Behaviour of Impurities during Electron Beam Melting of Copper Technogenic Material
Abstract
:1. Introduction
2. Materials and Methods
3. Results
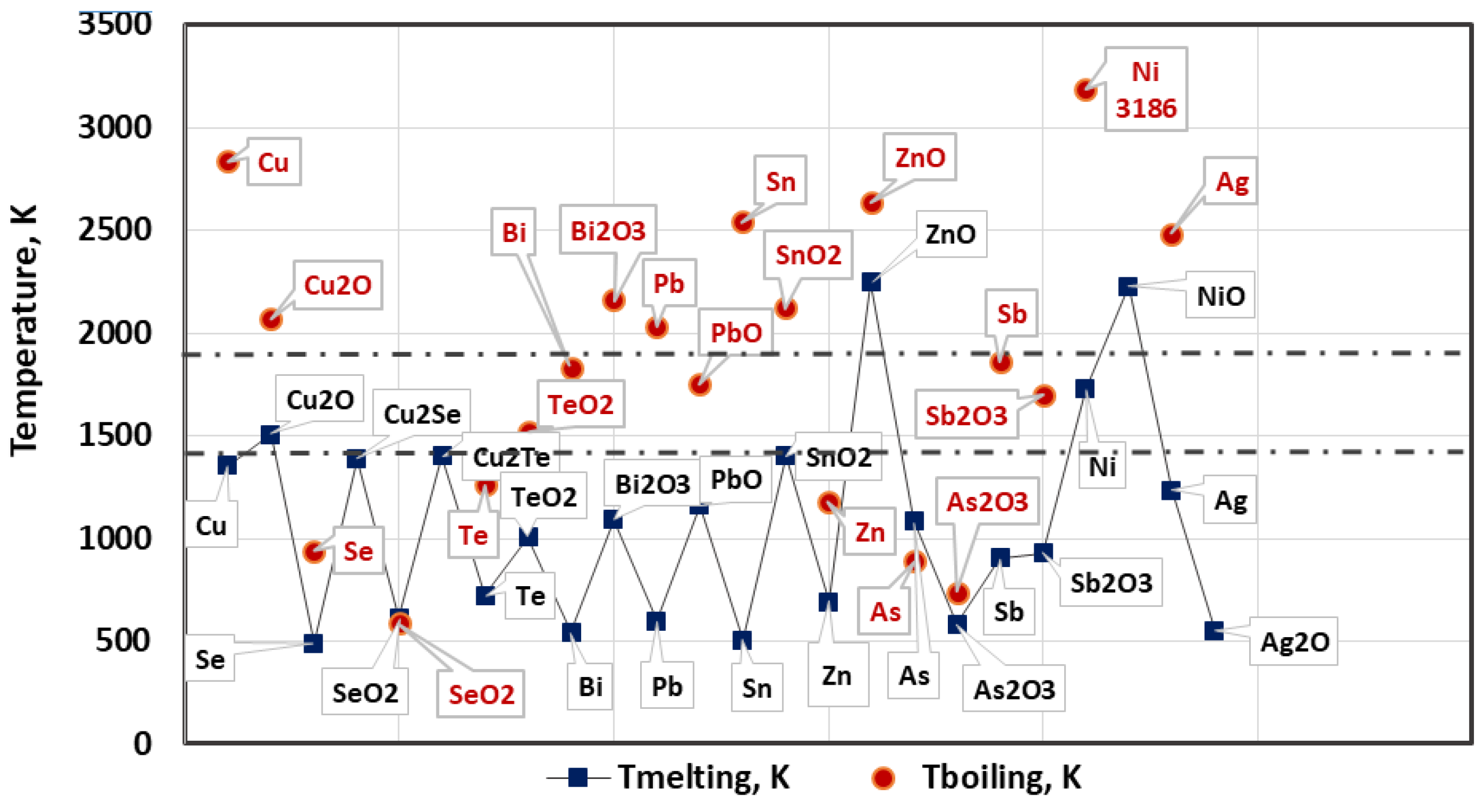
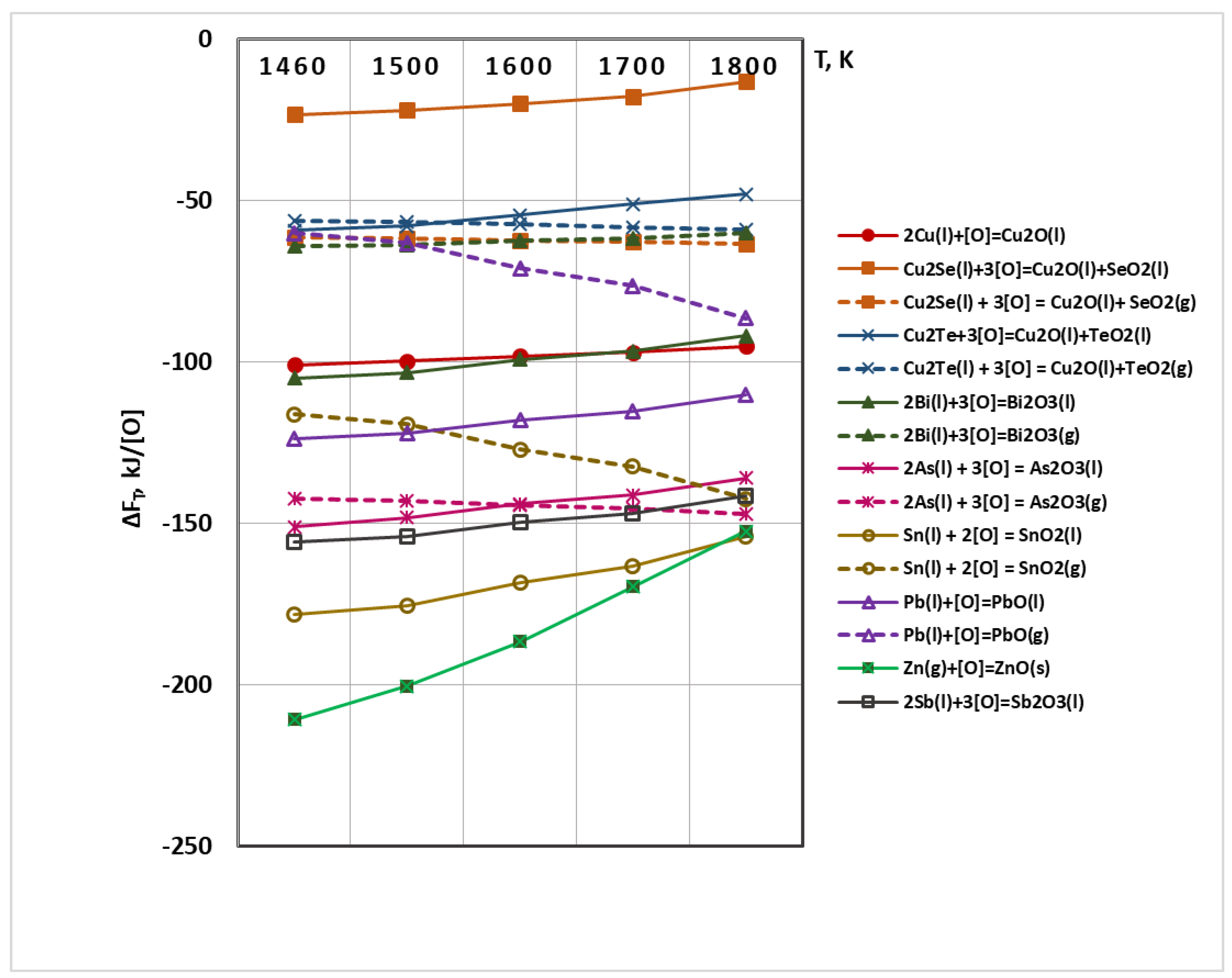
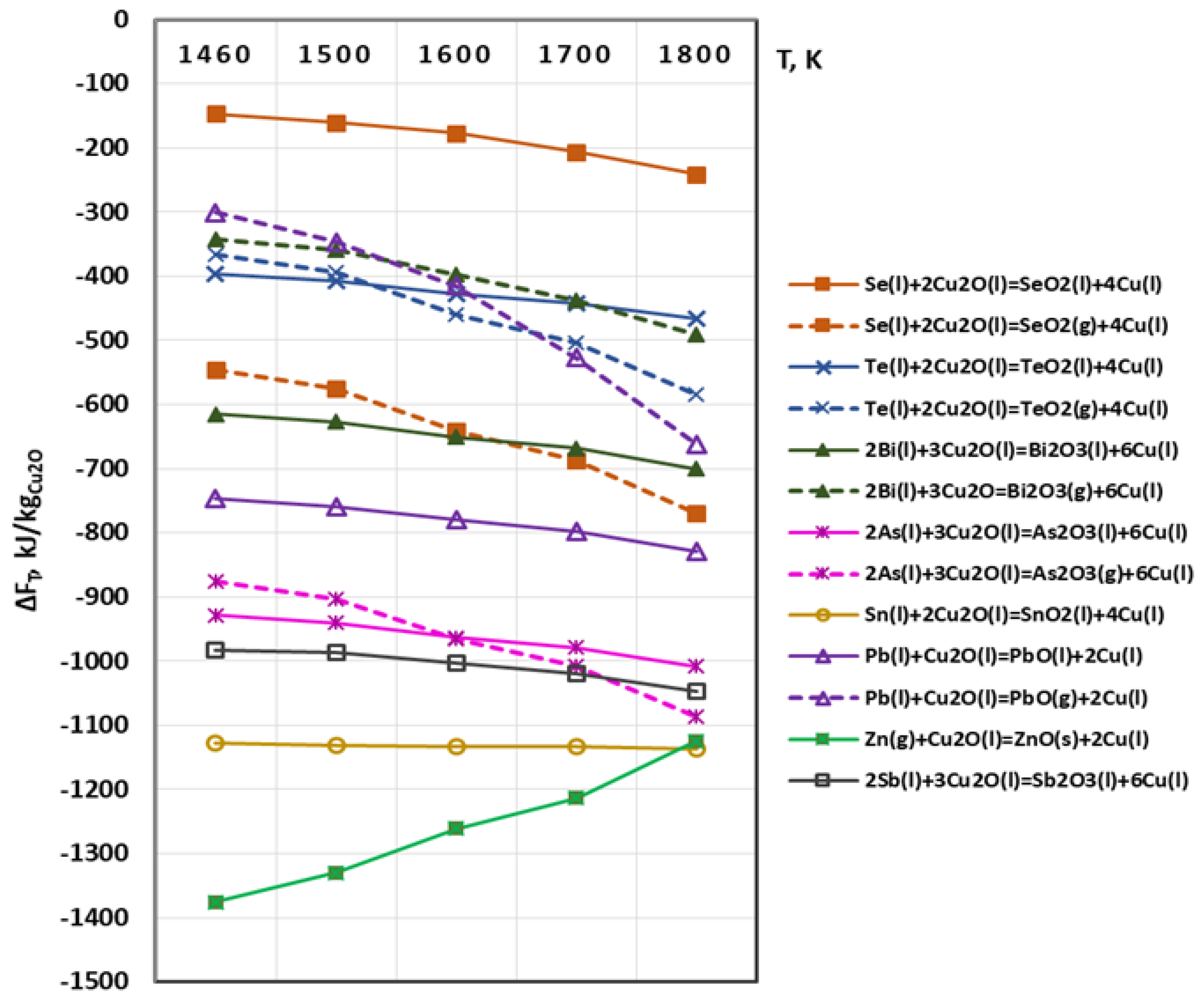
