Poly(vinyl alcohol)/Plant Extracts Films: Preparation, Surface Characterization and Antibacterial Studies against Gram Positive and Gram Negative Bacteria
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Obtainment of Plant Extracts
2.3. Films Preparation
2.4. Determination of Total Polyphenol and Flavonoids Content
2.5. UV-VIS Spectra
2.6. Fourier-Transform Infrared Spectroscopy Analysis
2.7. Contact Angle and Surface Free Energy
2.8. Profilometry Analysis
2.9. Raman Microscopy Analysis
2.10. Mechanical Properties
2.11. Scanning Electron Microscopy (SEM)
2.12. Antibacterial Analysis
2.13. Cytotoxicity Assay
2.13.1. Cell Culture Conditions
2.13.2. MTT Assay
3. Results and Discussion
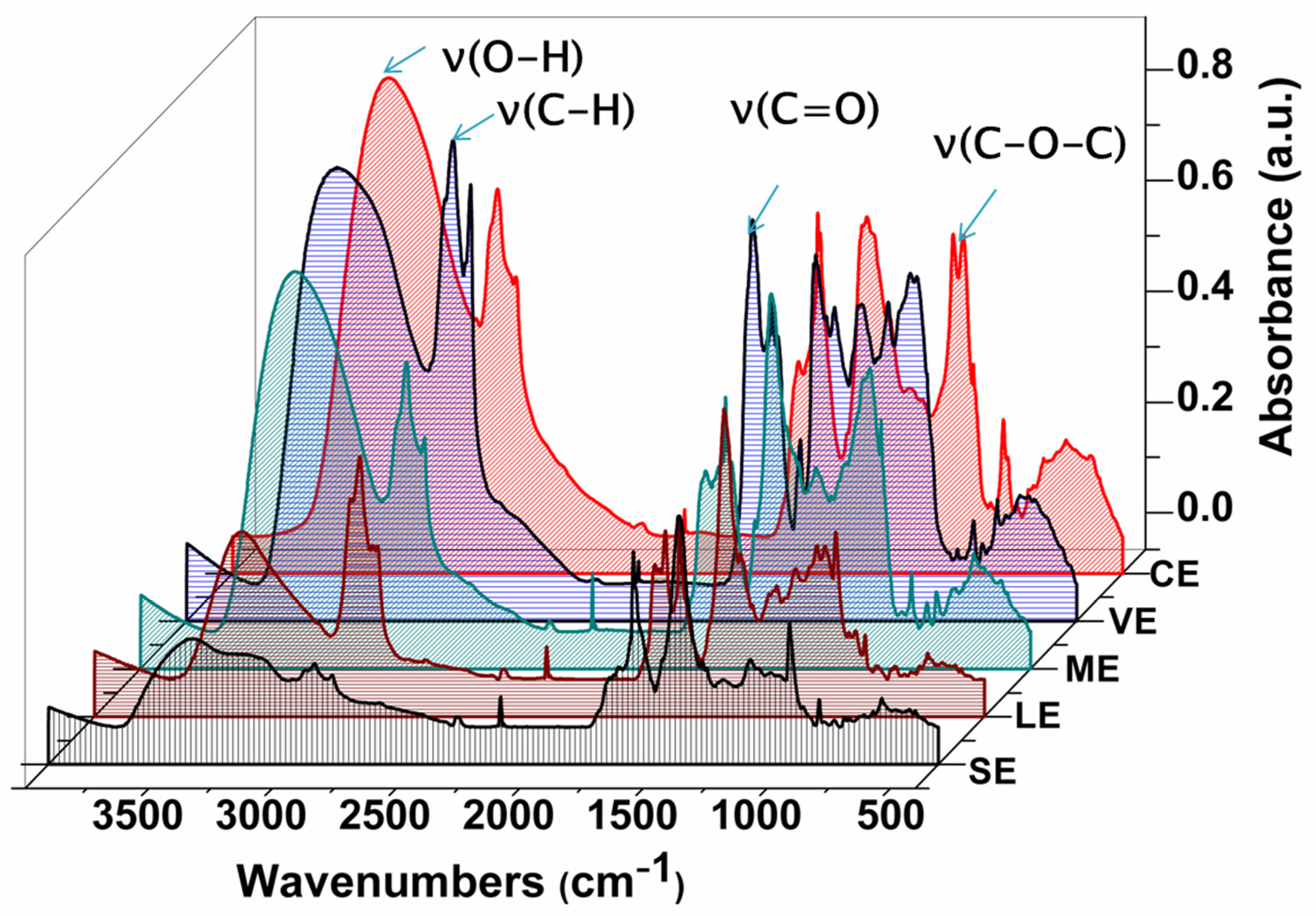
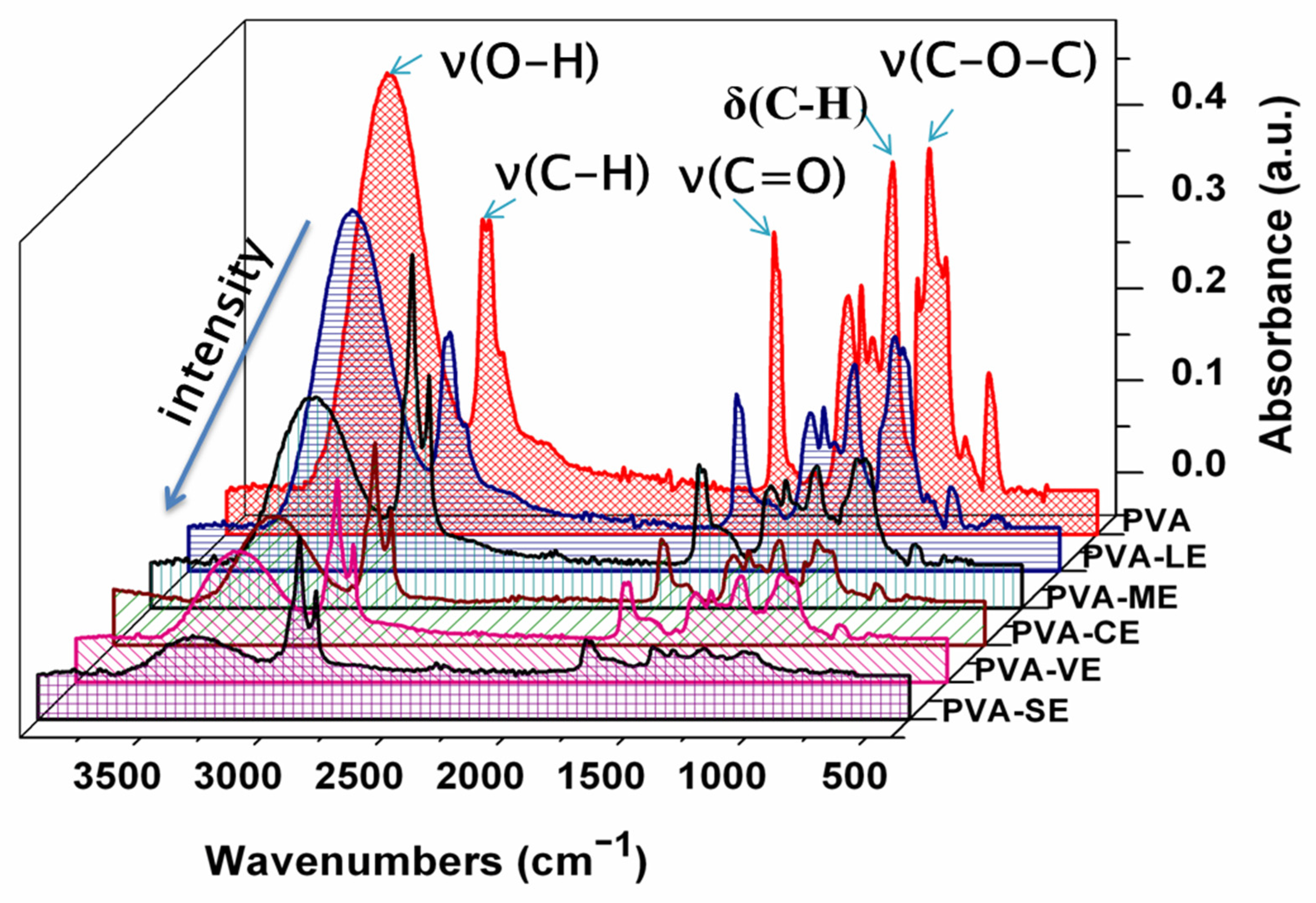
3.1. ATR-FTIR Analysis
3.2. UV-Vis Spectroscopy and the Phenols and Flavonoids Content of Extracts
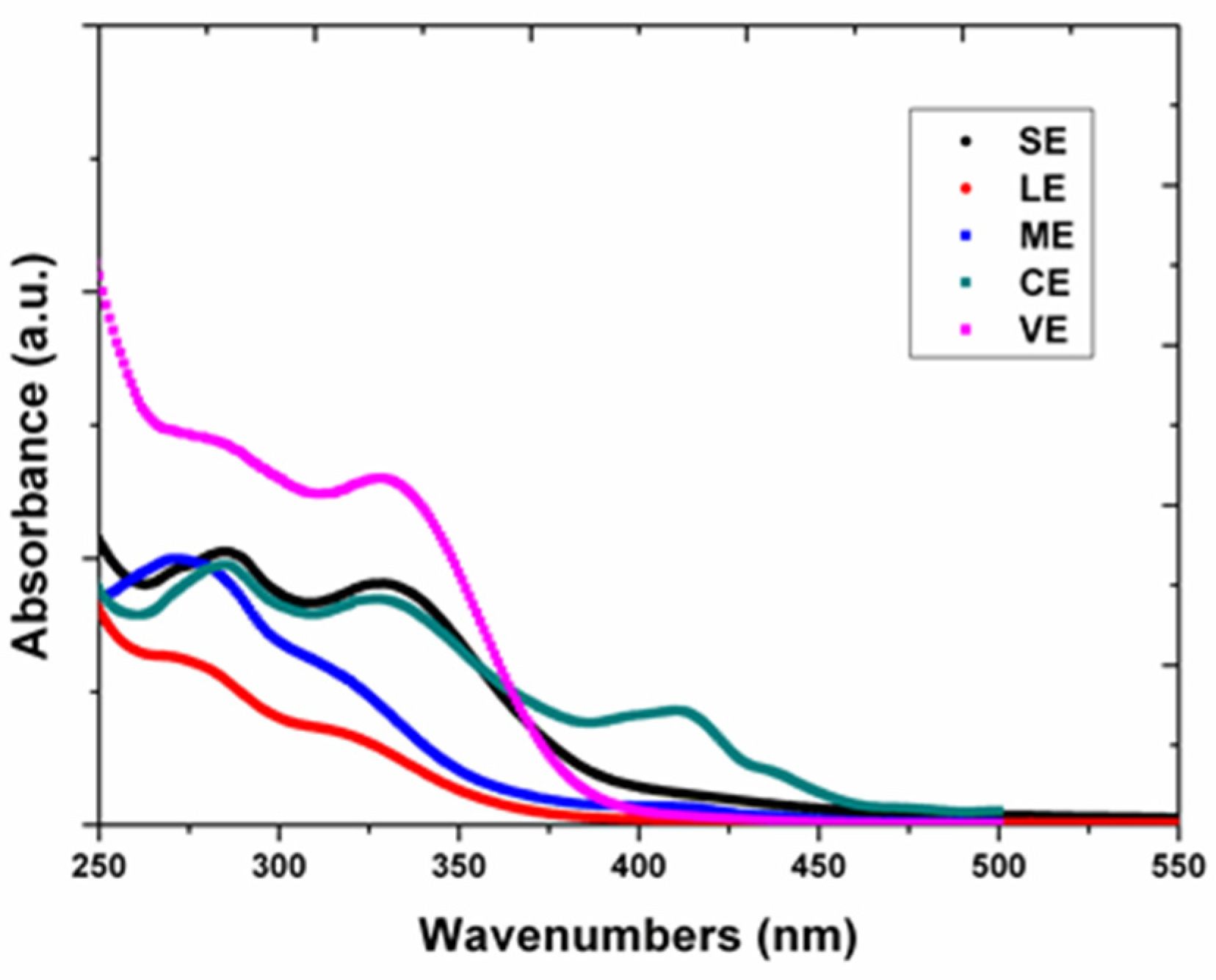
3.2.1. UV-Vis Spectroscopy Analysis
3.2.2. Determination of Total Phenols and Flavonoids Content of Extracts
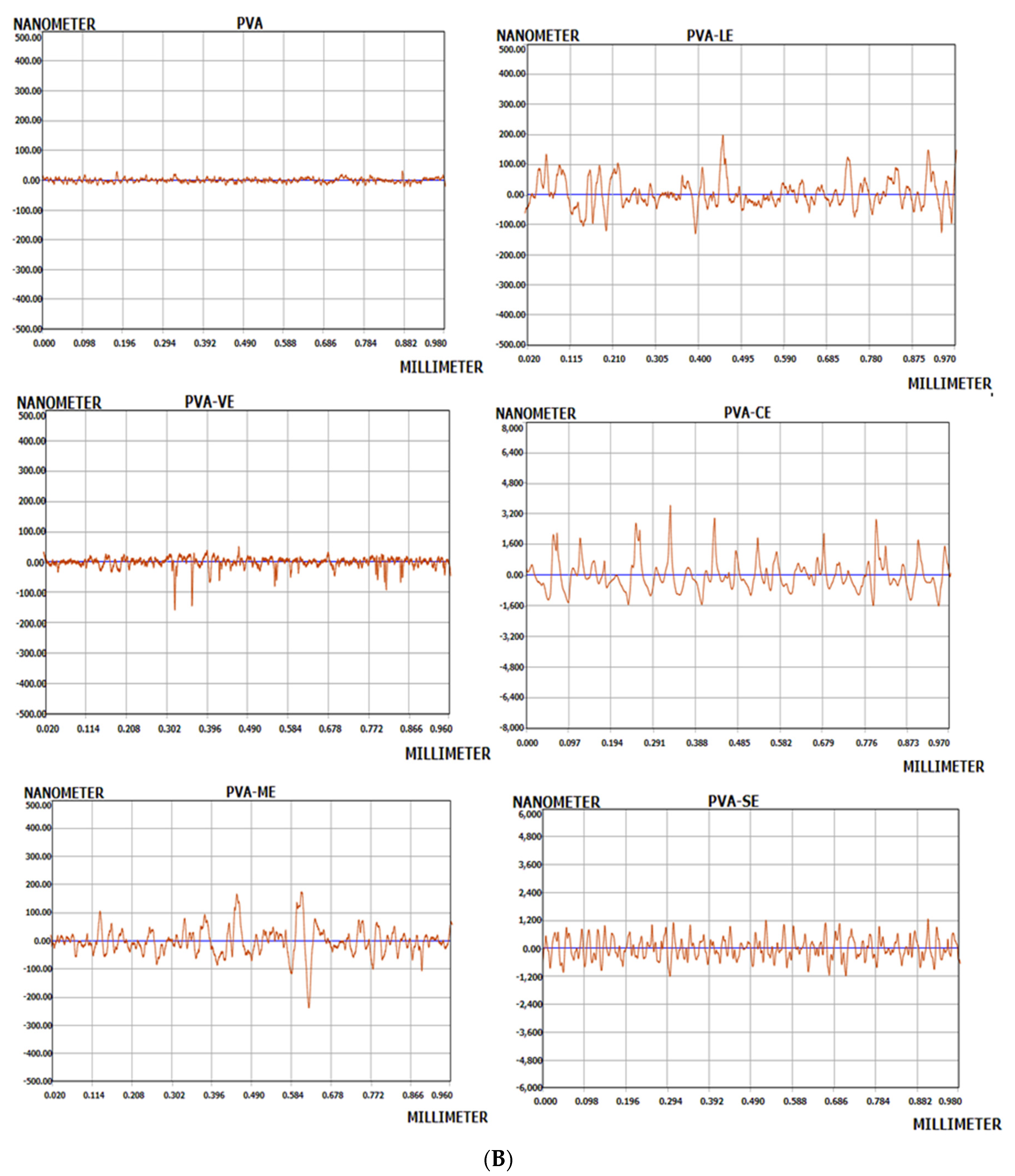
3.3. Surface Morphology and Surface Roughness Analysis of Films
3.4. Surface Free Energy Analysis of Films
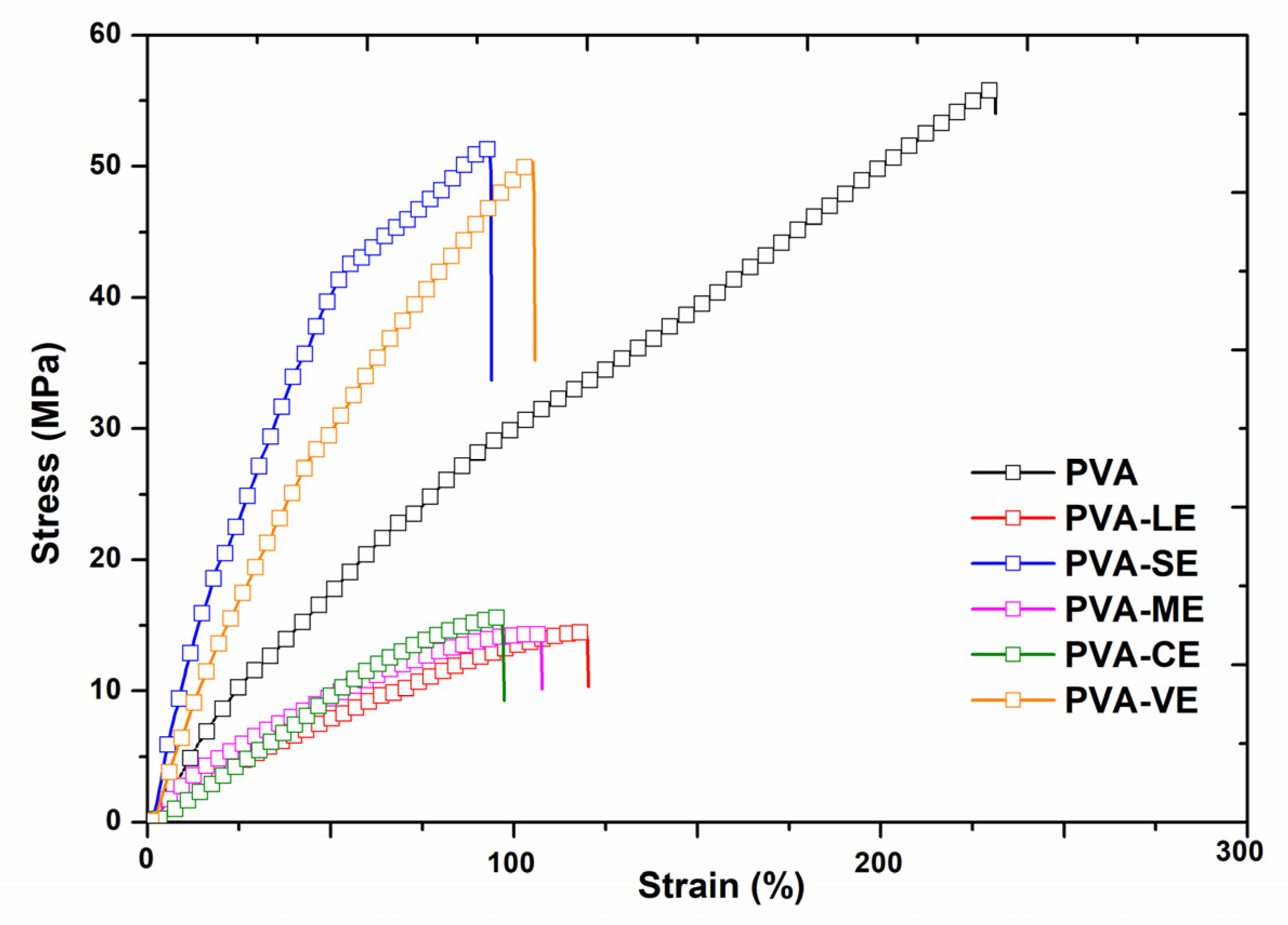
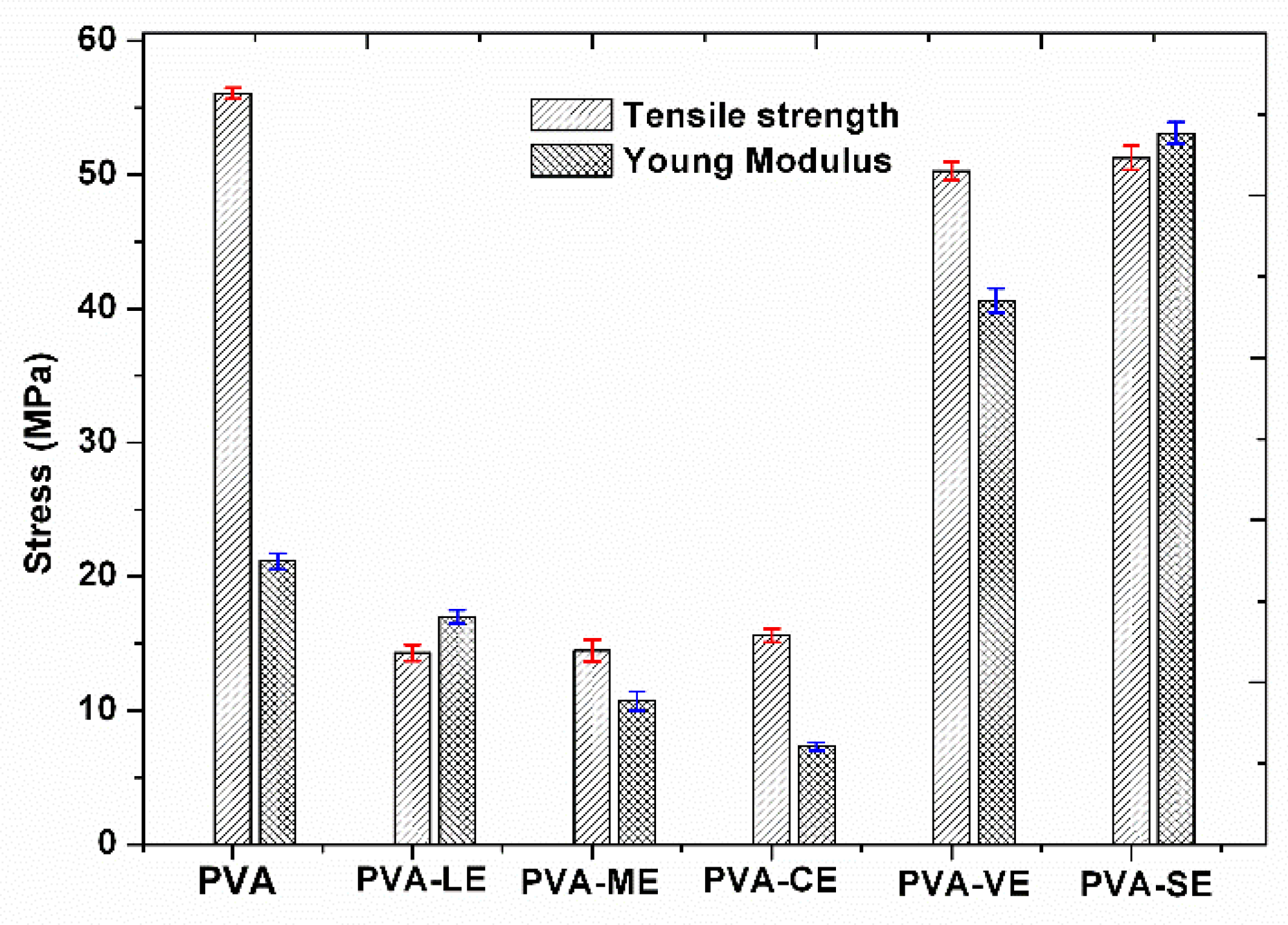
3.5. Mechanical Properties
3.6. Antibacterial Activity Analysis
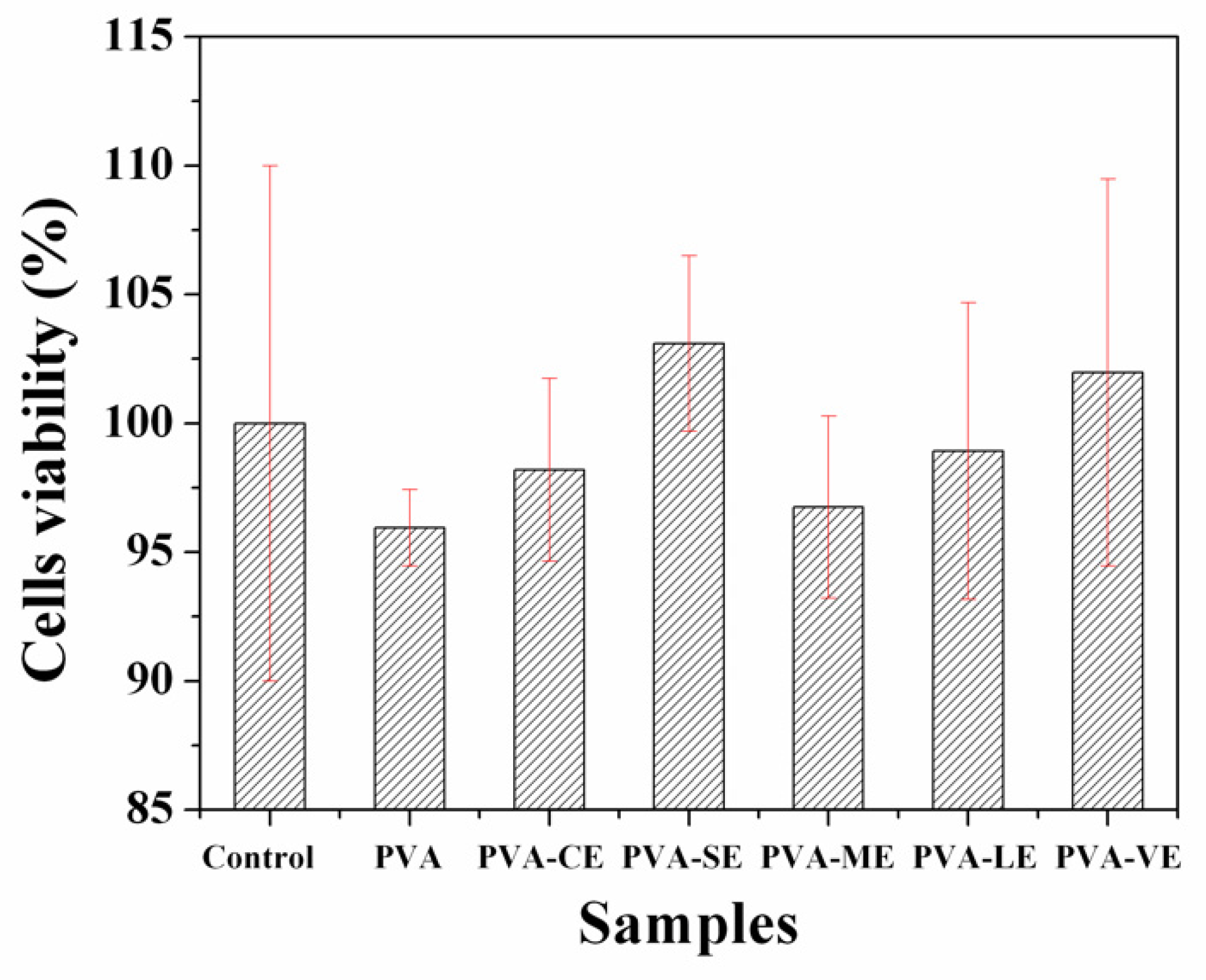
3.7. In Vitro Evaluation of Cytotoxicity—MTT Assay of the Films
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Predoi, D.; Iconaru, S.L.; Buton, N.; Badea, M.L.; Marutescu, L.J.N. Antimicrobial activity of new materials based on lavender and basil essential oils and hydroxyapatite. Nanomaterials 2018, 8, 291. [Google Scholar] [CrossRef] [PubMed]
- Tiemersma, E.W.; Bronzwaer, S.L.; Lyytikäinen, O.; Degener, J.E.; Schrijnemakers, P.; Bruinsma, N.; Monen, J.; Witte, W.; Grundmann, H.; European Antimicrobial Resistance Surveillance System Participants. Methicillin-resistant Staphylococcus aureus in Europe, 1999–2002. Emerg. Infect. Dis. 2004, 10, 1627. [Google Scholar] [CrossRef] [PubMed]
- Barbalata-Mandru, M.; Bercea, M.; Gradinaru, L.M.; Aflori, M.; Albulescu, R. Preparation and surface characterization of polyurethane hydrogels. Rev. Roum. Chim. 2021, 66, 87–93. [Google Scholar] [CrossRef]
- Brunchi, C.-E.; Bercea, M.; Morariu, S.; Avadanei, M. Investigations on the interactions between xanthan gum and poly(vinyl alcohol) in solid state and aqueous solutions. Eur. Polym. J. 2016, 84, 161–172. [Google Scholar] [CrossRef]
- Bonilla, J.; Fortunati, E.; Atarés, L.; Chiralt, A.; Kenny, J.M. Physical, structural and antimicrobial properties of poly vinyl alcohol–chitosan biodegradable films. Food Hydrocoll. 2014, 35, 463–470. [Google Scholar] [CrossRef]
- Chen, J.; Wei, D.; Gong, W.; Zheng, A.; Guan, Y. Hydrogen-Bond Assembly of Poly(vinyl alcohol) and Polyhexamethylene Guanidine for Nonleaching and Transparent Antimicrobial Films. ACS Appl. Mater. Interfaces 2018, 10, 37535–37543. [Google Scholar] [CrossRef]
- Ceja, I.; González-Íñiguez, K.J.; Carreón-Álvarez, A.; Landazuri, G.; Barrera, A.; Casillas, L.-C.; Fernández-Escamilla, V.V.A.; Aguilar, J. Characterization and Electrical Properties of PVA Films with Self-Assembled Chitosan-Au NPs/ SWCNT-COOH Nanostructures. Materials 2020, 13, 4138. [Google Scholar] [CrossRef]
- Ranjbarian, M.; Ma, X.; Mechtcherine, V. Influence of Crack Width in Alternating Tension–Compression Regimes on Crack-Bridging Behaviour and Degradation of PVA Microfibres Embedded in Cement-Based Matrix. Materials 2020, 13, 4189. [Google Scholar] [CrossRef]
