Polymeric Fibrous Materials for Procoagulant and Anticoagulant Applications: A Review of Molecular Blood–Material Mechanisms and Strategies
Abstract
1. Introduction
2. Fundamentals of the Blood Coagulation Cascade
2.1. Plasma Proteins, Platelets, and the Contact Pathway
2.2. Intrinsic, Extrinsic, and Common Pathways
2.3. Cellular Model and Spatial Regulation of Thrombin on Fibrous Materials
2.4. Analytical Tests Characterizing the Interaction of Hemostatic Materials with Blood
3. Interactions of Polymers with Proteins Regulating Blood Coagulation
3.1. The Role of Surface Charge, Hydration Force, and Zeta Potential Between Polymers and Plasma Proteins
3.2. Nanoscale Fiber Topography, Diameter, Porosity, and Mechanical Strength
3.3. Contact Activation at Polymer Interfaces: FXII/HMWK/Prekallikrein Microenvironment
3.4. Platelet Adhesion, Activation, Spreading, and Membrane Support for Enzymatic Complexes on Fibrous Materials
3.5. Mapping Interfacial Phenomena to Coagulation Metrics of Fibrous Materials
3.6. The Influence of Hemorheology on Coagulation Process
4. Procoagulant Fiber-Forming Polymers
4.1. Natural Polysaccharides with Intrinsic Hemostatic Potential
4.1.1. Chitosan: A Cationic Polymer with Strong Platelet-Interactive Properties
4.1.2. Alginate: An Anionic Polysaccharide Enabling Ca2+-Mediated Gelation and Fibrin Reinforcement
4.1.3. Cellulose-Based Fibers
4.2. Synthetic Polymers Engineered for Rapid Hemostasis
4.3. Metal–Polymer Coordination Systems: Ca2+, Zn2+, and Cu2+ as Accelerators of Coagulation Microenvironments
4.4. Crosslinking Strategies, Surface Charge Tuning, and Microenvironment Engineering
4.5. Electrospun Prohemostatic Composites: Synergistic Fiber Systems
4.6. Translational Considerations for Procoagulant Fiber Design
5. Strategies for Designing Anticoagulant and Antithrombogenic Fiber-Based Materials
5.1. Heparin-Functionalized Fibers: Localized Suppression of Enzymatic Propagation
5.2. Nitric Oxide (NO)-Releasing Coatings: Biomimetic Signaling
5.3. Zwitterionic and Ultra-Hydrophilic Interfaces: Preventing Protein Adsorption and Contact Activation
5.4. Hybrid Fiber Strategies: Synergy Through Multi-Level Control
5.5. Fiber Architecture and Morphology
5.6. Regulation of Protein–Material Molecular Interactions: Initiation, Amplification, and Propagation Phases
5.7. Quantitative Mechanistic Assessment Indicators
5.8. Comparative Mechanisms and Translational Matrix
5.9. Interfacial Chemistry and Morphological Drivers
6. Logic and Perspectives of Functional Material Design
The Clinical Implementation of Hemostatic Biomaterials
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ADP | Adenosine Diphosphate |
| anti-Xa | Heparin Assay |
| aPTT | Kaolin–Cephalin Time |
| ATIII | Antithrombin III |
| CB | Carboxybetaine |
| ETP | Endogenous Thrombin Potential |
| GPIIb | Glycoprotein Receptor Inhibitor |
| H12 | γ-chain Dodecapeptide |
| HIT | Heparin-induced Thrombocytopenia |
| HMWK | High-Molecular-Weight Kininogen |
| IgG | Immunoglobulin G |
| LbL | Layer by Layer |
| ORC | Oxidized Regenerated Cellulose |
| PAC-1 | Procaspase-3 Activator |
| PC | Phosphorylcholine |
| PCL | Polycaprolactone |
| PEG | Polyethylene Glycol |
| PF4 | Platelet Factor 4 |
| PK | Prekallikrein |
| PLA | Poly(lactic acid) |
| PS | Phosphatidylserine |
| PT | Prothrombin Time |
| PU | Polyurethane |
| SB | Sulfobetaine |
| SEM | Scanning Electron Microscopy |
| TAFI | Thrombin Fibrinolysis Inhibitor |
| TAT | Thrombin–Antithrombin III |
| TF | Tissue Factor |
| TG | Thrombin Generation |
References
- Jaffer, I.H.; Weitz, J.I. The Blood Compatibility Challenge. Part 1: Blood-Contacting Medical Devices: The Scope of the Problem. Acta Biomater. 2019, 94, 2–10. [Google Scholar] [CrossRef]
- Brash, J.L.; Horbett, T.A.; Latour, R.A.; Tengvall, P. The Blood Compatibility Challenge. Part 2: Protein Adsorption Phenomena Governing Blood Reactivity. Acta Biomater. 2019, 94, 11–24. [Google Scholar] [CrossRef]
- Park, S.; Park, J.K. Back to Basics: The Coagulation Pathway. Blood Res. 2024, 59, 35. [Google Scholar] [CrossRef]
- Hoffman, M.; Monroe, D.M. A Cell-Based Model of Hemostasis. Thromb. Haemost. 2001, 85, 958–965. [Google Scholar] [CrossRef] [PubMed]
- Monroe, D.M.; Hoffman, M. What Does It Take to Make the Perfect Clot? Arterioscler. Thromb. Vasc. Biol. 2006, 26, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Becker, R.C. Cell-Based Models of Coagulation: A Paradigm in Evolution. J. Thromb. Thrombolysis 2005, 20, 65–68. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.Y.; Lim, S.M.; Albertorio, F.; Kim, G.; Gurau, M.C.; Yang, R.D.; Holden, M.A.; Cremer, P.S. The Vroman Effect: A Molecular Level Description of Fibrinogen Displacement. J. Am. Chem. Soc. 2003, 125, 12782–12786. [Google Scholar] [CrossRef]
