Magnetite Nanoparticles: Synthesis and Applications in Optics and Nanophotonics
Abstract
:1. Introduction
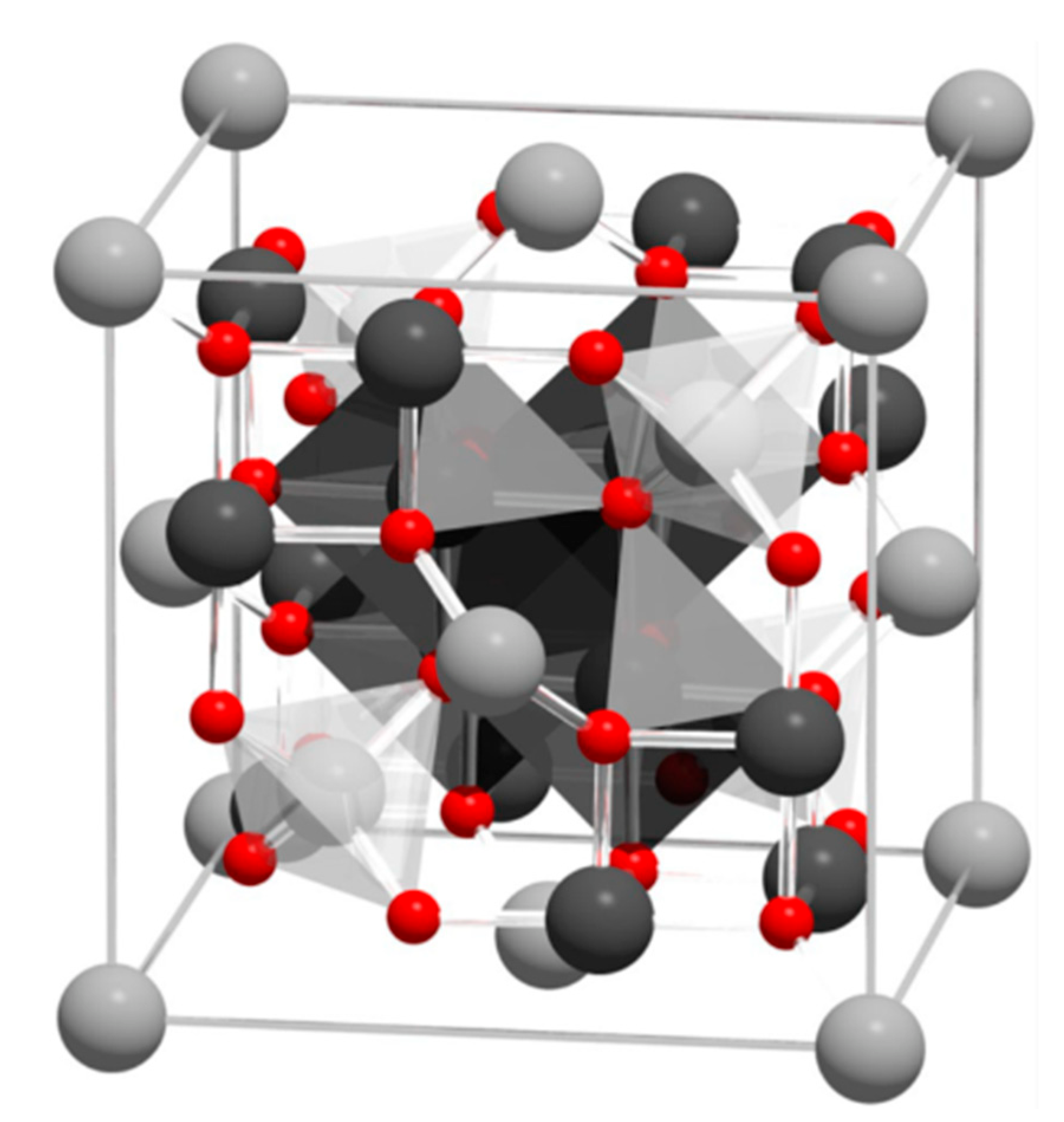
2. Structure and Properties of Magnetite
3. Synthesis and Characterization of Magnetite Nanoparticles
3.1. Chemical Synthesis of Magnetite Nanoparticles
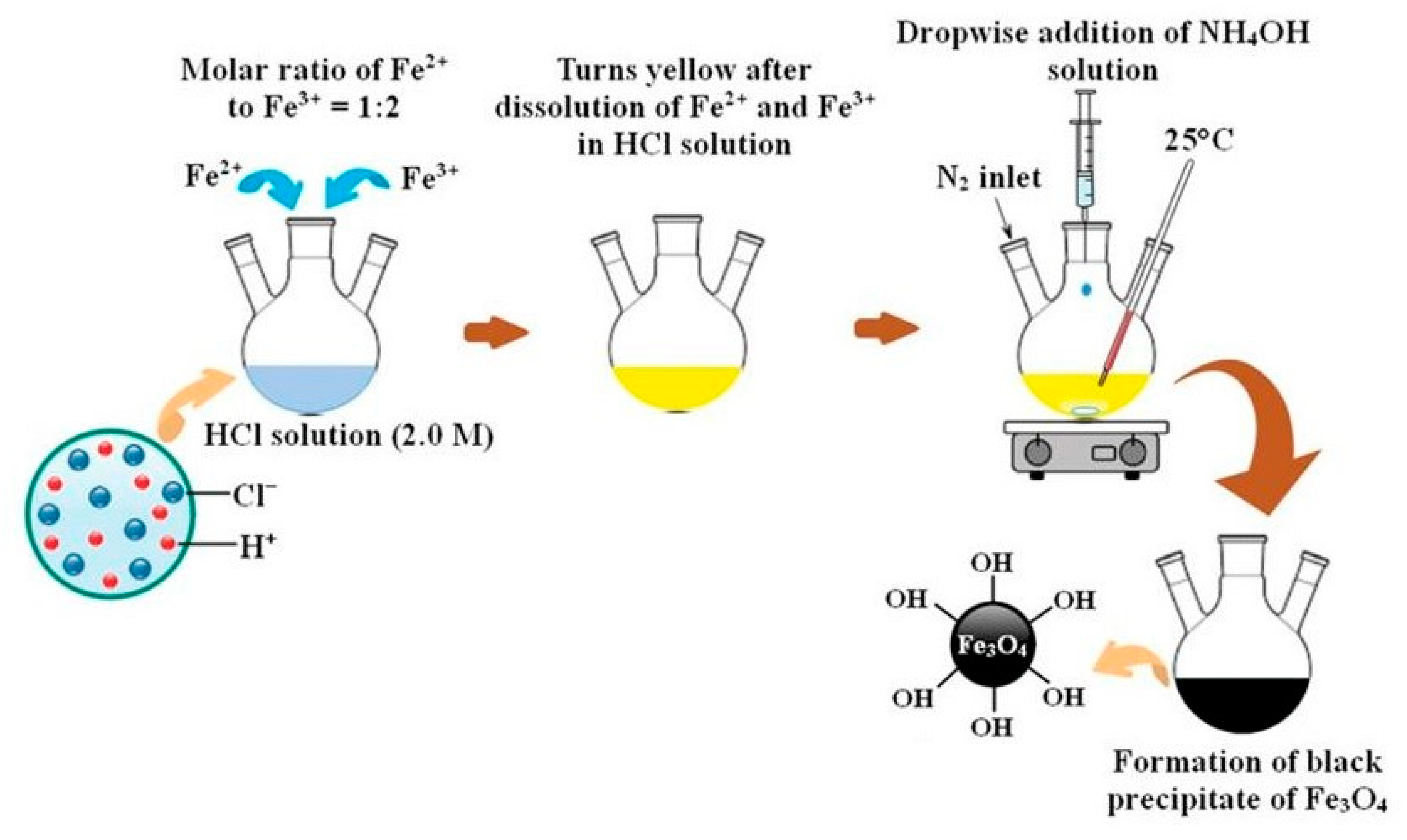
3.1.1. Co-Precipitation Method
3.1.2. Partial Oxidation of Ferrous Hydroxide
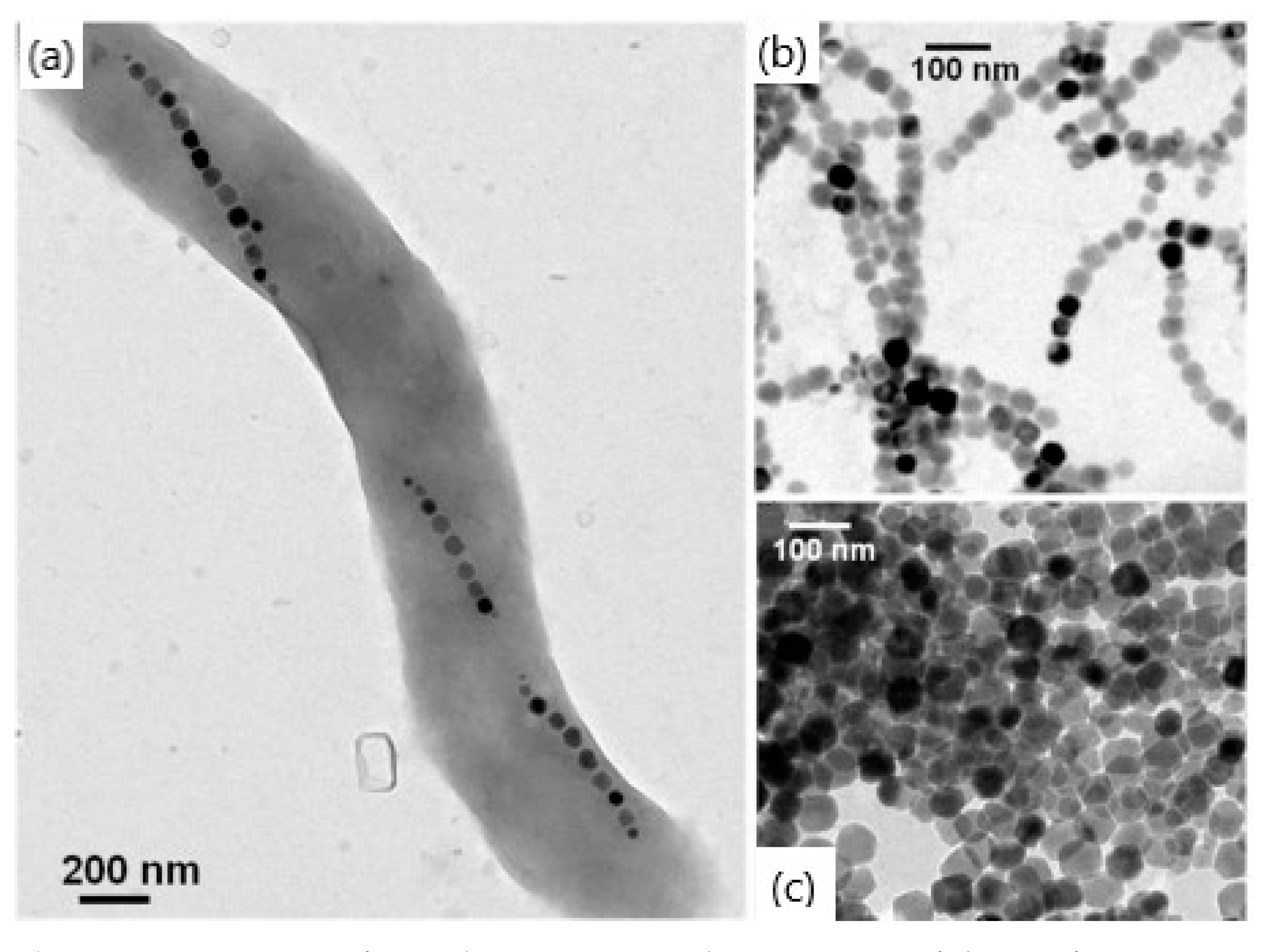
3.1.3. Reaction in Constrained Environments
3.1.4. Hydrothermal or High-Temperature Reactions
3.1.5. Polyol Method
3.1.6. Sol–Gel Synthesis
3.1.7. Sonochemical Synthesis
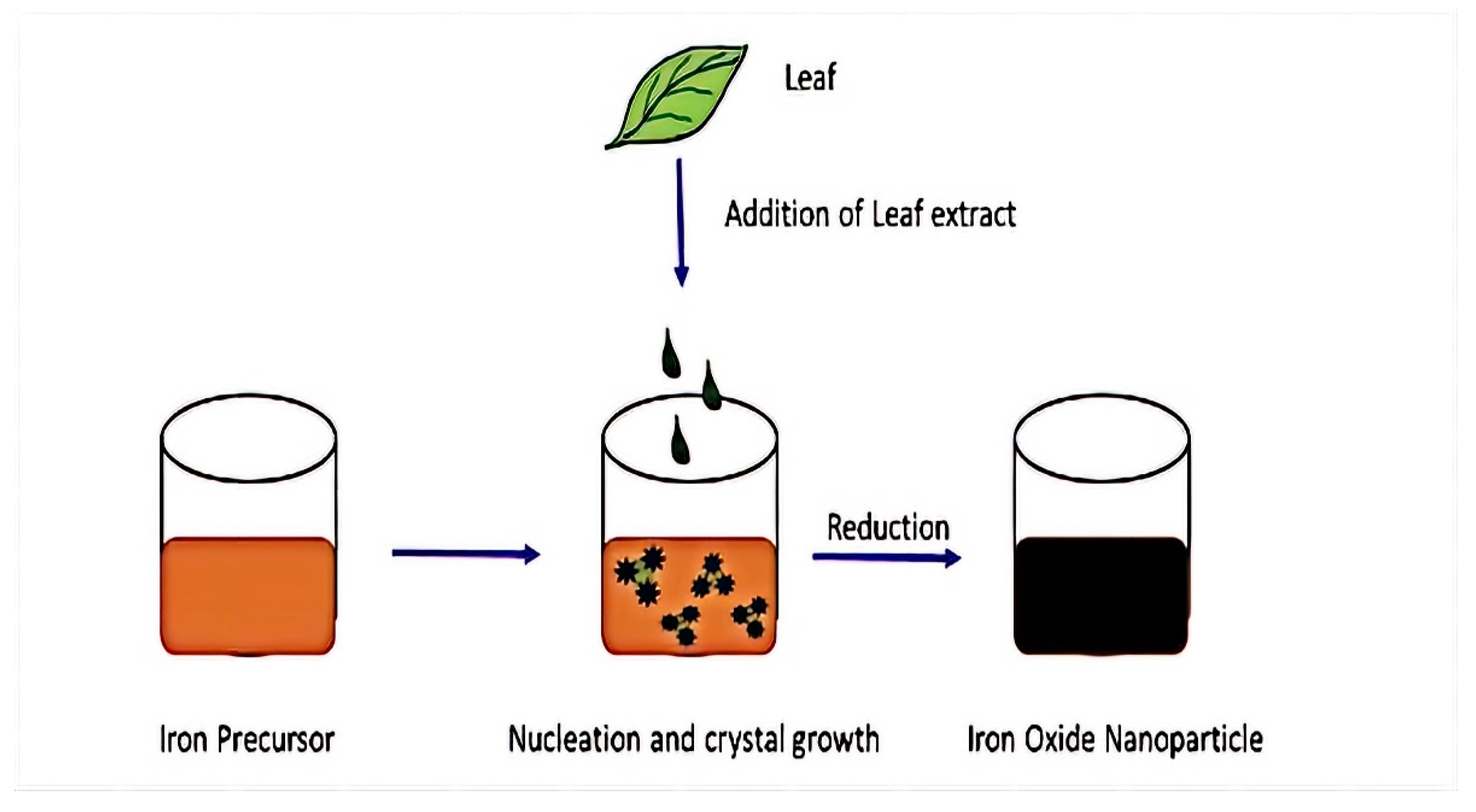
3.2. Biological Synthesis of Magnetite Nanoparticles
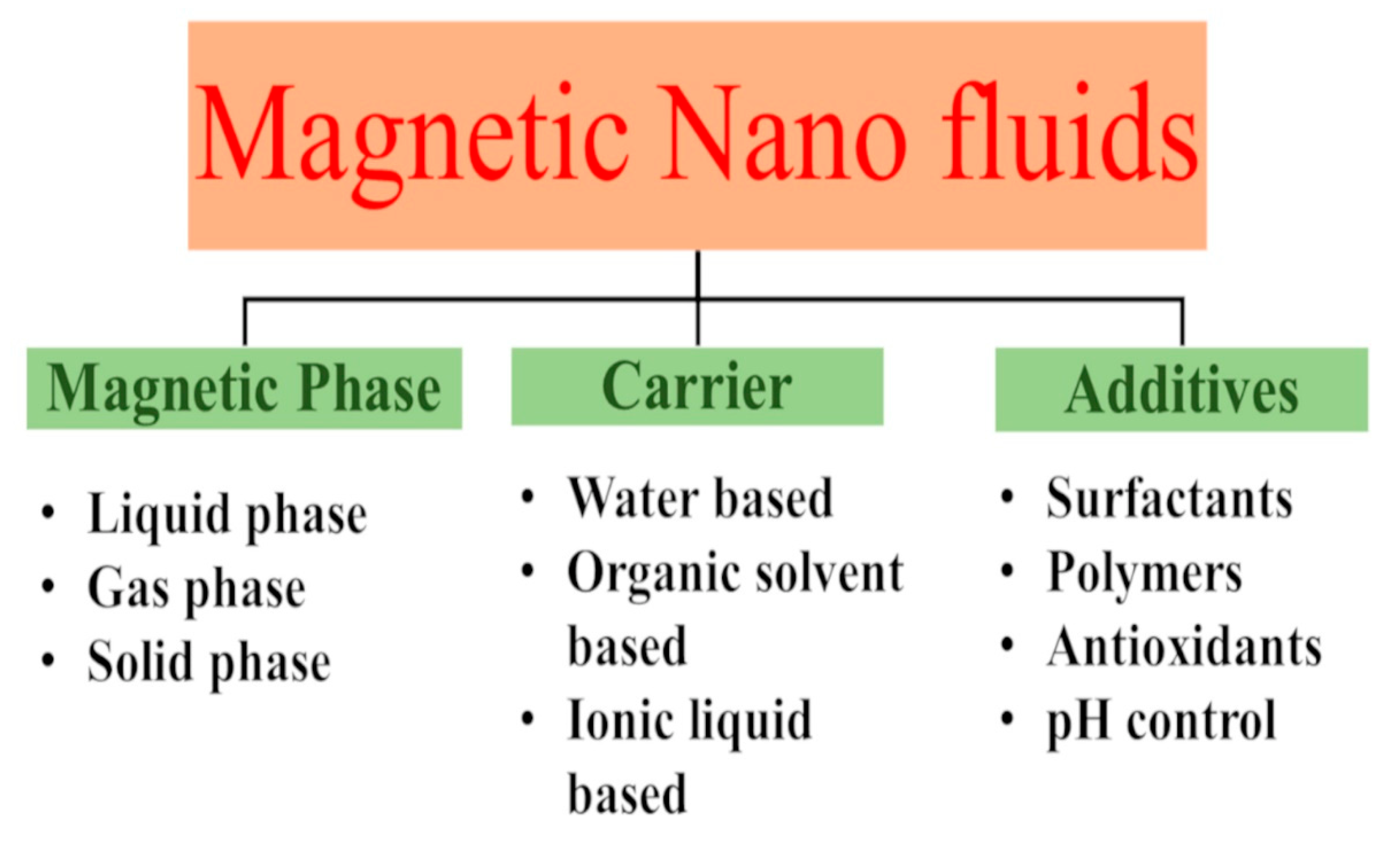
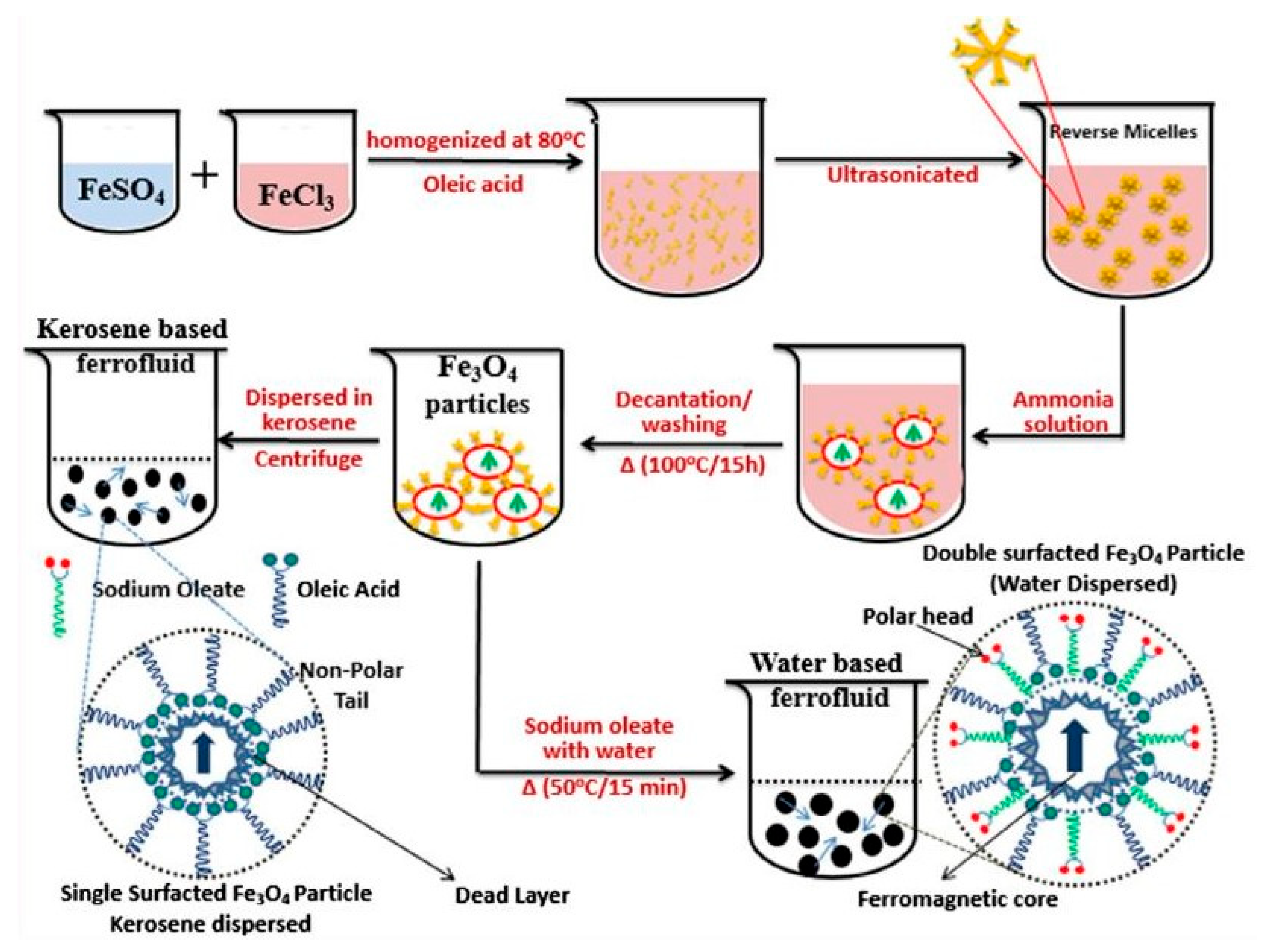
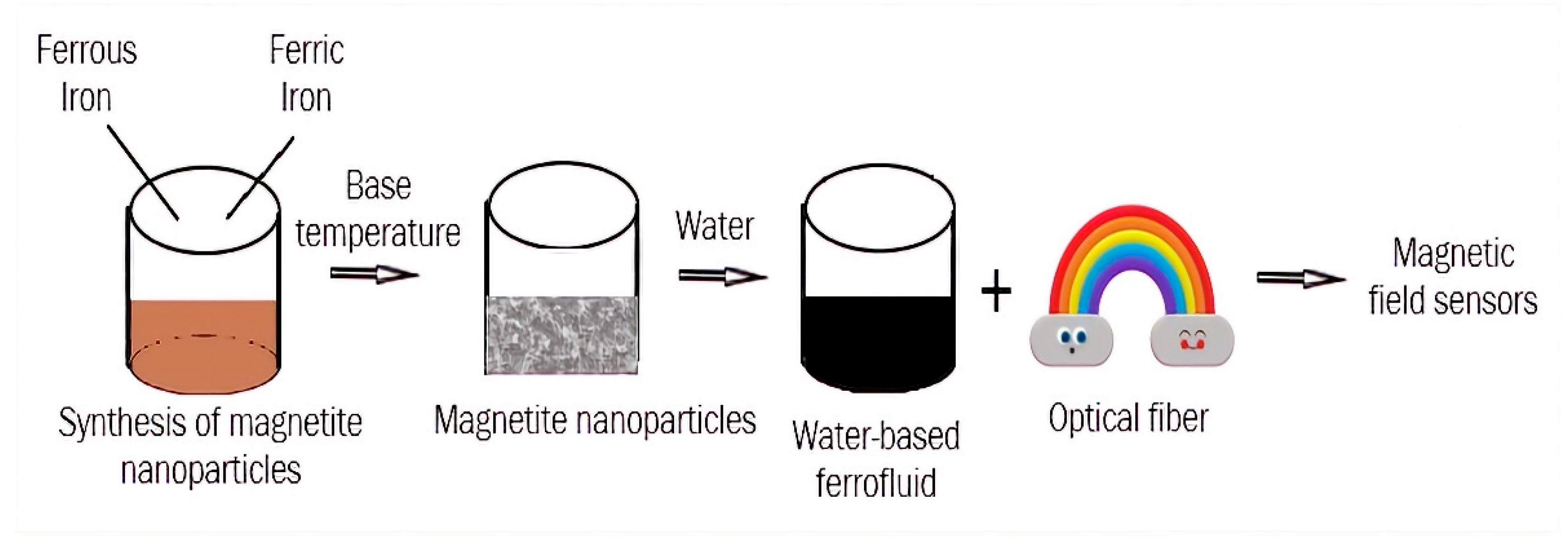
4. Preparation of Magnetic Ferrofluids
5. Application of Magnetic Ferrofluids in Optics and Nanophotonics
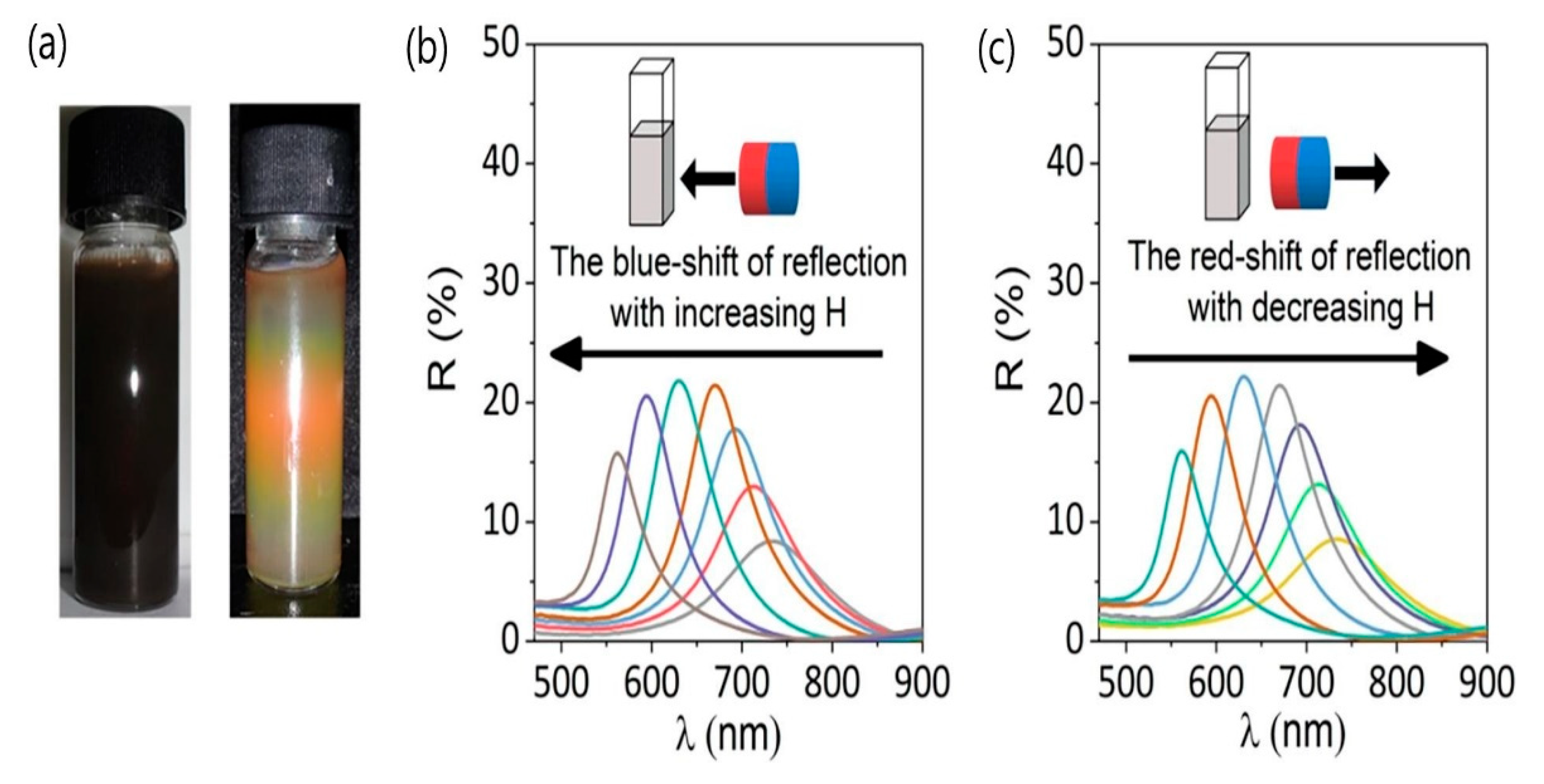
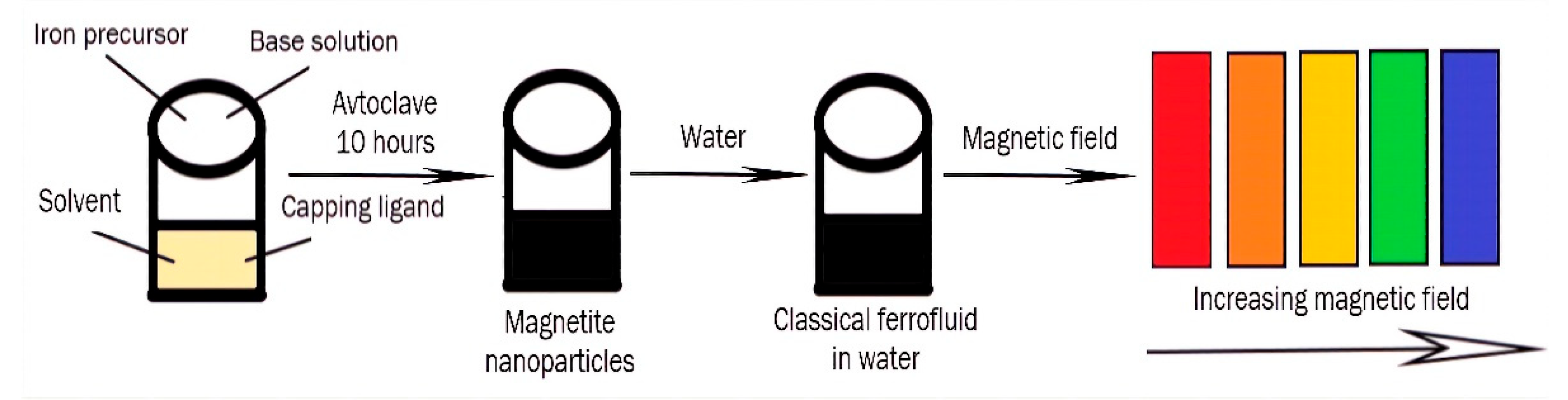
5.1. Photonic Materials
5.2. Organic Light-Emitting Diodes (OLEDs)
5.3. Magnetic Field Sensors
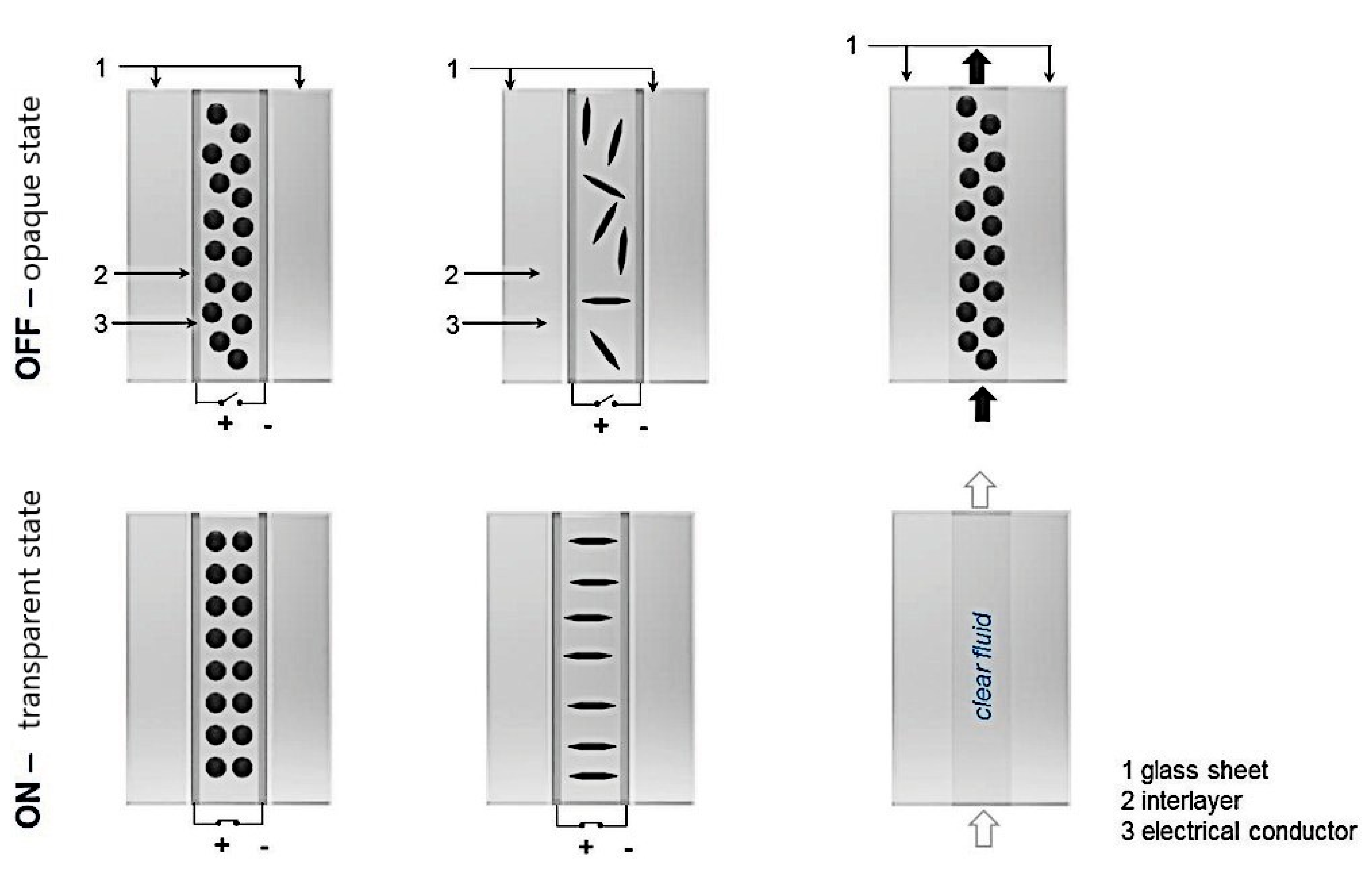
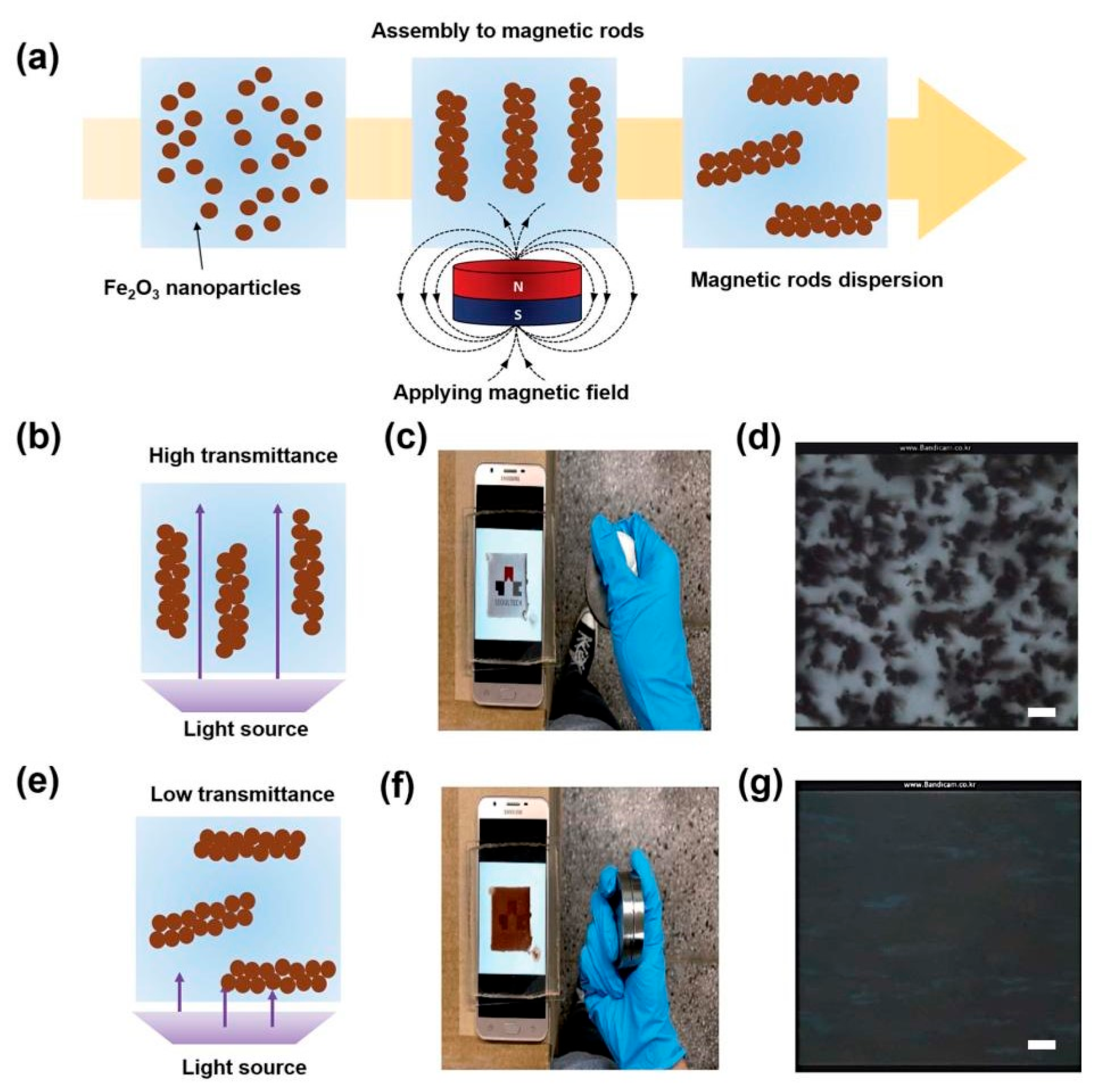
5.4. “Smart” Windows
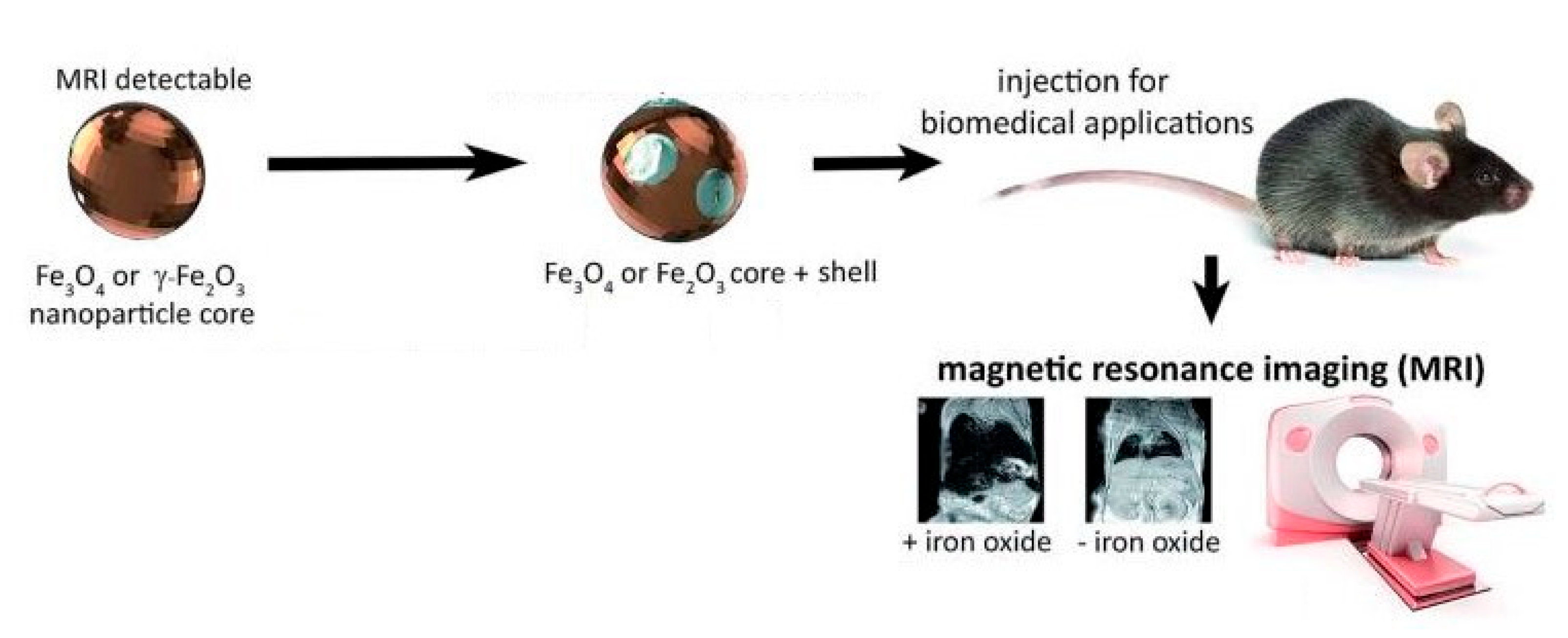
5.5. Magnetic Resonance Imaging
5.6. Solar Energy Harvesting
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Piracha, S.; Saleem, S.; Momil, G.; Anjum, A.; Yaseen, Z. Nanoparticle: Role in Chemical Industries, Potential Sources and Chemical Catalysis Applications. Sch. Int. J. Chem. Mater. Sci. 2020, 4, 40–45. [Google Scholar] [CrossRef]
- Subhan, A.; Choudhury, K.P.; Neogi, N. Advances with Molecular Nanomaterials in Industrial Manufacturing Applications. Int. J. Nanomanufacturing 2021, 1, 75–97. [Google Scholar] [CrossRef]
- Pawar, S.; Duadi, H.; Fleger, Y.; Fixler, D. Carbon Dots-Based Logic Gates. Nanomaterials 2021, 11, 232. [Google Scholar] [CrossRef] [PubMed]
- Prasad, R.; Bhattacharyya, A.; Nguyen, Q.D. Nanotechnology in Sustainable Agriculture: Recent Developments, Challenges, and Perspectives. Front. Microbiol. 2017, 8, 124–127. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Usman, M.; Farooq, M.; Wakeel, A.; Nawaz, A.; Alam Cheema, S.A.; Rehman, H.U.; Ashraf, I.; Sanaullah, M. Nanotechnology in agriculture: Current status, challenges and future opportunities. Sci. Total. Environ. 2020, 721, 137778. [Google Scholar] [CrossRef]
- Barnoy, E.A.; Popovtzer, R.; Fixler, D. Fluorescence for biological logic gates. J. Biophotonics 2020, 13, e202000158. [Google Scholar] [CrossRef]
- Finke, J.H.; Juhnke, M.; Kwade, A.; Bunjes, H.; Cornier, J.; Owen, A.; Van de Voorde, M. Overview of techniques and description of established processes. In Pharmaceutical Nanotechnology: Innovation and Production, 1st. ed.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2017; p. 772. [Google Scholar]
- Serrano, E.; Rus, G.; García-Martínez, J. Nanotechnology for sustainable energy. Renew. Sustain. Energy Rev. 2009, 13, 2373–2384. [Google Scholar] [CrossRef]
- Mabrouk, M.; Das, D.; Salem, Z.; Beherei, H. Nanomaterials for Biomedical Applications: Production, Characterisations, Recent Trends and Difficulties. Molecules 2021, 26, 1077. [Google Scholar] [CrossRef]
- Materón, E.M.; Miyazaki, C.M.; Carr, O.; Joshi, N.; Picciani, P.H.; Dalmaschio, C.J.; Davis, F.; Shimizu, F.M. Magnetic nanoparticles in biomedical applications: A review. Appl. Surf. Sci. Adv. 2021, 6, 100163. [Google Scholar] [CrossRef]
- Aflori, M. Smart Nanomaterials for Biomedical Applications—A Review. Nanomaterials 2021, 11, 396. [Google Scholar] [CrossRef]
- Arcudi, F.; Đorđević, L.; Prato, M. Design, Synthesis, and Functionalization Strategies of Tailored Carbon Nanodots. Acc. Chem. Res. 2019, 52, 2070–2079. [Google Scholar] [CrossRef] [PubMed]
- An, L.; Wang, Y.; Tian, Q.; Yang, S. Small Gold Nanorods: Recent Advances in Synthesis, Biological Imaging, and Cancer Therapy. Materials 2017, 10, 1372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parente, M.; Van Helvert, M.; Hamans, R.F.; Verbroekken, R.; Sinha, R.; Bieberle-Hütter, A.; Baldi, A. Simple and Fast High-Yield Synthesis of Silver Nanowires. Nano Lett. 2020, 20, 5759–5764. [Google Scholar] [CrossRef] [PubMed]
- Vorobjova, A.; Tishkevich, D.; Shimanovich, D.; Zubar, T.; Astapovich, K.; Kozlovskiy, A.; Zdorovets, M.; Zhaludkevich, A.; Lyakhov, D.; Michels, D.; et al. The influence of the synthesis conditions on the magnetic behaviour of the densely packed arrays of Ni nanowires in porous anodic alumina membranes. RSC Adv. 2021, 11, 3952–3962. [Google Scholar] [CrossRef]
- Gupta, N.; Gupta, S.M.; Sharma, S.K. Carbon nanotubes: Synthesis, properties and engineering applications. Carbon Lett. 2019, 29, 419–447. [Google Scholar] [CrossRef]
