Advances in Titanium-Based Biomaterial for Human Bone Scaffolds: Narrative Review on Design, Fabrication, Surface Engineering, Implantation, and Biological Evaluation
Abstract
1. Introduction
1.1. Introduction to Biomaterials and Metal Alloys for Bone Scaffolds


| Class | Material | Young’s Modulus (GPa) | Tensile Strength (MPa) | Biocompatibility | Source |
|---|---|---|---|---|---|
| Metallic | Ti-40Nb Alloy | 30–50 | _ | Excellent | [27] |
| TiAlV | 110–120 | _ | High | [28] | |
| Ti-10Mn Alloy | 76 | 860 | Low | [29] | |
| Co-Cr-Mo Alloy | 200–250 | 900–1540 | Low | [30] | |
| 316L Stainless Steel | 190–210 | 540–1000 | Low | [31] | |
| Mg-Based Alloys | 150–400 | 150–400 | High | [32] | |
| Ceramic | β-Tricalcium Phosphate (β-TCP) | 5–10 | _ | High | [33] |
| Bioactive Glass (45S5) | 35–50 | _ | High | [34] | |
| Zirconia (ZrO2) | 200–210 | _ | High | [35] | |
| Keratin/Hydroxyapatite | _ | _ | High | [36] | |
| HA/HDPE Composite | High | [37] | |||
| Mg-Ca/β-TCP composite | 45 | >300 | High | [38] | |
| Polymer (Synthetic) | PLA/PLGA (Poly-lactic/glycolic acid) | 1–3 | 50–70 | High | [39] |
| PCL composites | 0.004–3 Adjustable | Variable | High | [40] | |
| HA/PCL/gelatin composite | 0.0045 | 1.93 | High | [41] | |
| Polymer (Natural) | Silk Fibroin Composites | 5–15 | _ | High | [42] |
| Chitosan composites | 5.2–100 | _ | High | [43] |
1.2. Titanium Alloy Use for Human Bone
- Investigate the production and processing techniques used for titanium alloys in orthopedic applications.
- Evaluate 3D-modeled scaffold structures and their production methods.
- Find out how different ways of changing the surface of scaffolds affect their mechanical and biological behavior.
- Summarize findings from in vitro mechanical tests and in vivo assessments of osteogenesis and cellular proliferation.
- Figure out what significant changes have happened and what problems still need to be resolved to make implants work better and improve clinical results.

2. Materials and Methods
2.1. Material Examined in Reviewed Studies
2.2. Methodology
2.2.1. Method of Collecting Data
2.2.2. Limitation
2.2.3. Method of Bone Scaffold Preparation
2.2.4. Method of Manufacturing Bone Scaffold
3. Discussion of Results
3.1. Implantation
3.2. In Vivo Evaluation of Scaffold Effectiveness
3.2.1. Effectiveness Evaluation
3.2.2. Osteogenic and Cell Proliferation
3.3. In Vitro Evaluation of Scaffold Effectiveness
Mechanical Test Result
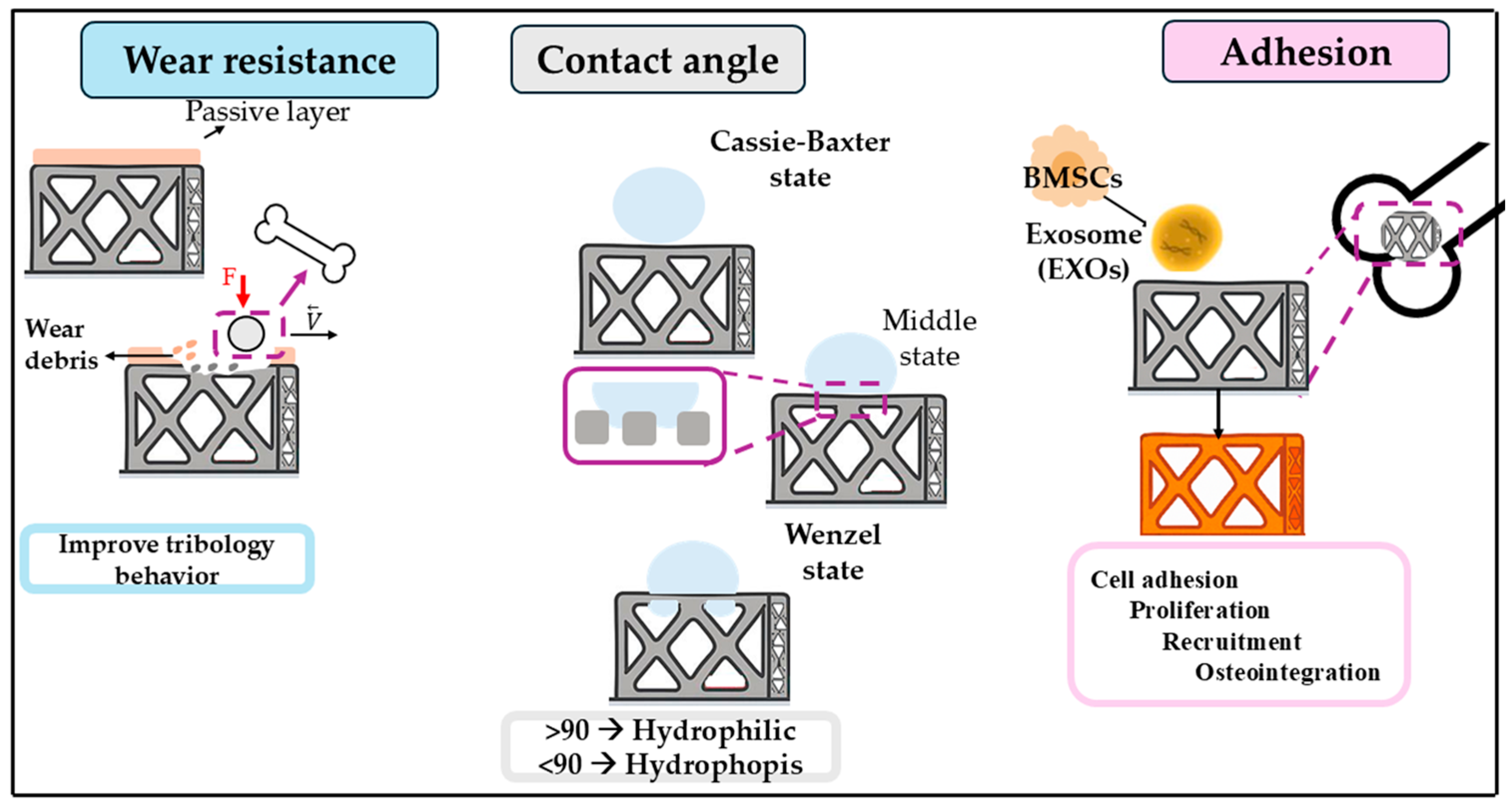
3.4. Result of Surface Modification on Bone Scaffold
3.5. Commercial Titanium Scaffold System
3.6. Challenges and Future Directions in Metal Alloy Bone Scaffold Development
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AD | Anno Domini |
| AM | Additive Manufacturing |
| ASTM | American Society for Testing and Materials |
| BCC | Body-Centered-Cubic |
| BIC | Bone-Implant contact |
| BMP-2 | Bone Morphogenetic Protein-2 |
| BMSC | Marrow–Derived Mesenchymal Stem Cells |
| BT/TV | Bone Tissue to Total Volume ratio |
| C | Circular |
| CAD | Computer-Aided Design |
| CD34 | Cluster of Differentiation 34 |
| CD44 | Cluster of Differentiation 44 |
| CD45 | Cluster of Differentiation 45 |
| CD90 | Cluster of Differentiation 90 (Thy-1) |
| CD105 | Cluster of Differentiation 105 (Endoglin) |
| CP-Ti | Commercially Pure Titanium |
| CT/MRI | Computed Tomography or Magnetic Resonance Imaging |
| CU | Cube |
| DIA | Diamond |
| DOPA | Dopamine |
| ELI | Extra Low Interstitials, Grade 23 |
| FEA | Finite Element Analysis |
| G | Gyroid |
| HCP | Hexagonal-close-packed |
| HPDLSCs | Human Periodontal Ligament Stem Cells |
| ISO | International Organization for Standardizationfacturing |
| LPBF | Laser Powder Bed Fusion |
| Micro-CT | Micro-Computed Tomography |
| MTT assay | (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) |
| P | Primitive |
| PDA-3D PPT | Polydopamine-coated 3D-printed porous titanium |
| PDLSCs | Periodontal Ligament Stem Cells |
| PE | Polyethylene |
| PLA | Polylactic acid |
| SLM | Selective Laser Melting |
| TPMS | Triply Periodic Minimal Surface |
| TC | Truncated cube |
| TC | Tetrahedral close-packed (TC) |
| VEGF | Vascular Endothelial Growth Factor |
| 3D | Three-Dimensional |
| 3D PPT | 3D-printed porous titanium |
| 5Gel-5Alg (S) | 5% gelatin and 5% alginate (5G5A) scaffold |
| 8Gel-2Alg (S) | 8% gelatin and 2% alginate scaffolds |
References
- Troy, E.; Tilbury, M.A.; Power, A.M.; Wall, J.G. Nature-Based Biomaterials and Their Application in Biomedicine. Polymers 2021, 13, 3321. [Google Scholar] [CrossRef]
- Nava, A.; Lugli, F.; Romandini, M.; Badino, F.; Evans, D.; Helbling, A.H.; Oxilia, G.; Arrighi, S.; Bortolini, E.; Delpiano, D.; et al. Early Life of Neanderthals. Proc. Natl. Acad. Sci. USA 2020, 117, 28719–28726. [Google Scholar] [CrossRef]
- Photos-Jones, E. Open Access: From Mine to Apothecary: An Archaeo-Biomedical Approach to the Study of the Greco-Roman Lithotherapeutics Industry. In The Archaeology of Medicine and Healthcare; Routledge: Oxford, UK, 2022; pp. 54–69. [Google Scholar]
- Wallner, C.; Moormann, E.; Lulof, P.; Drysch, M.; Lehnhardt, M.; Behr, B. Burn Care in the Greek and Roman Antiquity. Medicina 2020, 56, 657. [Google Scholar] [CrossRef] [PubMed]
- Aksoy, U.; Aksoy, S.; Kırmızı, D.; Orhan, K. Pierre Fauchard (1678–1761): Pioneering Dental Surgeon of the Enlightenment Age. Cureus 2024, 16, e69563. [Google Scholar] [CrossRef] [PubMed]
- Khan, H.; Barkham, B.; Trompeter, A. The Use of Bioabsorbable Materials in Orthopaedics. Orthop. Trauma 2021, 35, 289–296. [Google Scholar] [CrossRef]
- Kiradzhiyska, D.D.; Mantcheva, R.D. Overview of Biocompatible Materials and Their Use in Medicine. Folia Med. 2019, 61, 34–40. [Google Scholar] [CrossRef]
- Balamurugan, A.; Rajeswari, S.; Balossier, G.; Rebelo, A.H.S.; Ferreira, J.M.F. Corrosion Aspects of Metallic Implants—An Overview. Mater. Corros. 2008, 59, 855–869. [Google Scholar] [CrossRef]
- Navarro, M.; Michiardi, A.; Castaño, O.; Planell, J.A. Biomaterials in Orthopaedics. J. R. Soc. Interface 2008, 5, 1137–1158. [Google Scholar] [CrossRef]
- Huebsch, N.; Mooney, D.J. Inspiration and Application in the Evolution of Biomaterials. Nature 2009, 462, 426–432. [Google Scholar] [CrossRef]
- Vallet-Regí, M. Evolution of Biomaterials. Front. Mater. 2022, 9, 864016. [Google Scholar] [CrossRef]
- Marin, E.; Boschetto, F.; Pezzotti, G. Biomaterials and Biocompatibility: An Historical Overview. J. Biomed. Mater. Res. A 2020, 108, 1617–1633. [Google Scholar] [CrossRef]
- Zhang, X.; Williams, D. Definitions of Biomaterials for the Twenty-First Century; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Dec, P.; Modrzejewski, A.; Pawlik, A. Existing and Novel Biomaterials for Bone Tissue Engineering. Int. J. Mol. Sci. 2022, 24, 529. [Google Scholar] [CrossRef]
- Sun, Z. The Use of Metallic Elements in Bone Repair Scaffolds. Theor. Nat. Sci. 2024, 63, 38–44. [Google Scholar] [CrossRef]
- Todros, S.; Todesco, M.; Bagno, A. Biomaterials and Their Biomedical Applications: From Replacement to Regeneration. Processes 2021, 9, 1949. [Google Scholar] [CrossRef]
- Chen, Y.; Frith, J.E.; Dehghan-Manshadi, A.; Attar, H.; Kent, D.; Soro, N.D.M.; Bermingham, M.J.; Dargusch, M.S. Mechanical Properties and Biocompatibility of Porous Titanium Scaffolds for Bone Tissue Engineering. J. Mech. Behav. Biomed. Mater. 2017, 75, 169–174. [Google Scholar] [CrossRef]
- Ferraz, M.P. An Overview on the Big Players in Bone Tissue Engineering: Biomaterials, Scaffolds and Cells. Int. J. Mol. Sci. 2024, 25, 3836. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, D.; Zhao, X.; Pakvasa, M.; Tucker, A.B.; Luo, H.; Qin, K.H.; Hu, D.A.; Wang, E.J.; Li, A.J.; et al. Stem Cell-Friendly Scaffold Biomaterials: Applications for Bone Tissue Engineering and Regenerative Medicine. Front. Bioeng. Biotechnol. 2020, 8, 598607. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Li, K.; Wu, T.; Chen, J.; Li, S.; Zhang, X. Research Progress in Degradable Metal-based Multifunctional Scaffolds for Bone Tissue Engineering. MedComm-Biomater. Appl. 2023, 2, e60. [Google Scholar] [CrossRef]
- Johnston, A.; Callanan, A. Recent Methods for Modifying Mechanical Properties of Tissue-Engineered Scaffolds for Clinical Applications. Biomimetics 2023, 8, 205. [Google Scholar] [CrossRef]
- Karageorgiou, V.; Kaplan, D. Porosity of 3D Biomaterial Scaffolds and Osteogenesis. Biomaterials 2005, 26, 5474–5491. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, J.L.; Woodrow, K.A. Medical Applications of Porous Biomaterials: Features of Porosity and Tissue-Specific Implications for Biocompatibility. Adv. Healthc. Mater. 2022, 11, 2102087. [Google Scholar] [CrossRef]
- Wang, C.; Xu, D.; Li, S.; Yi, C.; Zhang, X.; He, Y.; Yu, D. Effect of Pore Size on the Physicochemical Properties and Osteogenesis of Ti6Al4V Porous Scaffolds with Bionic Structure. ACS Omega 2020, 5, 28684–28692. [Google Scholar] [CrossRef] [PubMed]
- Wieding, J.; Jonitz, A.; Bader, R. The Effect of Structural Design on Mechanical Properties and Cellular Response of Additive Manufactured Titanium Scaffolds. Materials 2012, 5, 1336–1347. [Google Scholar] [CrossRef]
- Kurowiak, J.; Klekiel, T.; Będziński, R. Biodegradable Polymers in Biomedical Applications: A Review—Developments, Perspectives and Future Challenges. Int. J. Mol. Sci. 2023, 24, 16952. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Han, P.; Dehghan-Manshadi, A.; Kent, D.; Ehtemam-Haghighi, S.; Jowers, C.; Bermingham, M.; Li, T.; Cooper-White, J.; Dargusch, M.S. Sintering and Biocompatibility of Blended Elemental Ti-xNb Alloys. J. Mech. Behav. Biomed. Mater. 2020, 104, 103691. [Google Scholar] [CrossRef]
- Wang, C.; Wu, J.; Liu, L.; Xu, D.; Liu, Y.; Li, S.; Hou, W.; Wang, J.; Chen, X.; Sheng, L.; et al. Improving Osteoinduction and Osteogenesis of Ti6Al4V Alloy Porous Scaffold by Regulating the Pore Structure. Front. Chem. 2023, 11, 1190630. [Google Scholar] [CrossRef]
