Effects of Two Dental Implant Micromotor Systems for Dental Implant Placement on Implant Stability and Removal Torque: An Animal Experiment
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Preparation
2.2. Evaluation of Implant Stability Quotient
2.3. Evaluation of the Bone–Implant Interface Gap by Micro-CT 3D Deviation
2.4. Evaluation of Removal Torque of Placed Implants
2.5. Statistical Analysis
3. Results
3.1. Implant Stability
3.2. Removal Torque
3.3. Bone–Implant Interface Gap (Micro-CT 3D Deviation)
3.4. Equivalence Testing, Variance Components, and Ancillary Association
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Raffat, E.M.; Shady, M.; Elkashty, A.A.R.; Syad, M.E. Comparative analysis of implant survival, peri-implant health, and patient satisfaction among three treatment modalities in atrophic posterior mandibles: A randomized clinical study. BMC Oral Health 2025, 25, 939. [Google Scholar] [CrossRef]
- Son, K.; Son, Y.T.; Kim, J.Y.; Lee, J.M.; Yu, W.J.; Kim, J.W.; Lee, K.B. Effect of repeated use of an implant handpiece on an output torque: An in-vitro study. J. Adv. Prosthodont. 2021, 13, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Haas, O.L., Jr.; Scolari, N.; Meirelles, L.D.S.; Becker, O.E.; Melo, M.F.S.; Viegas, V.N.; de Oliveira, R.B. Intraoral technique for locking reconstruction plate fixation using an implant handpiece with adapted drills. Head Neck 2016, 38, 1436–1439. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Son, K.; Son, Y.T.; Kim, Y.G.; Suh, J.Y.; Lee, K.B.; Lee, J.M. Comparison of osseointegration of dental implants placed in rabbit tibia using two dental laser and implant handpiece systems. Bioengineering 2022, 9, 681. [Google Scholar] [CrossRef] [PubMed]
- Becker, O.E.; Avelar, R.L.; do Nascimento Dolzan, A.; Göelzer, J.G.; Haas, O.L., Jr.; de Oliveira, R.B. Implant handpiece with adapted drills in orthognathic surgery: Preventing facial scars. J. Craniofac. Surg. 2012, 23, e529–e530. [Google Scholar] [CrossRef]
- Siegmund, B.J.; Winter, K.; Meyer-Marcotty, P.; Rustemeyer, J. Reconstruction of the temporomandibular joint: A comparison between prefabricated and customized alloplastic prosthetic total-joint systems. Int. J. Oral Maxillofac. Surg. 2019, 48, 1066–1071. [Google Scholar] [CrossRef]
- Farré-Pagès, N.; Augé-Castro, M.L.; Alaejos-Algarra, F.; Mareque-Bueno, J.; Ferrés-Padró, E.; Hernández-Alfaro, F. A novel trephine design for sinus-lift lateral approach: Case report. Med. Oral Patol. Oral Cir. Bucal 2011, 16, e79–e82. [Google Scholar] [CrossRef]
- Dastouri, E.; Heck, T.Y.; Wang, H.L. Accidental ingestion of a dental object during a periodontal surgical procedure on an IV-sedated patient: Case management. Clin. Adv. Periodontics 2023, 13, 156–162. [Google Scholar] [CrossRef]
- Duarte, N.D.; Mulinari-Santos, G.; Batista, F.R.D.S.; Gomes, M.B.; Monteiro, N.G.; Silva, A.C.E.D.; Gruber, R.; Lisboa-Filho, P.N.; Gomes-Ferreira, P.H.S.; Okamoto, R. Sonification of deproteinized bovine bone functionalized with genistein enhances bone repair in peri-implant bone defects in ovariectomized rats. J. Funct. Biomater. 2024, 15, 328. [Google Scholar] [CrossRef]