- Chen, H.; Zhang, L.; Li, M.; Xie, G. Synthesis of Core–Shell Micro/Nanoparticles and Their Tribological Application: A Review. Materials 2020, 13, 4590. [Google Scholar] [CrossRef]
- Kianfar, E. Magnetic Nanoparticles in Targeted Drug Delivery: A Review. J. Supercond. Nov. Magn. 2021, 34, 1709–1735. [Google Scholar] [CrossRef]
- Price, P.M.; Mahmoud, W.E.; Al-Ghamdi, A.A.; Bronstein, L.M. Magnetic Drug Delivery: Where the Field Is Going. Front. Chem. 2018, 6, 619. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.-L.; Chen, D.; Shang, P.; Yin, D.-C. A review of magnet systems for targeted drug delivery. J. Control. Release 2019, 302, 90–104. [Google Scholar] [CrossRef]
- Chacón-Torres, J.C.; Reinoso, C.; Navas-León, D.G.; Briceño, S.; González, G. Optimized and scalable synthesis of magnetic nanoparticles for RNA extraction in response to developing countries’ needs in the detection and control of SARS-CoV-2. Sci. Rep. 2020, 10, 19004. [Google Scholar] [CrossRef]
- Khatami, F.; Najafi, F.; Yari, F.; Khavari-Nejad, R.A. Magnetic nanoparticles: A promising component in RNA extraction process. Prog. Biol. Sci. 2017, 7, 47–52. [Google Scholar] [CrossRef]
- Tang, C.; He, Z.; Liu, H.; Xu, Y.; Huang, H.; Yang, G.; Xiao, Z.; Li, S.; Liu, H.; Deng, Y.; et al. Application of magnetic nanoparticles in nucleic acid detection. J. Nanobiotechnol. 2020, 18, 62. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grzeskowiak, B.; Sánchez-Antequera, Y.; Hammerschmid, E.; Döblinger, M.; Eberbeck, D.; Woźniak, A.; Słomski, R.; Plank, C.; Mykhaylyk, O. Nanomagnetic Activation as a Way to Control the Efficacy of Nucleic Acid Delivery. Pharm. Res. 2015, 32, 103–121. [Google Scholar] [CrossRef] [PubMed]
- Zuvin, M.; Kuruoglu, E.; Kaya, V.O.; Unal, O.; Kutlu, O.; Acar, H.Y.; Gozuacik, D.; Koşar, A. Magnetofection of Green Fluorescent Protein Encoding DNA-Bearing Polyethyleneimine-Coated Superparamagnetic Iron Oxide Nanoparticles to Human Breast Cancer Cells. ACS Omega 2019, 4, 12366–12374. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalubowilage, M.; Janik, K.; Bossmann, S.H. Magnetic Nanomaterials for Magnetically-Aided Drug Delivery and Hyperthermia. Appl. Sci. 2019, 9, 2927. [Google Scholar] [CrossRef] [Green Version]
- Rajan, A.; Sahu, N.K. Review on magnetic nanoparticle-mediated hyperthermia for cancer therapy. J. Nanopart. Res. 2020, 22, 319. [Google Scholar] [CrossRef]
- Chang, D.; Lim, M.; Goos, J.; Qiao, R.; Ng, Y.Y.; Mansfeld, F.M.; Jackson, M.; Davis, T.P.; Kavallaris, M. Biologically Targeted Magnetic Hyperthermia: Potential and Limitations. Front. Pharmacol. 2018, 9, 831. [Google Scholar] [CrossRef] [Green Version]
- Fatima, H.; Charinpanitkul, T.; Kim, K.-S. Fundamentals to Apply Magnetic Nanoparticles for Hyperthermia Therapy. Nanomaterials 2021, 11, 1203. [Google Scholar] [CrossRef]
- Avasthi, A.; Caro, C.; Pozo-Torres, E.; Leal, M.P.; García-Martín, M.L. Magnetic Nanoparticles as MRI Contrast Agents. Top. Curr. Chem. 2020, 378, 49–91. [Google Scholar] [CrossRef]
- Fernández-Barahona, I.; Muñoz-Hernando, M.; Ruiz-Cabello, J.; Herranz, F.; Pellico, J. Iron Oxide Nanoparticles: An Alternative for Positive Contrast in Magnetic Resonance Imaging. Inorganics 2020, 8, 28. [Google Scholar] [CrossRef] [Green Version]
- Haghighi, A.H.; Khorasani, M.T.; Faghih, Z.; Farjadian, F. Effects of different quantities of antibody conjugated with magnetic nanoparticles on cell separation efficiency. Heliyon 2020, 6, e03677. [Google Scholar] [CrossRef] [PubMed]
- Bragg, W.H. The Structure of Magnetite and the Spinels. Nature 1915, 95, 561. [Google Scholar] [CrossRef]
- Moreno, R.; Poyser, S.; Meilak, D.; Meo, A.; Jenkins, S.; Lazarov, V.K.; Vallejo-Fernandez, G.; Majetich, S.; Evans, R.F.L. The role of faceting and elongation on the magnetic anisotropy of magnetite Fe3O4 nanocrystals. Sci. Rep. 2020, 10, 2722. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Shah, T.; Ullah, R.; Zhou, P.; Guo, M.; Ovais, M.; Tan, Z.; Rui, Y. Review on Recent Progress in Magnetic Nanoparticles: Synthesis, Characterization, and Diverse Applications. Front. Chem. 2021, 9, 629054. [Google Scholar] [CrossRef]
- Wallyn, J.; Anton, N.; Vandamme, T.F. Synthesis, Principles, and Properties of Magnetite Nanoparticles for In Vivo Imaging Applications—A Review. Pharmaceutics 2019, 11, 601. [Google Scholar] [CrossRef] [Green Version]
- Shukla, S.; Khan, R.; Daverey, A. Synthesis and characterization of magnetic nanoparticles, and their applications in wastewater treatment: A review. Environ. Technol. Innov. 2021, 24, 101924. [Google Scholar] [CrossRef]
- Hernández-Hernández, A.A.; Aguirre-Álvarez, G.; Cariño-Cortés, R.; Mendoza-Huizar, L.H.; Jiménez-Alvarado, R. Iron oxide nanoparticles: Synthesis, functionalization, and applications in diagnosis and treatment of cancer. Chem. Pap. 2020, 74, 3809–3824. [Google Scholar] [CrossRef]
