Controlled Synthesis and Formation Mechanism of Uniformly Sized Spherical CeO2 Nanoparticles
Abstract
1. Introduction
2. Experimental Procedure
2.1. Materials
2.2. Synthesis of CeO2 NPs
2.3. Characterization
2.4. Data Analysis and Statistical Methods
3. Results and Discussion
3.1. Thermal Analysis
- Dehydration Stage:Ce(SO4)2•4H2O → Ce(SO4)2 + 4H2O ↑
- Ion Exchange Stage:Ce(SO4)2 + 4NO3− → Ce(NO3)4 + 2SO42−
- Thermal Decomposition Stage:Ce(NO3)4 → CeO2 + 4NO ↑ + 3O2 ↑Ce(NO3)4 → CeO2 + 4NO2 ↑ + O2 ↑
3.2. Effect of Reaction Conditions on CeO2 NPs
3.2.1. Influence of Cerium Salt and Surfactant Type
3.2.2. Influence of Calcination Process
- I.
- Effects of Heating Rate
- -
- At a heating rate of 2 °C/min, a part of extremely fine particles existed in the product.
- -
- At a rate of 10 °C/min, a small number of large particles with dimensions greater than 100 nm were found.
- -
- The optimal heating rate was 6 °C/min. Under this rate, the proportion of undersized particles decreased, resulting in a uniform particle size with a narrow distribution.
- II.
- Effects of Soaking Time
- -
- However, when the holding time was extended to 30 min, a large number of fine particles in the range of 10–20 nm were observed in the product.
- -
- When the holding time was extended to 6 h, abnormal growth of some particles occurred, resulting in some oversized particles with diameters greater than 100 nm.
- III.
- Optimal Calcination Parameters
3.2.3. Influence of F127 Dosage
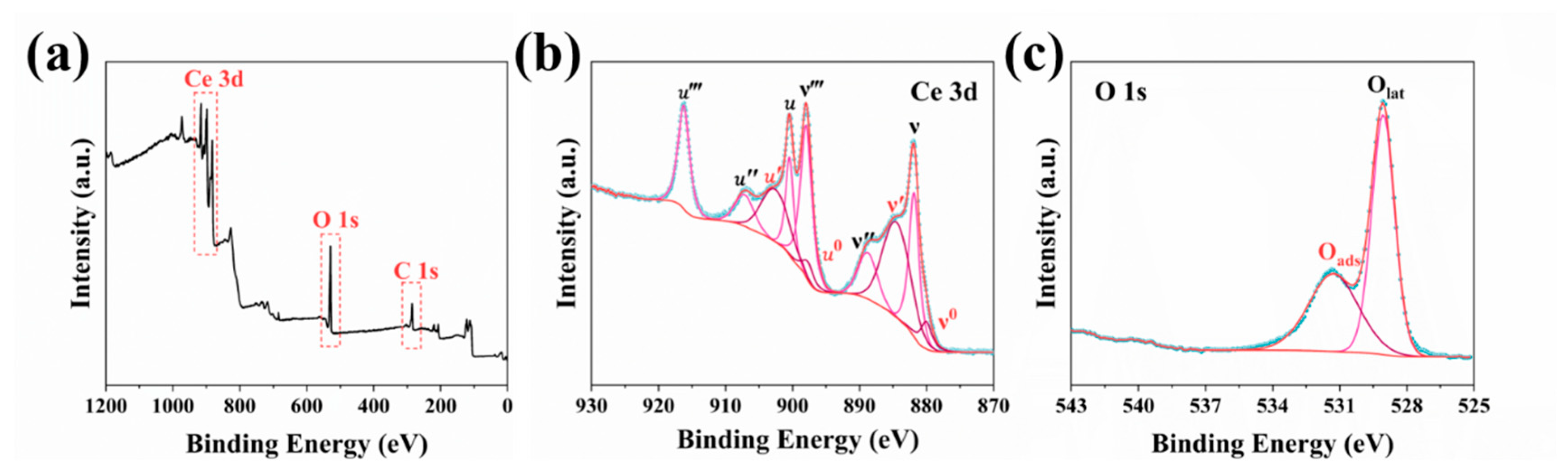
3.3. Characterization of Spherical CeO2 NPs Synthesized Under Optimal Conditions
3.4. Formation Mechanism of Spherical CeO2 NPs
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhao, X.; Zhao, J.; Wang, F.; Tan, B.; Shi, Y.; Han, X.; Qi, W.; Geng, J. Effect of hydroxyl and aldehyde groups on the polishing of silicon dioxide and silicon nitride by cerium oxide dispersions. Mater. Sci. Semicond. Process 2025, 199, 109872. [Google Scholar] [CrossRef]
- An, S.; Chen, P.; Hou, F.; Wang, Q.; Pan, H.; Chen, X.; Lu, X.; Zhao, Y.; Huang, Q.; Zhang, X. Cerium-doped indium oxide transparent electrode for semi-transparent perovskite and perovskite/silicon tandem solar cells. Sol. Energy 2020, 196, 409–418. [Google Scholar] [CrossRef]
- Zhang, L.; Che, Z.; Shang, J.; Wang, Q.; Cao, M.; Zhou, Y.; Zhou, Y.; Liu, F. Cerium-doped indium oxide as a top electrode of semitransparent perovskite solar cells. ACS Appl. Mater. Interfaces 2023, 15, 10838–10846. [Google Scholar] [CrossRef]
- Pesaran, A.; Jaiswal, A.; Ren, Y.; Wachsman, E.D. Development of a new ceria/yttria-ceria double-doped bismuth oxide bilayer electrolyte low-temperature SOFC with higher stability. Ionics 2019, 25, 3153–3164. [Google Scholar] [CrossRef]
- Charbgoo, F.; Ramezani, M.; Darroudi, M. Bio-sensing applications of cerium oxide nanoparticles: Advantages and disadvantages. Biosens. Bioelectron. 2017, 96, 33–43. [Google Scholar] [CrossRef]
- Hoang, V.A.; Yoshizuka, K.; Nishihama, S. Oxidative adsorption of arsenic from water environment by activated carbon modified with cerium oxide/hydroxide. Chem. Eng. Res. Des. 2022, 186, 161–173. [Google Scholar] [CrossRef]