- Xia, Y.; Yang, R.; Wang, H.; Li, Y.; Fu, C. Application of Chitosan-Based Materials in Surgical or Postoperative Hemostasis. Front. Mater. 2022, 9, 994265. [Google Scholar] [CrossRef]
- Hassanzadeh-Tabrizi, S.A. Alginate Based Hemostatic Materials for Bleeding Management: A Review. Int. J. Biol. Macromol. 2024, 274, 133218. [Google Scholar] [CrossRef]
- Kudzin, M.H.; Kaczmarek, A.; Mrozinska, Z.; Hernandez, C.; Piekarska, K.; Wozniak, K.; Juszczak, M.; Krol, P. Hybrid Alginate–Graphene Composites: Biochemical Features and Biomedical Potential. Mar. Drugs 2025, 23, 323. [Google Scholar] [CrossRef]
- Bukatuka, C.F.; Mbituyimana, B.; Xiao, L.; Qaed Ahmed, A.A.; Qi, F.; Adhikari, M.; Shi, Z.; Yang, G. Recent Trends in the Application of Cellulose-Based Hemostatic and Wound Healing Dressings. J. Funct. Biomater. 2025, 16, 151. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, Y.; Gao, Z.; Mao, X.; Cheng, J.; Huang, L.; Tang, J. Advances in Wound Dressing Based on Electrospinning Nanofibers. J. Appl. Polym. Sci. 2024, 141, e54746. [Google Scholar] [CrossRef]
- Gupta, S.; Puttaiahgowda, Y.M.; Deiglmayr, L. Recent Advances in the Design and Immobilization of Heparin for Biomedical Application: A Review. Int. J. Biol. Macromol. 2024, 264, 130743. [Google Scholar] [CrossRef]
- Kalugin, D.; Bahig, J.; Shoker, A.; Abdelrasoul, A. Heparin-Immobilized Polyethersulfone for Hemocompatibility Enhancement of Dialysis Membrane: In Situ Synchrotron Imaging, Experimental, and Ex Vivo Studies. Membranes 2023, 13, 718. [Google Scholar] [CrossRef] [PubMed]
- Yadav, C.J.; Yadav, U.; Afrin, S.; Lee, J.Y.; Kamel, J.; Park, K.M. Heparin Immobilization Enhances Hemocompatibility, Re-Endothelization, and Angiogenesis of Decellularized Liver Scaffolds. Int. J. Mol. Sci. 2024, 25, 12132. [Google Scholar] [CrossRef]
- Laschewsky, A. Structures and Synthesis of Zwitterionic Polymers. Polymers 2014, 6, 1544–1601. [Google Scholar] [CrossRef]
- Bhattacharjee, A.; Savargaonkar, A.V.; Tahir, M.; Sionkowska, A.; Popat, K.C. Surface Modification Strategies for Improved Hemocompatibility of Polymeric Materials: A Comprehensive Review. RSC Adv. 2024, 14, 7440. [Google Scholar] [CrossRef]
- Ma, Q.; Zhang, W.; Mou, X.; Huang, N.; Wang, H.; Zhang, H.; Yang, Z. Bioinspired Zwitterionic Block Polymer-Armored Nitric Oxide-Generating Coating Combats Thrombosis and Biofouling. Research 2024, 7, 0423. [Google Scholar] [CrossRef] [PubMed]
- Chaudhry, R.; Killeen, R.B.; Babiker, H.M.; Usama, S.M.; Babiker, H.M. Physiology, Coagulation Pathways; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Gailani, D.; Renné, T. Intrinsic Pathway of Coagulation and Arterial Thrombosis. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 2507–2513. [Google Scholar] [CrossRef]
- Weisel, J.W.; Litvinov, R.I. Fibrin Formation, Structure and Properties. In Fibrous Proteins: Structures and Mechanisms; Subcellular Biochemistry; Springer: Berlin/Heidelberg, Germany, 2017; Volume 82, pp. 405–456. [Google Scholar]
- Noh, H.; Vogler, E.A. Volumetric Interpretation of Protein Adsorption: Partition Coefficients, Interphase Volumes, and Free Energies of Adsorption to Hydrophobic Surfaces. Biomaterials 2006, 27, 5780–5793. [Google Scholar] [CrossRef]
- Hemker, H.C.; Beguin, S. Thrombin Generation in Plasma: Its Assessment via the Endogenous Thrombin Potential. Thromb. Haemost. 1995, 74, 134–138. [Google Scholar] [CrossRef]
- Hemker, H.C.; Giesen, P.; Al Dieri, R.; Regnault, V.; De Smedt, E.; Wagenvoord, R.; Lecompte, T.; Béguin, S. Calibrated Automated Thrombin Generation Measurement in Clotting Plasma. Pathophysiol. Haemost. Thromb. 2003, 33, 4–15. [Google Scholar] [CrossRef]
- Bagot, C.N.; Leishman, E. Establishing a Reference Range for Thrombin Generation Using a Standard Plasma Significantly Improves Assay Precision. Thromb. Res. 2015, 136, 139–143. [Google Scholar] [CrossRef]
- Long, A.T.; Kenne, E.; Jung, R.; Fuchs, T.A.; Renné, T. Contact System Revisited: An Interface between Inflammation, Coagulation, and Innate Immunity. J. Thromb. Haemost. 2016, 14, 427–437. [Google Scholar] [CrossRef]
- Gheorghiță, D.; Moldovan, H.; Robu, A.; Bița, A.I.; Grosu, E.; Antoniac, A.; Corneschi, I.; Antoniac, I.; Bodog, A.D.; Băcilă, C.I. Chitosan-Based Biomaterials for Hemostatic Applications: A Review of Recent Advances. Int. J. Mol. Sci. 2023, 24, 10540. [Google Scholar] [CrossRef]
- Gailani, D.; Broze, G.J. Factor XI Activation in a Revised Model of Blood Coagulation. Science 1991, 253, 909–912. [Google Scholar] [CrossRef]
- Li, H.; Guo, Q.; Tu, Q.; Xiong, K.; Wang, W.; Lu, L.; Zhang, W.; Huang, N.; Yang, Z. Nitric Oxide-Generating Self-Assembling Peptide Hydrogel Coating for Enhancing Hemocompatibility of Blood-Contacting Devices. J. Mater. Sci. Technol. 2022, 131, 106–114. [Google Scholar] [CrossRef]
