Defect-Selective Luminescence in Hydroxyapatite Under Electron and Gallium Ion Beams
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Crystallinity and Morphology
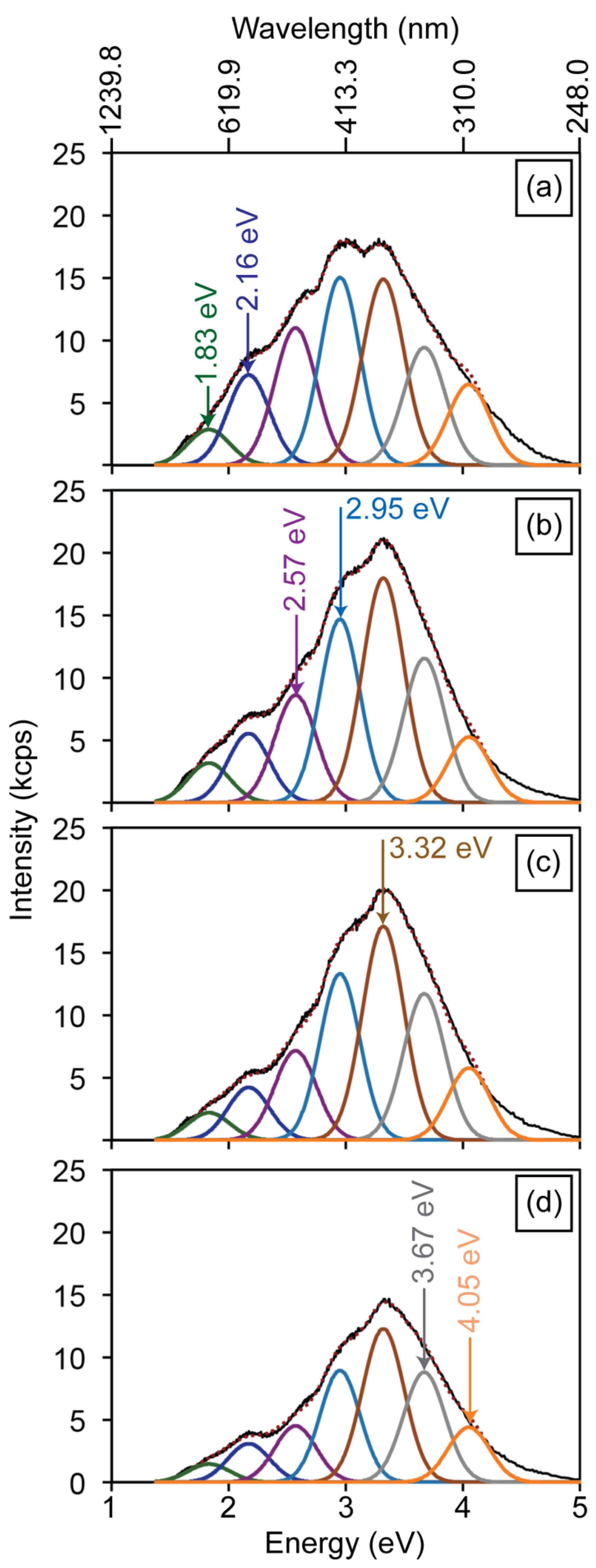
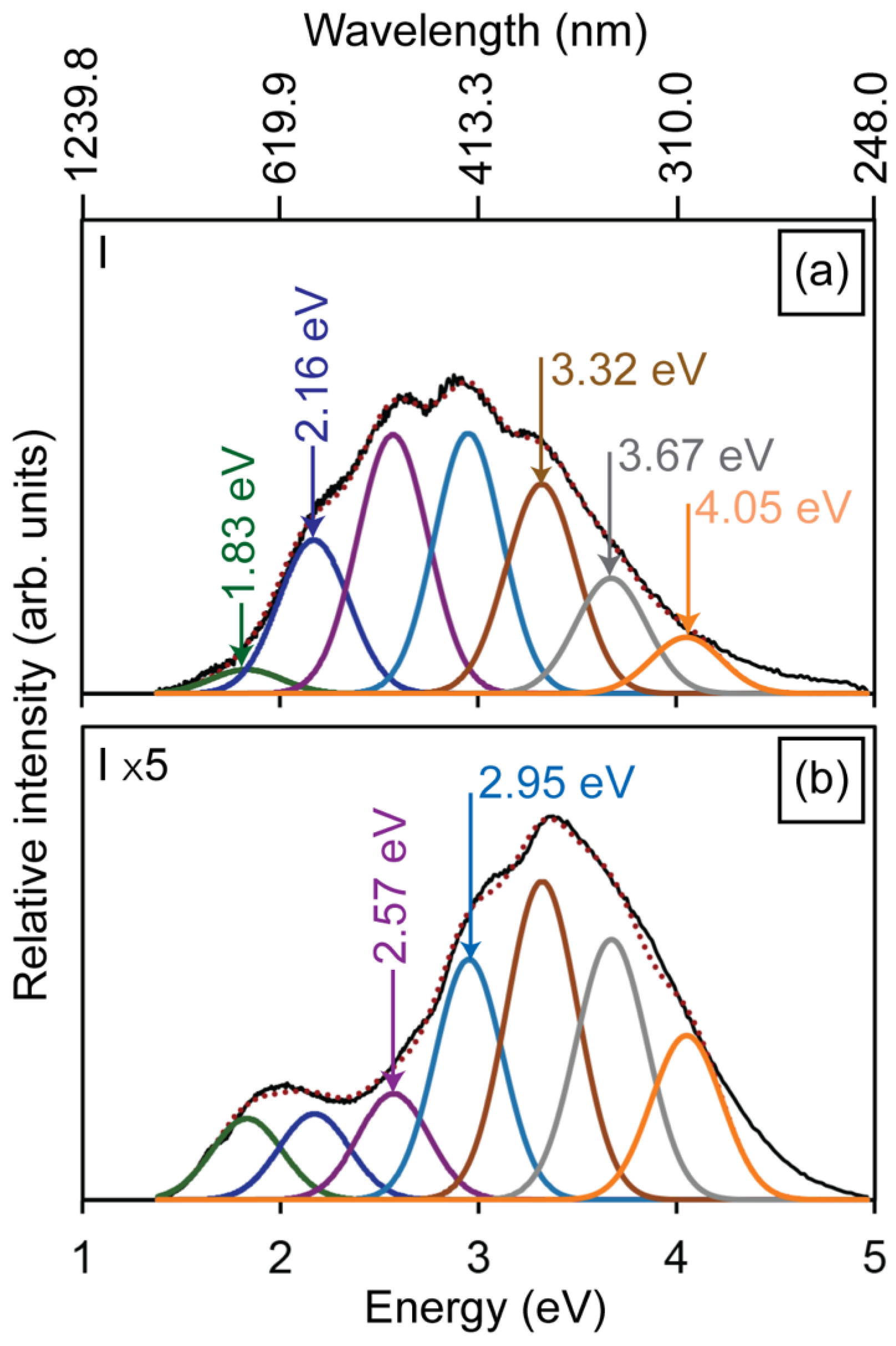
3.2. Defect-Related Luminescence
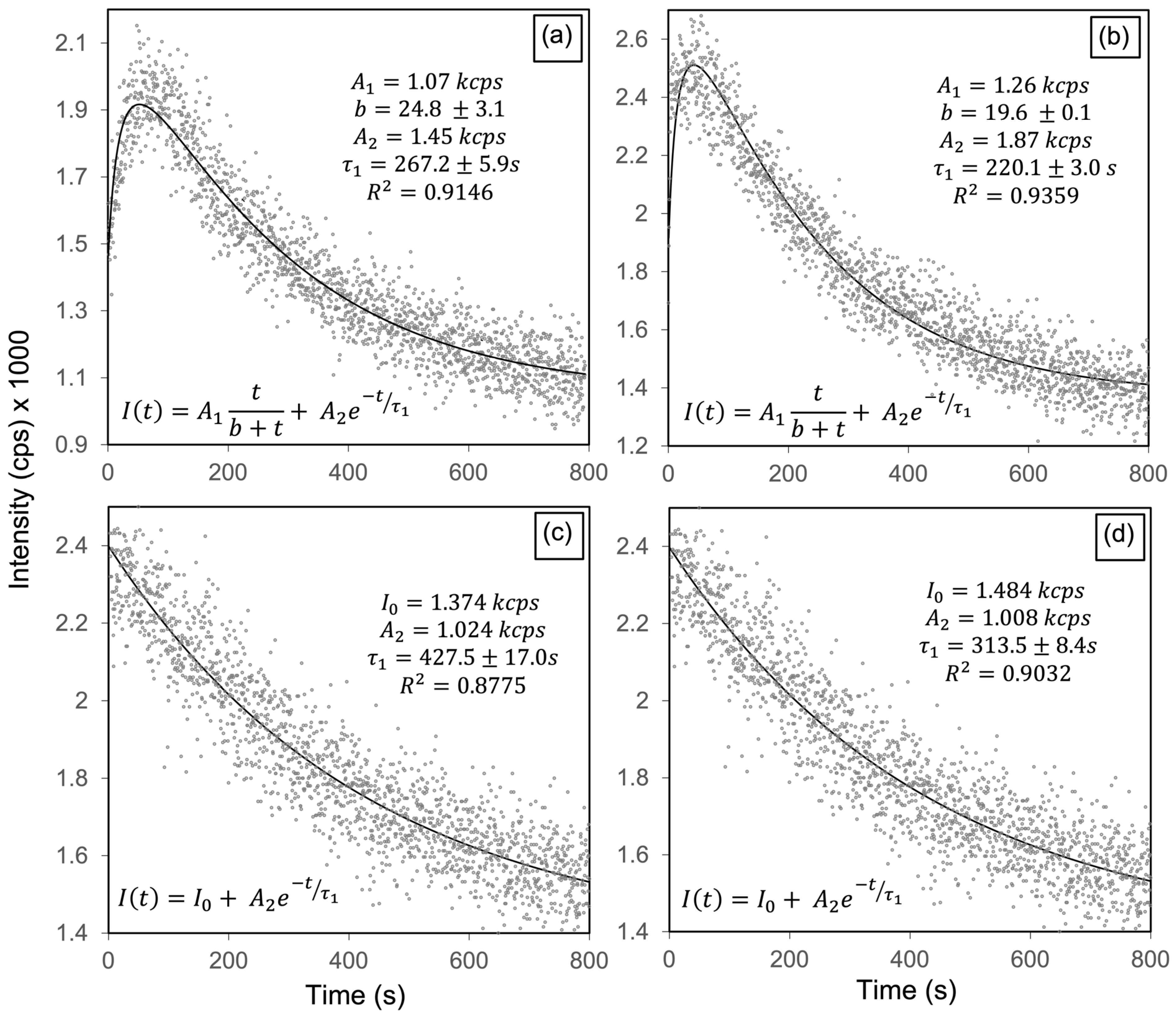
3.3. Low-Energy Electron Beam Irradiation
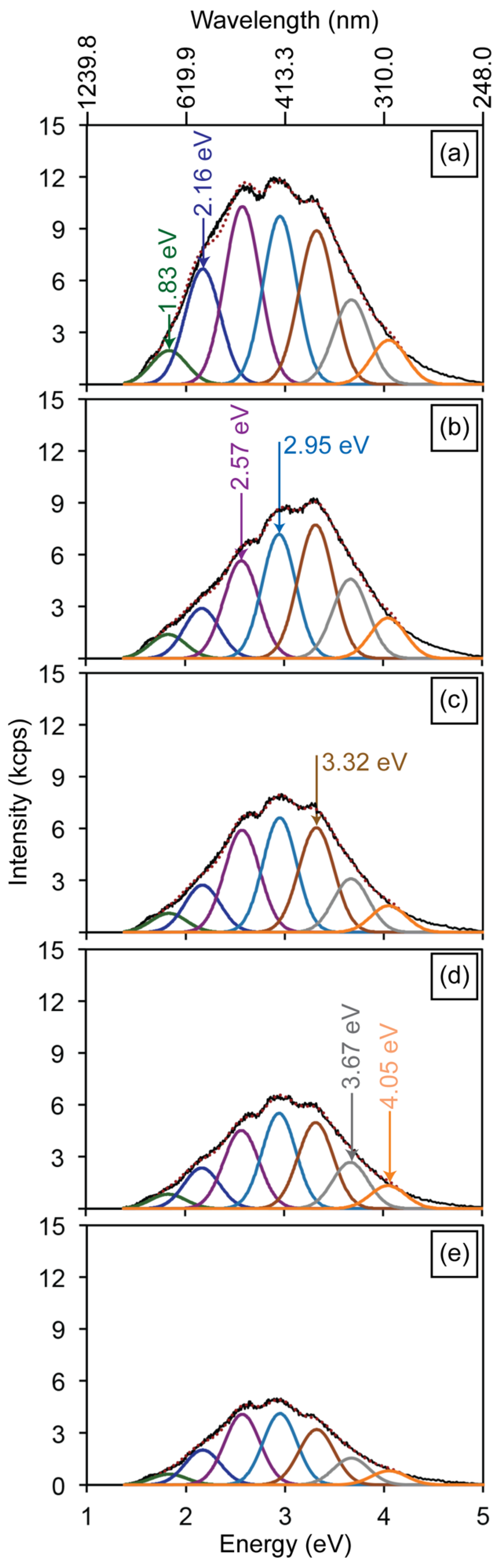
3.4. Gallium Ion Irradiations
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Venkatesan, J.; Anchan, R.V.; Murugan, S.S.; Anil, S.; Kim, S.-K. Natural hydroxyapatite-based nanobiocomposites and their biomaterials-to-cell interaction for bone tissue engineering. Discov. Nano 2024, 19, 169. [Google Scholar] [CrossRef]
- Bordea, I.R.; Candrea, S.; Alexescu, G.T.; Bran, S.; Băciuț, M.; Băciuț, G.; Lucaciu, O.; Dinu, S.M.; Todea, D.A. Nano-hydroxyapatite use in dentistry: A systematic review. Drug Metab. Rev. 2020, 52, 319–332. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Lee, J. Nanoscale hydroxyapatite particles for bone tissue engineering. Acta Biomater. 2011, 7, 2769–2781. [Google Scholar] [CrossRef] [PubMed]
- Haider, A.; Haider, S.; Han, S.S.; Kang, I.-K. Recent advances in the synthesis, functionalization and biomedical applications of hydroxyapatite: A review. RSC Adv. 2017, 7, 7442–7458. [Google Scholar] [CrossRef]
- Martinez-Pallares, F.; Herrera, M.; Graeve, O.A. Decomposition of luminescent hydroxyapatite scaffolds in simulated body fluid. ACS Appl. Bio Mater. 2024, 7, 3136–3142. [Google Scholar] [CrossRef]
