3.1. Phase Composition and Surface Morphology of MgAlTiVFeCo Powder
Figure 1 shows the XRD patterns of the alloy powders after different milling times. As seen in
Figure 1, diffraction peaks corresponding to Al
12Mg
17 from the MgAl alloy powder, as well as Al, Ti, V, Fe, and Co, are all clearly visible in the unmilled powder. After 20 h of milling, the intensity of all diffraction peaks significantly decreased, and a BCC phase was formed. With increasing milling time, the diffraction peaks of the BCC phase gradually broadened. Finally, after 100 h of milling, all other peaks disappeared, and a single BCC solid solution phase was formed. The corresponding crystallographic plane indices are (110), (200), and (211).
The crystallite size was calculated using Scherrer’s formula:
where
D is the crystallite size, λ is the X-ray wavelength,
β is the full width at half maximum (FWHM) of the diffraction peak, and 2
θ is the diffraction angle. Assuming that microstrain is neglected, the calculated crystallite sizes of the alloy powders are summarized in
Table 1. As the milling time increases, the crystallite size gradually decreases. The crystallite size corresponding to the (110) plane of the powder milled for 100 h is 17.66 nm.
It should be noted that the Scherrer formula does not account for the contribution of microstrain to diffraction peak broadening. Given that the high-energy ball milling process introduces a substantial number of crystal defects, the Williamson–Hall method was further employed to analyze the diffraction data of the powders ball-milled for 100 h. This method is based on the following equation:
where
ε represents the microstrain.
The results indicate that both significant grain refinement and microstrain coexist in the powders, with an estimated microstrain value of approximately 0.38%. After separating the strain contribution, the corrected crystallite size is about 22 nm. The values derived from the Scherrer formula in
Table 1 can be regarded as lower-bound estimates of the crystallite size, whereas the evolutionary trend that “the crystallite size decreases monotonically with ball-milling time” remains valid regardless of the analytical model used.
Figure 2 shows the microscopic morphology of the alloy powders after different ball-milling durations. With increasing milling time, significant changes in the morphology, size, and uniformity of the powders are observed. After 20 h of milling (
Figure 2a), the powder particles are relatively large, exhibit irregular shapes with numerous edges and flaky structures, indicating that the powder is still in the initial stage of fracture and plastic deformation. After 40 h of milling (
Figure 2b), the particle size decreases noticeably, edges become rounded, and some particles undergo cold-welding and agglomeration, leading to a more uniform morphology. Upon further milling to 80 h (
Figure 2c), the powder is further refined, the particle-size distribution becomes narrower, the shapes approach equiaxed, and agglomeration is somewhat alleviated, reflecting the continuous energy accumulation and structural evolution during the milling process. When the milling time is extended to 100 h (
Figure 2d), the powder exhibits a more homogeneous fine-particle morphology with clear particle boundaries and reduced agglomeration, demonstrating that prolonged milling promotes sufficient refinement and homogenization of the powder, which is beneficial for subsequent densification and the formation of alloy phases [
20].
Figure 3 shows the particle-size distribution and elemental mapping of the alloy powders ball-milled for 100 h. The particle-size distribution histogram reveals a relatively broad unimodal profile, with the main distribution ranging from 0.1 μm to 45 μm, along with a small fraction of coarse particles exceeding 50 μm. This distribution indicates that after prolonged milling for 100 h, a dynamic equilibrium has been established between powder refinement and particle cold-welding/agglomeration. The median particle size (D50 = 11.10 μm) is expected to be significantly reduced compared to the initial mixed powders, yet complete nanoscale refinement was not achieved. This can be attributed to the high work-hardening rate of the high-entropy alloy system, as well as the coexistence of ductile components (e.g., Al, Fe) and hard phases (e.g., oxides formed by O with Mg and V introduced during long-term milling). These factors enhance the cold-welding tendency of particles after reaching a certain size, thereby hindering further homogeneous refinement. The elemental mapping displays a uniform distribution of all components, demonstrating that after 100 h of high-energy ball milling, thorough mechanical alloying has been achieved, with extensive inter-diffusioninterdiffusion among the elements, leading to the preliminary formation of a compositionally homogeneous solid-solution precursor.
To analyze the evolution of oxygen introduced during mechanical alloying in the subsequent sintering process, energy-dispersive X-ray spectroscopy (EDS) was employed to perform statistical point analysis of the oxygen content in the 100 h ball-milled powder and the bulk samples sintered at different temperatures. The average oxygen content (in atomic percent, at%) for each sample was calculated based on multiple independent measurements, as presented in
Table 2. It should be noted that EDS provides semi-quantitative data for light elements such as oxygen. Nevertheless, compared with the as-milled powder, the average bulk oxygen content of all sintered samples decreased significantly, indicating redistribution or partial loss of oxygen during sintering. Within the sintering temperature range of 850 °C to 1000 °C, the oxygen content of the samples did not exhibit a simple monotonic variation. This non-monotonic trend suggests that sintering temperature not only influences the diffusion and reaction rates of oxygen but may also complexly affect the existing forms and distribution of oxygen by altering the densification behavior and phase composition of the alloy.
