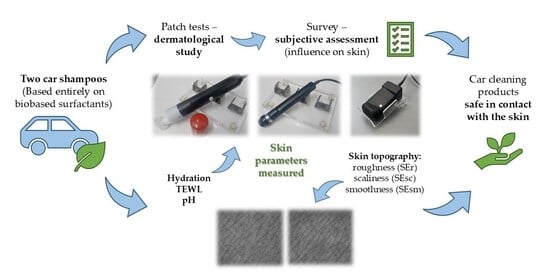
In Vivo Characterisation of Skin Response to Sustainable Car Cleaning Products
Abstract
1. Introduction
2. Materials and Methods
2.1. Characterisation of Car Shampoos
2.2. Dermatological Tests
2.3. Application and Instrumental Tests
2.3.1. Characteristics of the Group
2.3.2. Equipment for Analysing Skin Parameters
2.4. Statistical Analysis
3. Results
3.1. Dermatological Study—The Patch Test
3.2. In Vivo Application Studies
3.2.1. Effect on Skin Hydration
3.2.2. Effect on Transepidermal Water Loss
3.2.3. Effect on Skin pH Value
3.2.4. Changes in Skin Topography
3.2.5. Survey Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| TEWL | Transepidermal Water Loss |
| APGs | Alkyl Polyglycosides |
| FAEs | Fatty Alcohol Ethoxylates |
| NMF | Natural Moisturising Factor |
| SELS | Surface Evaluation of Living Skin |
| SEr | Skin Roughness |
| SEsm | Skin Smoothness |
| SEsc | Skin Scaliness |
References
- Lechuga, M.; Fernandez-Serrano, M.; Nunez-Olea, J.; Martinez-Gallegos, J.F.; Rios, F. Optimization of Toxicity, Biodegradability, and Skin Irritation in Formulations Containing Mixtures of Anionic and Nonionic Surfactants Combined with Silica Nanoparticles. Toxics 2025, 13, 43. [Google Scholar] [CrossRef]
- Schreiner, T.B.; Dias, M.M.; Barreiro, M.F.; Pinho, S.P. Saponins as Natural Emulsifiers for Nanoemulsions. J. Agric. Food Chem. 2022, 70, 6573–6590. [Google Scholar] [CrossRef]
- Nagtode, V.S.; Cardoza, C.; Yasin, H.K.A.; Mali, S.N.; Tambe, S.M.; Roy, P.; Singh, K.; Goel, A.; Amin, P.D.; Thorat, B.R.; et al. Green Surfactants (Biosurfactants): A Petroleum-Free Substitute for Sustainability—Comparison, Applications, Market, and Future Prospects. ACS Omega 2023, 8, 11674–11699. [Google Scholar] [CrossRef]
- Sochacki, M.; Michorczyk, P.; Vogt, O. Foam Fractionation as an Efficient Method for the Separation and Recovery of Surfactants and Surface-Inactive Agents: State of the Art. ACS Omega 2024, 10, 55–75. [Google Scholar] [CrossRef] [PubMed]
- Dutta, A.K. Introductory Chapter: Surfactants in Household and Personal Care Formulations—An Overview. In Surfactants and Detergents; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Li, L.; Su, Q.; Xu, K.; Ren, L.; Zhao, Q.; Wu, Q.; Xia, J.; Lu, X.; Wang, L. Self-Assembly of Cinnamic Acid-Modified Chitosan Surfactants: Synthesis, Characterization, and Applications in Emulsification. ACS Sustain. Chem. Eng. 2024, 12, 7541–7552. [Google Scholar] [CrossRef]
- Woźniak, B.; Wawrzyńczak, A.; Nowak, I. Przegląd produktów do pielęgnacji samochodów. Chemik 2025, 74, 24–27. [Google Scholar] [CrossRef]
- Woźniak, B.; Garstka, A.; Wawrzyńczak, A.; Nowak, I. Nowoczesne surfaktanty—Czym są oraz co je wyróżnia? Przemysł Chem. 2024, 103, 1000–1015. [Google Scholar] [CrossRef]
- Ye, Z.; Wen, Q.; Liu, Y.; Zhou, K.; Shang, Y.; Wang, F.; Lian, C.; Liu, H. Microscopic Origin of Surfactant Irritation: An Experimental and Computational Study. Langmuir 2024, 40, 23102–23110. [Google Scholar] [CrossRef]
- Abdollahi, H.; Ghanbarlou, S.; Babapoor, A.; Kalashgarani, M.Y.; Valadez, I.; Zarrintaj, P.; Davachi, S.M. Surfactant-Containing Detergents: Impacts on Dermal Health. Colloids Surf. B Biointerfaces 2025, 256, 115026. [Google Scholar] [CrossRef]
