3.2. Initial Permeability
Initial permeability
µi is a key performance indicator for MnZn ferrites, reflecting the material’s ability to respond to an applied magnetic field. To identify processing routes that enhance
µi, an orthogonal analysis was performed on samples produced under an L9 design as a main-effects screening strategy. The results are summarised in
Table 2. For each factor,
K denotes the mean
µi at a given level (
K1,
K2,
K3 for temperature levels), and
R is the range for that factor, defined as the maximum minus the minimum level average (
R = max
Ki − min
Ki). The
R value, therefore, indicates the influence of a factor on
µi, whereas a larger
K at a specific level identifies the preferred level for that factor within the investigated parameter window. In
Table 2, the relation R(B) > R(A) > R(C) indicates that factor B, namely the oxygen partial pressure, exerts the greatest impact on the initial permeability
µi. This ranking is further supported by a main-effects ANOVA of the L9 dataset (
Supplementary Information, Table S3), which confirms that oxygen partial pressure contributes most strongly to
µi, followed by sintering temperature, while holding time has the smallest contribution within the investigated window. Meanwhile, the
K value reaches its maximum when factor A is at level 1, factor B is at level 3, and factor C is at level 3. Accordingly, it can be inferred that MnZn ferrites are more likely to achieve a higher
µi under the A1B3C3 condition, corresponding to a sintering temperature of 1250 °C, an oxygen partial pressure of 3.5%, and a holding time of 4 h. This inference is consistent with the experimental result for sample E3, which exhibited a remarkably high initial permeability of 3453 compared with other samples in the orthogonal experiment, supporting the validity of the orthogonal screening. The L9 design primarily captures main effects, while a more rigorous quantification of interaction and statistical uncertainty would require additional runs and repeats. Such temperature–oxygen interactions may arise because temperature accelerates defect equilibration and diffusion kinetics, thereby strengthening or weakening the influence of oxygen partial pressure on oxygen non-stoichiometry, grain-boundary resistivity, and ultimately permeability and power loss.
In summary, the relative importance of the three factors affecting µi can be ranked as: oxygen partial pressure > sintering temperature > holding time. Consistent with this ranking, µi reached a maximum at an intermediate oxygen partial pressure (3.5% ) compared with 2% or 5% . In MnZn spinel ferrites, the sintering atmosphere may modify the defect chemistry and redox equilibrium, which can affect magnetic softness through changes in magnetocrystalline anisotropy, magnetostriction and internal stress. However, because Ms, K1, and the Fe2+/Fe3+ ratio were not quantitatively determined for all E1–E9 samples, the atmosphere-related interactions in this work are qualitative, and we therefore focus on experimentally observable structure–property correlations.
Except for oxygen partial pressure, the sintering temperature also plays an essential role in determining the initial permeability (
µi) of MnZn ferrites. Within the investigated L9 parameter window, the
µi of samples sintered at 1250 °C tended to be higher than that of samples sintered at 1280 °C and 1310 °C. This trend can be attributed to the fact that the sintering temperature not only provides the energy required for solid-phase reactions but also influences the microstructure of MnZn ferrites, including grain morphology, grain size, and density. As shown in
Figure 5, the samples sintered at 1250 °C exhibited a more uniform microstructure with clearer grain boundaries compared with those sintered at 1280 °C and 1310 °C, which is favourable for domain-wall motion and can reduce pinning sites [
23]. Since the initial permeability is the combined contribution of domain wall movement and spin rotation, expressed as
, such uniformity and reduced defects are conducive to enhancing
µi. However, as the sintering temperature further increased, grain coarsening occurred, leading to the formation of abnormal grains (
Figure 6) and a broader grain size distribution. This microstructural heterogeneity is expected to increase pinning and internal stress, consistent with reduced magnetic softness (e.g., higher coercivity) and therefore lower
µi [
24]. Therefore, an appropriately lower sintering temperature (e.g., 1250 °C) is beneficial for achieving higher
µi in the present study.
3.3. Power Loss
To minimise the power loss (
Pcv) under the targeted operating conditions, an orthogonal analysis was carried out using the measured
Pcv values at 100 kHz/200 mT (
Table 3) and 500 kHz/50 mT (
Table S4) for the L9 sample set. The analysis procedure follows that used for
µi, where
K denotes the mean response at each factor level, and
R represents the range (
R = max
Ki − min
Ki). A larger
R value indicates that the corresponding factor exerts a stronger influence on
Pcv within the investigated parameter window. The results showed that the oxygen partial pressure was the most significant factor affecting
Pcv during sintering, followed by the sintering temperature, while the holding time had the least effect. This ranking is further supported by a main-effects ANOVA of the L9 dataset at 100 kHz/200 mT (
Supplementary Information, Table S5), which confirms that oxygen partial pressure and temperature contribute more strongly to
Pcv than holding time within the investigated window. This ranking differs from that for
µi, reflecting the different sensitivities of permeability and loss to processing variables and the trade-off between performance metrics.