- Ali, A.; Zafar, H.; Zia, M.; ul Haq, I.; Phull, A.R.; Ali, J.S.; Hussain, A. Synthesis, characterization, applications, and challenges of iron oxide nanoparticles. Nanotechnol. Sci. Appl. 2016, 9, 49–67. [Google Scholar] [CrossRef] [Green Version]
- Massart, R. Preparation of aqueous magnetic liquids in alkaline and acidic media. IEEE Trans. Magn. 1981, 17, 1247–1248. [Google Scholar] [CrossRef]
- Fadli, A.; Komalasari; Adnan, A.; Iwantono; Rahimah; Addabsi, A.S. Synthesis of Magnetite Nanoparticles via Co-precipitation Method. IOP Conf. Ser. Mater. Sci. Eng. 2019, 622, 012013. [Google Scholar] [CrossRef]
- Rahmayanti, M. Synthesis of Magnetite Nanoparticles Using Reverse Co-precipitation Method with NH4OH as Precipitating Agent and Its Stability Test at Various pH. Nat. Sci. J. Sci. Technol. 2020, 9, 54–58. [Google Scholar] [CrossRef]
- Rashid, H.; Mansoor, M.A.; Haider, B.; Nasir, R.; Hamid, S.B.A.; Abdulrahman, A. Synthesis and characterization of magnetite nano particles with high selectivity using in-situ precipitation method. Sep. Sci. Technol. 2020, 55, 1207–1215. [Google Scholar] [CrossRef] [Green Version]
- Askaripour, H.; Vossoughi, M.; Khajeh, K.; Alemzadeh, I. Magnetite nanoparticle as a support for stabilization of chondroitinase ABCI. Artif. Cells Nanomed. Biotechnol. 2019, 47, 2721–2728. [Google Scholar] [CrossRef] [PubMed]
- Mascolo, M.C.; Pei, Y.; Ring, T.A. Room Temperature Co-Precipitation Synthesis of Magnetite Nanoparticles in a Large pH Window with Different Bases. Materials 2013, 6, 5549–5567. [Google Scholar] [CrossRef] [Green Version]
- Sugimoto, T.; Matijević, E. Formation of uniform spherical magnetite particles by crystallization from ferrous hydroxide gels. J. Colloid Interface Sci. 1980, 74, 227–243. [Google Scholar] [CrossRef]
- Vereda, F.; de Vicente, J.; Hidalgo-Álvarez, R. Influence of a Magnetic Field on the Formation of Magnetite Particles via Two Precipitation Methods. Langmuir 2007, 23, 3581–3589. [Google Scholar] [CrossRef]
- Vereda, F.; de Vicente, J.; Hidalgo-Alvarez, R. Oxidation of ferrous hydroxides with nitrate: A versatile method for the preparation of magnetic colloidal particles. J. Colloid Interface Sci. 2013, 392, 50–56. [Google Scholar] [CrossRef]
- Zurgil, N.; Shafran, Y.; Afrimzon, E.; Fixler, D.; Shainberg, A.; Deutsch, M. Concomitant real-time monitoring of intracellular reactive oxygen species and mitochondrial membrane potential in individual living promonocytic cells. J. Immunol. Methods 2006, 316, 27–41. [Google Scholar] [CrossRef]
- Yu, J.; Zhang, T.; Xu, H.; Dong, X.; Cai, Y.; Pan, Y.; Cao, C. Thermostable iron oxide nanoparticle synthesis within recombinant ferritins from the hyperthermophile Pyrococcus yayanosii CH1. RSC Adv. 2019, 9, 39381–39393. [Google Scholar] [CrossRef] [Green Version]
- Jung, E.; Kim, S.-W.; Cho, A.; Kim, Y.-J.; Jeong, G.-J.; Kim, J.; Bhang, S.H.; Yu, T. Synthesis of Sub 3 nm-Sized Uniform Magnetite Nanoparticles Using Reverse Micelle Method for Biomedical Application. Materials 2019, 12, 3850. [Google Scholar] [CrossRef] [Green Version]
- Guo, Y.; Chen, B.; Zhao, Y.; Yang, T. Fabrication of the magnetic mesoporous silica Fe-MCM-41-A as efficient adsorbent: Performance, kinetics and mechanism. Sci. Rep. 2021, 11, 2612. [Google Scholar] [CrossRef] [PubMed]
- Salvador, M.; Gutiérrez, G.; Noriega, S.; Moyano, A.; Blanco-López, M.C.; Matos, M. Microemulsion Synthesis of Superparamagnetic Nanoparticles for Bioapplications. Int. J. Mol. Sci. 2021, 22, 427. [Google Scholar] [CrossRef] [PubMed]
- Meldrum, F.C.; Heywood, B.R.; Mann, S. Magnetoferritin: In Vitro Synthesis of a Novel Magnetic Protein. Science 1992, 257, 522–523. [Google Scholar] [CrossRef] [PubMed]
- Xue, L.; Deng, D.; Sun, J. Magnetoferritin: Process, Prospects, and Their Biomedical Applications. Int. J. Mol. Sci. 2019, 20, 2426. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Uchida, M.; Qazi, S.; Edwards, E.; Douglas, T. Use of Protein Cages as a Template for Confined Synthesis of Inorganic and Organic Nanoparticles. Methods Mol. Biol. 2014, 1252, 17–25. [Google Scholar] [CrossRef]
- Uchida, M.; Kang, S.; Reichhardt, C.; Harlen, K.; Douglas, T. The ferritin superfamily: Supramolecular templates for materials synthesis. Biochim. Biophys. Acta (BBA) Gen. Subj. 2010, 1800, 834–845. [Google Scholar] [CrossRef] [Green Version]
- Gomez, N.T.; Nava, O.; Argueta-Figueroa, L.; García-Contreras, R.; Baeza-Barrera, A.; Vilchis-Nestor, A.R. Shape Tuning of Magnetite Nanoparticles Obtained by Hydrothermal Synthesis: Effect of Temperature. J. Nanomater. 2019, 2019, 7921273. [Google Scholar] [CrossRef] [Green Version]