- Okasaka, M.; Kubota, K.; Yamasaki, E.; Yang, J.; Takata, S. Evaluation of Anionic Surfactants Effects on the Skin Barrier Function Based on Skin Permeability. Pharm. Dev. Technol. 2018, 24, 99–104. [Google Scholar] [CrossRef]
- Abdurrahman, M.; Kamal, M.S.; Ramadhan, R.; Daniati, A.; Arsad, A.; Rahman, A.F.A.; Rita, N. Ecofriendly Natural Surfactants in the Oil and Gas Industry: A Comprehensive Review. ACS Omega 2023, 8, 41004–41021. [Google Scholar] [CrossRef]
- Lechuga, M.; Avila-Sierra, A.; Lobato-Guarnido, I.; Garcia-Lopez, A.I.; Rios, F.; Fernandez-Serrano, M. Mitigating the Skin Irritation Potential of Mixtures of Anionic and Non-Ionic Surfactants by Incorporating Low-Toxicity Silica Nanoparticles. J. Mol. Liq. 2023, 383, 122021. [Google Scholar] [CrossRef]
- Woźniak, B.; Wawrzyńczak, A.; Nowak, I. Analysis of Foaming Properties, Foam Stability, and Basic Physicochemical and Application Parameters of Bio-Based Car Shampoos. Coatings 2025, 15, 907. [Google Scholar] [CrossRef]
- Thompson, C.J.; Ainger, N.; Starck, P.; Mykhaylyk, O.O.; Ryan, A.J. Shampoo Science: A Review of the Physiochemical Processes behind the Function of a Shampoo. Macromol. Chem. Phys. 2022, 224, 2200420. [Google Scholar] [CrossRef]
- Chen, C.; Xi, H.; Li, Z.; Zhang, H.; Lin, X.; Wang, Y. Removal of uranium by APG/TAS antifreeze foam detergent with high foaming property. Colloids Surf. A 2022, 650, 129589. [Google Scholar] [CrossRef]
- Wasilewski, T.; Hordyjewicz-Baran, Z.; Sabura, E.; Malorna, K.; Dresler, E.; Zegarski, M.; Stanek-Wandzel, N. Application of Alkyl Polyglucosides as Components of the Extraction Medium in the Preparation of a Shampoo Cosmetic Formulation Containing Substances Isolated from Red Grape Pomace. Molecules 2025, 30, 3817. [Google Scholar] [CrossRef]
- Markovic, D.B.; Tasic-Kostov, M.; Lukic, M.; Isailovic, T.; Krstonosic, V.; Daniels, R.; Savic, S. Physicochemical Characterization and In Vivo Skin Performance of a Novel Alkyl Polyglucoside Emulsifier in Natural Cosmetic Cream-Bases. Tenside Surfactants Deterg. 2014, 51, 133–145. [Google Scholar] [CrossRef]
- Cirin, D.; Milutinov, J.; Krstonosic, V. Occurrence of Alkyl Glucosides in Rinse-Off Cosmetics Marketed as Hypoallergenic or for Sensitive Skin. Toxicol. Ind. Health 2024, 40, 306–311. [Google Scholar] [CrossRef] [PubMed]
- Bubić Pajić, N.Z.; Todosijević, M.N.; Vuleta, G.M.; Cekić, N.D.; Dobričić, V.D.; Vucen, S.R.; Calija, B.R.; Lukić, M.Z.; Ilić, T.M.; Savić, S.D. Alkyl Polyglucoside vs. Ethoxylated Surfactant-Based Microemulsions as Vehicles for Two Poorly Water-Soluble Drugs: Physicochemical Characterization and In Vivo Skin Performance. Acta Pharm. 2021, 67, 415–439. [Google Scholar] [CrossRef] [PubMed]
- Fouda, A.S.; El-Maksoud, S.A.A.; El-Habab, A.T.; Ibrahim, A.R. Synthesis and Characterization of Novel Fatty Alcohol Ethoxylate Surfactants for Corrosion Inhibition of Mild Steel. J. Bio- Tribo-Corros. 2021, 7, 18. [Google Scholar] [CrossRef]
- Rincon-Romero, J.F.; Rios, F.; Reyes-Requena, A.; Luzon-Gonzalez, G.; Garcia-Lopez, A.I. Surface and Thermodynamics Properties of Commercial Fatty-Alcohol Ethoxylate Surfactants. J. Mol. Liq. 2023, 376, 121396. [Google Scholar] [CrossRef]
- Hartwig, A.; MAK Commission. Fatty Alcohol Ethoxylates, C16–18 and C18. MAK Collect. Occup. Health Saf. 2024, 9, Doc061. [Google Scholar] [CrossRef]
