Effective BMP-2 Release and Mineralization on a Graphene Oxide/Polyvinylpyrrolidone Hydrogel Forming Poly (ε-Caprolactone) Nanofibrous Scaffolds
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
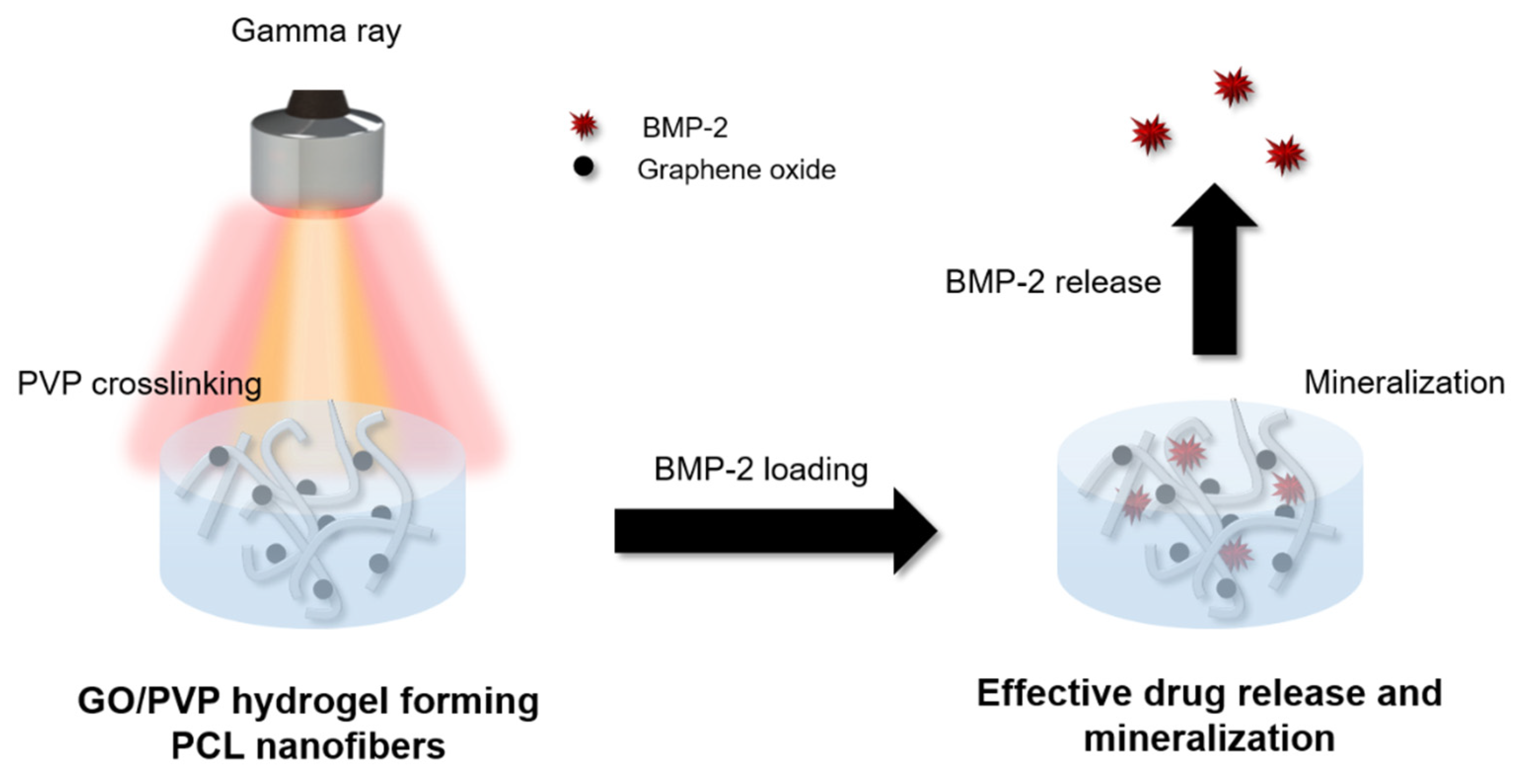
2.2. Preparation of the GO/PVP Hydrogel Forming the PCL Nanofibrous Scaffolds
2.3. Characterization of the GO/PVP Hydrogel Forming PCL Nanofibrous Scaffolds
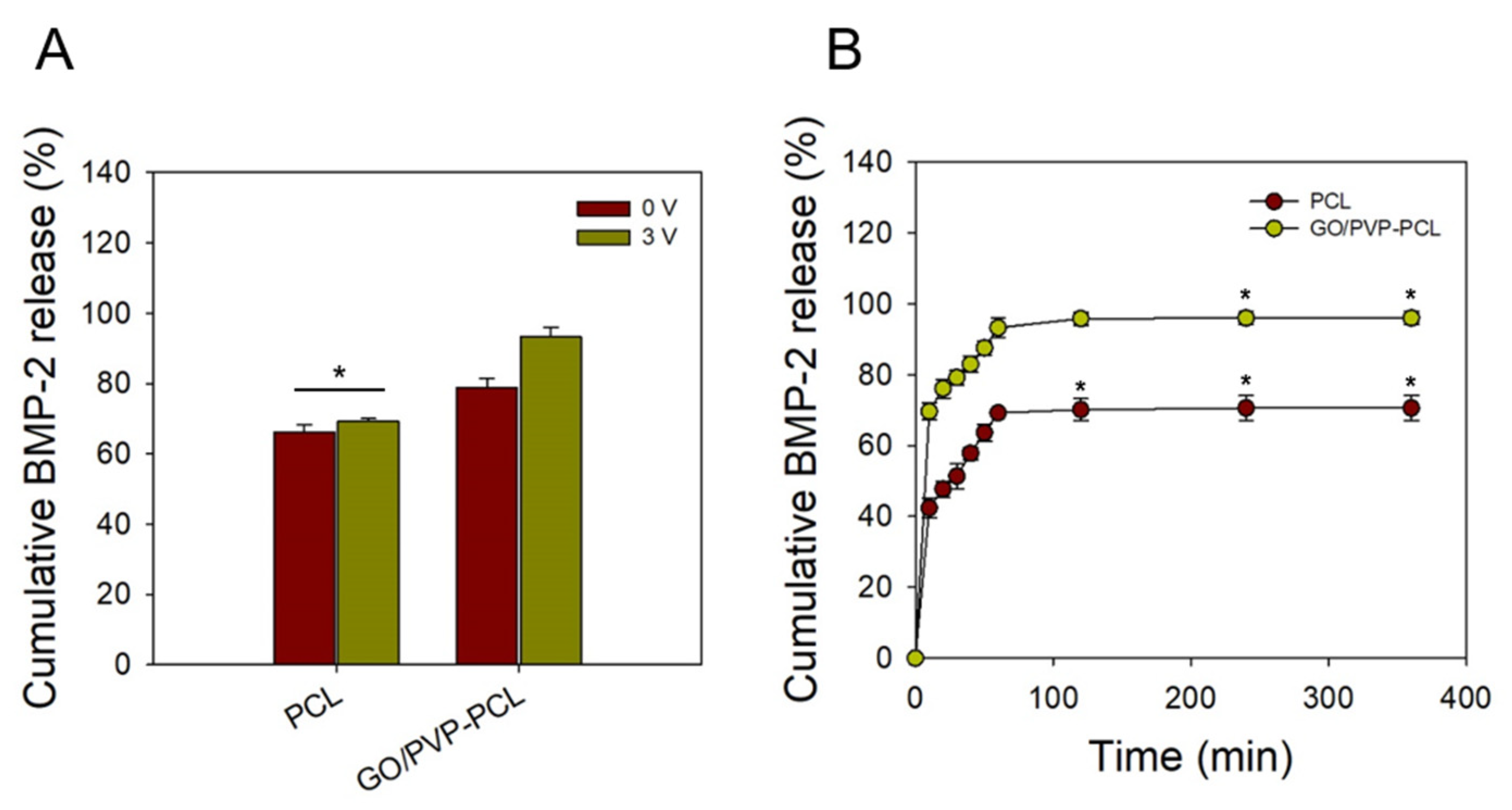
2.4. BMP-2 Release Test
2.5. In Vitro Cytocompatibility
2.6. Mineralization and Alkaline phosphatase (ALP) Activity Test
2.7. Statistical Analysis
3. Results and Discussion
3.1. Preparation of the GO/PVP Hydrogel Forming the PCL Nanofibrous Scaffolds
3.2. Characterization of the GO/PVP Hydrogel Forming PCL Nanofibrous Scaffolds
3.3. In Vitro Cytocompatibility
3.4. BMP-2 Release Test
3.5. Mineralization and ALP Activity Test
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Liao, S.; Li, B.; Ma, Z.; Wei, H.; Chan, C.; Ramakrishna, S. Biomimetic Electrospun Nanofibers for Tissue Regeneration. Biomed. Mater. 2006, 1, R45–R53. [Google Scholar] [CrossRef] [PubMed]
- Barnes, C.P.; Sell, S.A.; Boland, E.D.; Simpson, D.G.; Bowlin, G.L. Nanofiber Technology: Designing the next Generation of Tissue Engineering Scaffolds. Adv. Drug Deliv. Rev. 2007, 59, 1413–1433. [Google Scholar] [CrossRef] [PubMed]
- Venugopal, J.; Low, S.; Choon, A.T.; Ramakrishna, S. Interaction of Cells and Nanofiber Scaffolds in Tissue Engineering. J. Biomed. Mater. Res. Part B Appl. Biomater. 2008, 84B, 34–48. [Google Scholar] [CrossRef]
- Smith, L.A.; Ma, P.X. Nano-Fibrous Scaffolds for Tissue Engineering. Colloids Surf. B Biointerfaces 2004, 39, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Miszuk, J.M.; Zhao, Y.; Sun, H.; Fong, H. Electrospun Polycaprolactone 3D Nanofibrous Scaffold with Interconnected and Hierarchically Structured Pores for Bone Tissue Engineering. Adv. Healthc. Mater. 2015, 4, 2238–2246. [Google Scholar] [CrossRef]