- Rabe, M.; Verdes, D.; Seeger, S. Understanding Protein Adsorption Phenomena at Solid Surfaces. Adv. Colloid Interface Sci. 2011, 162, 87–106. [Google Scholar] [CrossRef]
- Litvinov, R.I.; Farrell, D.H.; Weisel, J.W.; Bennett, J.S. The Platelet Integrin AIIbβ3 Differentially Interacts with Fibrin Versus Fibrinogen. J. Biol. Chem. 2016, 291, 7858. [Google Scholar] [CrossRef] [PubMed]
- Noh, H.; Vogler, E.A. Volumetric Interpretation of Protein Adsorption: Competition from Mixtures and the Vroman Effect. Biomaterials 2007, 28, 405–422. [Google Scholar] [CrossRef] [PubMed]
- Sin, M.C.; Chen, S.H.; Chang, Y. Hemocompatibility of Zwitterionic Interfaces and Membranes. Polym. J. 2014, 46, 436–443. [Google Scholar] [CrossRef]
- Bashir, S.; Hina, M.; Iqbal, J.; Rajpar, A.H.; Mujtaba, M.A.; Alghamdi, N.A.; Wageh, S.; Ramesh, K.; Ramesh, S. Fundamental Concepts of Hydrogels: Synthesis, Properties, and Their Applications. Polymers 2020, 12, 2702. [Google Scholar] [CrossRef]
- Dargaud, Y.; Wolberg, A.S.; Gray, E.; Negrier, C.; Hemker, H.C. Proposal for Standardized Preanalytical and Analytical Conditions for Measuring Thrombin Generation in Hemophilia: Communication from the SSC of the ISTH. J. Thromb. Haemost. 2017, 15, 1704–1707. [Google Scholar] [CrossRef] [PubMed]
- Depasse, F.; Binder, N.B.; Mueller, J.; Wissel, T.; Schwers, S.; Germer, M.; Hermes, B.; Turecek, P.L. Thrombin Generation Assays Are Versatile Tools in Blood Coagulation Analysis: A Review of Technical Features, and Applications from Research to Laboratory Routine. J. Thromb. Haemost. 2021, 19, 2907–2917. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.C.; Wo, Y.; Meyerhoff, M.E.; Siedlecki, C.A. Inhibition of Bacterial Adhesion and Biofilm Formation by Dual Functional Textured and Nitric Oxide Releasing Surfaces. Acta Biomater. 2017, 51, 53–65. [Google Scholar] [CrossRef]
- Xu, L.C.; Meyerhoff, M.E.; Siedlecki, C.A. Blood Coagulation Response and Bacterial Adhesion to Biomimetic Polyurethane Biomaterials Prepared with Surface Texturing and Nitric Oxide Release. Acta Biomater. 2019, 84, 77–87. [Google Scholar] [CrossRef]
- Tomizawa, Y. Clinical Benefits and Risk Analysis of Topical Hemostats: A Review. J. Artif. Organs 2005, 8, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Zhang, T.; Huang, X.; Huan, X.; Li, Y. Evaluating the impact of transient shear stress on platelet activation, adhesion, and aggregation with microfluidic chip technique. Artif. Organs 2023, 58, 28–36. [Google Scholar] [CrossRef]
- Wang, S.; Griffith, B.P.; Wu, Z.J. Device-induced hemostatic disorders in mechanically assisted circulation. Clin. Appl. Thromb. Hemost. 2021, 27, 1–14. [Google Scholar] [CrossRef]
- Affeld, K.; Schaller, J.; Wölken, T.; Krabatsch, T.; Kertzscher, U. Role of flow for the deposition of platelets. Biointerphases 2016, 11, 029804. [Google Scholar] [CrossRef]
- Rooij, B.J.M.; Zavodszky, G.; Hoekstra, A.G.; Ku, D.N. Haemodynamic flow conditions at the initiation of high-shear platelet aggregation: A combined in vitro and cellular in silico study. Interface Focus 2021, 11, 20190126. [Google Scholar] [CrossRef]
- Zhao, Y.C.; Vatankhah, P.; Goh, T.; Michelis, R.; Kyanian, K.; Zhang, Y.; Li, Z.; Ju, L.A. Hemodynamic analysis for stenosis microfluidic model of thrombosis with refined computational fluid dynamics simulation. Sci. Rep. 2021, 11, 6875. [Google Scholar] [CrossRef]
- Rana, A.; Westein, E.; Niego, B.; Hagemeyer, C.E. Shear-Dependent Platelet Aggregation: Mechanisms and Therapeutic Opportunities. Front. Cardiovasc. Med. 2019, 6, 141. [Google Scholar] [CrossRef]
- Chen, Z.; Zhang, J.; Li, T.; Tran, D.; Griffith, B.P.; Wu, Z.J. The impact of shear stress on device-induced platelet hemostatic dysfunction relevant to thrombosis and bleeding in mechanically assisted circulation. Artif. Organs 2020, 44, E201–E213. [Google Scholar] [CrossRef] [PubMed]
- DaShawn, A.; Hickman, C.L.; Pawlowski, U.D.S.; Sekhon, J.M.; Gupta, A.S. Biomaterials and Advanced Technologies for Hemostatic Management of Bleeding. Adv. Mater. 2018, 30, 1700859. [Google Scholar] [CrossRef]
- Kuchinka, J.; Willems, C.; Telyshev, D.V.; Groth, T. Control of Blood Coagulation by Hemocompatible Material Surfaces—A Review. Bioengineering 2021, 8, 215. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Liu, S.; Lau, S.; Li, J. Hemostatic biomaterials to halt non-compressible hemorrhage. J. Mater. Chem. B 2022, 37, 7239–7259. [Google Scholar] [CrossRef]
- Thomas, S. Alginate Dressings in Surgery and Wound Management–Part 1. J. Wound Care 2000, 9, 56–60. [Google Scholar] [CrossRef]
- Khoshmohabat, H.; Paydar, S.; Kazemi, H.M.; Dalfardi, B. Overview of Agents Used for Emergency Hemostasis. Trauma Mon. 2016, 21, e26023. [Google Scholar] [CrossRef] [PubMed]