- Elias, C.N.; Oshida, Y.; Lima, J.H.C.; Muller, C.A. Relationship between surface properties (roughness, wettability and morphology) of titanium and dental implant removal torque. J. Mech. Behav. Biomed. Mater. 2008, 1, 234–242. [Google Scholar] [CrossRef]
- Arshad, M.; Mazidi, Y.; Chiniforush, N.; Shirvani, A.; Bassir, S.H. The effect of photobiomodulation therapy on implant stability: A systematic review and meta-analysis. Photochem. Photobiol. 2025, 101, 709–727. [Google Scholar] [CrossRef] [PubMed]
- Shenoy, A.; Ganapathy, D.; Maiti, S. Effectiveness of photobiomodulation with low-level laser therapy on the implant stability quotient at different time intervals: A randomized clinical trial. Cureus 2024, 16, e54577. [Google Scholar] [CrossRef] [PubMed]
- Nakanishi, Y.; Wang, P.L.; Ochi, M.; Nakanishi, K.; Matsubara, H. Low-intensity pulsed ultrasound stimulation significantly enhances promotion of bone formation around dental implants. J. Hard Tissue Biol. 2011, 20, 139–146. [Google Scholar] [CrossRef][Green Version]
- Xailani, K.K.; Hamad, S.A. Effect of photobiomodulation therapy of overprepared dental implant bed on torque removal and implant stability quotient: An experimental study in sheep. Minerva Dent. Oral Sci. 2023, 72, 96–101. [Google Scholar] [CrossRef] [PubMed]
- Bardyn, T.; Gédet, P.; Hallermann, W.; Büchler, P. Prediction of dental implant torque with a fast and automatic finite-element analysis: A pilot study. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2010, 109, 594–603. [Google Scholar] [CrossRef]
- Fernandes, D.J.; Marques, R.G.; Elias, C.N. Influence of acid treatment on surface properties and in vivo performance of Ti6Al4V alloy for biomedical applications. J. Mater. Sci. Mater. Med. 2017, 28, 164. [Google Scholar] [CrossRef]
- Sohn, S.H.; Cho, S.A. Comparison of removal torques for implants with hydroxyapatite-blasted and sandblasted and acid-etched surfaces. Implant Dent. 2016, 25, 581–587. [Google Scholar] [CrossRef]
- Gill, T.; Ooi, H.; Tezulas, E.; Petrie, A.; Rawlinson, S.; Roccuzzo, M.; Shahdad, S. Osseointegration in the absence of primary stability: An experimental preclinical mandibular minipig overpreparation in vivo model. Clin. Oral Implant. Res. 2025, in press. [Google Scholar] [CrossRef]
- Makary, C.; Menhall, A.; Lahoud, P.; Yang, K.R.; Park, K.B.; Razukevicius, D.; Traini, T. Bone-to-implant contact in implants with plasma-treated nanostructured calcium-incorporated surface (XPEEDActive) compared to non-plasma-treated implants (XPEED): A human histologic study at 4 weeks. Materials 2024, 17, 2331. [Google Scholar] [CrossRef]
- Nguyen, A.T.M.; Landao-Bassonga, E.; Kontogiorgos, E.D.; Lee, C.; Cheng, T.; Ngo, H.C.; Allan, B.; Goonewardene, M.; Opperman, L.A.; Zheng, M. The efficacy of a novel porcine-derived collagen membrane on guided bone regeneration: A comparative study in canine model. BMC Oral Health 2025, 25, 850. [Google Scholar] [CrossRef]