For
Pcv, the optimum level corresponds to the minimum
K value since the experimental objective was to minimise power loss. This criterion was also applicable to the
Pcv data at 500 kHz/50 mT. Importantly, the minimum-loss sample depends on the excitation condition. The lowest
Pcv at 100 kHz/200 mT was obtained for E4 (1280 °C, 5%
, and 3.5 h), whereas at 500 kHz/50 mT the minimum
Pcv was achieved by E1 (1250 °C, 5%
, and 3 h) (
Table S4). This observation highlights that the optimal sintering condition should be selected based on the targeted operating points and the required balance between
µi and
Pcv. Nevertheless, the orthogonal analysis consistently indicates that a higher oxygen partial pressure (5%
) is favourable for reducing
Pcv within the investigated window.
At the operating condition of this work (100 kHz/200 mT), loss separation was further performed for all samples (E1–E9) to clarify the origin of the variations in the total power loss
Pcv. As shown in
Figure 7,
Pcv is decomposed into the
Ph and
Pe under the same measurement conditions. A key mechanistic outcome is that, at 100 kHz/200 mT, the differences in
Pcv across the L9 set are predominantly
Ph-driven. Specifically, E4 exhibited the minimum
Pcv mainly because
Ph is markedly reduced, whereas
Pe changes only modestly. This
Ph-dominant behaviour is consistent with the microstructural features associated with magnetic softness, where reduced domain-wall pinning (e.g., fewer pores or defects), together with a more uniform grain-size distribution and lower porosity, suppresses irreversible magnetisation dissipation. Overall, the loss-separation provides direct, mechanism-level evidence that the minimum-loss case (E4) is governed primarily by
Ph reduction.
Consistent with the factor ranking, increasing the oxygen partial pressure to 5% provides a robust route to reduce
Pcv in the present processing window. In practice, the sintering atmosphere can influence defect chemistry and oxidation equilibria and thereby affect microstructural development and magnetic softness. These defect-chemistry-related interpretations are qualitative in the present work, as oxygen non-stoichiometry and Fe
2+/Fe
3+ equilibrium were not directly quantified for the full L9 sample set. The present loss-separation result further shows that, at 100 kHz/200 mT, the reduction in
Pcv is mainly driven by a decrease in
Ph, while
Pe plays a secondary role in differentiating the total loss among E1–E9. In addition,
Pe generally correlates with electrical resistivity and characteristic eddy-current path length, which depend on densification and grain size. However, in the current dataset,
Pe is not the dominant contribution at 100 kHz/200 mT.
where
Kh and
Ke are loss-related constants,
f is the excitation frequency,
Bm is the peak flux density,
D is the grain size, and
ρ is the electrical resistivity. At 100 kHz/200 mT,
Ph constitutes a major fraction of
Pcv for these soft ferrites, consistent with the loss-separation results. Therefore, processing routes that improve magnetic softness (e.g., reduced pinning and improved microstructural uniformity) are expected to effectively suppress
Ph.
Another important aspect of the sintering temperature is its impact on Pe through microstructural evolution. As previously discussed, the grain size of MnZn ferrites sintered at 1250 °C was generally smaller than that of samples sintered at higher temperatures, thereby reducing Pe according to Equation (1). However, with increasing sintering temperature, grain growth became more pronounced and abnormal grain growth may occur, which can unfavourably affect both Pe and magnetic softness. In summary, an appropriate combination of oxygen partial pressure and sintering temperature is essential for reducing Pcv under the targeted operating conditions.
Given that holding time shows the smallest
R value in the orthogonal analysis, its effect on
Pcv is secondary within the 3–4 h range investigated here and is therefore not over-interpreted. Additionally, since the orthogonal analysis suggests a preference for a lower temperature level within the investigated window, it raises the question of whether further lowering the sintering temperature would continue to improve performance. To verify the validity of 1250 °C as the optimum sintering temperature, toroidal samples were sintered at 1230 °C, with a holding time of 3.5 h under 5% oxygen partial pressure, and the results are presented in
Table S6. It was observed that both
µi and
Pcv deteriorated when the sintering temperature decreased, which may be attributed to the insufficient thermal driving force for adequate microstructural development and magnetic softness. This result supports the selection of 1250 °C as a practical low-loss temperature within the investigated window, particularly when performance at higher frequency (500 kHz/50 mT) is prioritised.