- Lewis, K.M.; Spazierer, D.; Urban, M.D.; Lin, L.; Redl, H.; Goppelt, A. Comparison of Regenerated and Non-Regenerated Oxidized Cellulose Hemostatic Agents. Eur. Surg. 2013, 45, 213–220. [Google Scholar] [CrossRef]
- Kozen, B.G.; Kircher, S.J.; Henao, J.; Godinez, F.S.; Johnson, A.S. An Alternative Hemostatic Dressing: Comparison of CELOX, HemCon, and QuikClot. Acad. Emerg. Med. 2008, 15, 74–81. [Google Scholar] [CrossRef]
- Roach, P.; Farrar, D.; Perry, C.C. Interpretation of Protein Adsorption: Surface-Induced Conformational Changes. J. Am. Chem. Soc. 2005, 127, 8168–8173. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Zheng, Y.; Zhang, K.; Yao, Y.; Wang, L.; Li, X.; Yu, J.; Ding, B. Electrospun Nanofibrous Materials for Wound Healing. Adv. Fiber Mater. 2020, 2, 212–227. [Google Scholar] [CrossRef]
- Mrozinska, Z.; Kudzin, M.H.; Ponczek, M.B.; Kaczmarek, A.; Krol, P.; Lisiak-Kucinska, A.; Zylla, R.; Walawska, A. Biochemical Approach to Poly(Lactide)–Copper Composite—Impact on Blood Coagulation Processes. Materials 2024, 17, 608. [Google Scholar] [CrossRef]
- Gaharwar, A.K.; Singh, I.; Khademhosseini, A. Engineered Biomaterials for in Situ Tissue Regeneration. Nat. Rev. Mater. 2020, 5, 686–705. [Google Scholar] [CrossRef]
- Satchanska, G.; Davidova, S.; Petrov, P.D. Natural and Synthetic Polymers for Biomedical and Environmental Applications. Polymers 2024, 16, 1159. [Google Scholar] [CrossRef]
- Tawade, P.; Tondapurkar, N.; Jangale, A. Biodegradable and biocompatible synthetic polymers for applications in bone and muscle tissue engineering. J. Med. Sci. 2022, 91, e712. [Google Scholar] [CrossRef]
- Heidari, B.S.; Ruan, R.; Vahabli, E.; Chen, P.; De-Juan-Pardo, E.M.; Zheng, M.; Doyle, B. Natural, synthetic and commercially-available biopolymers used to regenerate tendons and ligaments. Bioact. Mater. 2023, 19, 179–197. [Google Scholar] [CrossRef]
- Prete, S.; Dattilo, M.; Patitucci, F.; Pezzi, G.; Parisi, O.I.; Puoci, F. Natural and Synthetic Polymeric Biomaterials for Application in Wound Management. J. Funct. Biomater. 2023, 14, 455. [Google Scholar] [CrossRef] [PubMed]
- Riaz, S.; Waheed, H.; Ahmad, F.; Khan, M.I.; Shanableh, A. Natural and synthetic biomaterials, structural matrices-based wound dressings: Key properties, material correlation, and adaptability. Regenes. Repair Rehabil. 2025, 1, 47–65. [Google Scholar] [CrossRef]
- Donnaloja, F.; Jacchetti, E.; Soncini, M.; Raimondi, M.T. Natural and Synthetic Polymers for Bone Scaffolds Optimization. Polymers 2020, 12, 905. [Google Scholar] [CrossRef]
- Zamani, S.; Ehterami, A.; Vaez, A.; Naeiji, M.; Maghsoodifar, H.; Douki, S.A.H.S.; Abad, M.M.E.; Arabpour, Z.; Baheiraei, N.; Farahani, A.; et al. Natural and synthetic polymers in burn wound healing. J. Biomater. Sci. Polym. Ed. 2025, 30, 118–183. [Google Scholar] [CrossRef] [PubMed]
- Gorbet, M.B.; Sefton, M.V. Biomaterial-Associated Thrombosis: Roles of Coagulation Factors, Complement, Platelets and Leukocytes. Biomaterials 2004, 25, 5681–5703. [Google Scholar] [CrossRef] [PubMed]
- Manivasagam, V.K.; Sabino, R.M.; Kantam, P.; Popat, K.C. Surface modification strategies to improve titanium hemo-compatibility: A comprehensive review. Mater. Adv. 2021, 2, 5824–5842. [Google Scholar] [CrossRef]
- Weber, M.; Steinle, H.; Golombek, S.; Hann, L.; Schlensak, C.; Wendel, H.P.; Avci-Adali, M. Blood-Contacting Biomaterials: In Vitro Evaluation of the Hemocompatibility. Front. Bioeng. Biotechnol. 2018, 6, 99. [Google Scholar] [CrossRef]
- Biran, R.; Pond, D. Heparin Coatings for Improving Blood Compatibility of Medical Devices. Adv. Drug Deliv. Rev. 2017, 112, 12–23. [Google Scholar] [CrossRef] [PubMed]
- Abdelrasoul, A.; Kalugin, D.; Shoker, A. Recent Developments and Current Challenges of Heparin-Grafted Hemodialysis Membranes. J. Compos. Sci. 2022, 6, 244. [Google Scholar] [CrossRef]
- Ishihara, K. Blood-Compatible Surfaces with Phosphorylcholine-Based Polymers for Cardiovascular Medical Devices. Langmuir 2018, 35, 1778–1787. [Google Scholar] [CrossRef] [PubMed]
- Kodama, K.; Pasche, B.; Olsson, P.; Swedenborg, J.; Adolfsson, L.; Larm, O.; Riesenfeld, J. Antithrombin III Binding to Surface Immobilized Heparin and Its Relation to F Xa Inhibition. Thromb. Haemost. 1987, 58, 1064–1067. [Google Scholar] [CrossRef] [PubMed]
- Nalezinková, M. In Vitro Hemocompatibility Testing of Medical Devices. Thromb. Res. 2020, 195, 146–150. [Google Scholar] [CrossRef] [PubMed]
- Begovac, P.C.; Thomson, R.C.; Fisher, J.L.; Hughson, A.; Gällhagen, A. Improvements in GORE-TEX® Vascular Graft Performance by Carmeda® Bioactive Surface Heparin Immobilization. Eur. J. Vasc. Endovasc. Surg. 2003, 25, 432–437. [Google Scholar] [CrossRef]
