Rare Earth Group Separation after Extraction Using Sodium Diethyldithiocarbamate/Polyvinyl Chloride from Lamprophyre Dykes Leachate
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Instrumentations
2.2. Preparation of Sodium Diethyldithiocarbamate Trihydrate/Polyvinyl Chloride
2.3. REE Sorption Studies
2.4. Desorption Studies
2.5. Precipitation of REEs
2.6. Group Separation of REEs by Three-Liquid-Phase Technique
3. Results and Discussion
3.1. Characterization
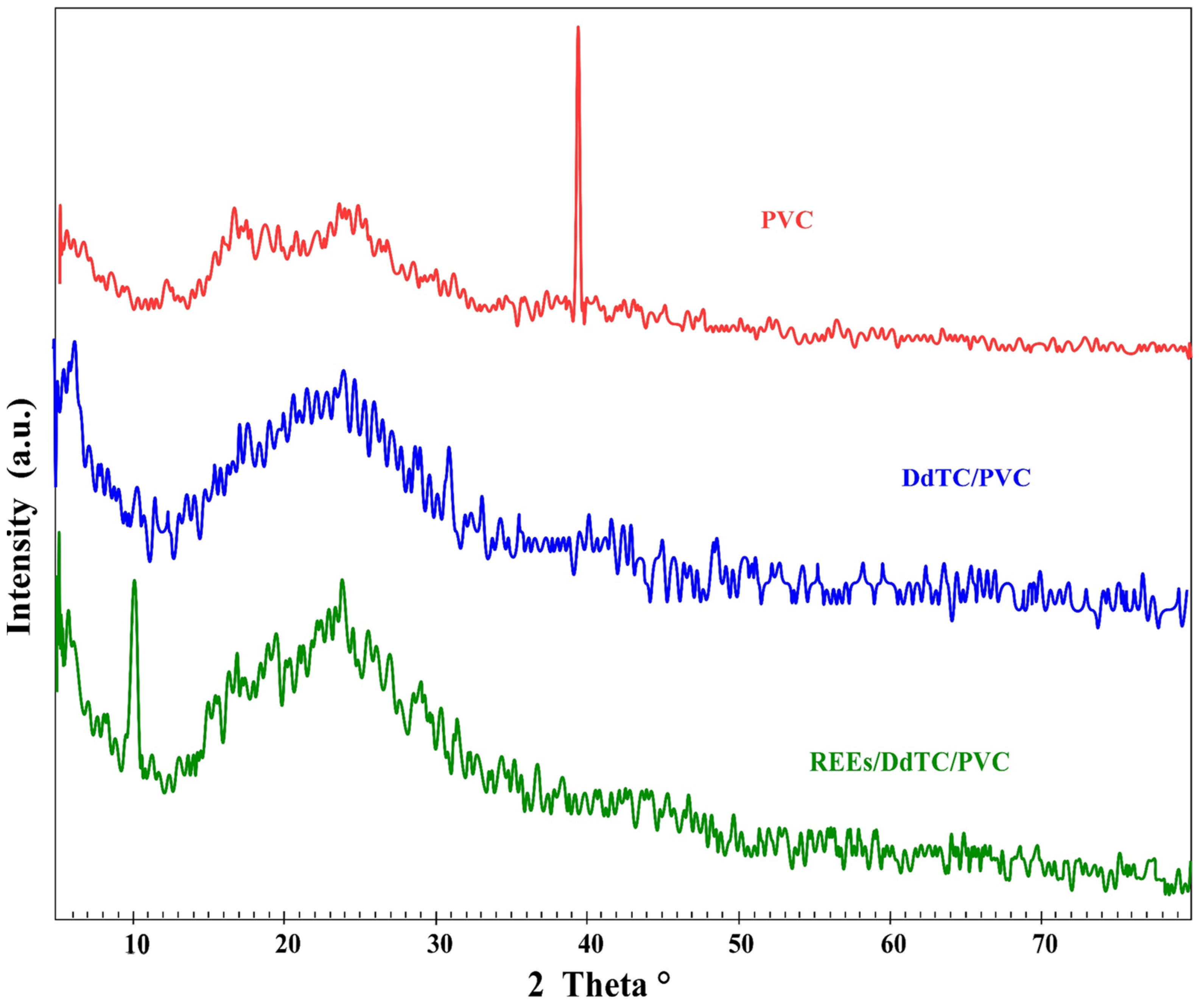
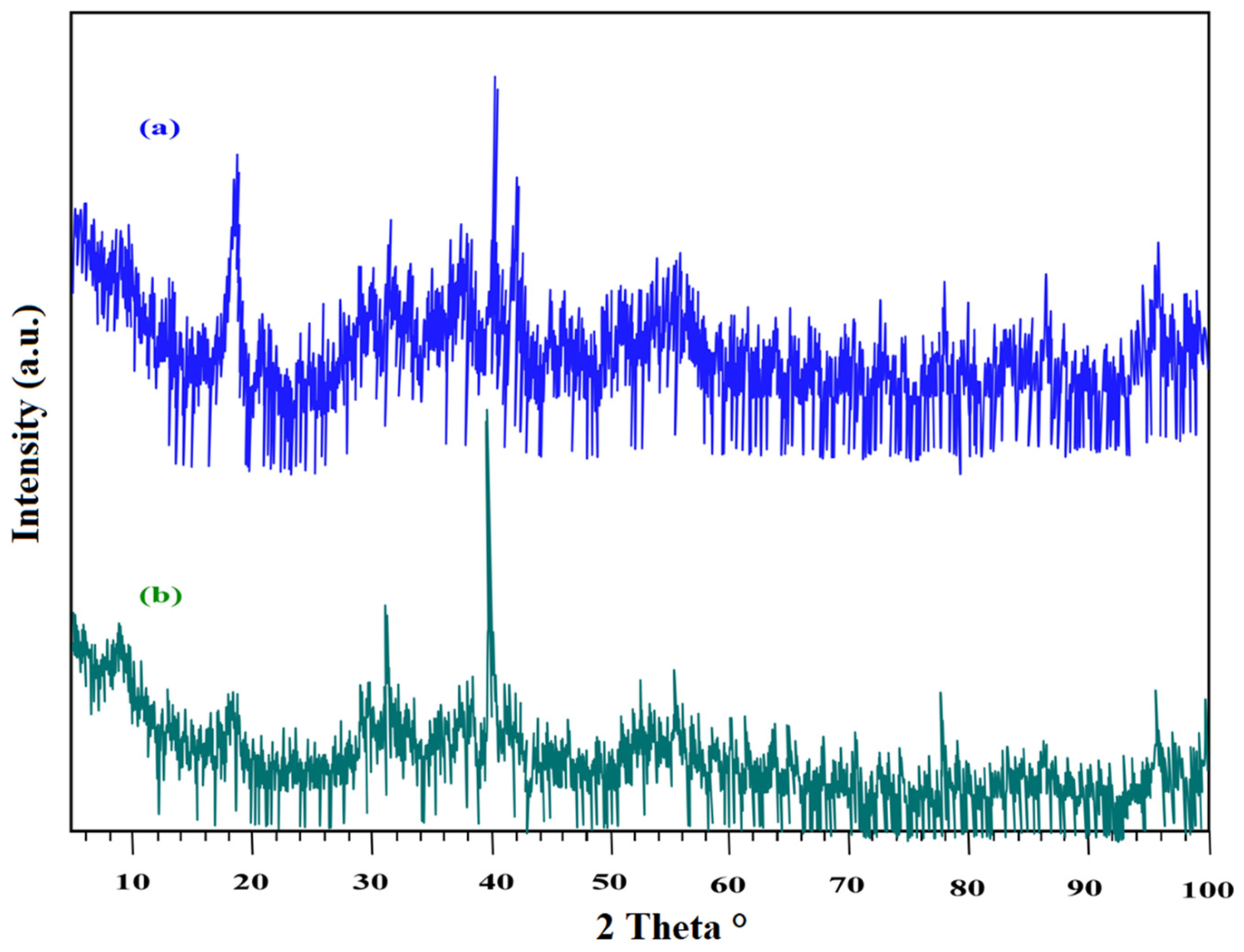
3.1.1. XRD Analysis
3.1.2. SEM/EDX Analysis
3.1.3. BET Surface Analysis
3.1.4. FTIR Analysis
3.1.5. Thermal Analysis
3.2. REE Adsorption
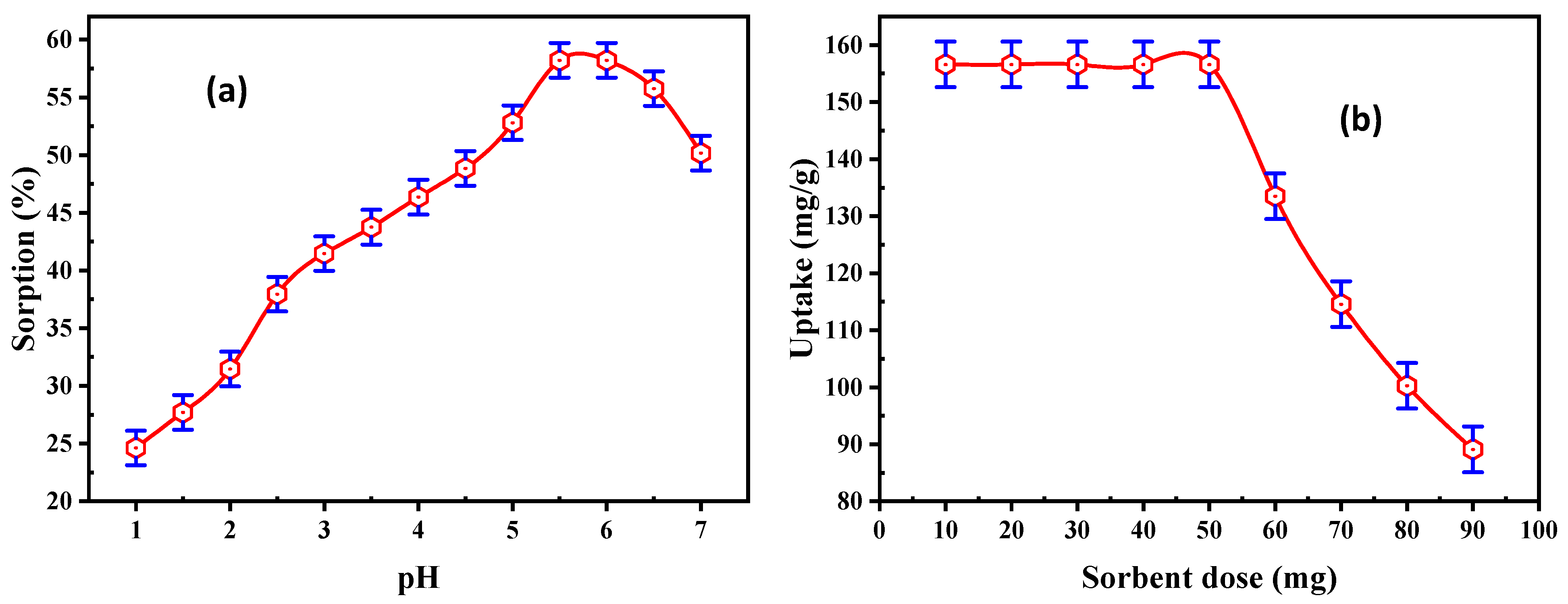
3.2.1. Effect of pH
3.2.2. Effect of Adsorbent Dose
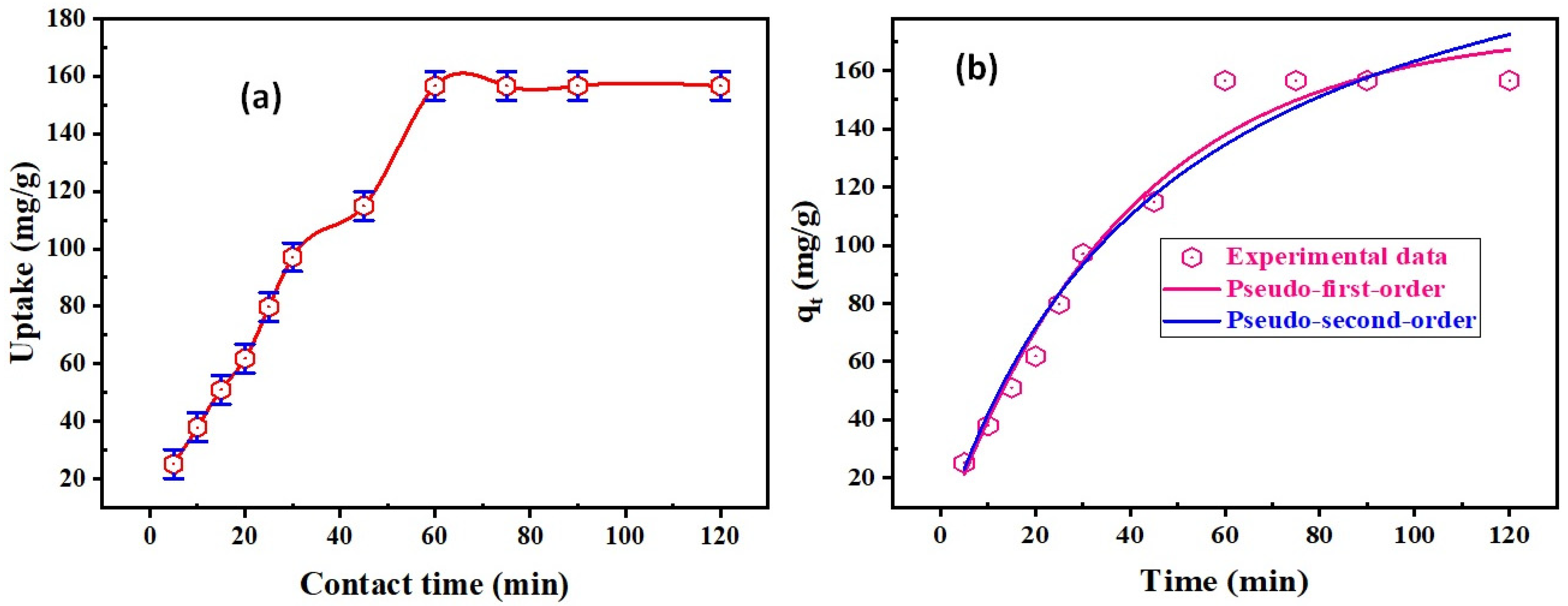
3.2.3. Contact Time and Kinetics
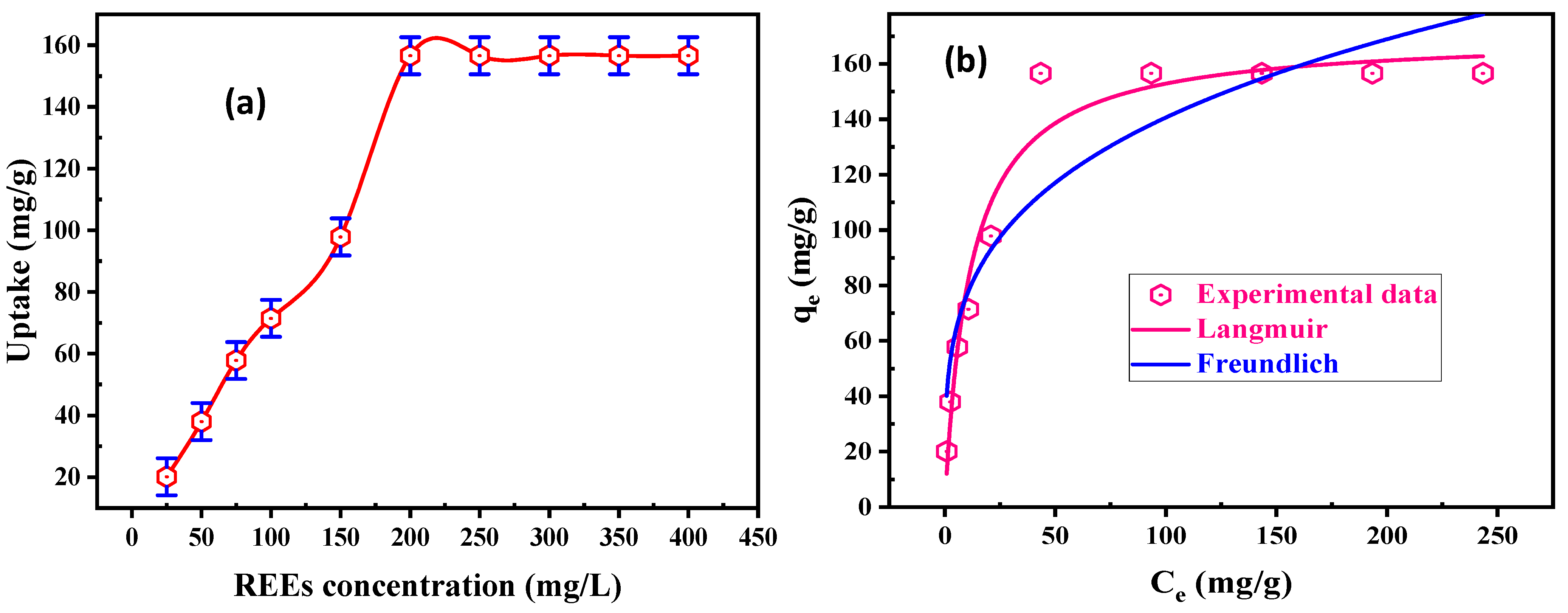
3.2.4. Initial REE Concentration and Equilibrium Isotherm
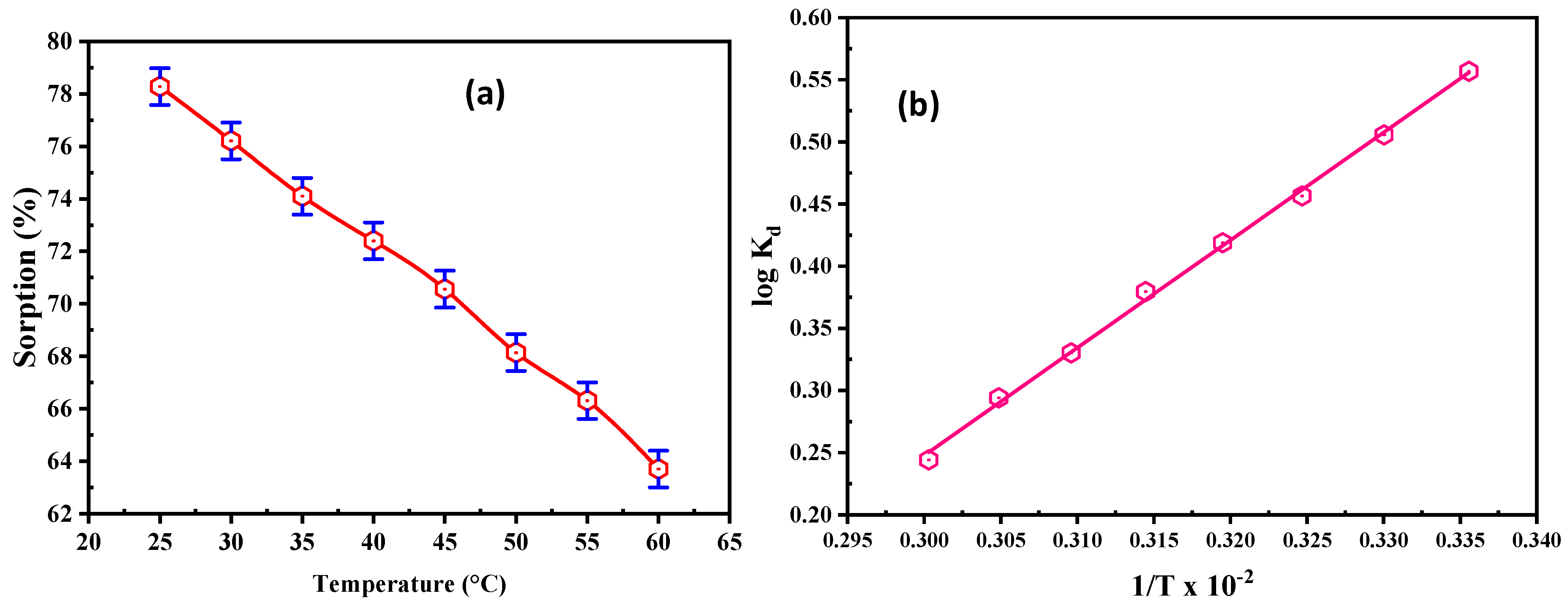
3.2.5. Effect of Temperature and Thermodynamics
3.3. Rare Earth Desorption
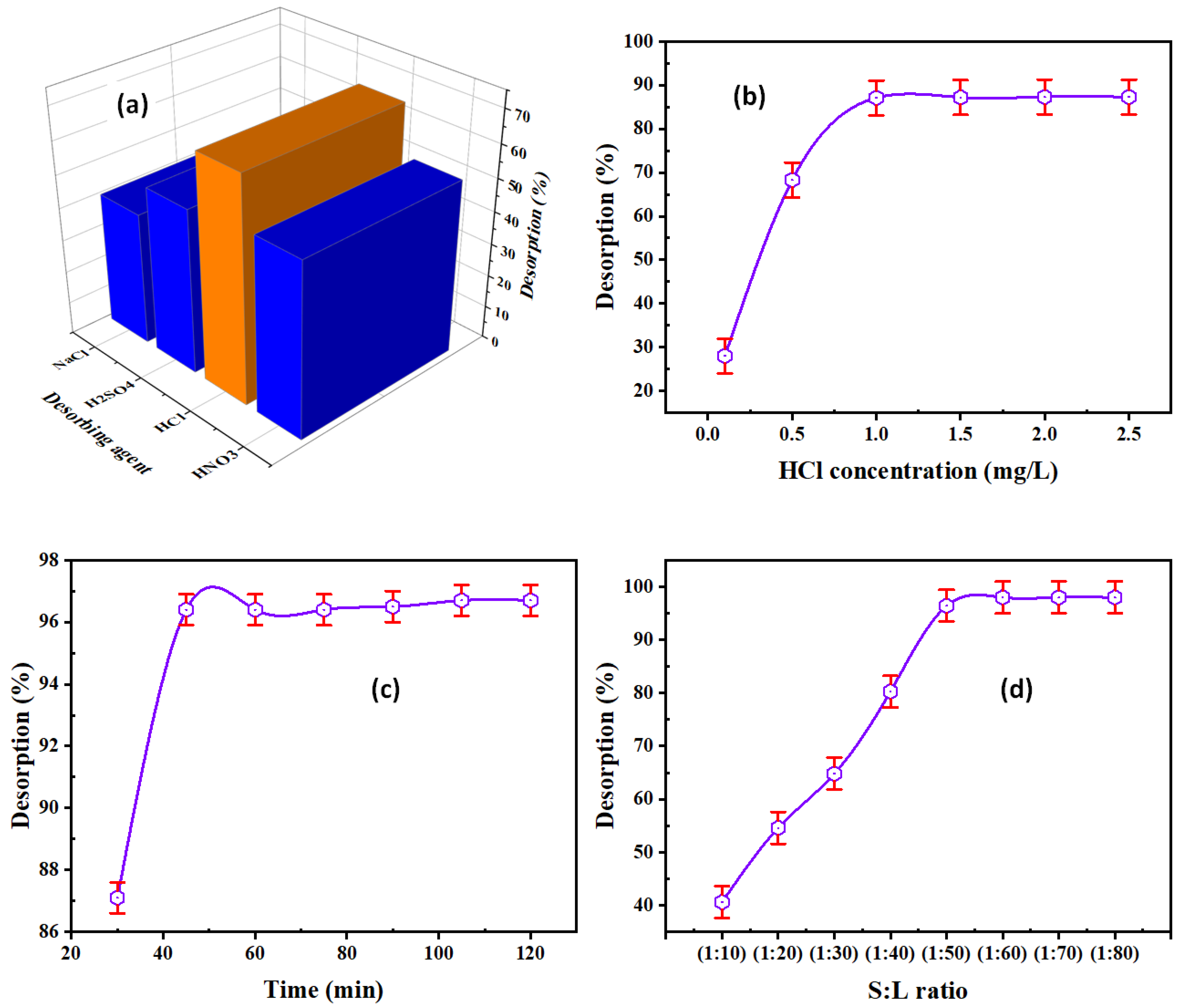
3.3.1. Desorbing Agent Type
3.3.2. HCl Concentration
3.3.3. Contact Time
3.3.4. S:L Phase Ratio
3.4. Regeneration of DdTC/PVC Adsorbent
3.5. Leaching Studies
3.5.1. Characterization
3.5.2. Leaching Investigation
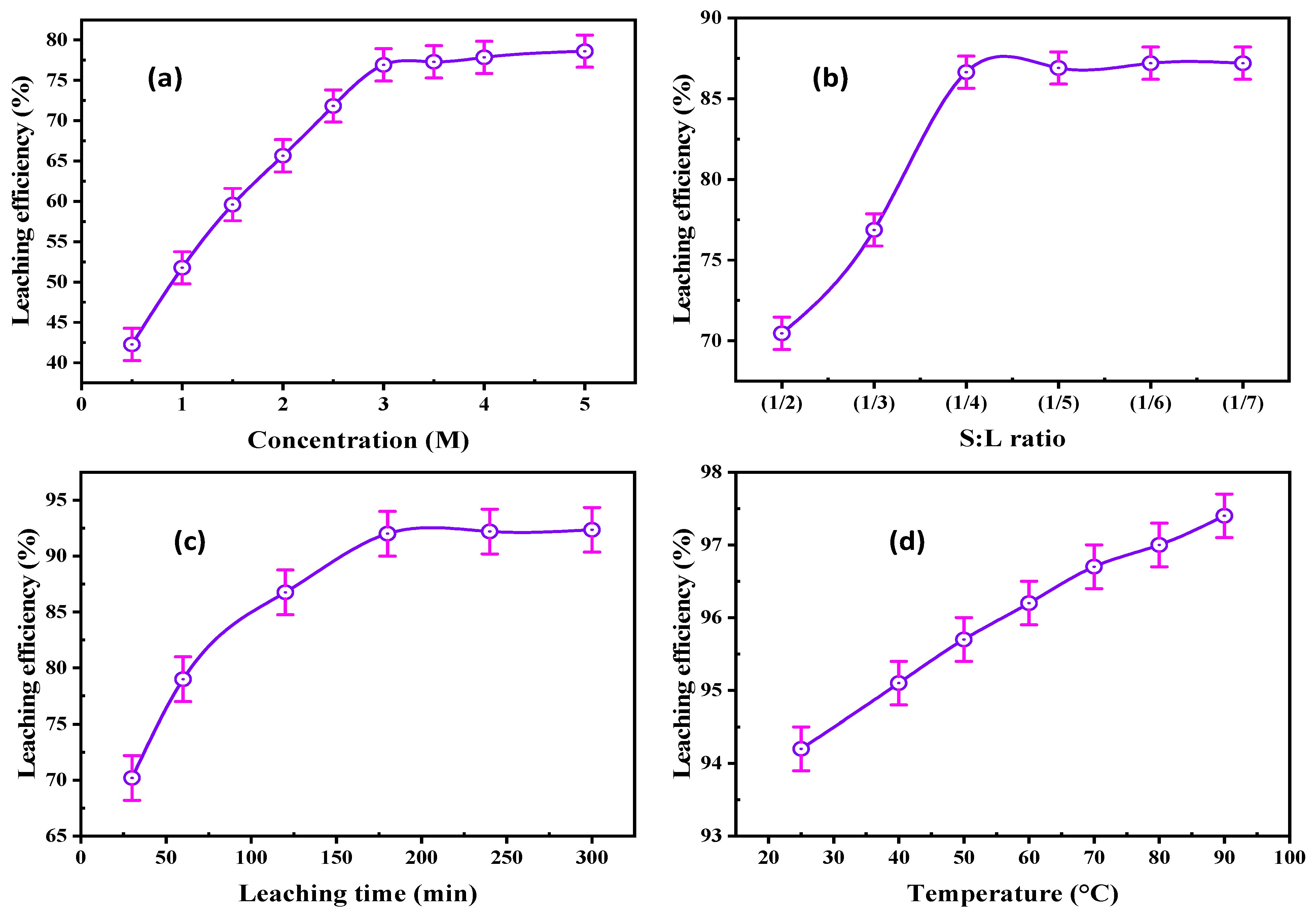
HCl Concentration
Solid/Liquid Ratio
Agitation Time
Leaching Temperature
3.6. Application
3.6.1. Recovery of REEs from Leach Liquor
3.6.2. Precipitation of REEs by Oxalic Acid
3.7. Group Separation of REEs by Three-Liquid-Phase Extraction
3.7.1. REE Partition Parameters on the Three-Liquid Phase
Effect of pH on the Molar Ratio of RE Separation
Effect of DTPA on the Molar Ratio of RE Extraction
Effect of the Type of Polymers
(NH4)2SO4 Content Impact
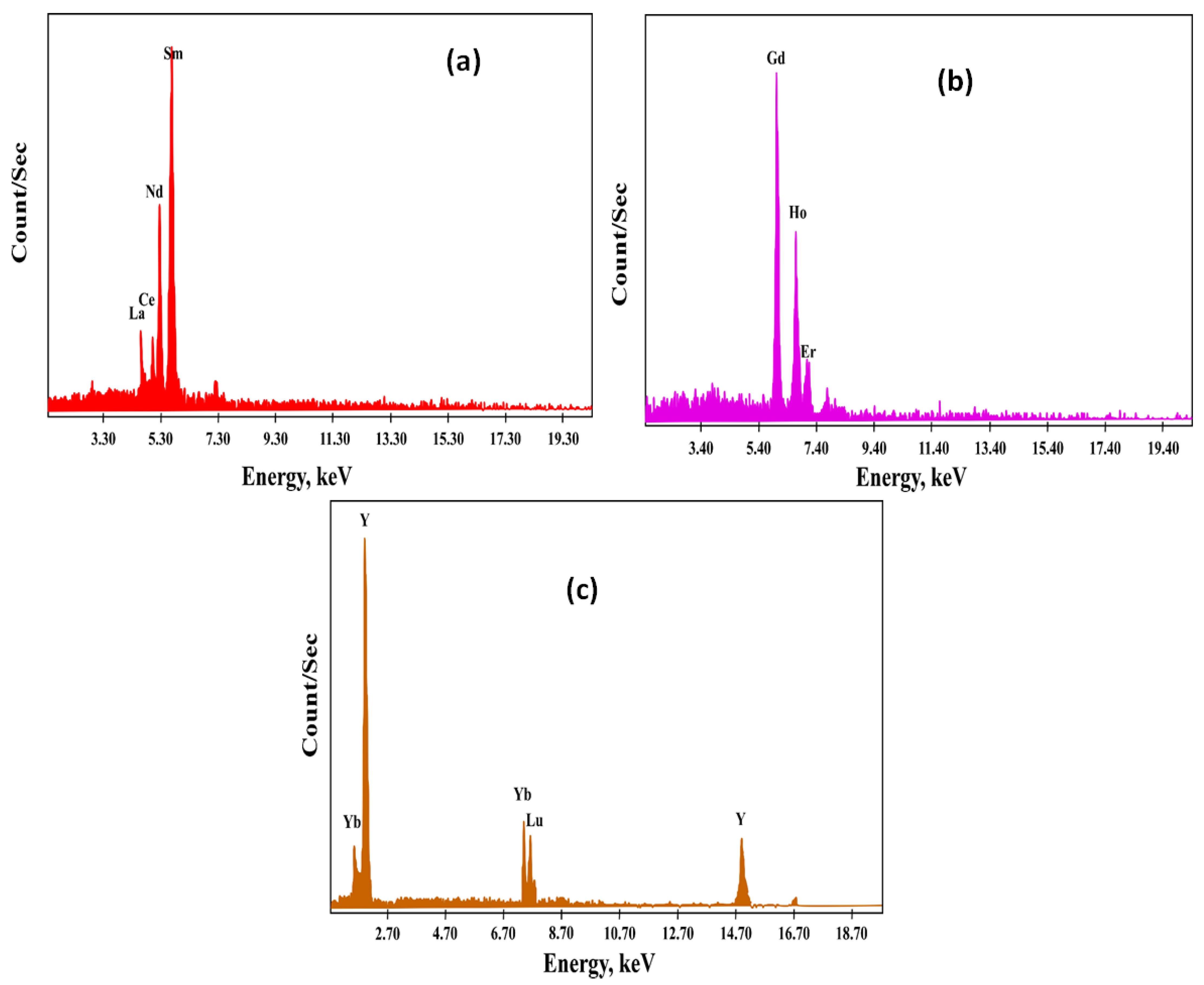
3.7.2. Light, Middle, Heavy RE Ion Recovery
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cheira, M.F. Characteristics of uranium recovery from phosphoric acid by an aminophosphonic resin and application to wet process phosphoric acid. Eur. J. Chem. 2015, 6, 48–56. [Google Scholar] [CrossRef] [Green Version]
- El-Mottaleb, M.A.; Cheira, M.F.; Gouda, G.A.R.; Saiyd, A.A. Leaching of rare earth elements from Egyptian western desert phosphate rocks using HCl. Chem. Adv. Mater. 2016, 1, 33–40. [Google Scholar]
- Cheira, M.F.; Zidan, I.H.; Manaa, E.A. Potentiality of white sand for the purification of wet process phosphoric acid from some metallic elements (U, Zn, Cd). Chem. Technol. Indian J. 2014, 9, 224–233. [Google Scholar]
