Influence of Pd Coating Thickness and Pd Content in Sn-Based Solders on Interfacial IMC Formation and Microstructural Evolution in Solder/Ni Joints
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Sn/Au/Pd/Ni Reactions
3.1.1. Sn (50 µm)/Au/Pd (70 nm)/Ni System
3.1.2. Sn (50 µm)/Au/Pd (200 nm)/Ni System
3.1.3. Sn (50 µm)/Au/Pd (1 µm)/Ni System
3.1.4. Sn (20 µm)/Au/Pd (200 nm)/Ni System
3.1.5. Sn (20 µm)/Au/Pd (1 µm)/Ni System
3.1.6. Sn (10 µm)/Au/Pd (70 nm)/Ni System
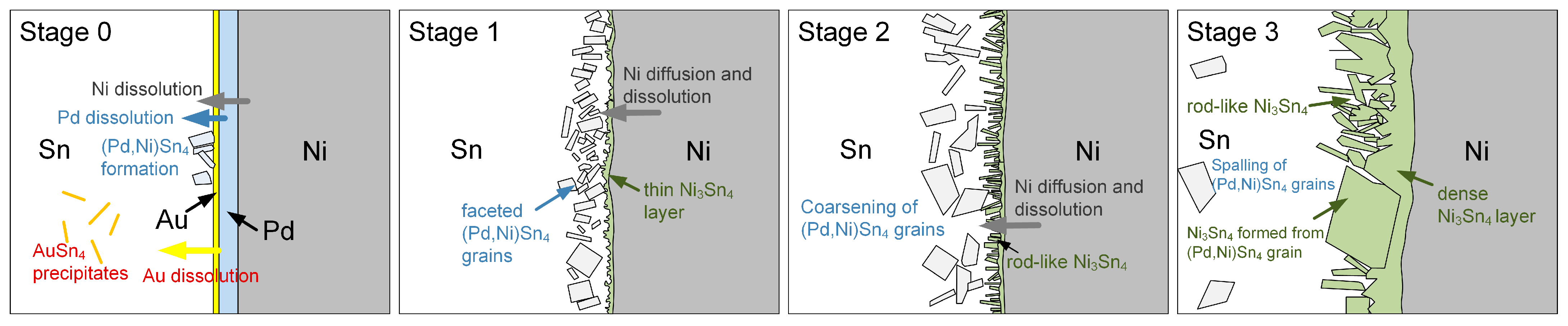
- Stage I: Initial Au and Pd dissolution and (Pd,Ni)Sn4 formation
- Stage II: Concurrent formation of (Pd,Ni)Sn4 and Ni3Sn4 grains
- Stage III: Transformation of (Pd,Ni)Sn4 into thermodynamically stable Ni3Sn4
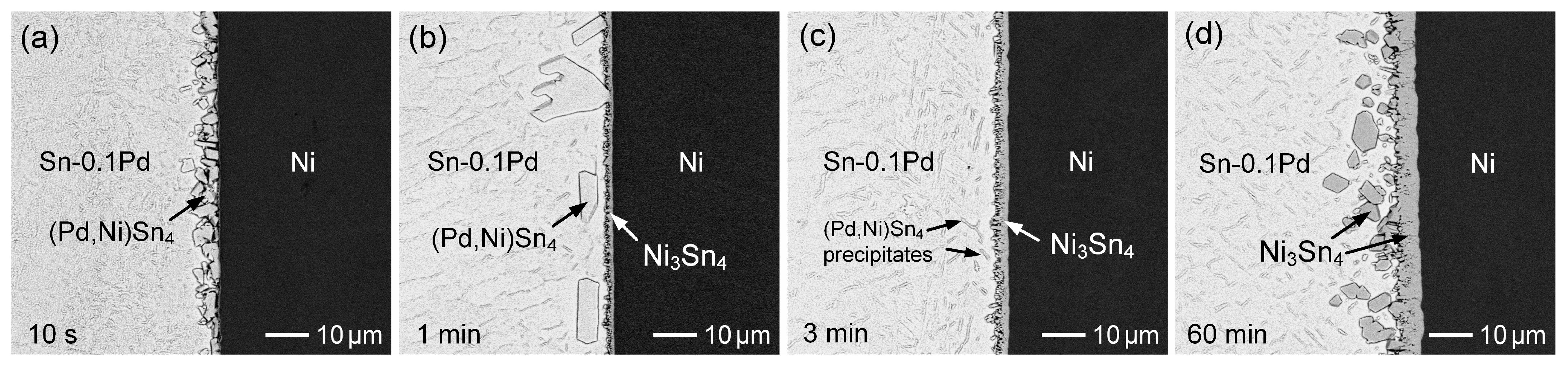
3.2. Sn-Pd/Ni Reactions
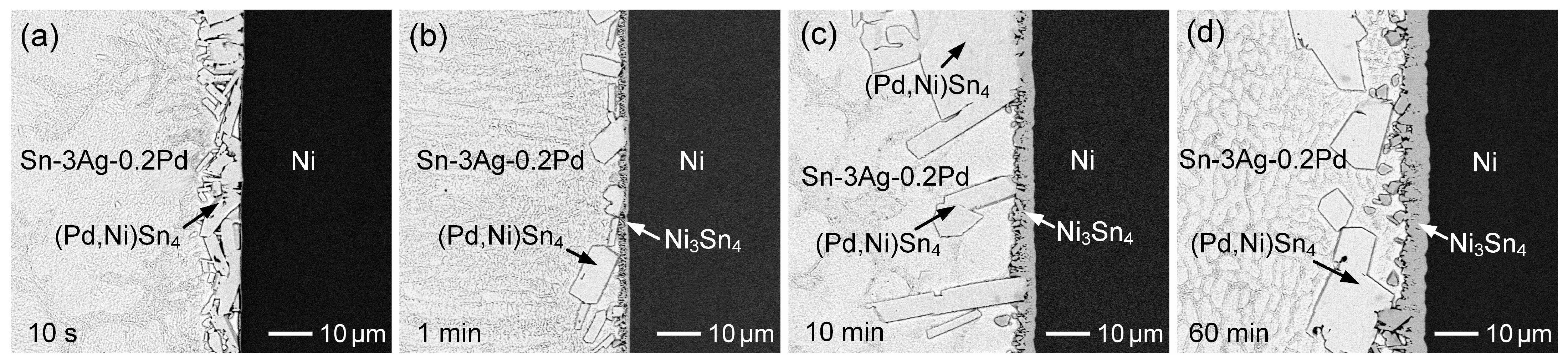
3.3. Sn-3Ag-Pd/Ni Reactions
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gan, C.L.; Huang, C.Y. Advanced Flip Chip Packaging. In Interconnect Reliability in Advanced Memory Device Packaging; Springer Series in Reliability Engineering; Springer: Cham, Switzerland, 2023. [Google Scholar]
- Tang, C.; Zhu, W.; Chen, Z.; Wang, L. Thermomechanical reliability of a Cu/Sn-3.5Ag solder joint with a Ni insertion layer in flip chip bonding for 3D interconnection. J. Mater. Sci. Mater. Electron. 2021, 32, 11893–11909. [Google Scholar] [CrossRef]
- Ho, C.E.; Yang, S.P.; Lee, P.T.; Lee, C.Y.; Chen, C.C.; Kuo, T.T. IMC microstructure modification and mechanical reinforcement of Sn–Ag–Cu/Cu microelectronic joints through an advanced surface finish technique. J. Mater. Res. Technol. 2021, 11, 1895–1910. [Google Scholar] [CrossRef]
- Wang, C.H.; Li, K.T.; Yen, Y.W. Effects of minor Ga addition on interfacial reactions between Sn-Ga solders and Cu. JOM 2024, 76, 2731–2740. [Google Scholar] [CrossRef]