- Wu, Y.; Zhou, Z.; Meyerhoff, M.E. In Vitro Platelet Adhesion on Polymeric Surfaces with Varying Fluxes of Continuous Nitric Oxide Release. J. Biomed. Mater. Res. Part A 2007, 81, 956–963. [Google Scholar] [CrossRef]
- Reynolds, M.M.; Frost, M.C.; Meyerhoff, M.E. Nitric Oxide-Releasing Hydrophobic Polymers: Preparation, Characterization, and Potential Biomedical Applications. Free Radic. Biol. Med. 2004, 37, 926–936. [Google Scholar] [CrossRef] [PubMed]
- Annich, G.M.; Meinhardt, J.P.; Mowery, K.A.; Ashton, B.A.; Merz, S.I.; Hirschl, R.B.; Meyerhoff, M.E.; Bartlett, R.H. Reduced Platelet Activation and Thrombosis in Extracorporeal Circuits Coated with Nitric Oxide Release Polymers. Crit. Care Med. 2000, 28, 915–920. [Google Scholar] [CrossRef]
- Amoako, K.A.; Sundaram, H.S.; Suhaib, A.; Jiang, S.; Cook, K.E. Multimodal, Biomaterial-Focused Anticoagulation via Superlow Fouling Zwitterionic Functional Groups Coupled with Anti-Platelet Nitric Oxide Release. Adv. Mater. Interfaces 2016, 3, 1500646. [Google Scholar] [CrossRef]
- Blackman, L.D.; Gunatillake, P.A.; Cass, P.; Locock, K.E.S. An Introduction to Zwitterionic Polymer Behavior and Applications in Solution and at Surfaces. Chem. Soc. Rev. 2019, 48, 757–770. [Google Scholar] [CrossRef] [PubMed]
- Shao, Q.; Jiang, S. Molecular Understanding and Design of Zwitterionic Materials. Adv. Mater. 2015, 27, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Simpson, A.; Shukla, A.; Brown, A.C. Biomaterials for Hemostasis. Annu. Rev. Biomed. Eng. 2022, 24, 111–135. [Google Scholar] [CrossRef]
- Kizhakkedathu, J.N.; Conway, E.M. Biomaterial and Cellular Implants: Foreign Surfaces Where Immunity and Coagulation Meet. Blood 2022, 139, 1987–1998. [Google Scholar] [CrossRef]
- Chang, Y.; Liao, S.C.; Higuchi, A.; Ruaan, R.C.; Chu, C.W.; Chen, W.Y. A Highly Stable Nonbiofouling Surface with Well-Packed Grafted Zwitterionic Polysulfobetaine for Plasma Protein Repulsion. Langmuir 2008, 24, 5453–5458. [Google Scholar] [CrossRef]
- Nazari, S.; Abdelrasoul, A. Impact of Membrane Modification and Surface Immobilization Techniques on the Hemocompatibility of Hemodialysis Membranes: A Critical Review. Membranes 2022, 12, 1063. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, B.; Ekdahl, K.N.; Mollnes, T.E.; Lambris, J.D. The Role of Complement in Biomaterial-Induced Inflammation. Mol. Immunol. 2007, 44, 82–94. [Google Scholar] [CrossRef]
- Mödinger, Y.; Teixeira, G.Q.; Neidlinger-Wilke, C.; Ignatius, A. Role of the Complement System in the Response to Orthopedic Biomaterials. Int. J. Mol. Sci. 2018, 19, 3367. [Google Scholar] [CrossRef]
- Ghimire, S.; Sarkar, P.; Rigby, K.; Maan, A.; Mukherjee, S.; Crawford, K.E.; Mukhopadhyay, K. Polymeric Materials for Hemostatic Wound Healing. Pharmaceutics 2021, 13, 2127. [Google Scholar] [CrossRef]
- Hu, Z.; Zhang, D.Y.; Lu, S.T.; Li, P.W.; Li, S.D. Chitosan-Based Composite Materials for Prospective Hemostatic Applications. Mar. Drugs 2018, 16, 273. [Google Scholar] [CrossRef] [PubMed]
- Cui, G.; Guo, X.; Deng, L. Preparation Strategies of Mussel-Inspired Chitosan-Based Biomaterials for Hemostasis. Front. Pharmacol. 2024, 15, 1439036. [Google Scholar] [CrossRef] [PubMed]
- Major, T.C.; Handa, H.; Annich, G.M.; Bartlett, R.H. Development and Hemocompatibility Testing of Nitric Oxide Releasing Polymers Using a Rabbit Model of Thrombogenicity. J. Biomater. Appl. 2014, 29, 479–501. [Google Scholar] [CrossRef]
- Major, T.C.; Handa, H.; Brisbois, E.J.; Reynolds, M.M.; Annich, G.M.; Meyerhoff, M.E.; Bartlett, R.H. The Mediation of Platelet Quiescence by NO-Releasing Polymers via CGMP-Induced Serine 239 Phosphorylation of Vasodilator-Stimulated Phosphoprotein. Biomaterials 2013, 34, 8086–8096. [Google Scholar] [CrossRef]
- Dargaud, Y.; Wolberg, A.S.; Luddington, R.; Regnault, V.; Spronk, H.; Baglin, T.; Lecompte, T.; Ten Cate, H.; Negrier, C. Evaluation of a Standardized Protocol for Thrombin Generation Measurement Using the Calibrated Automated Thrombogram: An International Multicentre Study. Thromb. Res. 2012, 130, 929–934. [Google Scholar] [CrossRef]
- Murugesan, S.; Xie, J.; Linhardt, R.J. Immobilization of Heparin: Approaches and Applications. Curr. Top. Med. Chem. 2008, 8, 80. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, Q.; Du, J.; Zhu, T.; Chen, D.; Liu, F.; Dong, Y. Nanofibers with Homogeneous Heparin Distribution and Protracted Release Profile for Vascular Tissue Engineering. Front. Bioeng. Biotechnol. 2023, 11, 1187914. [Google Scholar] [CrossRef]
- Zhao, X.; Yang, S.; Lei, R.; Duan, Q.; Li, J.; Meng, J.; Sun, L. Clinical Study on the Feasibility of New Thrombus Markers in Predicting Massive Cerebral Infarction. Front. Neurol. 2023, 13, 942887. [Google Scholar] [CrossRef]