3.4. Sintering Optimisation
A verification sample was prepared independently from the L9 set using a representative condition from the low-loss window suggested by orthogonal screening (1250 °C, 5%
, and 3.5 h). This condition retains the preferred high oxygen partial pressure for loss reduction and the low-temperature tendency within the investigated window, while the holding time was set at the middle level, given its minor main effect over 3–4 h. The sample exhibits
µi = 2678 and low power losses of 400 mW/cm
3 at 100 kHz/200 mT and 182 mW/cm
3 at 500 kHz/50 mT, supporting the orthogonal conclusions and confirming that this process window provides a favourable balance between permeability and loss over the targeted frequency range. Duplicate preparations of this optimised low-loss condition show good reproducibility, as summarised in
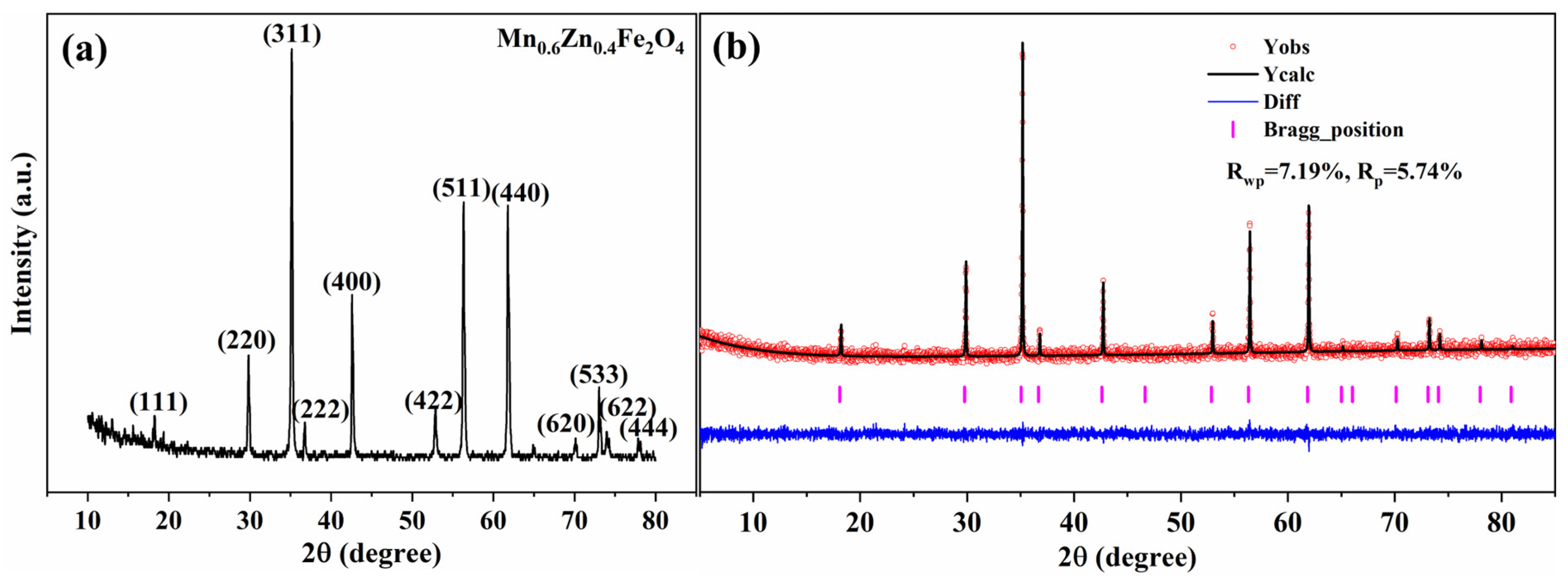
Table S7. The XRD analysis (
Figure 8a) shows that all diffraction peaks can be indexed to the cubic spinel Mn
0.6Zn
0.4Fe
2O
4 phase (PDF#74-2401), and no additional reflections attributable to secondary phases are observed within the detection limit of laboratory XRD. To further strengthen the reliability of the phase and lattice analysis, Rietveld refinement was performed for this representative sample (
Figure 8b). The calculated profile matches the experimental pattern well, with
Rwp = 7.19% and
Rp = 5.74%, a flat difference curve, and correctly located Bragg positions, supporting the formation of a well-crystallised single-phase spinel under the selected sintering scheme. These results provide a solid structural basis for the subsequent discussion of magnetic properties.