- Ratzker, M.; Pearl, A.; Osterman, M.; Pecht, M.; Milad, G. Review of capabilities of the ENEPIG surface finish. J. Electron. Mater. 2014, 43, 3885–3897. [Google Scholar] [CrossRef]
- Wang, S.; Chen, X.; Luo, K.; Zhou, H.; Li, R.; He, P.; Paik, K.W.; Zhang, S. The design of low-temperature solder alloys and the comparison of mechanical performance of solder joints on ENIG and ENEPIG interface. J. Mater. Res. Technol. 2023, 27, 5332–5339. [Google Scholar] [CrossRef]
- Shen, C.; Hai, Z.; Zhao, C.; Zhang, J.; Evans, J.L.; Bozack, M.J.; Suhling, J.C. Packaging reliability effect of ENIG and ENEPIG surface finishes in board level thermal test under long-term aging and cycling. Materials 2017, 10, 451. [Google Scholar] [CrossRef]
- Chaillot, A.; Venet, N.; Tegehall, P.E.; Hokka, J.; Lortal, J.L. ENEPIG finish: An alternative solution for space printed circuit boards (PCB). In Proceedings of the 2013 Eurpoean Microelectronics Packaging Conference (EMPC), Grenoble, France, 9–12 September 2013; pp. 1–6. [Google Scholar]
- Xiao, J.; Wang, F.; Li, J.; Chen, Z. Comparison of interfacial reactions and isothermal aging of cone Ni-P and flat Ni-P with Sn3.5Ag solders. Appl. Surf. Sci. 2023, 625, 157219. [Google Scholar] [CrossRef]
- Shang, M.; Yao, J.; Xing, J.; Liu, X.; Wang, Y.; Ma, H.; Ma, H. Ni and Ni–P substrates inhibit Bi phase segregation and IMC overgrowth during the soldering process of Sn–Bi solder. Mater. Chem. Phys. 2024, 325, 129726. [Google Scholar] [CrossRef]
- Choi, H.; Kim, C.-L.; Sohn, Y. Diffusion barrier properties of the intermetallic compound layers formed in the Pt nanoparticles alloyed Sn-58Bi solder joints reacted with ENIG and ENEPIG surface finishes. Materials 2022, 15, 8419. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.H.; Li, K.T.; Lin, C.Y. Minor Ga addition to effectively inhibit PdSn4 growth between Sn solder and Pd substrate. Intermetallics 2015, 67, 102–110. [Google Scholar] [CrossRef]