- Nasab, M.E.; Sam, A.; Milani, S.A. Determination of optimum process conditions for the separation of thorium and rare earth elements by solvent extraction. Hydrometallurgy 2011, 106, 141–147. [Google Scholar] [CrossRef]
- Ronda, C.R.; Jüstel, T.; Nikol, H. Rare earth phosphors: Fundamentals and applications. J. Alloys Compd. 1998, 275–277, 669–676. [Google Scholar] [CrossRef]
- Talan, D.; Huang, Q. Separation of thorium, uranium, and rare earths from a strip solution generated from coarse coal refuse. Hydrometallurgy 2020, 197, 105446. [Google Scholar] [CrossRef]
- Wang, X.; Lei, Y.; Ge, J.; Wu, S. Production forecast of China’s rare earths based on the generalized Weng model and policy recommendations. Resour. Policy 2015, 43, 11–18. [Google Scholar] [CrossRef] [Green Version]
- Xie, F.; Zhang, T.A.; Dreisinger, D.; Doyle, F. A critical review on solvent extraction of rare earths from aqueous solutions. Miner. Eng. 2014, 56, 10–28. [Google Scholar] [CrossRef]
- Ye, S.; Xiao, F.; Pan, Y.X.; Ma, Y.Y.; Zhang, Q.Y. Phosphors in phosphor-converted white light-emitting diodes: Recent advances in materials, techniques and properties. Mater. Sci. Eng. R Rep. 2010, 71, 1–34. [Google Scholar] [CrossRef]
- Sakr, A.K.; Cheira, M.F.; Hassanin, M.A.; Mira, H.I.; Mohamed, S.A.; Khandaker, M.U.; Osman, H.; Eed, E.M.; Sayyed, M.I.; Hanfi, M.Y. Adsorption of Yttrium Ions on 3-Amino-5-Hydroxypyrazole Impregnated Bleaching Clay, a Novel Sorbent Material. Appl. Sci. 2021, 11, 10320. [Google Scholar] [CrossRef]
- Nazarov, M.; Noh, D.Y. Rare earth double activated phosphors for different applications. J. Rare Earths 2010, 28, 1–11. [Google Scholar] [CrossRef]
- Rapaport, A.; Milliez, J.; Bass, M.; Cassanho, A.; Jenssen, H. Review of the properties of up-conversion phosphors for new emissive displays. J. Disp. Technol. 2006, 2, 68. [Google Scholar] [CrossRef]
- Hasad, A.H.; Afifi, S.Y.; Cheira, M.F.; Ghonaim, A.K. Measurement of Sm/Nd isotope ratios in geological samples after rare earth separation using cation and anion exchange resins by inductively coupled plasma mass spectrometry. Arab J. Nucl. Sci. Appl. 2006, 39, 94–108. [Google Scholar]
- Helaly, O.S.; Abd El-Ghany, M.S.; Moustafa, M.I.; Abuzaid, A.H.; Abd El-Monem, N.M.; Ismail, I.M. Extraction of cerium(IV) using tributyl phosphate impregnated resin from nitric acid medium. Trans. Nonferr. Met. Soc. China 2012, 22, 206–214. [Google Scholar] [CrossRef]
- Dave, S.R.; Kaur, H.; Menon, S.K. Selective solid-phase extraction of rare earth elements by the chemically modified Amberlite XAD-4 resin with azacrown ether. React. Funct. Polym. 2010, 70, 692–698. [Google Scholar] [CrossRef]
