Comparative Analysis of Industrial Fused Magnesia from Natural and Flotation-Processed Magnesite: Associations Among CaO/SiO2 Ratio, Silicate Phase Formation, and Microcracking
Highlights
- Reverse flotation coincided with higher CaO/SiO2 ratio (0.68 to 2.85).
- Higher CaO/SiO2 coincided with C2S dominance; a lower ratio coincided with CMS.
- More C2S coincided with more microcracks and slightly lower densification.
- Beneficiation may benefit from controlling oxide balance, not only impurities.
- Tailoring CaO/SiO2 may help avoid conditions linked to C2S and microcracking.
- The findings are derived from industrial comparison; causality requires controlled experiments.
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Materials
2.2. Preparation of Fused Magnesia
2.3. Characterization
3. Results
3.1. Physicochemical Properties of Raw Materials
3.2. Effect of Impurity Occurrence
3.3. Microstructure and Densification Behavior of Fused Magnesia
4. Discussion
5. Conclusions
- (1)
- The impurities in Dashiqiao magnesite exhibit diverse modes of occurrence. Calcium is predominantly found as dolomite, occurring either as intergranular grains or within crystal grains. Silicon and aluminum are present in silicate minerals, including quartz, talc, and chlorite. Iron substitutes isomorphically within the magnesite lattice in the form of a solid solution, while phosphorus and fluorine are primarily hosted within fluorapatite.
- (2)
- Reverse flotation proves highly effective in removing intergranular silicate impurities, achieving a silica (SiO2) removal efficiency exceeding 97%. However, it shows limited efficacy in eliminating intragranular dolomite and iron incorporated in solid solution within the magnesite lattice. This selective removal is associated with the change in the CaO/SiO2 (C/S) ratio, which increases from 0.68 to 2.85 after flotation.
- (3)
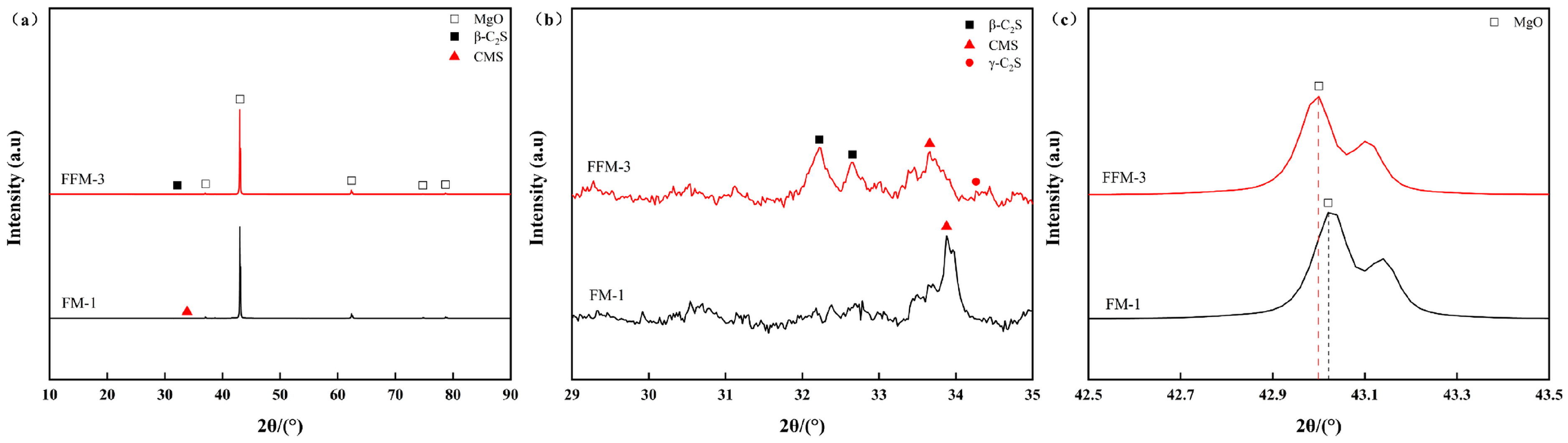
- In the fused magnesia derived from the flotation concentrate, the elevated C/S ratio is associated with the precipitation of β-C2S. Microcrack formation was observed, consistent with the volumetric expansion associated with the β → γ C2S transformation during cooling.
- (4)
- The results suggest that for the flotation–fusion route, managing the CaO/SiO2 (C/S) ratio in addition to total impurities may help avoid conditions associated with transformation-prone Ca–silicates and potentially improve microstructural stability.