- Li, H.; Fan, X.; Tao, B.; Wang, W.; Wu, Y.; Chen, X. ZygoPlanner: A three-stage graphics-based framework for optimal preoperative planning of zygomatic implant placement. Med. Image Anal. 2025, 101, 103401. [Google Scholar] [CrossRef]
- Park, J.Y.; Kwon, Y.H.; Song, Y.W.; Cha, J.K.; Jung, U.W.; Jung, R.E.; Thoma, D.S. Flapless early implant placement into the uncalcified provisional matrix—Does it lead to osseointegration of the implant? A preclinical study. J. Clin. Periodontol. 2024, 51, 766–773. [Google Scholar] [CrossRef] [PubMed]
- Mangano, C.; Tumedei, M.; Piattelli, A.; Mangano, F.G.; Romasco, T.; Di Pietro, N.; Iezzi, G. A human self-locking cone Morse connection retrieved after 30 years: A histological and histomorphometric case report. Eng 2025, 6, 58. [Google Scholar] [CrossRef]
- Deng, B.; Tan, K.B.; Liu, G.R.; Lu, Y. Influence of osseointegration degree and pattern on resonance frequency in the assessment of dental implant stability using finite-element analysis. Int. J. Oral Maxillofac. Implant. 2008, 23, 1082. [Google Scholar]
- Bezdjian, A.; Mikolajewicz, N.; Denton, C.; Bouchard, A.; Rummler, M.; Gludovatz, B.; Mihee, S.; Elizabeth, Z.; Samer, S.; Daniel, S.J.; et al. Resonance frequency analysis identifies implant- and host-related factors associated with bone-anchored hearing implant stability. Otol. Neurotol. 2024, 45, 676–683. [Google Scholar] [CrossRef]
- Lee, J.; Lim, Y.J.; Ahn, J.S.; Kim, B.; Baek, Y.W.; Lim, B.S. Correlation of two different devices for evaluation of primary implant stability depending on dental implant length and bone density: An in vitro study. PLoS ONE 2024, 19, e0290595. [Google Scholar] [CrossRef]
- Fu, P.S.; Du, J.K.; Tseng, F.C.; Hsu, H.J.; Lan, T.H.; Kung, J.C.; Lai, P.-L.; Lee, K.-T.; Liu, C.-T.; Chen, W.-C.; et al. Comparison of implant stability and marginal bone loss between osseodensification and conventional osteotomy at adjacent implant sites. J. Dent. Sci. 2025, 20, 1002–1007. [Google Scholar] [CrossRef]
- Tofan, N.H.; Hussaini, A.H.A. Evaluation of dental implant stability in narrow alveolar ridges utilizing osseodensification technique versus screw expansion: A randomized single-blinded clinical trial. Dent. Hypotheses 2024, 15, 37–40. [Google Scholar] [CrossRef]
- Sghaireen, M.G.; Alam, M.K.; Mohamedeissa, A.A.S.; Kazma, J.M. In-vitro study of osseointegration: Evaluating the influence of surface modifications on dental implant stability. J. Pharm. Bioallied Sci. 2024, 16 (Suppl. S3), S2518–S2520. [Google Scholar] [CrossRef]
- Kim, H.; Noh, G.; Kim, H.; Han, H.S.; Kim, S.; Cho, Y.D. In vitro evaluation of prosthesis-level implant stability using ‘BracketPeg’. J. Periodontal Implant Sci. 2025, 55, 232–239. [Google Scholar] [CrossRef]
- Silva-Henao, J.D.; Schober, S.; Pahr, D.H.; Reisinger, A.G. Critical loss of primary implant stability in osteosynthesis locking screws under cyclic overloading. Med. Eng. Phys. 2024, 126, 104143. [Google Scholar] [CrossRef]
- Rugova, S.; Abboud, M. Comparison of one-drill protocol to sequential drilling in vitro and in vivo. Bioengineering 2025, 12, 51. [Google Scholar] [CrossRef]