- Khajavi, R.; Abbasipour, M.; Bahador, A. Electrospun Biodegradable Nanofibers Scaffolds for Bone Tissue Engineering. J. Appl. Polym. Sci. 2016, 42883. [Google Scholar] [CrossRef]
- Gao, Y.; Shao, W.; Qian, W.; He, J.; Zhou, Y.; Qi, K.; Wang, L.; Cui, S.; Wang, R. Biomineralized Poly (l-Lactic-Co-Glycolic Acid)-Tussah Silk Fibroin Nanofiber Fabric with Hierarchical Architecture as a Scaffold for Bone Tissue Engineering. Mater. Sci. Eng. C 2018, 84, 195–207. [Google Scholar] [CrossRef]
- Lin, W.; Chen, M.; Qu, T.; Li, J.; Man, Y. Three-Dimensional Electrospun Nanofibrous Scaffolds for Bone Tissue Engineering. J. Biomed. Mater. Res. Part B Appl. Biomater. 2020, 108, 1311–1321. [Google Scholar] [CrossRef]
- Yao, Q.; Cosme, J.G.L.; Xu, T.; Miszuk, J.M.; Picciani, P.H.S.; Fong, H.; Sun, H. Three Dimensional Electrospun PCL/PLA Blend Nanofibrous Scaffolds with Significantly Improved Stem Cells Osteogenic Differentiation and Cranial Bone Formation. Biomaterials 2017, 115, 115–127. [Google Scholar] [CrossRef]
- Ye, K.; Liu, D.; Kuang, H.; Cai, J.; Chen, W.; Sun, B.; Xia, L.; Fang, B.; Morsi, Y.; Mo, X. Three-Dimensional Electrospun Nanofibrous Scaffolds Displaying Bone Morphogenetic Protein-2-Derived Peptides for the Promotion of Osteogenic Differentiation of Stem Cells and Bone Regeneration. J. Colloid Interface Sci. 2019, 534, 625–636. [Google Scholar] [CrossRef]
- Pilipchuk, S.P.; Plonka, A.B.; Monje, A.; Taut, A.D.; Lanis, A.; Kang, B.; Giannobile, W.V. Tissue Engineering for Bone Regeneration and Osseointegration in the Oral Cavity. Dent. Mater. 2015, 31, 317–338. [Google Scholar] [CrossRef]
- Koons, G.L.; Diba, M.; Mikos, A.G. Materials Design for Bone-Tissue Engineering. Nat. Rev. Mater. 2020, 5, 584–603. [Google Scholar] [CrossRef]
- Li, J.J.; Ebied, M.; Xu, J.; Zreiqat, H. Current Approaches to Bone Tissue Engineering: The Interface between Biology and Engineering. Adv. Healthc. Mater. 2018, 7, 1701061. [Google Scholar] [CrossRef] [PubMed]