- Aina, V.; Bergandi, L.; Lusvardi, G.; Malavasi, G.; Imrie, F.E.; Gibson, I.R.; Cerrato, G.; Ghigo, D. Sr-containing hydroxyapatite: Morphologies of HA crystals and bioactivity on osteoblast cells. Mater. Sci. Eng. C 2013, 33, 1132–1142. [Google Scholar] [CrossRef]
- Huang, P.; Zhou, B.; Zheng, Q.; Tian, Y.; Wang, M.; Wang, L.; Li, J.; Jiang, W. Nano wave plates structuring and index matching in transparent hydroxyapatite-YAG: Ce composite ceramics for high luminous efficiency white light-emitting diodes. Adv. Mater. 2020, 32, 1905951. [Google Scholar] [CrossRef]
- Deshmukh, K.; Shaik, M.M.; Ramanan, S.R.; Kowshik, M. Self-activated fluorescent hydroxyapatite nanoparticles: A promising agent for bioimaging and biolabeling. ACS Biomater. Sci. Eng. 2016, 2, 1257–1264. [Google Scholar] [CrossRef]
- Lara-Ochoa, S.; Ortega-Lara, W.; Guerrero-Beltrán, C.E. Hydroxyapatite nanoparticles in drug delivery: Physicochemistry and applications. Pharmaceutics 2021, 13, 1642. [Google Scholar] [CrossRef]
- Kabilan, N.; Babu, K.D.; Karthikeyan, N.; Chinnakali, K. Optical nonlinear properties of hydroxyapatite based materials. Optik 2022, 265, 169562. [Google Scholar] [CrossRef]
- Huerta, V.J.; Fernández, P.; Gómez, V.; Graeve, O.A.; Herrera, M. Defect-related luminescence properties of hydroxyapatite nanobelts. Appl. Mater. Today 2020, 21, 100822. [Google Scholar] [CrossRef]
- Huerta, V.; Murillo, E.; Chaikina, E.; Graeve, O.A.; Herrera, M. Defect-mediated electrical conduction and piezoelectricity in hydroxyapatite nanofibers. J. Phys. Chem. B 2025, 129, 8428–8435. [Google Scholar] [CrossRef]