- Cho, K.; Niinomi, M.; Nakai, M.; Hieda, J.; Santos, P.F.; Itoh, Y.; Hattori, T.; Ikeda, M. Mechanical and Biological Biocompatibilityof Novel β-Type Ti-Mn Alloys for Biomedical Applications. In Materials Science Forum; Trans Tech Publications Ltd.: Bäch, Switzerland, 2014; Volume 783, pp. 1232–1237. [Google Scholar]
- Wanniarachchi, C.T.; Arjunan, A.; Baroutaji, A.; Singh, M. Mechanical Performance of Additively Manufactured Cobalt-Chromium-Molybdenum Auxetic Meta-Biomaterial Bone Scaffolds. J. Mech. Behav. Biomed. Mater. 2022, 134, 105409. [Google Scholar] [CrossRef]
- Čapek, J.; Machová, M.; Fousová, M.; Kubásek, J.; Vojtěch, D.; Fojt, J.; Jablonská, E.; Lipov, J.; Ruml, T. Highly Porous, Low Elastic Modulus 316L Stainless Steel Scaffold Prepared by Selective Laser Melting. Mater. Sci. Eng. C 2016, 69, 631–639. [Google Scholar] [CrossRef] [PubMed]
- Antoniac, I.; Manescu, V.; Paltanea, G.; Antoniac, A.; Nemoianu, I.V.; Petrescu, M.I.; Dura, H.; Bodog, A.D. Additive Manufactured Magnesium-Based Scaffolds for Tissue Engineering. Materials 2022, 15, 8693. [Google Scholar] [CrossRef]
- Bohner, M.; Santoni, B.L.G.; Döbelin, N. β-Tricalcium Phosphate for Bone Substitution: Synthesis and Properties. Acta Biomater. 2020, 113, 23–41. [Google Scholar] [CrossRef]
- Chen, Q.Z.; Thompson, I.D.; Boccaccini, A.R. 45S5 Bioglass®-Derived Glass–Ceramic Scaffolds for Bone Tissue Engineering. Biomaterials 2006, 27, 2414–2425. [Google Scholar] [CrossRef]
- He, Z.; Jiao, C.; Zhang, H.; Xie, D.; Ge, M.; Yang, Y.; Wu, G.; Liang, H.; Shen, L.; Wang, C. Fabrication of a Zirconia/Calcium Silicate Composite Scaffold Based on Digital Light Processing. Ceram. Int. 2022, 48, 25923–25932. [Google Scholar] [CrossRef]
- Feroz, S.; Muhammad, N.; Ullah, R.; Nishan, U.; Cathro, P.; Dias, G. Mechanical Properties, and in Vitro Biocompatibility Assessment of Biomimetic Dual Layered Keratin/Hydroxyapatite Scaffolds. Front. Bioeng. Biotechnol. 2023, 11, 1304147. [Google Scholar] [CrossRef]
- Wang, M.; Au, C.L.; Lai, P.K.; Bonfield, W. Tensile and Compressive Behaviours and Properties of a Bone Analogue Biomaterial. Key Eng. Mater. 2005, 284, 693–696. [Google Scholar] [CrossRef]
- Watanabe, H.; Ikeo, N.; Mukai, T. Processing and Mechanical Properties of a Tricalcium Phosphate-Dispersed Magnesium-Based Composite. Mater. Trans. 2019, 60, 105–110. [Google Scholar] [CrossRef]
- Zhao, D.; Zhu, T.; Li, J.; Cui, L.; Zhang, Z.; Zhuang, X.; Ding, J. Poly (Lactic-Co-Glycolic Acid)-Based Composite Bone-Substitute Materials. Bioact. Mater. 2021, 6, 346–360. [Google Scholar] [CrossRef]
- Rezania, N.; Asadi-Eydivand, M.; Abolfathi, N.; Bonakdar, S.; Mehrjoo, M.; Solati-Hashjin, M. Three-Dimensional Printing of Polycaprolactone/Hydroxyapatite Bone Tissue Engineering Scaffolds Mechanical Properties and Biological Behavior. J. Mater. Sci. Mater. Med. 2022, 33, 31. [Google Scholar] [CrossRef] [PubMed]
- MK, A.S.; Hadi, S.; Sari, Y.W.; Cahyati, N.; Yusuf, Y.; Abdullah, C.A.C. Fabrication and Biocompatibility Evaluation of Hydroxyapatite-Polycaprolactone-Gelatin Composite Nanofibers as a Bone Scaffold. RSC Adv. 2024, 14, 24815–24827. [Google Scholar]
- Johari, N.; Khodaei, A.; Samadikuchaksaraei, A.; Reis, R.L.; Kundu, S.C.; Moroni, L. Ancient Fibrous Biomaterials from Silkworm Protein Fibroin and Spider Silk Blends: Biomechanical Patterns. Acta Biomater. 2022, 153, 38–67. [Google Scholar] [CrossRef] [PubMed]
- Saravanan, S.; Leena, R.S.; Selvamurugan, N. Chitosan Based Biocomposite Scaffolds for Bone Tissue Engineering. Int. J. Biol. Macromol. 2016, 93, 1354–1365. [Google Scholar] [CrossRef]
- Nikita, N.; Kamlesh, W.; Milind, U. An Overview on Biomaterials: Pharmaceutical and Biomedical Applications. J. Drug Deliv. Ther. 2021, 11, 154–161. [Google Scholar] [CrossRef]
- Agrawal, R.; Singh, S.; Saxena, K.K.; Buddhi, D. A Role of Biomaterials in Tissue Engineering and Drug Encapsulation. Proc. Inst. Mech. Eng. Part E J. Process Mech. Eng. 2023, 239, 1626–1636. [Google Scholar] [CrossRef]
- Mahmud, R.U.; Rahman, M.Z. Recent Advances in Biobased Materials and Their Applications; Elsevier: Amsterdam, The Netherlands, 2024. [Google Scholar]
- Abraham, A.M.; Venkatesan, S. A Review on Application of Biomaterials for Medical and Dental Implants. Proc. Inst. Mech. Eng. Part L J. Mater. Des. Appl. 2023, 237, 249–273. [Google Scholar] [CrossRef]
- Ansari, M.A.A.; Golebiowska, A.A.; Dash, M.; Kumar, P.; Jain, P.K.; Nukavarapu, S.P.; Ramakrishna, S.; Nanda, H.S. Engineering Biomaterials to 3D-Print Scaffolds for Bone Regeneration: Practical and Theoretical Consideration. Biomater. Sci. 2022, 10, 2789–2816. [Google Scholar] [CrossRef] [PubMed]
- Owida, H.A.; Al-Ayyad, M.; Turab, N.M.; Al-Nabulsi, J.I. Recent Biomaterial Developments for Bone Tissue Engineering and Potential Clinical Application: Narrative Review of the Literature. Int. J. Online Biomed. Eng. 2023, 19, 4. [Google Scholar]
- Besisa, N.H.; Yajima, T. Titanium-Based Alloys: Classification and Diverse Applications. In Titanium-Based Alloys-Characteristics and Applications; IntechOpen: London, UK, 2024. [Google Scholar]
- Siemers, C.; Haase, F.; Klinge, L. From Titanium Ore Extraction and Processing to Its Applications in the Transportation Industry—An Overview. CIM J. 2023, 14, 158–168. [Google Scholar] [CrossRef]
- Warnke, P.H.; Douglas, T.; Wollny, P.; Sherry, E.; Steiner, M.; Galonska, S.; Becker, S.T.; Springer, I.N.; Wiltfang, J.; Sivananthan, S. Rapid Prototyping: Porous Titanium Alloy Scaffolds Produced by Selective Laser Melting for Bone Tissue Engineering. Tissue Eng. Part C Methods 2009, 15, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Sarraf, M.; Rezvani Ghomi, E.; Alipour, S.; Ramakrishna, S.; Liana Sukiman, N. A State-of-the-Art Review of the Fabrication and Characteristics of Titanium and Its Alloys for Biomedical Applications. Bio-Des. Manuf. 2022, 5, 371–395. [Google Scholar] [CrossRef]
- Gosavi, S.; Gosavi, S.; Alla, R. Titanium: A Miracle Metal in Dentistry. Trends Biomater. Artif. Organs 2013, 27, 42–46. [Google Scholar]
- Van Noort, R. Titanium: The Implant Material of Today. J. Mater. Sci. 1987, 22, 3801–3811. [Google Scholar] [CrossRef]
- Abd-Elaziem, W.; Darwish, M.A.; Hamada, A.; Daoush, W.M. Titanium-Based Alloys and Composites for Orthopedic Implants Applications: A Comprehensive Review. Mater. Des. 2024, 241, 112850. [Google Scholar] [CrossRef]
- Froes, F.; Qian, M. Titanium in Medical and Dental Applications; Woodhead Publishing: Cambridge, UK, 2018. [Google Scholar]
- Jafari, M.; Vaezzadeh, M.; Noroozizadeh, S. Thermal Stability of α Phase of Titanium by Using X-Ray Diffraction. Metall. Mater. Trans. A 2010, 41, 3287–3290. [Google Scholar] [CrossRef]
- Gialanella, S.; Malandruccolo, A. Titanium and Titanium Alloys. In Aerospace Alloys; Topics in Mining, Metallurgy and Materials Engineering; Springer International Publishing: Cham, Switzerland, 2020; pp. 129–189. ISBN 978-3-030-24439-2. [Google Scholar]
- Wang, C.; Lei, Y.; Li, C. Achieving an Excellent Strength and Ductility Balance in Additive Manufactured Ti-6Al-4V Alloy through Multi-Step High-to-Low-Temperature Heat Treatment. Materials 2023, 16, 6947. [Google Scholar] [CrossRef]
- An, F.; Zhang, B.; Yan, Y.; Wang, L. Effect of Vanadium Contents on Microstructure and Mechanical Properties of Ti–6Al–xV Components Produced by Wire + Arc Additive Manufacturing. Mater. Trans. 2021, 62, 1071–1078. [Google Scholar] [CrossRef]
- Buzatu, M.; Geantă, V.; Ştefănoiu, R.; Buţu, M.; Petrescu, M.-I.; Buzatu, M.; Antoniac, I.; Iacob, G.; Niculescu, F.; Ghica, Ş.-I. Investigations into Ti-15Mo-W Alloys Developed for Medical Applications. Materials 2019, 12, 147. [Google Scholar] [CrossRef] [PubMed]
- Ishii, A. Ab Initio Stability Prediction of β Titanium and α and ω Precipitates in β Titanium Matrix for Titanium Alloys Using Density Functional Theory and Micromechanics. Mater. Today Commun. 2024, 38, 107708. [Google Scholar] [CrossRef]
- Moroz, L.S.; Razuvaeva, I.N. Effect of β Stabilizers on the Mechanical Properties of Titanium Alloys with an α Structure. Met. Sci. Heat Treat. 1971, 13, 212–216. [Google Scholar] [CrossRef]
- Siemers, C.; Bäker, M.; Brunke, F.; Wolter, D.; Sibum, H. Aluminum-and Vanadium-Free Titanium Alloys for Application in Medical Engineering. In Titanium in Medical and Dental Applications; Elsevier: Amsterdam, The Netherlands, 2018; pp. 477–492. [Google Scholar]
- Geetha, M.; Singh, A.K.; Asokamani, R.; Gogia, A.K. Ti Based Biomaterials, the Ultimate Choice for Orthopaedic Implants—A Review. Prog. Mater. Sci. 2009, 54, 397–425. [Google Scholar] [CrossRef]
- Challa, V.S.A.; Mali, S.; Misra, R.D.K. Reduced Toxicity and Superior Cellular Response of Preosteoblasts to Ti-6Al-7Nb Alloy and Comparison with Ti-6Al-4V. J. Biomed. Mater. Res. A 2013, 101A, 2083–2089. [Google Scholar] [CrossRef]
- Niinomi, M.; Nakai, M. Titanium-Based Biomaterials for Preventing Stress Shielding between Implant Devices and Bone. Int. J. Biomater. 2011, 2011, 836587. [Google Scholar] [CrossRef]
- Marin, E.; Lanzutti, A. Biomedical Applications of Titanium Alloys: A Comprehensive Review. Materials 2023, 17, 114. [Google Scholar] [CrossRef]
- Michalcová, A.; Vojtěch, D.; Vavřík, J.; Bartha, K.; Beran, P.; Drahokoupil, J.; Džugan, J.; Palán, J.; Čížek, J.; Lejček, P. Structure and Properties of High-Strength Ti Grade 4 Prepared by Severe Plastic Deformation and Subsequent Heat Treatment. Materials 2020, 13, 1116. [Google Scholar] [CrossRef]
- Taleuzzaman, M.; Rahat, I. Patent Application Preparation and Filing. In Intellectual Property Rights-Patent; IntechOpen: London, UK, 2020. [Google Scholar]
- Sordi, V.L.; Ferrante, M.; Kawasaki, M.; Langdon, T.G. Microstructure and Tensile Strength of Grade 2 Titanium Processed by Equal-Channel Angular Pressing and by Rolling. J. Mater. Sci. 2012, 47, 7870–7876. [Google Scholar] [CrossRef]