- Huang, Y.; Liu, H.; Zhao, Y.; Chen, H.; Li, Q.; Li, X.; Hua, S.; Cao, D.; Chang, Y. Disrupting Redox Homeostasis for Tumor Therapy Based on PDT/Chemo/Ferroptosis Therapeutic Hybrid Liposomes. RSC Adv. 2024, 14, 20152–20162. [Google Scholar] [CrossRef]
- Wang, H.; Yang, L. Applications of injectable hemostatic materials in wound healing: Principles, strategies, performance requirements, and future perspectives. Theranostics 2023, 13, 4615–4635. [Google Scholar] [CrossRef]
- Sung, Y.K.; Lee, D.R.; Chung, D.J. Advances in the development of hemostatic biomaterials for medical application. Biomater. Res. 2021, 25, 37. [Google Scholar] [CrossRef]
- Bernard, M.; Jubeli, E.; Pungente, M.D.; Yagoubia, N. Biocompatibility of polymer-based biomaterials and medical devices —Regulations, in vitro screening and risk-management. Biomater. Sci. 2018, 6, 2025–2053. [Google Scholar] [CrossRef]
- Braune, S.; Lendlein, A.; Jung, F. Developing standards and test protocols for testing the hemocompatibility of biomaterials. In Hemocompatibility of Biomaterials for Clinical Applications; Siedlecki, C.A., Ed.; Elsevier Ltd.: Amsterdam, The Netherlands, 2018; Chapter 3; pp. 51–76. ISBN 978-0-08-100497-5. [Google Scholar] [CrossRef]
- Guo, Y.; Cheng, N.; Sun, H.; Hou, J.; Zhang, Y.; Wang, D.; Zhang, W.; Chen, Z. Advances in the development and optimization strategies of the hemostatic biomaterials. Front. Bioeng. Biotechnol. 2023, 10, 1062676. [Google Scholar] [CrossRef] [PubMed]
- Saha, A.K.; Zhen, M.Y.S.; Erogbogbo, F.; Ramasubramanian, A.K. Design considerations and assays for hemocompatibility of FDA-approved nanoparticles. Semin. Thromb. Hemost. 2020, 46, 637–652. [Google Scholar] [CrossRef] [PubMed]
- Sperling, C.; Fischer, M.; Maitz, M.F.; Werner, C. Blood Coagulation on Biomaterials Requires the Combination of Distinct Activation Processes. Biomaterials 2009, 30, 4447–4456. [Google Scholar] [CrossRef] [PubMed]
- Gorbet, M.; Sperling, C.; Maitz, M.F.; Siedlecki, C.A.; Werner, C.; Sefton, M.V. The Blood Compatibility Challenge. Part 3: Material Associated Activation of Blood Cascades and Cells. Acta Biomater. 2019, 94, 25–32. [Google Scholar] [CrossRef]
- Kudzin, M.H.; Mrozinska, Z.; Kaczmarek, A.; Chrusciel, J.; Pinar, A.; Sulak, E.; Shah, S.A.R.; Juszczak, M.; Wozniak, K.; Ponczek, M.B. Development of Iron-Modified Cotton Material: Surface Characterization, Biochemical Activity, and Cytotoxicity Assessment. Coatings 2025, 15, 663. [Google Scholar] [CrossRef]
- Kudzin, M.H.; Mrozinska, Z.; Kaczmarek, A.; Chrusciel, J.; Gloc, M.; Zylla, R. Preparation and Biochemical and Microbial Behavior of Poly(Lactide) Composites with Polyethersulfone and Copper-Complexed Cellulose Phosphate. Materials 2025, 18, 2954. [Google Scholar] [CrossRef] [PubMed]
- Ong, S.Y.; Wu, J.; Moochhala, S.M.; Tan, M.H.; Lu, J. Development of a Chitosan-Based Wound Dressing with Improved Hemostatic and Antimicrobial Properties. Biomaterials 2008, 29, 4323–4332. [Google Scholar] [CrossRef]
- Fischer, T.H.; Bode, A.P.; Demcheva, M.; Vournakis, J.N. Hemostatic Properties of Glucosamine-Based Materials. J. Biomed. Mater. Res. Part A 2007, 80, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Iwasaki, Y.; Yamasaki, A.; Ishihara, K. Platelet Compatible Blood Filtration Fabrics Using a Phosphorylcholine Polymer Having High Surface Mobility. Biomaterials 2003, 24, 3599–3604. [Google Scholar] [CrossRef]
- Mollahosseini, A.; Bahig, J.; Shoker, A.; Abdelrasoul, A.; Mollahosseini, A.; Bahig, J.; Shoker, A.; Abdelrasoul, A. Aminolysis-Based Zwitterionic Immobilization on Polyethersulfone Membranes for Enhanced Hemocompatibility: Experimental, Computational, and Ex Vivo Investigations. Biomimetics 2024, 9, 320. [Google Scholar] [CrossRef]
- Handa, H.; Major, T.C.; Brisbois, E.J.; Amoako, K.A.; Meyerhoff, M.E.; Bartlett, R.H. Hemocompatibility Comparison of Biomedical Grade Polymers Using Rabbit Thrombogenicity Model for Preparing Nonthrombogenic Nitric Oxide Releasing Surfaces. J. Mater. Chem. B. Mater. Biol. Med. 2014, 2, 1059. [Google Scholar] [CrossRef]
- Bohl, K.S.; West, J.L. Nitric Oxide-Generating Polymers Reduce Platelet Adhesion and Smooth Muscle Cell Proliferation. Biomaterials 2000, 21, 2273–2278. [Google Scholar] [CrossRef]
- Fleser, P.S.; Nuthakki, V.K.; Malinzak, L.E.; Callahan, R.E.; Seymour, M.L.; Reynolds, M.M.; Merz, S.I.; Meyerhoff, M.E.; Bendick, P.J.; Zelenock, G.B.; et al. Nitric Oxide-Releasing Biopolymers Inhibit Thrombus Formation in a Sheep Model of Arteriovenous Bridge Grafts. J. Vasc. Surg. 2004, 40, 803–811. [Google Scholar] [CrossRef]
- Qiu, X.; Lee, B.L.P.; Ning, X.; Murthy, N.; Dong, N.; Li, S. End-Point Immobilization of Heparin on Plasma-Treated Surface of Electrospun Polycarbonate-Urethane Vascular Graft. Acta Biomater. 2017, 51, 138–147. [Google Scholar] [CrossRef]