3.1. Thermodynamic Analysis of Possible Chemical Interactions during Electron Beam Melting and Refining (EBMR)
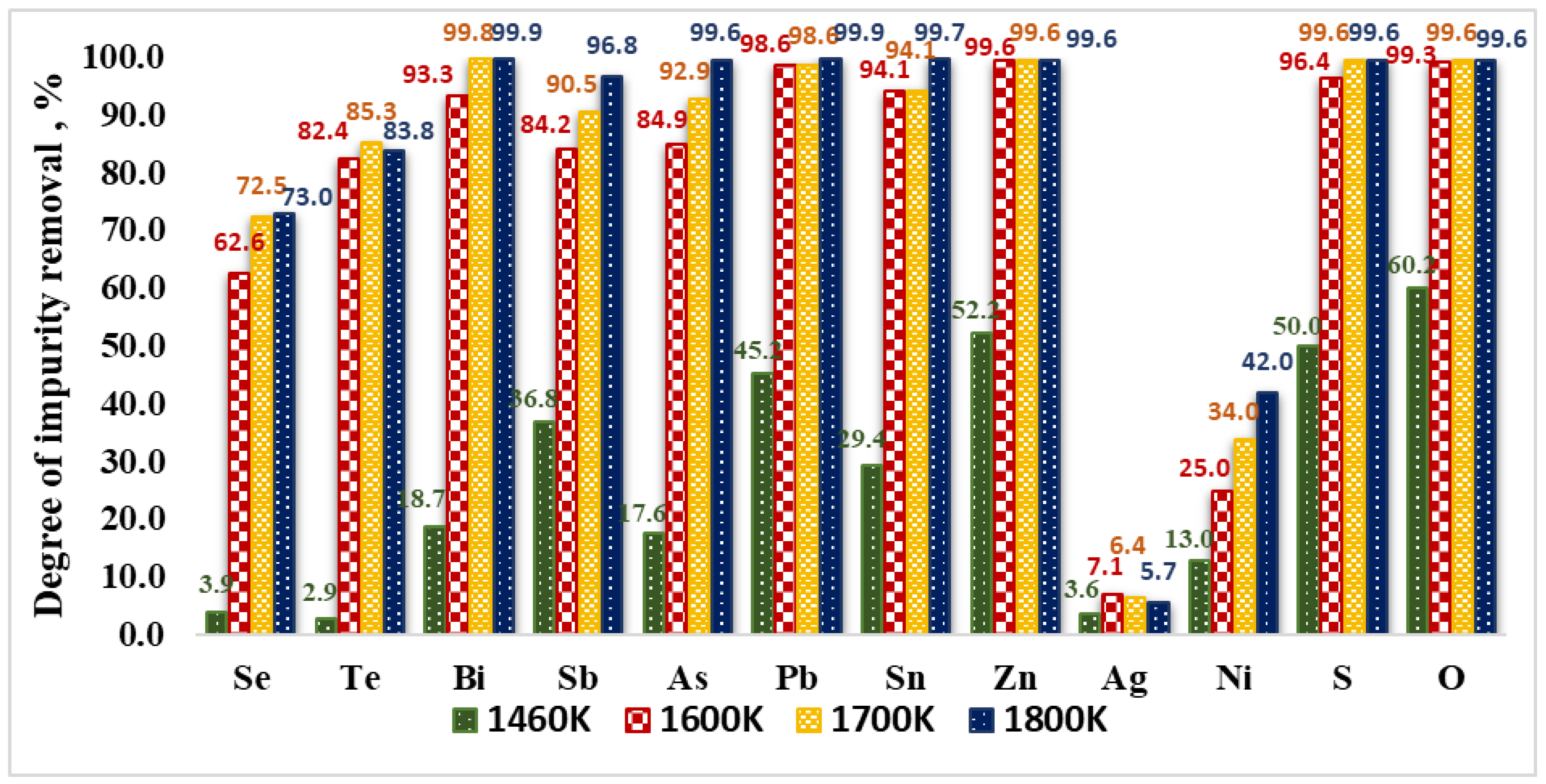
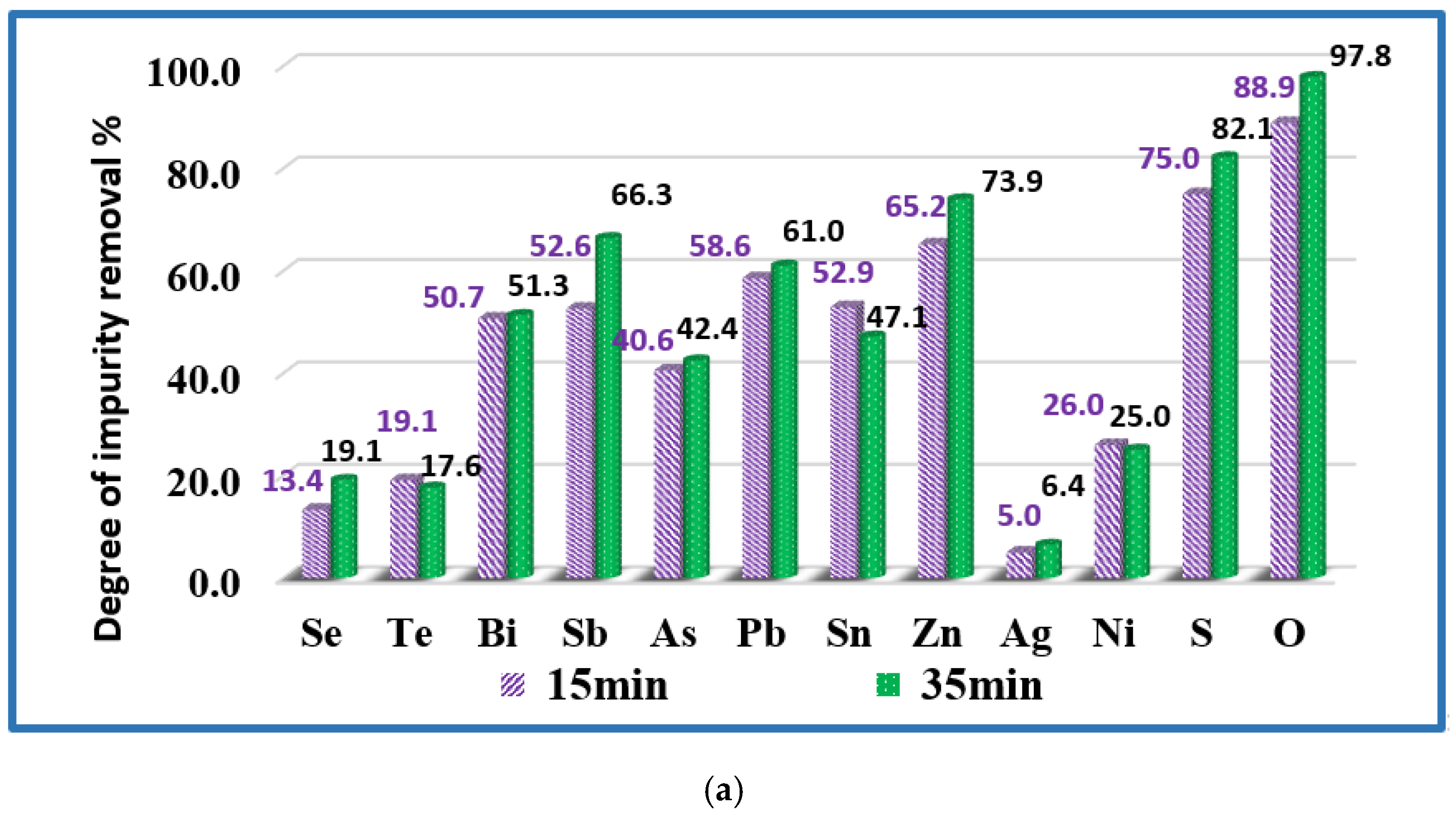
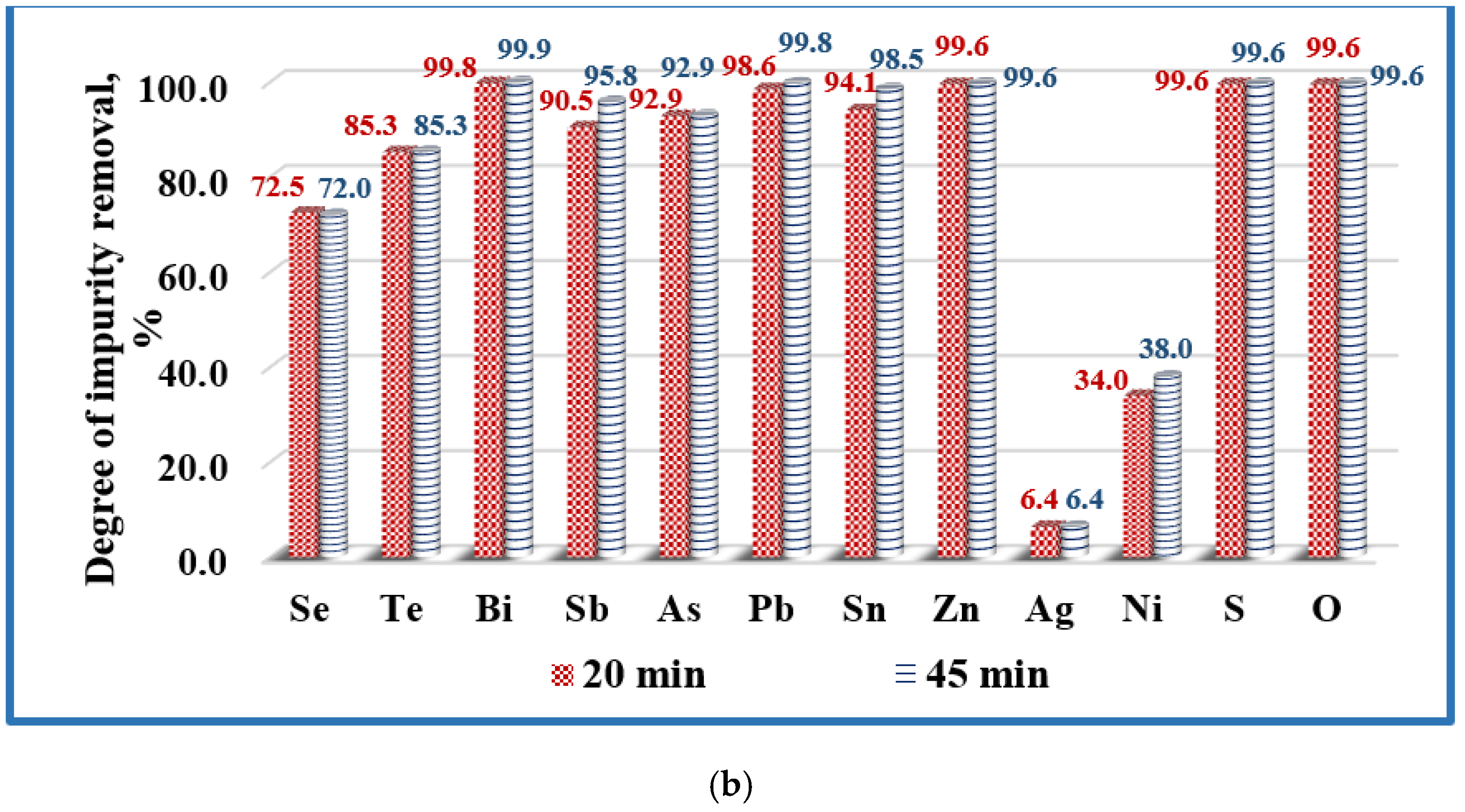
3.2. Refining Efficiency and Microstructures of Obtained Copper
4. Conclusions
- The results obtained show that the electron beam melting method can be successfully applied for refining copper technogenic material with a high content of impurities, which in the conditions of EBM are in a gaseous state (such as Bi, Pb, Zn, As, Sb, Sn) and reach nearly 100% removal degree and ~97% for Sb.
- Oxygen and sulphur also reach a high degree of removal (≥99%). Under the studied conditions, the maximum degree of refining of Se and Te is 73% and 85.3%, respectively, which is due to the fact that Se and Te form intermetallic compounds with copper, which are more difficult to oxidize. This corresponds to significantly lower values of the Gibbs energy of the oxidation reactions of these impurities.