- Cursaru, L.M.; Piticescu, R.M.; Dragut, D.V.; Morel, R.; Thébault, C.; Carrière, M.; Joisten, H.; Dieny, B. One-Step Soft Chemical Synthesis of Magnetite Nanoparticles under Inert Gas Atmosphere. Magnetic Properties and In Vitro Study. Nanomaterials 2020, 10, 1500. [Google Scholar] [CrossRef]
- Kumar, P.; Khanduri, H.; Pathak, S.; Singh, A.; Basheed, G.A.; Pant, R.P. Temperature selectivity for single phase hydrothermal synthesis of PEG-400 coated magnetite nanoparticles. Dalton Trans. 2020, 49, 8672–8683. [Google Scholar] [CrossRef]
- Wan, J.; Cai, W.; Meng, X.; Liu, E. Monodisperse water-soluble magnetite nanoparticles prepared by polyol process for high-performance magnetic resonance imaging. Chem. Commun. 2007, 47, 5004–5006. [Google Scholar] [CrossRef]
- Oh, A.H.; Park, H.-Y.; Jung, Y.-G.; Choi, S.-C.; An, G.S. Synthesis of Fe3O4 nanoparticles of various size via the polyol method. Ceram. Int. 2020, 46, 10723–10728. [Google Scholar] [CrossRef]
- Hachani, R.; Lowdell, M.; Birchall, M.; Hervault, A.; Mertz, D.; Begin-Colin, S.; Thanh, N.T.K. Polyol synthesis, functionalisation, and biocompatibility studies of superparamagnetic iron oxide nanoparticles as potential MRI contrast agents. Nanoscale 2016, 8, 3278–3287. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ansari, S.A.M.K.; Ficiarà, E.; Ruffinatti, F.A.; Stura, I.; Argenziano, M.; Abollino, O.; Cavalli, R.; Guiot, C.; D’Agata, F. Magnetic Iron Oxide Nanoparticles: Synthesis, Characterization and Functionalization for Biomedical Applications in the Central Nervous System. Materials 2019, 12, 465. [Google Scholar] [CrossRef] [Green Version]
- Shaker, S.; Zafarian, S.; Chakra, C.H.S.; Rao, K.V. Preparation and characterization of magnetite nanoparticles by sol-gel method for water treatment. Int. J. Innov. Res. Sci. Eng. Technol. 2013, 2, 2969–2973. [Google Scholar]
- Takai, Z.I.; Mustafa, M.K.; Asman, S.; Sekak, K.A. Preparation and Characterization of Magnetite (Fe3O4) nanoparticles By Sol-Gel Method. Int. J. Nanoelectron. Mater. 2019, 12, 37–46. [Google Scholar]
- De Freitas, J.C.; Branco, R.M.; Lisboa, I.G.O.; Da Costa, T.P.; Campos, M.G.N.; Junior, M.J.; Marques, R.F.C. Magnetic Nanoparticles Obtained by Homogeneous Coprecipitation Sonochemically Assisted. Mater. Res. 2015, 18, 220–224. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Nkurikiyimfura, I.; Pan, Z. Sonochemical Synthesis of Magnetic Nanoparticles. Chem. Eng. Commun. 2015, 202, 616–621. [Google Scholar] [CrossRef]
- Fuentes-García, J.A.; Alavarse, A.C.; Maldonado, A.C.M.; Toro-Córdova, A.; Ibarra, M.R.; Goya, G.F. Simple Sonochemical Method to Optimize the Heating Efficiency of Magnetic Nanoparticles for Magnetic Fluid Hyperthermia. ACS Omega 2020, 5, 26357–26364. [Google Scholar] [CrossRef]
- Samrot, A.V.; Sahithya, C.S.; A, J.S.; Purayil, S.K.; Ponnaiah, P. A review on synthesis, characterization and potential biological applications of superparamagnetic iron oxide nanoparticles. Curr. Res. Green Sustain. Chem. 2021, 4, 100042. [Google Scholar] [CrossRef]
- Yew, Y.P.; Shameli, K.; Miyake, M.; Ahmad Khairudin, N.B.B.; Mohamad, S.E.B.; Naiki, T.; Lee, K.X. Green biosynthesis of superparamagnetic magnetite Fe3O4 nanoparticles and biomedical applications in targeted anticancer drug delivery system: A review. Arab. J. Chem. 2020, 13, 2287–2308. [Google Scholar] [CrossRef]
- Blakemore, R. Magnetotactic bacteria. Science 1975, 190, 377–379. [Google Scholar] [CrossRef] [PubMed]
- Gareev, K.; Grouzdev, D.; Kharitonskii, P.; Kosterov, A.; Koziaeva, V.; Sergienko, E.; Shevtsov, M. Magnetotactic Bacteria and Magnetosomes: Basic Properties and Applications. Magnetochemistry 2021, 7, 86. [Google Scholar] [CrossRef]
- Alphandéry, E. Applications of Magnetosomes Synthesized by Magnetotactic Bacteria in Medicine. Front. Bioeng. Biotechnol. 2014, 2, 5. [Google Scholar] [CrossRef] [PubMed]
- Papell, S.S. Low Viscosity Magnetic Fluid Obtained by the Colloidal Suspension of Magnetic Particles. U.S. Patent 3,215,572, 2 November 1965. [Google Scholar]
- Genc, S.; Derin, B. Synthesis and rheology of ferrofluids: A review. Curr. Opin. Chem. Eng. 2014, 3, 118–124. [Google Scholar] [CrossRef]
- Hong, R.; Pan, T.; Li, H. Microwave synthesis of magnetic Fe3O4 nanoparticles used as a precursor of nanocomposites and ferrofluids. J. Magn. Magn. Mater. 2006, 303, 60–68. [Google Scholar] [CrossRef]
- Laokul, P.; Amornkitbamrung, V.; Seraphin, S.; Maensiri, S. Characterization and magnetic properties of nanocrystalline CuFe2O4, NiFe2O4, ZnFe2O4 powders prepared by the Aloe vera extract solution. Curr. Appl. Phys. 2011, 11, 101–108. [Google Scholar] [CrossRef]
- Andrade, L.; Valente, M.A.; Ferreira, J.M.; Fabris, J.D. Preparation of size-controlled nanoparticles of magnetite. J. Magn. Magn. Mater. 2012, 324, 1753–1757. [Google Scholar] [CrossRef] [Green Version]