3.2. Phase Composition of the MgAlTiVFeCo High-Entropy Alloy
Figure 4 shows the XRD patterns of the alloy powder milled for 100 h after sintering at different temperatures. As shown in
Figure 4, the position and profile of the BCC phase diffraction peaks remain consistent across samples sintered at different temperatures. However, for the LW1000 sample, the intensity of the main BCC phase diffraction peak increases and the peaks become sharper, indicating an increase in grain size and more complete crystallinity at the 1000 °C sintering temperature. Compared to the phase structure of the as-milled powder, the sintered samples exhibit the precipitation of secondary phases. This demonstrates that the nanocrystalline precursor powder formed via mechanical alloying successfully crystallized during the rapid SPS process, resulting in a high-entropy alloy solid solution with a BCC matrix. By comparison with standard PDF cards, the secondary phases are identified as various spinel-type oxides (MgV
2O
4 (PDF#65-3109), MgAl
2O
4 (PDF#82-2424), FeAl
2O
4 (PDF#82-0584), AlV
2O
4 (PDF#77-2131)), oxides (V
3O
4 (PDF#34-0615), TiO (PDF#08-0117)), and an ilmenite-type oxide (FeTiO
3 (PDF#75-0519)). The presence of oxides in the alloy can be attributed to two main factors. First, during the weighing process, the raw materials inevitably came into contact with atmospheric oxygen, leading to the formation of a thin oxide film on the surface of some elemental powders. Second, during high-energy ball milling, the powder particles underwent severe plastic deformation and friction both on the surface and internally. This accelerated elemental diffusion, providing the driving force for oxide precipitation, while the defects and stresses generated during mechanical alloying supplied nucleation sites, thereby promoting oxide formation [
21].
Studies have shown that magnesium vanadium spinel (MgV
2O
4) and magnesium aluminate spinel (MgAl
2O
4) can form at relatively low temperatures, whereas spinels such as AlV
2O
4 and FeAl
2O
4, as well as FeTiO
3, require higher temperatures to precipitate stably. As shown in
Figure 4, with increasing sintering temperature, the diffraction peak intensities of the MgV
2O
4 and MgAl
2O
4 spinel phases increase significantly. This indicates that at lower temperatures, sintering primarily activates short-range diffusion at the powder surfaces, allowing the pre-formed Mg, Al, and V oxides from the milling process to undergo complex reactions, crystallize, and grow. However, when the sintering temperature rises to 1000 °C, phases such as AlV
2O
4, FeAl
2O
4, and FeTiO
3 precipitate. This is attributed to the fact that, within the severe lattice distortion characteristic of high-entropy alloys, sufficiently high temperature and sintering pressure provide atoms with extremely high diffusion activity, promoting oxide growth and elemental redistribution. V
3O
4 is an intermediate-valence oxide, and its formation is closely related to the multivalent nature of vanadium and the local oxygen partial pressure. During the SPS process, trace oxygen adsorbed on powder surfaces or in inter-particle gaps preferentially reacts with the more reactive V. Due to V’s strong affinity for oxygen and its relatively fast diffusion rate, V tends to accumulate at grain boundaries or defects to form V
3O
4. In contrast, Ti diffuses more slowly in the alloy and is prone to competing for oxygen with more reactive elements such as Al and V; hence, the oxidation of Ti often remains in a low-valence state rather than being fully oxidized to TiO
2. When the sintering temperature reaches 1000 °C, the TiO phase disappears in the LW1000 sample, suggesting that at sufficiently high temperatures, the extreme atomic mobility may temporarily overcome diffusion barriers, causing partial dissolution of oxides and driving the system metastably toward a more homogeneous single-phase solid solution. Therefore, the phase evolution in the alloy samples is the result of a competition between thermodynamic driving forces and kinetic diffusion rates under the rapid sintering conditions of SPS. Thermodynamically, elements such as Mg, Al, Ti, and V have a very strong affinity for oxygen, making oxide formation energetically favorable. Kinetically, the rapid heating/cooling characteristics of the SPS process, combined with the sluggish diffusion kinetics (the “sluggish diffusion effect”) typical of high-entropy alloys, significantly influence the final outcome of this process.
3.3. Microstructure of the MgAlTiVFeCo High-Entropy Alloy
Figure 5 shows the SEM images of the alloys sintered at different temperatures. As shown in
Figure 5, the surface of the LW850 (850 °C) alloy exhibits a relatively loose structure with noticeable pores. Compared to LW850, the LW900 (900 °C) alloy shows a slight reduction in porosity and gradually blurred particle boundaries, but the overall morphology remains inhomogeneous. The densification of the LW950 (950 °C) alloy is significantly improved, with a marked decrease in porosity and the appearance of locally dense regions, although a small amount of finely dispersed precipitated phases is still visible. The surface of the LW1000 (1000 °C) alloy is further densified, forming a continuous matrix with a more uniform distribution of precipitated phases.