- Silver and most of the nickel remain in copper. Under vacuum conditions and at the temperature range studied, silver does not oxidize or evaporate. The low degree of refining of nickel (34–42%) from copper can be explained by the good solubility of this impurity in copper. In addition, nickel is also not oxidized in the studied temperature range. It was found that raising the temperature above 1700 K, as well as extending the melting time over 20 min hardly change the purity and structure of the resulting refined metal.
- At temperatures in the range 1600–1800 K, the achieved refining efficiency is 78–85% and the purity of copper after EBM is 99.9%. The highest total refining efficiency of 84.6% is seen at a beam power of 19.5 kW for 20 min melting time and the best purification of copper technogenic material (99.92%) is achieved.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- International Copper Study Group. The World Copper Factbook; 2020; Available online: http://www.icsg.org/index.php/component/jdownloads/finish/170/3046 (accessed on 23 January 2022).
- European Copper Institute Copper: A Circular Material for a Resource Efficient Europe. Available online: https://copperalliance.eu/benefits-of-copper/recycling/ (accessed on 3 December 2021).
- Biswas, A.K.; Davenport, W.G. Extractive Metallurgy of Copper, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2013; p. 518. [Google Scholar]
- Hanusch, K.; Bussmann, H. Behavior and removal of associated metals in the secondary metallurgy of copper. In Proceedings of the 3rd International Symposium on Recycling of Materials and Engineered Materials, Point Clear, AL, USA, 12–16 November 1995; pp. 171–188. [Google Scholar]
- Antrekowitsch, H.; Wenzl, C.; Filzwieser, I.; Offenthaler, D. Pyrometallurgical Refining of Copper in an Anode Furnace; TMS, Ed.; The Minerals, Metals & Materials Society: Warrendale, PA, USA, 2005; pp. 1–6. [Google Scholar]
- Asghari, H.; Safarzadeh, M.S.; Asghari, G.; Moradkham, D. The effect of impurities on the extraction of copper from sulfate medium using LIX®984N in kerosene. Russ. J. Non-Ferr. Met. 2009, 50, 89–96. [Google Scholar] [CrossRef]
- Navarro, P.; Vargas, C.; Castillo, J.; Sepulveda, R. Experimental Study of Phase Entrainment in Copper Solvent Extraction. DYNA 2020, 87, 85–90. [Google Scholar] [CrossRef]
- Mackey, P.J.; Wraith, J. Development of copper quality: An historical perspective. Miner. Process. Extr. Metall 2004, 113, 25–37. [Google Scholar] [CrossRef]
- Mladenov, G.; Koleva, E.; Vutova, K.; Vassileva, V. Practical Aspects and Application of Electron Beam Irradiation; Memtanu, M., Brasoveanu, M., Eds.; Transworld Research Network: Trivandrum, India, 2011; pp. 43–93. [Google Scholar]
- Kalugin, A. Electron Beam Melting of Metals; Metallurgy Publishing House: Moscow, Russia, 1980. (In Russian) [Google Scholar]