- Jain, V.K.; Pandya, R.A.; Pillai, S.G.; Agrawal, Y.K.; Kanaiya, P.H. Solid-phase extractive preconcentration and separation of lanthanum(III) and cerium(III) using a polymer-supported chelating calix (4) arene resin. J. Anal. Chem. 2007, 62, 104–112. [Google Scholar] [CrossRef]
- Cheira, M.F.M. Analysis of Rare Earth Elements and Some of Their Isotopes in Geologic Samples Using Inductively Coupled Plasma Mass Spectrometry. Master’s Thesis, Zagazig University, Zagazig, Egypt, 2005. [Google Scholar]
- Nishihama, S.; Kohata, K.; Yoshizuka, K. Separation of lanthanum and cerium using a coated solvent-impregnated resin. Sep. Purif. Technol. 2013, 118, 511–518. [Google Scholar] [CrossRef]
- Shetty, S.; Chernyshova, I.V.; Ponnurangam, S. Foam flotation of rare earth elements by conventional and green surfactants. Miner. Eng. 2020, 158, 106585. [Google Scholar] [CrossRef]
- Jordens, A.; Marion, C.; Grammatikopoulos, T.; Hart, B.; Waters, K.E. Beneficiation of the Nechalacho rare earth deposit: Flotation response using benzohydroxamic acid. Miner. Eng. 2016, 99, 158–169. [Google Scholar] [CrossRef]
- Han, G.; Wen, S.-M.; Wang, H.; Feng, Q.-C. Enhanced sulfidization flotation of cuprite by surface modification with hydrogen peroxide. Trans. Nonferr. Met. Soc. China 2021, 31, 3564–3578. [Google Scholar] [CrossRef]
- Han, G.; Wen, S.; Wang, H.; Feng, Q. Sulfidization regulation of cuprite by pre-oxidation using sodium hypochlorite as an oxidant. Int. J. Min. Sci. Technol. 2021, 31, 1117–1128. [Google Scholar] [CrossRef]
- Wang, Z.H.; Ma, G.X.; Lu, J.; Liao, W.P.; Li, D.Q. Separation of heavy rare earth elements with extraction resin containing 1-hexyl-4-ethyloctyl isopropylphosphonic acid. Hydrometallurgy 2002, 66, 95–99. [Google Scholar] [CrossRef]
- Yadav, K.K.; Singh, D.K.; Anitha, M.; Varshney, L.; Singh, H. Studies on separation of rare earths from aqueous media by polyethersulfone beads containing D2EHPA as extractant. Sep. Purif. Technol. 2013, 118, 350–358. [Google Scholar] [CrossRef]
- Lee, G.S.; Uchikoshi, M.; Mimura, K.; Isshiki, M. Separation of major impurities Ce, Pr, Nd, Sm, Al, Ca, Fe, and Zn from La using bis(2-ethylhexyl)phosphoric acid (D2EHPA)-impregnated resin in a hydrochloric acid medium. Sep. Purif. Technol. 2010, 71, 186–191. [Google Scholar] [CrossRef]
- Soe, N.N.; Shwe, L.T.; Lwin, K.T. Study on Extraction of Lanthanum Oxide from Monazite Concentrate. World Acad. Sci. Eng. Technol. Int. J. Chem. Mol. Nucl. Mater. Metall. Eng. 2008, 2, 226–229. [Google Scholar]
- Jorjani, E.; Shahbazi, M. The production of rare earth elements group via tributyl phosphate extraction and precipitation stripping using oxalic acid. Arab. J. Chem. 2016, 9, S1532–S1539. [Google Scholar] [CrossRef]