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zheng, Z. Review of the metallogenic regularity of magnesite deposits in China. Acta Geol. Sin. Engl. Ed. 2015, 89, 1761. (In Chinese) [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, G. A technology of preparing honeycomb-like structure MgO from low grade magnesite. Int. J. Miner. Process. 2014, 126, 35–40. [Google Scholar] [CrossRef]
- Ghosh, C.; Sinhamahapatra, S.; Tripathi, H.S.; Sarkar, U. Reverse flotation of natural magnesite and process optimization using response surface methodology. Trans. Indian Ceram. Soc. 2020, 79, 23–29. [Google Scholar] [CrossRef]
- Liu, Z.; Cheng, K.; Qiu, W.; Zhang, R.; Pan, S.; Gao, Y.; Wen, T.; Zhang, L.; Yuan, L.; Yu, J. Comprehensive review of the corrosion behavior of magnesia–based refractories by molten steel slag. J. Ind. Eng. Chem. 2025, 145, 123–143. [Google Scholar] [CrossRef]
- Ghosh, A.; Haldar, M.K.; Das, S.K. Effect of MgO and ZrO2 additions on the properties of magnesite-chrome composite refractory. Ceram. Int. 2007, 33, 821–825. [Google Scholar] [CrossRef]
- Gokce, A.S.; Gurcan, C.; Ozgen, S.; Aydin, S. The effect of antioxidants on the oxidation behaviour of magnesia–carbon refractory bricks. Ceram. Int. 2008, 34, 323–330. [Google Scholar] [CrossRef]
- Xiao, H.; Cheng, Y.; Yang, Q.; Senda, T. Mechanical and tribological properties of calcia–magnesia–alumina–silica-based glass–ceramics prepared by in situ crystallization. Mater. Sci. Eng. A 2006, 423, 170–174. [Google Scholar] [CrossRef]
- Li, Q.; Yin, W.Z.; Zhu, D.S.; Lv, Z.F. Flotation and purification research on low grade magnesite in Kuandian of Liaoning. Adv. Mater. Res. 2010, 92, 97–102. [Google Scholar] [CrossRef]
- Han, J.H.; Li, X.A.; Dai, S.J.; Wang, Z.Y.; Yu, L.T. Experimental study on purification of magnesite ore in Liaoning Province. Adv. Mater. Res. 2013, 826, 48–52. [Google Scholar] [CrossRef]
- Li, C.; Sun, C.Y.; Wang, Y.L.; Fu, Y.F.; Xu, P.Y.; Yin, W.Z. Research on new beneficiation process of low-grade magnesite using vertical roller mill. J. Iron Steel Res. Int. 2020, 27, 11. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, W.; Han, C.; Hao, H. Effects of monohydric alcohols on the flotation of magnesite and dolomite by sodium oleate. J. Mol. Liq. 2017, 249, 1060–1067. [Google Scholar] [CrossRef]
- Tang, Y.; Yin, W.; Kelebek, S. Selective flotation of magnesite from calcite using potassium cetyl phosphate as a collector in the presence of sodium silicate. Miner. Eng. 2019, 146, 106154. [Google Scholar] [CrossRef]
- Assanelli, M.; Luoni, P.; Rebay, G.; Roda, M.; Spalla, M.I. Tectono-metamorphic evolution of serpentinites from Lanzo Valleys subduction complex (Piemonte—Sesia-Lanzo Zone boundary, Western Italian Alps). Minerals 2020, 10, 985. [Google Scholar] [CrossRef]
- Chan, C.J.; Kriven, W.M.; Young, J.F. Physical stabilization of the β→γ transformation in dicalcium silicate. J. Am. Ceram. Soc. 1992, 75, 1621–1627. [Google Scholar] [CrossRef]
- Yao, J.; Hou, Y.; Wang, Y.L.; Zhong, W.X.; Yin, W.Z. Research on the floatability of magnesite and its gangue minerals with sodium oleate and lauryl amine as collectors. Adv. Mater. Res. 2013, 798–799, 328–332. [Google Scholar] [CrossRef]
- Aslani, S.; Hashemi, H.R.S.B.; Arianpour, F. Beneficiation of Iranian magnesite ores by reverse flotation process and its effects on shaped and unshaped refractories properties. Bull. Mater. Sci. 2010, 33, 697–705. [Google Scholar] [CrossRef]