- Zilm, M.E.; Chen, L.; Sharma, V.; McDannald, A.; Jain, M.; Ramprasad, R.; Wei, M. Hydroxyapatite substituted by transition metals: Experiment and theory. Phys. Chem. Chem. Phys. 2016, 18, 16457. [Google Scholar] [CrossRef] [PubMed]
- Korneev, A.V.; Sergienko, E.S.; Kuz’mina, M.A.; Pavlychev, A.A.; Brykalova, K.O.; Kalkura, S.N.; Frank-Kamenetskaya, O.V. Crystal-chemical design of hydroxyapatite magnetic nanoparticles with transition metals. Cryst. Growth Des. 2025, 25, 3481–3496. [Google Scholar] [CrossRef]
- Yang, K.; Li, C.; Liu, X.; Du, X.; Zhu, Y.; Zhao, J.; Zhang, Y.; Huang, L. Unveiling promotional effect of non-calcium ionic defects in VOCs catalytic oxidation over modified hydroxyapatite. Mol. Catal. 2025, 586, 115452. [Google Scholar] [CrossRef]
- Novitskaya, E.; Amachraa, M.; Martínez-Pallares, F.; Güell, F.; Gómez-Vidales, V.; Ong, S.P.; Herrera, M.; Graeve, O.A. Barium vacancies as the origin of triboluminescence in hexacelsian ceramics: An ab initio and experimental investigation. ACS Appl. Opt. Mater. 2024, 2, 585–594. [Google Scholar] [CrossRef]
- Wang, M.; Wang, X.-F.; Bao, W.-C.; Chen, Y.; He, T.; Lv, S.-Z.; Jiang, D.-Y.; Wang, X.-G. Crystal defects induced grain refinement and enhanced mechanical properties of hydroxyapatite ceramics. J. Am. Ceram. Soc. 2026, 109, e70412. [Google Scholar] [CrossRef]
- Miao, Y.; Tian, W.; Han, J.; Li, N.; Chen, D.; Xu, Q.; Lu, J. Oxygen vacancy-induced hydroxyl dipole reorientation in hydroxyapatite for enhanced piezocatalytic activity. Nano Energy 2022, 100, 107473. [Google Scholar] [CrossRef]