- Pislaru-Danescu, L.; Morega, A.; Telipan, G.; Stoica, V. Nanoparticles of ferrofluid Fe3O4 synthetised by coprecipitation method used in microactuation process. Optoelectron. Adv. Mater. Rap. Commun. 2010, 4, 1182–1186. [Google Scholar]
- Chaitoglou, S.; Sanaee, M.R.; Aguayo, N.A.; Bertran, E. Arc-Discharge Synthesis of Iron Encapsulated in Carbon Nanoparticles for Biomedical Applications. J. Nanomater. 2014, 2014, 178524. [Google Scholar] [CrossRef]
- Darvina, Y.; Yulfriska, N.; Rifai, H.; Dwiridal, L.; Ramli, R. Synthesis of magnetite nanoparticles from iron sand by ball-milling. J. Phys. Conf. Ser. 2019, 1185, 012017. [Google Scholar] [CrossRef]
- López-López, M.; Durán, J.; Delgado, A.; González-Caballero, F. Stability and magnetic characterization of oleate-covered magnetite ferrofluids in different nonpolar carriers. J. Colloid Interface Sci. 2005, 291, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Arco, L.R.; Lopez-Lopez, M.T.; Caballero, F.G.; Durán, J.D. Steric repulsion as a way to achieve the required stability for the preparation of ionic liquid-based ferrofluids. J. Colloid Interface Sci. 2011, 357, 252–254. [Google Scholar] [CrossRef] [PubMed]
- Fauconnier, N.; Bée, A.; Roger, J.; Pons, J. Synthesis of aqueous magnetic liquids by surface complexation of maghemite nanoparticles. J. Mol. Liq. 1999, 83, 233–242. [Google Scholar] [CrossRef]
- Kroell, M.; Pridoehl, M.; Zimmermann, G.; Pop, L.; Odenbach, S.; Hartwig, A. Magnetic and rheological characterization of novel ferrofluids. J. Magn. Magn. Mater. 2004, 289, 21–24. [Google Scholar] [CrossRef]
- Donadel, K.; Felisberto, M.D.; Fávere, V.T.; Rigoni, M.; Batistela, N.J.; Laranjeira, M.C. Synthesis and characterization of the iron oxide magnetic particles coated with chitosan biopolymer. Mater. Sci. Eng. C 2008, 28, 509–514. [Google Scholar] [CrossRef]
- Khosroshahi, M.; Ghazanfari, L. Preparation and rheological studies of uncoated and PVA-coated magnetite nanofluid. J. Magn. Magn. Mater. 2012, 324, 4143–4146. [Google Scholar] [CrossRef]
- Brullot, W.; Reddy, N.; Wouters, J.; Valev, V.; Goderis, B.; Vermant, J.; Verbiest, T. Versatile ferrofluids based on polyethylene glycol coated iron oxide nanoparticles. J. Magn. Magn. Mater. 2012, 324, 1919–1925. [Google Scholar] [CrossRef]
- Pathak, S.; Jain, K.; Kumar, P.; Wang, X.; Pant, R. Improved thermal performance of annular fin-shell tube storage system using magnetic fluid. Appl. Energy 2019, 239, 1524–1535. [Google Scholar] [CrossRef]
- Imran, M.; Alam, M.; Hussain, S.; Abutaleb, A.; Aziz, A.; Chandan, M.R.; Irshad, K.; Al-Hagri, A.M.A.; Bakather, O.Y.; Khan, A. Colloidal Fe3O4 nanoparticles-based oil blend ferro-nanofluid for heat transfer application. Eur. Phys. J. Plus 2021, 136, 752. [Google Scholar] [CrossRef]
- Elsaidy, A.; Vallejo, J.P.; Salgueiriño, V.; Lugo, L. Tuning the thermal properties of aqueous nanofluids by taking advantage of size-customized clusters of iron oxide nanoparticles. J. Mol. Liq. 2021, 344, 117727. [Google Scholar] [CrossRef]
- Mohapatra, D.K.; Zubarev, A.; Safronov, A.; Philip, J. Reconfiguring nanostructures in magnetic fluids using pH and magnetic stimulus for tuning optical properties. J. Magn. Magn. Mater. 2021, 539, 168351. [Google Scholar] [CrossRef]
- Scherer, C.; Neto, A.M.F. Ferrofluids: Properties and applications. Braz. J. Phys. 2005, 35, 718–727. [Google Scholar] [CrossRef]
- Kole, M.; Khandekar, S. Engineering applications of ferrofluids: A review. J. Magn. Magn. Mater. 2021, 537, 168222. [Google Scholar] [CrossRef]
- Ge, J.; Hu, Y.; Yin, Y. Highly Tunable Superparamagnetic Colloidal Photonic Crystals. Angew. Chem. Int. Ed. 2007, 46, 7428–7431. [Google Scholar] [CrossRef]
- Yang, P.; Li, H.; Zhang, S.; Chen, L.; Zhou, H.; Tang, R.; Zhou, T.; Bao, F.; Zhang, Q.; He, L.; et al. Gram-scale synthesis of superparamagnetic Fe3O4nanocrystal clusters with long-term charge stability for highly stable magnetically responsive photonic crystals. Nanoscale 2016, 8, 19036–19042. [Google Scholar] [CrossRef]
- Kostopoulou, A.; Tsiaoussis, I.; Lappas, A. Magnetic iron oxide nanoclusters with tunable optical response. Photon.-Nanostruct. Fundam. Appl. 2011, 9, 201–206. [Google Scholar] [CrossRef]
- He, L.; Wang, M.; Ge, J.; Yin, Y. Magnetic Assembly Route to Colloidal Responsive Photonic Nanostructures. Acc. Chem. Res. 2012, 45, 1431–1440. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.; Li, Q.; Zheng, A.; Li, X.; Pan, Z.; Jiang, J.; Zhang, L.; Hong, R.; Zhuang, L. Superparamagnetic iron oxide nanoparticles for full-color photonic materials with tunable properties. Results Phys. 2019, 14, 102366. [Google Scholar] [CrossRef]
- Zhang, D.; Xu, J.; Sun, H. Toward High Efficiency Organic Light-Emitting Diodes: Role of Nanoparticles. Adv. Opt. Mater. 2021, 9, 2001710. [Google Scholar] [CrossRef]