- Zhang, R.; Liu, Z.; Yu, J. Removal of silicon from magnesite by flotation: Influence of particle size and mechanical mechanism. Materials 2023, 16, 6095. [Google Scholar] [CrossRef]
- Dai, S.J.; Li, X.A.; Yang, S.Y.; Yu, L.T. The flotation experiment study on a high silicon low-grade magnesite in Liaoning Province. Adv. Mater. Res. 2012, 454, 352–356. [Google Scholar] [CrossRef]
- Hoda, S.; Rashani, S.; Berisha, K.; Shaqiri, M. Enriching of magnesite ore containing dolomite. Kem. Ind. 2010, 59, 489–497. [Google Scholar]
- Li, M.; Yang, C.; Wu, Z.; Gao, X.; Tong, X.; Yu, X.; Long, H. Selective depression action of taurine in flotation separation of specularite and chlorite. J. Min. Sci. Technol. Engl. Ed. 2022, 3, 32. (In Chinese) [Google Scholar] [CrossRef]
- Chen, G.L.; Tao, D. Reverse flotation of magnesite by dodecyl phosphate from dolomite in the presence of sodium silicate. Sep. Sci. Technol. 2005, 39, 377–390. [Google Scholar] [CrossRef]
- Gence, N.; Zda, H. Surface properties of magnesite and surfactant adsorption mechanism. Int. J. Miner. Process. 1995, 43, 37–47. [Google Scholar] [CrossRef]
- Cilek, E.C. The effect of hydrodynamic conditions on true flotation and entrainment in flotation of a complex sulphide ore. Int. J. Miner. Process. 2009, 90, 35–44. [Google Scholar] [CrossRef]
- Bentli, I.; Erdogan, N.; Elmas, N.; Kaya, M. Magnesite concentration technology and caustic-calcined product from Turkish magnesite middlings by calcination and magnetic separation. Sep. Sci. Technol. 2017, 52, 1129–1142. [Google Scholar] [CrossRef]
- Qin, W.; Hu, J.; Zhu, J.C. Effect of depressants on flotation separation of magnesite from dolomite and calcite. Int. J. Min. Sci. Technol. 2023, 33, 83–91. [Google Scholar] [CrossRef]
- Chen, J.H.; Zhu, Y.G. Study of semi-constrained properties of metal ions on mineral surface of flotation system. J. China Univ. Min. Technol. 2021, 50, 1181–1188. (In Chinese) [Google Scholar]
- Yin, X.; Gupta, V.; Du, H.; Wang, X.; Miller, J.D. Surface charge and wetting characteristics of layered silicate minerals. Adv. Colloid Interface Sci. 2012, 179–182, 43–50. [Google Scholar] [CrossRef]
- Xu, L.; Tian, J.; Wu, H.; Fang, S.; Lu, Z.; Ma, C.; Sun, W.; Hu, Y. Anisotropic surface chemistry properties and adsorption behavior of silicate mineral crystals. Adv. Colloid Interface Sci. 2018, 256, 340–351. [Google Scholar] [CrossRef]
- Bai, J.; Wang, J.; Yin, W.; Chen, X. Influence of sodium phosphate salts with different chain length on the flotation behavior of magnesite and dolomite. Minerals 2020, 10, 1031. [Google Scholar] [CrossRef]
- Li, M.Y.; Lian, D.; Li, H.; Chen, W.X.; Hu, Y.M.; Gao, X.P.; Tong, X. Study on the anisotropy of chlorite crystals and its relationship with their floatability. Met. Mine 2020, 6, 56–61. [Google Scholar] [CrossRef]
- Yao, J.; Yin, W.; Gong, E. Depressing effect of fine hydrophilic particles on magnesite reverse flotation. Int. J. Miner. Process. 2016, 149, 84–93. [Google Scholar] [CrossRef]
- You, J.; Zhao, X.; Qian, G.; Zhang, X.F.; Zhang, G.; Ma, B. Development status of fused magnesia in the southern Liaoning region. Refractories 2024, 58, 357–361. (In Chinese) [Google Scholar]
- GB/T 2999-2016; Refractory Materials—Determination of Bulk Density of Granular Materials. Standardization Administration of China (SAC): Beijing, China, 2016.
