifically targeting wolframite was proposed.
- The directional migration and fixation mechanisms of impurity elements were elucidated.
- The discharge of high-salinity wastewater inherent in conventional processes was avoided.
2. Experimental Methods
2.1. Materials
2.2. Experiment Procedure
2.2.1. Conventional Alkali Fusion
2.2.2. Alkali Fusion with Phase Separation
2.3. Analysis Methods
3. Results and Discussion
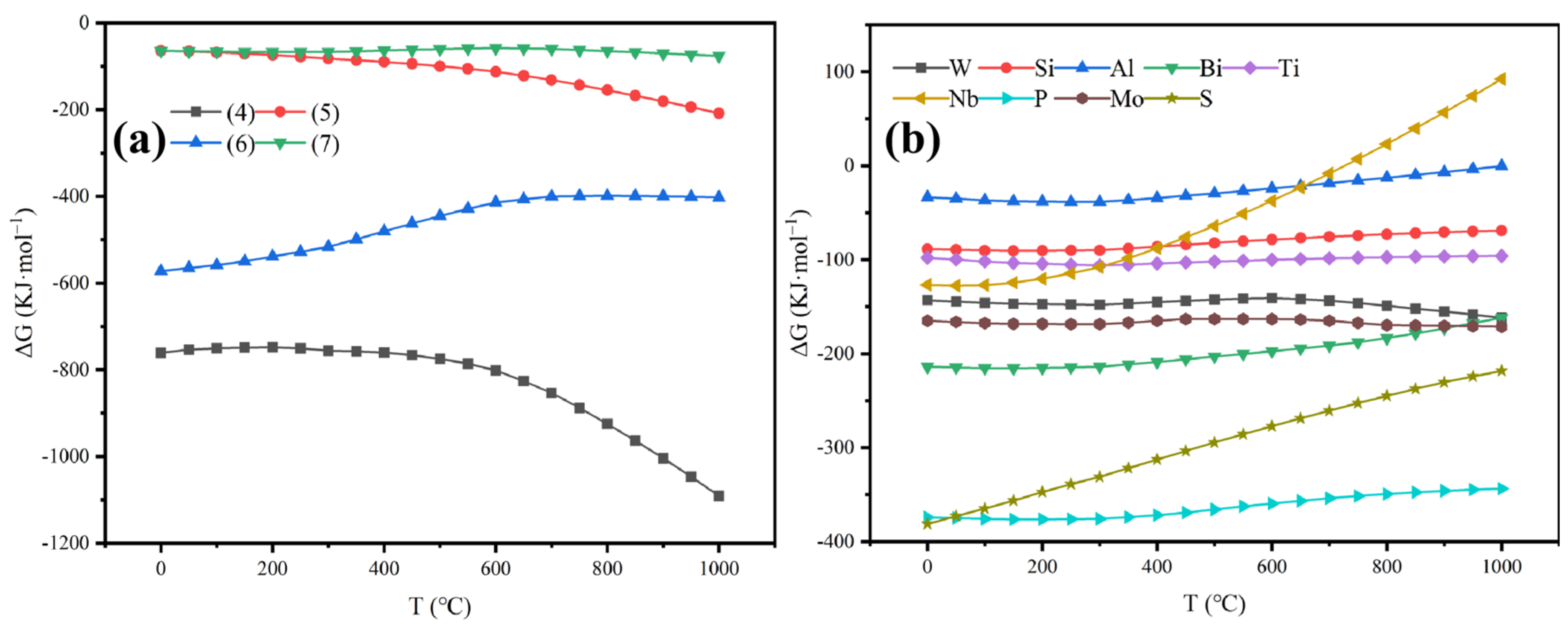
3.1. Thermodynamic Calculations
3.1.1. Thermodynamic Calculations for Wolframite Reactions
3.1.2. Thermodynamic Calculation of Phase Diagram
3.2. Alkali Fusion Process
3.2.1. Effect of NaOH Stoichiometric Ratio
3.2.2. Effect of Alkali Fusion Temperature
3.2.3. Effect of Holding Time
3.3. Phase Separation of Sodium Tungstate at Moderate Temperature
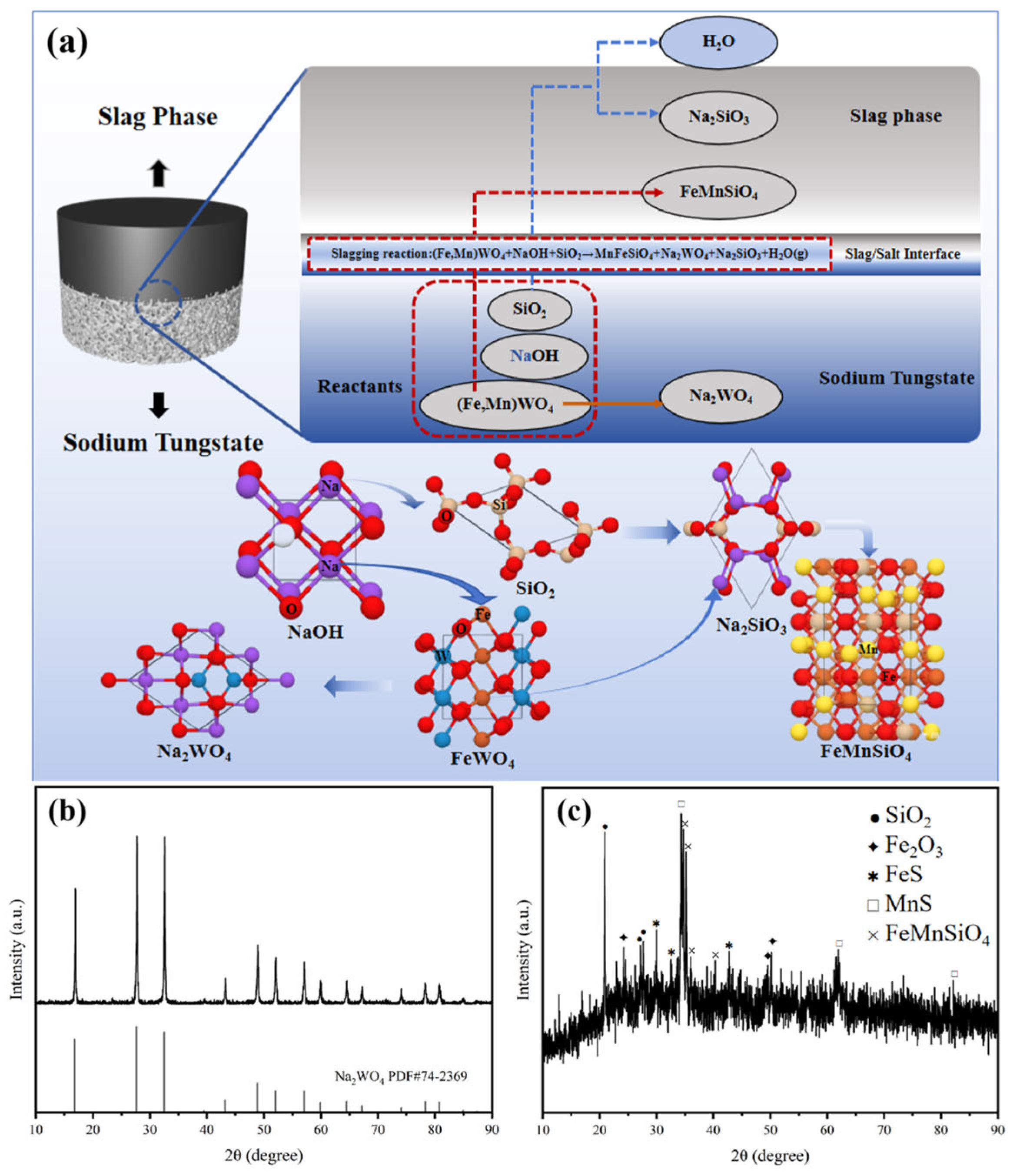
3.3.1. Slag–Salt Separation Process
3.3.2. Trends in Impurity Elements in Wolframite
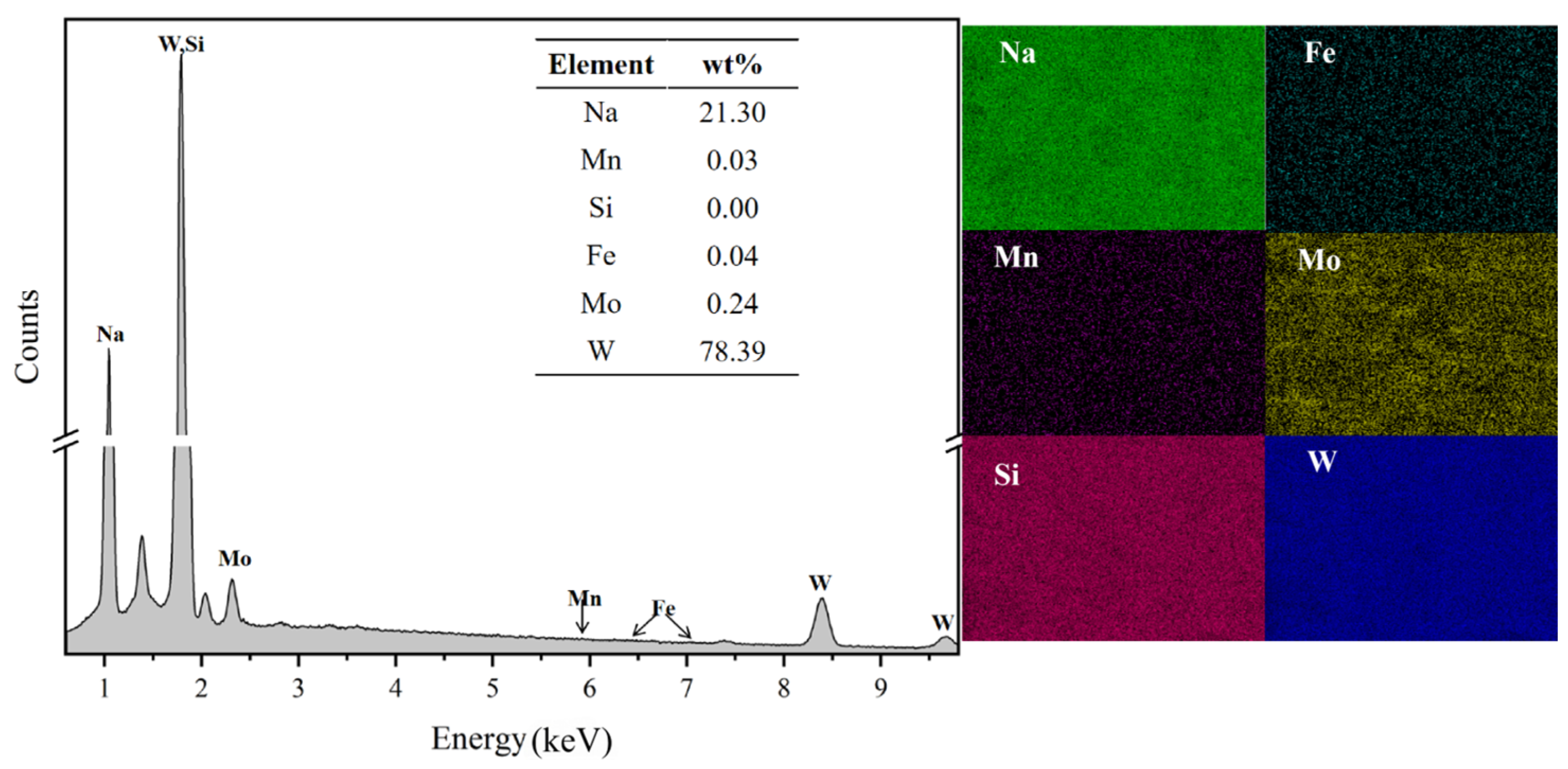
3.3.3. Product Characterization
3.4. Summary and Evaluation of Process Advantages
3.5. Preliminary Assessment for Scale-Up and Industrial Application
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Li, T.-X.; Miao, J.-W.; Guo, E.-Y.; Huang, H.; Wang, J.; Lu, Y.-P.; Wang, T.-M.; Cao, Z.-Q.; Li, T.-J. Tungsten-containing high-entropy alloys: A focused review of manufacturing routes, phase selection, mechanical properties, and irradiation resistance properties. Tungsten 2021, 3, 181–196. [Google Scholar] [CrossRef]