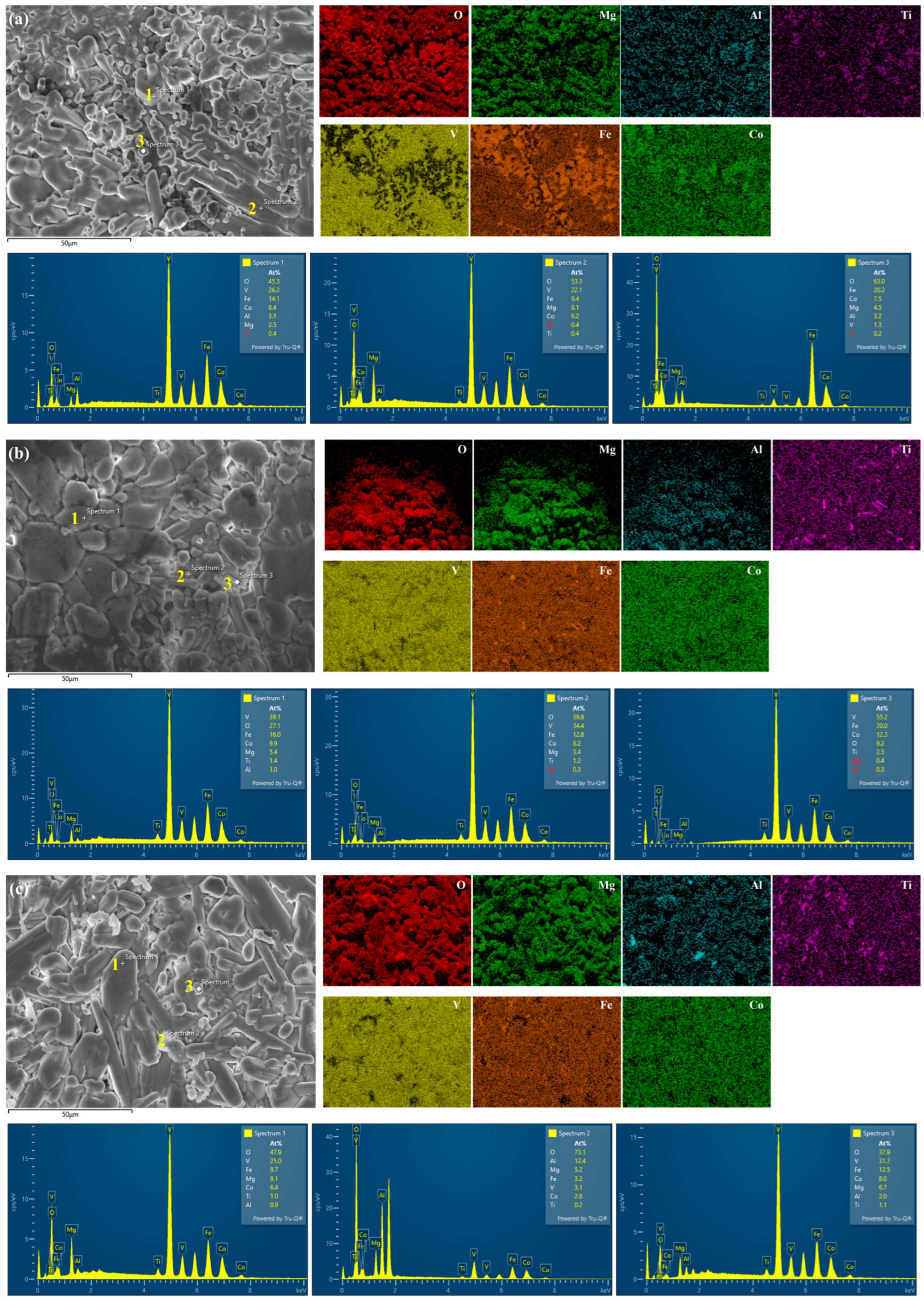
To establish a clear correspondence among the microscopic morphology, local chemical composition, and overall phase constitution, a multi-scale correlation analysis was employed. First, based on the atomic-number contrast in back-scattered electron (BSE) images, typical regions exhibiting distinct gray-level intensities (such as Regions 1, 2, and 3 illustrated in
Figure 6) were identified. Subsequently, high-resolution energy-dispersive X-ray spectroscopy (EDS) point analyses and small-area elemental mapping were performed on these specific regions to obtain their precise chemical compositions (
Table 3). Finally, these local composition data were systematically compared and correlated with the crystal structure information determined by large-area X-ray diffraction (XRD).
In the BSE images, regions displaying light-gray contrast (labeled as Region 1) show a relatively uniform distribution of the principal elements (Mg, Al, Ti, V, Fe, Co) according to EDS, with typically elevated contents of Fe and Co (
Table 3). The compositional characteristics of this region are consistent with the dominant body-centered cubic (BCC) solid-solution phase identified in the XRD patterns. The ability of the BCC structure to accommodate elements with considerably different atomic radii, together with the lattice parameters calculated from the diffraction peak positions (
Figure 4), which lie between those of the major constituent elements, further supports that this phase is a multi-principal-element substitutional solid solution rather than a single intermetallic compound. Regions exhibiting dark-gray contrast (labeled as Region 2) show strongly overlapping and intense signals of Mg, Al, and O in their EDS spectra. For example, in Region 2 of sample LW1000, the combined atomic percentages of Mg, Al, and O exceed 90%. This Mg-, Al-, and O-rich composition perfectly matches the diffraction peaks of various spinel-type structures (AB
2O
4), such as MgAl
2O
4 and MgV
2O
4, detected by XRD. The spinel phase allows a range of divalent and trivalent cations (e.g., Mg
2+, Fe
2+, Al
3+, V
3+) to occupy its tetrahedral (A) and octahedral (B) sites, leading to slight variations in the cation ratios measured by EDS across different samples, which explains the minor compositional differences observed in Region 2 of various specimens. Regions with a deep-gray contrast (labeled as Region 3) are identified by EDS analysis as Ti-rich (or V-rich) areas associated with high oxygen content. This compositional feature corresponds clearly to the diffraction peaks of simple oxide phases detected by XRD, such as TiO (PDF#08-0117) or V
3O
4 (PDF#34-0615). The precipitation of these phases is attributed to local micro-heterogeneities in oxygen partial pressure and elemental diffusion kinetics.
In multi-component alloy systems, the spinel structure (AB2O4) possesses higher configurational entropy and structural stability. During the SPS process, the alloy remains under relatively low-oxygen (or locally oxygen-deficient) conditions. According to the Ellingham diagram, under a given oxygen partial pressure, the oxidation tendency of the metals follows the order: Mg > Al > Ti > V > Fe > Co. However, in the MgAlTiVFeCo high-entropy alloy, although Mg and Al have strong oxidation tendencies, they are more likely to combine with other trivalent metals (e.g., Al3+, Fe3+, V3+) to form spinels rather than individually forming MgO or Al2O3. In contrast, the oxidation tendencies of Fe and Co are relatively weak. In the limited oxygen environment, oxygen is preferentially consumed by Mg, Al, Ti, and V, thereby suppressing the formation of FeO and CoO. This indicates that during the sintering of multi-component lightweight high-entropy alloys, trace oxygen can lead to the formation of complex oxide phases through selective oxidation, which in turn influences the microstructure and properties of the alloy.