- Liu, C.Y.; Wei, Y.S.; Lin, E.J.; Hsu, Y.C.; Tang, Y.K. Study of Sn and SnAgCu solders wetting reaction on Ni/Pd/Au substrates. J. Electron. Mater. 2016, 45, 6079–6085. [Google Scholar] [CrossRef]
- Kim, Y.M.; Park, J.Y.; Kim, Y.H. Effect of Pd thickness on the interfacial reaction and shear strength in solder joints between Sn-3.0Ag-0.5Cu solder and electroless nickel/electroless palladium/immersion Gold (ENEPIG) surface finish. J. Electron. Mater. 2012, 41, 763–773. [Google Scholar] [CrossRef]
- Lee, C.Y.; Yang, S.P.; Yang, C.H.; Lu, M.K.; Kuo, T.T.; Ho, C.E. Influence of Pd(P) thickness on the Pd-free solder reaction between eutectic Sn-Ag alloy and Au/Pd(P)/Ni(P)/Cu multilayer. Surf. Coat. Technol. 2020, 395, 125879. [Google Scholar]
- Wu, Y.S.; Lee, P.T.; Hsieh, W.Z.; Kuo, T.T.; Ho, C.E. Interfacial reaction and mechanical reliability between Sn-3Ag-0.5Cu alloy and ultrathin-Ni(P)-type Au/Pd(P)/Ni(P)/Cu metallization pad. Surf. Coat. Technol. 2019, 359, 374–383. [Google Scholar] [CrossRef]
- Kim, J.; Back, J.H.; Jung, S.B.; Yoon, J.W. Interfacial reactions and mechanical properties of Sn–3.0Ag–0.5Cu solder with pure Pd or Pd(P) layers containing thin-Au/Pd/Ni(P) surface-finished PCBs during aging. J. Mater. Sci. Mater. Electron. 2020, 31, 4027–4039. [Google Scholar] [CrossRef]
- Yoon, J.W.; Back, J.H.; Jung, S.B. Comparative study of ENEPIG and thin ENEPIG as surface finishes for SAC305 solder joints. J. Mater. Sci. Mater. Electron. 2018, 29, 4724–4731. [Google Scholar]