- Zhang, D.; Tian, Y.; Zou, S.; Tang, J.; Li, Y. Highly efficient organic light-emitting devices based on multifunctional nanoparticles. Opt. Lett. 2019, 44, 2462–2465. [Google Scholar] [CrossRef]
- Lian, H.; Tang, Z.; Guo, H.; Zhong, Z.; Wu, J.; Dong, Q.; Zhu, F.; Wei, B.; Wong, W.-Y. Magnetic nanoparticles/PEDOT:PSS composite hole-injection layer for efficient organic light-emitting diodes. J. Mater. Chem. C 2018, 6, 4903–4911. [Google Scholar] [CrossRef]
- Chen, Y.; Sun, W.; Zhang, Y.; Liu, G.; Luo, Y.; Dong, J.; Zhong, Y.; Zhu, W.; Yu, J.; Chen, Z. Magnetic Nanoparticles Functionalized Few-Mode-Fiber-Based Plasmonic Vector Magnetometer. Nanomaterials 2019, 9, 785. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, Y.; Liu, X.; Lv, R.-Q.; Zhang, Y.-N.; Wang, Q. Review on Optical Fiber Sensors Based on the Refractive Index Tunability of Ferrofluid. J. Light. Technol. 2017, 35, 3406–3412. [Google Scholar] [CrossRef]
- Cennamo, N.; Arcadio, F.; Marletta, V.; Baglio, S.; Zeni, L.; Ando, B. A Magnetic Field Sensor Based on SPR-POF Platforms and Ferrofluids. IEEE Trans. Instrum. Meas. 2020, 70, 9504010. [Google Scholar] [CrossRef]
- Zheng, J.; Dong, X.; Zu, P.; Ji, J.; Su, H.; Shum, P.P. Intensity-modulated magnetic field sensor based on magnetic fluid and optical fiber gratings. Appl. Phys. Lett. 2013, 103, 183511. [Google Scholar] [CrossRef]
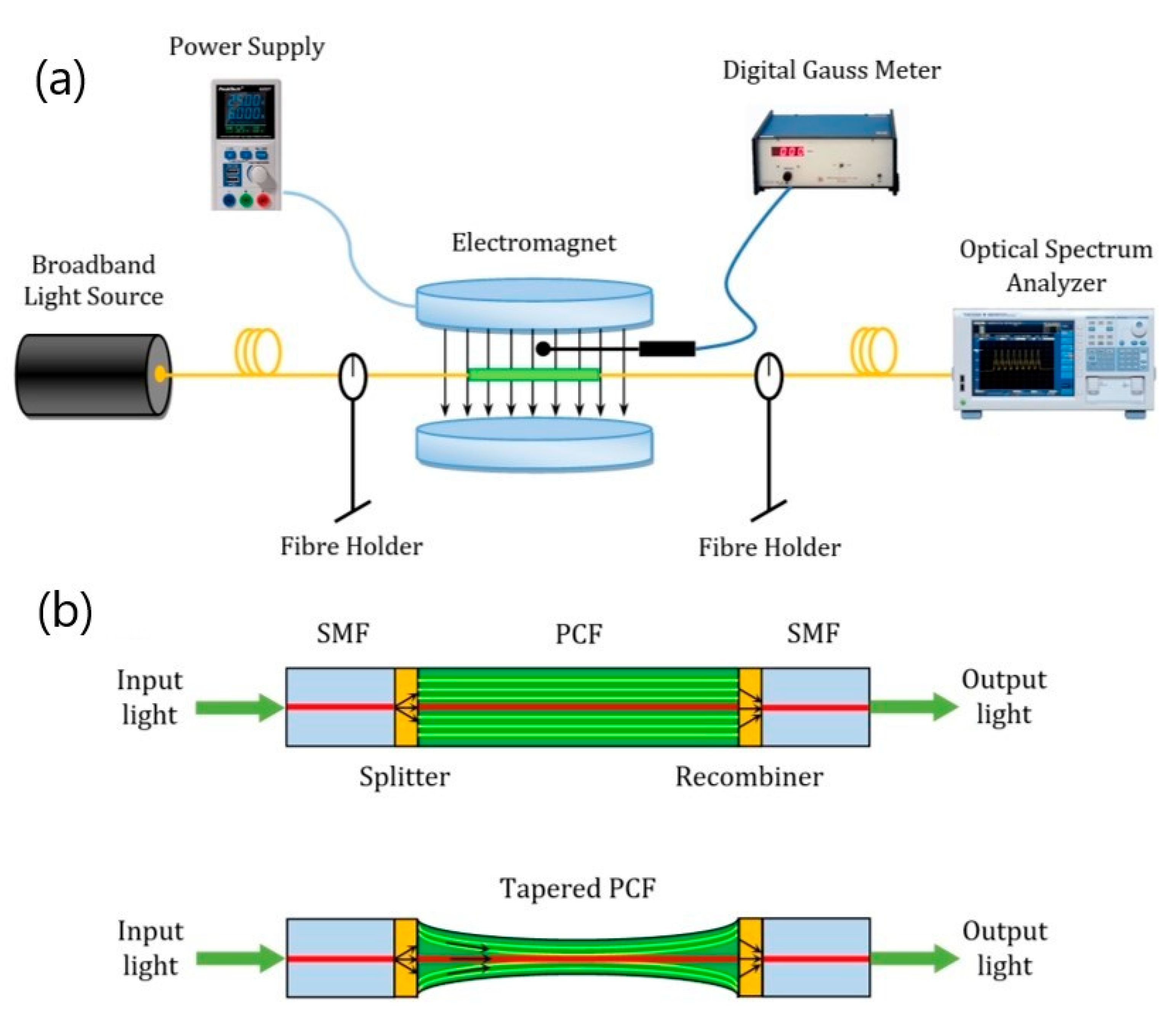
- Taghizadeh, M.; Bozorgzadeh, F.; Ghorbani, M. Designing magnetic field sensor based on tapered photonic crystal fibre assisted by a ferrofluid. Sci. Rep. 2021, 11, 14325. [Google Scholar] [CrossRef]
- Li, Y.; Pu, S.; Hao, Z.; Yan, S.; Zhang, Y.; Lahoubi, M. Vector magnetic field sensor based on U-bent single-mode fiber and magnetic fluid. Opt. Express 2021, 29, 5236–5246. [Google Scholar] [CrossRef]
- Brzezicki, M. A Systematic Review of the Most Recent Concepts in Smart Windows Technologies with a Focus on Electrochromics. Sustainability 2021, 13, 9604. [Google Scholar] [CrossRef]
- Heiz, B.P.V.; Pan, Z.; Su, L.; Le, S.T.; Wondraczek, L. A Large-Area Smart Window with Tunable Shading and Solar-Thermal Harvesting Ability Based on Remote Switching of a Magneto-Active Liquid. Adv. Sustain. Syst. 2018, 2, 1700140. [Google Scholar] [CrossRef] [Green Version]
- Seo, Y.J.; Lee, H.G.; Yang, J.S.; Jeong, H.; Han, J.; Kim, J.-H.; Koo, H.-J.; Yoon, H. Manipulation of light transmission from stable magnetic microrods formed by the alignment of magnetic nanoparticles. RSC Adv. 2021, 11, 2390–2396. [Google Scholar] [CrossRef]
- Patrick, P.S.; Bogart, L.K.; Macdonald, T.J.; Southern, P.; Powell, M.J.; Zaw-Thin, M.; Voelcker, N.H.; Parkin, I.P.; Pankhurst, Q.A.; Lythgoe, M.F.; et al. Surface radio-mineralisation mediates chelate-free radiolabelling of iron oxide nanoparticles. Chem. Sci. 2019, 10, 2592–2597. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bao, Y.; Sherwood, J.A.; Sun, Z. Magnetic iron oxide nanoparticles asT1contrast agents for magnetic resonance imaging. J. Mater. Chem. C 2018, 6, 1280–1290. [Google Scholar] [CrossRef]
- Nguyen, K.-L.; Yoshida, T.; Kathuria-Prakash, N.; Zaki, I.H.; Varallyay, C.G.; Semple, S.I.; Saouaf, R.; Rigsby, C.; Stoumpos, S.; Whitehead, K.K.; et al. Multicenter Safety and Practice for Off-Label Diagnostic Use of Ferumoxytol in MRI. Radiology 2019, 293, 554–564. [Google Scholar] [CrossRef] [PubMed]
- Marashdeh, M.; Ababneh, B.; Lemine, O.; Alsadig, A.; Omri, K.; El Mir, L.; Sulieman, A.; Mattar, E. The significant effect of size and concentrations of iron oxide nanoparticles on magnetic resonance imaging contrast enhancement. Results Phys. 2019, 15, 102651. [Google Scholar] [CrossRef]
- Tao, C.; Zheng, Q.; An, L.; He, M.; Lin, J.; Tian, Q.; Yang, S. T1-Weight Magnetic Resonance Imaging Performances of Iron Oxide Nanoparticles Modified with a Natural Protein Macromolecule and an Artificial Macromolecule. Nanomaterials 2019, 9, 170. [Google Scholar] [CrossRef] [Green Version]
- Tao, C.; Chen, Y.; Wang, D.; Cai, Y.; Zheng, Q.; An, L.; Lin, J.; Tian, Q.; Yang, S. Macromolecules with Different Charges, Lengths, and Coordination Groups for the Coprecipitation Synthesis of Magnetic Iron Oxide Nanoparticles as T1 MRI Contrast Agents. Nanomaterials 2019, 9, 699. [Google Scholar] [CrossRef] [Green Version]
- Wei, H.; Bruns, O.T.; Kaul, M.G.; Hansen, E.C.; Barch, M.; Wisniowska, A.E.; Chen, O.; Chen, Y.; Li, N.; Okada, S.; et al. Exceedingly small iron oxide nanoparticles as positive MRI contrast agents. Proc. Natl. Acad. Sci. USA 2017, 114, 2325–2330. [Google Scholar] [CrossRef] [Green Version]
- Alipour, A.; Erdem, Z.S.; Utkur, M.; Sharma, V.K.; Algin, O.; Saritas, E.U.; Demir, H.V. A new class of cubic SPIONs as a dual-mode T1 and T2 contrast agent for MRI. Magn. Reson. Imaging 2018, 49, 16–24. [Google Scholar] [CrossRef] [Green Version]