- Shen, S.; Chang, Z.; Sun, X.; Hu, X.; Liu, H. Application of block copolymer in three-liquid-phase extraction system. Tsinghua Sci. Technol. 2006, 11, 248–251. [Google Scholar] [CrossRef]
- Yu, P.; Huang, K.; Zhang, C.; Xie, K.; He, X.; Zhao, J.; Deng, F.; Liu, H. Block copolymer micellization induced microphase mass transfer: Partition of Pd(II), Pt(IV) and Rh(III) in three-liquid-phase systems of S201–EOPO–Na2SO4–H2O. J. Colloid Interface Sci. 2011, 362, 228–234. [Google Scholar] [CrossRef]
- Zhang, C.; Huang, K.; Yu, P.; Liu, H. Salting-out induced three-liquid-phase separation of Pt(IV), Pd(II) and Rh(III) in system of S201-acetonitrile-NaCl-water. Sep. Purif. Technol. 2011, 80, 81–89. [Google Scholar] [CrossRef]
- Sui, N.; Huang, K.; Zhang, C.; Wang, N.; Wang, F.; Liu, H. Light, middle, and heavy rare-earth group separation: A new approach via a liquid–liquid–liquid three-phase system. Ind. Eng. Chem. Res. 2013, 52, 5997–6008. [Google Scholar] [CrossRef]
- Lahsen, T.L.; Mohamed, S.A.; Cheira, M.F.; Zaki, D.I.; Allam, E.M. Leaching and recovery of rare earth elements from altered alkaline granite rock from Nusab El-Balgum area, south western desert, Egypt. Int. J. Adv. Res. 2016, 4, 787–801. [Google Scholar]
- Cheira, M.F. Performance of poly sulfonamide/nano-silica composite for adsorption of thorium ions from sulfate solution. SN Appl. Sci. 2020, 2, 398. [Google Scholar] [CrossRef] [Green Version]
- Cheira, M.F.; Rashed, M.N.; Mohamed, A.E.; Hussein, G.M.; Awadallah, M.A. Removal of some harmful metal ions from wet-process phosphoric acid using murexide-reinforced activated bentonite. Mater. Today Chem. 2019, 14, 100176. [Google Scholar] [CrossRef]
- Cheira, M.F.; Rashed, M.N.; Mohamed, A.E.; Zidan, I.H.; Awadallah, M.A. The performance of Alizarin impregnated bentonite for the displacement of some heavy metals ions from the wet phosphoric acid. Sep. Sci. Technol. 2020, 55, 3072–3088. [Google Scholar] [CrossRef]
- Ahmad, N.; Kausar, A.; Muhammad, B. An investigation on 4-aminobenzoic acid modified polyvinyl chloride/graphene oxide and PVC/graphene oxide based nanocomposite membranes. J. Plast. Film Sheeting 2015, 32, 419–448. [Google Scholar] [CrossRef]
- Abd El-Rahem, K.A.; Taher, M.A.; Cheira, M.F. Peculiarities of U(VI) adsorption from acidic solution using chitin-derived chitosan as a low-priced bio-adsorbent. Al-Azhar Bull. Sci. 2021, 32, 57–70. [Google Scholar] [CrossRef]
- Góbi, S.; Reva, I.; Tarczay, G.; Fausto, R. Amorphous and crystalline thioacetamide ice: Infrared spectra as a probe for temperature and structure. J. Mol. Struct. 2020, 1220, 128719. [Google Scholar] [CrossRef]