- Oda, Y.; Fukumuro, N.; Yae, S. Intermetallic compound growth between electroless nickel/electroless palladium/immersion gold surface finish and Sn-3.5Ag or Sn-3.0Ag-0.5Cu solder. J. Electron. Mater. 2018, 47, 2507–2511. [Google Scholar]
- Wang, C.H.; Li, K.T. Study of electromigration resistance of (Pd,Ni)Sn4 phase in lead-free solder joints. J. Alloys Compds. 2016, 654, 546–553. [Google Scholar] [CrossRef]
- Ho, C.E.; Lin, S.W.; Lin, Y.C. Effects of Pd concentration on the interfacial reaction and mechanical reliability of the Sn–Pd/Ni system. J. Alloys Compds. 2011, 509, 7749–7757. [Google Scholar] [CrossRef]
- Ho, C.E.; Hsu, L.H.; Lin, S.W.; Rahman, M.A. Influence of Pd concentration on the interfacial reaction and mechanical reliability of the Ni/Sn-Ag-Cu-xPd System. J. Electron. Mater. 2012, 41, 2–10. [Google Scholar]
- Belyakov, S.A.; Gourlay, C.M. NiSn4 formation in as-soldered Ni-Sn and ENIG-Sn couples. J. Electron. Mater. 2012, 41, 3331–3341. [Google Scholar] [CrossRef]
- Belyakov, S.A.; Gourlay, C.M. NiSn4 formation during the solidification of Sn–Ni alloys. Intermetallics 2012, 25, 48–59. [Google Scholar] [CrossRef]
- Rahman, M.A.; Ho, C.E.; Gierlotka, W.; Kuo, J.C. Experimental determination and thermodynamic modeling of the Sn-rich corner of the ternary Ni-Pd-Sn phase diagram at 250 °C. J. Electron. Mater. 2014, 43, 4582–4593. [Google Scholar] [CrossRef]
- Horváth, B.; Illés, B.; Shinohara, T. Growth of intermetallics between Sn/Ni/Cu, Sn/Ag/Cu and Sn/Cu layered structures. Thin Solid Films 2014, 556, 345–353. [Google Scholar] [CrossRef]
- Jandl, I.; Richter, K.W. The Sn-rich corner of the system Ni-Pd-Sn: A phase diagram study. J. Alloys Compds. 2017, 697, 310–317. [Google Scholar] [CrossRef]
- Wang, C.H.; Liu, J.L. Effects of Sn thickness on morphology and evolution of Ni3Sn4 grains formed between molten Sn and Ni substrate. Intermetallics 2015, 61, 9–15. [Google Scholar] [CrossRef]