- Szkliniarz, A.; Szkliniarz, W. Microstructure and Properties of Ti-5Al-2.5 Sn Alloy with Higher Carbon Content. Coatings 2025, 15, 224. [Google Scholar] [CrossRef]
- Nikiel, P.; Wróbel, M.; Szczepanik, S.; Stępień, M.; Wierzbanowski, K.; Baczmański, A. Microstructure and Mechanical Properties of Titanium Grade 23 Produced by Selective Laser Melting. Arch. Civ. Mech. Eng. 2021, 21, 152. [Google Scholar] [CrossRef]
- Niinomi, M. Mechanical Properties of Biomedical Titanium Alloys. Mater. Sci. Eng. A 1998, 243, 231–236. [Google Scholar] [CrossRef]
- Polmear, I.; StJohn, D.; Nie, J.-F.; Qian, M. Light Alloys: Metallurgy of the Light Metals; Butterworth-Heinemann: Oxford, UK, 2017. [Google Scholar]
- Destro, F.; Borgatti, M.; Iafelice, B.; Gavioli, R.; Braun, T.; Bauer, J.; Böttcher, L.; Jung, E.; Bocchi, M.; Guerrieri, R.; et al. Effects of Biomaterials for Lab-on-a-Chip Production on Cell Growth and Expression of Differentiated Functions of Leukemic Cell Lines. J. Mater. Sci. Mater. Med. 2010, 21, 2653–2664. [Google Scholar] [CrossRef]
- Chauhan, S.R.; Dass, K. Dry Sliding Wear Behaviour of Titanium (Grade 5) Alloy by Using Response Surface Methodology. Adv. Tribol. 2013, 2013, 272106. [Google Scholar] [CrossRef]
- Maehara, K.; Doi, K.; Matsushita, T.; Sasaki, Y. Application of Vanadium-Free Titanium Alloys to Artificial Hip Joints. Mater. Trans. 2002, 43, 2936–2942. [Google Scholar] [CrossRef]
- Ronoh, K.; Mwema, F.; Dabees, S.; Sobola, D. Advances in Sustainable Grinding of Different Types of the Titanium Biomaterials for Medical Applications: A Review. Biomed. Eng. Adv. 2022, 4, 100047. [Google Scholar] [CrossRef]
- Okazaki, Y. A New Ti–15Zr–4Nb–4Ta Alloy for Medical Applications. Curr. Opin. Solid State Mater. Sci. 2001, 5, 45–53. [Google Scholar] [CrossRef]
- Bocchetta, P.; Chen, L.-Y.; Tardelli, J.D.C.; dos Reis, A.C.; Almeraya-Calderón, F.; Leo, P. Passive Layers and Corrosion Resistance of Biomedical Ti-6Al-4V and β-Ti Alloys. Coatings 2021, 11, 487. [Google Scholar] [CrossRef]
- Tamilselvi, S.; Raman, V.; Rajendran, N. Corrosion Behaviour of Ti–6Al–7Nb and Ti–6Al–4V ELI Alloys in the Simulated Body Fluid Solution by Electrochemical Impedance Spectroscopy. Electrochim. Acta 2006, 52, 839–846. [Google Scholar] [CrossRef]
- Muhammad, F.; Sari, S.N.; Dilasari, B. The Effect of Titanium Nanostructure on Corrosion Resistance as Dental Implants: A Review. Indones. J. Mater. Res. 2024, 2, 7–15. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, M.; Li, K.; Hu, J. Study on the Surface Properties and Biocompatibility of Nanosecond Laser Patterned Titanium Alloy. Opt. Laser Technol. 2021, 139, 106987. [Google Scholar] [CrossRef]
- Ezura, A.; Katahira, K.; Komotori, J. Generation of Biocompatible Titanium Alloy Surfaces Including Calcium and Phosphorus Elements by Laser-Induced Mist Spraying Wet Treatment. Int. J. Autom. Technol. 2020, 14, 575–581. [Google Scholar] [CrossRef]
- Fan, X.; Fan, W. Preparation and Biological Activity Study of Ti13Nb13Zr Surface Nanotubes. Biosurf. Biotribol. 2024, 10, 159–166. [Google Scholar] [CrossRef]
- Markhoff, J.; Krogull, M.; Schulze, C.; Rotsch, C.; Hunger, S.; Bader, R. Biocompatibility and Inflammatory Potential of Titanium Alloys Cultivated with Human Osteoblasts, Fibroblasts and Macrophages. Materials 2017, 10, 52. [Google Scholar] [CrossRef]
- Shaik, M.; Subramanyam, G. Stem Cell Scaffolds for Tissue Engineering. Curr. Trends Biomed. Eng. Biosci. 2017, 9, 3–6. [Google Scholar] [CrossRef]
- Goswami, R.; Ghosh, S.; Dasgupta, S.; Dey, A.; Banerjee, R.; Basak, I.; Bhuniya, T. Recent Advances in Tissue Engineering Scaffolds and Commercial Applications. YMER 2022, 21, 865–882. [Google Scholar] [CrossRef]
- González-Carrasco, J.L. Metals as Bone Repair Materials. In Bone Repair Biomaterials; Elsevier: Amsterdam, The Netherlands, 2009; pp. 154–193. [Google Scholar]
- Elsayed, H.S. Biodegradable and Bioactive Porous Polymer/Inorganic Nanocomposites Scaffolds for Biomedical Applications. Ph.D. Thesis, University of Padua, Padua, Italy, 2017. [Google Scholar]
- Santos-Rosales, V.; Iglesias-Mejuto, A.; García-González, C.A. Solvent-Free Approaches for the Processing of Scaffolds in Regenerative Medicine. Polymers 2020, 12, 533. [Google Scholar] [CrossRef]
- Wu, H.; Yang, S.; Li, J.; Ma, T.; Yang, K.; Liao, T.; Feng, W.; Zhou, B.; Yong, X.; Zhou, K.; et al. Current Status and Challenges of Shape Memory Scaffolds in Biomedical Applications. MedComm-Biomater. Appl. 2024, 3, e95. [Google Scholar] [CrossRef]
- Shi, Q.; Shui, H.; Chen, Q.; Li, Z.-Y. How Does Mechanical Stimulus Affect the Coupling Process of the Scaffold Degradation and Bone Formation: An in Silico Approach. Comput. Biol. Med. 2020, 117, 103588. [Google Scholar] [CrossRef]
- Tajvar, S.; Hadjizadeh, A.; Samandari, S.S. Scaffold Degradation in Bone Tissue Engineering: An Overview. Int. Biodeterior. Biodegrad. 2023, 180, 105599. [Google Scholar] [CrossRef]
- Scott, T.E.; Guelcher, S.A. Advanced Scaffold Fabrication Using Additive Manufacturing. In Biomaterial Based Approaches to Study the Tumour Microenvironment; Royal Society of Chemistry: London, UK, 2022. [Google Scholar]
- Babbar, A.; Jain, V.; Gupta, D.; Sharma, A.; Prakash, C.; Kumar, V.; Goyal, K.K. Additive Manufacturing for the Development of Biological Implants, Scaffolds, and Prosthetics. In Additive Manufacturing Processes in Biomedical Engineering; CRC Press: Boca Raton, FL, USA, 2022; pp. 27–46. [Google Scholar]
- Patiño, M.D.; Chávez, M.; Bribiesca, M.R.; Cisneros, G.V. Development of Biomaterials with Therapeutically Medicinal Application. MRS Online Proc. Libr. OPL 2009, 1244, 1. [Google Scholar] [CrossRef]
- Jain, A.K. Research in Orthopedics: A Necessity. Indian J. Orthop. 2009, 43, 315–317. [Google Scholar] [CrossRef]
- Hooper, G. The Ageing Population and the Increasing Demand for Joint Replacement. J. N. Z. Med. Assoc. 2013, 126, 5–6. [Google Scholar]
- Goncalves, A.D.; Balestri, W.; Reinwald, Y. Biomedical Implants for Regenerative Therapies. Biomaterials 2020, 9, 1–36. [Google Scholar]
- Rodrigues, L.M.; Lima Zutin, E.A.; Sartori, E.M.; Mendonça, D.B.S.; Mendonça, G.; Carvalho, Y.R.; Reis De Vasconcellos, L.M. Influence of Titanium Alloy Scaffolds on Enzymatic Defense against Oxidative Stress and Bone Marrow Cell Differentiation. Int. J. Biomater. 2020, 2020, 1708214. [Google Scholar] [CrossRef] [PubMed]
- Guillemot, F. Recent Advances in the Design of Titanium Alloys for Orthopedic Applications. Expert Rev. Med. Devices 2005, 2, 741–748. [Google Scholar] [CrossRef] [PubMed]
- Taddei, E.B.; Henriques, V.A.R.; Silva, C.R.M.; Cairo, C.A.A. Production of New Titanium Alloy for Orthopedic Implants. Mater. Sci. Eng. C 2004, 24, 683–687. [Google Scholar] [CrossRef]
- Oliveira, V.; Chaves, R.R.; Bertazzoli, R.; Caram, R. Preparation and Characterization of Ti-Al-Nb Alloys for Orthopedic Implants. Braz. J. Chem. Eng. 1998, 15, 326–333. [Google Scholar] [CrossRef]
- Jin, W.; Chu, P.K. Orthopedic Implants. In Encyclopedia of Biomedical Engineering; Elsevier: Amsterdam, The Netherlands, 2019; pp. 425–439. ISBN 978-0-12-805144-3. [Google Scholar]
- Afonso, C.R.M.; Aleixo, G.T.; Ramirez, A.J.; Caram, R. Influence of Cooling Rate on Microstructure of Ti–Nb Alloy for Orthopedic Implants. Mater. Sci. Eng. C 2007, 27, 908–913. [Google Scholar] [CrossRef]
- Castagnini, F.; Bordini, B.; Stea, S.; Calderoni, P.P.; Masetti, C.; Busanelli, L. Highly Porous Titanium Cup in Cementless Total Hip Arthroplasty: Registry Results at Eight Years. Int. Orthop. 2019, 43, 1815–1821. [Google Scholar] [CrossRef]
- Sass, J.-O.; Kebbach, M.; Lork, C.; Johannsen, J.; Weinmann, M.; Stenzel, M.; Bader, R. Computational Biomechanical Study on Hybrid Implant Materials for the Femoral Component of Total Knee Replacements. J. Mech. Behav. Biomed. Mater. 2024, 158, 106681. [Google Scholar] [CrossRef]
- Apostolopoulos, V.; Tomáš, T.; Boháč, P.; Marcián, P.; Mahdal, M.; Valoušek, T.; Janíček, P.; Nachtnebl, L. Biomechanical Analysis of All-Polyethylene Total Knee Arthroplasty on Periprosthetic Tibia Using the Finite Element Method. Comput. Methods Programs Biomed. 2022, 220, 106834. [Google Scholar] [CrossRef] [PubMed]
- Campanelli, L.C. A Review on the Recent Advances Concerning the Fatigue Performance of Titanium Alloys for Orthopedic Applications. J. Mater. Res. 2021, 36, 151–165. [Google Scholar] [CrossRef]
- Olin, C. Titanium in Cardiac and Cardiovascular Applications. In Titanium in Medicine: Material Science, Surface Science, Engineering, Biological Responses and Medical Applications; Brunette, D.M., Tengvall, P., Textor, M., Thomsen, P., Eds.; Springer: Berlin/Heidelberg, Germany, 2001; pp. 889–907. ISBN 978-3-642-56486-4. [Google Scholar]
- Beshchasna, N.; Ho, A.Y.K.; Saqib, M.; Kraśkiewicz, H.; Wasyluk, Ł.; Kuzmin, O.; Duta, O.C.; Ficai, D.; Trusca, R.D.; Ficai, A. Surface Evaluation of Titanium Oxynitride Coatings Used for Developing Layered Cardiovascular Stents. Mater. Sci. Eng. C 2019, 99, 405–416. [Google Scholar] [CrossRef]
- Grenadyorov, A.S.; Zhulkov, M.O.; Solovyev, A.A.; Oskomov, K.V.; Semenov, V.A.; Chernyavskiy, A.M.; Sirota, D.A.; Karmadonova, N.A.; Malashchenko, V.V.; Litvinova, L.S. Surface Characterization and Biological Assessment of Corrosion-Resistant aC: H: SiOx PACVD Coating for Ti-6Al-4V Alloy. Mater. Sci. Eng. C 2021, 123, 112002. [Google Scholar] [CrossRef] [PubMed]
- Nagaraja, S.; Brown, R.; Saylor, D.; Undisz, A. Oxide Layer Formation, Corrosion, and Biocompatibility of Nitinol Cardiovascular Devices. Shape Mem. Superelasticity 2022, 8, 45–63. [Google Scholar] [CrossRef]
- Norlin, A.; Pan, J.; Leygraf, C. Investigation of Pt, Ti, TiN, and Nano-Porous Carbon Electrodes for Implantable Cardioverter-Defibrillator Applications. Electrochim. Acta 2004, 49, 4011–4020. [Google Scholar] [CrossRef]