- DZ/T 0348-2020; Specifications for Magnesite and Dolomite Mineral Exploration. Geological Publishing House: Beijing, China, 2020. (In Chinese)
- Xi, Y.; Dai, S.J.; Guo, X.F. Experimental study on reverse flotation of magnesite ores in Haicheng. Non. Met. Mines 2018, 41, 76–78. (In Chinese) [Google Scholar]
- Qi, X.; Luo, X.D.; Li, Z.; Zhou, Y.P.; Pan, Z.Z. Research progress on mineral processing purification and application of high-silica magnesite. Bull. Chin. Ceram. Soc. 2021, 40, 485–492. (In Chinese) [Google Scholar]
- Ma, Z.C.; Ma, Y.Q.; Yang, C.Y. Investigation on reverse flotation for removing silicate from a low-grade magnesite with high silicate. Non Ferr. Min. Metall. 2018, 34, 26–29. (In Chinese) [Google Scholar]
- Brand, O.P.R.G.; Poling, G.W. Anionic flotation of magnesite. Can. Metall. Q. 1982, 21, 211–220. [Google Scholar] [CrossRef]
- Wang, W.; Gu, H.; Zhai, Y.; Bai, J.; Deng, W. Separation of Mg and Si from low-grade magnesite through sulfuric acid leaching method. In Proceedings of the 2011 International Conference on Remote Sensing, Environment and Transportation Engineering, Nanjing, China, 24–26 June 2011; pp. 7819–7822. [Google Scholar] [CrossRef]
- Yao, J.; Sun, H.; Yang, B.; Zhou, Y.; Yin, W.; Zhu, Z. Selective co-adsorption of a novel mixed collector onto magnesite surface to improve the flotation separation of magnesite from dolomite. Powder Technol. 2020, 371, 180–189. [Google Scholar] [CrossRef]
- Sun, W.; Liu, W.; Dai, S.; Yang, T.; Duan, H.; Liu, W. Effect of Tween 80 on flotation separation of magnesite and dolomite using NaOL as the collector. J. Mol. Liq. 2020, 315, 113712. [Google Scholar] [CrossRef]
- Chen, G.; Tao, D. Effect of solution chemistry on flotability of magnesite and dolomite. Int. J. Miner. Process. 2004, 74, 343–357. [Google Scholar] [CrossRef]
- Wright, K.; Cygan, R.T.; Slater, B. Structure of the (104) surfaces of calcite, dolomite and magnesite under wet and dry conditions. Phys. Chem. Chem. Phys. 2000, 3, 839–844. [Google Scholar] [CrossRef]
- Azizi, D.; Larachi, F. Surface interactions and flotation behavior of calcite, dolomite and ankerite with alkyl hydroxamic acid bearing collector and sodium silicate. Colloids Surf. A 2018, 537, 127–138. [Google Scholar] [CrossRef]
- Chen, Z.H.; Guo, X.F.; Wang, Q.Q. Iron and silicon removal test on a low-grade magnesite in Haicheng, Liaoning Province. Mod. Min. 2017, 33, 53–54+57. (In Chinese) [Google Scholar]
- Yao, J.; Xue, J.; Li, D.; Fu, Y.; Gong, E.; Yin, W. Effects of fine–coarse particles interaction on flotation separation and interaction energy calculation. Particul. Sci. Technol. 2016, 35, 733–741. [Google Scholar] [CrossRef]
- Richards, R.G.; Jones, T.A. The Kelsey centrifugal Jig—An update on technology and application. Miner. Metall. Process. 2004, 21, 179–182. [Google Scholar] [CrossRef]
- Guo, W.; Zhu, Y.; Han, Y.; Li, Y.; Yuan, S. Flotation performance and adsorption mechanism of a new collector 2-(carbamoylamino) lauric acid on quartz surface. Miner. Eng. 2020, 153, 106343. [Google Scholar] [CrossRef]
- Gao, G.; Sun, H.; Wang, Y.; Sun, Y.; Han, H.; Yuan, Z. Study on the separation effects of the novel collector ODD on magnesite and quartz. Physicochem. Probl. Miner. Process. 2024, 60, 12. [Google Scholar] [CrossRef]
- Silvester, E.J.; Bruckard, W.J.; Woodcock, J.T. Surface and chemical properties of chlorite in relation to its flotation and depression. Miner. Process. Extr. Metall. 2013, 120, 65–70. [Google Scholar] [CrossRef]