- Rugova, S.; Abboud, M. Thermal evaluation of bone drilling with a one-drill protocol. Bioengineering 2024, 11, 1022. [Google Scholar] [CrossRef]
- Bera, T.; Kukreja, B.J.; Sharma, C.; Gupta, V.V.; Patel, P.; Singhal, P.; Jadhav, M.S.; Pattnaik, N. Relative contribution of trabecular and cortical bone to primary implant stability: An in vitro model study. J. Pharm. Bioallied Sci. 2024, 16 (Suppl. S4), S3427–S3429. [Google Scholar] [CrossRef] [PubMed]
- Zieliński, R.; Puszkarz, A.K.; Piętka, T.; Sowiński, J.; Sadowska-Sowińska, M.; Kołkowska, A.; Simka, W. Comparative Evaluation of Bone–Implant Contact in Various Surface-Treated Dental Implants Using High-Resolution Micro-CT in Rabbits’ Bone. Materials 2024, 17, 5396. [Google Scholar] [CrossRef] [PubMed]
- Andersen, O.Z.; Bellón, B.; Lamkaouchi, M.; Brunelli, M.; Wei, Q.; Procter, P.; Pippenger, B.E. Determining primary stability for adhesively stabilized dental implants. Clin. Oral Investig. 2023, 27, 3741–3748. [Google Scholar] [CrossRef]
- Gehrke, S.A.; Marin, G.W. Biomechanical Evaluation of Dental Implants with Three Different Designs: Removal Torque and Resonance Frequency Analysis in Rabbits. Ann. Anat. 2015, 199, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Lee, S.K.; Kim, H.S.; Kim, W.J.; Ryu, J.H.; Ji, M.K.; Lim, H.P. Atomic layer deposition of zirconia on titanium implants improves osseointegration in rabbit bones. Int. J. Nanomed. 2024, 19, 5011–5020. [Google Scholar] [CrossRef]
- Imber, J.C.; Roccuzzo, A.; Irani, D.R.; Bellón, B.; Bosshardt, D.D.; Sculean, A.; Pippenger, B.E. Histological evaluation of osseointegration between conventional and novel bone-level tapered implants in healed bone: A preclinical study. J. Periodontal Res. 2024, 59, 1210–1219. [Google Scholar] [CrossRef]
- Kuwano, K.; Canullo, L.; Botticelli, D.; Xavier, S.P.; Silva, E.R.; Kusano, K.; Baba, S. Ablative and expansive protocols for bone osteotomy in rabbits. Dent. J. 2025, 13, 118. [Google Scholar] [CrossRef]
- Jar, C.; Archibald, A.; Gibson, M.; Westover, L. Evaluation of a vibration modeling technique for the in-vitro measurement of dental implant stability. J. Mech. Behav. Biomed. Mater. 2024, 154, 106537. [Google Scholar] [CrossRef]
- Meredith, N.; Alleyne, D.; Cawley, P. Quantitative determination of the stability of the implant-tissue interface using resonance frequency analysis. Clin. Oral Implant. Res. 1996, 7, 261–267. [Google Scholar] [CrossRef]
- Wakamatsu, K.; Doi, K.; Kobatake, R.; Oki, Y.; Tsuga, K. Evaluation of bone density for primary implant stability using a newly designed drill: An in vitro study on polyurethane bone blocks. Clin. Exp. Dent. Res. 2024, 10, e70048. [Google Scholar] [CrossRef] [PubMed]
- Al Madhoun, A.; Meshal, K.; Carrió, N.; Ferrés-Amat, E.; Ferrés-Amat, E.; Barajas, M.; Jiménez-Escobar, A.L.; Al-Madhoun, A.S.; Saber, A.; Abou Alsamen, Y.; et al. Randomized clinical trial: Bone bioactive liquid improves implant stability and osseointegration. J. Funct. Biomater. 2024, 15, 293. [Google Scholar] [CrossRef]
