1. Introduction
Orthopedics, cranial and maxillofacial reconstructions represent a major sector of surgical procedures at present. Furthermore, the aging of the population in recent decades has generated a significant increase in patients needing skeletal repair [
1,
2]. This led to an increase in age-related pathologies and accentuated skeletal fragility, as well as problems of dependence when there is a physical functional limitation (e.g., due to skeletal diseases or traumas) with an inability to perform daily life activities. Engineered biomaterials are developed as bone substitutes, which are potentially multifunctional, to improve the quality of patients’ life and to allow for them to recover normal physical activity as quickly as possible.
For bone repair, although autologous bone graft has long been considered the gold standard, it has limitations in terms of amount of available graft tissue and it increases the morbidity and risks of surgical site infection and pain. Instead, the development of synthetic bone substitutes is now clearly favored, in combination with several advantages such as unlimited resources, the absence of transferred pathogens and the possibility to tailor their shape, size and composition to address specific clinical needs. Among bone biomaterials, calcium phosphates (CaP) and bioactive glasses are by far the two main groups of compounds used at industrial and clinical scales [
3,
4,
5]. CaPs are directly inspired by the composition of natural bone, which involves ca. 70 wt.% of a CaP apatitic phase [
6], and, as such, they exhibit intrinsic biocompatibility, making them very adequate for bone regeneration purposes. Among CaPs, the development of high-temperature and sintering processing approaches [
7] has favored the development of well-crystallized high-temperature phases, such as stoichiometric hydroxyapatite (HA, Ca
10(PO
4)
6(OH)
2) and β-tricalcium phosphate (β-TCP, Ca
3(PO
4)
2) [
8,
9,
10]. One limitation of HA is its very low solubility (pK
sp~117 for the Ca
10 stoichiometry) [
11,
12], leading to poor ion release kinetics (and related bioactivity) and a non-resorbable character. Combinations of HA with β-TCP could seen as a way to artificially increase the resorption rate of BCPs. Indeed, although β-TCP has a solubility product (pK
sp~29) [
13] greater than that of body fluids such as blood (pK
sp~25–26) [
14], this CaP phase is rather readily resorbable upon osteoclast cell activity [
15]. BCPs are commonly produced commercially and used clinically in orthopedics and dentistry—e.g., in the form of porous or dense scaffolds—for filling bone defects (e.g., after bone tumors or traumas), and to provide support for bone healing in prosthesis revision and joint arthrodesis [
16,
17]. Such BCPs have several appealing assets such as ease of fabrication (despite the need to generate high temperatures) in different shapes and with tailorable porosity, as well as relevant mechanical properties, especially for bone sites under moderate pressure solicitations. However, these two high-temperature phases, HA and β-TCP, although osteoconductive, present limited bioactivity related to their low specific surface areas and poor ability to release ions. Contrary to natural bone mineral (or its biomimetic synthetic analogs) [
6,
18,
19], these phases do not allow for fast ion exchanges with body fluids or extensive molecular adsorption possibilities to potentially accelerate bone regeneration.
Strategies are increasingly being explored to increase biomaterials’ bioactivity, which may occur via bioactive calcium phosphate phases. Such strategies gather “bio-activation” approaches, aiming to favor natural healing pathways and increase biomedical potentialities by conveying additional properties (e.g., antimicrobial) [
20,
21]. Concerning BCPs, the “bio-activation” strategies have been based on the deposition of more bioactive CaP phases, leading to a bio-inspired coating capable of improving the biological behavior of the scaffold [
22]. Wet processes allowed, for example, to deposit biomimetic apatites or their reactive hydrolysable precursors such as brushite (dicalcium phosphate dihydrate, DCPD, CaHPO
4.2H
2O) or octacalcium phosphate (OCP, Ca
8(PO
4)
4(HPO
4)
2.5H
2O). A significant amount of research has been conducted in this direction, inspired by work such as that from Kokubo’s group [
23]. The substrate was covered by a highly reactive apatite layer formed from a metastable solution. Agents such as CO
2 can then be used to modify the pH of the solution and modify the composition of the precipitated phases [
24]. Such a coating process has the advantage of allowing for the treatment of surfaces of complex shapes but suffers from difficulties related to their use at an industrial scale, with uncontrolled phases’ precipitation being the main issue. Other works have proposed to coat porous BCP ceramics with a reactive “nanoporous” biomimetic apatite coating using an impregnation-drying process from a calcium phosphate nanogel [
25,
26]. The presence of a biomimetic apatite coating on the BCP porosity allowed for significant increase in the molecular adsorption capacity of the ceramic (illustrated with the BMP-2 growth factor) while also providing a net increase in new bone formation and unveiling osteoinductive properties—which was not observed in stoichiometric HA nor β-TCP. Biomimetic apatites showed great bone regeneration capabilities in vivo [
26,
27,
28], ich was wh not reached with high-temperature CaP phases. This coating approach has some limitations, especially regarding the adhesion of the coating on the substrate.
Consequently, at this stage, several of the studies cited above have reported that exposing biomimetic apatite (or its metastable/hydrolysable precursors such as OCP) on BCP surface led to a clear increase in in vivo bone neoformation and enhanced significantly the osteointegration of the implants by conferring an osteoinductive character. The technological challenge that remains lies in the strategy to successfully expose such bioactive CaPs compounds at the surface of BCPs while preserving the overall integrity of the scaffolds, not blocking their porous network, without a lack of adherence to the substrate, and with an easily industrializable process.
Furthermore, bacterial infections are a major concern in orthopedic and dental surgery [
29,
30]. While the use of antibiotics might generate bacterial resistance mechanisms [
31], some ionic species, such as copper ions (Cu
2+), are well known for their antibacterial intrinsic properties [
32]. Copper ions thus proved to be very relevant in the biomedical field and bone engineering thanks to their antibacterial and pro-angiogenic properties, as well as for their ability to potentially stimulate osteoblast cell activity [
33,
34].