- Li, C.; Gao, Z. Tune surface physicochemical property of fluorite particles by regulating the exposure degree of crystal surfaces. Miner. Eng. 2018, 128, 123–132. [Google Scholar] [CrossRef]
- Li, C.; Gao, Z. Effect of grinding media on the surface property and flotation behavior of scheelite particles. Powder Technol. 2017, 322, 386–392. [Google Scholar] [CrossRef]
- Yehia, A.; Al-Wakeel, M. Role of ore mineralogy in selecting beneficiation route for magnesite-dolomite separation. Physicochem. Probl. Miner. Process. 2013, 49, 525–534. [Google Scholar] [CrossRef]
- Yin, W.Z.; Tang, Y. Interactive effect of minerals on complex ore flotation: A brief review. Int. J. Miner. Metall. Mater. 2020, 27, 571–583. [Google Scholar] [CrossRef]
- Nayak, N.P.; Pal, B.K. Separation behaviour of iron ore fines in Kelsey Centrifugal Jig. J. Miner. Mater. Charact. Eng. 2013, 1, 85–89. [Google Scholar] [CrossRef][Green Version]
- Jung, I.H.; Decterov, S.A.; Pelton, A.D. Critical thermodynamic evaluation and optimization of the CaO–MgO–SiO2 system. J. Eur. Ceram. Soc. 2005, 25, 313–333. [Google Scholar] [CrossRef]
- Lampropoulou, P.G.; Katagas, C.G.; Papamantellos, D.C. Composition of periclase and calcium-silicate phases in magnesia refractories derived from natural microcrystalline magnesite. J. Am. Ceram. Soc. 2005, 88, 1568–1574. [Google Scholar] [CrossRef]
- Hao, S.; Luo, G.P.; Lu, Y.Y.; Chai, Y.F.; An, S.L.; Zang, B.K. Effect of fluorine and aluminum doping on the polymorphic transformation and equilibrium phase composition of C2S. J. Funct. Mater. 2023, 54, 5230–5236. (In Chinese) [Google Scholar]








| Sample NO. | MgO | CaO | Al2O3 | SiO2 | Fe2O3 | LOI | C/S (n) |
|---|---|---|---|---|---|---|---|
| DSQLM-1 | 46.92 | 0.78 | 0.48 | 1.06 | 0.57 | 50.19 | 0.79 |
| DSQLM-2 | 45.90 | 0.80 | 0.06 | 3.07 | 0.65 | 49.52 | 0.24 |
| DSQLM-3 | 41.48 | 1.18 | 0.76 | 8.68 | 0.88 | 47.02 | 0.15 |
| F-DSQLM-3 | 47.55 | 0.70 | 0.14 | 0.21 | 0.29 | 51.11 | 3.57 |
| Weight Percentage of Elements, wt.% | Phase Composition | Distribution Characteristics | Flotation Difficulty | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mg | Al | Ca | Si | Fe | C | O | P | F | ||||
| 1 | 13.99 | — | 22.71 | — | — | 12.24 | 51.07 | — | — | Dolomite | Intergranular granular | Difficult |
| 2 | 13.52 | — | 21.28 | — | — | 13.00 | 52.20 | — | — | Dolomite | Fine-grained intergranular | Very Difficult |
| 3 | 28.27 | — | — | — | 0.20 | 14.35 | 57.18 | — | — | Iron-rich magnesite | Intragranular | Very Difficult |
| 4 | — | — | — | 45.75 | — | — | 54.25 | — | — | Quartz | Fracture filling | Simple |
| 5 | 25.87 | 7.89 | — | 16.96 | — | — | 48.39 | — | 0.89 | Fluorine-containing chlorite | Fracture filling | Difficult |
| 6 | 14.00 | — | 22.71 | — | — | 12.24 | 51.05 | — | — | Dolomite | Intergranular granular | Difficult |
| 7 | 14.19 | — | 20.35 | — | — | 13.05 | 52.41 | — | — | Dolomite | Intragranular | Very Difficult |
| 8 | 28.27 | — | — | — | 0.19 | 14.35 | 57.19 | — | — | Iron-rich magnesite | Intragranular | Very Difficult |
| 9 | — | — | — | 46.70 | — | — | 53.30 | — | — | Quartz | Fracture filling | Simple |
| 10 | — | — | — | 47.02 | — | — | 52.98 | — | — | Quartz | Fracture filling | Simple |
| 11 | 18.16 | 6.71 | 0.19 | 16.16 | — | 5.57 | 53.21 | — | — | chlorite | Fracture filling | Difficult |