- Görlich, J.; Baither, D.; Schmitz, G. Reaction kinetics of Ni/Sn soldering reaction. Acta Mater. 2010, 58, 3187–3197. [Google Scholar] [CrossRef]















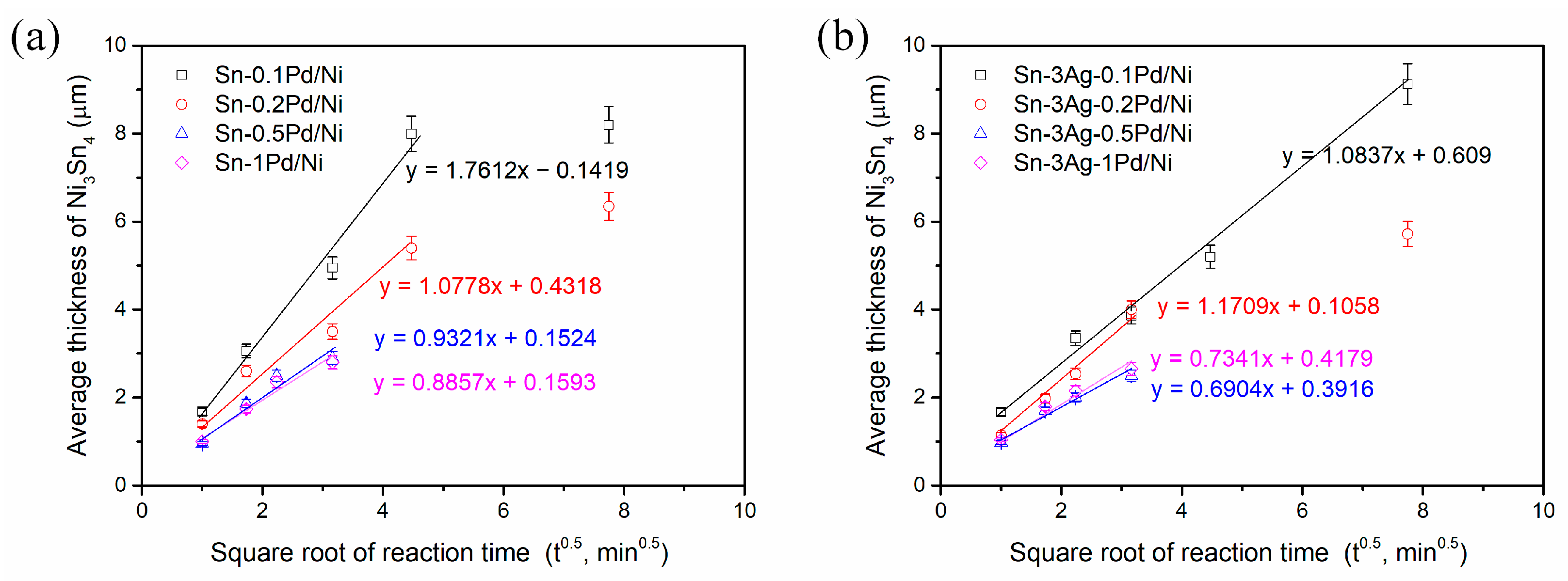
| Sn/Au/Pd/Ni Systems | Sn-Pd/Ni Systems | Sn-3Ag-Pd/Ni Systems |
|---|---|---|
| Sn (50 µm)/Au/Pd (70 nm)/Ni | Sn-0.1 wt.%Pd | Sn-3Ag-0.1Pd/Ni (wt.%) |
| Sn (50 µm)/Au/Pd (200 nm)/Ni | Sn-0.2 wt.%Pd | Sn-3Ag-0.2Pd/Ni |
| Sn (50 µm)/Au/Pd (1 µm)/Ni | Sn-0.5 wt.%Pd | Sn-3Ag-0.5Pd/Ni |
| Sn (20 µm)/Au/Pd (200 nm)/Ni | Sn-1 wt.%Pd | Sn-3Ag-1Pd/Ni |
| Sn (20 µm)/Au/Pd (1 µm)/Ni | ||
| Sn (10 µm)/Au/Pd (70 nm)/Ni |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Wang, C.-H.; Li, C.-A.; Li, K.-T.; Chiu, H.-W. Influence of Pd Coating Thickness and Pd Content in Sn-Based Solders on Interfacial IMC Formation and Microstructural Evolution in Solder/Ni Joints. Materials 2026, 19, 526. https://doi.org/10.3390/ma19030526
Wang C-H, Li C-A, Li K-T, Chiu H-W. Influence of Pd Coating Thickness and Pd Content in Sn-Based Solders on Interfacial IMC Formation and Microstructural Evolution in Solder/Ni Joints. Materials. 2026; 19(3):526. https://doi.org/10.3390/ma19030526
Chicago/Turabian StyleWang, Chao-Hong, Chu-An Li, Kuan-Ting Li, and Hsuan-Wei Chiu. 2026. "Influence of Pd Coating Thickness and Pd Content in Sn-Based Solders on Interfacial IMC Formation and Microstructural Evolution in Solder/Ni Joints" Materials 19, no. 3: 526. https://doi.org/10.3390/ma19030526
APA StyleWang, C.-H., Li, C.-A., Li, K.-T., & Chiu, H.-W. (2026). Influence of Pd Coating Thickness and Pd Content in Sn-Based Solders on Interfacial IMC Formation and Microstructural Evolution in Solder/Ni Joints. Materials, 19(3), 526. https://doi.org/10.3390/ma19030526