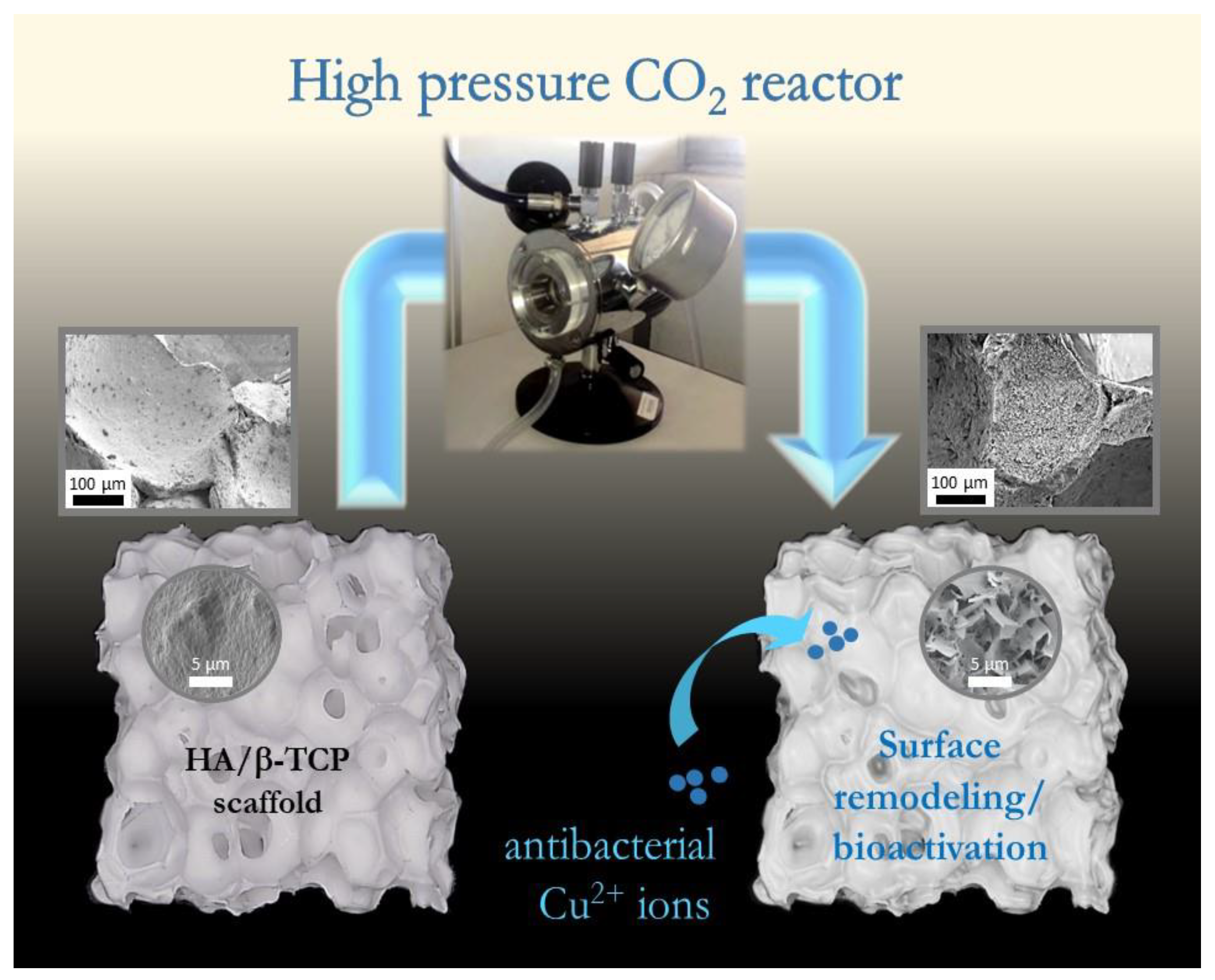
In the present work, we have conceptualized a novel strategy to address these questions, based on an original coating-from instead of a coating-to approach. In contrast to depositing an external CaP coating, this concept lies in the direct chemical modification of the surface of BCPs upon treatment with high-pressure CO
2 (using a supercritical CO
2 reactor). CO
2 is particularly relevant for its three functions of (1) pH modifier, (2) carbonate ions’ provider and (3) sterilizing agent. Moreover, various industrial processes already use high-pressure CO
2, for example, as a “green” and biocompatible solvent [
35], including for biomedical applications [
35,
36,
37], evidencing clear scale-up possibilities.
The innovative approach of this study resides in the use of high-pressure (supercritical) CO
2 to allow for water-immersed BCP surface remodeling. In simple terms, this may be seen as a “
coating-from” rather than a “
coating-to” approach, as no addition of external solid substance is needed, conversely to traditional coating strategies [
22,
23,
25]. Such a strategy could thus be applied to existing, commercial BCPs, in order to significantly enhance their bioactivity and renew their clinical relevance.
We will here describe this proof-of-concept and validate it on actual commercial BCPs. We will explore the properties of the newly formed phases using complementary techniques such as X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FTIR), scanning electron microscopy (FEG-SEM), solid-state Nuclear Magnetic Resonance (NMR), specific surface area measurements (BET) and mercury intrusion porosimetry, as well as indirect tests as ion exchange experiments. We will then evidence the possibility of doping such remodeled BCPs surfaces with bioactive Cu2+ ions and demonstrate their antibacterial features toward two major bacterial strains, as well as the absence of acute cytotoxicity to osteoblast cells.
3. Results and Discussion
The innovative approach of this study resides in the use of high-pressure (supercritical) CO
2 to allow for water-immersed BCP surface remodeling via a dissolution/reprecipitation mechanism (
Figure 1). In simple terms, this may be seen as a “
coating-from” rather than a “
coating-to” approach, by the fact that no addition of external solid substance is needed, conversely to traditional coating strategies [
23,
25,
26,
39].
The general concept of the study is based on the idea that both constituents of BCPs, namely, HA and β-TCP, become soluble in acidic conditions, making the solution pH a powerful tool for controlling CaP phases dissolution and precipitation. In addition to its already approved use at the industrial scale, including in the biomedical field [
37], the choice of CO
2 is correlated to its formation in solution of HCO
3− and CO
32− ions, thus generating an acidification effect through the following scheme:
The degree of acidification is linked to the solubility of CO
2 in water, itself related to the working temperature and pressure of the cell [
40]. In the conditions used in this work (37 °C, 80 bar), a theoretical minimal pH of 3.2 may be expected, which is in the right pH domain to allow for both HA and β-TCP dissolution [
41]. In a preliminary work [
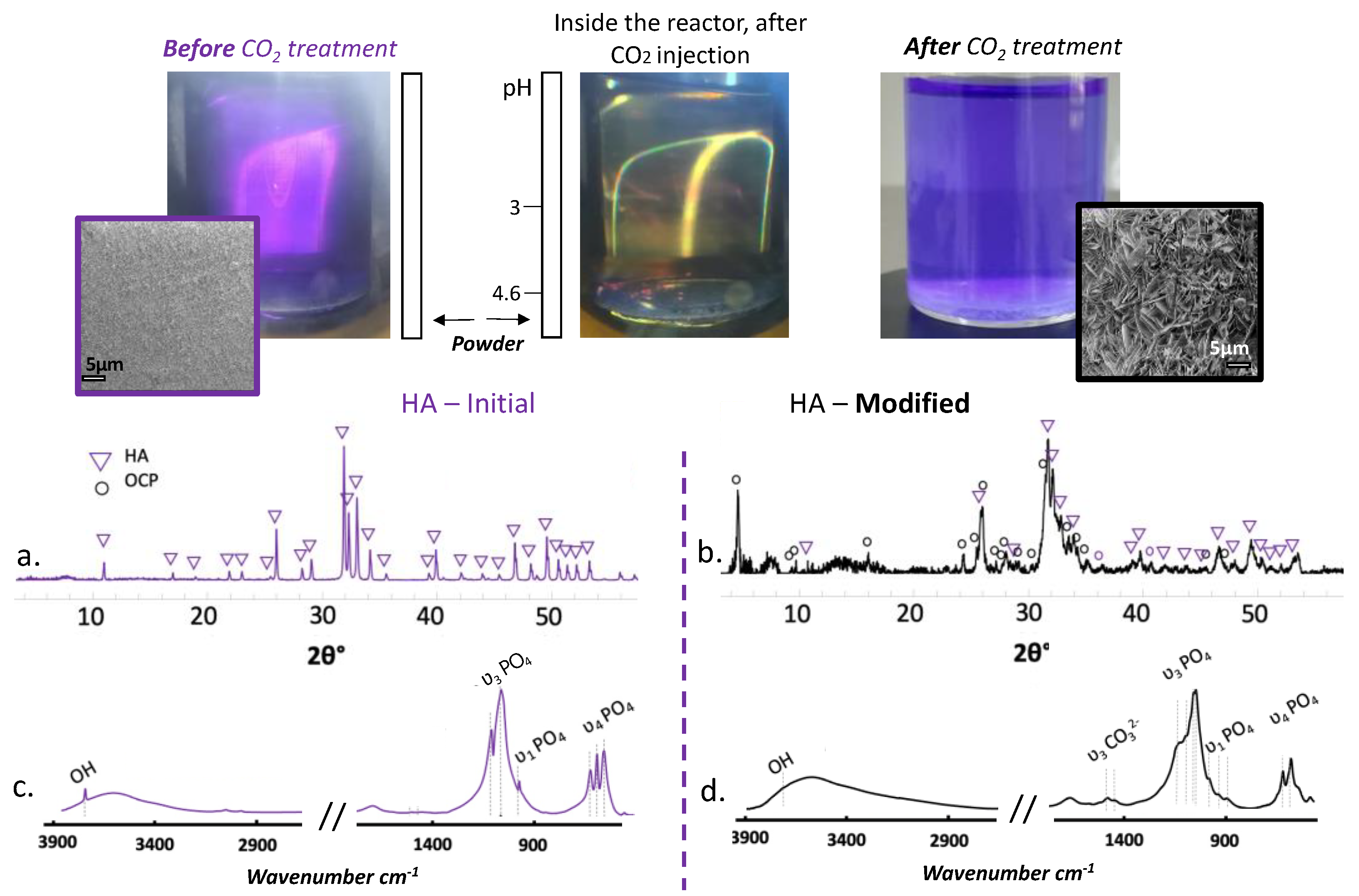
42], we verified that a “simple” HA powder, i.e., the most insoluble of the two phases and easily characterized by FTIR and XRD, underwent a progressive dissolution in these conditions.