- Wang, X.; Huang, H.; Shi, J.; Xu, H.-Y.; Meng, D.-Q. Recent progress of tungsten-based high-entropy alloys in nuclear fusion. Tungsten 2021, 3, 143–160. [Google Scholar] [CrossRef]
- Shen, L.; Li, X.; Lindberg, D.; Taskinen, P. Tungsten extractive metallurgy: A review of processes and their challenges for sustainability. Miner. Eng. 2019, 142, 105934. [Google Scholar] [CrossRef]
- Liu, H.; Liu, H.; Nie, C.; Zhang, J.; Steenari, B.-M.; Ekberg, C. Comprehensive treatments of tungsten slags in China: A critical review. J. Environ. Manag. 2020, 270, 110927. [Google Scholar] [CrossRef]
- Han, Z.; Golev, A.; Edraki, M. A Review of Tungsten Resources and Potential Extraction from Mine Waste. Minerals 2021, 11, 701. [Google Scholar] [CrossRef]
- Chen, Y.-L.; Guo, X.-Y.; Wang, Q.-M.; Tian, Q.-H.; Huang, S.-B.; Zhang, J.-X. Tungsten and arsenic substance flow analysis of a hydrometallurgical process for tungsten extracting from wolframite. Tungsten 2021, 3, 348–360. [Google Scholar] [CrossRef]
- Ren, H.; Li, J.; Tang, Z.; Zhao, Z.; Chen, X.; Liu, X.; He, L. Sustainable and efficient extracting of tin and tungsten from wolframite—Scheelite mixed ore with high tin content. J. Clean. Prod. 2020, 269, 122282. [Google Scholar] [CrossRef]
- Alguacil, F.J.; Alonso, M. Recovery of Tungsten from Raw and Secondary Materials Using Hydrometallurgical Processing. Metals 2025, 15, 799. [Google Scholar] [CrossRef]