- Nazirkar, G.; Singh, S.; Dole, V.; Nikam, A. Effortless Effort in Bone Regeneration: A Review. J. Int. Oral Health 2014, 6, 120–124. [Google Scholar] [PubMed]
- Matassi, F.; Nistri, L.; Chicon Paez, D.; Innocenti, M. New Biomaterials for Bone Regeneration. Clin. Cases Min. Bone Metab. 2011, 8, 21–24. [Google Scholar]
- Wen, Y.; Xun, S.; Haoye, M.; Baichuan, S.; Peng, C.; Xuejian, L.; Kaihong, Z.; Xuan, Y.; Jiang, P.; Shibi, L. 3D Printed Porous Ceramic Scaffolds for Bone Tissue Engineering: A Review. Biomater. Sci. 2017, 5, 1690–1698. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, L.; Fan, Y.; Feng, Q.; Cui, F.-Z.; Watari, F. Nanostructured Scaffolds for Bone Tissue Engineering. J. Biomed. Mater. Res. Part A 2013, 101A, 2424–2435. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Webster, T.J. Mesoporous Silica Based Nanostructures for Bone Tissue Regeneration. Front. Mater. 2021, 8, 692309. [Google Scholar] [CrossRef]
- Amini, A.R.; Laurencin, C.T.; Nukavarapu, S.P. Bone Tissue Engineering: Recent Advances and Challenges. Crit. Rev. Biomed. Eng. 2012, 40, 363–408. [Google Scholar] [CrossRef]
- Sill, T.J.; von Recum, H.A. Electrospinning: Applications in Drug Delivery and Tissue Engineering. Biomaterials 2008, 29, 1989–2006. [Google Scholar] [CrossRef]
- Zhang, Y.; Su, B.; Venugopal, J.; Ramakrishna, S.; Lim, C. Biomimetic and Bioactive Nanofibrous Scaffolds from Electrospun Composite Nanofibers. Int. J. Nanomed. 2007, 2, 623–638. [Google Scholar]
- Zhang, Q.; Li, Y.; Lin, Z.Y.W.; Wong, K.K.Y.; Lin, M.; Yildirimer, L.; Zhao, X. Electrospun Polymeric Micro/Nanofibrous Scaffolds for Long-Term Drug Release and Their Biomedical Applications. Drug Discov. Today 2017, 22, 1351–1366. [Google Scholar] [CrossRef] [PubMed]
- Bayer, I.S. A Review of Sustained Drug Release Studies from Nanofiber Hydrogels. Biomedicines 2021, 9, 1612. [Google Scholar] [CrossRef] [PubMed]