Figure 2 shows how the surface morphology of HA particles (powder) is modified upon such CO
2 treatment and also shows, thanks to the addition of bromophenol blue in the medium, the clear acidification of the solution under treatment. The formation of plate-like features is a clear indication of the chemical remodeling of the surface of the HA particles. XRD analyses (
Figure 2a,b) showed the presence of OCP and FTIR spectral data (
Figure 2c,d) indicated the presence of carbonates, pointing to the co-presence of a carbonated apatite phase (since OCP is not known to be able to accommodate easily carbonate species). These results could be an indicator of the mechanism at stake while modifying the BCP.
We may remark that the low temperature of 37 °C is not only relevant to reaching the supercritical state of CO
2 into the chamber’s atmosphere, but also in view of biomedical applications (physiological temperature) and allows for the preservation of metastable CaPs-targeted phases such as biomimetic apatite or lower-Ca/P precursors like OCP [
38,
43]. Upon depressurization of the reactor, the progressive degassing of dissolved CO
2 is expected to induce a gradual increase in the pH until it reverted to neutrality. Therefore, the whole process can be seen as being composed of two essential steps: (i) CO
2 pressurization of the cell, allowing for a pH drop and CaP partial dissolution and (ii) depressurization of the chamber, allowing for a pH rise and reprecipitation of metastable CaP phases (indeed, at temperatures as low as 37 °C, neither stoichiometric HA nor β-TCP can reform). It may be noted that, in case of the dissolution of HA/β-TCP phases, the release of PO
43− and OH
− ions in the medium also plays a role in the rise in pH due to their propensity to combine with protons.
In comparison to CaP powders, 3D BCP scaffolds exhibit a lower specific surface area and a cohesive character with an interconnected porous network. The possibility of modifying the surface of BCP porous scaffolds thanks to this high-pressure CO2 process thus needs specific attention compared to treating powders, and has been evaluated here using different methods.
- (i)
Characterization of BCP modified by supercritical CO2
As shown above, SEM is a particularly suited to detecting the presence or absence of surface chemical remodeling.
Figure 3 shows typical surface features of both initial and CO
2-treated BCP scaffolds (37 °C, 80 bar, 4 h). A clear remodeling of the surface of the initially smooth scaffold pores walls was achieved upon treatment. This is evidenced by the formation of plate-like particles covering the surface, with typical dimensions of around 1–5 µm on the external part of the 3 mm diameter BCP cubes and with smaller particle sizes of around 0.5 µm in the deeper porosity (red arrow on
Figure 3). Despite this change in particle dimensions across the scaffold depth, the whole open porous structure appeared to be successfully remodeled. It may be noted that the (sub)micron scale of this modification throughout the outside scaffold surface (
Figure 3) is well below the mean size of live cells (>10 µm), thus providing them with a renewed “covered” surface state for cell activity after implantation.
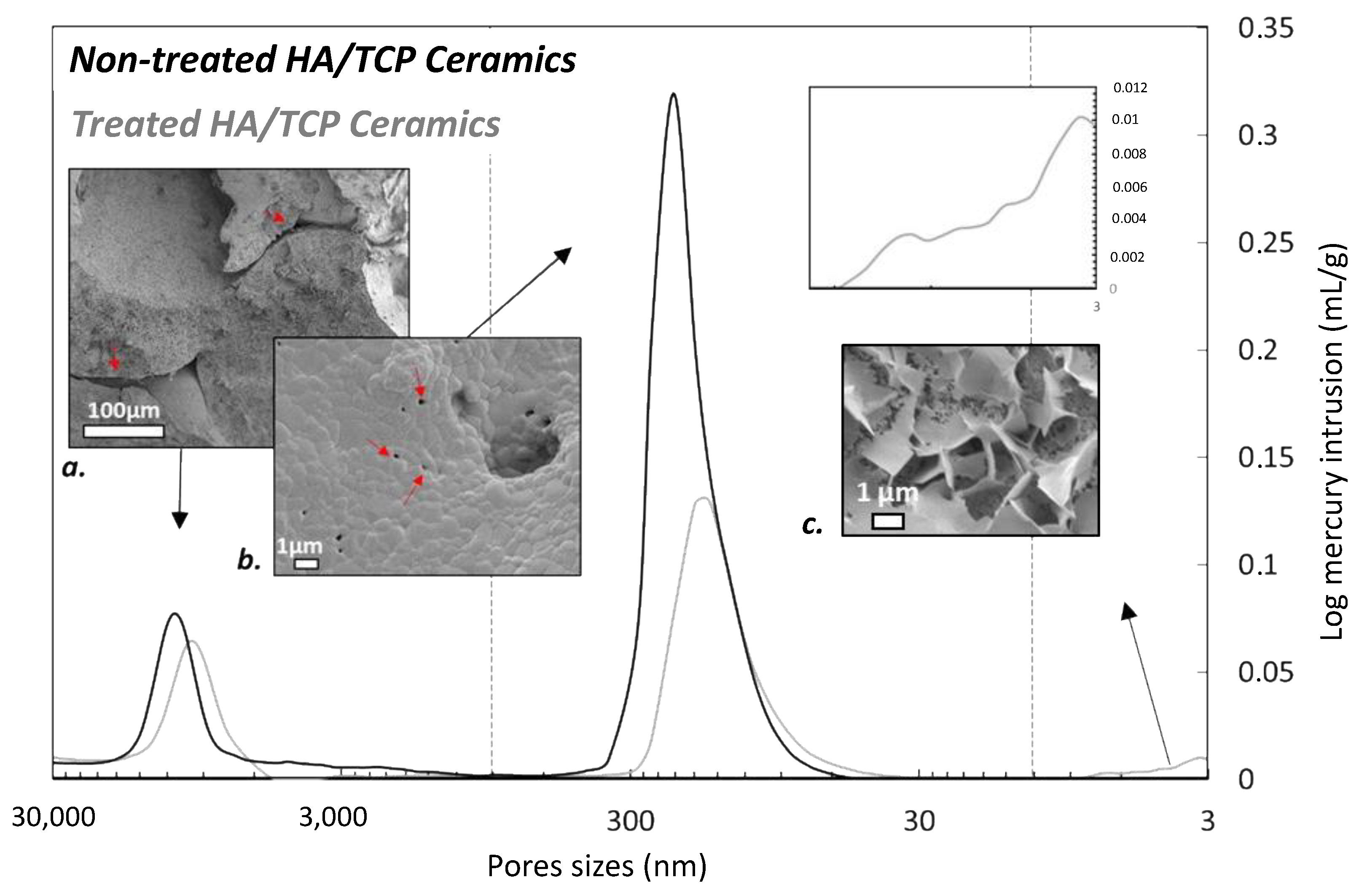
BET measurements showed a reproducible light increase in the specific surface area of the porous BCP cubes after high-pressure CO
2 treatment, from 0.66 ± 0.03 to 0.76 ± 0.04 m
2/g, thus allowing for us to expect a more extended contact surface with the surrounding medium. Mercury intrusion porosimetry analyses were carried out to analyze the modifications to the porous network in more detail (
Figure 4). Two distinct pore populations are visible on initial BCP scaffolds. The first one, centered at around 10.5 µm, could match the gaps between pores, as shown in
Figure 4a. The second (most prominent) pore population is observed close to 200 nm, itself composed of several subpopulations, as indicated from the analysis of the derivative function, which shows several inflexion points in this area (
Supplementary Materials, Figure S1). This could be due to the sintering defects (red arrow,
Figure 4a,b). In contrast, several modifications are seen for the treated BCP. The two main populations that were previously observed on untreated BCP are downshifted toward slightly smaller pore sizes, of around 10.5 µm and 150 nm, respectively. This slight decrease in main pore diameters is indicative of the surface remodeling observed by SEM and unveiling of newly precipitated phases/crystals covering the surface, thus generating a decreasing tendency for the original pore diameters. In addition, a new mesoporosity was observed on the treated scaffold, with nanometer-sized pores (~3–9 nm). This new nanometer-scale porosity could be due to the network formed by the newly precipitated entangled platelets detected by SEM (
Figure 4c). The occurrence of nanosized pores has already been reported in biomimetic apatites and attributed to the 3D organization of apatite nanocrystals [
25].