3.4. Physical Properties of the MgAlTiVFeCo High-Entropy Alloy
According to the rule of mixtures based on the density of each constituent element, the theoretical density of the alloy can be calculated using Equation (3):
where
Ai is the atomic weight of element I,
ci is its atomic concentration, and
ρi is the density of element
i. The calculated theoretical density of the MgAlTiVFeCo high-entropy alloy is 5.304 g/cm
3, which meets the design criterion for lightweight high-entropy alloys (density < 6 g/cm
3). The measured actual densities of the LW850, LW900, LW950, and LW1000 alloy samples are 4.165 g/cm
3, 4.553 g/cm
3, 4.641 g/cm
3, and 5.127 g/cm
3 (
Figure 7a), respectively. The corresponding relative densities, calculated from these values, are 85.8%, 87.5%, 89.3%, and 96.7%.
Figure 7b shows the microhardness and relative density of the alloys sintered at different temperatures. As illustrated in
Figure 7b, the variation trends of microhardness and relative density for the alloy samples are consistent, both reaching their peak values at the sintering temperature of 1000 °C. The maximum hardness of LW1000 is 1235.5 HV, which is significantly higher than that of reported high-entropy alloys such as AlCrCuFeTi (714 HV) and AlCrCuFeV (565 HV) [
22], as well as traditional hard alloys [
23]. The relative density of this alloy reaches 96.7%. LW1000 thus exhibits excellent physical properties characterized by high hardness and high density.
The sintering densification process Is closely related to the evolution of pore structure. At lower sintering temperatures (850 °C and 900 °C), material transport is dominated by surface diffusion, and atomic mobility is limited. Consequently, the interconnected pore channels formed between the initial powder particles are difficult to eliminate, leading to slow densification progress. The residual porosity primarily consists of open pores, which can serve as rapid pathways for corrosive media or oxygen penetration. As the temperature rises to 950 °C or above, bulk diffusion and grain-boundary diffusion mechanisms become activated. The sintering driving force is significantly enhanced, accelerating atomic migration and neck growth between particles, which promotes gradual spheroidization, shrinkage, and closure of pores. As a result, the pore structure transitions to one dominated by isolated closed pores. At 1000 °C, this process proceeds most completely, thereby yielding the highest relative density (96.7%).
As can be seen from
Figure 7b, during low- to medium-temperature sintering (850–950 °C), the alloy hardness increases only gradually with rising temperature, primarily due to reduced porosity and improved matrix continuity. At the high sintering temperature of 1000 °C, the alloy’s hardness increases substantially. Besides the further improved densification, this significant enhancement is closely related to the abundant precipitation of oxide secondary phases. Spinel-type oxides (e.g., MgAl
2O
4, FeAl
2O
4) possess high hardness and high modulus; their dispersed distribution can effectively hinder dislocation motion. Simple oxides such as V
3O
4 and TiO, as well as the ilmenite-type oxide FeTiO
3, precipitate at grain boundaries or phase boundaries, pinning grain boundaries and refining grains, thereby contributing to precipitation strengthening and grain-refinement strengthening. LW1000 thus exhibits physical properties characterized by high hardness and high densification.
3.5. High-Temperature Oxidation Resistance of the MgAlTiVFeCo High-Entropy Alloy
Figure 8 shows the oxidation kinetics curves of the MgAlTiVFeCo high-entropy alloy oxidized at 900 °C for 60 h. According to the weight gain per unit area curves in
Figure 8a, all samples follow a parabolic law during the 60 h oxidation at 900 °C, which is consistent with Wagner’s theory of high-temperature oxidation [
24]. This indicates that the oxidation process is solid-state diffusion-controlled. The oxidation weight gain can be divided into two stages: the first stage (0–25 h), where the oxidation rate is relatively fast, and the weight gain increases rapidly with time, corresponding to the initial formation and growth of the oxide scale, and the second stage (25–60 h), where the oxidation rate slows down and stabilizes, corresponding to the thickening of the oxide scale and the establishment of a diffusion equilibrium. After 60 h of oxidation, the weight gains for LW850, LW900, LW950, LW1000, and LW1050 are 290.43, 284.08, 301.46, 130.76, and 140.45 mg/cm
2, respectively.
The parabolic rate constant (k
p) can be calculated using Equation (4):
where Δ
m is the mass gain,
S is the surface area of the sample, and
t is the oxidation time.
Figure 8b shows the relationship between (Δm/S)
2 and t or the MgAlTiVFeCo high-entropy alloy. The obtained oxidation rate constants (k
p1, k
p2) and the corresponding goodness-of-fit coefficients (R
2) are listed in
Table 4. As can be seen from the figure and
Table 4, during the first oxidation stage (0–25 h), the order of kp1 values is k
p1(LW1000) < k
p1(LW900) < k
p1(LW950) < k
p1(LW850). In the second oxidation stage (25–60 h), the order is k
p2(LW850) < k
p2(LW1000) < k
p2(LW950) < k
p2(LW900). The k
p values exhibit a non-monotonic variation with increasing sintering temperature, reflecting that the sintering temperature influences the oxidation diffusion path and rate by altering the intrinsic structure of the alloy. The oxidation rate constant of the MgAlTiVFeCo high-entropy alloy is comparable to that of the CrNbTiVAlx high-entropy alloy reported by Zhu et al. [
25] (
Table 4), indicating that the alloy exhibits favorable high-temperature oxidation resistance at 900 °C.