- Mitchell, A.; Wang, T. Electron beam melting technology review. In Proceedings of the Conference Electron Beam Melting and Refining, Reno, NV, USA, 29–31 October 2000; pp. 2–13. [Google Scholar]
- Vassileva, V.; Mladenov, G.; Vutova, K.; Nikolov, T.; Georgieva, E. Oxygen removal during electron beam drip melting and refining. Vacuum 2005, 77, 429–436. [Google Scholar] [CrossRef]
- Vutova, K.; Vassileva, V.; Koleva, E.; Munirathnam, N.; Amalnerkar, D.; Tanaka, T. Investigation of Tantalum Recycling by Electron Beam Melting. Metals 2016, 6, 287. [Google Scholar] [CrossRef] [Green Version]
- Yue, H.; Peng, H.; Li, R.; Gao, R.; Wang, X.; Chen, Y. High-temperature microstructure stability and fracture toughness of TiAl alloy prepared via electron beam smelting and selective electron beam melting. Intermetallics 2021, 136, 107259. [Google Scholar] [CrossRef]
- You, X.; Tan, Y.; Cui, H.; Zhang, H.; Zhuang, X.; Zhao, L.; Niu, S.; Li, Y.; Li, P. Microstructure evolution of an Inconel 718 alloy prepared by electron beam smelting. Mater. Charact. 2021, 173, 110925. [Google Scholar] [CrossRef]
- Vutova, K.; Vassileva, V.; Stefanova, V.; Amalnerkar, D.; Tanaka, T. Effect of electron beam method on processing of titanium technogenic material. Metals 2019, 9, 683. [Google Scholar] [CrossRef] [Green Version]
- Yazawa, A.; Nakazawa, S.; Takeda, Y. Distribution behavior of various elements in copper smelting Systems. In Proceedings of the International Sulfide Smelting Symposium: Advances in Sulfide Smelting, San Francisco, CA, USA, 6–9 November 1983; Volume 1, pp. 99–117. [Google Scholar]
- Kamberovic, J.; Ranitovic, M.; Korac, M.; Jovanovic, N.; Tomovic, B. Pyro-Refining of Mechanically Treated Waste Printed Circuit Boards in DC Arc-Furnace. J. Sustain. Metall. 2018, 4, 251–259. [Google Scholar] [CrossRef]
- Klementtinen, L.; Avarmaa, K.; O’Brien, H.; Taskinen, P. Behavior of Tin and Antimony in Secondary Copper Smelting Process. Minerals 2019, 9, 39. [Google Scholar] [CrossRef] [Green Version]
- Vassileva, V.; Vutova, K. Influence of process parameters on quality of copper in electron-beam melting. J. Phys. Conf. Ser. 2020, 1492, 012014. [Google Scholar] [CrossRef]
- Roine, A.; Kotiranta, T.; Esrola, K.; Lamberg, P. HSC Chemistry v. 7.1; Metso Outotec: Helsinki, Finland, 2011. [Google Scholar]
- Vassileva, V.; Vutova, K.; Mladenov, G. Analysis of the Thermodynamics of Refining during Electron Beam Melting of Refractory Metals. Mater. Und Werkst. 2006, 37, 613–618. [Google Scholar] [CrossRef]
- Phase Diagram of the Cu-Se System. Available online: http://www.himikatus.ru/art/phase-diagr1/Cu-Se.php (accessed on 23 January 2022).
- Phase Diagram of the Cu-Te System. Available online: http://www.himikatus.ru/art/phase-diagr1/Cu-Te.php (accessed on 23 January 2022).