These cumulated data, arising from SEM, BET and Hg-porosimetry, demonstrate that BCPs subjected to this high-pressure CO
2 process undergo a surface remodeling with newly formed crystals, whose morphology, size and 3D organization are close to those reported for biomimetic nanocrystalline apatites. To shed more light on the nature of the formed precipitates, XRD and FTIR analyses were carried out (
Figure S2a,b). However, the data obtained via these bulk characterization techniques did not allow for the detection of substantial new phases. Nonetheless, a decrease of the HA/β-TCP weight ratio from 70/30 (experimental starting values) to 85/15 was evaluated (
Figure S2c, estimated error of 5% on mass% assignments), pointing to a significant loss of β-TCP, the most soluble of the two phases in the conditions of the process. Contrary to the powders, BCP scaffolds exhibit a low surface area and an overall mechanical cohesion that limits the extent of the chemical alterations generated by the CO
2 treatment to only the surface of the scaffold’s open porosity. This fits well with our goals aiming at remodeling only the surface of BCPs while preserving their general integrity and porous character, in view of subsequent uses in previously identified bone applications. Nonetheless, in these conditions, neither XRD nor FTIR offers enough resolution to access the top surface modifications generated here. Therefore, more advanced analyses were performed with less conventional approaches, such as solid-state NMR. Indeed, NMR proved to be a particularly suitable method for characterizing bone mineral and CaP phases [
43,
44,
45,
46,
47,
48,
49,
50,
51], and presents the advantage of allowing for biomimetic apatites to be distinguished from OCP that otherwise exhibits rather close features using XRD and FTIR [
43].
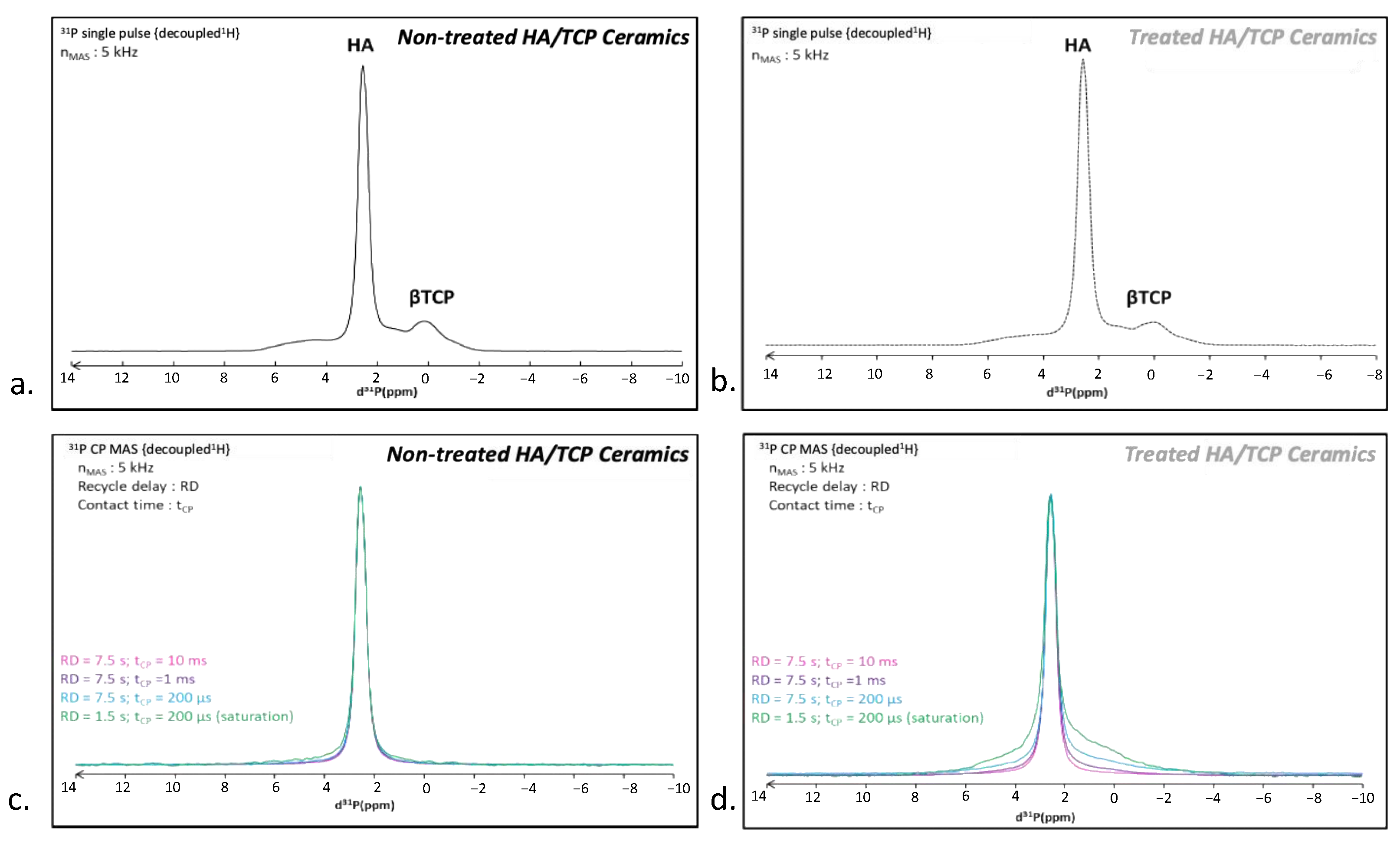
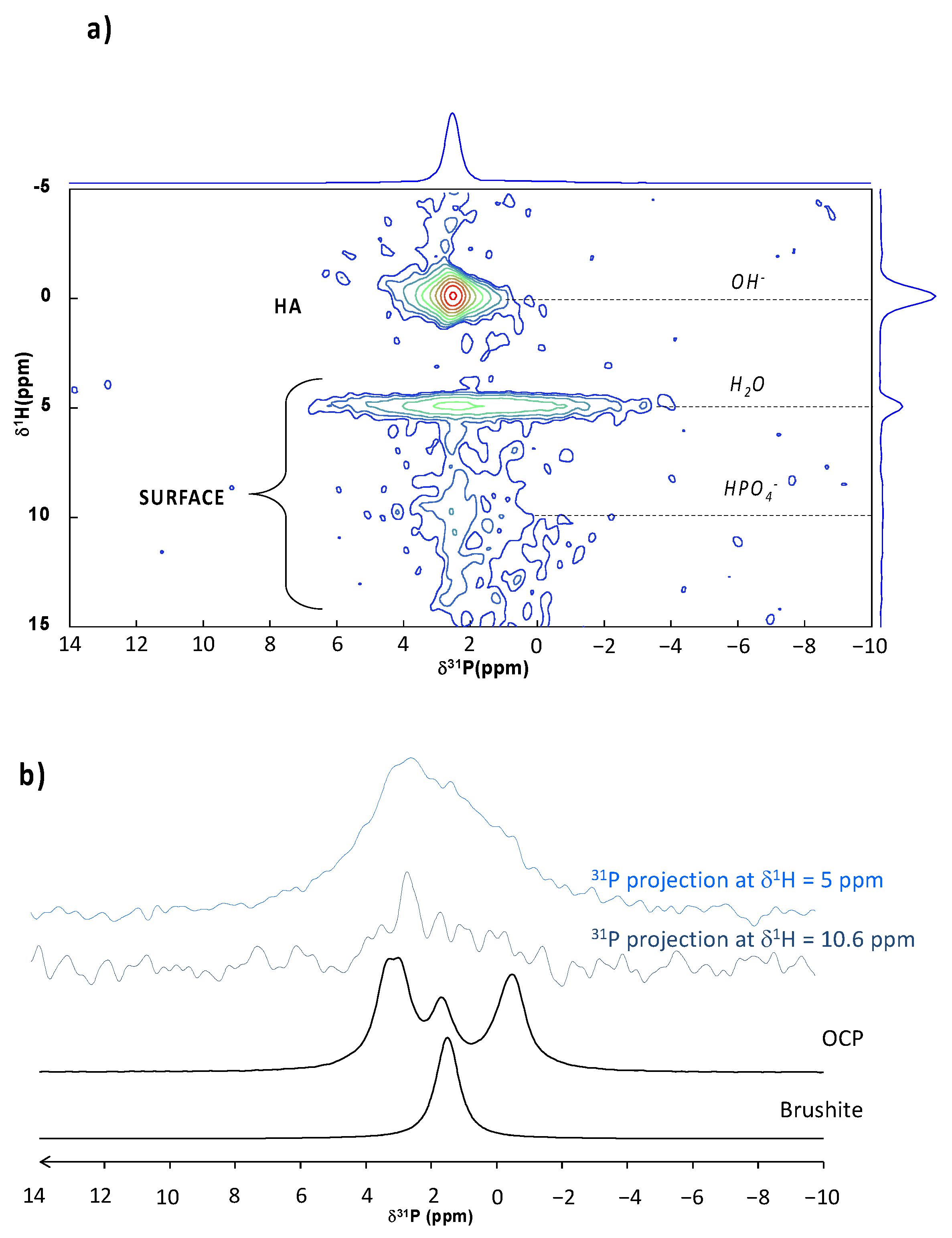
A
31P single pulse MAS NMR spectrum was recorded on the untreated BCP scaffold (
Figure 5a). Two main signals were detected, at 2.6 and a broad one spanning from 6 to 0 ppm. The most intense 2.6 ppm peak can be assigned to the HA component [
45] while the 6–0 ppm broad signal corresponds to the β-TCP phase [
46,
47], in agreement with the initial composition of these biphasic scaffolds. These two components were also detected on the CO
2-treated BCP without major visible alteration (
Figure 5b). In order to enhance the response of protonated phases, additional measurements were made in
1H →
31P cross polarization mode. Note that, in this configuration, the β-TCP phase (exempt of protons) cannot be visualized. Several measurements were made by decreasing the contact time (from 10 ms down to 200 µs), allowing for us to probe the nearest environment of phosphorus nuclei. As expected, the
1H-
31P CP MAS NMR spectrum of the untreated scaffold displayed the single 2.6 ppm peak characteristic of HA [
45], whatever the contact time (
Figure 5c). In contrast, the CO
2-treated BCP behaved differently (