The relative density of the alloy increases significantly with the sintering temperature. The LW850 sample, containing numerous pores and interconnected channels, provides fast pathways for oxygen in-diffusion during the initial stage of high-temperature oxidation, accelerating internal oxidation and resulting in a higher kp value in the first stage. Conversely, high-density alloys like LW1000, with fewer pores and tightly bonded grain boundaries, effectively impede oxygen diffusion into the interior, lowering the initial oxidation rate. The various secondary oxide phases (spinel-type, simple oxides, ilmenite-type) formed during sintering play different roles during high-temperature oxidation. Spinel-type oxides (e.g., MgAl2O4, FeAl2O4), possessing excellent structural stability and low oxygen diffusion coefficients, are uniformly distributed in the LW1000 microstructure. During oxidation, they act as diffusion barriers, inhibiting further oxygen penetration. The content of non-protective oxides like V3O4 and TiO gradually decreases with increasing sintering temperature, reducing their detrimental effect on the continuity of the oxide scale. When ilmenite-type oxides such as FeTiO3 coexist with spinel phases, they can form multi-layered or composite oxide scales, improving the scale’s densification and adherence, thereby further enhancing the high-temperature oxidation resistance of LW1000. The high-temperature oxidation process is controlled by grain boundary diffusion. Alloys sintered at lower temperatures have finer grains and a higher density of grain boundaries, providing rapid pathways for oxygen diffusion along the boundaries, which is detrimental to oxidation resistance. In contrast, alloys sintered at high temperatures exhibit grain growth and a reduced density of grain boundaries. Furthermore, protective oxides may become enriched at these boundaries, hindering oxygen diffusion and thus leading to a lower oxidation rate.
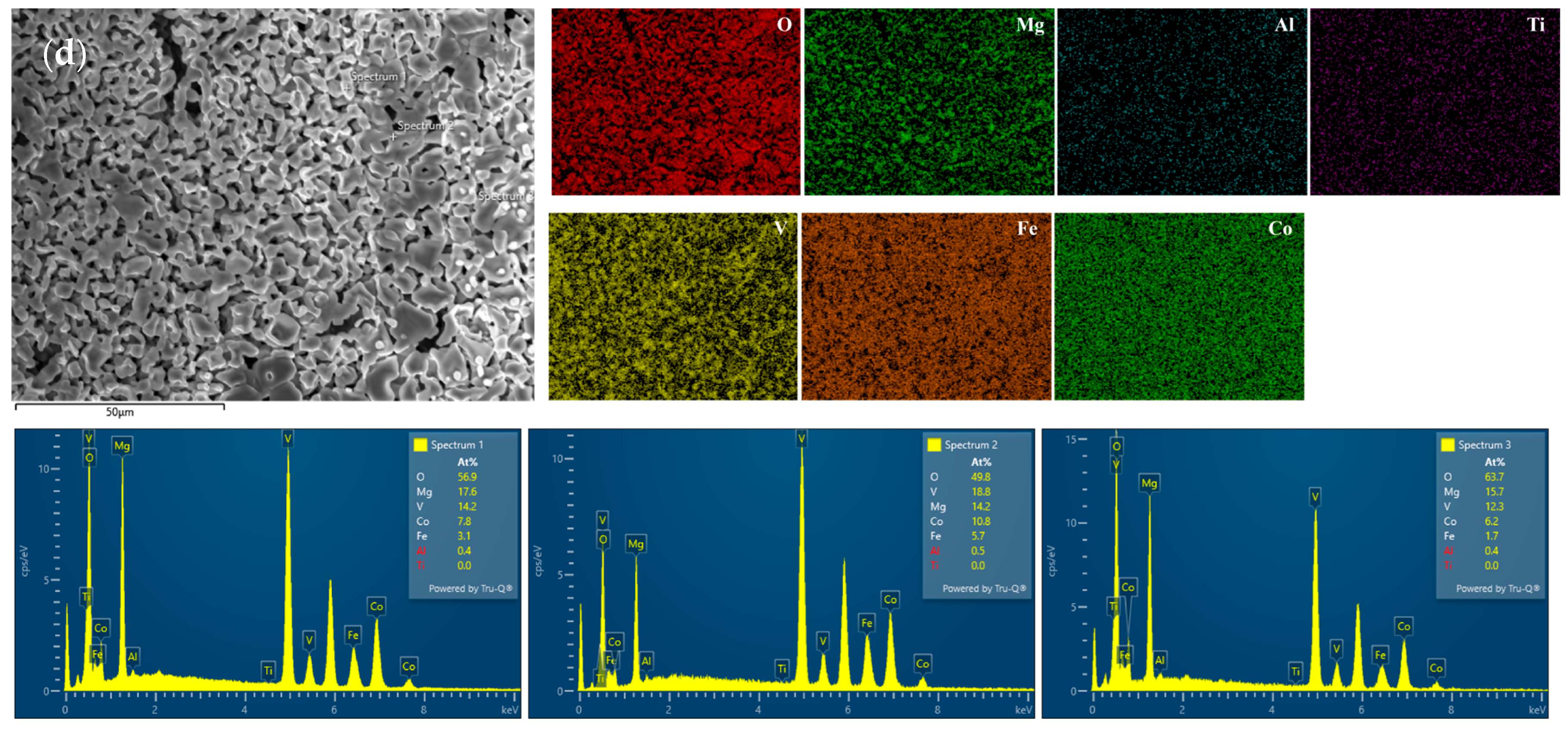
Figure 9 shows the surface oxide-layer morphology and the corresponding elemental mapping of the MgAlTiVFeCo high-entropy alloy after oxidation at 900 °C for 60 h. As the sintering temperature increases, the surface morphology after oxidation exhibits significant systematic changes. The oxide layer on the LW850 sample (
Figure 9a) appears rough, with a loose accumulation of granular oxides and noticeable micro-cracks and pores. This porous and poorly coherent structure provides fast diffusion channels for oxygen inward transport, which is detrimental to oxidation resistance. The surface compactness of the LW900 sample (
Figure 9b) improves to some extent, but the oxide particles remain non-uniform in size, and defects with poor continuity are still present in local areas. When the sintering temperature rises to 950 °C (
Figure 9c) and 1000 °C (
Figure 9d), a fundamental transformation occurs in the oxide-layer morphology. The surfaces become relatively smooth and dense, forming continuous and well-covered oxide layers. In particular, the LW1000 sample displays the most uniform and compact surface morphology, with micro-cracks and pores nearly eliminated. Such a dense oxide layer effectively hinders further inward diffusion of oxygen into the substrate, representing a key microstructural feature for excellent oxidation resistance.