- Lv, Y.; Liu, J.; Luo, Z.; Wang, H.; Wei, Z. Construction of chain segment structure models, and effects on the initial stage of the thermal degradation of poly(vinyl chloride). RSC Adv. 2017, 7, 37268–37275. [Google Scholar] [CrossRef] [Green Version]
- Roy, K.J.; Anjali, T.V.; Sujith, A. Asymmetric membranes based on poly(vinyl chloride): Effect of molecular weight of additive and solvent power on the morphology and performance. J. Mater. Sci. 2017, 52, 5708–5725. [Google Scholar] [CrossRef]
- Ho, Y.-S. Effect of pH on lead removal from water using tree fern as the sorbent. Bioresour. Technol. 2005, 96, 1292–1296. [Google Scholar] [CrossRef] [PubMed]
- Chairat, M.; Rattanaphani, S.; Bremner, J.B.; Rattanaphani, V. An adsorption and kinetic study of lac dyeing on silk. Dye. Pigment. 2005, 64, 231–241. [Google Scholar] [CrossRef]
- Cheira, M.F. Synthesis of aminophosphonate-functionalised ZnO/polystyrene-butadiene nanocomposite and its characteristics for uranium adsorption from phosphoric acid. Int. J. Environ. Anal. Chem. 2021, 101, 1710–1734. [Google Scholar] [CrossRef]
- Hassanin, M.A.; El-Gendy, H.S.; Cheira, M.F.; Atia, B.M. Uranium ions extraction from the waste solution by thiosemicarbazide anchored cellulose acetate. Int. J. Environ. Anal. Chem. 2021, 101, 351–369. [Google Scholar] [CrossRef]
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef] [Green Version]
- Abdien, H.G.; Cheira, M.F.; Abd-Elraheem, M.; El-Naser, T.A.S.; Zidan, I.H. Extraction and pre-concentration of uranium using activated carbon impregnated trioctyl phosphine oxide. Elixir Appl. Chem. 2016, 100, 43462–43469. [Google Scholar]
- Gomaa, H.; Cheira, M.F.; Abd-Elmottaleb, M.; El-Naser, T.A.S.; Zidan, I.H. Removal of uranium from acidic solution using activated carbon impregnated with tri butyl phosphate. Biol. Chem. Res. 2016, 2016, 313–340. [Google Scholar]
- Gomaa, H.; Shenashen, M.A.; Elbaz, A.; Kawada, S.; Seaf El-Nasr, T.A.; Cheira, M.F.; Eid, A.I.; El-Safty, S.A. Inorganic-organic mesoporous hybrid segregators for selective and sensitive extraction of precious elements from urban mining. J. Colloid Interface Sci. 2021, 604, 61–79. [Google Scholar] [CrossRef]
- Cui, J.; Wang, Q.; Gao, J.; Guo, Y.; Cheng, F. The selective adsorption of rare earth elements by modified coal fly ash based SBA-15. Chin. J. Chem. Eng. 2021. [Google Scholar] [CrossRef]
- Chen, L.; Dong, H.; Pan, W.; Dai, J.; Dai, X.; Pan, J. Poly (vinyl alcohol-co-ethylene) (EVOH) modified polymer inclusion membrane in heavy rare earths separation with advanced hydrophilicity and separation property. Chem. Eng. J. 2021, 426, 131305. [Google Scholar] [CrossRef]
- Shu, Q.; Liao, C.-F.; Zou, W.-Q.; Xu, B.-Q.; Tan, Y.-H. Recovery of rare earth element ytterbium(III) by dried powdered biomass of spirulina: Adsorption isotherm, kinetic and thermodynamic study. Trans. Nonferr. Met. Soc. China 2021, 31, 1127–1139. [Google Scholar] [CrossRef]