- Bercea, M.; Morariu, S.; Rusu, D.J.S.M. In situ gelation of aqueous solutions of entangled poly (vinyl alcohol). Soft Matter 2013, 9, 1244–1253. [Google Scholar] [CrossRef]
- Hajji, S.; Chaker, A.; Jridi, M.; Maalej, H.; Jellouli, K.; Boufi, S.; Nasri, M.J.E.S.; Research, P. Structural analysis, and antioxidant and antibacterial properties of chitosan-poly (vinyl alcohol) biodegradable films. Environ. Sci. Pollut. Res. 2016, 23, 15310–15320. [Google Scholar] [CrossRef]
- Santamaria-Echart, A.; Fernandes, I.; Barreiro, F.; Retegi, A.; Arbelaiz, A.; Corcuera, M.A.; Eceiza, A. Development of waterborne polyurethane-ureas added with plant extracts: Study of different incorporation routes and their influence on particle size, thermal, mechanical and antibacterial properties. Prog. Org. Coat. 2018, 117, 76–90. [Google Scholar] [CrossRef]
- Duda, S.C.; Mărghitaş, L.A.; Dezmirean, D.; Duda, M.; Mărgăoan, R.; Bobiş, O. Products. Changes in major bioactive compounds with antioxidant activity of Agastache foeniculum, Lavandula angustifolia, Melissa officinalis and Nepeta cataria: Effect of harvest time and plant species. Ind. Crops Prod. 2015, 77, 499–507. [Google Scholar] [CrossRef]
- Sofi, H.S.; Akram, T.; Tamboli, A.H.; Majeed, A.; Shabir, N.; Sheikh, F.A. Novel lavender oil and silver nanoparticles simultaneously loaded onto polyurethane nanofibers for wound-healing applications. Int. J. Pharm. 2019, 569, 118590. [Google Scholar] [CrossRef] [PubMed]
- Vieira, S.F.; Ferreira, H.; Neves, N.M. Antioxidant and anti-inflammatory activities of cytocompatible Salvia officinalis extracts: A comparison between traditional and Soxhlet extraction. Antioxidants 2020, 9, 1157. [Google Scholar] [CrossRef] [PubMed]
- Alibi, S.; Crespo, D.; Navas, J. Plant-Derivatives Small Molecules with Antibacterial Activity. Antibiotics 2021, 10, 231. [Google Scholar] [CrossRef] [PubMed]
- Hirshfield, I.N.; Terzulli, S.; O’Byrne, C. Weak organic acids: A panoply of effects on bacteria. Sci. Prog. 2003, 86, 245–269. [Google Scholar] [CrossRef]
- Cowan, M.M. Plant products as antimicrobial agents. Clin. Microbiol. Rev. 1999, 12, 564. [Google Scholar] [CrossRef]
- Araniti, F.; Landi, M.; Lupini, A.; Sunseri, F.; Guidi, L.; Abenavoli, M. Origanum vulgare essential oils inhibit glutamate and aspartate metabolism altering the photorespiratory pathway in Arabidopsis thaliana seedlings. J. Plant Physiol. 2018, 231, 297–309. [Google Scholar] [CrossRef]
- Elhendawy, M.A.; Wanas, A.S.; Radwan, M.M.; Azzaz, N.A.; Toson, E.S.; ElSohly, M.A. Chemical and Biological Studies of Cannabis sativa Roots. Med. Cannabis Cannabinoids 2018, 1, 104–111. [Google Scholar] [CrossRef]
- Zaidi, S.; Dahiya, P. In vitro antimicrobial activity, phytochemical analysis and total phenolic content of essential oil from Mentha spicata and Mentha piperita. Int. Food Res. J. 2015, 22, 2040–2445. [Google Scholar]
- Giovannini, D.; Gismondi, A.; Basso, A.; Canuti, L.; Braglia, R.; Canini, A.; Mariani, F.; Cappelli, G. Lavandula angustifolia Mill. essential oil exerts antibacterial and anti-inflammatory in macrophage mediated immune response to Staphylococcus aureus. Immunol. Investig. 2016, 45, 11–28. [Google Scholar]
- Trevisan, S.C.C.; Menezes, A.P.P.; Barbalho, S.M.; Guiguer, E.L. Properties of Mentha piperita: A Brief Review. World, J. Pharm. Med. Res. 2017, 3, 309–313. [Google Scholar]