Figure 5d) as the
1H-
31P CP MAS NMR spectrum revealed a wide halo at the peak basis, which increases in intensity as the contact times decreases. A shortened recycling delay (RD) (from 7.5 s down to 1.5 s) made this large signal even more obvious. Through comparison with the untreated sample, we assigned this broad resonance to surface phosphate species induced by the CO
2 treatment. The enlargement is indicative of a distribution of chemical shifts and, thus, the presence of disordered phase(s) at the surface involving strong
1H-
31P interactions at short range, thus pointing to the presence of phase(s) rich in H
2O and/or HPO
42− ions. This is characteristic of crystallographic disorder and/or of a high degree of hydration compared to HA [
42,
48].
To try to further identify the disordered CaP phase(s) composing the BCP remodeled surface, a 2D
1H-
31P HETCOR experiments was performed (
Figure 6a). This analysis aims to provide information on the proximity and interaction between
31P and
1H nuclei. Two distinct correlation zones can be identified. A first correlation (denoted as HA in
Figure 6a) shows the interaction between a rather narrow correlation between
31P and protons at 0 ppm, which is characteristic of PO
43−/OH
− interactions within the HA phase [
43,
44,
45]. In addition, other correlations can also be seen (denoted SURFACE in
Figure 6a) between the broad
31P signal from surface species and
1H arising from H
2O molecules (δ
1H = 5 ppm) but also, to a lesser extent, from HPO
42− ions (centered at δ
1H = 10.6 ppm), confirming the presence of at least one hydrated phase including some hydrogen phosphate ions after CO
2 treatment. The
31P contributions (projections from the 2D HETCOR spectrum) from phosphate surface species have been plotted in
Figure 6b to better visualize the underlying contributions, compared with the reference spectra of OCP and DCPD (internal collection). Thus, despite the presence of H
2O and HPO
42−, the presence of DCPD—which exhibits a highly ordered structure—at the surface of the treated samples can be ruled out (or is present in small amount). On the other hand, we note that the
31P surface signal spanned same range as the OCP reference spectrum and correlated with H
2O and HPO
42− species similar to OCP. Thus, this analysis might suggest the presence of a poorly crystallized OCP-like phase (e.g., in the process of crystallization) at the surface of the treated scaffold. These findings are in coherence with previously reported data of HA and ACP powders treated with the same process [
42]. However, if the co-presence of biomimetic apatite was clearly identified in this previous work using powders, its presence on the treated BCP scaffold cannot be asserted here on the sole basis of solid-state NMR. Indeed, since the BCP bulk is mainly composed of HA, an eventual trace of biomimetic apatite at the surface could not be detected in these conditions due to the superposition of the corresponding signals.