- Fontes Pereira, J.; Costa, R.; Nunes Vasques, M.; Relvas, M.; Braga, A.C.; Salazar, F.; Infante da Câmara, M. The effectiveness of osseodensification drilling versus the conventional surgical technique on implant stability: A clinical trial. J. Clin. Med. 2024, 13, 2912. [Google Scholar] [CrossRef]
- do Vale Souza, J.P.; de Moraes Melo Neto, C.L.; Piacenza, L.T.; da Silva, E.V.F.; de Melo Moreno, A.L.; Penitente, P.A.; Brunetto, J.L.; dos Santos, D.M.; Goiato, M.C. Relation Between Insertion Torque and Implant Stability Quotient: A Clinical Study. Eur. J. Dent. 2021, 15, 618–623. [Google Scholar] [CrossRef]
- Lima Monteiro, F.; Moreira, C.L.; Arias Pecorari, V.G.; Orth, C.C.; Joly, J.C.; Peruzzo, D. Biomechanical and histomorphometric analysis of osseodensification drilling versus conventional technique: A systematic review and meta-analysis. Quintessence Int. 2024, 55, 212. [Google Scholar]
- Galletti, F.; D’Angelo, T.; Fiorillo, L.; Lo Giudice, P.; Irrera, N.; Rizzo, G.; Cervino, G. Micro-CT structure analysis on dental implants: Preliminary in vitro trial. Prosthesis 2024, 6, 1437–1447. [Google Scholar] [CrossRef]
- Morandini, L.; Heath, T.; Sheakley, L.S.; Avery, D.; Grabiec, M.; Friedman, M.; Martin, R.K.; Boyd, J.; Olivares-Navarrete, R. Adoptive transfer of immunomodulatory macrophages reduces the pro-inflammatory microenvironment and increases bone formation on titanium implants. Acta Biomater. 2024, 188, 432–445. [Google Scholar] [CrossRef]
- Berger, M.B.; Bosh, K.; Deng, J.; Jacobs, T.W.; Cohen, D.J.; Boyan, B.D.; Schwartz, Z. Wnt16 increases bone-to-implant contact in an osteopenic rat model by increasing proliferation and regulating the differentiation of bone marrow stromal cells. Ann. Biomed. Eng. 2024, 52, 1744–1762. [Google Scholar] [CrossRef]
- Perez, A.; Martinelli-Klay, C.P.; Lombardi, T. Increased peri-implant bone formation around simultaneously grafted hydrophilic microrough titanium implants: An exploratory human histometric analysis in four patients. Int. J. Periodontics Restor. Dent. 2024, 44, e103–e111. [Google Scholar] [CrossRef]
- Xiao, Y.; Lv, L.; Xu, Z.; Zhou, L.; Lin, Y.; Lin, Y.; Guo, J.; Chen, J.; Ou, Y.; Lin, L.; et al. Correlation between peri-implant bone mineral density and primary implant stability based on artificial intelligence classification. Sci. Rep. 2024, 14, 3009. [Google Scholar] [CrossRef]
- Khan, M.W.; Inayat, N.; Zafar, M.S.; Zaigham, A.M. A resonance frequency analysis to investigate the impact of implant size on primary and secondary stability. Pak. J. Med. Sci. 2024, 40, 1261–1266. [Google Scholar] [CrossRef] [PubMed]
- Mohyuddin, G.R.; Jobe, A.; Thomas, L. Does use of high-fidelity simulation improve resident physician competency and comfort identifying and managing bradyarrhythmias? Cureus 2020, 12, e6943. [Google Scholar] [CrossRef] [PubMed]
- Matta, R.E.; Berger, L.; Loehlein, M.; Leven, L.; Taxis, J.; Wichmann, M.; Motel, C. Stress distribution within the peri-implant bone for different implant materials obtained by digital image correlation. Materials 2024, 17, 2161. [Google Scholar] [CrossRef] [PubMed]