- Park, S.J.; Lee, Y.J.; Heo, D.N.; Kwon, I.K.; Yun, K.-S.; Kang, J.Y.; Lee, S.H. Functional Nerve Cuff Electrode with Controllable Anti-Inflammatory Drug Loading and Release by Biodegradable Nanofibers and Hydrogel Deposition. Sens. Actuators B Chem. 2015, 215, 133–141. [Google Scholar] [CrossRef]
- Pawłowska, S.; Rinoldi, C.; Nakielski, P.; Ziai, Y.; Urbanek, O.; Li, X.; Kowalewski, T.A.; Ding, B.; Pierini, F. Ultraviolet Light-Assisted Electrospinning of Core–Shell Fully Cross-Linked P(NIPAAm-Co-NIPMAAm) Hydrogel-Based Nanofibers for Thermally Induced Drug Delivery Self-Regulation. Adv. Mater. Interfaces 2020, 7, 2000247. [Google Scholar] [CrossRef]
- Kopeček, J.; Yang, J. Hydrogels as Smart Biomaterials. Polym. Int. 2007, 56, 1078–1098. [Google Scholar] [CrossRef]
- Mathur, A.M.; Moorjani, S.K.; Scranton, A.B. Methods for Synthesis of Hydrogel Networks: A Review. J. Macromol. Sci. Part C 1996, 36, 405–430. [Google Scholar] [CrossRef]
- Ahmed, E.M. Hydrogel: Preparation, Characterization, and Applications: A Review. J. Adv. Res. 2015, 6, 105–121. [Google Scholar] [CrossRef]
- Liu, M.; Zeng, X.; Ma, C.; Yi, H.; Ali, Z.; Mou, X.; Li, S.; Deng, Y.; He, N. Injectable Hydrogels for Cartilage and Bone Tissue Engineering. Bone Res. 2017, 5, 17014. [Google Scholar] [CrossRef]
- Drury, J.L.; Mooney, D.J. Hydrogels for Tissue Engineering: Scaffold Design Variables and Applications. Biomaterials 2003, 24, 4337–4351. [Google Scholar] [CrossRef]
- Zhu, J.; Marchant, R.E. Design Properties of Hydrogel Tissue-Engineering Scaffolds. Expert Rev. Med. Devices 2011, 8, 607–626. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.; Marra, K.G. Injectable, Biodegradable Hydrogels for Tissue Engineering Applications. Materials 2010, 3, 1746–1767. [Google Scholar] [CrossRef]
- Jeong, J.-O.; Park, J.-S.; Kim, Y.-A.; Yang, S.-J.; Jeong, S.-I.; Lee, J.-Y.; Lim, Y.-M. Gamma Ray-Induced Polymerization and Cross-Linking for Optimization of PPy/PVP Hydrogel as Biomaterial. Polymers 2020, 12, 111. [Google Scholar] [CrossRef]