- Hirshorn, M.S.; Holley, L.K.; Money, D.K.; Spector, M.; Young, F.A.; Hales, J.R.S. Histological Evaluation of Porous Titanium Cardiac Pacemaker Electrode Tips. J. Biomed. Mater. Res. 1984, 18, 47–60. [Google Scholar] [CrossRef] [PubMed]
- Prima, F.; Sun, F.; Vermaut, P.; Gloriant, T.; Mantovani, D.; Jacques, P.J. High Performance Beta Titanium Alloys as a New Material Perspective for Cardiovascular Applications. Mater. Sci. Forum 2012, 706–709, 578–583. [Google Scholar] [CrossRef]
- Ye, S.-H.; Johnson, C.A., Jr.; Woolley, J.R.; Snyder, T.A.; Gamble, L.J.; Wagner, W.R. Covalent Surface Modification of a Titanium Alloy with a Phosphorylcholine-Containing Copolymer for Reduced Thrombogenicity in Cardiovascular Devices. J. Biomed. Mater. Res. A 2009, 91A, 18–28. [Google Scholar] [CrossRef]
- Mohanta, M.; Thirugnanam, A. Commercial Pure Titanium—A Potential Candidate for Cardiovascular Stent. Mater. Werkst. 2022, 53, 1518–1543. [Google Scholar] [CrossRef]
- Asirvatham, A.; Devadoss, D.; Kujur, A.; Selvam, A.; Devi, J.N.; Mary, S.J. Anti Corrosion Activity of CRF (Cardiac Risk Free) Drug for SS316L, Ni–Ti, and Ti-6Al-4V in Artificial Blood Plasma. Chem. Afr. 2024, 7, 891–903. [Google Scholar] [CrossRef]
- Ji, H.; Xie, X.; Jiang, Z.; Wu, X. Wear and Corrosion of Titanium Alloy Spinal Implants in Vivo. Sci. Rep. 2024, 14, 16847. [Google Scholar] [CrossRef]
- Lah, N.A.C.; Hussin, M.H. Titanium and Titanium Based Alloys as Metallic Biomaterials in Medical Applications—Spine Implant Case Study. Pertanika J. Sci. Technol. 2019, 27, 459–472. [Google Scholar]
- Massaad, E.; Fatima, N.; Kiapour, A.; Hadzipasic, M.; Shankar, G.M.; Shin, J.H. Polyetheretherketone versus Titanium Cages for Posterior Lumbar Interbody Fusion: Meta-Analysis and Review of the Literature. Neurospine 2020, 17, 125. [Google Scholar] [CrossRef]
- Dick, J.C.; Bourgeault, C.A. Notch Sensitivity of Titanium Alloy, Commercially Pure Titanium, and Stainless Steel Spinal Implants. Spine 2001, 26, 1668–1672. [Google Scholar] [CrossRef]
- Kasai, Y.; Iida, R.; Uchida, A. Metal Concentrations in the Serum and Hair of Patients with Titanium Alloy Spinal Implants. Spine 2003, 28, 1320–1326. [Google Scholar] [CrossRef]
- Fukutake, K.; Wada, A.; Kamakura, D.; Nakamura, K.; Tsuge, S.; Hasegawa, K.; Takahashi, H. Evaluation of Percutaneous Pedicle Screw Fixation in Patients with Pyogenic Spondylitis of the Thoracolumbar Spine. Open J. Orthop. 2020, 10, 303–312. [Google Scholar] [CrossRef]
- Kim, H.-D.; Kim, K.-S.; Ki, S.-C.; Choi, Y.-S. Electron Microprobe Analysis and Tissue Reaction around Titanium Alloy Spinal Implants. Asian Spine J. 2007, 1, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Wang, D.; Dou, W.; Wang, Y.; Yang, Y.; Wang, J.; Chen, J. Design and Manufacture of Bionic Porous Titanium Alloy Spinal Implant Based on Selective Laser Melting (SLM). Comput. Model. Eng. Sci. 2020, 124, 1099–1117. [Google Scholar] [CrossRef]
- Gegner, J. Tribology: Fundamentals and Advancements; BoD—Books on Demand: Norderstedt, Germany, 2013; ISBN 978-953-51-1135-1. [Google Scholar]
- Vancleef, S.; Wesseling, M.; Duflou, J.R.; Nijs, S.; Jonkers, I.; Vander Sloten, J. Thin Patient-specific Clavicle Fracture Fixation Plates Can Mechanically Outperform Commercial Plates: An in Silico Approach. J. Orthop. Res. 2022, 40, 1695–1706. [Google Scholar] [CrossRef]
- Zhang, S.; Yu, Y.; Wang, H.; Ren, L.; Yang, K. Study on Mechanical Behavior of Cu-Bearing Antibacterial Titanium Alloy Implant. J. Mech. Behav. Biomed. Mater. 2022, 125, 104926. [Google Scholar] [CrossRef]
- Perren, S.M.; Regazzoni, P.; Fernandez, A.A. How to Choose between the Implant Materials Steel and Titanium in Orthopedic Trauma Surgery: Part 2–Biological Aspects. Acta Chir. Orthop. Traumatol. Cech. 2017, 84, 85–90. [Google Scholar] [CrossRef]
- Zigterman, B.G.R.; Van den Borre, C.; Braem, A.; Mommaerts, M.Y. Titanium Surface Modifications and Their Soft-Tissue Interface on Nonkeratinized Soft Tissues—A Systematic Review (Review). Biointerphases 2019, 14, 040802. [Google Scholar] [CrossRef]
- Abrahamsson, I.; Cardaropoli, G. Peri-Implant Hard and Soft Tissue Integration to Dental Implants Made of Titanium and Gold. Clin. Oral Implant. Res. 2007, 18, 269–274. [Google Scholar] [CrossRef]
- Suchenski, M.; McCarthy, M.B.; Chowaniec, D.; Hansen, D.; McKinnon, W.; Apostolakos, J.; Arciero, R.; Mazzocca, A.D. Material Properties and Composition of Soft-Tissue Fixation. Arthrosc. J. Arthrosc. Relat. Surg. 2010, 26, 821–831. [Google Scholar] [CrossRef]
- Corvino, E.; Pesce, P.; Mura, R.; Marcano, E.; Canullo, L. Influence of Modified Titanium Abutment Surface on Peri-Implant Soft Tissue Behavior: A Systematic Review of in Vitro Studies. Int. J. Oral Maxillofac. Implant. 2020, 35, 503–519. [Google Scholar] [CrossRef]
- Liu, R.; Chen, S.; Huang, P.; Liu, G.; Luo, P.; Li, Z.; Xiao, Y.; Chen, Z.; Chen, Z. Immunomodulation-Based Strategy for Improving Soft Tissue and Metal Implant Integration and Its Implications in the Development of Metal Soft Tissue Materials. Adv. Funct. Mater. 2020, 30, 1910672. [Google Scholar] [CrossRef]
- Shemyatovsky, K.A.; Azimov, R.H.; Alekhin, A.I.; Kazantsev, A.A.; Alekhin, A.A. Computed Tomography Options in the Evaluation of Hernia Repair Outcomes Using “Titanium Silk” Mesh Implants. J. Tissue Eng. Regen. Med. 2020, 14, 684–689. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, I.; Kluge, M. Investigation of an Increased Particle Size Distribution of Ti-6Al-4V Powders Used for Laser-Based Powder Bed Fusion of Metals. Materials 2024, 17, 2942. [Google Scholar] [CrossRef]
- Pattanayak, D.K.; Fukuda, A.; Matsushita, T.; Takemoto, M.; Fujibayashi, S.; Sasaki, K.; Nishida, N.; Nakamura, T.; Kokubo, T. Bioactive Ti Metal Analogous to Human Cancellous Bone: Fabrication by Selective Laser Melting and Chemical Treatments. Acta Biomater. 2011, 7, 1398–1406. [Google Scholar] [CrossRef]
- Guo, Y.; Ren, L.; Xie, K.; Wang, L.; Yu, B.; Jiang, W.; Zhao, Y.; Hao, Y. Functionalized TiCu/Ti-Cu-N-Coated 3D-Printed Porous Ti6Al4V Scaffold Promotes Bone Regeneration through BMSC Recruitment. Adv. Mater. Interfaces 2020, 7, 1901632. [Google Scholar] [CrossRef]
- Deng, F.; Liu, L.; Li, Z.; Liu, J. 3D Printed Ti6Al4V Bone Scaffolds with Different Pore Structure Effects on Bone Ingrowth. J. Biol. Eng. 2021, 15, 4. [Google Scholar] [CrossRef]
- Wang, C.; Xu, D.; Lin, L.; Li, S.; Hou, W.; He, Y.; Sheng, L.; Yi, C.; Zhang, X.; Li, H.; et al. Large-Pore-Size Ti6Al4V Scaffolds with Different Pore Structures for Vascularized Bone Regeneration. Mater. Sci. Eng. C 2021, 131, 112499. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Zhang, Y.; Wu, Y.; Zhang, Z.; Jiang, D.; Jia, R.; Wang, X.; Liu, Z. In Vitro and in Vivo Analysis of the Effects of 3D-Printed Porous Titanium Alloy Scaffold Structure on Osteogenic Activity. BioMed Res. Int. 2022, 2022, 8494431. [Google Scholar] [CrossRef]
- Chen, C.; Hao, Y.; Bai, X.; Ni, J.; Chung, S.-M.; Liu, F.; Lee, I.-S. 3D Printed Porous Ti6Al4V Cage: Effects of Additive Angle on Surface Properties and Biocompatibility; Bone Ingrowth in Beagle Tibia Model. Mater. Des. 2019, 175, 107824. [Google Scholar] [CrossRef]
- Chen, Z.; Yan, X.; Yin, S.; Liu, L.; Liu, X.; Zhao, G.; Ma, W.; Qi, W.; Ren, Z.; Liao, H.; et al. Influence of the Pore Size and Porosity of Selective Laser Melted Ti6Al4V ELI Porous Scaffold on Cell Proliferation, Osteogenesis and Bone Ingrowth. Mater. Sci. Eng. C 2020, 106, 110289. [Google Scholar] [CrossRef]
- Gryko, A.; Prochor, P.; Sajewicz, E. Finite Element Analysis of the Influence of Porosity and Pore Geometry on Mechanical Properties of Orthopaedic Scaffolds. J. Mech. Behav. Biomed. Mater. 2022, 132, 105275. [Google Scholar] [CrossRef]
- Tilton, M.; Lewis, G.S.; Hast, M.W.; Fox, E.; Manogharan, G. Additively Manufactured Patient-Specific Prosthesis for Tumor Reconstruction: Design, Process, and Properties. PLoS ONE 2021, 16, e0253786. [Google Scholar] [CrossRef]
- Antounian, F.; Avagyan, H.; Ghaltaghchyan, T.; Holovenko, Y.; Khachatryan, H.; Aghayan, M. Designing and Additive Manufacturing of Talus Implant for Post-Traumatic Talus Avascular Necrosis: A Case Study. J. Orthop. Surg. Res. 2024, 19, 501. [Google Scholar] [CrossRef] [PubMed]
- Hindy, A.; Farahmand, F.; Pourdanesh, F.; Torshabi, M.; Al Janabi, A.H.; Rasoulianboroujeni, M.; Tayebi, L.; Tabatabaei, F.S. Synthesis and Characterization of 3D-Printed Functionally Graded Porous Titanium Alloy. J. Mater. Sci. 2020, 55, 9082–9094. [Google Scholar] [CrossRef]
- Sun, C.; Dong, E.; Chen, J.; Zheng, J.; Kang, J.; Jin, Z.; Liu, C.; Wang, L.; Li, D. The Promotion of Mechanical Properties by Bone Ingrowth in Additive-Manufactured Titanium Scaffolds. J. Funct. Biomater. 2022, 13, 127. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Zhao, P.; Lin, L.; Qin, L.; Huan, Z.; Leeflang, S.; Zadpoor, A.A.; Zhou, J.; Wu, L. Surface-Treated 3D Printed Ti-6Al-4V Scaffolds with Enhanced Bone Regeneration Performance: An In Vivo Study. Ann. Transl. Med. 2021, 9, 39. [Google Scholar] [CrossRef]
- Zhong, W.; Li, J.; Hu, C.; Quan, Z.; Jiang, D.; Huang, G.; Wang, Z. 3D-Printed Titanium Implant-Coated Polydopamine for Repairing Femoral Condyle Defects in Rabbits. J. Orthop. Surg. 2020, 15, 102. [Google Scholar] [CrossRef]
- Ma, L.; Wang, X.; Zhou, Y.; Ji, X.; Cheng, S.; Bian, D.; Fan, L.; Zhou, L.; Ning, C.; Zhang, Y. Biomimetic Ti–6Al–4V Alloy/Gelatin Methacrylate Hybrid Scaffold with Enhanced Osteogenic and Angiogenic Capabilities for Large Bone Defect Restoration. Bioact. Mater. 2021, 6, 3437–3448. [Google Scholar] [CrossRef]
- Crovace, A.M.; Lacitignola, L.; Forleo, D.M.; Staffieri, F.; Francioso, E.; Di Meo, A.; Becerra, J.; Crovace, A.; Santos-Ruiz, L. 3D Biomimetic Porous Titanium (Ti6Al4V ELI) Scaffolds for Large Bone Critical Defect Reconstruction: An Experimental Study in Sheep. Animals 2020, 10, 1389. [Google Scholar] [CrossRef]