- Siddiqui, M.A.S.; Mia, M.P.; Hossain, M.A.K.; Rabbi, M.S.; Butt, H.; Alam, F. Recent Advances in Crystalline Architecture and Defect Engineering of 3D-Printed Metals for Biomedical Breakthroughs: A Comprehensive Review. Adv. Eng. Mater. 2025, 27, 2500257. [Google Scholar] [CrossRef]
- Yuan, M.; Kermanian, M.; Agarwal, T.; Yang, Z.; Yousefiasl, S.; Cheng, Z.; Ma, P.; Maleki, A. Defect Engineering in Biomedical Sciences. Adv. Mater. 2023, 35, 2304176. [Google Scholar] [CrossRef]
- Zhu, H.; Guo, D.; Zang, H.; Hanaor, D.; Yu, S.; Schmidt, F.; Xu, K. Enhancement of hydroxyapatite dissolution through structure modification by Krypton ion irradiation. J. Mater. Sci. Technol. 2020, 38, 148–158. [Google Scholar] [CrossRef]
- Raj, A.K.V.; Banerjee, R.H.; Sanwal, J.; Pathak, N.; Chaudhary, N.; Arya, A.; Sengupta, P. Ordering of oxygen vacancies in hydroxyapatite under electron irradiation. Mater. Chem. Phys. 2024, 322, 129609. [Google Scholar] [CrossRef]
- Makhov, V.N.; Lushchik, A.; Lushchik, C.B.; Kirm, M.; Vasil’chenko, E.; Vielhauer, S.; Harutunyan, V.V.; Aleksanyan, E. Luminescence and radiation defects in electron-irradiated Al2O3 and Al2O3:Cr. Nucl. Instrum. Methods Phys. Res. Sect. B 2008, 266, 2949–2952. [Google Scholar] [CrossRef]
- Bystrov, V.S.; Piccirillo, C.; Tobaldi, D.M.; Castro, P.M.L.; Coutinho, J.; Kopyl, S.; Pullar, R.C. Oxygen vacancies, the optical band gap (Eg) and photocatalysis of hydroxyapatite: Comparing modelling with measured data. Appl. Catal. B 2016, 196, 100–107. [Google Scholar] [CrossRef]