- Kim, Y.-A.; Jeong, J.-O.; Park, J.-S. Preparation and Characterization of Ionic Conductive Poly(Acrylic Acid)-Based Silicone Hydrogels for Smart Drug Delivery System. Polymers 2021, 13, 406. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Yin, Z.; Wu, S.; Qi, X.; He, Q.; Zhang, Q.; Yan, Q.; Boey, F.; Zhang, H. Graphene-Based Materials: Synthesis, Characterization, Properties, and Applications. Small 2011, 7, 1876–1902. [Google Scholar] [CrossRef] [PubMed]
- Georgakilas, V.; Tiwari, J.N.; Kemp, K.C.; Perman, J.A.; Bourlinos, A.B.; Kim, K.S.; Zboril, R. Noncovalent Functionalization of Graphene and Graphene Oxide for Energy Materials, Biosensing, Catalytic, and Biomedical Applications. Chem. Rev. 2016, 116, 5464–5519. [Google Scholar] [CrossRef] [PubMed]
- Khatir, N.M.; Fatoorehchi, H.; Ahmadi, A.; Khoshnoodfar, A.; Faghihnasiri, M. Investigating the Adsorption of the Thyroid Stimulating Hormones Molecules on Graphene Sheets by the Density Functional Theory for Possible Nano-Biosensor Applications. J. Chem. Pet. Eng. 2021, 55, 385–392. [Google Scholar]
- Khatir, N.M.; Ahmadi, A.; Taghizade, N.; Motevali Khameneh, S.; Faghihnasiri, M. Electronic Transport Properties of Nanoribbons of Graphene and ψ-Graphene -Based Lactate Nanobiosensor. Superlattices Microstruct. 2020, 145, 106603. [Google Scholar] [CrossRef]
- Dinescu, S.; Ionita, M.; Ignat, S.-R.; Costache, M.; Hermenean, A. Graphene Oxide Enhances Chitosan-Based 3D Scaffold Properties for Bone Tissue Engineering. Int. J. Mol. Sci. 2019, 20, 5077. [Google Scholar] [CrossRef]
- Shadjou, N.; Hasanzadeh, M. Graphene and Its Nanostructure Derivatives for Use in Bone Tissue Engineering: Recent Advances. J. Biomed. Mater. Res. Part A 2016, 104, 1250–1275. [Google Scholar] [CrossRef]
- Nie, W.; Peng, C.; Zhou, X.; Chen, L.; Wang, W.; Zhang, Y.; Ma, P.X.; He, C. Three-Dimensional Porous Scaffold by Self-Assembly of Reduced Graphene Oxide and Nano-Hydroxyapatite Composites for Bone Tissue Engineering. Carbon 2017, 116, 325–337. [Google Scholar] [CrossRef]