- Li, L.; Shi, J.; Zhang, K.; Yang, L.; Yu, F.; Zhu, L.; Liang, H.; Wang, X.; Jiang, Q. Early Osteointegration Evaluation of Porous Ti6Al4V Scaffolds Designed Based on Triply Periodic Minimal Surface Models. J. Orthop. Transl. 2019, 19, 94–105. [Google Scholar] [CrossRef] [PubMed]
- Fan, B.; Guo, Z.; Li, X.; Li, S.; Gao, P.; Xiao, X.; Wu, J.; Shen, C.; Jiao, Y.; Hou, W. Electroactive Barium Titanate Coated Titanium Scaffold Improves Osteogenesis and Osseointegration with Low-Intensity Pulsed Ultrasound for Large Segmental Bone Defects. Bioact. Mater. 2020, 5, 1087–1101. [Google Scholar] [CrossRef] [PubMed]
- Ragone, V.; Canciani, E.; Arosio, M.; Olimpo, M.; Piras, L.A.; Von Degerfeld, M.M.; Augusti, D.; D’Ambrosi, R.; Dellavia, C. In Vivo Osseointegration of a Randomized Trabecular Titanium Structure Obtained by an Additive Manufacturing Technique. J. Mater. Sci. Mater. Med. 2020, 31, 17. [Google Scholar] [CrossRef]
- Liu, L.; Duan, J.; Shi, Q.; Chen, Q.; Yao, Q.; Li, Z. Mechanical Effect on the Evolution of Bone Formation during Bone Ingrowth into a 3D-Printed Ti-Alloy Scaffold. Mater. Lett. 2020, 273, 127921. [Google Scholar] [CrossRef]
- Li, L.; Li, Y.; Yang, L.; Yu, F.; Zhang, K.; Jin, J.; Shi, J.; Zhu, L.; Liang, H.; Wang, X.; et al. Polydopamine Coating Promotes Early Osteogenesis in 3D Printing Porous Ti6Al4V Scaffolds. Ann. Transl. Med. 2019, 7, 240. [Google Scholar] [CrossRef]
- Luan, H.; Wang, L.; Ren, W.; Chu, Z.; Huang, Y.; Lu, C.; Fan, Y. The Effect of Pore Size and Porosity of Ti6Al4V Scaffolds on MC3T3-E1 Cells and Tissue in Rabbits. Sci. China Technol. Sci. 2019, 62, 1160–1168. [Google Scholar] [CrossRef]
- Yu, A.; Zhang, C.; Xu, W.; Zhang, Y.; Tian, S.; Liu, B.; Zhang, J.; He, A.; Su, B.; Lu, X. Additive Manufacturing of Multi-Morphology Graded Titanium Scaffolds for Bone Implant Applications. J. Mater. Sci. Technol. 2023, 139, 47–58. [Google Scholar] [CrossRef]
- Mustafa, N.S.; Akhmal, N.H.; Izman, S.; Ab Talib, M.H.; Shaiful, A.I.M.; Omar, M.N.B.; Yahaya, N.Z.; Illias, S. Application of Computational Method in Designing a Unit Cell of Bone Tissue Engineering Scaffold: A Review. Polymers 2021, 13, 1584. [Google Scholar] [CrossRef]
- Al-Hammadi, A.S.S.; Saidin, S.; Ramlee, M.H. Simulation Analyses Related to Human Bone Scaffold: Utilisation of Solidworks® Software in 3D Modelling and Mechanical Simulation Analyses. J. Hum. Centered Technol. 2022, 1, 97–104. [Google Scholar] [CrossRef]
- Sanas, M.M.; Mulay, A.V. Assessment on Effect of Physical Parameters of Scaffold on Its Strength through Finite Element Analysis. In Proceedings of the 9th National Conference on Recent Developments in Mechanical Engineering [RDME 2021], Pune, India, 18 August 2021; AIP Publishing: Melville, NY, USA, 2022; Volume 2469. [Google Scholar]
- Uth, N.P. Computational Design and Optimization of Bone Tissue Engineering Scaffold Topology. Master’s Thesis, Miami University, Oxford, OH, USA, 2016. [Google Scholar]
- Li, Y.; Yang, C.; Zhao, H.; Qu, S.; Li, X.; Li, Y. New Developments of Ti-Based Alloys for Biomedical Applications. Materials 2014, 7, 1709–1800. [Google Scholar] [CrossRef]
- Bai, L.; Zhang, J.; Xiong, Y.; Chen, X.; Sun, Y.; Gong, C.; Pu, H.; Wu, X.; Luo, J. Influence of Unit Cell Pose on the Mechanical Properties of Ti6Al4V Lattice Structures Manufactured by Selective Laser Melting. Addit. Manuf. 2020, 34, 101222. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, L.; Liu, L.; Lv, L.; Gao, L.; Liu, N.; Wang, X.; Ye, J. Mechanical Behavior of a Titanium Alloy Scaffold Mimicking Trabecular Structure. J. Orthop. Surg. 2020, 15, 40. [Google Scholar] [CrossRef]
- Calazans Neto, J.V.; da Costa Valente, M.L.; do Reis, A.C. Effect of Pores on Cell Adhesion to Additively Manufactured Titanium Implants: A Systematic Review. J. Prosthet. Dent. 2025, 133, 990–997. [Google Scholar] [CrossRef]
- Günther, F.; Wagner, M.; Pilz, S.; Gebert, A.; Zimmermann, M. Design Procedure for Triply Periodic Minimal Surface Based Biomimetic Scaffolds. J. Mech. Behav. Biomed. Mater. 2022, 126, 104871. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.C.; Wang, J.; Liu, Y.; Jia, Z.; Liang, S.X. Additive Manufacturing of Titanium Alloys. In Encyclopedia of Materials: Metals and Alloys; Elsevier: Amsterdam, The Netherlands, 2021; pp. 256–274. [Google Scholar]
- Das, M.; Balla, V.K. Additive Manufacturing of Titanium and Alloys. In Springer Handbook of Additive Manufacturing; Pei, E., Bernard, A., Gu, D., Klahn, C., Monzón, M., Petersen, M., Sun, T., Eds.; Springer Handbooks; Springer International Publishing: Cham, Switzerland, 2023; pp. 671–698. ISBN 978-3-031-20751-8. [Google Scholar]
- Dutta, B.; Froes, F.H.S. The Additive Manufacturing (AM) of Titanium Alloys. In Titanium Powder Metallurgy; Elsevier: Amsterdam, The Netherlands, 2015; pp. 447–468. [Google Scholar]
- Rahmani, R.; Lopes, S.I.; Prashanth, K.G. Selective Laser Melting and Spark Plasma Sintering: A Perspective on Functional Biomaterials. J. Funct. Biomater. 2023, 14, 521. [Google Scholar] [CrossRef] [PubMed]
- Wen, C. Structural Biomaterials: Properties, Characteristics, and Selection; Woodhead Publishing: Cambridge, UK, 2021. [Google Scholar]
- Yang, Y.; Wang, G.; Liang, H.; Gao, C.; Peng, S.; Shen, L.; Shuai, C. Additive Manufacturing of Bone Scaffolds. Int. J. Bioprint. 2018, 5, 148. [Google Scholar] [CrossRef]
- Nune, K.C.; Li, S.; Misra, R.; Kumar, D. Advancements in Three-Dimensional Titanium Alloy Mesh Scaffolds Fabricated by Electron Beam Melting for Biomedical Devices: Mechanical and Biological Aspects. Sci. China Mater. 2018, 61, 455–474. [Google Scholar] [CrossRef]
- Gaur, B.; Soman, D.; Ghyar, R.; Bhallamudi, R. Ti6Al4V Scaffolds Fabricated by Laser Powder Bed Fusion with Hybrid Volumetric Energy Density. Rapid Prototyp. J. 2023, 29, 67–79. [Google Scholar] [CrossRef]
- Entezari, A.; Wu, Q.; Mirkhalaf, M.; Lu, Z.; Roohani, I.; Li, Q.; Dunstan, C.R.; Jiang, X.; Zreiqat, H. Unraveling the Influence of Channel Size and Shape in 3D Printed Ceramic Scaffolds on Osteogenesis. Acta Biomater. 2024, 180, 115–127. [Google Scholar] [CrossRef]
- Hu, Z.; Lin, H.; Wang, Z.; Yi, Y.; Zou, S.; Liu, H.; Han, X.; Rong, X. 3D Printing Hierarchical Porous Nanofibrous Scaffold for Bone Regeneration. Small 2025, 21, 2405406. [Google Scholar] [CrossRef]
- Jung, H.-D. Titanium and Its Alloys for Biomedical Applications. Metals 2021, 11, 1945. [Google Scholar] [CrossRef]
- Castellani, C.; Lindtner, R.A.; Hausbrandt, P.; Tschegg, E.; Stanzl-Tschegg, S.E.; Zanoni, G.; Beck, S.; Weinberg, A.-M. Bone–Implant Interface Strength and Osseointegration: Biodegradable Magnesium Alloy versus Standard Titanium Control. Acta Biomater. 2011, 7, 432–440. [Google Scholar] [CrossRef]
- Pestel, M.; Alphonsus, J.; Toegel, S.; Strassl, A.; Herold, J.; Windhager, R.; Benca, E. Improving Accuracy in Assessing Osseointegration in Small Animal Bone Using Specimen-Specific Additively-Manufactured Fixtures Based on Clinical CT Imaging. J. Mech. Behav. Biomed. Mater. 2025, 165, 106941. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Li, G.; Ren, L.; Kong, X.; Wang, Y.; Han, X.; Jiang, W.; Dai, K.; Yang, K.; Hao, Y. Nano-Copper-Bearing Stainless Steel Promotes Fracture Healing by Accelerating the Callus Evolution Process. Int. J. Nanomed. 2017, 12, 8443–8457. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.; Ren, L.; Xie, K.; Wang, L.; Jiang, W.; Guo, Y.; Hao, Y. Functionalized TiCu/TiCuN Coating Promotes Osteoporotic Fracture Healing by Upregulating the Wnt/β-Catenin Pathway. Regen. Biomater. 2023, 10, rbac092. [Google Scholar] [CrossRef]
- Morgan, E.F.; Bayraktar, H.H.; Keaveny, T.M. Trabecular Bone Modulus–Density Relationships Depend on Anatomic Site. J. Biomech. 2003, 36, 897–904. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Wang, T.; Li, F.; Mao, X. Surface Modifications of Biomaterials in Different Applied Fields. RSC Adv. 2023, 13, 20495–20511. [Google Scholar] [CrossRef]
- Molina, N.; González, A.; Monopoli, D.; Mentado, B.; Becerra, J.; Santos-Ruiz, L.; Vida, Y.; Perez-Inestrosa, E. Dendritic Scaffold onto Titanium Implants. A Versatile Strategy Increasing Biocompatibility. Polymers 2020, 12, 770. [Google Scholar] [CrossRef]
- Long, S.; Zhu, J.; Jing, Y.; He, S.; Cheng, L.; Shi, Z. A Comprehensive Review of Surface Modification Techniques for Enhancing the Biocompatibility of 3D-Printed Titanium Implants. Coatings 2023, 13, 1917. [Google Scholar] [CrossRef]
- El-Bassyouni, G.T.; Mouneir, S.M.; El-Shamy, A.M. Advances in Surface Modifications of Titanium and Its Alloys: Implications for Biomedical and Pharmaceutical Applications. Multiscale Multidiscip. Model. Exp. Des. 2025, 8, 265. [Google Scholar] [CrossRef]
- Paxton, N.C.; Woodruff, M.A. Measuring Contact Angles on Hydrophilic Porous Scaffolds by Implementing a Novel Raised Platform Approach: A Technical Note. Polym. Adv. Technol. 2022, 33, 3759–3765. [Google Scholar] [CrossRef]
- Wu, Y.; Wan, K.; Lu, J.; Yuan, C.; Cui, Y.; Duan, R.; Yu, J. Research Progress on Surface Modification of Titanium Implants. Coatings 2025, 15, 229. [Google Scholar] [CrossRef]
- Xu, J.; Zhang, J.; Shi, Y.; Tang, J.; Huang, D.; Yan, M.; Dargusch, M.S. Surface Modification of Biomedical Ti and Ti Alloys: A Review on Current Advances. Materials 2022, 15, 1749. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; De Barros, N.R.; Zheng, T.; Gomez, A.; Doyle, M.; Zhu, J.; Nanda, H.S.; Li, X.; Khademhosseini, A.; Li, B. 3D Printing and Surface Engineering of Ti6Al4V Scaffolds for Enhanced Osseointegration in an in Vitro Study. Biomimetics 2024, 9, 423. [Google Scholar] [CrossRef] [PubMed]