- Beigi, M.; Torki-Harchegani, M.; Pirbalouti, A.G. Quantity and chemical composition of essential oil of peppermint (Mentha piperita L.) leaves under different drying methods. Int. J. Food Prop. 2018, 21, 267–276. [Google Scholar]
- Chalchat, J.-C.; Garry, R.-P.; Michet, A. Variation of the Chemical Composition of Essential Oil of Mentha piperita L. during the Growing Time. J. Essent. Oil Res. 1997, 9, 463–465. [Google Scholar] [CrossRef]
- Lowe, H.; Toyang, N.; Steele, B.; Bryant, J.; Ngwa, W. The endocannabinoid system: A potential target for the treatment of various diseases. Int. J. Mol. Sci. 2021, 22, 9472. [Google Scholar] [CrossRef] [PubMed]
- Giuliano, C.; Francavilla, M.; Ongari, G.; Petese, A.; Ghezzi, C.; Rossini, N.; Blandini, F.; Cerri, S. Neuroprotective and Symptomatic Effects of Cannabidiol in an Animal Model of Parkinson’s Disease. Int. J. Mol. Sci. 2021, 22, 8920. [Google Scholar] [CrossRef]
- Khan, B.A.; Warner, P.; Wang, H. Antibacterial properties of hemp and other natural fibre plants: A review. BioRes. 2014, 9, 3642–3659. [Google Scholar] [CrossRef]
- Kubica, P.; Szopa, A.; Dominiak, J.; Luczkiewicz, M.; Ekiert, H. Verbena officinalis (Common Vervain)–A Review on the Investigations of This Medicinally Important Plant Species. Planta Med. 2020, 17, 1241–1257. [Google Scholar] [CrossRef]
- Deepak, M.; Handa, S.S. Quantitative determination of the major constituents of Verbena officinalis using high performance thin layer chromatography and high pressure liquid chromatography. Phytochem. Anal. 2000, 11, 351–355. [Google Scholar] [CrossRef]
- Casanova, E.; Garcia-Mina, J.; Calvo, M. Antioxidant and Antifungal Activity of Verbena officinalis L. leaves. Qual. Plant. Mater. Veg. 2008, 63, 93–97. [Google Scholar] [CrossRef]
- Martins, N.; Barros, L.; Santos-Buelga, C.; Henriques, M.; Silva, S.; Ferreira, I.C.F.R. Evaluation of bioactive properties and phenolic compounds in different extracts prepared from Salvia officinalis L. Food. Chem. 2015, 170, 378–385. [Google Scholar] [CrossRef] [PubMed]
- Ghorbanpour, M.; Hatami, M.; Kariman, K.; Dahaji, P.A. Phytochemical variations and enhanced efficiency of antioxidant and antimicrobial ingredients in Salvia officinalis as inoculated with different rhizobacteria. Chem. Biodivers. 2016, 13, 319–330. [Google Scholar] [CrossRef] [PubMed]
- Salevic, A.; Prieto, C.; Cabedo, L.; Nedović, V.; Lagaron, J.M. Physicochemical, antioxidant and antimicrobial properties of electrospun poly (ε-caprolactone) films containing a solid dispersion of sage (Salvia officinalis L.) extract. Nanomaterials 2019, 9, 270. [Google Scholar] [CrossRef] [PubMed]
- Zheljazkov, V.D.; Astatkie, T.; Hristov, A.N. Lavender and hyssop productivity, oil content, and bioactivity as a function of harvest time and drying. Ind. Crops Prod. 2012, 36, 222–228. [Google Scholar] [CrossRef]
- Wang, H.; Liu, Y.; Wei, S.; Yan, Z. Comparative seasonal variation and chemical composition of essential oils from the leaves and stems of Schefflera heptaphylla using microwave-assisted and conventional hydrodistillation. Ind. Crops Prod. 2012, 36, 229–237. [Google Scholar] [CrossRef]
- Slota, D.; Florkiewicz, W.; Piętak, K.; Szwed, A.; Włodarczyk, M.; Siwińska, M.; Rudnicka, K.; Sobczak-Kupiec, A. Preparation, Characterization, and Biocompatibility Assessment of Polymer-Ceramic Composites Loaded with Salvia officinalis Extract. Materials 2021, 14, 6000. [Google Scholar] [CrossRef]