| 12 | 1.69 | — | 36.06 | — | — | 12.73 | 49.52 | — | — | Calcite | Fracture filling | Simple |
| 13 | — | — | — | 46.82 | — | — | 53.18 | — | — | Quartz | Fracture filling | Simple |
| 14 | 27.48 | — | 0.53 | — | — | 14.60 | 57.39 | — | — | Calcium-rich magnesite | — | — |
| 15 | — | — | — | — | 70.71 | — | 29.29 | — | — | Limonite | Fracture filling | Difficult |
| 16 | — | — | 28.22 | — | — | 5.12 | 45.18 | 15.70 | 5.78 | Fluorapatite | Fine-grained intergranular | Difficult |
| 17 | 27.66 | — | — | — | — | 14.77 | 57.56 | — | — | magnesite | — | — |
| 18 | 13.81 | — | 20.51 | — | — | 13.21 | 52.47 | — | — | Dolomite | Intragranular | Very Difficult |
| 19 | 15.22 | — | — | 27.73 | — | 4.22 | 52.84 | — | — | Talc | Fracture filling | Difficult |
| 20 | 22.75 | 8.88 | 0.27 | 20.70 | — | — | 45.56 | — | 0.84 | Fluor–chlorite | Fracture filling | Difficult |
| 21 | — | — | — | 43.33 | — | — | 56.67 | — | — | Quartz | Fracture filling | Simple |
| 22 | 27.66 | — | — | — | 0.19 | 14.77 | 57.37 | — | — | Iron-rich magnesite | Intragranular | Difficult |
| Sample | MgO | CaO | Al2O3 | SiO2 | Fe2O3 | LOI | B.D. (g/cm3) | C/S (n) | Number of Specimens | Statistical Significance |
|---|---|---|---|---|---|---|---|---|---|---|
| FM-1 | 97.25 | 0.73 | 0.20 | 1.15 | 0.53 | 0.14 | 3.46 | 0.68 | 5 | No |
| FFM-3 | 97.61 | 1.09 | 0.18 | 0.41 | 0.56 | 0.15 | 3.45 | 2.85 | 5 | No |
| C/S (n) | Phase Composition and Initial Melting Temperatures of Impurity Phases |
|---|---|
| <1 | MgO, M2S (1890), CMS (1495) |
| 1–1.5 | MgO, CMS (1495), C3MS2 (1550) |
| 1.5–2 | MgO, C3MS2 (1550), C2S (2130) |
| >2 | MgO, C2S (2130) |
| 2–3 | MgO, C2S (2130), C3S (1900) |
| MgO | CMS | Ca3(PO4)2 | β-C2S | γ-C2S | wRp | |
|---|---|---|---|---|---|---|
| FM-1 | 95.83 | 3.12 | 1.05 | — | — | 5.75 |
| FFM-3 | 98.24 | — | — | 1.51 | 0.25 | 6.25 |
| Mg | Si | Ca | P | O | Possible Compositions | |
|---|---|---|---|---|---|---|
| 1 | 17.42 | 18.56 | 22.46 | — | 41.57 | CMS |
| 2 | — | 1.35 | 39.02 | 18.56 | 41.07 | Ca3(PO4)2 |
| 3 | 1.02 | 15.01 | 46.03 | 0.78 | 37.16 | C2S |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Wang, C.; Luan, J.; Yang, Z.; Ma, Q.; Wang, G.; Zang, X. Comparative Analysis of Industrial Fused Magnesia from Natural and Flotation-Processed Magnesite: Associations Among CaO/SiO2 Ratio, Silicate Phase Formation, and Microcracking. Materials 2026, 19, 463. https://doi.org/10.3390/ma19030463
Wang C, Luan J, Yang Z, Ma Q, Wang G, Zang X. Comparative Analysis of Industrial Fused Magnesia from Natural and Flotation-Processed Magnesite: Associations Among CaO/SiO2 Ratio, Silicate Phase Formation, and Microcracking. Materials. 2026; 19(3):463. https://doi.org/10.3390/ma19030463
Chicago/Turabian StyleWang, Chunyan, Jian Luan, Zhitao Yang, Qigang Ma, Gang Wang, and Ximin Zang. 2026. "Comparative Analysis of Industrial Fused Magnesia from Natural and Flotation-Processed Magnesite: Associations Among CaO/SiO2 Ratio, Silicate Phase Formation, and Microcracking" Materials 19, no. 3: 463. https://doi.org/10.3390/ma19030463
APA StyleWang, C., Luan, J., Yang, Z., Ma, Q., Wang, G., & Zang, X. (2026). Comparative Analysis of Industrial Fused Magnesia from Natural and Flotation-Processed Magnesite: Associations Among CaO/SiO2 Ratio, Silicate Phase Formation, and Microcracking. Materials, 19(3), 463. https://doi.org/10.3390/ma19030463