- Mofazali, P.; Atapour, M.; Nakamura, M.; Sheikholeslam, M.; Galati, M.; Saboori, A. Surface Modification of Additive Manufactured Ti6Al4V Scaffolds with Gelatin/Alginate-IGF-1 Carrier: An Effective Approach for Healing Bone Defects. Int. J. Biol. Macromol. 2024, 265, 131125. [Google Scholar] [CrossRef]
- Gallo, J.; Holinka, M.; Moucha, C.S. Antibacterial Surface Treatment for Orthopaedic Implants. Int. J. Mol. Sci. 2014, 15, 13849–13880. [Google Scholar] [CrossRef]
- Wang, W.; Liu, H.; Guo, Z.; Hu, Z.; Wang, K.; Leng, Y.; Yuan, C.; Li, Z.; Ge, X. Various Antibacterial Strategies Utilizing Titanium Dioxide Nanotubes Prepared via Electrochemical Anodization Biofabrication Method. Biomimetics 2024, 9, 408. [Google Scholar] [CrossRef] [PubMed]
- Wilk, K.; Szewczenko, J. Research on the Influence of Anodic Oxidation Parameters on the Corrosion Resistance of Titanium Alloys. In Innovations in Biomedical Engineering; Gzik, M., Paszenda, Z., Piętka, E., Tkacz, E., Milewski, K., Jurkojć, J., Eds.; Lecture Notes in Networks and Systems; Springer International Publishing: Cham, Switzerland, 2023; Volume 409, pp. 225–232. ISBN 978-3-030-99111-1. [Google Scholar]
- Poquillon, D.; Parrens, C.; Pugliara, A.; Perrais, M.; Malard, B. Oxidation of Ti–6Al–4V Alloy between 450 and 600 °C. Evolution of Microstructure and Mechanical Properties. MATEC Web Conf. 2020, 321, 06009. [Google Scholar] [CrossRef]
- Casadebaigt, A.; Monceau, D.; Hugues, J. High Temperature Oxidation of Ti-6Al-4V Alloy Fabricated by Additive Manufacturing. Influence on Mechanical Properties. In Proceedings of the 14th World Conference on Titanium (Ti 2019), Nantes, France, 10–14 June 2019; EDP Sciences: Les Ulis, France, 2020; Volume 321, p. 03006. [Google Scholar]
- Gu, Y.; Liu, Y.; Jacobs, R.; Wei, L.; Sun, Y.; Tian, L.; Liu, Y.; Politis, C. BMP-2 Incorporated into a Biomimetic Coating on 3D-Printed Titanium Scaffold Promotes Mandibular Bicortical Bone Formation in a Beagle Dog Model. Mater. Des. 2023, 228, 111849. [Google Scholar] [CrossRef]
- Zhang, B.; Li, J.; He, L.; Huang, H.; Weng, J. Bio-Surface Coated Titanium Scaffolds with Cancellous Bone-like Biomimetic Structure for Enhanced Bone Tissue Regeneration. Acta Biomater. 2020, 114, 431–448. [Google Scholar] [CrossRef]
- Woźniak, A.; Smok, W.; Szewczenko, J.; Staszuk, M.; Chladek, G. Influence of Hybrid Surface Modification on Biocompatibility and Physicochemical Properties of Ti-6Al-4V ELI Titanium. J. Funct. Biomater. 2024, 15, 52. [Google Scholar] [CrossRef] [PubMed]
- Laubach, M.; Hildebrand, F.; Suresh, S.; Wagels, M.; Kobbe, P.; Gilbert, F.; Kneser, U.; Holzapfel, B.M.; Hutmacher, D.W. The Concept of Scaffold-Guided Bone Regeneration for the Treatment of Long Bone Defects: Current Clinical Application and Future Perspective. J. Funct. Biomater. 2023, 14, 341. [Google Scholar] [CrossRef]
- Popov, V.V.; Muller-Kamskii, G.; Kovalevsky, A.; Dzhenzhera, G.; Strokin, E.; Kolomiets, A.; Ramon, J. Design and 3D-Printing of Titanium Bone Implants: Brief Review of Approach and Clinical Cases. Biomed. Eng. Lett. 2018, 8, 337–344. [Google Scholar] [CrossRef]
- Băbțan, A.-M.; Timuș, D.; Sorițău, O.; Boșca, B.A.; Barabas, R.; Ionel, A.; Petrescu, N.B.; Feurdean, C.N.; Bordea, I.R.; Saraci, G. Tissue Integration and Biological Cellular Response of SLM-Manufactured Titanium Scaffolds. Metals 2020, 10, 1192. [Google Scholar] [CrossRef]
- Chen, Y.; Frith, J.E.; Dehghan-Manshadi, A.; Kent, D.; Bermingham, M.; Dargusch, M. Biocompatible Porous Titanium Scaffolds Produced Using a Novel Space Holder Technique. J. Biomed. Mater. Res. B Appl. Biomater. 2018, 106, 2796–2806. [Google Scholar] [CrossRef]
- Visai, L. Topological, Mechanical and Biological Properties of Ti6Al4V Scaffolds for Bone Tissue Regeneration Fabricated with Reused Powders via Electron Beam Melting. Materials 2021, 14, 224. [Google Scholar]
- Wysocki, B.; Idaszek, J.; Szlązak, K.; Strzelczyk, K.; Brynk, T.; Kurzydłowski, K.J.; Święszkowski, W. Post Processing and Biological Evaluation of the Titanium Scaffolds for Bone Tissue Engineering. Materials 2016, 9, 197. [Google Scholar] [CrossRef]
- Lee, S.S.; Du, X.; Smit, T.; Bissacco, E.G.; Seiler, D.; de Wild, M.; Ferguson, S.J. 3D-Printed LEGO®-Inspired Titanium Scaffolds for Patient-Specific Regenerative Medicine. Biomater. Adv. 2023, 154, 213617. [Google Scholar] [CrossRef]
- Liu, Z.; Xu, Z.; Wang, X.; Zhang, Y.; Wu, Y.; Jiang, D.; Jia, R. Construction and Osteogenic Effects of 3D-Printed Porous Titanium Alloy Loaded with VEGF/BMP-2 Shell-Core Microspheres in a Sustained-Release System. Front. Bioeng. Biotechnol. 2022, 10, 1028278. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Wu, J.; Xie, K.; Tan, J.; Yang, Y.; Zhao, S.; Wang, L.; Jiang, W.; Hao, Y. Study of Bone Regeneration and Osteointegration Effect of a Novel Selective Laser-Melted Titanium-Tantalum-Niobium-Zirconium Alloy Scaffold. ACS Biomater. Sci. Eng. 2019, 5, 6463–6473. [Google Scholar] [CrossRef] [PubMed]
- Kelly, C.N.; Wang, T.; Crowley, J.; Wills, D.; Pelletier, M.H.; Westrick, E.R.; Adams, S.B.; Gall, K.; Walsh, W.R. High-Strength, Porous Additively Manufactured Implants with Optimized Mechanical Osseointegration. Biomaterials 2021, 279, 121206. [Google Scholar] [CrossRef]
- Martinez-Marquez, D.; Delmar, Y.; Sun, S.; Stewart, R.A. Exploring Macroporosity of Additively Manufactured Titanium Met amaterials for Bone Regeneration with Quality by Design: A Systematic Literature Review. Materials 2020, 13, 4794. [Google Scholar] [CrossRef]
- Safavi, S.; Yu, Y.; Robinson, D.L.; Gray, H.A.; Ackland, D.C.; Lee, P.V.S. Additively Manufactured Controlled Porous Orthopedic Joint Replacement Designs to Reduce Bone Stress Shielding: A Systematic Review. J. Orthop. Surg. 2023, 18, 42. [Google Scholar] [CrossRef] [PubMed]
- Toosi, S.; Javid-Naderi, M.J.; Tamayol, A.; Ebrahimzadeh, M.H.; Yaghoubian, S.; Mousavi Shaegh, S.A. Additively Manufactured Porous Scaffolds by Design for Treatment of Bone Defects. Front. Bio. Biotechnol. 2024, 11, 1252636. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Sun, N.; Zhu, M.; Qiu, Q.; Zhao, P.; Zheng, C.; Bai, Q.; Zeng, Q.; Lu, T. The Contribution of Pore Size and Porosity of 3D Printed Porous Titanium Scaffolds to Osteogenesis. Mater. Sci. Eng. C 2022, 133, 112651. [Google Scholar] [CrossRef]
- Rao, X.; Yang, J.; Feng, X.; Chen, Z.; Yuan, Y.; Yong, B.; Chu, C.; Tan, X.; Song, Q. Replication and Bioactivation of Ti-Based Alloy Scaffold Macroscopically Identical to Cancellous Bone from Polymeric Template with TiNbZr Powders. J. Mech. Behav. Biomed. Mater. 2018, 88, 296–304. [Google Scholar] [CrossRef]
- Wang, X.; Li, Y.; Hodgson, P.D.; Wen, C. Biomimetic Modification of Porous TiNbZr Alloy Scaffold for Bone Tissue Engineering. Tissue Eng. Part A 2010, 16, 309–316. [Google Scholar] [CrossRef]
- Moshokoa, N.A.; Raganya, M.L.; Machaka, R.; Obadele, B.A.; Mamookho, M.E. Microstructure and Bending Properties of Solution-Treated Ti-Mo Binary Alloys for Biomedical Applications. MATEC Web Conf. 2022, 370, 03014. [Google Scholar] [CrossRef]
- Romero-Resendiz, L.; Rossi, M.C.; Seguí-Esquembre, C.; Amigó-Borrás, V. Development and Characterization of a New Predominantly β Ti–15Mo–5In Alloy for Biomedical Applications. J. Mater. Sci. 2023, 58, 15828–15844. [Google Scholar] [CrossRef]
- Gouvea, E.d.S.; Rossi, M.C.; Escuder, A.V.; Afonso, C.R.M.; Borras, V.A. Relation between Mechanical Milling Parameters in Phase Transformation and Oxygen Content in Ti–Nb–Mo Powders for Posterior Sintering. Metals 2022, 12, 1238. [Google Scholar] [CrossRef]
- Yue, Y.; Qi, M.; Song, T.; Chen, B.; Tang, Y.; Xia, C. Enhanced Mechanical and Corrosion Properties via Annealing Treatment on the Hot-Rolled Ti-Zr-Mo Alloy. Materials 2023, 16, 2597. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wang, C.; Zhang, S.; Yu, X.; Zhou, G.; Zhang, Z.; Chen, L. Effect of Secondary α Phase on Stress Corrosion Cracking of a Novel Metastable β Titanium Alloy in 3.5% NaCl Solution. Crystals 2022, 12, 1849. [Google Scholar] [CrossRef]
- Zhang, H.; Sun, J.; Zhou, G.; Yu, X.; Wang, C.; Gao, J. Influence of Phase Composition on Stress-Corrosion Cracking of Ti-6Mo-5V-3Al-2Fe-2Zr Alloy in 3.5% NaCl Solution. Crystals 2022, 12, 1794. [Google Scholar] [CrossRef]
- Zeng, G.; Hu, B.; Shi, Y.; Lan, X.; Yin, H. Corrosion Resistance Investigation of the Ti–Al–Mo System Supported by CALPHAD Approach and Key Experiments. Corros. Sci. 2024, 229, 111857. [Google Scholar] [CrossRef]
- Wang, X.; Pan, Y.; Yang, J.; Zhu, R.; Zhou, Y.; Yuan, Z.; Chu, H.; Hu, P.; Li, L. Corrosion Behavior of Ti-0.3 Mo-0.8 Ni (TA10) Alloy in Proton Exchange Membrane Fuel Cell Environment: Experimental and Theoretical Studies. Int. J. Electrochem. Sci. 2023, 18, 100239. [Google Scholar] [CrossRef]
- Hong, S.H.; Park, S.W.; Park, C.H.; Yeom, J.-T.; Kim, K.B. Relationship between Phase Stability and Mechanical Properties on near/Metastable β-Type Ti–Cr-(Mn) Cast Alloys. J. Alloys Compd. 2020, 821, 153516. [Google Scholar] [CrossRef]
- Li, D.; Chen, Y.; Li, R.; Chen, Z.; Qian, K.; Zou, Z.; Li, W.; Chen, Q. Preparation of Biomimetic MAO Coating on β-Titanium Alloy Surface and Its Bone Regeneration Property in Diabetic Environment. Mater. Des. 2024, 239, 112808. [Google Scholar] [CrossRef]
- Lekszycki, T.; dell’Isola, F. A Mixture Model with Evolving Mass Densities for Describing Synthesis and Resorption Phenomena in Bones Reconstructed with Bio-resorbable Materials. ZAMM-J. Appl. Math. Mech. Z. Angew. Math. Mech. 2012, 92, 426–444. [Google Scholar] [CrossRef]
- Allena, R.; Scerrato, D.; Bersani, A.; Giorgio, I. Simulating Bone Healing with Bio-Resorbable Scaffolds in a Three-Dimensional System: Insights into Graft Resorption and Integration. C. R. Méc. 2025, 353, 479–497. [Google Scholar] [CrossRef]
- ASTM F136; Standard Specification for Wrought Titanium-6Aluminum-4Vanadium ELI Alloy for Surgical Implant Applications. ASTM International: West Conshohocken, PA, USA, 2013.