To elucidate the microstructural origin of the low-loss behaviour, the optimised sample was examined by SEM, and the grain size was quantified using Nano-Measurer (
Figure 9). The micrographs show a relatively uniform polycrystalline microstructure with clear grain boundaries and no obvious abnormal grain growth. Importantly, the grain-size distribution (
Figure 9c) is narrow and shows a single main peak, giving an average grain size of 9.02 ± 0.25 µm, supporting good microstructural homogeneity under the selected sintering condition. In the frequency range investigated here (100–500 kHz), the total power loss is commonly discussed in terms of the hysteresis- and eddy-current-related contributions, and their relative importance depends on both excitation conditions and microstructural and stress-related factors [
25,
26,
27]. At 100 kHz/200 mT,
Pcv is primarily associated with a decrease in
Ph, whereas the change in
Pe is comparatively less pronounced. The improved grain-size uniformity observed in
Figure 9 is consistent with reduced domain-wall pinning and reduced hysteresis-related dissipation, as reported for fine and homogeneous microstructure in low-loss MnZn ferrites [
25,
28,
29,
30]. In addition, the optimised sample shows a modest increase in electrical resistivity (from 24.692 to 26.647 Ω·m), which is expected to further suppress the eddy-current contribution according to Equation (1). Overall, the SEM observations, together with the resistivity measurement and loss separation, provide an experimentally supported explanation for the reduced
Pcv achieved under the optimised sintering scheme [
31,
32].
Figure 10 shows the
B-
H hysteresis loop of the MnZn ferrites prepared under the optimised sintering scheme. The loop yields a saturation flux density (
Bs) of 500 mT, a remanence (
Br) of 66 mT, and a coercivity (
Hc) of 12 A/m. The low
Hc together with the narrow loop indicates good magnetic softness and a reduced field requirement for magnetisation reversal under alternating excitation. In practical power-magnetic components, a relatively high
Bs is beneficial because it provides higher usable flux swing before approaching saturation, which supports higher power density and improves tolerance to transient over-excitation. The modest
Br is also advantageous for high-frequency operation, as it reduces residual magnetisation and helps mitigate DC-bias sensitivity and waveform distortion, thereby contributing to more stable inductance and lower hysteresis-related dissipation [
33,
34,
35]. For completeness, the hysteresis parameters (
Bs,
Hc, and
Br) of all L9 samples (E1–E9) measured using the same protocol are summarised in
Table S8, allowing comparison across the processing window.
3.5. Sintering Mechanism
Magnetic and loss properties of MnZn ferrites are closely tied to phase evolution, densification, and microstructure development during sintering. The heating stage mainly removes volatile/organic species and advances solid-state reactions, whereas the high temperature holding stage governs densification and grain growth under controlled oxygen partial pressure. The orthogonal screening above identifies oxygen partial pressure as the dominant factor, followed by temperature and then holding time, based on a lateral comparison across the L9 set (E1–E9). However, such a main-effects analysis does not directly reveal how phase formation, densification and microstructure evolve along the sintering process under a given atmosphere-temperature schedule. Therefore, a step-sintering verification was conducted by decomposing the optimised scheme into key temperature/time nodes. This process-path analysis provides mechanistic support for the orthogonal conclusions and helps rationalise why temperature-driven phase formation and densification exert a stronger influence than holding time adjustments within the investigated range. From an implementation perspective, the step-sintering strategy is compatible with industrial programmable furnaces (batch kilns or multi-zone continuous furnaces), as it mainly requires a multi-stage temperature-time profile with controlled atmosphere setpoints. Scale-up would primarily involve ensuring temperature/atmosphere uniformity across larger thermal masses and may require modest re-tuning of ramp/soak times, which can be addressed through pilot trials and standard process monitoring.