- Braghirolli, D.I.; Helfer, V.E.; Chagastelles, P.C.; Dalberto, T.P.; Gamba, D.; Pranke, P. Electrospun Scaffolds Functionalized with Heparin and Vascular Endothelial Growth Factor Increase the Proliferation of Endothelial Progenitor Cells. Biomed. Mater. 2017, 12, 025003. [Google Scholar] [CrossRef] [PubMed]
- Ninivaggi, M.; Apitz-Castro, R.; Dargaud, Y.; De Laat, B.; Hemker, H.C.; Lindhout, T. Whole-Blood Thrombin Generation Monitored with a Calibrated Automated Thrombogram-Based Assay. Clin. Chem. 2012, 58, 1252–1259. [Google Scholar] [CrossRef] [PubMed]





| Polymer Property | Mechanistic Effect | Biological Consequence | Ref. |
|---|---|---|---|
| Positive surface charge (e.g., chitosan) | Attracts fibrinogen/HMWK; promotes FXII binding | Shortened aPTT; enhanced thrombin generation | [4,9,27] |
| Negative charge (e.g., alginate and ORC) | Ca2+ binding; moderate FXII activation | Rapid fibrin polymerization | [26,39] |
| High surface curvature | Induces fibrinogen unfolding; platelet clustering | Accelerated platelet activation | [33,40] |
| Hydration/zwitterionic shell | Prevents protein adsorption | Prolonged aPTT; reduced platelet adhesion | [8,16] |
| Soft mechanical compliance | Limits platelet spreading | Reduced PS exposure | [23] |
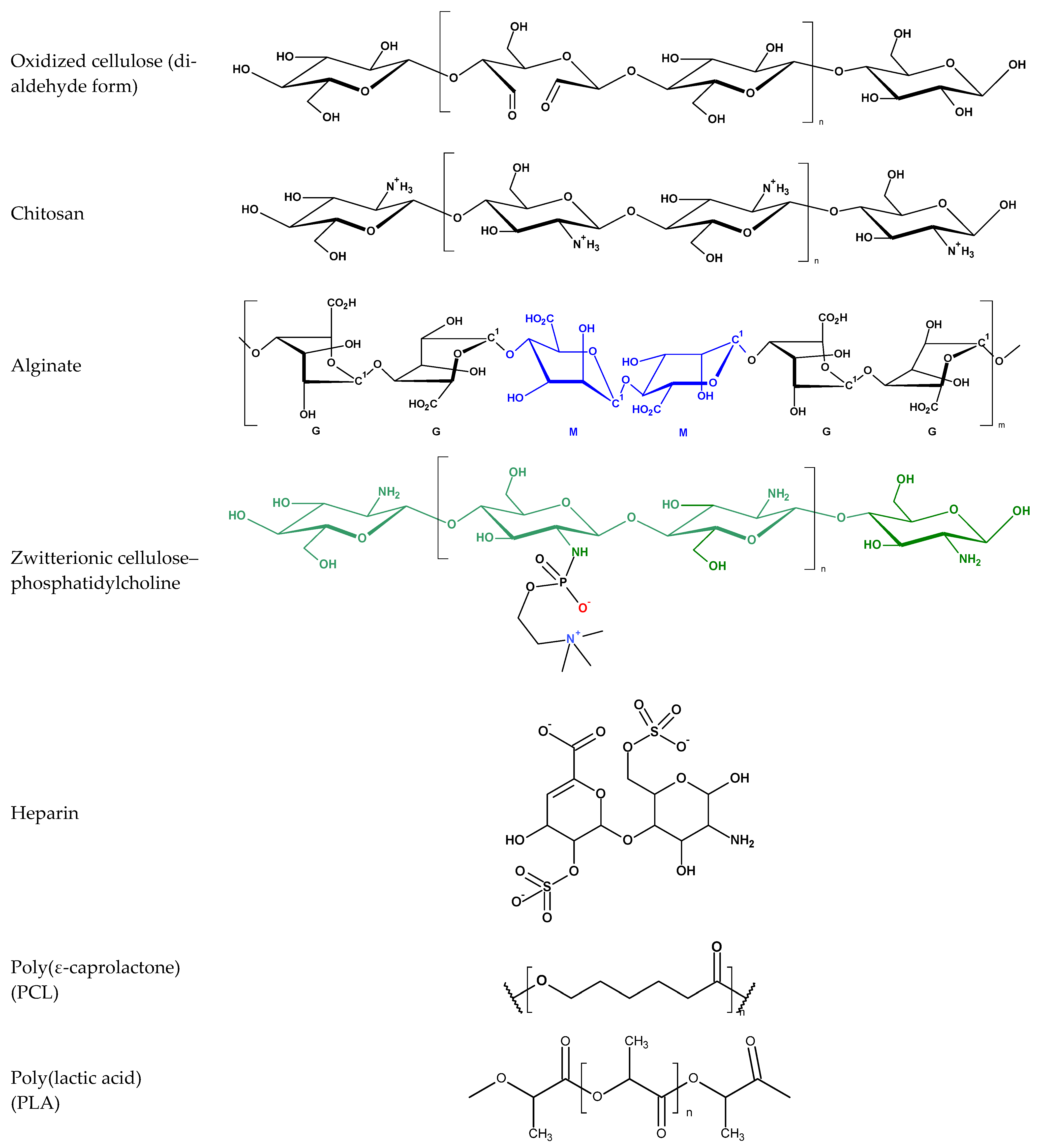
| Polymer | Dominant Functional Group(s) | Typical Surface Charge (pH 7.4) | Biological Effect on Coagulation | Ref. |
|---|---|---|---|---|
| Chitosan | -NH2 (primary amine, protonated) | Positive (cationic) | Promotes platelet adhesion and activation; shortens aPTT; enhances fibrin formation via FXII/HMWK binding | [4,8,9,27,50,51] |
| Alginate | -COO− (carboxylate, Ca2+-crosslinked) | Negative (anionic) | Releases Ca2+; accelerates FX activation and fibrin polymerization; may trigger mild contact activation | [26,27,52,53] |
| Oxidized cellulose (ORC) | -COOH (oxidized hydroxyl groups) | Negative (anionic) | Concentrates plasma and coagulation proteins; promotes fibrin network anchoring; mild FXII activation | [4,26,39,54] |
| PLA/PCL fibers | -COOR (ester), hydrophobic backbone | Neutral/slightly negative | Promotes fibrinogen unfolding and platelet adhesion on curved submicron fibers; enhances thrombin generation | [4,50,55] |
| Heparin | -SO3−, -COO− (sulfated polysaccharide) | Strongly negative | Binds antithrombin III; inhibits FXa and FIIa; prolongs aPTT; suppresses thrombin generation | [4,55,56,57] |
| Zwitterionic polymers (PC, SB, and CB) | -N+(CH3)3/-SO3− or -COO− (dipolar groups) | Net neutral | Forms stable hydration layer; suppresses protein adsorption, FXII/HMWK recruitment, and platelet activation | [8,16,57,58] |
| Characteristic | Natural Polymers | Synthetic Polymers | Ref. |
|---|---|---|---|
| Mechanical Properties | Typically weak; low tensile strength and brittleness | High and configurable; high strength and flexibility | [58,59] |
| Degradation | Enzymatic, rapid, and often difficult to precisely control | Hydrolytic, predictable, and controllable (from weeks to years) | [59,60] |
| Hydration (Hydrophilicity) | Usually high (form hydrogels); excellent water binding | Variable (often hydrophobic) but modifiable | [58,60,61] |
| Rate of Hemostasis | Very high; possess natural protein and platelet-binding motifs (e.g., RGD) | Low; function primarily mechanically unless chemically modified | [59,61,62] |
| Cost | Often lower (renewable raw materials) but with high medical purification costs | High synthesis and certification costs for chemical processes | [62,63] |
| Clinical Readiness | High (widely used in dressings and natural threads) | High for specific groups (e.g., FDA-approved PLA/PGA threads) | [61,63,64] |
| Hybrid Composition | Main Components/Coatings | Mechanistic Synergy | Observed Effect (aPTT/TG/Platelet) | Application | Ref. |
|---|---|---|---|---|---|
| Heparin + Zwitterion | Heparinized base + phosphorylcholine brush | ATIII-mediated enzyme inhibition + antifouling hydration | Prolonged aPTT and ↓ TG peak | Vascular grafts | [68,70,78] |
| NO + Zwitterion | S-nitrosothiol donor in sulfobetaine matrix | Platelet suppression + protein resistance | ↓ P-selectin and delayed TG lag | Catheters and stents | [74,76,77] |
| Heparin + NO | Covalently coupled layers | Enzymatic inhibition + platelet quiescence | ↓ TAT and prolonged TG lag | Dialysis membranes | [71,74,75] |
| Heparin + NO + Zwitterion | Multilayer copolymer | Full control of initiation/amplification/ propagation | Endothelium-like hemocompatibility | Long-term implants | [68,74,77,78,79] |
| Coagulation Metric | Pathway/ Mechanistic Target | Procoagulant Behavior | Anticoagulant Behavior | [Ref.] |
|---|---|---|---|---|
| aPTT (activated partial thromboplastin time) | Intrinsic/contact activation (FXII/HMWK) | Shortened due to enhanced FXII autoactivation and contact factor binding | Prolonged; suppressed FXII binding and reduced HMWK interaction | [33,85,95] |
