Effect of Alumina on Crystallization Behavior of Calcium Ferrite in Fe2O3-CaO-SiO2-Al2O3 System
Abstract
1. Introduction
2. Experimental Procedures
2.1. Preparation of Samples
2.2. Sinter Process
2.3. Phase Determination
3. Results and Discussion
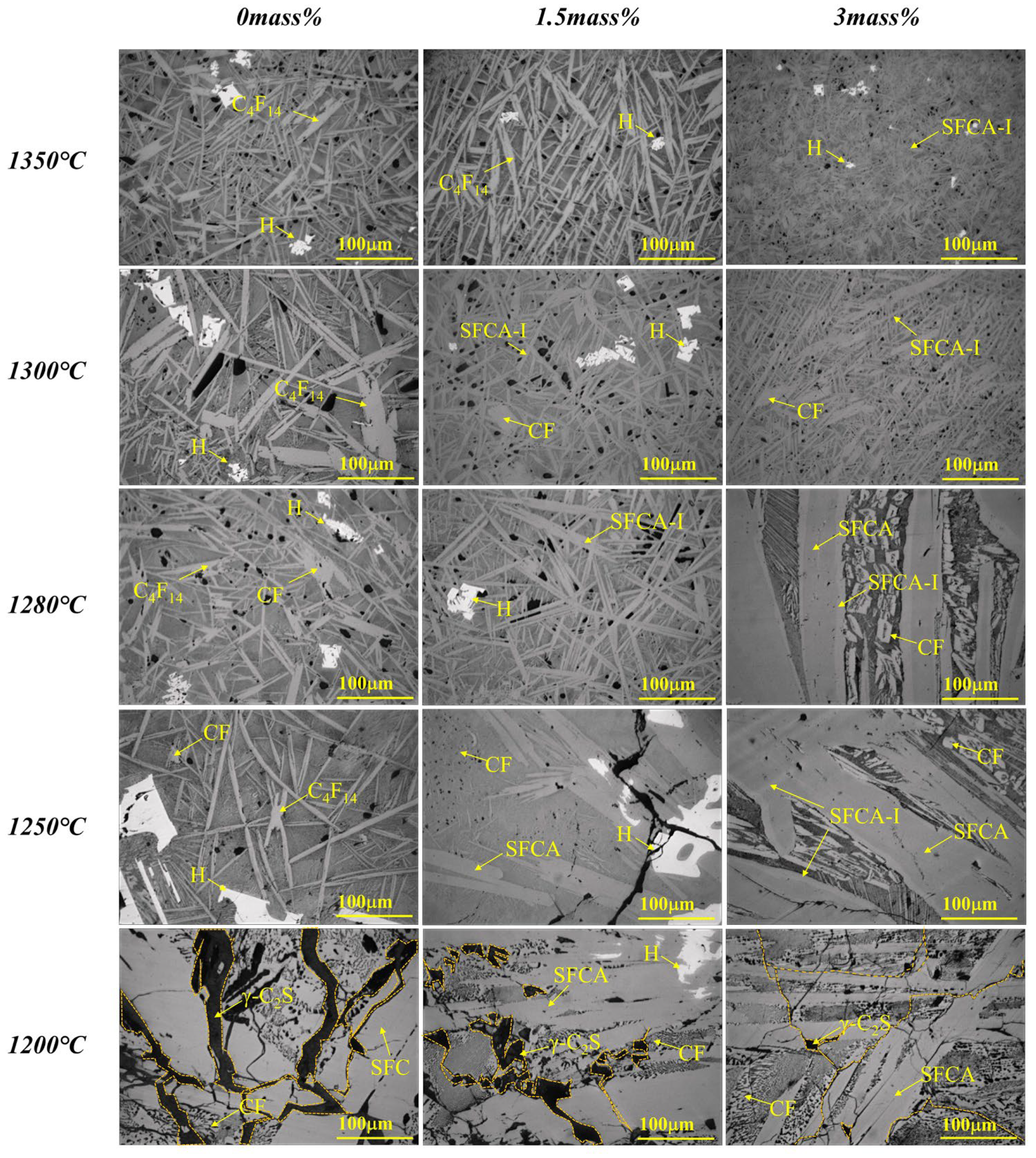
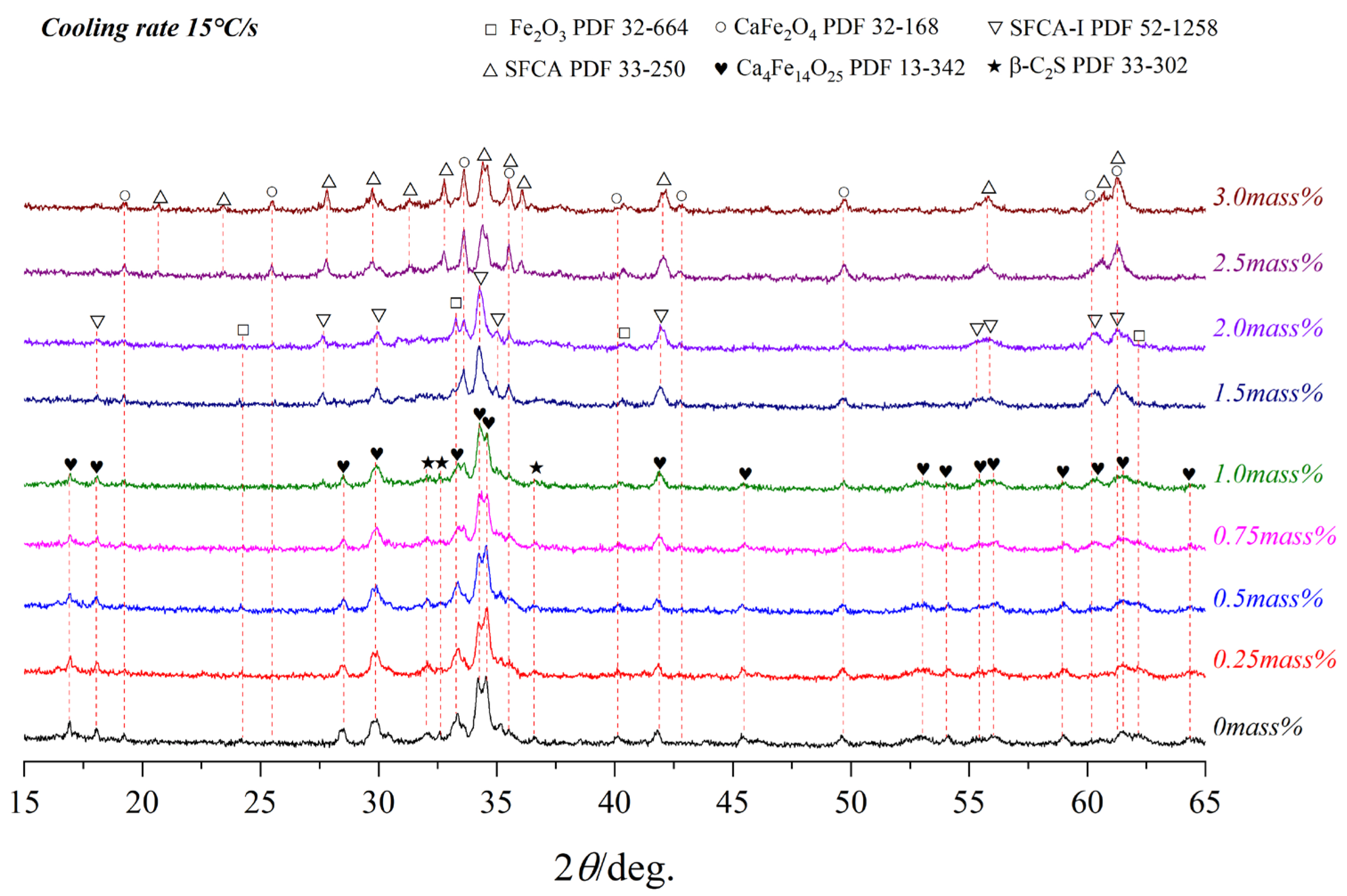
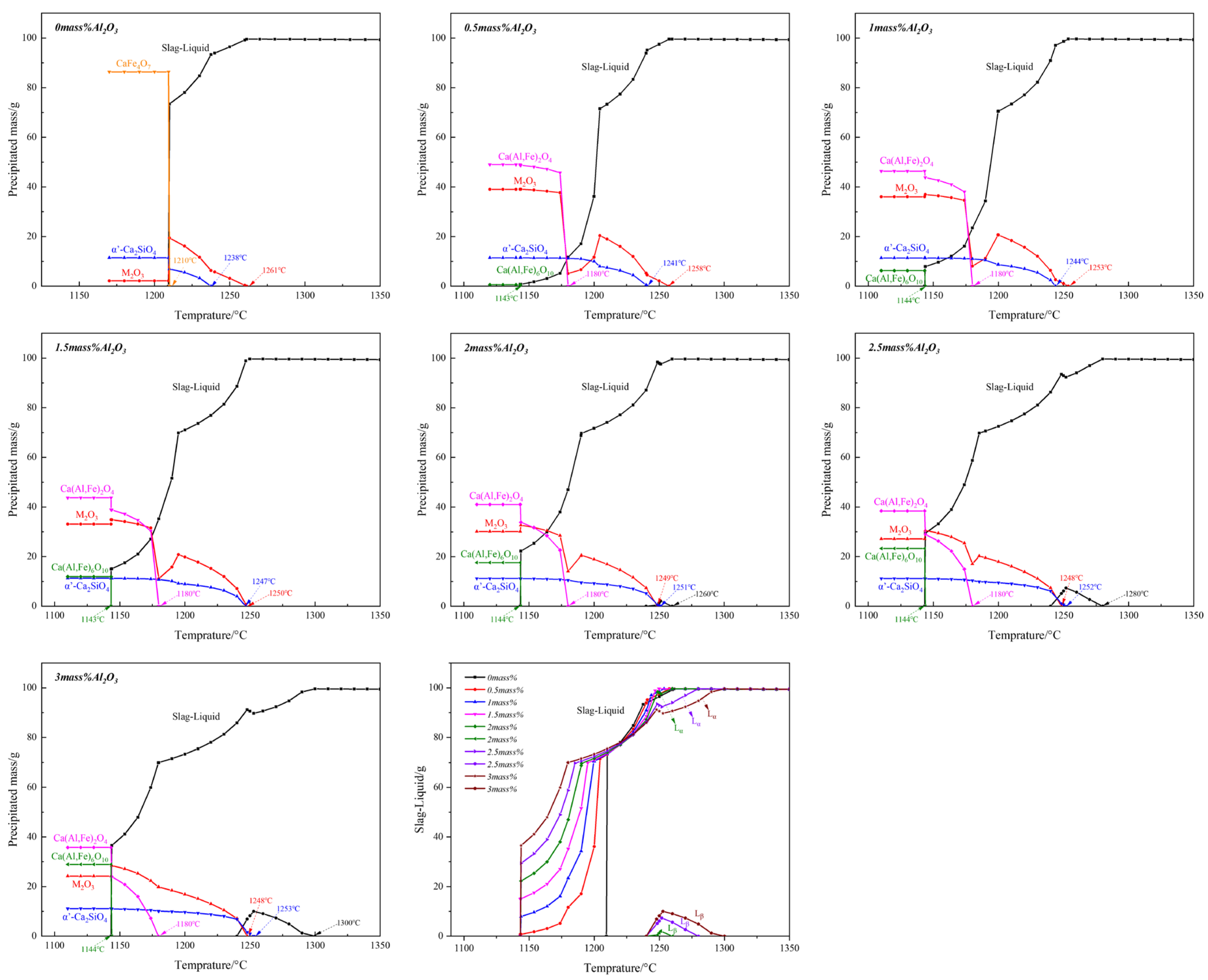
3.1. Effect of Al2O3
- (1)
- When Al2O3 was not added, Ca2+ reacted with Fe3+ and O2− to form C4F14 and CF, while Ca2+ reacted with Si4+ and O2− to form γ-C2S;
- (2)
- When Al2O3 reached 0.5 mass pct, C4F14 disappeared, and CF had gradually increased, indicating that the preferentially crystallized C4F14 reacted with Al3+ and Si4+ to form SFCA-I;
- (3)
- When Al2O3 reached 2.0 mass pct, CF and Fe2O3 had gradually decreased, γ-C2S had not changed significantly, and SFCA-I increased gradually. It shows that CF also participated in the generation of SFCA-I.
- (4)
- When Al2O3 reached 2.5 mass pct, the iron-rich SFCA-I was transformed into SFCA (high Si, high Al). Simultaneously, it promoted the precipitation of Fe2O3. Fe2O3 and CF increased, and Si4+ was mainly involved in generating SFCA, resulting in the decrease of γ-C2S.
- (5)
- When Al2O3 reached 3.0 mass pct, CF and SFCA continued to increase, while γ-C2S decreased and Fe2O3 disappeared.