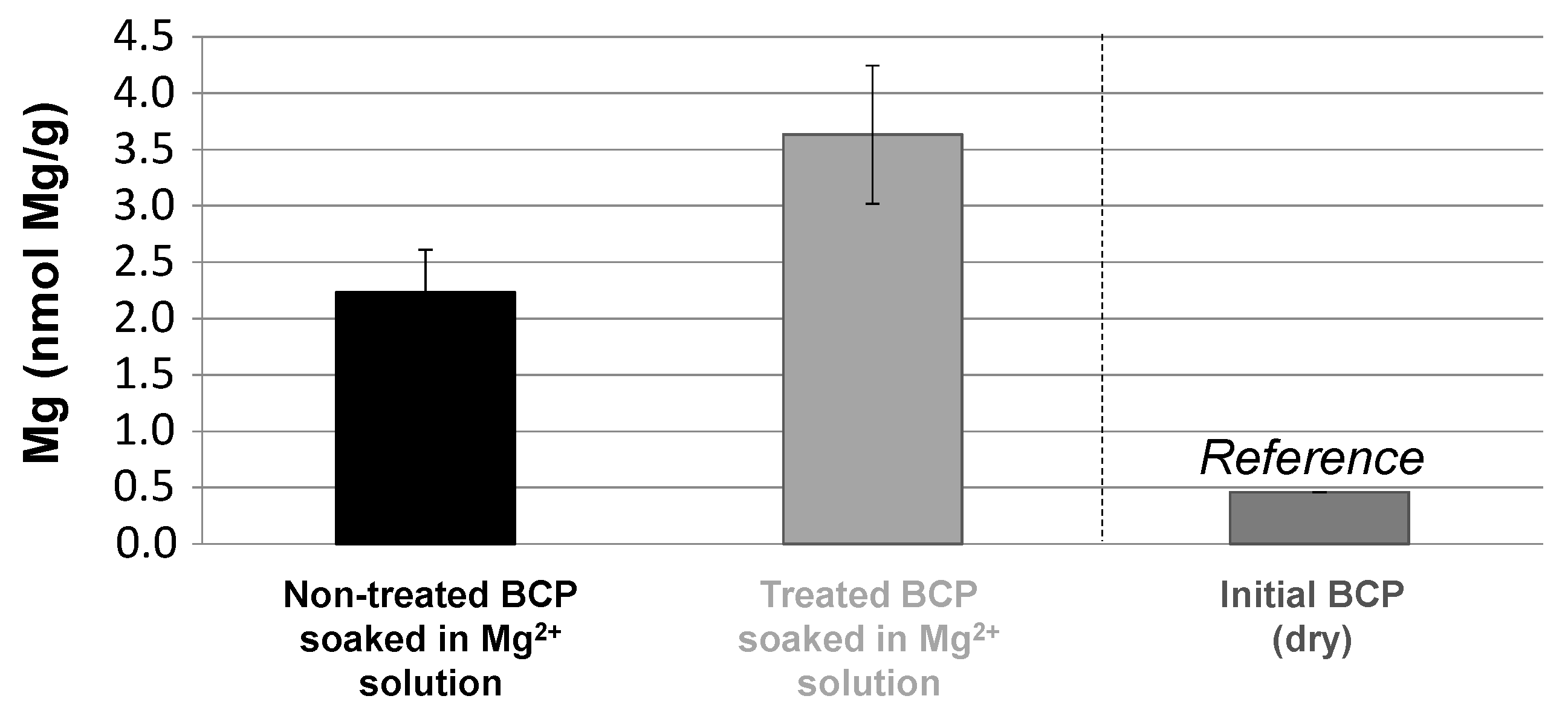
To test the co-presence of biomimetic apatite on the CO
2-treated BCP remodeled surface, fast Mg
2+ → Ca
2+ ion exchange experiments were carried out in solution. Indeed, one feature of bone-like biomimetic apatites is their very high surface ion mobility allowing for fast exchanges (within few seconds/minutes) with various types of ions contained in the surrounding aqueous medium. This property is naturally used by bone mineral in vivo to play an active role in homeostasis [
52]. Such fast ion exchanges were, for instance, reported in the example of the replacement of surface Ca
2+ ions (contained in the hydrated layer on biomimetic apatite nanocrystals) by Mg
2+ or Sr
2+ ions [
18]. This approach may be seen as an indirect way of testing presence of biomimetic apatite, since OCP or DCPD are not known to share this property. Indeed, although ion-doped OCP [
53,
54] and DCPD [
55,
56] have been reported, this necessitates directly adding the doping ions during synthesis. Treated and untreated BCP scaffolds were, thus, immersed in a Mg
2+-containing solution (10 min, magnesium chloride 1M) followed by thorough washing with DI water, and the Mg contents were titrated by AAS. Results are reported in
Figure 7 (expressed per gram of ceramic). Titration on raw BCPs indicates an initial Mg content close to 0.5 nmol Mg/g, with magnesium being a frequent impurity in other alkaline-earth sources, such as calcium ores. The Mg content on CO
2-untreated BCP cubes simply immersed in the Mg
2+ solution led to an Mg content of 2.2 ± 0.4 nmol/g, which is presumably assignable to retention by the HA phase present in the BCP composition. Although some ion sorption ability of HA is significantly lower than that for biomimetic apatites, the possibility of HA capturing ions to some extent has been reported, especially for the remediation of polluting cations [
57,
58]. It is then interesting to compare this Mg content value to the CO
2-treated BCP scaffolds, which was found to be noticeably higher (by a factor 1.6) and reaching 3.6 ± 0.6 nmol Mg/g. The CO
2 treatment thus allowed an additional Mg incorporation of 1.4 ± 0.7 nmol/g. This important increase is in accordance with our hypothesis of the presence of a biomimetic apatite phase besides OCP (a precursor of biomimetic apatite formation) in the treated sample, which is coherent with the NMR results. These conclusions are also similar to those obtained in our preliminary study on the modification of HA and ACP powders by high-pressure CO
2 [
42]. It is likely that the mechanism during the process leading to the precipitation of those phases is similar. Additionally, these two phases (biomimetic apatite and OCP) are known to form plate-like particles, as was observed on the new surface layer shown in
Figure 3. In contrast, the presence of an amorphous CaP phase on the CO
2-treated BCPs seems improbable from the absence of globular morphology typical of ACPs. Concerning the biomimetic apatite phase, it is logical to suspect carbonation (as in bone), considering the presence of carbonate ions in water during the high-pressure CO
2 treatment. To further investigate this latter point, a test was carried out in the CO
2 cell on an “initially non-carbonated” biomimetic apatite gel, precipitated following a previously reported protocol but without final freeze-drying [
38]. As shown by FTIR analyses (
Figure S2c), a clear development of two vibrational bands characteristic of carbonate species in apatite was detected in the domains 800–900 cm
−1 and 1350–1600 cm
−1, corresponding, respectively, to the vibration modes ν
2CO
3 and ν
3CO
3 [
59]. These findings thus indicate that, in the presence of high-pressure CO
2, any nanocrystalline biomimetic apatite formed upon reprecipitation would readily get carbonated, thus approaching the features of natural bone apatite.
Considering the findings presented above, the overall surface remodeling scheme may presumably be summarized by the following main steps:
Partial surface dissolution of the HA: Ca10(PO4)6(OH)2 and β-TCP: Ca3(PO4)2 phases upon acidification under high-pressure CO2.
Local generation of dissolved calcium and phosphate ions (plus OH− ions recombining with H+ to form H2O), besides carbonate ions arising from dissolved CO2.
Local re-increase in the pH linked to the partial dissolution of the phosphate ions present on the surface of the ceramics and during depressurization. Local supersaturation.
Re-precipitation of nonstoichiometric carbonated biomimetic apatite:
It should be noticed that possible precipitation occurred very locally during the process, due to the release of ionic species and the modified pH (linked to the phosphate release). Thus, several local cycles of dissolution/precipitation could occur during the CO2 treatment.
In this scheme, note that we hypothesized a chemical composition for biomimetic apatite that is generic and is often considered in first approximation. In actual nanocrystals, especially those with a poor crystallinity, the presence of a hydrated ionic layer on the nanocrystals surface probably leads to a departure from this generic formula in a way that is still under exploration. This formula, however, aims to highlight the nonstoichiometric character of such biomimetic apatite, which also explains its metastability and high related reactivity [
60]. Note also that the OCP phase itself may depart somewhat from this chemical formula in the present study, taking into account the NMR data suggesting a concerning OCP phase, which could be in the course of crystallization or transformation into biomimetic apatite. Since the initial Ca/P ratio of the BCP surface (assuming a homogeneous initial distribution of the HA and β-TCP components on the commercial scaffolds) is lower than 1.67—it can be calculated at 1.60 for the BCP used here—then the re-crystallization of CaP phases with a lower Ca/P ratio such as biomimetic apatite and OCP (Ca/P = 1.33) is totally coherent. According to our previous experiments on powders [
42] and the literature [
61], it is possible that polynuclear germs could form, followed by crystal growth, corresponding to OCP formation. The HA original grains are likely more stable than β-TCP during the dissolution phenomenon, and might act as nucleation points for the newly precipitated phases. Indeed, no signs of homogeneous precipitation could be detected in the CO
2 cell (no blurring is observed in the medium). The precipitation of biomimetic apatite might occur directly from the supersaturated medium as for OCP, but may also form in a subsequent stage, upon hydrolysis of OCP. In any case, the presence of these phases (with OCP being a known precursor in vitro and in vivo of nanocrystalline apatite) validates the concept pursued in the present work, aiming to modify the surface of commercial BCPs to expose such highly reactive CaP phases for an increased bioactivity, based on previous biological evidence from the literature [
22,
25].