- Hu, X.; Carter, J.; Ge, T.; Liao, M.; Stephens, A.M.; McInnes, E.F.; Padia, F.; Lu, J.R. Impacts of Chain and Head Lengths of Nonionic Alkyl Ethoxylate Surfactants on Cytotoxicity to Human Corneal and Skin Cells in Agri-Spraying Processes. J. Colloid Interface Sci. 2022, 628, 162–173. [Google Scholar] [CrossRef]
- Choi, E.H.; Kang, H. Importance of Stratum Corneum Acidification to Restore Skin Barrier Function in Eczematous Diseases. Ann. Dermatol. 2024, 36, 1–8. [Google Scholar] [CrossRef]
- Rinaldi, A.O.; Li, M.; Barletta, E.; D’Avino, P.; Yazici, D.; Pat, Y.; Ward, S.; Burla, D.; Tan, G.; Askary, N.; et al. Household Laundry Detergents Disrupt Barrier Integrity and Induce Inflammation in Mouse and Human Skin. Allergy 2023, 79, 128–141. [Google Scholar] [CrossRef]
- Vater, C.; Apanovic, A.; Riethmuller, C.; Litschauer, B.; Wolzt, M.; Valenta, C.; Klang, V. Changes in Skin Barrier Function after Repeated Exposition to Phospholipid-Based Surfactants and Sodium Dodecyl Sulfate In Vivo and Corneocyte Surface Analysis by Atomic Force Microscopy. Pharmaceutics 2021, 13, 436. [Google Scholar] [CrossRef] [PubMed]
- OECD. Test No. 301: Ready Biodegradability; OECD Guidelines for the Testing of Chemicals, Section 3; OECD Publishing: Paris, France, 1992; Available online: https://www.oecd.org/en/publications/test-no-301-ready-biodegradability_9789264070349-en.html (accessed on 1 January 2026).
- Courage + Khazaka. Scientific Products (Online). Available online: https://www.courage-khazaka.com/en/scientific-products/all-products (accessed on 5 November 2025).
- Courage + Khazaka. Corneometer CM 825; Manual Instruction; Courage + Khazaka: Köln, Germany, 2014. [Google Scholar]
- Berardesca, E.; EEMCO Group. EEMCO Guidance for the Assessment of Stratum Corneum Hydration. Ski. Res. Technol. 1997, 3, 126–132. [Google Scholar] [CrossRef]
- Huygen, L.; Thys, P.M.; Wollenberg, A.; Gutermuth, J.; Krohn, I.K. Skin Barrier Function Assessment: Electrical Impedance Spectroscopy Is Less Influenced by Daily Routine Activities Than Transepidermal Water Loss. Ann. Dermatol. 2024, 36, 99–111. [Google Scholar] [CrossRef]
- Courage + Khazaka. Tewameter TM 300; Manual Instruction; Courage + Khazaka: Köln, Germany, 2014. [Google Scholar]
- Berardesca, E.; Loden, M.; Serup, J.; Masson, P.; Rodrigues, L.M. Revised EEMCO Guidance for Water in Skin Measurement. Ski. Res. Technol. 2018, 24, 351–358. [Google Scholar] [CrossRef]
- Rogiers, V.; EEMCO Group. EEMCO Guidance for the Assessment of TEWL. Ski. Pharmacol. Appl. Ski. Physiol. 2001, 14, 117–128. [Google Scholar] [CrossRef] [PubMed]
- Courage + Khazaka. Visioscan VC 98; Manual Instruction; Courage + Khazaka: Köln, Germany, 2016. [Google Scholar]
- Lévêque, J.L. EEMCO Guidance for Skin Topography Measurement. J. Eur. Acad. Dermatol. Venereol. 1999, 12, 103–114. [Google Scholar] [CrossRef] [PubMed]
- Courage + Khazaka. Skin-pH-Meter PH 905; Manual Instructions; Courage + Khazaka: Köln, Germany, 2020. [Google Scholar]
- Parra, J.L.; Paye, M.; EEMCO Group. EEMCO Guidance for the Assessment of Skin Surface pH. Ski. Pharmacol. Appl. Ski. Physiol. 2003, 16, 188–202. [Google Scholar] [CrossRef]
- Lemery, E.; Briançon, S.; Chevalier, Y.; Oddos, T.; Gohier, A.; Boyron, O.; Bolzinger, M.A. Surfactants Have Multifold Effects on Skin Barrier Function. Eur. J. Dermatol. 2015, 25, 424–435. [Google Scholar] [CrossRef]