- Prundeanu, M.; Brezoiu, A.-M.; Deaconu, M.; Gradisteanu Pircalabioru, G.; Lincu, D.; Matei, C.; Berger, D.J.M. Mesoporous Silica and Titania-Based Materials for Stability Enhancement of Polyphenols. Materials 2021, 14, 6457. [Google Scholar] [CrossRef]
- Zenkiewicz, M. Comparative study on the surface free energy of a solid calculated by different methods. Polym. Test. 2007, 26, 14–19. [Google Scholar] [CrossRef]
- Weinstein, M.P. Performance Standards for Antimicrobial Disk Susceptibility Tests, 30th ed.; CLSI: Wayne, NJ, USA, 2018; p. 92. [Google Scholar]
- Florkiewicz, W.; Pluta, C.; Dagmara, M.; Rudnicka, K.; Żywicka, A.; Guigou, M.D.; Tyliszczak, B.; Sobczak-Kupiec, A. Investigations on green synthesis, biocompatibility and antibacterial activity of silver nanoparticles prepared using Cistus incanus. Materials 2021, 14, 5028. [Google Scholar] [CrossRef]
- Pavlic, B.; Teslic, N.; Vidakovi, A.; Vidovic, S.; Velicanski, A.; Versari, A.; Radosavljevic, R.; Zekovic, Z. Sage processing from by-product to high quality powder: I. Bioactive potential. Ind. Crop Prod. 2017, 107, 81–89. [Google Scholar] [CrossRef]
- Masłowski, M.; Aleksieiev, A.; Miedzianowska, J.; Strzelec, K. Potential Application of Peppermint (Mentha piperita L.), German Chamomile (Matricaria chamomilla L.) and Yarrow (Achillea millefolium L.) as Active Fillers in Natural Rubber Biocomposites. Int. J. Mol. Sci. 2021, 22, 7530. [Google Scholar] [CrossRef] [PubMed]
- Balasubramanian, K.; Kodam, K.M. Encapsulation of therapeutic lavender oil in an electrolyte assisted polyacrylonitrile nanofibres for antibacterial applications. RSC Adv. 2014, 4, 54892–54901. [Google Scholar] [CrossRef]
- Das, S.; Das, A.; Selvan, T.R.; Ananda, R.S.; Bindhu, J. The antibacterial and aroma finishing of cotton fabrics by Menthe pipertia extract. J. Text. Inst. 2021, 112, 1180–1190. [Google Scholar] [CrossRef]
- Fernandez-Tendero, E.; Day, A.; Legros, S.; Habrant, A.; Hawkins, S.; Chabbert, B. Changes in hemp secondary fiber production related to technical fiber variability revealed by light microscopy and attenuated total reflectance Fourier transform infrared spectroscopy. PLoS ONE 2017, 12, e0179794. [Google Scholar] [CrossRef]
- Samfira, I.; Rodino, S.; Petrache, P.; Cristina, R.; Butu, M.; Butnariu, M. Characterization and identity confirmation of essential oils by mid infrared absorption spectrophotometry. Digest. J. Nanomater. Biostruct. 2015, 10, 557–566. [Google Scholar]
- Guillen, M.D.; Cabo, N. Infrared spectroscopy in the study of edible oils and fats. J. Sci. Food Agric. 1997, 75, 1–11. [Google Scholar] [CrossRef]
- Cirrincione, M.; Saladini, B.; Brighenti, V.; Salamone, S.; Mandrioli, R.; Pollastrio, F.; Pellati, F.; Protti, M.; Mercolini, L. Discriminating different Cannabis sativa L. chemotypes using attenuated total reflectance-infrared (ATR-FTIR) spectroscopy: A proof of concept. J. Pharm. Biomed. 2021, 204, 114270. [Google Scholar] [CrossRef]
- Smith, B. Infrared Spectra Interpretation, A Systematic Approach, 1st ed.; CRC Press: Boca Raton, FL, USA, 1999; p. 304. [Google Scholar]
- Schonbichler, S.A.; Bittner, L.K.H.; Pallua, J.D.; Popp, M.; Abel, G.; Bonn, G.K.; Huck, C.W. Simultaneous quantification of verbenalin and verbascoside in Verbena officinalis by ATR-IR and NIR spectroscopy. J. Pharm. Biomed. 2013, 84, 97–102. [Google Scholar] [CrossRef]
- Kucharska-Ambrozej, K.; Martyna, A.; Karpinska, J.; Kieltyka-Dadasiewicz, A.; Kubat-Sikorska, A. Quality control of mint species based on UV-VIS and FTIR spectral data supported by chemometric tools. Food Control. 2021, 129, 108228. [Google Scholar] [CrossRef]