- Holt, B.D.; Wright, Z.M.; Arnold, A.M.; Sydlik, S.A. Graphene Oxide as a Scaffold for Bone Regeneration. WIREs Nanomed. Nanobiotechnol. 2017, 9, e1437. [Google Scholar] [CrossRef]
- Kim, S.; Jeong, J.-O.; Lee, S.; Park, J.-S.; Gwon, H.-J.; Jeong, S.I.; Hardy, J.G.; Lim, Y.-M.; Lee, J.Y. Effective Gamma-Ray Sterilization and Characterization of Conductive Polypyrrole Biomaterials. Sci. Rep. 2018, 8, 3721. [Google Scholar] [CrossRef]
- Jeong, J.-O.; Jeong, S.I.; Park, J.-S.; Gwon, H.-J.; Ahn, S.-J.; Shin, H.; Lee, J.Y.; Lim, Y.-M. Development and Characterization of Heparin-Immobilized Polycaprolactone Nanofibrous Scaffolds for Tissue Engineering Using Gamma-Irradiation. RSC Adv. 2017, 7, 8963–8972. [Google Scholar] [CrossRef]
- Yang, S.-J.; Jeong, J.-O.; Lim, Y.-M.; Park, J.-S. Synthesis and Characterization of PVP Microneedle Patch Using Metal Bioelectrodes for Novel Drug Delivery System. Mater. Des. 2021, 201, 109485. [Google Scholar] [CrossRef]
- Prall, W.C.; Haasters, F.; Heggebö, J.; Polzer, H.; Schwarz, C.; Gassner, C.; Grote, S.; Anz, D.; Jäger, M.; Mutschler, W.; et al. Mesenchymal Stem Cells from Osteoporotic Patients Feature Impaired Signal Transduction but Sustained Osteoinduction in Response to BMP-2 Stimulation. Biochem. Biophys. Res. Commun. 2013, 440, 617–622. [Google Scholar] [CrossRef] [PubMed]
- Rickard, D.J.; Sullivan, T.A.; Shenker, B.J.; Leboy, P.S.; Kazhdan, I. Induction of Rapid Osteoblast Differentiation in Rat Bone Marrow Stromal Cell Cultures by Dexamethasone and BMP-2. Dev. Biol. 1994, 161, 218–228. [Google Scholar] [CrossRef]
- James, A.W.; LaChaud, G.; Shen, J.; Asatrian, G.; Nguyen, V.; Zhang, X.; Ting, K.; Soo, C. A Review of the Clinical Side Effects of Bone Morphogenetic Protein-2. Tissue Eng. Part B Rev. 2016, 22, 284–297. [Google Scholar] [CrossRef]
- Fu, C.; Bai, H.; Zhu, J.; Niu, Z.; Wang, Y.; Li, J.; Yang, X.; Bai, Y. Enhanced Cell Proliferation and Osteogenic Differentiation in Electrospun PLGA/Hydroxyapatite Nanofibre Scaffolds Incorporated with Graphene Oxide. PLoS ONE 2017, 12, e0188352. [Google Scholar] [CrossRef]