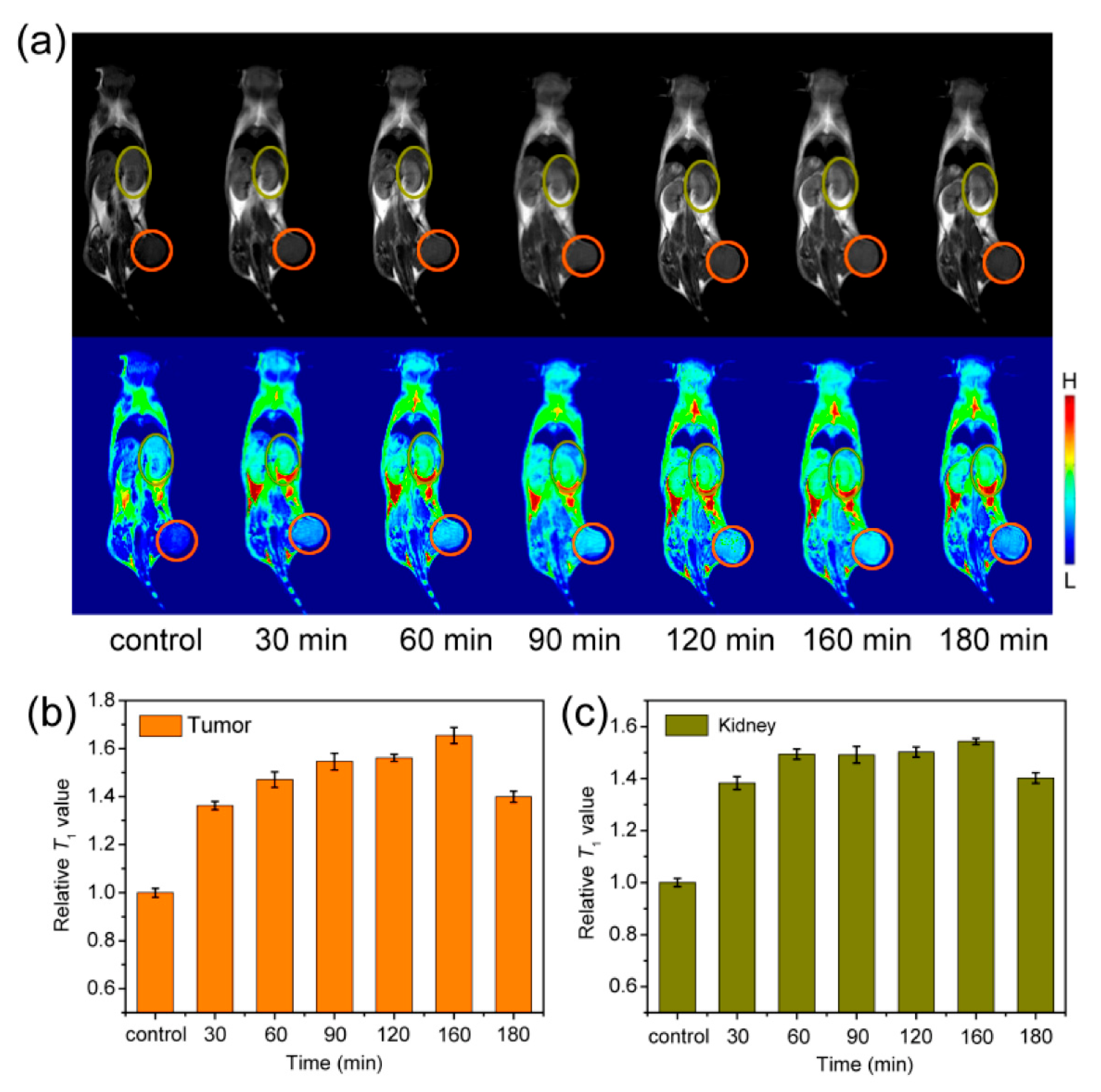
- Li, F.; Liang, Z.; Liu, J.; Sun, J.; Hu, X.; Zhao, M.; Liu, J.; Bai, R.; Kim, D.; Sun, X.; et al. Dynamically Reversible Iron Oxide Nanoparticle Assemblies for Targeted Amplification of T1-Weighted Magnetic Resonance Imaging of Tumors. Nano Lett. 2019, 19, 4213–4220. [Google Scholar] [CrossRef]
- Shen, Z.; Chen, T.; Ma, X.; Ren, W.; Zhou, Z.; Zhu, G.; Zhang, A.; Liu, Y.; Song, J.; Li, Z.; et al. Multifunctional Theranostic Nanoparticles Based on Exceedingly Small Magnetic Iron Oxide Nanoparticles for T1-Weighted Magnetic Resonance Imaging and Chemotherapy. ACS Nano 2017, 11, 10992–11004. [Google Scholar] [CrossRef]
- Vangijzegem, T.; Stanicki, D.; Boutry, S.; Paternoster, Q.; Elst, L.V.; Muller, R.N.; Laurent, S. VSION as high field MRI T1 contrast agent: Evidence of their potential as positive contrast agent for magnetic resonance angiography. Nanotechnology 2018, 29, 265103. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Li, L.; Zhang, D.; Gan, L.; Zhao, P.; Zhang, Y.; Zhang, Q.; Hua, M.; Jia, C. Gadolinium-doped carbon quantum dots loaded magnetite nanoparticles as a bimodal nanoprobe for both fluorescence and magnetic resonance imaging. Magn. Reson. Imaging 2020, 68, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Das, B.; Girigoswami, A.; Dutta, A.; Pal, P.; Dutta, J.; Dadhich, P.; Srivas, P.K.; Dhara, S. Carbon Nanodots Doped Super-paramagnetic Iron Oxide Nanoparticles for Multimodal Bioimaging and Osteochondral Tissue Regeneration via External Magnetic Actuation. ACS Biomater. Sci. Eng. 2019, 5, 3549–3560. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Shen, J.; Li, Y.; Wei, Z.; Cao, G.; Gai, Z.; Hong, K.; Banerjee, P.; Zhou, S. Magnetic iron oxide–fluorescent carbon dots integrated nanoparticles for dual-modal imaging, near-infrared light-responsive drug carrier and photothermal therapy. Biomater. Sci. 2014, 2, 915–923. [Google Scholar] [CrossRef] [PubMed]
- Tembhare, S.P.; Barai, D.P.; Bhanvase, B.A. Performance evaluation of nanofluids in solar thermal and solar photovoltaic systems: A comprehensive review. Renew. Sustain. Energy Rev. 2022, 153, 111738. [Google Scholar] [CrossRef]
- Sani, E.; Martina, M.; Salez, T.; Nakamae, S.; Dubois, E.; Peyre, V. Multifunctional Magnetic Nanocolloids for Hybrid Solar-Thermoelectric Energy Harvesting. Nanomaterials 2021, 11, 1031. [Google Scholar] [CrossRef]
- Grosu, Y.; Faik, A.; Ortega-Fernández, I.; D’Aguanno, B. Natural Magnetite for thermal energy storage: Excellent thermophysical properties, reversible latent heat transition and controlled thermal conductivity. Sol. Energy Mater. Sol. Cells 2017, 161, 170–176. [Google Scholar] [CrossRef]
- Kannan, U.; Giribabu, L.; Jammalamadaka, S.N. Demagnetization field driven charge transport in a TiO2 based dye sensitized solar cell. Sol. Energy 2019, 187, 281–289. [Google Scholar] [CrossRef]
- Cao, Y.; Hamidvand, S.; Bezaatpour, M.; Ebadollahi, M.; Ghaebi, H. Microporous foam, magnetic nanoparticles, and revolutionary tubes: Sophisticated combination of three solar energy materials in flat plate solar collectors. Sol. Energy Mater. Sol. Cells 2022, 235, 111464. [Google Scholar] [CrossRef]
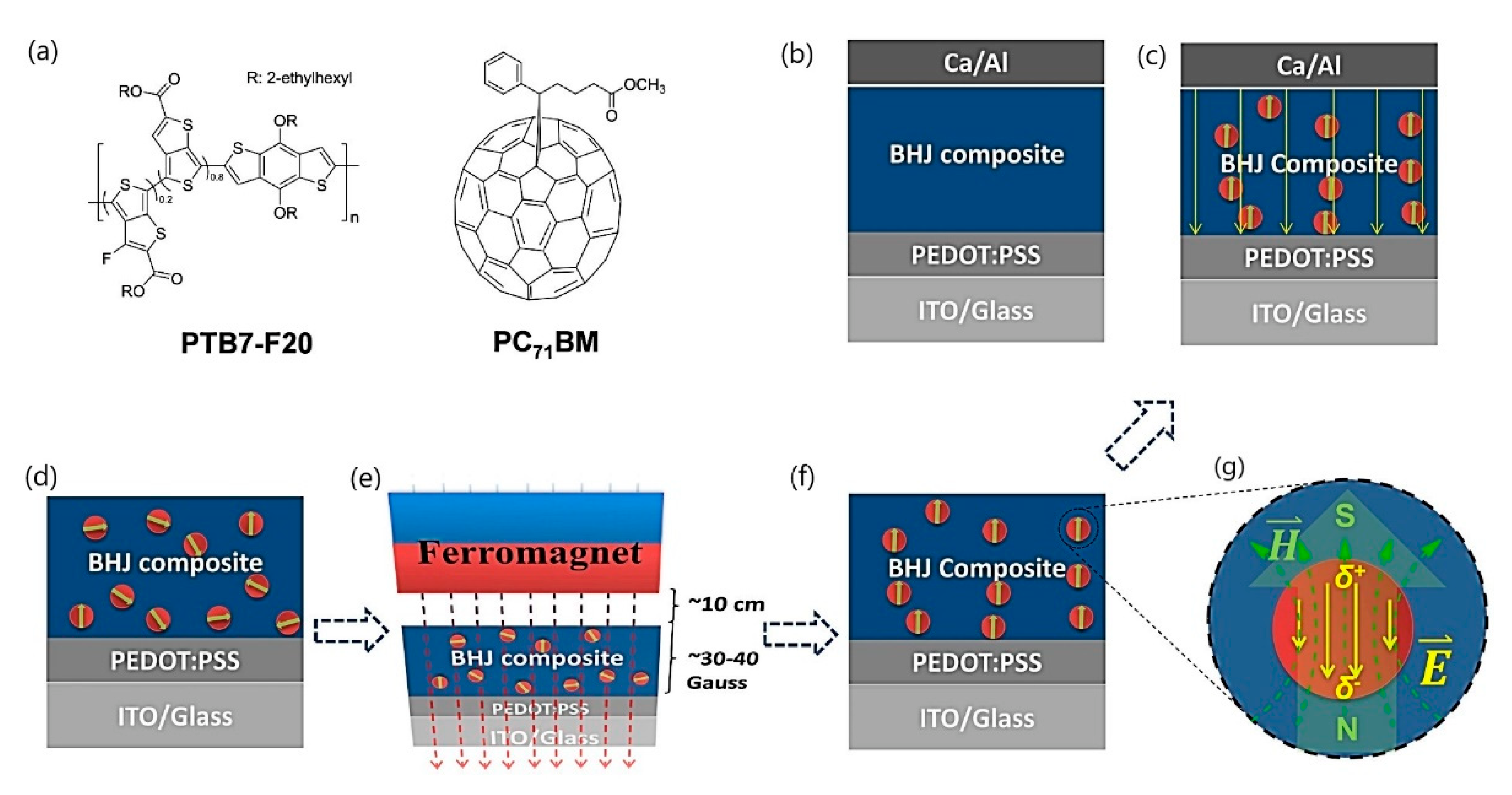
- Wang, K.; Yi, C.; Liu, C.; Hu, X.; Chuang, S.; Gong, X. Effects of Magnetic Nanoparticles and External Magnetostatic Field on the Bulk Heterojunction Polymer Solar Cells. Sci. Rep. 2015, 5, 9265. [Google Scholar] [CrossRef] [Green Version]
- Ansari, F.; Salavati-Niasari, M.; Amiri, O.; Mir, N.; Abdollahi Nejand, B.; Ahmadi, V. Magnetite as Inorganic Hole Transport Material for Lead Halide Perovskite-Based Solar Cells with Enhanced Stability. Ind. Eng. Chem. Res. 2020, 59, 743–750. [Google Scholar] [CrossRef]




| Properties | Magnetite |
|---|---|
| Molecular formula | Fe3O4 |
| Crystal structure | Cubic |
| Density (g/cm3 ) | 5.18 |
| Melting point (°C) | 1583–1597 |
| Boiling point (°C) | 2623 |
| Color | black |
| Hardness | 5.5 |
| Type of magnetism | ferrimagnetic |
| Curie temperature (K) | 580 |
| Ms at 300 K (A·m2/kg) | 92–100 |
| Magnetism (nanoparticles) | Superparamagnetic |
| Lustre | Metallic |
| Diaphaneity | Opaque |
| Crystal System | Isometric |
| Birefringence | Isotropic minerals have no birefringence |
| Refractive Index values | n = 2.42 |
| Method | Conditions | Advantages | Disadvantages | Challenges | |
|---|---|---|---|---|---|
| Chemical routes | Co-precipitation | Co-precipitation of ferrous and ferric salts in water with a strong basic solution at room temperature or higher | Simple, large quantity of nanoparticles | Poor morphology and non-stoichiometric magnetite | Size- and shape-controlled synthesis with reproducibility |