- Shen, L.; Li, X.; Zhou, Q.; Peng, Z.; Liu, G.; Qi, T.; Taskinen, P. Kinetics of Scheelite Conversion in Sulfuric Acid. JOM 2018, 70, 2499–2504. [Google Scholar] [CrossRef]
- Yin, C.; Ji, L.; Chen, X.; Liu, X.; Zhao, Z. Efficient leaching of scheelite in sulfuric acid and hydrogen peroxide solution. Hydrometallurgy 2020, 192, 105292. [Google Scholar] [CrossRef]
- Shen, L.; Li, X.; Zhou, Q.; Peng, Z.; Liu, G.; Qi, T.; Taskinen, P. Wolframite Conversion in Treating a Mixed Wolframite–Scheelite Concentrate by Sulfuric Acid. JOM 2017, 70, 161–167. [Google Scholar] [CrossRef]
- Luo, Y.; Chen, X.; Zhao, Z.; Liu, X.; Li, J.; He, L.; Sun, F. Pressure leaching of wolframite using a sulfuric-phosphoric acid mixture. Miner. Eng. 2021, 169, 106941. [Google Scholar] [CrossRef]
- Liu, D.-G.; Liao, C.-F.; Liu, Y.-X.; Xu, G.-Z.; Zhou, F.; He, B.-X.; Liang, Y. Efficient extracting of tungsten from scheelite via NaOH-SiO2 roasting followed by water leaching. J. Cent. South Univ. 2023, 30, 1856–1864. [Google Scholar] [CrossRef]
- Gaur, R.P.S. Modern hydrometallurgical production methods for tungsten. JOM 2006, 58, 45–49. [Google Scholar] [CrossRef]
- Baimbetov, B.; Moldabayeva, G.; Yeleuliyeva, A.; Jumankulova, S.; Taimassova, A.; Adilzhan, Z.; Baisultanov, R.; Yakob, E.; Serikbayev, V. Prospects of Processing Tungsten Ores from the Akchatau Deposit. Processes 2023, 12, 77. [Google Scholar] [CrossRef]