- Devaiah, D.; Reddy, L.H.; Park, S.; Reddy, B.M. Ceria-zirconia mixed oxides: Synthetic methods and applications. Catal. Rev.-Sci. Eng. 2018, 60, 177–277. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, N.; Wang, X.; Zhao, Z.; Dong, C.; Tan, X.; Zheng, Y.; Feng, Z.; Zhong, X.; Yang, J.; et al. Enhanced chemical mechanical polishing (CMP) performance of porous self-assembled spherical cerium oxide via RE(la/pr/nd) doping. Appl. Surf. Sci. 2025, 679, 161236. [Google Scholar] [CrossRef]
- Zhao, G.; Wang, C.X.; Chen, J.X.; Huang, Y.H.; Huang, R.C. Advances in chemical mechanical polishing endpoint detection technology for integrated circuits manufacturing. China Surf. Eng. 2025, 38, 83–98. [Google Scholar]
- Ma, J.; Xu, N.; Cheng, J.; Pu, Y. A review on the development of ceria for chemical mechanical polishing. Powder Technol. 2024, 444, 119989. [Google Scholar] [CrossRef]
- Xu, N.; Ma, J.; Liu, Q.; Luo, Y.; Pu, Y. Preparation of CeO2 abrasives by reducing atmosphere-assisted molten salt method for enhancing their chemical mechanical polishing performance on SiO2 substrates. J. Rare Earths 2023, 41, 1627–1635. [Google Scholar] [CrossRef]
- Kim, E.; Hong, J.; Hong, S.; Kanade, C.; Seok, H.; Kim, H.; Kim, T. Improvement of oxide removal rate in chemical mechanical polishing by forming oxygen vacancy in ceria abrasives via ultraviolet irradiation. Mater. Chem. Phys. 2021, 273, 124967. [Google Scholar] [CrossRef]
- Lee, J.; Kim, E.; Bae, C.; Seok, H.; Cho, J.; Aydin, K.; Kim, T. Improvement of oxide chemical mechanical polishing performance by increasing ce3+/ce4+ ratio in ceria slurry via hydrogen reduction. Mater. Sci. Semicond. Process. 2023, 159, 107349. [Google Scholar] [CrossRef]
- Wang, Q.; Jia, W.; Liu, B.; Zhao, W.; Li, C.; Zhang, J.; Xu, G. Controllable synthesis of nearly monodisperse spherical aggregates of CeO2 nanocrystals and their catalytic activity for HCHO oxidation. Chem.-Asian J. 2012, 7, 2258–2267. [Google Scholar] [CrossRef]
- Taniguchi, A.; Kumabe, Y.; Kan, K.; Ohtani, M.; Kobiro, K. Ce3+-enriched spherical porous ceria with an enhanced oxygen storage capacity. RSC Adv. 2021, 11, 5609–5617. [Google Scholar] [CrossRef]
- Suresh, R.; Ponnuswamy, V.; Mariappan, R. Effect of annealing temperature on the microstructural, optical and electrical properties of CeO2 nanoparticles by chemical precipitation method. Appl. Surf. Sci. 2013, 273, 457–464. [Google Scholar] [CrossRef]
- Bao, Z.; Li, K.; Wang, S.; Gao, K.; Zhang, D.; Li, M. Preparation and characterization of submicron-cerium oxide by hypergravity coprecipitation method. Adv. Powder Technol. 2021, 32, 1611–1618. [Google Scholar] [CrossRef]
- Ren, G.; Wang, L.; Wang, S. Innovative synthesis of CeO2 nanoparticles for advanced chemical mechanical polishing. Colloid Surf. A-Physicochem. Eng. Asp. 2025, 705, 135764. [Google Scholar] [CrossRef]
- He, Y.; Yang, B.; Cheng, G. Controlled synthesis of CeO2 nanoparticles from the coupling route of homogenous precipitation with microemulsion. Mater. Lett. 2003, 57, 1880–1884. [Google Scholar] [CrossRef]