The EDS elemental mapping results (elemental distribution maps In
Figure 9a–d) further clarify the chemical composition and formation mechanism of the oxide layer. In all specimens, oxygen (O) exhibits a uniform planar distribution that closely coincides with the oxide-layer regions, confirming the extensive formation of surface oxides. Magnesium (Mg) and aluminum (Al) are significantly enriched in the oxide layer, particularly in the LW950 and LW1000 specimens, where their enriched regions are more continuous. This is attributed to the high affinity of Mg and Al for oxygen (more negative standard Gibbs free energy, ΔG
ϴ), which drives their preferential outward diffusion during high-temperature oxidation, leading to the formation of MgO, Al
2O
3, or their composite oxides on the surface.
The Ccross-sectional morphology of specimens sintered at different temperatures (LW850, LW900, LW950, LW1000) after oxidation at 900 °C for 60 h is shown in
Figure 10. In all four specimens, the oxide layer is clearly distinguishable and resides on top of the dense alloy substrate. The oxide layer of the LW850 specimen (
Figure 10a) is the widest, exhibiting a thick and non-uniform multi-layer structure. Within the oxide layer, numerous longitudinal cracks and pores are observed, and the interface with the substrate is undulating. Local areas show extensive spallation of the oxide layer, indicating poor adhesion. The oxide layer of the LW900 specimen (
Figure 10b) is somewhat thinner, but still displays a distinct continuous oxide layer along with internal defects. In the LW950 specimen (
Figure 10c), the oxide layer becomes relatively thin and continuous, with reduced fragmentation and a flatter interface with the substrate. In contrast, the oxide-layer region of the LW1000 specimen (
Figure 10d) is the narrowest and thinnest among the four. Although micro-cracks were introduced during sample preparation, the overall continuity of the oxide layer remains good, with strong adhesion to the substrate and a sharp, planar interface.
The thick oxide layers observed in LW850 and LW900 originate from their porous and defect-rich initial matrix structures. During oxidation, oxygen readily penetrates into such matrices, leading to intense internal oxidation and the formation of thick, loose oxide scales. These oxide scales possess high internal stress and brittleness, making them prone to cracking and spallation under thermal stress or mechanical stress during sample preparation, which is consistent with the rough and porous morphologies observed on their surfaces. Repeated spallation of the oxide layer exposes fresh substrate, resulting in oxidation kinetics that follow an approximately linear, rapid rate. In contrast, for the LW950 and LW1000 specimens, the high-density matrix promotes predominantly external oxidation. Elemental diffusion leads to the formation of a thin, dense, and protective oxide layer on the surface, primarily consisting of spinel phases such as (Mg,Fe)(Al,V)2O4. Although these spinel phases are inherently brittle and micro-cracks may still appear during cross-sectional sample preparation, the oxide layer itself is thinner, denser, and exhibits relatively lower internal stress, thereby maintaining better overall integrity. In particular, the thinnest oxide layer of LW1000 provides direct evidence of its superior oxidation resistance, as the oxidation reaction is effectively confined to a near-surface region.
3.6. Electrochemical Corrosion Behavior of the MgAlTiVFeCo High-Entropy Alloy
Figure 11 presents the potentiodynamic polarization curves and electrochemical impedance spectra (EIS) of the MgAlTiVFeCo high-entropy alloys sintered at different temperatures in a 3.5 wt% NaCl solution. From the polarization curves in
Figure 11a, it can be observed that LW850, LW900, and LW950 all exhibit active dissolution behavior in the 3.5 wt% NaCl solution without a distinct passivation region. In contrast, LW1000 shows clear active and passivation characteristics, indicating the formation of a dense passive film on its surface, which reduces the anodic dissolution rate of the alloy. The corrosion potential (E
corr) and corrosion current density (I
corr) obtained by Tafel extrapolation are listed in
Table 5. A higher corrosion current density corresponds to a faster corrosion rate and thus poorer corrosion resistance. The corrosion potential (E
corr) reflects the tendency of the alloy to corrode; a more negative value indicates a greater susceptibility to corrosion. As shown in
Table 5, compared to the alloys sintered at medium-low temperatures (850–950 °C), namely LW850, LW900, and LW950, the high-temperature (1000 °C) sintered alloy LW1000 possesses a more positive corrosion potential (−0.438 V) and a lower corrosion current density (1.07 × 10
−6 A·cm
−2). Through comparison with the electrochemical parameters of lightweight high-entropy alloys reported in the literature [
26,
27,
28,
29], it can be concluded that the high-temperature sintered LW1000 exhibits excellent corrosion resistance in a 3.5 wt% NaCl solution.