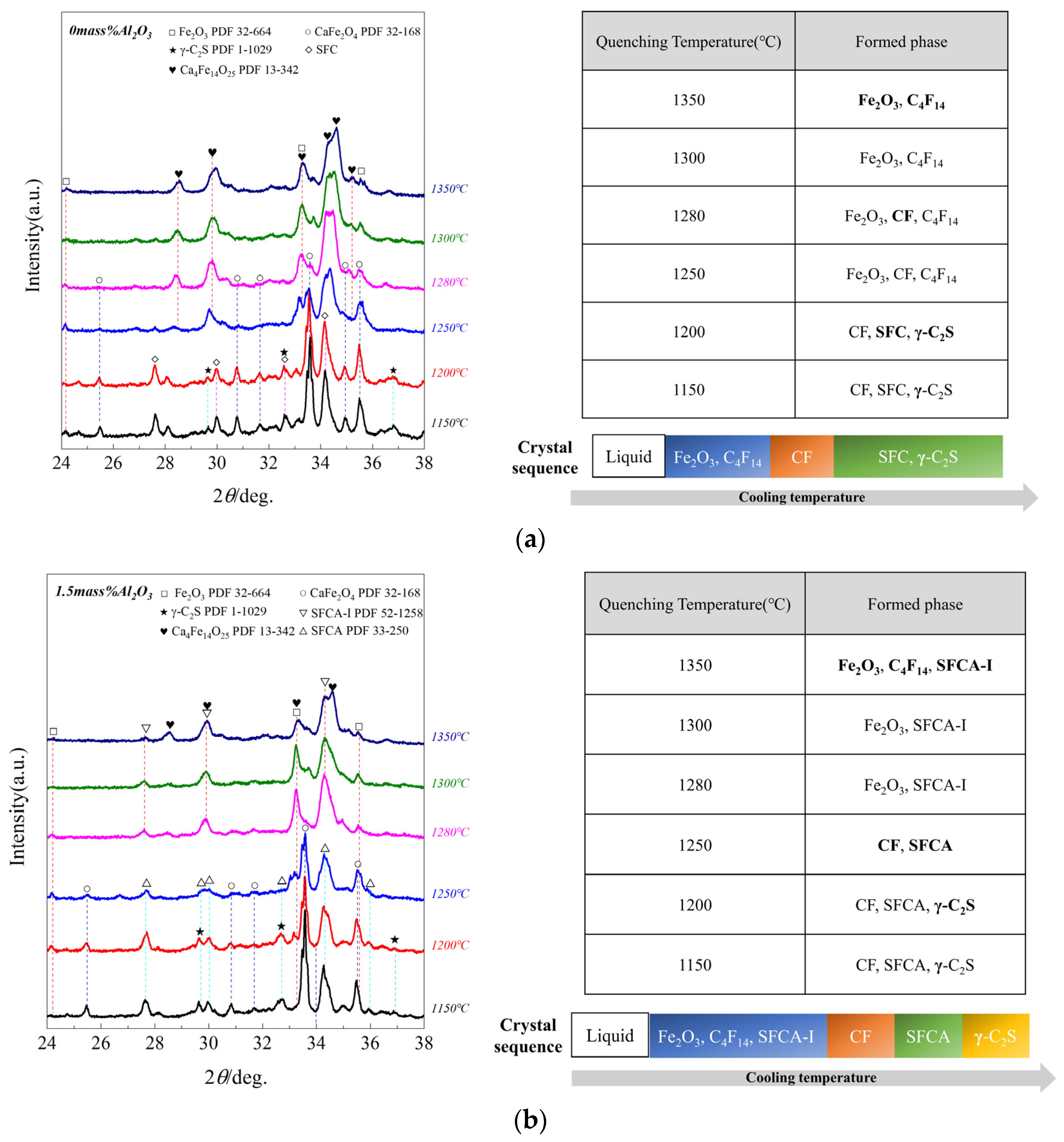
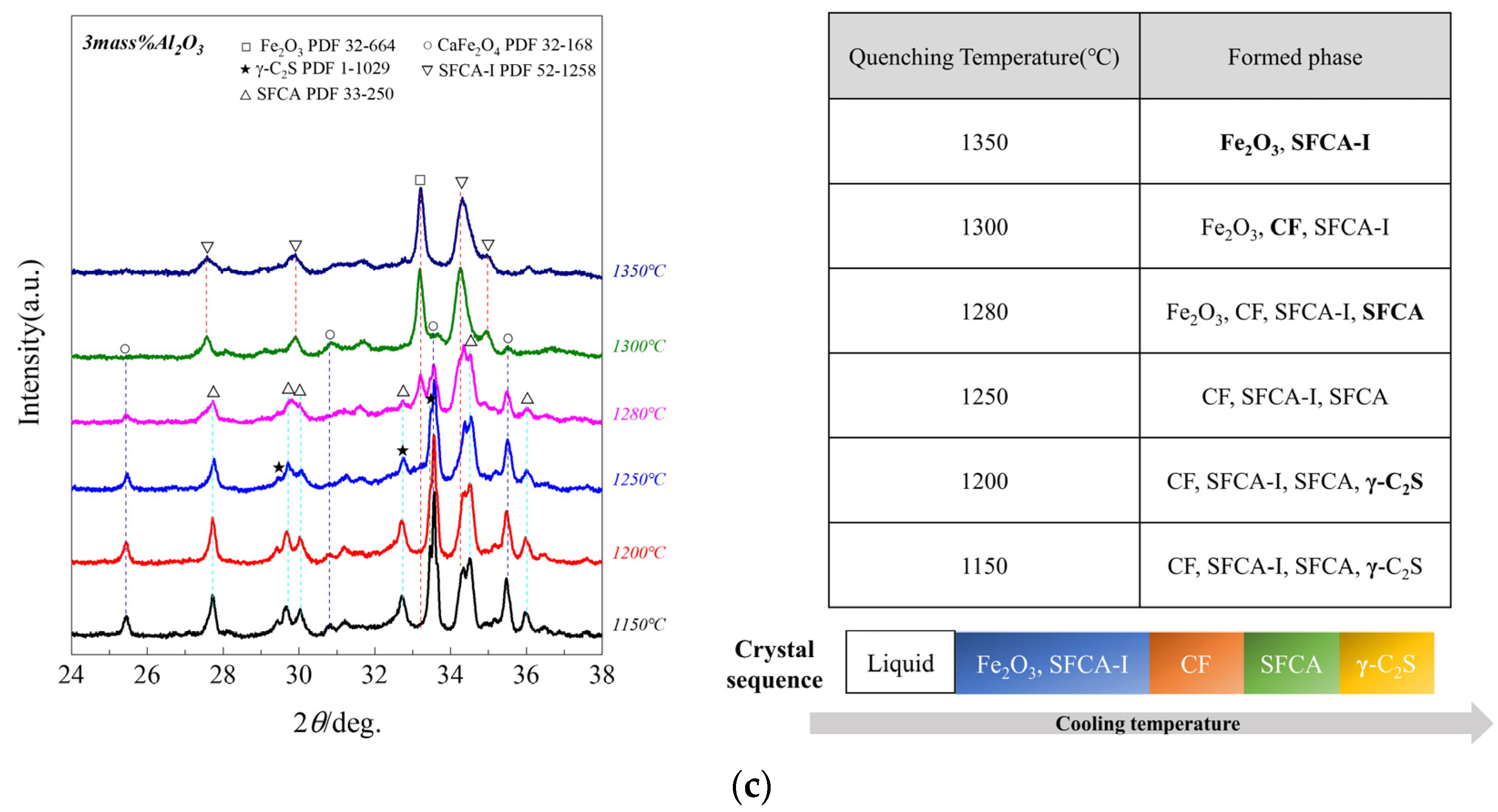
3.2. The Sequence of Crystallization Phase
- (1)
- When Al2O3 was not added, the crystalline phase of quenched samples was Fe2O3 and C4F14 at 1350 °C, while Fe2O3 and C4F14 increased at 1300 °C, simultaneously CF appeared; Fe2O3 and C4F14 increased at 1280 °C, while Fe2O3 decreased. CF increased at 1250 °C, simultaneously SFC and γ-C2S appeared while Fe2O3 disappeared; CF increased at 1200 °C, and CF, SFC, and γ-C2S increased at 1150 °C. So it can be considered the crystallization sequence was (Fe2O3, C4F14) → CF → (SFC, γ-C2S).