- (ii)
Influence of operating parameters
At this stage, it was interesting to investigate the possible effect of the main parameters of the supercritical CO
2 cell and experimental conditions. The first parameter investigated was the resting time of BCP after the process into the aqueous environment (set to 1 h in the reference protocol). Samples obtained without any resting time showed partial re-precipitation; therefore, nucleation starts before the beginning of the resting time, probably during the outgassing step. In contrast, samples left for a longer resting time (e.g., 24 h) showed an increased specific surface area (from 0.76 ± 0.03 to 0.98 ± 0.03 m
2/g) and a denser surface coating as seen by SEM (
Figure S3), both revealing a higher degree of conversion. Temperature (T) and pressure (P) were two other studied parameters. Upon T increase at 80 bar, the pH of water increases due to the lower solubilization of CO
2 in that phase (as evidenced by PHREEQC calculations,
Figure S4a), which leads to HA and β-TCP solubility decrease (retrograde solubility behavior of several CaP phases). Although reaction kinetics may be favored due to the T increase, these effects do not thermodynamically favor the surface remodeling of the BCP cubes of interest here, which is experimentally seen by a limited change in surface features upon T increase from 37 to 50 and 80 °C (
Figure S5). Conversely, a pressure increase at 37 °C tends to facilitate CO
2 solubilization in water (thus the related pH drop) and slightly increase HA/β-TCP solubility (
Figure S4b). Indeed, experiments performed at 100 bar (rather than 80) suggest that higher pressure favors the surface remodeling, as evidenced by the occurrence of holes of about 1 µm wide, suggesting the disappearance of some initial CaP grains (
Figure S6). Another relevant parameter to follow is the time spent under CO
2 pressure. As expected, allowing for more time (e.g., 100 h rather than 4 h) for BCP in an acidic environment favors an important surface remodeling (
Figure S7). The amount of water in the cell, as determined from the L/S mass ratio, also appears as a key parameter, as it is directly related to the CO
2 dissolution and pH drop. The absence of water or a simple humidification of the BCP samples were not found to allow for BCP surface dissolution/re-precipitation. In contrast, too high a water content (e.g., L/S = 20 compared to 2 in the reference protocol) dilutes the released ions and disfavors CaP reprecipitation by limiting the local supersaturation, thus leading to less efficient remodeling (
Figure S8). Finally, the depressurization speed (to get back to atmospheric pressure) regulates the kinetics of pH change, and is also likely to influence the BCP surface remodeling. Indeed, the medium pH only significantly increases toward the end of the depressurization step, at around 25 bar. Noticeable signs of surface remodeling were effectively seen by modulating the duration of the depressurization step from 2 min to 24 h (
Figure S9).
- (iii)
Functionalization
Beyond this point, we can also propose further widening this concept by exploiting the high reactivity of biomimetic apatite, by adding some biologically active ions to the supercritical CO2 cell ions. Indeed, such ions could then be incorporated during the reprecipitation step upon depressurization (step #4 in the above scheme), thus allowing for new functionalities to be adjoined to the scaffolds, such as antibacterial properties.
In this study, we investigated this point by dissolving copper acetate in the solution containing the BCP scaffolds to be treated in supercritical CO
2. Copper ions indeed proved to be very relevant in the biomedical field and, in particular, for bone engineering thanks to their antibacterial and pro-angiogenic properties, as well as for potentially stimulating osteoblast cell activity [
33,
34]. Cu
2+ ions have also shown an ability to integrate, at least to a certain extent, the apatite lattice, typically in replacement of calcium ions [
62,
63]. Being able to incorporate copper into the structure during our “coating-from” process could be highly relevant to add bioactivity to the BCP “CO
2-activated” scaffolds. Indeed, integration of this element into the scaffold could be of interest to allow for the in vivo release of the antibacterial/bioactive Cu
2+ ions after implantation and/or upon resorption.
In order to evaluate the amount of copper (Cu
2+) integrated into the scaffold after the process, ICP measurements were carried out on the dissolved solids, and also by titration of the medium before and after CO
2 treatment (the incorporated copper content being then determined from the difference between these two values). The two sets of data proved to be in agreement with each other. Tests were carried out for three copper acetate concentrations, denoted
Cu Min,
Cu Int and
Cu Max, as described in the experimental section. ICP results led to the doping rates reported in
Table 1.
It is important to note, however, that the introduction of a copper salt (here copper acetate) to the high-pressure cell before CO
2 treatment may impact the pH value (initial but also along the process), depending on its concentration. As it is not possible to directly measure the pH inside the reactor during the process because of the high pressure, the pH at CO
2 saturation was estimated using the PHREEQC software (version 2) for the three different initial copper concentrations studied here (
Figure S10). As may be seen, upon Cu increases in the medium, the pH at saturation tends to become less acidic, negatively influencing the BCP surface remodeling. Indeed, only very partial dissolution/re-precipitation seems to have occurred in the
Cu Max condition. In contrast, as expected, for the lower copper concentrations
Cu Int and
Cu Min, surface remodeling was clearly visible by SEM (
Figure S11)—although full surface coverage was not reached in these tested conditions. Hg porosimetry also showed a significant downshift of the 210 nm pore size peak to 162 nm, thus illustrating the effective surface remodeling of the BCP porous scaffolds.
We may also notice that, for the highest concentration of
Cu Max, at least one new phase was formed, as seen by SEM observations (
Figure S11), in the form of flower-like structures. EDX analyses revealed that they were mainly composed of copper and phosphorus (
Figure S12), suggesting the possible additional formation of copper phosphate(s) Cu
3(PO
4)
2. nH
2O.