- Wang, C.; Zheng, Y.; Qiao, K.; Xie, Y.; Zhou, X. An environmentally friendly preparation and characterization of waterborne polyurethane hydrogels by polyvinyl alcohol physical cross-linking to improve water absorption. RSC Adv. 2015, 5, 73882–73891. [Google Scholar] [CrossRef]
- Lima, G.G.; de Souza, R.O.; Bozzi, A.D.; Poplawska, M.A.; Devine, D.M.; Nugent, M.J.D. Extraction method plays critical role in antibacterial activity of propolis-loaded hydrogels. J. Pharm. Sci. 2016, 105, 1248–1257. [Google Scholar] [CrossRef] [PubMed]
- Olewnik-Kruszkowska, E.; Gierszewska, M.; Jakubowska, E.; Tarach, I.; Sedlarik, V.; Pummerova, M. Antibacterial Films Based on PVA and PVA–Chitosan Modified with Poly(Hexamethylene Guanidine). Polymers 2019, 11, 2093. [Google Scholar] [CrossRef] [PubMed]
- Mandru, M.; Bercea, M.; Gradinaru, L.M.; Ciobanu, C.; Drobota, M.; Vlad, S.; Albulescu, R. Polyurethane/poly(vinyl alcohol) hydrogels: Preparation, characterization and drug delivery. Eur. Polym. J. 2019, 118, 137–145. [Google Scholar] [CrossRef]
- Daferera, D.J.; Tarantilis, P.A.; Polissiou, M.G. Characterization of essential oils from lamiaceae species by fourier transform Raman spectroscopy. J. Agric. Food Chem. 2002, 50, 5503–5507. [Google Scholar] [CrossRef]
- Noh, C.H.C.; Azmin, N.F.M.; Amid, A.J.A. Principal component analysis application on flavonoids characterization. Adv. Sci. Technol. Eng. 2017, 2, 435–440. [Google Scholar] [CrossRef]
- Ciko, L.; Andoni, A.; Ylli, F.; Plaku, E.; Taraj, K.; Çomo, A. Extraction of Essential Oil from Albanian Salvia officinalis L. and its Characterization by FTIR Spectroscopy. Int. J. Ecosyst. Ecol. Sci. 2016, 28, 1401–1402. [Google Scholar] [CrossRef]
- Barbin, D.F.; Felicio, A.L.d.S.M.; Sun, D.-W.; Nixdorf, S.L.; Hirooka, E.Y. Application of infrared spectral techniques on quality and compositional attributes of coffee: An overview. Int. Fodd. Res. J. 2014, 61, 23–32. [Google Scholar] [CrossRef]
- Mabry, T.J.; Markham, K.R.; Thomas, M.D. The Ultraviolet Spectra of Flavones and Flavonols. In The Systematic Identification of Flavonoid, 1st ed.; Springer: Berlin/Heidelberg, Germany, 1970; pp. 41–164. [Google Scholar]
- Parlinska-Wojtan, M.; Kus-Liskiewicz, M.; Depciuch, J.; Sadik, O. Green synthesis and antibacterial effects of aqueous colloidal solutions of silver nanoparticles using camomile terpenoids as a combined reducing and capping agent. Bioprocess. Biosyst. Eng. 2016, 39, 1213–1223. [Google Scholar] [CrossRef]
- Wyrostek, J.; Kowalski, R.; Pankiewicz, U.; Solarska, E. Estimation of the Content of Selected Active Substances in Primary and Secondary Herbal Brews by UV-VIS and GC-MS Spectroscopic Analyses. J. Anal. Methods Chem. 2020, 11, 8891855. [Google Scholar] [CrossRef]
- Musetti, B.; González-Ramos, H.; González, M.; Bahnson, E.M.; Varela, J.; Thomson, L. Cannabis sativa extracts protect LDL from Cu2+-mediated oxidation. J. Cannabis Res. 2020, 2, 37. [Google Scholar] [CrossRef]
- Atobe, S.; Saga, K.; Maeyama, H.; Fujiwara, K.; Okada, S.; Imou, K. Culture of the green microalga Botryococcus braunii Showa with LED irradiation eliminating violet light enhances hydrocarbon production and recovery. Biosci. Biotechnol. Biochem. 2014, 78, 1765–1771. [Google Scholar] [CrossRef] [PubMed]