- Sun, Q.; Zhu, S.; Shen, Z.; Liu, Y.; Wu, C.; Kang, L.; Yang, Y. Molten-salt assisted synthesis of two-dimensional materials and energy storage application. Mater. Today Chem. 2023, 29, 101419. [Google Scholar] [CrossRef]
- Lan, Y.; Sohn, H.Y.; Mohassab, Y.; Liu, Q.; Xu, B. Nanoceria synthesis in the KCl-LiCl salt system: Crystal formation and properties. J. Am. Ceram. Soc. 2017, 100, 1863–1875. [Google Scholar] [CrossRef]
- Lan, Y.; Sohn, H.Y. Nanoceria synthesis in molten KOH-NaOH mixture: Characterization and oxygen vacancy formation. Ceram. Int. 2018, 44, 3847–3855. [Google Scholar] [CrossRef]
- Mao, X.; Xia, X.; Ning, D.; Li, J.; Chen, C.; Lan, Y. Characterizations and photocatalytic activity of ceria nanoparticles synthesized in KCl-LiCl/KOH-NaOH molten flux from different precursors. J. Nanopart. Res. 2021, 23, 12. [Google Scholar] [CrossRef]
- Ma, J.; Xu, N.; Luo, Y.; Liu, Q.; Pu, Y. Defect generation and morphology transformation mechanism of CeO2 particles prepared by molten salt method. Ceram. Int. 2023, 49, 4929–4943. [Google Scholar] [CrossRef]
- Liu, X.; Fechler, N.; Antonietti, M. Salt melt synthesis of ceramics, semiconductors and carbon nanostructures. Chem. Soc. Rev. 2013, 42, 8237–8265. [Google Scholar] [CrossRef]
- Kong, W.; Shen, B.; Ma, J.; Kong, J.; Feng, S.; Wang, Z.; Xiong, L. Pyrolysis of spirulina platensis, tetradesmus obliquus and chlorella vulgaris by TG-FTIR and py-GC/MS: Kinetic analysis and pyrolysis behaviour. Energy 2022, 244, 123165. [Google Scholar] [CrossRef]
- Ming, X.; Xu, F.; Jiang, Y.; Zong, P.; Wang, B.; Li, J.; Qiao, Y.; Tian, Y. Thermal degradation of food waste by TG-FTIR and py-GC/MS: Pyrolysis behaviors, products, kinetic and thermodynamic analysis. J. Clean. Prod. 2020, 244, 118713. [Google Scholar] [CrossRef]
- Madarasz, J.; Varga, P.P.; Pokol, G. Evolved gas analyses (TG/DTA-MS and TG-FTIR) on dehydration and pyrolysis of magnesium nitrate hexahydrate in air and nitrogen. J. Anal. Appl. Pyrolysis 2007, 79, 475–478. [Google Scholar] [CrossRef]
- Patel, D.; Gawali, S.L.; Kuperkar, K.; Hassan, P.A.; Bahadur, P. Co-micellization conduct and structural dynamics of block copolymers in water and salt solution environment for drug solubilization enhancement. Colloid Polym. Sci. 2023, 301, 919–931. [Google Scholar] [CrossRef]
- Gupta, S.K.; Mao, Y. A review on molten salt synthesis of metal oxide nanomaterials: Status, opportunity, and challenge. Prog. Mater. Sci. 2021, 117, 100734. [Google Scholar] [CrossRef]
- Kravtsov, A.A.; Blinov, A.V.; Nagdalian, A.A.; Gvozdenko, A.A.; Golik, A.B.; Pirogov, M.A.; Kolodkin, M.A.; Alharbi, N.S.; Kadaikunnan, S.; Thiruvengadam, M.; et al. Acid-base and photocatalytic properties of the CeO2-ag nanocomposites. Micromachines 2023, 14, 12. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, W.; Zhang, B.; Li, H.; Zhang, Q. Controllable synthesis of spherical cerium oxide particles. RSC Adv. 2016, 6, 30956–30962. [Google Scholar] [CrossRef]
- Aminzare, M.; Amoozegar, Z.; Sadrnezhaad, S.K. An investigation on the influence of milling time and calcination temperature on the characterization of nano cerium oxide powder synthesized by mechanochemical route. Mater. Res. Bull. 2012, 47, 3586–3591. [Google Scholar] [CrossRef]