- Lukić, M.; Pantelić, I.; Savić, S.D. Towards Optimal pH of the Skin and Topical Formulations: From the Current State of the Art to Tailored Products. Cosmetics 2021, 8, 69. [Google Scholar] [CrossRef]
- Barel, A.O.; Lambrecht, R.; Clarys, P.; Morrison, B.M.; Paye, M. Comparative Study on the Skin Effects of Bar Soap and Syndet Cleansing Bars. Ski. Res. Technol. 2001, 7, 98–104. [Google Scholar] [CrossRef]
- Hawkins, S.; Dasgupta, B.R.; Ananthapadmanabhan, K.P. Role of pH in Skin Cleansing. Int. J. Cosmet. Sci. 2021, 43, 474–483. [Google Scholar] [CrossRef]
- Lee, E.; An, S.; Im, M.S.; Kim, H.K.; Lee, T.R. An Improved Method for the Measurement of Skin Roughness Changes Caused by Cleansing Products. Ski. Res. Technol. 2011, 17, 320–325. [Google Scholar] [CrossRef] [PubMed]
- Harding, C.R.; Watkinson, A.; Rawlings, A.V.; Scott, I.R. Dry Skin, Moisturization and Corneodesmolysis. Int. J. Cosmet. Sci. 2000, 22, 21–52. [Google Scholar] [CrossRef]
- Blaak, J.; Grabmann, S.; Simon, I.; Callaghan, T.; Staib, P. Five Dimensions of Cleansing: A Holistic View on the Facets and Importance of Skin Cleansing. Int. J. Cosmet. Sci. 2023, 45, 557–571. [Google Scholar] [CrossRef]
- Liu, M.; Mollica, L.; Regan, J.; Hawkins, S.; McGuiness, H.; Vetro, K.; Garczarek, U.; Shi, S.; Vasudevan, V.; Ananthapadmanabhan, K.P. Modified Corneosurfametry as a High-Throughput Method. Int. J. Cosmet. Sci. 2016, 38, 178–186. [Google Scholar] [CrossRef]
- Ananthapadmanabhan, K.P.; Moore, D.J.; Subramanyan, K.; Misra, M.; Meyer, F. Cleansing without Compromise: The Impact of Cleansers on the Skin Barrier and the Technology of Mild Cleansing. Dermatol. Ther. 2004, 17, 16–25. [Google Scholar] [CrossRef]
- Bhoyrul, B.; Solman, L.; Kirk, S.; Orton, D.; Wilkinson, M. Patch Testing with Alkyl Glucosides. Contact Dermat. 2018, 80, 286–290. [Google Scholar] [CrossRef] [PubMed]
- Leoty-Okombi, S.; Gillaizeau, F.; Leuillet, S.; Douillard, B.; Le Fresne-Languille, S.; Carton, T.; De Martino, A.; Moussou, P.; Bonnaud-Rosaye, C.; André, V. Effect of Sodium Lauryl Sulfate (SLS) Applied as a Patch on Human Skin Physiology and Its Microbiota. Cosmetics 2021, 8, 6. [Google Scholar] [CrossRef]
- Siqueira, R.A.G.; Hradkova, I.; Leite-Silva, V.R.; Andreo-Filho, N.; Lopes, P.S. Skin Lipids and Their Influence on Skin Microbiome and Skin Care. ACS Omega 2025, 10, 28534–28546. [Google Scholar] [CrossRef] [PubMed]







Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Woźniak, B.; Marzec, M.; Wawrzyńczak, A.; Nowak, I. In Vivo Characterisation of Skin Response to Sustainable Car Cleaning Products. Materials 2026, 19, 269. https://doi.org/10.3390/ma19020269
Woźniak B, Marzec M, Wawrzyńczak A, Nowak I. In Vivo Characterisation of Skin Response to Sustainable Car Cleaning Products. Materials. 2026; 19(2):269. https://doi.org/10.3390/ma19020269
Chicago/Turabian StyleWoźniak, Bartosz, Marta Marzec, Agata Wawrzyńczak, and Izabela Nowak. 2026. "In Vivo Characterisation of Skin Response to Sustainable Car Cleaning Products" Materials 19, no. 2: 269. https://doi.org/10.3390/ma19020269
APA StyleWoźniak, B., Marzec, M., Wawrzyńczak, A., & Nowak, I. (2026). In Vivo Characterisation of Skin Response to Sustainable Car Cleaning Products. Materials, 19(2), 269. https://doi.org/10.3390/ma19020269