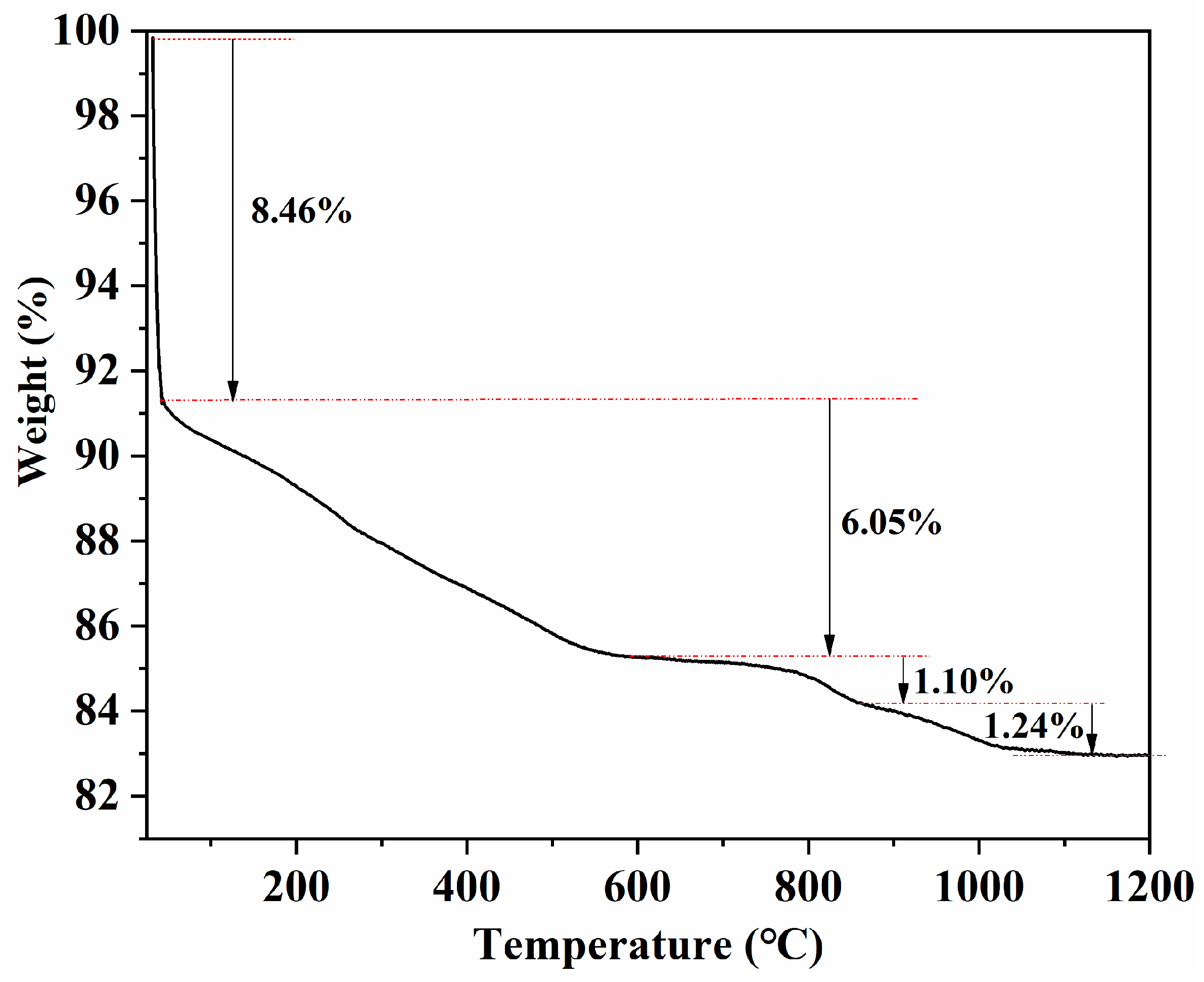
To support the discussion on thermal events and possible reactions during sintering, thermogravimetric analysis was conducted from room temperature to 1200 °C (
Figure 11). The sample exhibits a total mass loss of ~16.85%, which can be divided into four stages as indicated in
Figure 11: an initial rapid loss of ~8.46% at low temperature, followed by a gradual loss of ~6.05% up to ~800 °C, and two minor mass-loss steps of ~1.10% and ~1.24% at higher temperatures. The low-to-intermediate temperature mass loss is mainly associated with the removal/decomposition of volatile components (e.g., adsorbed species and organic residues introduced during granulation/pressing). The minor losses at elevated temperatures may be related to further thermally activated processes, such as oxygen non-stoichiometry adjustment and/or slight volatilisation. Notably, the mass becomes nearly stable above ~1100 °C, suggesting that the major mass-change events are largely completed before entering the final high-temperature sintering window. Although the TGA measurement was limited to 1200 °C and does not fully cover the highest sintering temperature used in this work, the near-constant mass at the upper end of the curve indicates that additional mass change above 1200 °C is expected to be limited.