- De Faria, A.F.; De Rosso, V.V.; Mercadante, A.Z. Carotenoid Composition of Jackfruit (Artocarpus heterophyllus), Determined by HPLC-PDA-MS/MS. Plant Foods Hum. Nutr. 2009, 64, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Thrane, J.-E.; Kyle, M.; Striebel, M.; Haande, S.; Grung, M.; Rohrlack, T.; Andersen, T. Spectrophotometric analysis of pigments: A critical assessment of a highthroughput method for analysis of algal pigment mixtures by spectral deconvolution. PLoS ONE 2015, 10, e0137645. [Google Scholar] [CrossRef] [PubMed]
- Karakaya, S. Bioavailability of phenolic compounds. Crit. Rev. Food Sci. Nutr. 2004, 44, 453–464. [Google Scholar] [CrossRef]
- Razna, K.; Sawinska, Z.; Ivanisova, E.; Vukovic, N.; Terentjeva, M.; Stricik, M.; Kowalczewski, P.L.; Hlavckova, L.; Rovna, K.; Ziarovska, J.; et al. Properties of Ginkgo biloba L. Antioxidant Characterization, Antimicrobial Activities, and Genomic MicroRNA Based Marker Fingerprints. Int. J. Mol. Sci. 2020, 21, 3087. [Google Scholar]
- Zohra, M. Impact of solvent extraction type on total polyphenols content and biological activity from Tamarix aphylla (L). Int. J. Pharm. Biol. 2011, 2, 609–615. [Google Scholar]
- Cocan, I.; Alexa, E.; Danciu, C.; Radulov, I.; Galuscan, A.; Obistioiu, D.; Morvay, A.A.; Sumalan, R.M.; Poiana, M.A.; Pop, G.; et al. Phytochemical screening and biological activity of Lamiaceae family plant extracts. Exp. Ther. Med. 2018, 15, 1863–1870. [Google Scholar] [CrossRef]
- Zagorska-Dziok, M.; Bujak, T.; Ziemlewska, A.; Niziol-Lukaszewska, Z. Positive Effect of Cannabis sativa L. Herb Extracts on Skin Cells and Assessment of Cannabinoid-Based Hydrogels Properties. Molecules 2021, 26, 802. [Google Scholar] [CrossRef]
- Benabdallah, A.; Rahmoune, C.; Boumendjel, M.; Aissi, O.; Messaoud, C. Total phenolic content and antioxidant activity of six wild Mentha species (Lamiaceae) from northeast of Algeria. Asian Pac. J. Trop. Biomed. 2016, 6, 760–766. [Google Scholar] [CrossRef]
- Wu, S.; Zhang, B.; Liu, Y.; Suo, X.; Li, H. Influence of surface topography on bacterial adhesion: A review. Biointerphases 2018, 13, 060801. [Google Scholar] [CrossRef]
- Hasan, J.; Chatterjee, K. Recent advances in engineering topography mediated antibacterial surfaces. Nanoscale 2015, 7, 15568–15575. [Google Scholar] [CrossRef] [PubMed]
- Cano, A.; Fortunati, E.; Cháfer, M.; Kenny, J.M. Properties and ageing behavior of pea starch flms as afected by blend with poly(vinyl alcohol). Food Hydrocoll. 2015, 48, 84–93. [Google Scholar] [CrossRef]
- Preedy, E.; Perni, S.; Nipic, D.; Bohinc, K.; Prokopovich, P. Surface roughness mediated adhesion forces between borosilicate glass and gram-positive bacteria. Langmuir 2014, 30, 9466–9476. [Google Scholar] [CrossRef] [PubMed]
- Kocijan, A.; Conradi, M.; Hocevar, M. The Influence of Surface Wettability and Topography on the Bioactivity of TiO2/Epoxy Coatings on AISI 316L Stainless Steel. Materials 2019, 12, 1877. [Google Scholar] [CrossRef]
- An, Y.H.; Friedman, R.J. Concise review of mechanisms of bacterial adhesion to biomaterial surfaces. J. Biomed. Mater. Res. 1998, 43, 338–348. [Google Scholar] [CrossRef]
- McAllister, E.W.; Carey, L.C.; Brady, P.G.; Heller, R.; Kovacs, S.G. The role of polymeric surface smoothness of biliary stents in bacterial adherence, biofilm deposition, and stent occlusion. Gastrointest. Endosc. 1993, 39, 422–425. [Google Scholar] [CrossRef]
- Unalan, I.; Slavik, B.; Buettner, A.; Goldmann, W.H.; Frank, G.; Boccaccini, A.R. Physical and Antibacterial Properties of Peppermint Essential Oil Loaded Poly (ε-caprolactone) (PCL) Electrospun Fiber Mats for Wound Healing. Front. Bioeng. Biotechnol. 2019, 7, 346. [Google Scholar] [CrossRef]