- Zhou, C.; Zhu, D.; Wang, D. Synthesis and characterization of cerium dioxide nanoparticles obtained by a novel soft mechanochemical method combined with sol-gel method. Nano 2017, 12, 1750020. [Google Scholar] [CrossRef]
- Ravindra, A.V.; Ju, S. Mesoporous CoFe2o4 nanocrystals: Rapid microwave-hydrothermal synthesis and effect of synthesis temperature on properties. Mater. Chem. Phys. 2023, 303, 127818. [Google Scholar] [CrossRef]
- He, L.; Li, J.; Feng, Z.; Sun, D.; Wang, T.; Li, R.; Xu, Y. Solvothermal synthesis and characterization of ceria with solid and hollow spherical and multilayered morphologies. Appl. Surf. Sci. 2014, 322, 147–154. [Google Scholar] [CrossRef]
- Penn, R.L.; Banfield, J.F. Imperfect oriented attachment: Dislocation generation in defect-free nanocrystals. Science 1998, 281, 969–971. [Google Scholar] [CrossRef]
- Chen, J.; Li, K.; Yang, J.; Gu, J. Bimetallic ordered large-pore MesoMOFs for simultaneous enrichment and dephosphorylation of phosphopeptides. ACS Appl. Mater. Interfaces 2021, 13, 60173–60181. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.; Umar, A.; Yunarti, R.T.; Bakri, R.; Putra, B.R.; Wahyuni, W.T.; Arifutzzaman, A.; Aroua, M.K.; Khalil, M. Mesoporous metal oxide via nanocasting: Recent advances on types of templates, properties, and catalytic activities. Mater. Today Commun. 2024, 40, 19. [Google Scholar] [CrossRef]
- Zou, Y.; Zhou, X.; Zhu, Y.; Cheng, X.; Zhao, D.; Deng, Y. Sp2-hybridized carbon-containing block copolymer templated synthesis of mesoporous semiconducting metal oxides with excellent gas sensing property. Acc. Chem. Res. 2019, 52, 714–725. [Google Scholar] [CrossRef]
- Zhang, D.; Fu, H.; Shi, L.; Pan, C.; Li, Q.; Chu, Y.; Yu, W. Synthesis of CeO2 nanorods via ultrasonication assisted by polyethylene glycol. Inorg. Chem. 2007, 46, 2446–2451. [Google Scholar] [CrossRef]
- Wang, Y.; Sukhishvili, S.A. Hydrogen-bonded polymer complexes and nanocages of weak polyacids templated by a pluronic® block copolymer. Soft Matter 2016, 12, 8744–8754. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.Y.; Hao, W.; Liu, H.Z.; Xu, Z.H. Temperature effects on block copolymer micelle based on the scaling model. Acta Phys.-Chim. Sin. 2004, 20, 38–42. [Google Scholar]
- Zandkarimi, B.; Poths, P.; Alexandrova, A.N. When fluxionality beats size selection: Acceleration of ostwald ripening of sub-nano clusters. Angew. Chem. Int. Ed. 2021, 60, 11973–11982. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Zhang, P.; Wu, Y.; Li, X.; Wang, X.; Wang, M.; Jin, S.; Bi, W.; Zhao, Y.; Zhou, W.; et al. Preparation of CeO2 polishing powder and its performance and mechanism for chemical mechanical polishing of optical glass. ACS Appl. Mater. Interfaces 2025, 17, 20394–20410. [Google Scholar] [CrossRef]
- Mao, X.; Xia, X.; Li, J.; Chen, C.; Zhang, J.; Ning, D.; Lan, Y. Homogenously rare-earth-ion-doped nanoceria synthesis in KOH-NaOH molten flux: Characterization and photocatalytic property. J. Mater. Eng. Perform. 2021, 30, 3795–3805. [Google Scholar] [CrossRef]