The excellent corrosion resistance of LW1000 Is closely associated with its phase composition, microstructural evolution, and the stability of the surface oxide film, as revealed by combined XRD, SEM, and EDS analyses. Spinel structures, such as MgAl2O4 and FeAl2O4, possess outstanding chemical stability and corrosion resistance. These phases contribute to the formation of a continuous and dense oxide film on the alloy surface, effectively blocking the penetration of Cl− ions. While TiO is present in the alloys sintered at low-to-medium temperatures and acts as an anodic dissolution site due to its lower corrosion potential, it dissolves or transforms into more stable oxides during sintering at 1000 °C, thereby reducing active sites for localized corrosion. The high-temperature sintering process promotes elemental diffusion and phase equilibration, minimizing compositional segregation and the inhomogeneous distribution of secondary phases. A more uniform microstructure implies fewer galvanic corrosion cells, consequently leading to a lower corrosion rate.
Figure 11b presents the Nyquist plots of the alloys. The corrosion resistance of the alloys is positively correlated with the diameter of the capacitive arc on the Nyquist curve. As shown in
Figure 11b, the diameter of the capacitive arc increases with rising sintering temperature, indicating enhanced corrosion resistance. LW850, LW900, and LW950 exhibit a single capacitive arc, while LW1000 displays two capacitive arcs. Furthermore, the arc diameter for the high-temperature (1000 °C) sintered alloy LW1000 is significantly larger than that of the alloys sintered at medium-low temperatures (850–950 °C). The single capacitive arc observed for LW850, LW900, and LW950 suggests that the corrosion process is primarily controlled by a single time constant, corresponding to charge transfer occurring on a heterogeneous surface. The phase composition of these alloys consists of a BCC matrix with various dispersed secondary phases (e.g., MgV
2O
4, TiO). This structure does not allow the formation of a complete, dense, and protective surface film, leaving the alloy surface relatively “exposed.” Consequently, corrosion occurs directly at the metal/solution interface, which promotes pitting mechanisms [
30]. High-temperature sintering promotes the surface enrichment of elements such as Al and Fe in LW1000. These elements combine with oxygen to form a dense, stable, and continuous (spinel-type) oxide film in situ on the alloy surface. The corrosion process thus proceeds in two steps: first, in the high-frequency region (the first capacitive arc), aggressive ions (e.g., Cl
−) undergo a slow transport process diffusing through the surface film. Second, in the low-frequency region (the second capacitive arc), only the few ions that manage to penetrate the film reach the underlying metal substrate, initiating substrate metal dissolution (charge transfer). The appearance of the double capacitive arc confirms the formation of a high-quality protective passive film on the LW1000 surface, shifting the corrosion mechanism from “direct attack” to a “penetration and defense” mode.
Figure 11c,d shows the Bode plots of the alloys. From the phase-angle plot in
Figure 11c and the impedance modulus plot in
Figure 11d, it can be observed that the phase angle and the impedance modulus in the low-frequency region for LW1000 are significantly greater than those of the other alloys. It has been shown that a higher impedance modulus in the low-frequency region indicates a lower corrosion rate and better corrosion resistance. The phase angle is typically related to capacitive behavior; a larger phase angle suggests less surface charge accumulation and higher capacitive performance of the alloy. These observations demonstrate that LW1000 exhibits the lowest corrosion rate and the best corrosion resistance, which is consistent with the analysis results obtained from the potentiodynamic polarization curves.
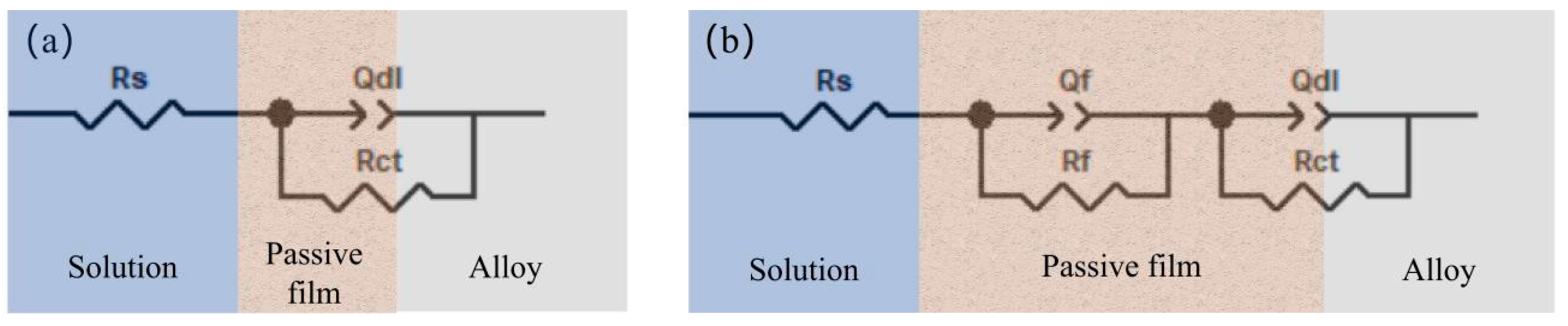
The EIS results of the MgAlTiVFeCo high-entropy alloy were fitted using equivalent circuits in Zview software to evaluate its corrosion resistance. The EIS data of LW850, LW900, and LW950 were fitted with the R
s-(Q
dl//R
ct) equivalent circuit model (
Figure 12a), while the EIS data of LW1000 were fitted with the R
s-(Q
f//R
f)-(Q
dl//R
ct) equivalent circuit model (
Figure 12b). Here, R
s represents the solution resistance, R
f the passive film resistance, R
ct the charge transfer resistance, and Q
f and Q
dl the capacitance of the passive film and the double layer, respectively. A constant phase element (CPE) is commonly used to represent non-ideal capacitive behavior. The impedance of a CPE can be expressed as
where
Y is the proportionality factor,
n the dispersion coefficient,
j the imaginary unit, and
ω the angular frequency.