- Javadian, H.; Ruiz, M.; Taghvai, M.; Sastre, A.M. Novel magnetic nanocomposite of calcium alginate carrying poly(pyrimidine-thiophene-amide) as a novel green synthesized polyamide for adsorption study of neodymium, terbium, and dysprosium rare-earth ions. Colloids Surf. A Physicochem. Eng. Asp. 2020, 603, 125252. [Google Scholar] [CrossRef]
- Alakhras, F.A.; Dari, K.A.; Mubarak, M.S. Synthesis and chelating properties of some poly(amidoxime-hydroxamic acid) resins toward some trivalent lanthanide metal ions. J. Appl. Polym. Sci. 2005, 97, 691–696. [Google Scholar] [CrossRef]
- Zhao, X.; Jiang, X.; Peng, D.; Teng, J.; Yu, J. Behavior and mechanism of graphene oxide-tris(4-aminophenyl)amine composites in adsorption of rare earth elements. J. Rare Earths 2021, 39, 90–97. [Google Scholar] [CrossRef]
- Atia, B.M.; Gado, M.A.; Cheira, M.F.; El-Gendy, H.S.; Yousef, M.A.; Hashem, M.D. Direct synthesis of a chelating carboxamide derivative and its application for thorium extraction from Abu Rusheid ore sample, South Eastern Desert, Egypt. Int. J. Environ. Anal. Chem. 2021, 1–24. [Google Scholar] [CrossRef]
- Atia, B.M.; Khawassek, Y.M.; Hussein, G.M.; Gado, M.A.; El-Sheify, M.A.; Cheira, M.F. One-pot synthesis of pyridine dicarboxamide derivative and its application for uranium separation from acidic medium. J. Environ. Chem. Eng. 2021, 9, 105726. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Insights into the modeling of adsorption isotherm systems. Chem. Eng. J. 2010, 156, 2–10. [Google Scholar] [CrossRef]
- Sayed, A.S.; Abdelmottaleb, M.; Cheira, M.F.; Abdel-Aziz, G.; Gomaa, H.; Hassanein, T.F. Date seed as an efficient, eco-friendly, and cost-effective bio-adsorbent for removal of thorium ions from acidic solutions. Aswan Univ. J. Environ. Stud. 2020, 1, 106–124. [Google Scholar] [CrossRef]
- Ibrahim, M.E.; Saleh, G.M.; Dawood, N.A.; Aly, G.M. Ocellar lamprophyre dyke bearing mineralization, Wadi Nugrus, eastern desert, Egypt: Geology, mineralogy and geochemical implications. Chin. J. Geochem. 2010, 29, 383–392. [Google Scholar] [CrossRef]
- Bakry, A.R.; Demerdash, M.; Manaa, E.A.; Abd El Fatah, A.I.L. Recovery of uranium and rare earth elements from western desert phosphate rocks with EDTA and nitric acid solutions. Radiochemistry 2021, 63, 297–306. [Google Scholar] [CrossRef]
- Eshuis, A.; van Elderen, G.R.A.; Koning, C.A.J. A descriptive model for the homogeneous precipitation of zinc sulfide from acidic zinc salt solutions. Colloids Surf. A Physicochem. Eng. Asp. 1999, 151, 505–512. [Google Scholar] [CrossRef]