- ASTM F2924; Standard Specification for Additive Manufacturing Titanium-6 Aluminum-4 Vanadium with Powder Bed Fusion. ASTM International: West Conshohocken, PA, USA, 2014.
- ASTM F3302; Standard Specification for Additive Manufacturing Finished Part Properties. ASTM International: West Conshohocken, PA, USA, 2018.
- ISO 5832; Implants for Surgery Metallic Materials Part 3: Wrought Titanium 6 Aluminum 4 Vanadium Alloy. ISO: Geneva, Switzerland, 2016.
- ISO 52900; Additive Manufacturing General Principles Terminology. ISO: Geneva, Switzerland, 2015.
- Martinez-Marquez, D.; Jokymaityte, M.; Mirnajafizadeh, A.; Carty, C.P.; Lloyd, D.; Stewart, R.A. Development of 18 Quality Control Gates for Additive Manufacturing of Error Free Patient-Specific Implants. Materials 2019, 12, 3110. [Google Scholar] [CrossRef] [PubMed]
- Sidambe, A.T. Biocompatibility of Advanced Manufactured Titanium Implants—A Review. Materials 2014, 7, 8168–8188. [Google Scholar] [CrossRef] [PubMed]



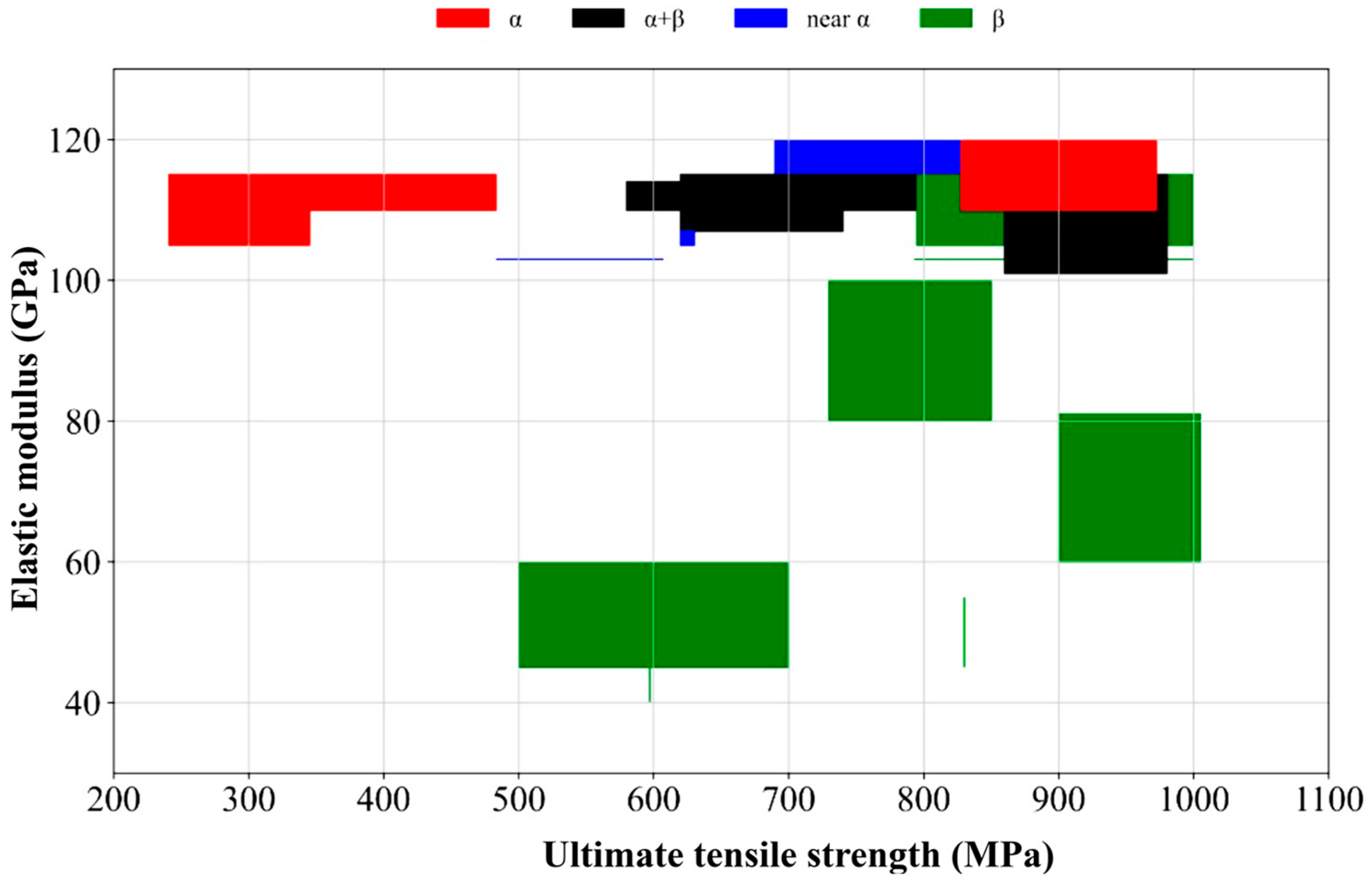









| S/No. | Grade | Name | Structure Type | Young’s Modulus (GPa) | Tensile Strength (MPa) | Fatigue Strength (MPa) | Hardness (HV) | Elongation (%) |
|---|---|---|---|---|---|---|---|---|
| CP-Ti | ||||||||
| 1 | Grade 1 | Titanium CP1 | alpha (α) | 103 | 240 | 160 | 120 | 24–28 |
| 2 | Grade 2 | Titanium CP2 | alpha (α) | 103 | 345 | 240 | 160 | 20 |
| 3 | Garde 3 | Titanium CP3 | alpha (α) | 103 | 450 | 310 | 200 | 18 |
| 4 | Grade 4 | Titanium CP4 | alpha (α) | 103 | 550 | 340 | 250 | 15 |
| aluminium vanadium titanium alloy | ||||||||
| 5 | Grade 5 | Ti-6Al-4V | α + β-type | 113–120 | 895–965 | 510–560 | 330–370 | 10–14 |
| 6 | Grade 8 | Ti-7.35Al-1Mo-1V | Near α | 110–120 | 690–830 | 550–620 | 300–350 | 10–15 |
| 7 | Grade 9 | Ti-3Al-2.5V | α + β-type | 110–115 | 620–900 | 400–480 | 300–360 | 10–15 |
| 8 | Grade 18 | Ti-3Al-2.5V-0.05Pd | α + β-type | 107–110 | 620–740 | 450–500 | 300–350 | 15–17 |
| 9 | Grade 19 | Ti-8V-6Cr-4Mo-4Zr-3Al | β-type | 103 | 793–1000 | 500–650 | 330–390 | 8–12 |
| 10 | Grade 20 | Ti-8V-6Cr-4Zr-4Mo-3Al-0.06Pd | 105–115 | 795–100 | 340–400 | 8 | ||
| 11 | Grade 23 | Ti-6Al-4 V ELI | α + β-type | 101–110 | 860–980 | 510–620 | 320–260 | 10 |
| 12 | Grade 24 | Ti-6Al-4V-0.06Pd | α + β-type | 110–115 | 860–980 | 10–14 | ||
| 13 | Grade 25 | Ti-6Al-4V-0.5Ni-0.06Pd | α + β-type | 105–115 | 860–980 | 10–15 | ||
| 14 | Grade 28 | Ti-3Al-2.5V-0.5Ru | near α-type | 105–107 | 620–630 | 290–340 | 10–18 | |
| 15 | Grade 29 | Ti-6AL-4V-0.1Ru ELI | α + β-type | 110 | 890 | 320–370 | 16 | |
| Others, titanium alloy | ||||||||
| 16 | Grade 6 | Ti-5Al-2.5Sn | α-type | 110–120 | 828–972 | 290–350 | 290 | 10–16 |
| 17 | Grade 7 | Ti-0.15Pd | α-type | 110 | 485 | 250–330 | 12–20 | |
| 18 | Grade 10 | Ti-11.5Mo-6Zr-4.5Sn | β-type | 80–100 | 730–850 | 380–400 | 330–380 | 13 |
| 19 | Grade 11 | Ti-0.15Pd | α-type | 103 | 345 | 250–330 | 20–37 | |
| 20 | Grade 12 | Ti-0.3Mo-0.8Ni | near α-type | 103 | 483–607 | 280–350 | 18–22 | |
| 21 | Grade 13 | Ti-0.5Ni-0.05Ru | α-type | 110–120 | 275 | 20–24 | ||
| 22 | Grade 14 | Ti-0.5Ni-0.05Ru | α-type | 105 | 410 | 12–20 | ||
| 22 | Grade 15 | Ti-0.5Ni-0.05Ru | α + β type | 110 | 484 | 19 | ||
| 23 | Grade 16 | Ti-0.06Pd | α-type | 115–110 | 345–483 | 160–230 | 20–30 | |
| 24 | Grade 17 | Ti-0.06Pd | α-type | 105–115 | 241–345 | 160–230 | 24–37 | |
| 25 | Grade 21 | Ti-15Mo-3Nb-3Al-0.2Si | β-type | 90–100 | 793 | 280–350 | 15 | |
| 26 | Grade 26 | Ti-0.1Ru | α-type | 105–115 | 345 | 20 | ||
| 27 | Grade 27 | Ti-0.1Ru | α-type | 105–115 | 300 | 20–30 | ||
| 28 | Grade 36 | 55Ti-45Nb | β-type | 45–60 | 500–700 | 22 | ||
| Non-grade biomedical Ti-alloys | ||||||||
| 29 | Ti-6Al-7Nb | α + β-type | 110–114 | 580–710 | 300–350 | 8.1–15 | ||
| 30 | Ti-13Nb-13Zr | β-type | 60–81 | 900–1005 | 490–550 | 12–13 | ||
| 31 | Ti-35Nb-7Zr-5Ta | β-type | 40–55 | 597 | 260–300 | 19–20 | ||
| 32 | Ti-29Nb-13Ta-4.6Zr | β-type | 60–80 | 912 | 13 | |||
| 33 | Ti-24Nb-4Zr-7.9Sn | β-type | 45–55 | 830 | 12–15 | |||
| 34 | Ti-15Mo | β-type | 78 | 874 | 21 | |||
| 36 | Ti-15Mo-2.8Nb-0.2Si | β-type | 83 | 990 | 16–18 | |||
| 37 | Ti-16Nb-10Hf | β-type | 81 | 852 | 11 | |||
| 38 | Ti–25Pd–5Cr | β-type | 110–120 | 880 | 5 | |||
| Titanium Alloy | Microstructure | Category of Use | Uses | Source |
|---|---|---|---|---|
| Pure Titanium, | α | Orthopedic implant | Parts of the joint (stems, cups, etc.), meshes, artificial bones, and fixation instruments | [56,104,105,106,107,108,109,110,111,112] |
| Ti6Al4V | α + β | |||
| Ti6Al7Nb | α + β | |||
| Ti13Nb13Zr | β | |||
| Ti15Mo | β | |||
| Pure titanium | α | Cardiovascular devices | Medical supplies such as ventricular assist devices, implantable defibrillators, clips, guidewires, catheters, and heart valves. | [113,114,115,116,117,118,119,120,121,122] |
| Ti15Mo | β | |||
| Ti6Al4V | α + β | |||
| Ti6Al7N | α + β | |||
| Pure titanium | α | Spinal implants | Fixing devices, discs, and cages | [123,124,125,126,127,128,129,130] |
| Ti6Al4V | α + β | |||
| Pure titanium | α | Trauma devices | Fixing plates, screws, and rods | [69,131,132,133,134] |
| Ti6Al7Nb | α + β | |||
| Ti6Al4V | α + β | |||
| Pure titanium | α | Soft tissue implant | Fixation apparatus, hernia devices, breast reconstruction prosthesis | [135,136,137,138,139,140] |
| Ti6Al7Nb | α + β | |||
| Ti6Al4V | α + β |
| Author, Date | Particle Size | Ti-Alloy Used | Vivo/Vitro Test | S-Manufacturing | Effectiveness After Implantation/of the Article | Positive Innovation |
|---|---|---|---|---|---|---|
| Tilton et al., 2021 [150] | _ | Ti-6Al-4V (Grade 5) spherical powder | Vitro | EBM | The patient-specific AM prosthesis showed enough biomechanical strength to avoid. | Patient-specific AM. prostheses could effectively restore substantial bone defects following tumor excision. |
| Antounian et al., 2024 [151] | 15–45 μm | Gas-atomized Ti-6Al-4V Grade 23 | Vitro and vivo | SLM | The implant was successfully integrated with the bone, and after 14 months, an X-ray was taken. | The patient’s functional status showed improvement, and limb shortening was efficiently minimized. |
| Hindy et al., 2020 [152] | 5–50 μm | spherical Ti-6Al-4V powder. | Vitro | SLM | Demonstrate the feasibility of using 3D printed functionally | Functionally graded samples with dense cores had a good match of Young’s |
| Deng et al., 2021 [144] | 15–45 µm | Ti6-Al-4V | Vivo | SLM | After the removal of the scaffold from the implant, it was found that a greater. | Uses 3D printing technology to produce functionally graded porous titanium alloys, which increases. |
| Wang et al., 2021 [145] | 20–50 µm | porous Ti-6Al-4V | vitro and vivo | EBM | Ti6Al4V scaffolds for vascularized bone regeneration were tested for biocompatibility and bone ingrowth | generating better vascularization and osseointegration for better bone regeneration in orthopedic applications |
| Gryko et al., 2022 [149] | _ | Ti-6Al-4V porosity and pore geometry | Focuses on FEA | _ | FEA simulates the mechanical properties of different pores | Improve scaffold design by showing how computational modeling may optimize |
| Xu et al., 2022 [146] | _ | Ti-6Al-4V powder | Vivo and Vitro | SLM | In vitro and in vivo investigations have shown that 3D-printed Ti6Al4V scaffolds enhance osteoblast activity, bone regeneration, and osseointegration. | Using VEFG/BMP 2 microspheres for sequential growth factor release with a 3D-printed porous titanium alloy scaffold to improve bone. |
| Sun et al., 2022 [153] | _ | Ti-6Al-4V powder | Vivo | LPBF | In vivo tests and FEM show that additive-manufactured Ti6Al4V | show that cortical bone ingrowth causes significant mechanical changes in additive-manufactured Ti6Al4V scaffolds, offering a predictive basis for optimizing porous implant designs. |