The fitting results are presented in
Table 6. The corrosion process of the alloys prepared at low-to-medium temperatures (LW850, LW900, LW950) is governed by a single time constant, which corresponds to the charge transfer at the relatively exposed alloy/solution interface. The magnitude of the R
ct value directly reflects the difficulty of the electrochemical corrosion reaction. The fitting results show that as the sintering temperature increases from 850 °C to 950 °C, the R
ct value increases significantly from 159.80 Ω·cm
2 to 695.10 Ω·cm
2, indicating a gradual increase in the kinetic resistance of the corrosion reaction and an improvement in corrosion resistance. However, these values remain at the order of 10
2 Ω·cm
2, which is relatively low and corresponds to the higher corrosion current densities observed in the polarization curves. The Q
dl value of the alloys drops sharply from 4.78 × 10
−2 Ω
−1·cm
−2·s
n for LW850 to 2.66 × 10
−3 Ω
−1·cm
−2·s
n for LW950. Q
dl is related to the true electrode surface area and the dielectric properties of the interfacial layer; its decrease by an order of magnitude indicates a reduction in the active area available for the corrosion reaction. Meanwhile, the n1 values range between 0.46 and 0.52, significantly less than 1, indicating that the double layer exhibits strong non-ideal capacitive behavior (dispersion effect). This arises from the high heterogeneity of the alloy surface—a multiphase structure composed of the BCC matrix phase and various dispersed secondary phases (e.g., MgV
2O
4, TiO)—resulting in non-uniform current and potential distribution and the absence of a complete, dense protective surface film. Corrosive ions (e.g., Cl
−) can readily reach the metal surface, triggering a “direct-attack” corrosion mechanism characterized by localized pitting.
The EIS of the high-temperature alloy LW1000 exhibits distinct dual capacitive arc characteristics. The fitting yields an extremely high passive film resistance, Rf = 2.29 × 104 Ω·cm2, which is 1–2 orders of magnitude greater than the Rct values of the medium- and low-temperature alloys. This demonstrates that after sintering at 1000 °C, a dense, continuous, and highly stable protective oxide film (an Al- and Fe-enriched (spinel-type) structure) has formed in situ on the alloy surface. This film acts as a robust physical and electrochemical barrier, significantly hindering the penetration and transport of aggressive ions (Cl−). Concurrently, the passive film capacitance, Qf (5.00 × 10−5 Ω−1·cm−2·sn), is much smaller than the Qdl values of the medium- and low-temperature alloys, and its dispersion coefficient, n2 = 0.78, is closer to 1. This indicates that the film possesses relatively uniform and dense dielectric properties. Under the protection of this passive film, the amount of corrosive medium reaching the substrate interface is significantly reduced, leading to a further decrease in the double-layer capacitance at this interface to Qdl = 2.28 × 10−4 Ω−1·cm−2·sn. However, the corresponding charge transfer resistance, Rct (85.42 Ω·cm2), is far lower than that of LW950. This suggests that once the corrosive medium penetrates the dense passive film through micropores or defects and contacts the fresh metal substrate, localized corrosion reactions can still proceed readily. Nevertheless, the extremely low Qdl and very high Rf collectively indicate that the number and area of such penetration sites are minimal, and the path for ions through the film is greatly obstructed. Therefore, the overall corrosion rate is dominated by ion diffusion within the film (controlled by Rf), rather than by the charge transfer reaction beneath the film.
Figure 13 shows the surface corrosion morphology of the MgAlTiVFeCo high-entropy alloy after potentiodynamic polarization testing in a 3.5 wt% NaCl solution. As shown in
Figure 13, the corrosion morphology of the LW850 and LW900 alloys exhibits typical severe general corrosion characteristics. The surface is extensively covered by a loose, non-uniform layer of corrosion products, with densely distributed deep and large pits visible. The corrosion morphology of the LW950 alloy shows some improvement compared to LW850 and LW900, but overall, it still belongs to non-uniform corrosion. The surface displays large, lumpy accumulations of corrosion products, while in some areas the substrate metal is exposed, revealing features of localized dissolution. It is evident that the increase in sintering temperature refines the microstructure to some extent or promotes surface diffusion of certain elements, slightly mitigating the surface chemical heterogeneity and reducing the severity of corrosion. The surface of the LW1000 alloy is flat and smooth, with only a few small, isolated pitting spots scattered across it, demonstrating excellent localized corrosion characteristics. High-temperature (1000 °C) sintering promotes the surface enrichment of elements such as Al and Fe, leading to the formation of a dense, continuous (spinel-type) passive film. This dense passive film (high R
f) effectively blocks the uniform penetration of aggressive Cl
− ions, confining corrosion to local weak points in the film. Once the film is locally ruptured and aggressive ions reach the substrate, pitting is initiated, forming the isolated pits seen in the figure. However, due to the extremely strong protective nature of the film (high R
f), the pits are prevented from propagating and connecting, thereby preserving the majority of the substrate. The extremely low double-layer capacitance Q
dl also indirectly confirms that the true area undergoing corrosion reactions is very small.