| PT (prothrombin time) | Extrinsic pathway (tissue factor) | Largely unchanged; minimal sensitivity to contact-driven changes | Largely unchanged | [24,91] |
| TG (thrombin generation) | Global coagulation kinetics (initiation → propagation) | High peak thrombin and ETP; short lag time | Low peak; delayed lag; reduced ETP | [24,93] |
| TAT complex | Cumulative thrombin exposure in vivo | Elevated (sustained thrombin activity) | Reduced (ATIII-mediated FXa/FIIa inhibition) | [92,93,94] |
| Platelet activation (P-selectin and PAC-1) | Amplification/ PS exposure | Strong upregulation; largely irreversible activation | Minimal; reversible adhesion only | [86,87,88,89,90] |
| Complement activation (C3a and SC5b-9) | Inflammatory cross-talk/ foreign surface recognition | Moderate to high | Negligible to low | [33,85] |
| Mechanistic Target | Representative Fiber Chemistry | Dominant Mode of Action | Coagulation Phase Affected | Key Analytical Metric(s) | Typical Application | Ref. |
|---|---|---|---|---|---|---|
| Contact activation inhibition | Zwitterionic (PC, SB, and CB), PEGylated coatings | Hydration-layer formation; suppression of FXII/HMWK adsorption and activation | Initiation | aPTT, FXIIa assays, and protein adsorption studies | Vascular grafts and catheters | [70,78,79] |
| Platelet activation control | Nitric oxide (NO)-releasing fibers | GPIIb/IIIa suppression, reduced PS exposure, inhibition of platelet aggregation | Amplification | TG lag time, P-selectin, and PAC-1 binding | Catheters and stents | [74] |
| Enzymatic propagation suppression | Heparin-functionalized fibers | Antithrombin III-mediated inhibition of FXa/FIIa | Propagation | TG (peak/ETP), TAT complex, and aPTT | Dialysis membranes and extracorporeal circuits | [68,71] |
| Multi-phase control (synergistic) | Hybrid NO–Heparin–Zwitterion fibers | Synergistic antifouling, platelet quiescence, and enzyme inhibition | All phases | aPTT, TG, TAT, and platelet and complement assays | Long-term implants and artificial organs | [68,72,73,74,75,76,77] |
| Design Parameter | Procoagulant Fibers | Anticoagulant/Antithrombogenic Fibers | [Ref.] |
|---|---|---|---|
| Representative Polymers | Chitosan, alginate, oxidized cellulose, and PLA/PCL composites | Heparinized polymers, zwitterionic copolymers, and NO-releasing systems | [33,65,96] |
| Dominant Surface Charge | Positive or moderately negative | Neutral or zwitterionic | [33,65,95] |
| Hydration and Protein Adsorption | Limited hydration; high fibrinogen/HMWK adsorption | Strong hydration; low adsorption (<10 ng cm−2) | [33,65,87] |
| Protein Corona Composition | Fibrinogen, HMWK, and FXII rich | Albumin dominated; native conformations | [33,48,91,92,93,94,95] |
| Platelet Interaction | Strong adhesion; PS exposure; aggregation ↑ | Weak, reversible adhesion; quiescent phenotype | [63,64,69,85] |
| Thrombin Activity | Accelerated generation; high ETP | ATIII-mediated inhibition of FXa/FIIa; low TAT | [72,92,93,94,95] |
| Mechanical Compliance | Stiff, porous matrices promoting clot anchoring | Soft, hydrated coatings preventing mechanotransduction | [48,63,78] |
| Typical Application | Topical hemostats, trauma pads, and surgical dressings | Catheters, vascular grafts, dialyzers, and long-term implants | [48,63,64,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85] |
| Dominant Biological Outcome | Rapid fibrin formation and localized bleeding control | Sustained hemocompatibility and thrombosis prevention | [33,68,72,86] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Kudzin, M.H.; Sikora, M.; Mrozińska, Z.; Chruściel, J.J. Polymeric Fibrous Materials for Procoagulant and Anticoagulant Applications: A Review of Molecular Blood–Material Mechanisms and Strategies. Materials 2026, 19, 539. https://doi.org/10.3390/ma19030539
Kudzin MH, Sikora M, Mrozińska Z, Chruściel JJ. Polymeric Fibrous Materials for Procoagulant and Anticoagulant Applications: A Review of Molecular Blood–Material Mechanisms and Strategies. Materials. 2026; 19(3):539. https://doi.org/10.3390/ma19030539
Chicago/Turabian StyleKudzin, Marcin H., Monika Sikora, Zdzisława Mrozińska, and Jerzy J. Chruściel. 2026. "Polymeric Fibrous Materials for Procoagulant and Anticoagulant Applications: A Review of Molecular Blood–Material Mechanisms and Strategies" Materials 19, no. 3: 539. https://doi.org/10.3390/ma19030539
APA StyleKudzin, M. H., Sikora, M., Mrozińska, Z., & Chruściel, J. J. (2026). Polymeric Fibrous Materials for Procoagulant and Anticoagulant Applications: A Review of Molecular Blood–Material Mechanisms and Strategies. Materials, 19(3), 539. https://doi.org/10.3390/ma19030539