- Zhang, W.; Chen, Y.; Che, J.; Wang, C.; Ma, B. Green leaching of tungsten from synthetic scheelite with sulfuric acid-hydrogen peroxide solution to prepare tungstic acid. Sep. Purif. Technol. 2020, 241, 116752. [Google Scholar] [CrossRef]
- Xu, L.; Zhao, B. A Fundamental Study on the Preparation of Sodium Tungstate from Wolframite via the Smelting Process. Metals 2024, 14, 299. [Google Scholar] [CrossRef]
- Gong, D.; Zhou, K.; Peng, C.; Li, J.; Chen, W. Sequential extraction of tungsten from scheelite through roasting and alkaline leaching. Miner. Eng. 2019, 132, 238–244. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, Y.; Wu, J.; Shen, C.; Wei, L. Efficient recovery of tungsten from copper tailings using sodium carbonate synergistic pyrometallurgical smelting. J. Environ. Chem. Eng. 2025, 13, 118525. [Google Scholar] [CrossRef]
- Zhao, B.; Su, K.; Ma, X. Experimental Determination of Phase Equilibria in the Na2O-SiO2-WO3 System. Metals 2021, 11, 2014. [Google Scholar] [CrossRef]








| Oxide | WO3 | Fe2O3 | MnO | SiO2 | PbO | CaO | Al2O3 | Bi2O3 | K2O |
|---|---|---|---|---|---|---|---|---|---|
| wt.% | 62.050 | 16.544 | 7.200 | 4.526 | 3.199 | 1.209 | 0.884 | 0.276 | 0.222 |
| Oxide | CuO | Na2O | TiO2 | ZnO | Nb2O5 | P2O5 | ZrO2 | MoO3 | Other |
| wt.% | 0.171 | 0.151 | 0.121 | 0.078 | 0.052 | 0.038 | 0.031 | 0.021 | 3.175 |
| No. | (Fe,Mn)WO4 | NaOH | SiO2 |
|---|---|---|---|