- Halim, A.; Luo, Q.; Ju, Y.; Song, G. A Mini Review Focused on the Recent Applications of Graphene Oxide in Stem Cell Growth and Differentiation. Nanomaterials 2018, 8, 736. [Google Scholar] [CrossRef]
- Rawat, S.; Jain, K.G.; Gupta, D.; Raghav, P.K.; Chaudhuri, R.; Pinky; Shakeel, A.; Arora, V.; Sharma, H.; Debnath, D.; et al. Graphene Nanofiber Composites for Enhanced Neuronal Differentiation of Human Mesenchymal Stem Cells. Nanomedicine 2021, 16, 1963–1982. [Google Scholar] [CrossRef] [PubMed]





| Ion | Na+ | K+ | Mg2+ | Ca2+ | Cl− | HCO3− | HPO42− | SO42− | pH | |
|---|---|---|---|---|---|---|---|---|---|---|
| Ion-content (mM) | Blood plasma | 142.0 | 5.0 | 1.5 | 2.5 | 103.0 | 27.0 | 1.0 | 0.5 | 7.2~7.4 |
| SBF | 142.0 | 5.0 | 1.5 | 2.5 | 147..8 | 4.2 | 1.0 | 0.5 | 7.4 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jeong, J.-O.; Jeong, S.-I.; Lim, Y.-M.; Park, J.-S. Effective BMP-2 Release and Mineralization on a Graphene Oxide/Polyvinylpyrrolidone Hydrogel Forming Poly (ε-Caprolactone) Nanofibrous Scaffolds. Materials 2022, 15, 8642. https://doi.org/10.3390/ma15238642
Jeong J-O, Jeong S-I, Lim Y-M, Park J-S. Effective BMP-2 Release and Mineralization on a Graphene Oxide/Polyvinylpyrrolidone Hydrogel Forming Poly (ε-Caprolactone) Nanofibrous Scaffolds. Materials. 2022; 15(23):8642. https://doi.org/10.3390/ma15238642
Chicago/Turabian StyleJeong, Jin-Oh, Sung-In Jeong, Youn-Mook Lim, and Jong-Seok Park. 2022. "Effective BMP-2 Release and Mineralization on a Graphene Oxide/Polyvinylpyrrolidone Hydrogel Forming Poly (ε-Caprolactone) Nanofibrous Scaffolds" Materials 15, no. 23: 8642. https://doi.org/10.3390/ma15238642
APA StyleJeong, J.-O., Jeong, S.-I., Lim, Y.-M., & Park, J.-S. (2022). Effective BMP-2 Release and Mineralization on a Graphene Oxide/Polyvinylpyrrolidone Hydrogel Forming Poly (ε-Caprolactone) Nanofibrous Scaffolds. Materials, 15(23), 8642. https://doi.org/10.3390/ma15238642