- Phase Diagram of the Cu-Ni System. Available online: http://www.himikatus.ru/art/phase-diagr1/Cu-Ni.php (accessed on 23 January 2022).
- Kawecki, A.; Knych, T.; Sieja-Smaga, E.; Mamala, A.; Kwasniewski, P.; Kiesiewicz, G.; Smyrak, B.; Pacewicz, A. Fabrication, properties, and microstructures of high strength and high conductivity copper-silver wires. Arch. Metall. Mater. 2012, 57, 4. [Google Scholar] [CrossRef]
- Linchevskiy, B.V. Thermodynamics and Kinetics of the Interaction of Gases with Liquid Metals; Metallurgy Publishing House: Moscow, Russia, 1986. (In Russian) [Google Scholar]
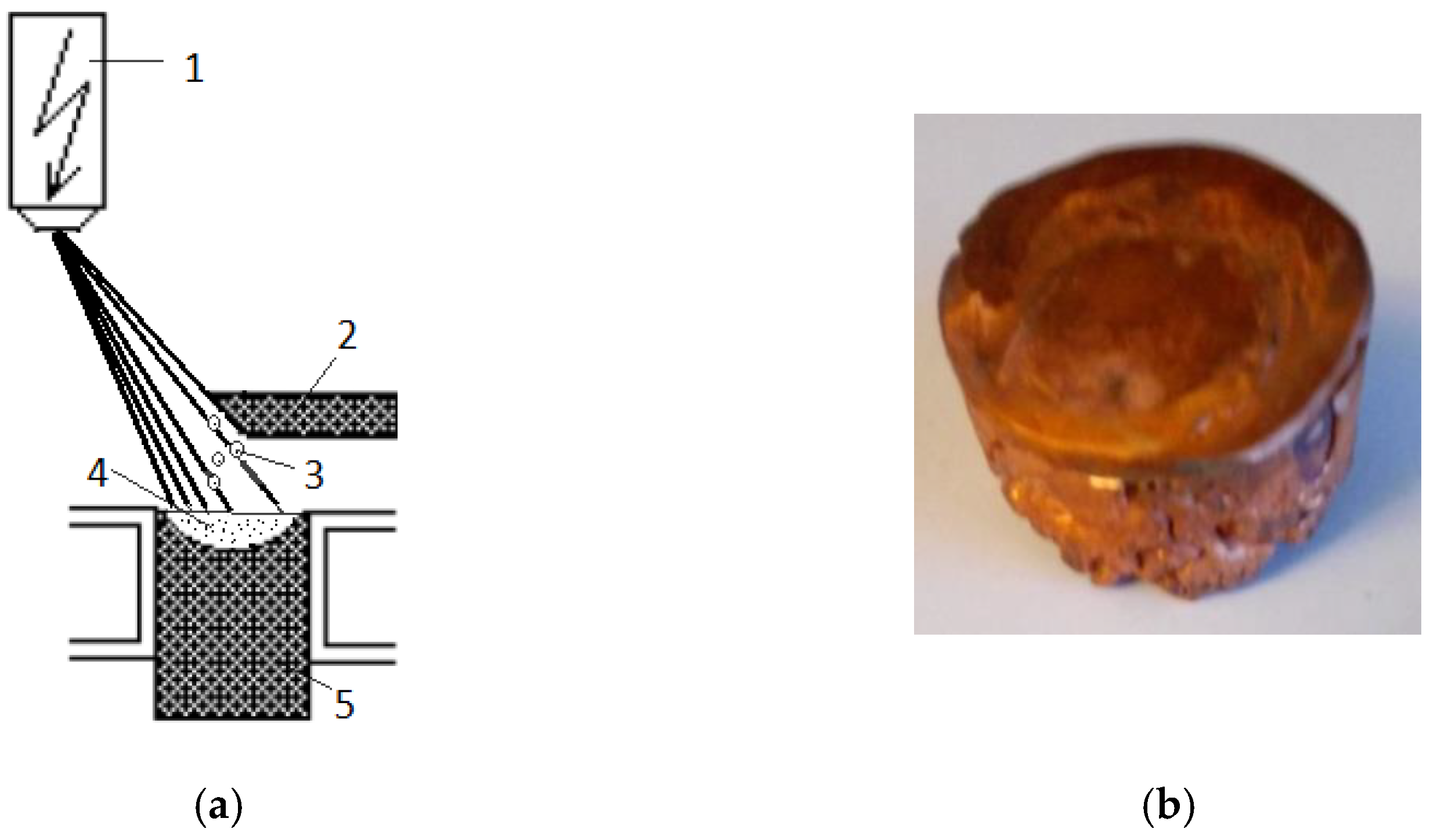



| Probe | Cu-00 | Cu-01 | Cu-02 | Cu-03 | Cu-04 | Cu-05 | Cu-06 | Cu-07 |
|---|---|---|---|---|---|---|---|---|
| Concentration before EBMR | Process Parameters | |||||||
| Type | ppm | T = 1460 K | T = 1500 K | T = 1600 K | T = 1700 K | T = 1800 K | ||
| t = 20 min | t = 15 min | t = 35 min | t = 20 min | t = 20 min | t = 45 min | t = 25 min | ||
| Se, ppm | 382 | 367 | 331 | 309 | 143 | 105 | 107 | 103 |
| Te, ppm | 68 | 66 | 55 | 56 | 12 | 10 | 10 | 11 |
| Bi, ppm | 150 | 122 | 74 | 73 | <10 | <1 | <1 | <1 |
| Pb, ppm | 210 | 115 | 87 | 82 | <3 | <3 | <1 | <1 |
| Sn, ppm | 34 | 24 | 16 | 18 | <2 | <2 | <1 | <1 |
| Sb, ppm | 95 | 60 | 45 | 32 | 15 | <9 | <4 | <3 |
| As, ppm | 700 | 577 | 416 | 403 | 106 | 50 | 50 | <3 |
| Ni, ppm | 1000 | 870 | 740 | 750 | 750 | 660 | 620 | 580 |
| Zn, ppm | 23 | 11 | 8 | 6 | <1 | <1 | <1 | <1 |
| Ag, ppm | 140 | 135 | 133 | 131 | 130 | 131 | 131 | 132 |
| O, ppm | 2251 | 897 | 250 | 50 | 15 | 10 | 10 | 10 |
| S, ppm | 28 | 14 | 7 | 5 | <1 | <1 | <1 | <1 |
| Cu,% | 99.454 | 99.675 | 99.784 | 99.809 | 99.892 | 99.902 | 99.907 | 99.916 |
| 1εtot,% | 40.5 | 60.4 | 65.0 | 78.3 | 82.1 | 83.0 | 84.6 | |
| Wloss,% | 0.42 | 0.86 | 2.07 | 1.63 | 2.93 | 3.56 | 3.06 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vutova, K.; Stefanova, V.; Vassileva, V.; Kadiyski, M. Behaviour of Impurities during Electron Beam Melting of Copper Technogenic Material. Materials 2022, 15, 936. https://doi.org/10.3390/ma15030936
Vutova K, Stefanova V, Vassileva V, Kadiyski M. Behaviour of Impurities during Electron Beam Melting of Copper Technogenic Material. Materials. 2022; 15(3):936. https://doi.org/10.3390/ma15030936
Chicago/Turabian StyleVutova, Katia, Vladislava Stefanova, Vania Vassileva, and Milen Kadiyski. 2022. "Behaviour of Impurities during Electron Beam Melting of Copper Technogenic Material" Materials 15, no. 3: 936. https://doi.org/10.3390/ma15030936
APA StyleVutova, K., Stefanova, V., Vassileva, V., & Kadiyski, M. (2022). Behaviour of Impurities during Electron Beam Melting of Copper Technogenic Material. Materials, 15(3), 936. https://doi.org/10.3390/ma15030936