| Partial oxidation of ferrous hydroxide | Precipitation of amorphous ferrous hydroxide from a ferrous sulphate solution with subsequent aging at 90 °C in the presence of nitrate ion | Versatility and hydrophilic particles | Goethite formation and extensive nanoparticle agglomeration | High oxidation of ultrafine iron oxide nanoparticles and aggregation of nanoparticles | |
| Reaction in constrained environments | Precipitation of iron oxides using synthetic and biological reactors (apoferritin protein cages, micelles, mesoporous templates, and microemulsion) | Nanoparticles with uniform dimensions preventing oxidation and particle interaction | Complicated conditions | Purification of nanoparticles and scale-up procedures | |
| Hydrothermal or high-temperature reactions | Wet-chemical method of magnetite nanoparticles obtained using reactor or autoclave, with a pressure of >6000 Pa and a temperature of >200 °C | Nanoparticles of different shapes and morphologies with high crystallinity | High temperatures and pressures | Need expensive facilities | |
| Polyol method | High-temperature decomposition of ferric precursors in the polyol medium | Low cost, hydrolytic stability, and nanoparticles with controlled shape and size | Usage of toxic solvents | Thermal instability and flammability | |
| Sol–gel synthesis | Wet-chemical process based on hydrolysis and polycondensation of iron precursors with the formation of a “sol” and further drying (“gel” formation) | Monodispersity, good control of particle size, control of microstructure, desirable shape and length of the products, high purity, and good crystallinity | Long completion time and toxic organic solvents | Contamination of the product with the matrix component | |
| Sonochemical synthesis | Chemical reaction occurs due to the application of ultrasound irradiation, which causes acoustic cavitation in the aqueous solutions | Nanoparticles with high crystallinity, saturation magnetization, and narrow size distribution | Shape and size of nanoparticles obtained are difficult to control | Mechanism of reaction is still not well understood | |
| Biological routes | Bacterial | Precipitation of magnetite nanoparticles inside a bacterial magnetosome | Non-toxic, biocompatible, and unique crystal shape | Culturing microorganisms takes more time and complicated equipment | Limited knowledge about biomineralization process and time-consuming |
| Plants | Mixing precursor salts with green substrates that act as reducing and limiting agents | Non-toxic, biocompatible, and eco-friendly | Difficulty controlling size and properties | Limited knowledge about the mechanism of the process and time-consuming | |
| Application | Description | Challenges |
|---|---|---|
| Photonic materials | Superparamagnetic magnetite nanocrystals or nanoclusters with tunable sizes from 30 to 200 nm in water | NPs with controlled diameters and stoichiometric structures |
| Organic light-emitting diodes (OLEDs) | Gold-doped magnetite nanoparticles | Size of magnetite nanoparticles and aggregation of nanoparticles |
| Magnetic field sensors | Combination of different shape of optical fibers and optical grating with magnetic FFs | Long-term colloidal stability of FFs |
| “Smart” windows | Nanoscale magnetite nanoparticles from 50 to 100 nm used for shading and the optical properties of the window controlled through remote switching of magnetic field | Long-term colloidal stability of FFs |
| Magnetic resonance imaging | Exceedingly small superparamagnetic iron oxide NPs (up to ~10 nm) with various surface modifications | Reproducibility, scalability of synthesis, and functionalization of the surface for specifically targeting to different tissues and cells |
| Solar energy harvesting | Sensitized solar cells based on MNPs | Nanoparticle sedimentation and aggregation, cost of nanomaterials, and toxicity |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dudchenko, N.; Pawar, S.; Perelshtein, I.; Fixler, D. Magnetite Nanoparticles: Synthesis and Applications in Optics and Nanophotonics. Materials 2022, 15, 2601. https://doi.org/10.3390/ma15072601
Dudchenko N, Pawar S, Perelshtein I, Fixler D. Magnetite Nanoparticles: Synthesis and Applications in Optics and Nanophotonics. Materials. 2022; 15(7):2601. https://doi.org/10.3390/ma15072601
Chicago/Turabian StyleDudchenko, Nataliia, Shweta Pawar, Ilana Perelshtein, and Dror Fixler. 2022. "Magnetite Nanoparticles: Synthesis and Applications in Optics and Nanophotonics" Materials 15, no. 7: 2601. https://doi.org/10.3390/ma15072601
APA StyleDudchenko, N., Pawar, S., Perelshtein, I., & Fixler, D. (2022). Magnetite Nanoparticles: Synthesis and Applications in Optics and Nanophotonics. Materials, 15(7), 2601. https://doi.org/10.3390/ma15072601