- Chen, Y.; Busscher, H.J.; van der Mei, H.C.; Norde, W. Statistical Analysis of Long- and Short-Range Forces Involved in Bacterial Adhesion to Substratum Surfaces as Measured Using Atomic Force Microscopy. Appl. Environ. Microbiol. 2011, 77, 5065–5070. [Google Scholar] [CrossRef]
- Aggarwal, K.K.; Khanuja, S.P.S.; Ahmad, A.; Kumar, T.R.S.; Gupta, V.K.; Kumar, S. Antimicrobial activity profiles of the two enantiomers of limonene and carvone isolated from the oils of Mentha spicata and Anethum sowa. Flavour Fragr. J. 2002, 17, 59–63. [Google Scholar] [CrossRef]
- Cetin-Karaca, H.; Newman, C.M. Antimicrobial Efficacy of Natural Phenolic Compounds against Gram Positive Foodborne Pathogens. J. Food Sci. 2015, 4, 6. [Google Scholar] [CrossRef]
- Atar´es, L.; Chiralt, A. Essential oils as additives in biodegradable films and coatings for active food packaging. Trends Food Sci. Technol. 2016, 48, 51–62. [Google Scholar] [CrossRef]
- Hammer, K.A.; Carson, C.F.; Riley, T.V. Antimicrobial activity of essential oils and other plant extracts. J. Appl. Microbiol. 1999, 86, 985–990. [Google Scholar] [CrossRef] [PubMed]
- Burt, S.A.; Reinders, R.D. Antibacterial activity of selected plant essential oils against Escherichia coli O157: H7. Lett. Appl. Microbiol. 2003, 36, 162–167. [Google Scholar] [CrossRef] [PubMed]
- Havsteen, B. Flavonoids, a class of natural products of high pharmacological potency. Biochem. Pharmacol. 1983, 7, 1141–1148. [Google Scholar] [CrossRef]
- McKay, D.L.; Blumberg, J.B. A review of the bioactivity and potential health benefits of peppermint tea (Mentha piperita L.). Phytother. Res. 2006, 20, 619–633. [Google Scholar] [CrossRef] [PubMed]
- Kligler, B.; Chaudhary, S. Peppermint oil. Am. Fam. Physician. 2007, 75, 1027–1030. [Google Scholar]
- Bautista, J.L.; Yu, S.; Tian, L. Flavonoids in Cannabis sativa: Biosynthesis, Bioactivities, and Biotechnology. ACS Omega 2021, 6, 5119–5123. [Google Scholar] [CrossRef]
- Wasim, K.; Haq, I.; Ashraf, M. Antimicrobial studies of the leaf of Cannabis sativa L. Pak J. Pharm. Sci. 1995, 8, 29–38. [Google Scholar]
- Ghorbani, A.; Esmaeilizadeh, M. Pharmacological properties of Salvia officinalis and its components. J. Tradit. Complement. Med. 2017, 7, 433–440. [Google Scholar] [CrossRef]
- Delamare, A.P.L.; Moschen-Pistorello, I.T.; Artico, L.; Atti-Serafini, L.; Echeverrigaray, S. Antibacterial activity of the essential oils of Salvia officinalis L. and Salvia triloba L. cultivated in South Brazil. Food Chem. 2007, 100, 603–608. [Google Scholar] [CrossRef]
- Liu, X.; Sun, Z.-L.; Jia, A.-R.; Shi, Y.-P.; Li, R.-H.; Yang, P.-M. Extraction, Preliminary Characterization and Evaluation of in Vitro Antitumor and Antioxidant Activities of Polysaccharides from Mentha piperita. Int. J. Mol. Sci. 2014, 15, 16302–16319. [Google Scholar] [CrossRef] [PubMed]
- Cavanagh, H.M.A.; Wilkinson, J.M. Biological Activities of Lavender Essential Oil. Phytother. Res. 2002, 16, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Wang, J.; Zhang, H.; Dong, M.; Li, L.; Jia, P.; Bu, T.; Wang, X.; Wang, L. Development of functional gelatin-based composite films incorporating oil-in-water lavender essential oil nano-emulsions: Effects on physicochemical properties and cherry tomatoes preservation. LWT 2021, 142, 110987. [Google Scholar] [CrossRef]