- Liu, Y.; Lin, Y.-M.; Yang, S.-F. A thermodynamic interpretation of the Monod equation. Curr. Microbiol. 2003, 46, 0233–0234. [Google Scholar] [CrossRef]
- Novitskaya, E.; Manheim, A.; Herrera, M.; Graeve, O.A. Effect of oxygen vacancies on the mechanoluminescence response of magnesium oxide. J. Phys. Chem. C 2021, 125, 854–864. [Google Scholar] [CrossRef]
- Dierre, B.; Yuan, X.L.; Sekiguchi, T. Luminescence evolution of ZnO single crystal under low-energy electron beam irradiation. J. Appl. Phys. 2008, 104, 043528. [Google Scholar] [CrossRef]
- Elrahoumi, R.; Zhu, L.; Wagner, E.; Maudez, W.; Benvenuti, G.; Phillips, M.R.; Ton-That, C. Doping-induced Ti3+ state and oxygen vacancies in TiO2: A single-chip combinatorial investigation. Mater. Chem. Phys. 2023, 308, 128283. [Google Scholar] [CrossRef]
- Guzmán, G.; Herrera, M. Cathodoluminescence of GaN nanorods and nanowires grown by thermal evaporation. Semicond. Sci. Technol. 2014, 29, 025001. [Google Scholar] [CrossRef]
- Pelaz, L.; Marqúes, L.A.; Barbolla, J. Ion-beam-induced amorphization and recrystallization in silicon. J. Appl. Phys. 2004, 96, 5947–5976. [Google Scholar] [CrossRef]
- Höflich, K.; Hobler, G.; Allen, F.I.; Wirtz, T.; Rius, G.; McElwee-White, L.; Krasheninnikov, A.V.; Schmidt, M.; Utke, I.; Klingner, N.; et al. Roadmap for focused ion beam technologies. Appl. Phys. Rev. 2023, 10, 041311. [Google Scholar] [CrossRef]