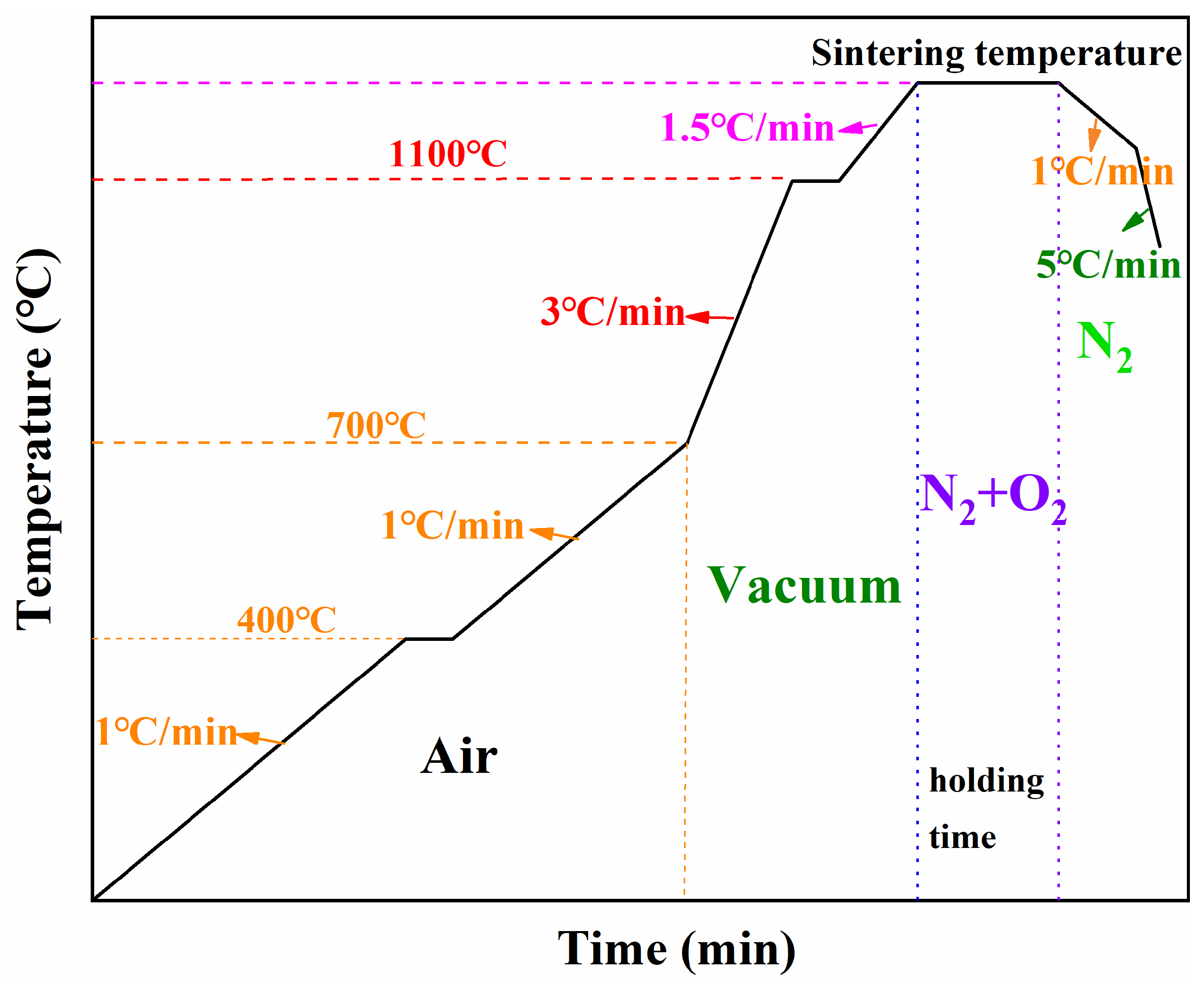
A stepwise sintering experiment was conducted to investigate the microstructural evolution and physicochemical changes throughout the entire sintering process.
Figure 12 shows the optimised sintering scheme used as the reference condition. Green toroidal compacts were heated to 400 °C, 700 °C, 900 °C, 1000 °C, 1100 °C, and 1250 °C as key sampling temperatures, and additional specimens were held at 1250 °C for different times (1 h and 3 h). The corresponding samples were denoted as S-400, S-700, S-900, S-1000, S-1100, S-1250, S-1250-1, and S-1250-3, respectively.
Taking S-400 as an example, the toroidal samples were heated in air at a rate of 1 °C/min to 400 °C. Upon reaching the set temperature, the run was terminated without an isothermal hold, and the samples were allowed to cool naturally to approximately 200 °C and removed from the furnace. This procedure was repeated for each designated sintering stage. The variations in mass loss, shrinkage, density, and initial permeability at each stage were systematically measured and are summarised in
Table 4.
The physical changes during sintering generally involve neck formation and densification, followed by grain growth [
36]. The evolution of particle contacts, sintering necks, pores, and the formation of spinel ferrite grains in MnZn ferrite samples at different sintering temperatures can be clearly observed in the SEM pictures shown in
Figure 13.