- Galas, A.; Banci, L.; Innocenti, B. The effects of different femoral component materials on bone and implant response in total knee arthroplasty: A finite-element analysis. Materials 2023, 16, 5605. [Google Scholar] [CrossRef]
- Liu, L.; Ma, S.; Zhang, Y.; Zhu, S.; Wu, S.; Liu, G.; Yang, G. Parametric design of porous structure and optimal porosity gradient distribution based on root-shaped implants. Materials 2024, 17, 1137. [Google Scholar] [CrossRef]








| Outcome/Time | NSK, Adjusted Mean (95% CI) | SAESHIN, Adjusted Mean (95% CI) | Difference (SAESHIN–NSK) | p-Value | Group × Time p |
|---|---|---|---|---|---|
| ISQ, immediate | 64.8 (61.0–68.6) | 71.7 (67.9–75.6) | +6.9 (+1.3–+12.5) | 0.018 | 0.011 |
| ISQ, 1 month | 79.8 (77.7–81.9) | 78.6 (76.4–80.8) | −1.2 (−4.3–+1.9) | 0.42 |
| Outcome | σ2_Rabbit (SE) | σ2_Residual (SE) | ICC |
|---|---|---|---|
| ISQ (both times) | 22.8 (8.1) | 40.7 (7.5) | 0.36 |
| Removal torque | 68.5 (24.3) | 152.0 (31.8) | 0.31 |
| BIG (total) | 3.2 (1.9) | 43.7 (8.6) | 0.07 |
| Outcome | NSK, Adjusted Mean (95% CI) | SAESHIN, Adjusted Mean (95% CI) | Difference (SAESHIN–NSK) | p-Value |
|---|---|---|---|---|
| Removal torque (N·cm) | 55.8 (47.4–64.2) | 60.1 (52.5–67.7) | +4.3 (−5.2–+13.8) | 0.36 |
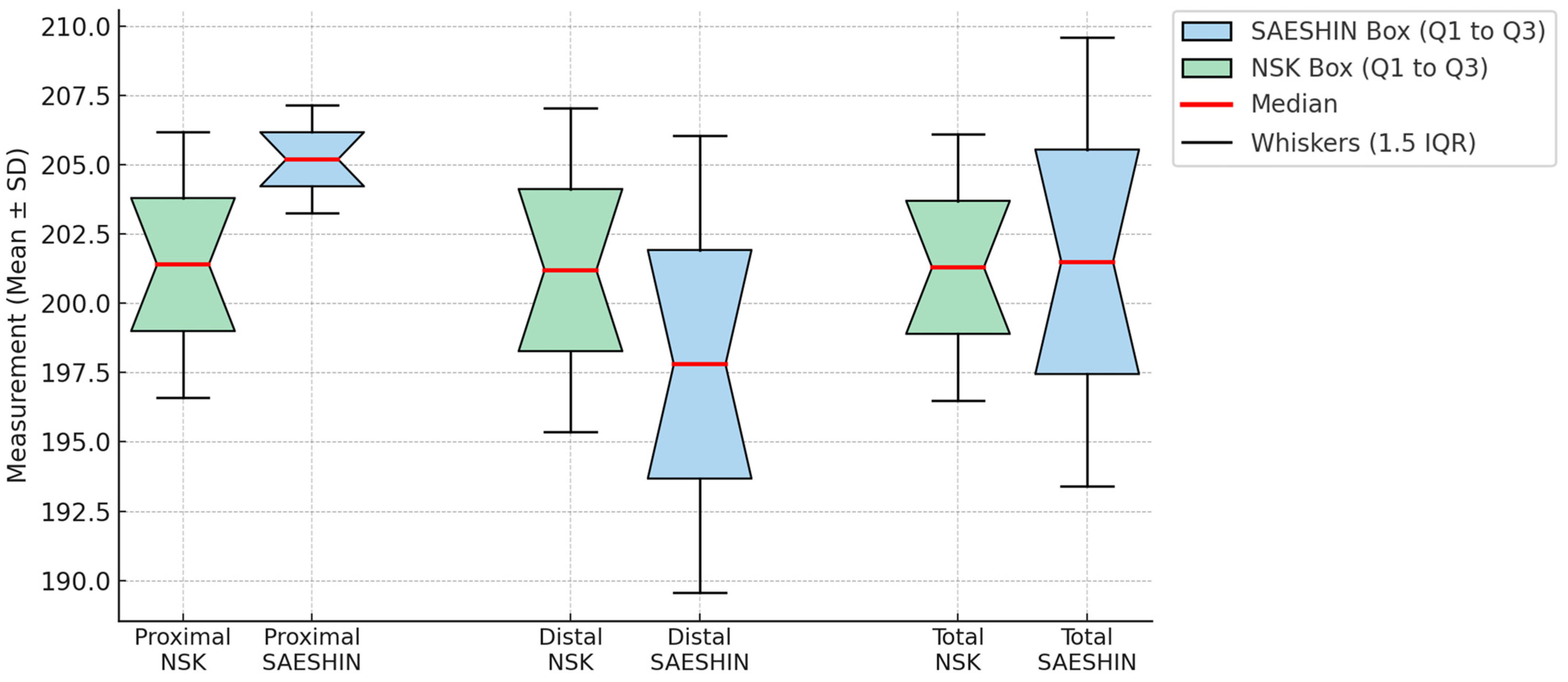
| Region | NSK, Adjusted Mean (95% CI) | SAESHIN, Adjusted Mean (95% CI) | Difference (SAESHIN–NSK) | p-Value |
|---|---|---|---|---|
| Proximal | 201.6 (199.7–203.5) | 204.9 (203.2–206.7) | +3.3 (−0.6–+7.2) | 0.10 |
| Distal | 201.0 (198.6–203.4) | 198.4 (195.9–200.8) | −2.6 (−7.0–+1.8) | 0.24 |
| Total | 201.3 (199.5–203.0) | 201.6 (199.7–203.4) | +0.3 (−3.5–+4.1) | 0.88 |
| Contrast | Equivalence Margin | p_Lower | p_Upper | Conclusion |
|---|---|---|---|---|
| SAESHIN–NSK at 1 month | ±5 ISQ | 0.024 | 0.019 | Equivalent (both p < 0.05) |
| Correlation | r_Partial (95% CI) | p-Value |
|---|---|---|
| ISQ (1 month) vs. removal torque | 0.41 (0.01–0.68) | 0.041 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Son, K.; Son, Y.-T.; Hwang, S.-M.; Lee, J.M.; Kim, J.-W.; Lee, K.-B. Effects of Two Dental Implant Micromotor Systems for Dental Implant Placement on Implant Stability and Removal Torque: An Animal Experiment. Materials 2025, 18, 4048. https://doi.org/10.3390/ma18174048
Son K, Son Y-T, Hwang S-M, Lee JM, Kim J-W, Lee K-B. Effects of Two Dental Implant Micromotor Systems for Dental Implant Placement on Implant Stability and Removal Torque: An Animal Experiment. Materials. 2025; 18(17):4048. https://doi.org/10.3390/ma18174048
Chicago/Turabian StyleSon, Keunbada, Young-Tak Son, Sung-Min Hwang, Jae Mok Lee, Jin-Wook Kim, and Kyu-Bok Lee. 2025. "Effects of Two Dental Implant Micromotor Systems for Dental Implant Placement on Implant Stability and Removal Torque: An Animal Experiment" Materials 18, no. 17: 4048. https://doi.org/10.3390/ma18174048
APA StyleSon, K., Son, Y.-T., Hwang, S.-M., Lee, J. M., Kim, J.-W., & Lee, K.-B. (2025). Effects of Two Dental Implant Micromotor Systems for Dental Implant Placement on Implant Stability and Removal Torque: An Animal Experiment. Materials, 18(17), 4048. https://doi.org/10.3390/ma18174048