| Powder Sample | EDS (at.%) | EDS Ca/P Atomic Ratio | ||
| O | Ca | P | ||
| HAp | 71.04 | 16.23 | 12.74 | 1.27 |
| XPS (at.%) | XPS Ca/P Atomic Ratio | |||
| O 1s | Ca 2p | P 2p | ||
| 56.31 | 24.48 | 19.21 | 1.27 | |
| Emission Energy (eV) | Electronic Transition | τ1 (s) | 1/τ1 (10−3 s−1) |
|---|---|---|---|
| 2.57 | VOH + Hi → VO in PO4 | 267.0 ± 6.0 | 3.75 ± 0.08 |
| 2.95 | VOH → VO in OH | 220.1 ± 3.0 | 4.54 ± 0.06 |
| 3.32 | CB → VO in PO4 | 427.5 ± 17.0 | 2.34 ± 0.09 |
| 3.67 | VOH → VB | 313.5 ± 8.4 | 3.19 ± 0.09 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Huerta, V.J.; Martínez, F.; Ochoa, H.M.; Graeve, O.A.; Herrera-Zaldívar, M. Defect-Selective Luminescence in Hydroxyapatite Under Electron and Gallium Ion Beams. Materials 2026, 19, 321. https://doi.org/10.3390/ma19020321
Huerta VJ, Martínez F, Ochoa HM, Graeve OA, Herrera-Zaldívar M. Defect-Selective Luminescence in Hydroxyapatite Under Electron and Gallium Ion Beams. Materials. 2026; 19(2):321. https://doi.org/10.3390/ma19020321
Chicago/Turabian StyleHuerta, Verónica J., Fabián Martínez, Hanna M. Ochoa, Olivia A. Graeve, and Manuel Herrera-Zaldívar. 2026. "Defect-Selective Luminescence in Hydroxyapatite Under Electron and Gallium Ion Beams" Materials 19, no. 2: 321. https://doi.org/10.3390/ma19020321
APA StyleHuerta, V. J., Martínez, F., Ochoa, H. M., Graeve, O. A., & Herrera-Zaldívar, M. (2026). Defect-Selective Luminescence in Hydroxyapatite Under Electron and Gallium Ion Beams. Materials, 19(2), 321. https://doi.org/10.3390/ma19020321