Sintering shrinkage (linear shrinkage). The bulk density and porosity of MnZn ferrites gradually improved with increasing linear shrinkage and mass loss. In the early stage of sintering, as shown in
Figure 13 (S-400), the solid particles were only in initial contact, and the pores were widely dispersed. As indicated in
Table 4, physically adsorbed/bound water and organic additives were removed when the temperature increased to about 700 °C, leading to reduced porosity, an increased linear shrinkage rate, and a slight rise in bulk density from 3.370 g/cm
3 to 3.442 g/cm
3.
In the middle stage of sintering, with further temperature increase, the interfaces between particles gradually merged, and the pores progressively closed, as observed in
Figure 13 (S-900, S-1000, and S-1100). The porosity decreased rapidly, while the linear shrinkage rate significantly increased to about 8% (relative to the green compact).
At the late stage of sintering, continued heating caused closed pores to contract further, and the bulk density increased substantially, reaching about 4.8 g/cm
3, indicating an advanced densification state. Overall, the total linear shrinkage of the ferrites exceeded 12%, as listed in
Table 4.
Polycrystalline grain growth. Before sintering, the solid particles in the powders were bonded by binder/adhesive additives to maintain a certain shape. As shown in
Figure 13 (S-400, S-700), during the early stage of sintering, some particles began to form fine crystalline domains, and the interparticle spacing gradually narrowed due to the volatilisation of organic residues and partial solid-phase reactions.
With increasing temperature, these microcrystals progressively merged to form sintering necks, as illustrated in
Figure 13 (S-1100), which is indicative of enhanced mass transport across particle contacts and grain-boundary diffusion. In the later stage of sintering, as the temperature further increased, the neck regions shrank significantly, while the grain size of the spinel crystals increased steadily, and the grain boundaries became more distinct. When the temperature reached approximately 1250 °C, the neck features became much less discernible, and the microstructure appeared uniform and complete, as observed in
Figure 13 (S-1250).
During the holding stage, the residual pores further diminished, and the grains continued to grow, as shown in
Figure 13 (S-1250-1 and S-1250-3). Notably, throughout the entire sintering process, the magnetic properties of the samples gradually emerged, as evidenced by the measured permeability, which can be attributed to the progressive formation of a continuous ferrite spinel network and the accompanying grain growth that reduces domain-wall pinning.
The phase composition of the samples obtained from the step-sintering experiment was identified by XRD, and the results are presented in
Figure 14. At the initial stage, Fe
2O
3, Mn
2O
3, and ZnFe
2O
4 phases were detected, which were nearly identical to those of the raw materials shown in
Figure 1b. Within the detection limit of XRD, no additional crystalline phases were observed at this stage, indicating that the material remained dominated by the pre-reacted/starting oxide components. This phase constitution is consistent with the SEM observation (
Figure 13), where particles are mainly in initial contact with limited neck development.
When the temperature was raised to 700 °C, the same phases were identified; however, the peak intensity of Fe
2O
3 exhibited a slight decrease compared with that at 400 °C. This can be attributed to enhanced ion diffusion at elevated temperatures, which increased the number of ions migrating between components and facilitated the participation of Fe
2O
3 in phase transformation. Previous studies have reported that zinc ferrites (ZnFe
2O
4) are typically formed at around 700 °C via Reaction (2) [
37]. The presence of ZnFe
2O
4 already at early stages in this work is attributed to the pre-firing step used in the commercial powder processing, which can alleviate the abnormal expansion associated with in situ ZnFe
2O
4 formation and promote subsequent ferrite phase development:
With the continuous increase in temperature, the solid-phase reactions proceeded through ion diffusion. Grain growth was promoted, and lattice defects were progressively eliminated, giving rise to a normal spinel structure. At ~1000 °C (S-1000), Mn-Fe spinel-related reflections became evident, indicating the formation of MnFe
2O
4-type ferrite according to Reaction (3), although the presence of residual Fe
2O
3 suggested that the reaction was incomplete owing to insufficient temperature:
When the samples were sintered at 1250 °C, XRD analysis showed that the dominant phase evolved into Mn
1-xZn
xFe
2O
4 spinel, consistent with the formation of a Mn-Zn ferrite solid solution through interdiffusion and spinel incorporation of MnFe
2O
4 and ZnFe
2O
4, as described by Reaction (4) [
38,
39]. With extended sintering time, the diffraction peak intensities became sharper and more intense, indicating improved crystallinity, increased grain size, and reduced porosity and defects. These findings were consistent with the microstructural features observed in the SEM images (
Figure 13), where densification and grain growth become more pronounced at the high-temperature and holding stages.