| 1 | 0.0261 mol | 0.15 mol | 0.0222 mol |
| 2 | 0.0261 mol | 0.15 mol | 0.0333 mol |
| 3 | 0.0261 mol | 0.15 mol | 0.0417 mol |
| 4 | 0.0261 mol | 0.15 mol | 0.05 mol |
| 5 | 0.0261 mol | 0.15 mol | 0.0583 mol |
| 6 | 0.0261 mol | 0.15 mol | 0.0667 mol |
| 7 | 0.0261 mol | 0.15 mol | 0.075 mol |
| 8 | 0.0261 mol | 0.15 mol | 0.0833 mol |
| 9 | 0.0261 mol | 0.15 mol | 0.0917 mol |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Bai, H.; Zhang, L.; Xi, X.; Nie, Z. One-Step Preparation of High-Purity Sodium Tungstate from Wolframite via Alkali Fusion and the Mechanism of Impurity Directional Migration. Materials 2026, 19, 932. https://doi.org/10.3390/ma19050932
Bai H, Zhang L, Xi X, Nie Z. One-Step Preparation of High-Purity Sodium Tungstate from Wolframite via Alkali Fusion and the Mechanism of Impurity Directional Migration. Materials. 2026; 19(5):932. https://doi.org/10.3390/ma19050932
Chicago/Turabian StyleBai, Hailong, Liwen Zhang, Xiaoli Xi, and Zuoren Nie. 2026. "One-Step Preparation of High-Purity Sodium Tungstate from Wolframite via Alkali Fusion and the Mechanism of Impurity Directional Migration" Materials 19, no. 5: 932. https://doi.org/10.3390/ma19050932
APA StyleBai, H., Zhang, L., Xi, X., & Nie, Z. (2026). One-Step Preparation of High-Purity Sodium Tungstate from Wolframite via Alkali Fusion and the Mechanism of Impurity Directional Migration. Materials, 19(5), 932. https://doi.org/10.3390/ma19050932