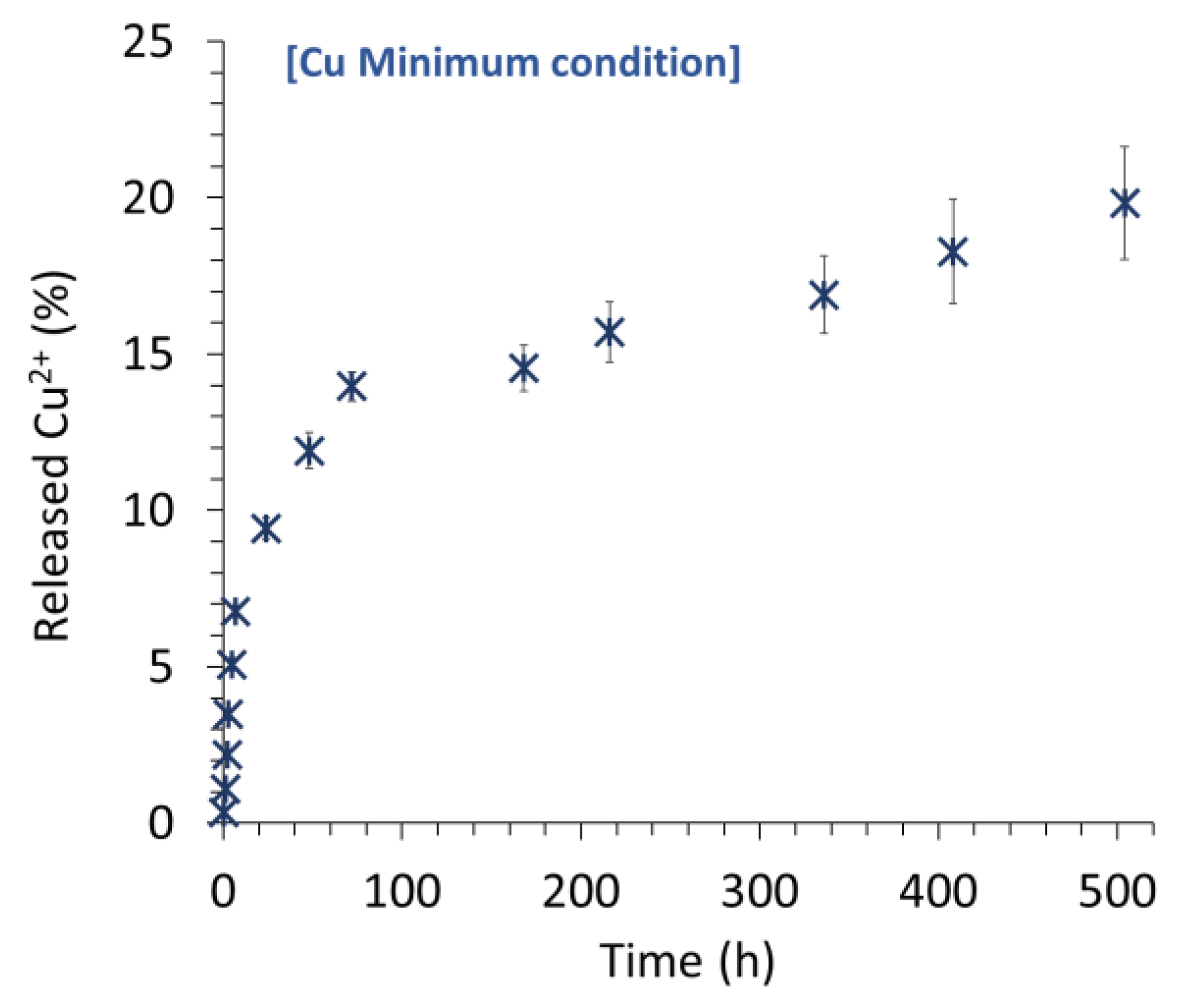
The Cu
2+ release profile of Cu-containing samples was explored in SBF 0.9x (e.g.,
Figure 8 for the example of
Cu Min). Several trends may be identified: a first significant “burst release” effect occurred in the first hours, followed by a decrease in the release kinetics until a rather steady plateau was reached. It may be noted that up to 20% of the copper ions included in the treated BCPs were released after three weeks in these cell-free conditions. These different release stages might reflect the different types of incorporation of copper ions in the modified BCPs, e.g., within the formed apatite nanocrystals (whether from the hydrated layer or the apatitic bulk), within the OCP, or in Cu-containing secondary phase such as copper phosphate(s) that may be more or less present as a function of the copper concentration. The clear identification of such phases is, however, quite delicate considering their top-surface localization, leading to characterization difficulties. Additionally, these “chemical” considerations must contain mechanisms of diffusion of released ionic species within the porous network of the scaffolds. However, these findings confirm the possibility of loading and further releasing copper ions in view of local action (e.g., antibacterial, pro-angiogenic or as osteoblast activator).
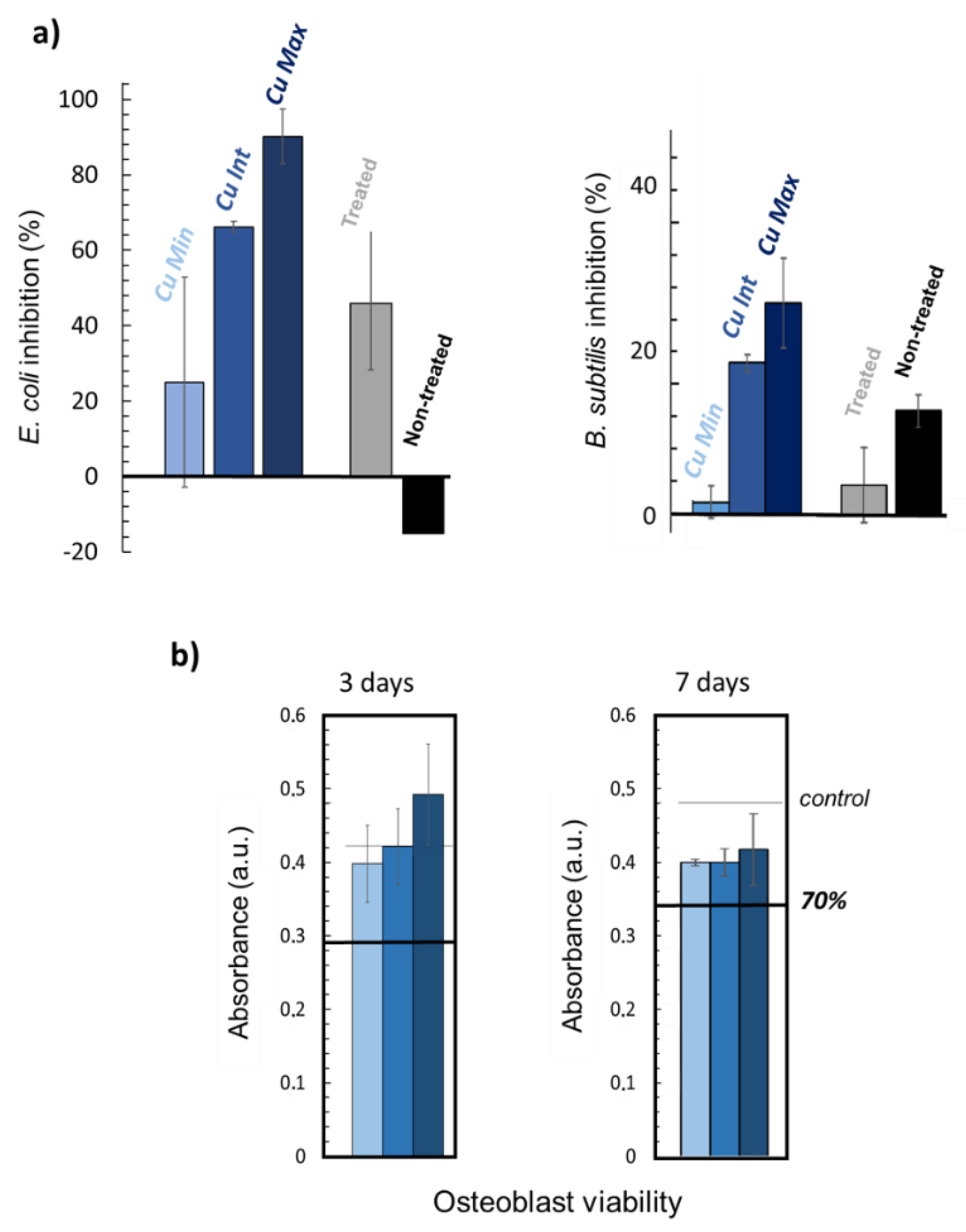
Biological tests were then carried out on the three types of scaffolds (
Cu Min,
Cu Int and
Cu Max). First, antibacterial tests showed an inhibitory effect of the simply modified scaffold without any supplementary ions on both
E. coli and
B. subtilis bacteria (
Figure 9a). This effect is likely related to the modification of the surface topography of the BCPs upon dissolution/reprecipitation, affecting bacteria survival/spreading. Copper-loaded samples demonstrated a greater inhibitory effect upon increasing the Cu content for both types of bacteria (although more markedly for the Gram negative
E. coli strain). This antibacterial effect could be due to a destabilization of the membrane, as already observed with copper ions [
64,
65]. These results thus confirm that doping with antimicrobial ions such as Cu
2+ during the CO
2 treatment process allows for additional properties to be conferred to the treated scaffolds, allowing for tailorable functionalized bone substitutes to be envisioned.
Finally, viability tests were performed on osteoblast cells contacted with the copper-loaded scaffolds (
Figure 9b). Although the viability at day 3 appears to be somewhat more related to the copper concentration than at day 7, this effect is not statistically significant when considering the larger error bars. At day 3, osteoblast viability is overall very satisfactory and close to the control (represented by the black line). At day 7, a slight decrease in viability can be noted compared to the control, but the samples do not exhibit a high cytotoxicity in any case.