- Elshafie, H.S.; Sakr, S.; Mang, S.M.; Belviso, S.; Feo, V.D.; Camele, I. Antimicrobial Activity and Chemical Composition of Three Essential Oils Extracted from Mediterranean Aromatic Plants. J. Med. Food 2016, 19, 1–8. [Google Scholar] [CrossRef]
- De Oliveira, J.R.; Vilela, P.; Almeida, R.B.d.A.; De Oliveira, F.E.; Carvalho, C.A.T.; Camargo, S.E.A.; Jorge, A.O.C.; de Oliveira, L.D. Antimicrobial activity of noncytotoxic concentrations of Salvia officinalis extract against bacterial and fungal species from the oral cavity. Gen. Dent. 2019, 67, 22–26. [Google Scholar] [PubMed]
- Jain, D.; Pathak, N.; Khan, S.; Raghuram, G.V.; Bhargava, A.; Samarth, R.; Mishra, P.K. Evaluation of cytotoxicity and anticarcinogenic potential of Mentha leaf extracts. Int. J. Toxicol. 2011, 30, 225–236. [Google Scholar] [CrossRef] [PubMed]



| Ethanolic Extract | UV | TPC | TFC |
|---|---|---|---|
| Absorbance (nm) | (mg GAE/g) | (mg QE/g) | |
| Lavandula angustifolia- (Flowers) | 278/318 | 69.61 ± 1.23 | 14.1 ± 0.56 |
| Verbena officinalis-(Leaves) | 285/331 | 102 ± 1.75 | 30.9 ± 1.24 |
| Salvia officinalis folium-(Leaves) | 287/333 | 297.5 ± 0.67 | 46.22 ± 0.97 |
| Hemp (Cannabis sativa L.)-(Leaves) | 286/332 | 76.88 ± 0.98 | 37.25 ± 1.56 |
| Mentha piperita-(Leaves) | 274/312 | 107.05 ± 1.89 | 26.80 ± 1.78 |
| Samples | Ra (nm) | θW (°) | Surface Free Energy (mN/m) | ||
|---|---|---|---|---|---|
| γpsv (mN/m) | γdsv (mN/m) | γsv (mN/m) | |||
| PVA | 6.47 ± 0.3 | 65.41 ± 0.7 | 22.89 | 16.72 | 39.61 |
| PVA-LE | 23.46 ± 0.5 | 57.67 ± 0.8 | 23.10 | 21.26 | 44.37 |
| PVA-ME | 22.81 ± 0.8 | 54.53 ± 0.2 | 27.42 | 18.56 | 45.99 |
| PVA-CE | 43.95 ± 0.6 | 44.01 ± 0.6 | 49.66 | 6.71 | 56.38 |
| PVA-VE | 12.27 ± 0.2 | 48.81 ± 0.4 | 38.07 | 12.26 | 50.33 |
| PVA-SE | 48.82 ± 0.4 | 36.76 ± 0.9 | 56.59 | 6.15 | 62.74 |
| Samples (0.5 mm) | Activity against +/- | Inhibition Zone mm | ||
|---|---|---|---|---|
| S. aureus | E. coli | S. aureus | E. coli | |
| PVA | - | - | 0 | 0 |
| PVA-LE | + | + | 8 ± 0.2 | 7 ± 0.2 |
| PVA-VE | ++ | + | 13 ± 0.3 | 9 ± 0.1 |
| PVA-SE | ++++ | + | 20 ± 0.1 | 7 ± 0.3 |
| PVA-CE | ++ | +++ | 10 ± 0.2 | 12 ± 0.4 |
| PVA-ME | ++ | ++ | 10 ± 0.1 | 9 ± 0.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barbălată-Mândru, M.; Serbezeanu, D.; Butnaru, M.; Rîmbu, C.M.; Enache, A.A.; Aflori, M. Poly(vinyl alcohol)/Plant Extracts Films: Preparation, Surface Characterization and Antibacterial Studies against Gram Positive and Gram Negative Bacteria. Materials 2022, 15, 2493. https://doi.org/10.3390/ma15072493
Barbălată-Mândru M, Serbezeanu D, Butnaru M, Rîmbu CM, Enache AA, Aflori M. Poly(vinyl alcohol)/Plant Extracts Films: Preparation, Surface Characterization and Antibacterial Studies against Gram Positive and Gram Negative Bacteria. Materials. 2022; 15(7):2493. https://doi.org/10.3390/ma15072493
Chicago/Turabian StyleBarbălată-Mândru, Mihaela, Diana Serbezeanu, Maria Butnaru, Cristina Mihaela Rîmbu, Alexandru Alin Enache, and Magdalena Aflori. 2022. "Poly(vinyl alcohol)/Plant Extracts Films: Preparation, Surface Characterization and Antibacterial Studies against Gram Positive and Gram Negative Bacteria" Materials 15, no. 7: 2493. https://doi.org/10.3390/ma15072493
APA StyleBarbălată-Mândru, M., Serbezeanu, D., Butnaru, M., Rîmbu, C. M., Enache, A. A., & Aflori, M. (2022). Poly(vinyl alcohol)/Plant Extracts Films: Preparation, Surface Characterization and Antibacterial Studies against Gram Positive and Gram Negative Bacteria. Materials, 15(7), 2493. https://doi.org/10.3390/ma15072493