- Sakr, A.; Mohamed, S.; Mira, H.; Cheira, M. Successive leaching of uranium and rare earth elements from El Sela mineralization. J. Sci. Eng. Res. 2018, 5, 95–111. [Google Scholar]
- Veeken, A.H.M.; Akoto, L.; Hulshoff Pol, L.W.; Weijma, J. Control of the sulfide (S2−) concentration for optimal zinc removal by sulfide precipitation in a continuously stirred tank reactor. Water Res. 2003, 37, 3709–3717. [Google Scholar] [CrossRef]
- Esposito, G.; Veeken, A.; Weijma, J.; Lens, P.N.L. Use of biogenic sulfide for ZnS precipitation. Sep. Purif. Technol. 2006, 51, 31–39. [Google Scholar] [CrossRef]
- Karmakar, R.; Sen, K. Aqueous biphasic extraction of metal ions: An alternative technology for metal regeneration. J. Mol. Liq. 2019, 273, 231–247. [Google Scholar] [CrossRef]
- Bulgariu, L.; Bulgariu, D. The partition behavior of Zn(II) using halide ions extractants in aqueous peg-based two-phase systems. Sep. Sci. Technol. 2007, 42, 1093–1106. [Google Scholar] [CrossRef]
- Tubío, G.; Pellegrini, L.; Nerli, B.B.; Picó, G.A. Liquid−liquid equilibria of aqueous two-phase systems containing poly(ethylene glycols) of different molecular weight and sodium citrate. J. Chem. Eng. Data 2006, 51, 209–212. [Google Scholar] [CrossRef]
- Silva, R.A.; Goulart Silva, G.; Moreira, R.L.; Pimenta, M.A. The effects of salt concentration on cation complexation in triblock-polyether electrolyte. Phys. Chem. Chem. Phys. 2003, 5, 2424–2428. [Google Scholar] [CrossRef]
- Wang, X.; Li, W.; Li, D. Extraction and stripping of rare earths using mixtures of acidic phosphorus-based reagents. J. Rare Earths 2011, 29, 413–415. [Google Scholar] [CrossRef]




| Materials | SBET, m2/g | Pore-Volume, cc/g | Pore-Size, nm |
|---|---|---|---|
| DdTC/PVC | 90.14 | 0.087 | 1.97 |
| REEs/DdTC/PVC | 86.53 | 0.084 | 1.89 |
| Adsorbent Materials | Uptake, mg/g | Reference |
|---|---|---|
| Modified coal fly ash-based SBA-15 | 32.0 | [49] |
| Poly (vinyl alcohol-co-ethylene) (EVOH) modified polymer | 22.05 | [50] |
| Powdered spirulina | 72.46 | [51] |
| Magnetic nanocomposite of calcium alginate carrying poly(pyrimidine-thiophene-amide) | 113.08 | [52] |
| Poly(amidoxime-hydroxamic acid) resin | 125.0 | [53] |
| Graphene oxide-tris(4-aminophenyl)amine composite | 30.88 | [54] |
| Sodium diethyldithiocarbamate/polyvinyl chloride (DdTC/PVC) | 156.6 | This study |
| Parameters | Temperature, K | DdTC/PVC |
|---|---|---|
| ΔG°, kJ/mol | 298 | −3.158 |
| 303 | −2.938 | |
| 308 | −2.718 | |
| 313 | −2.498 | |
| 318 | −2.278 | |
| 323 | −2.058 | |
| 328 | −1.838 | |
| 333 | −1.618 | |
| ΔH°, kJ/mol | −16.27 | |
| ΔS°, kJ/mol K | −0.0440 | |
| Major Oxides | Wt., % | Trace Metal Ions | mg/kg | REE Ions | mg/kg |