The EBSD analysis provides quantitative information on grain size and distribution, grain boundary characteristics, crystallographic orientations (texture), and phase identification [
40,
41,
42]. It should be stated that no EBSD results were obtained for the sample sintered at 400 °C, as the specimen was not sufficiently dense and tended to crumble during mechanical polishing, which is a prerequisite for EBSD characterisation. Consistent with the SEM observations (
Figure 15), a remarkable difference in grain size and distribution was observed with increasing sintering temperature. For the sample sintered at 700 °C, the grain size was mainly concentrated below 2 µm. At 1000 °C, most grains remained under 2 µm; however, the fraction of extremely fine grains decreased, with more grains distributed over a broader size range. At 1250 °C, the average grain size increased markedly, with most grains falling within the range of 2–8 µm. During the subsequent holding stage, continuous grain growth occurred, and the disparity between small and large grains became more pronounced. With an extended holding time of 3 h, grains ranging from 5 to 13 µm accounted for a larger proportion as compared with those in the S-1250-1 sample, indicating further coarsening and microstructural homogenisation as sintering proceeds.
Figure 16 illustrates the microstructural evolution of samples subjected to the step-sintering experiment from a complementary perspective. Between 700 °C and 1250 °C, the process involved particle contact, merging, and the subsequent formation of new phases and grains. The microstructure evolved from a disordered state at 700 °C (S-700) to a more uniform configuration at 1250 °C (S-1250), with well-defined grain boundaries and improved grain morphology. This evolution is consistent with curvature-driven grain-boundary migration, where the reduction of total grain-boundary area/curvature provides the driving force for grain growth, leading to an overall decrease in grain-boundary energy. Furthermore, extended holding time promoted the exclusion of pores, as evidenced by the comparison between S-1250-1 and S-1250-3.
The inverse pole figures (
Figure 16b) reveal the grain orientation distribution at different sintering stages. The colour follows the standard IPF colour key, where red and orange grains correspond to the (001) orientation, blue to (111), and green to (101). The orientation in S-700 appeared more complex than in S-1250-3, which is consistent with the higher microstructural heterogeneity at the early stage, compared with the predominantly single-spinel ferrite microstructure in S-1250-3. Additionally, the pole figures (
Figure 16c) show no obvious symmetry, and the maximum intensity values were below four, suggesting weak texture and predominantly random grain orientations, typical of polycrystalline ferrites [
43].
Figure 17 presents the misorientation angle distribution of samples sintered at different temperatures (S-700, S-1000, S-1250, S-1250-1, and S-1250-3). The samples at the intermediate stages (S-700 and S-1000) exhibited a high proportion of low-angle grain boundaries, with a pronounced peak below 10°, accompanied by several high-angle grain boundaries with peaks in the range of 40–55°. The predominance of low-angle grain boundaries at S-700/S-1000 is more reasonably associated with a sub-grain/dislocation-rich microstructure, rather than being directly assigned to a specific residual phase. Such low-angle boundaries are commonly related to dislocation arrays and therefore reflect higher stored strain energy. With increasing temperature, recovery and grain-boundary migration can reduce dislocation density and internal stress, thereby lowering the total interfacial energy and promoting microstructural stabilisation. Conversely, the existence of high-angle grain boundaries indicates the development of more mature grain boundaries as the ferrite microstructure progressively forms and coarsens, which agrees with the phase evolution identified by XRD in
Figure 14 and the microstructural observations in
Figure 16 (especially the IPF maps in
Figure 16b, where S-700 and S-1000 are finer and more fragmented, while S-1250-3 exhibits markedly coarser grains) [
44].
At higher temperatures, ferrite grains gradually formed, and similar misorientation angle profiles were observed for S-1250, S-1250-1, and S-1250-3, all showing strong peaks in the 40–55° range. This similarity implies that the phase compositions of these samples were essentially the same. More specifically, the consistent dominance of the 40–55° HAGB peak suggests that the grain-boundary character distribution becomes relatively stable once the predominant ferrite spinel microstructure is established. However, during the holding stage, grain growth, pore elimination and microstructural coarsening occurred to further stabilise the system. As a result, variations in grain size, grain boundary characteristics, and morphology were evident among S-1250, S-1250-1, and S-1250-3, which explains why the curves were not completely identical. To complement the above microstructural evidence, grain-growth kinetics were further evaluated using the TPRE model [
45,
46]. The fit yields n ≈ 1.81 and an apparent Q ≈ 200 kJ/mol [
47,
48], which are reported in the
Supplementary Information as preliminary estimates due to the limited data points.