- Chen, Y.; Li, P.; Wang, C.; Zhong, L.; Chen, A. Synthesis, characterization and CMP property of neat and doped mesoporous ceria as defective active particle abrasives. Surf. Interfaces 2025, 56, 105614. [Google Scholar] [CrossRef]
- Zhang, L.; Qi, Y.; Lai, J.; Hu, X.; Yu, B.; Li, Z.; Wang, S.; Luo, C. Electronic metal–support interaction–driven photocatalytic chemical mechanical polishing of silicon carbide wafers using ag-modified CeO2 slurry. Chem. Eng. J. 2025, 526, 171426. [Google Scholar] [CrossRef]
- Zhang, Z.; Guan, D.; Wang, N.; Tan, X.; Wang, X.; Feng, Z.; Zheng, Y.; Zhong, X.; Wang, X.; Yang, J.; et al. Controlling crystallization pathway for synthesizing high-polishing performance spherical cerium oxide. J. Rare Earths 2025, 43, 1046–1056. [Google Scholar] [CrossRef]
- Xiao, M.; Luo, B.; Konarova, M.; Wang, Z.; Wang, L. Molten salt synthesis of atomic heterogeneous catalysts: Old chemistry for advanced materials. Eur. J. Inorg. Chem. 2020, 2020, 2942–2949. [Google Scholar] [CrossRef]
- Ai, Y.; Yang, C.C.; Gai, T.Y.; Feng, J.Y.; Li, W. Mono-micelle directed assembly for the synthesis of mesoporous materials. Chin. Sci. Bull.-Chin. 2024, 69, 2110–2129. [Google Scholar] [CrossRef]
- Yang, P.D.; Zhao, D.Y.; Margolese, D.I.; Chmelka, B.F.; Stucky, G.D. Generalized syntheses of large-pore mesoporous metal oxides with semicrystalline frameworks. Nature 1998, 396, 152–155. [Google Scholar] [CrossRef]










| Temperature/°C | FWHM(111)/deg | d(111)/Å | A/Å | D/nm |
|---|---|---|---|---|
| 500 | 0.62 | 3.134 | 5.428 | 13.219 |
| 450 | 0.94 | 3.121 | 5.406 | 8.721 |
| 400 | 1.54 | 3.132 | 5.425 | 5.322 |
| 300 | 2.02 | 3.132 | 5.425 | 4.058 |
| 200 | 2.22 | 3.114 | 5.394 | 3.693 |
| 150 | 2.94 | 3.147 | 5.451 | 2.787 |
| Samples | Particle Size/nm | Ce3+ content/% | Oads Content/% | Refs. |
|---|---|---|---|---|
| Single-crystal CeO2 | 40 ± 5 | 22.03 | 24.02 | [18] |
| CeO2 NPs | 59 ± 7 | 40.22 | 41.18 | This work |
| Single-crystal CeO2 | 60 ± 12 | 29.37 | 30.73 | [18] |
| Porous CeO2 | 79 ± 9 | 10.74 | 76.67 | [47] |
| Porous CeO2 | 105 | 25.37 | \ | [48] |
| mCeO2 | 180 | 35.50 | \ | [49] |
| mCeO2 | 200–300 | 33.88 | 21.17 | [11] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Xie, J.; Feng, K.; Ye, R.; Wang, M.; Wang, Y.; Fan, X.; Liu, R. Controlled Synthesis and Formation Mechanism of Uniformly Sized Spherical CeO2 Nanoparticles. Materials 2026, 19, 211. https://doi.org/10.3390/ma19010211
Xie J, Feng K, Ye R, Wang M, Wang Y, Fan X, Liu R. Controlled Synthesis and Formation Mechanism of Uniformly Sized Spherical CeO2 Nanoparticles. Materials. 2026; 19(1):211. https://doi.org/10.3390/ma19010211
Chicago/Turabian StyleXie, Jiayue, Kai Feng, Rui Ye, Maokui Wang, Yunci Wang, Xing Fan, and Renlong Liu. 2026. "Controlled Synthesis and Formation Mechanism of Uniformly Sized Spherical CeO2 Nanoparticles" Materials 19, no. 1: 211. https://doi.org/10.3390/ma19010211
APA StyleXie, J., Feng, K., Ye, R., Wang, M., Wang, Y., Fan, X., & Liu, R. (2026). Controlled Synthesis and Formation Mechanism of Uniformly Sized Spherical CeO2 Nanoparticles. Materials, 19(1), 211. https://doi.org/10.3390/ma19010211