|---|---|---|---|---|---|
| SiO2 | 46.3 | U6+ | 400 | La3+ | 90 |
| Al2O3 | 16.8 | Ba2+ | 167 | Ce3+ | 160 |
| TiO2 | 3.4 | Pb2+ | 596 | Pr3+ | 170 |
| Fe2O3 | 15.36 | V5+ | 240 | Nd3+ | 143 |
| MnO | 0.65 | Cu2+ | 367 | Sm3+ | 26 |
| MgO | 0.06 | Ni2+ | 86.7 | Gd3+ | 60 |
| CaO | 2.3 | Cd2+ | 49.5 | Ho3+ | 171 |
| K2O | 2.8 | Zn2+ | 6348 | Er3+ | 145 |
| Na2O | 0.48 | Th4+ | 39 | Yb3+ | 550 |
| P2O5 | 1.02 | Lu3+ | 65 | ||
| LOI * | 8.75 | Y3+ | 3274 | ||
| Total | 97.92 |
| Metal Ions | g/L | Metal Ions | mg/L | RE Ions | mg/L |
|---|---|---|---|---|---|
| Si4+ | 1.35 | U6+ | 94 | La3+ | 21 |
| Al3+ | 2.34 | Ba2+ | 44 | Ce3+ | 37 |
| Ti4+ | 0.32 | Pb2+ | 103 | Pr3+ | 40 |
| Fe3+ | 3.57 | V5+ | 51 | Nd3+ | 34 |
| Mn2+ | 0.5 | Cu2+ | 85 | Sm3+ | 6 |
| Mg2+ | 0.03 | Ni2+ | 19 | Gd3+ | 14 |
| Ca2+ | 0.67 | Th4+ | 5 | Ho3+ | 40 |
| K+ | 0.73 | Zn2+ | 620 | Er3+ | 34 |
| Na+ | 0.1 | Yb3+ | 129 | ||
| P5+ | 0.12 | Lu3+ | 16 | ||
| Y3+ | 769 | ||||
| Metal Ions | % | Metal Ions | % |
|---|---|---|---|
| La3+ | 0.72 | Ho3+ | 1.38 |
| Ce3+ | 1.39 | Yb3+ | 4.57 |
| Pr3+ | 1.30 | Lu3+ | 0.57 |
| Nd3+ | 1.19 | Y3+ | 27.89 |
| Sm3+ | 0.21 | H2O (at 110 °C) | 13.52 |
| Gd3+ | 0.51 | CO2 (at 550 °C) | 43.61 |
| Total | 96.87 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Allam, E.M.; Lashen, T.A.; Abou El-Enein, S.A.; Hassanin, M.A.; Sakr, A.K.; Cheira, M.F.; Almuqrin, A.; Hanfi, M.Y.; Sayyed, M.I. Rare Earth Group Separation after Extraction Using Sodium Diethyldithiocarbamate/Polyvinyl Chloride from Lamprophyre Dykes Leachate. Materials 2022, 15, 1211. https://doi.org/10.3390/ma15031211
Allam EM, Lashen TA, Abou El-Enein SA, Hassanin MA, Sakr AK, Cheira MF, Almuqrin A, Hanfi MY, Sayyed MI. Rare Earth Group Separation after Extraction Using Sodium Diethyldithiocarbamate/Polyvinyl Chloride from Lamprophyre Dykes Leachate. Materials. 2022; 15(3):1211. https://doi.org/10.3390/ma15031211
Chicago/Turabian StyleAllam, Eman M., Taysser A. Lashen, Saeyda A. Abou El-Enein, Mohamed A. Hassanin, Ahmed K. Sakr, Mohamed F. Cheira, Aljawhara Almuqrin, Mohamed Y. Hanfi, and M. I. Sayyed. 2022. "Rare Earth Group Separation after Extraction Using Sodium Diethyldithiocarbamate/Polyvinyl Chloride from Lamprophyre Dykes Leachate" Materials 15, no. 3: 1211. https://doi.org/10.3390/ma15031211
APA StyleAllam, E. M., Lashen, T. A., Abou El-Enein, S. A., Hassanin, M. A., Sakr, A. K., Cheira, M. F., Almuqrin, A., Hanfi, M. Y., & Sayyed, M. I. (2022). Rare Earth Group Separation after Extraction Using Sodium Diethyldithiocarbamate/Polyvinyl Chloride from Lamprophyre Dykes Leachate. Materials, 15(3), 1211. https://doi.org/10.3390/ma15031211