| Zhang et al., 2021 [154] | 36 µm | Ti-6Al-4V powder | Vivo and Vitro | SLM | Bioactive glass and mesoporous bioactive glass-coated 3D-printed Ti-6Al-4V structures facilitate bone regeneration by promoting cell growth, bone conduction, and angiogenesis. | Mesoporous bioactive glass coatings on 3D-printed Ti-6Al-4V scaffolds enhance bone regeneration by promoting both osteogenesis and angiogenesis through careful structural design. |
| Chen et al., 2019 [147] | 30 μm | Ti-6Al-4V alloy powder | Vivo and Vitro | SLM | uses 3D-printed Ti6Al4V porous cages to show that surface modifications increase cellular behavior in vitro and bone ingrowth in vivo. | Adjusting additive angles promotes biocompatibility, osseointegration, and bone structure in 3D-printed porous Ti6Al4V scaffolds. |
| Zhong et.al., 2020 [155] | _ | Ti-6Al-4V | Vivo and vitro | SLM | Polydopamine-coated 3D-printed Ti-6Al-4V implants enhance cell adhesion, osteogenesis, bone regeneration, and osteointegration in vitro and in vivo. | A polydopamine coating on 3D-printed Ti-6Al-4V implants improves biocompatibility and osteointegration. |
| Guo et al., 2020 [143] | _ | Ti-6Al-4V | Vivo and Vitro | SLM | The success of TiCu/Ti-Cu-N-coated 3D-printed Ti6Al4V scaffolds in recruiting BMSCs for bone regeneration. | TiCu/Ti-Cu-N-coated 3D-printed Ti6Al4V scaffolds were developed to recruit BMSCs and promote osteogenic differentiation for bone regeneration. |
| Ma et al., 2021 [156] | _ | Ti-6Al-4V alloy and gelatin | Vivo and Vitro | SLM | In orthopaedic applications, this hybrid scaffold may improve bone defect repair and tissue regeneration. | Biomimetic hybrid scaffold combining Ti-6Al-4V’s mechanical strength with GelMA’s biocompatibility and bioactivity. |
| Crovace et al., 2020 [157] | 45–70 µm | Ti-6Al-4V | Vivo and vitro | EBM | Over one year, sheep models via EBM-sintered Ti6Al4V scaffolds showed better healing, defect restoration, and mechanical stability. | The scaffolds are intended to match bone mechanical properties, increasing biomechanical stability and healing. |
| Li et al., 2019 [158] | _ | Ti-6Al-4V | Vivo and Vitro | SLM | Enhanced scaffold porosity and mechanics improve osseointegration and early bone healing over traditional designs. | TPMS-designed Ti6Al4V scaffolds enhance porosity and mechanics, promoting osteointegration and bone regeneration. |
| Fan et al., 2020 [159] | _ | Ti-6Al-4V | Vivo and Vitro | SLM | Successful bone regeneration and osseointegration were found in animal models. | Electroactive BaTiO3-coated Ti6Al4V scaffolds with LIPUS stimulation enhance osteogenesis and osseointegration. |
| Ragone et al., 2020 [160] | _ | Ti-6Al-4V (Grade 5) | Vivo and Vitro | SLM | In animal models, the scaffold enhances bone ingrowth and integration, thereby supporting effective repair of bone defects. | AM-fabricated randomized trabecular titanium scaffolds enhance osseointegration and bone healing. |
| Chen et al., 2020 [148] | 22–51 μm | Ti-6Al-4V ELI, (Grade 23) | Vivo and vitro | SLM | Improved osteogenesis and bone ingrowth in Ti6Al4V ELI scaffolds with customized pore sizes and porosity. | Optimized pore size and porosity of Ti6Al4V ELI scaffolds via SLM improve scaffold performance. |
| Liu et al., 2020 [161] | 15–45 μm | Ti-6Al-4V | Vivo and vitro | SLM | 3D-printed Ti-6Al-4V scaffolds enable bone ingrowth and osseointegration, with mechanical stimulation boosting bone formation. | 3D-printed Ti-6Al-4V scaffolds with tailored mechanics enhance bone ingrowth and osseointegration. |
| Li et al., 2019 [162] | _ | Ti-6Al-4V | Vivo and vitro | SLM | PDA-coated 3D-printed Ti-6Al-4V scaffolds improve osteogenesis, cell adhesion, and osseointegration in vitro and in vivo | Polydopamine (PDA) coating on 3D-printed Ti-6Al-4V scaffolds boosts osteogenesis, cell adhesion, and bone regeneration. |
| Luan et al., 2019 [163] | _ | Ti-6Al-4V | Vivo and vitro | EBM | Pore size and porosity in Ti-6Al-4V scaffolds enhance cell proliferation, osteogenesis, bone regeneration, and osseointegration. | Optimizing the pore size and porosity of Ti-6Al-4V scaffolds enhances bone regeneration and osteogenesis. |
| Yu et al., 2023 [164] | _ | Ti-6Al-4V | vitro | SLM | Titanium scaffold with various kinds of shapes for improved cell growth and mechanical strength | Developed functionally graded titanium scaffolds with tailored porosity for mechanical strength and biological integration. |
| Study | Powder Size | Laser Power | Scan Speed | Layer Thickness | Powder Size Reported (Y/N) | Laser Power Reported (Y/N) | Scan Speed Reported (Y/N) | Layer Thickness Reported (Y/N) |
|---|---|---|---|---|---|---|---|---|
| Antounian et al. [151] | 15–45 µm | 80 W | 900 mm/s | 25 µm | Y | Y | Y | Y |
| Hindy et al. [152] | 5–50 µm | 175 W | 2000 mm/s | 30 µm | Y | Y | Y | Y |
| Wang et al. [145] | 45–100 μm | 720 W | 800, 900, and 1000 μm, | 30 µm | Y | Y | Y | Y |
| Deng et al. [144] | 15–45 µm | 500 W | 300 mm/s | 30 µm | Y | Y | Y | Y |
| Zhang et al. [154] | 36 μm | 400 W | 300 mm/s | 30 µm | Y | Y | Y | Y |
| Chen et al. [147] | 30 μm | - | - | - | Y | N | N | N |
| Crovac et al. [157] | 45–70 µm | - | - | 70 µm | Y | N | N | Y |
| Chen et al. [148] | 22–51 μm | 240 W | 240 mm/s | 30 µm | Y | Y | Y | Y |
| Liu et al. [161] | 15–45 µm | 180 W | 1250 mm/s | - | Y | Y | Y | N |
| Specimens | Compressive Strength (MPa) |
|---|---|
| Control | 149.7 ± 4.9 |
| Ti-NaOH | 150.5 ± 6.7 |
| Ti-DOPA | 155.8 ± 7.1 |
| Ti-NaOH + DOPA | 141.8 ± 4.6 |
| Alloy System | Major Alloying Element Composition wt (%) | B Phase Stabilizing Strength (wt) | Beta Phase Stability | Corrosion Potential (V vs. SCE) | Reference |
|---|---|---|---|---|---|
| Ti10Mo | Mo: 10.0 | 10 | Metastable β + α″ | −0.15 to −0.10 | Moshokoa et al. [223] |
| Ti-15Mo-5In | Mo: 15.0, In: 5.0 | 16.6 | Predominantly β + α + α″ | −0.10 to −0.06 | Romero-Resendiz et al. [224] |
| Ti-35Nb-6Mo | Nb: 35.0, Mo: 6.0 | 15.7 | β phase (increases with processing) | −0.08 to −0.03 | Gouvêa et al. [225] |
| Ti-20Zr-15Mo | Zr: 20.0, Mo: 15.0 | 16.8 | Single β (after 800 °C anneal) | −0.06 to −0.02 | Yue et al. [226] |
| Ti-6Mo-5V-3Al-2Fe | Mo: 6.0, V: 5.0, Al: 3.0, Fe: 2.0 | 11.8 | Metastable β + secondary α | −0.10 to −0.05 | Zhang et al. [227] |
| Ti-6Mo-5V-3Al-2Fe-2Zr | Mo: 6.0, V: 5.0, Al: 3.0, Fe: 2.0, Zr: 2.0 | 12.8 | β + variable α | −0.08 to −0.04 | Zhang et al. [228] |
| Ti55Al40Mo5 | Al: 40.0, Mo: 5.0 | 6 | bcc(Ti) single phase | −0.05 to 0.00 | Zeng et al. [229] |
| Ti-0.3Mo-0.8Ni (TA10) | Mo: 0.3, Ni: 0.8 | 1 | α phase dominant | −0.35 to −0.30 | Wang et al. [230] |
| Ti-Cr-Mn | Cr: 5–8, Mn: 2–4 | 8–12 | Near/metastable β + α″ + ω | −0.20 to −0.12 | Hong et al. [231] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fita, S.W.; Bonek, M.; Woźniak, A.; Sławski, S. Advances in Titanium-Based Biomaterial for Human Bone Scaffolds: Narrative Review on Design, Fabrication, Surface Engineering, Implantation, and Biological Evaluation. Materials 2025, 18, 5421. https://doi.org/10.3390/ma18235421
Fita SW, Bonek M, Woźniak A, Sławski S. Advances in Titanium-Based Biomaterial for Human Bone Scaffolds: Narrative Review on Design, Fabrication, Surface Engineering, Implantation, and Biological Evaluation. Materials. 2025; 18(23):5421. https://doi.org/10.3390/ma18235421
Chicago/Turabian StyleFita, Sichale W., Mirosław Bonek, Anna Woźniak, and Sebastian Sławski. 2025. "Advances in Titanium-Based Biomaterial for Human Bone Scaffolds: Narrative Review on Design, Fabrication, Surface Engineering, Implantation, and Biological Evaluation" Materials 18, no. 23: 5421. https://doi.org/10.3390/ma18235421
APA StyleFita, S. W., Bonek, M., Woźniak, A., & Sławski, S. (2025). Advances in Titanium-Based Biomaterial for Human Bone Scaffolds: Narrative Review on Design, Fabrication, Surface Engineering, Implantation, and Biological Evaluation. Materials, 18(23), 5421. https://doi.org/10.3390/ma18235421