- (2)
- When Al2O3 reached 1.5 mass pct, the crystalline phase of quenched samples was Fe2O3 and C4F14 at 1350 °C; SFCA-I appeared, simultaneously Fe2O3 increased at 1300 °C, but C4F14 disappeared; Fe2O3 and SFCA-I increased at 1280 °C; SFCA and CF appeared at 1250 °C, simultaneously Fe2O3 and SFCA-I decreased. At this time, SFCA was formed by the preferentially precipitated SFCA-I and Al3+ and Si4+ in the melt; CF and SFCA increased at 1200 °C, simultaneously γ-C2S appeared; CF, SFCA, and γ-C2S increased at 1150 °C. So it can be considered the crystallization sequence was (Fe2O3, C4F14) → SFCA-I → CF → SFCA → γ-C2S.
- (3)
- When Al2O3 reached 3.0 mass pct, the crystalline phase of quenched samples was Fe2O3 and SFCA-I at 1350 °C; SFCA-I increased while Fe2O3 decreased, simultaneously CF appeared at 1300 °C; Fe2O3 and SFCA-I decreased at 1280 °C while CF increased, simultaneously SFCA appeared; CF and SFCA increased at 1250 °C while SFCA-I decreased, simultaneously Fe2O3 disappeared; SFCA-I decreased at 1200 °C while CF and SFCA increased, simultaneously γ-C2S appeared; SFCA-I decreased at 1150 °C while CF, SFCA, and γ-C2S increased. So it can be considered the crystallization sequence was (Fe2O3, SFCA-I) → CF → SFCA → γ-C2S.
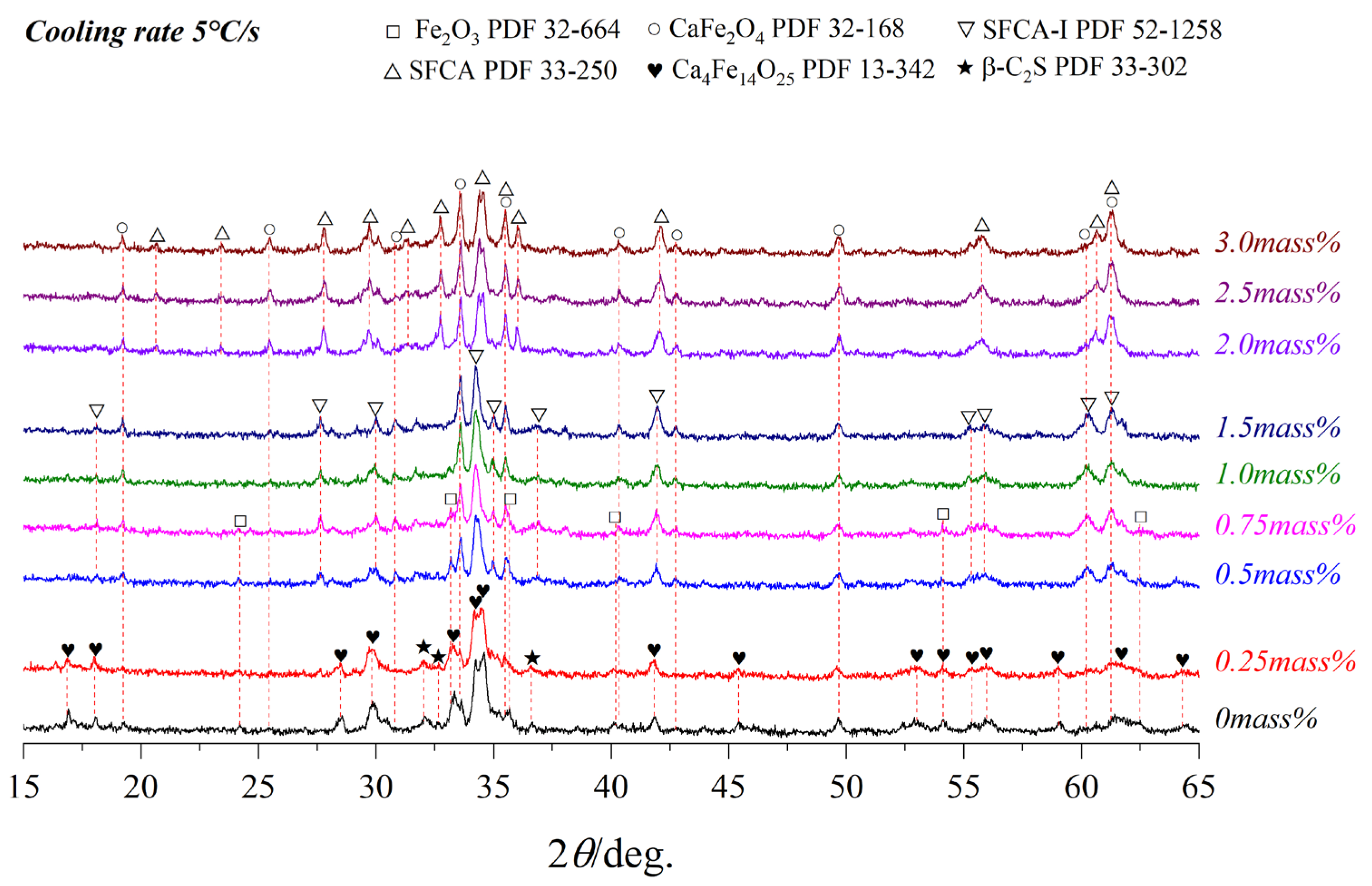
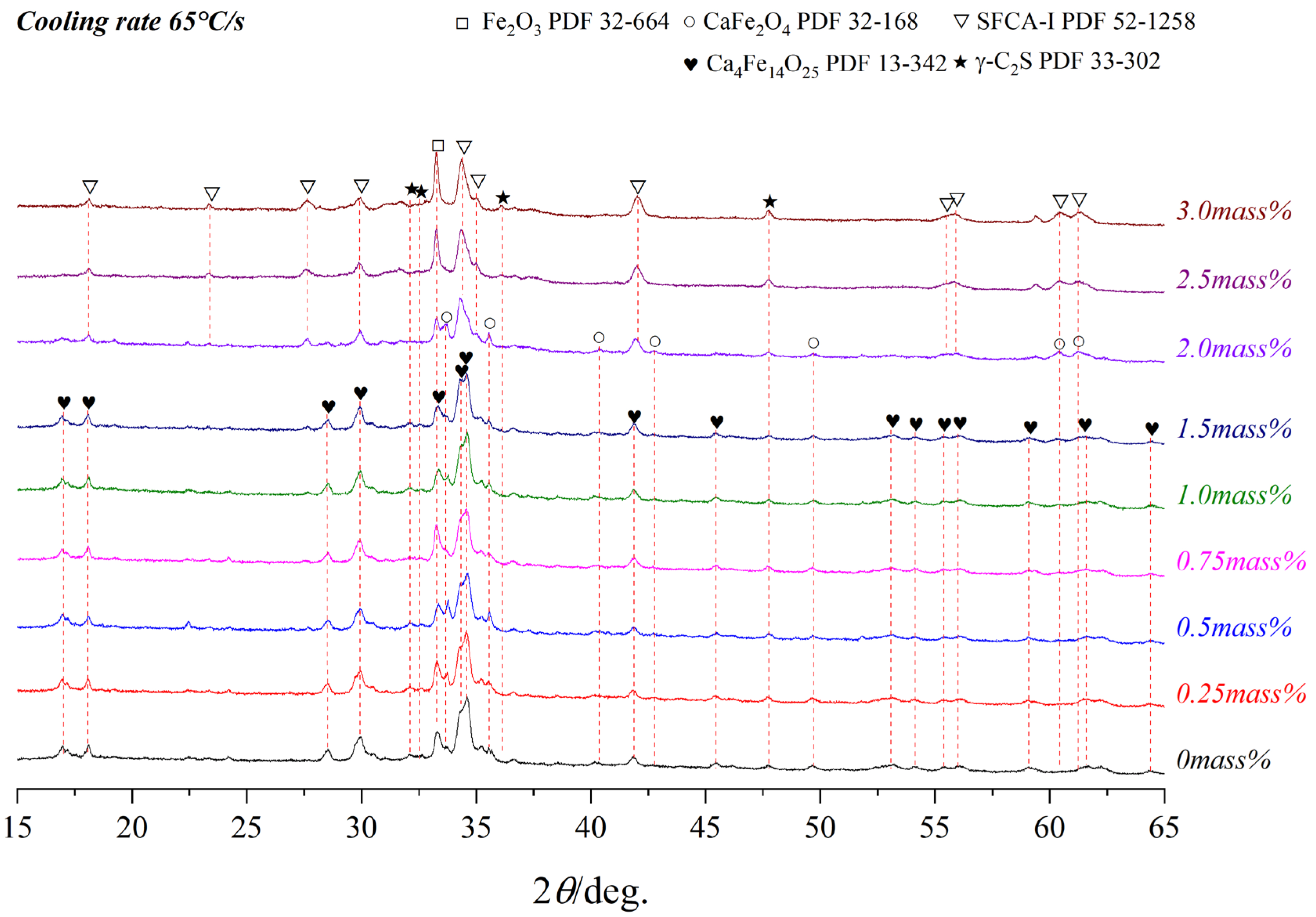
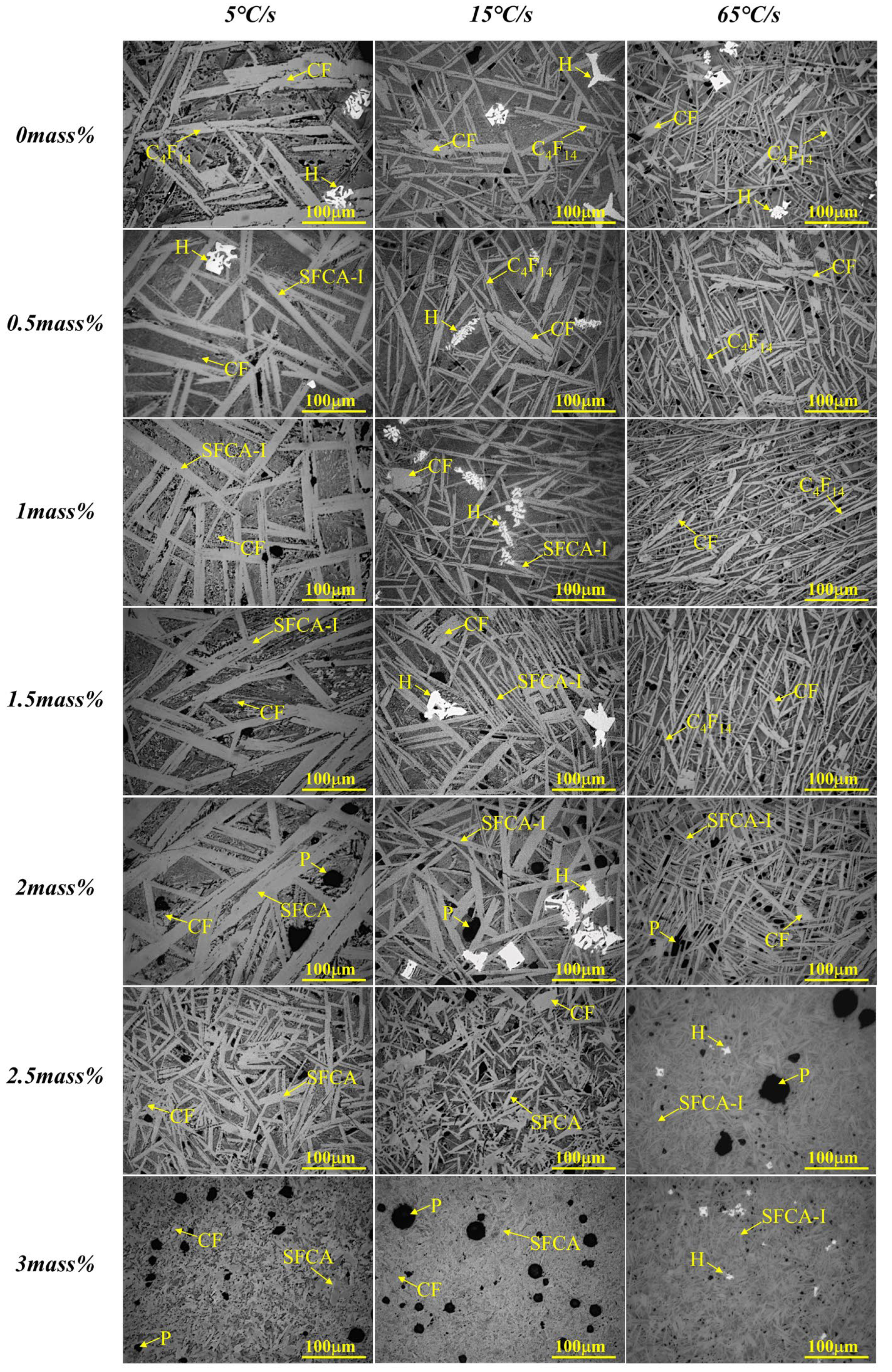
3.3. Effect of Cooling Rate on Crystallization
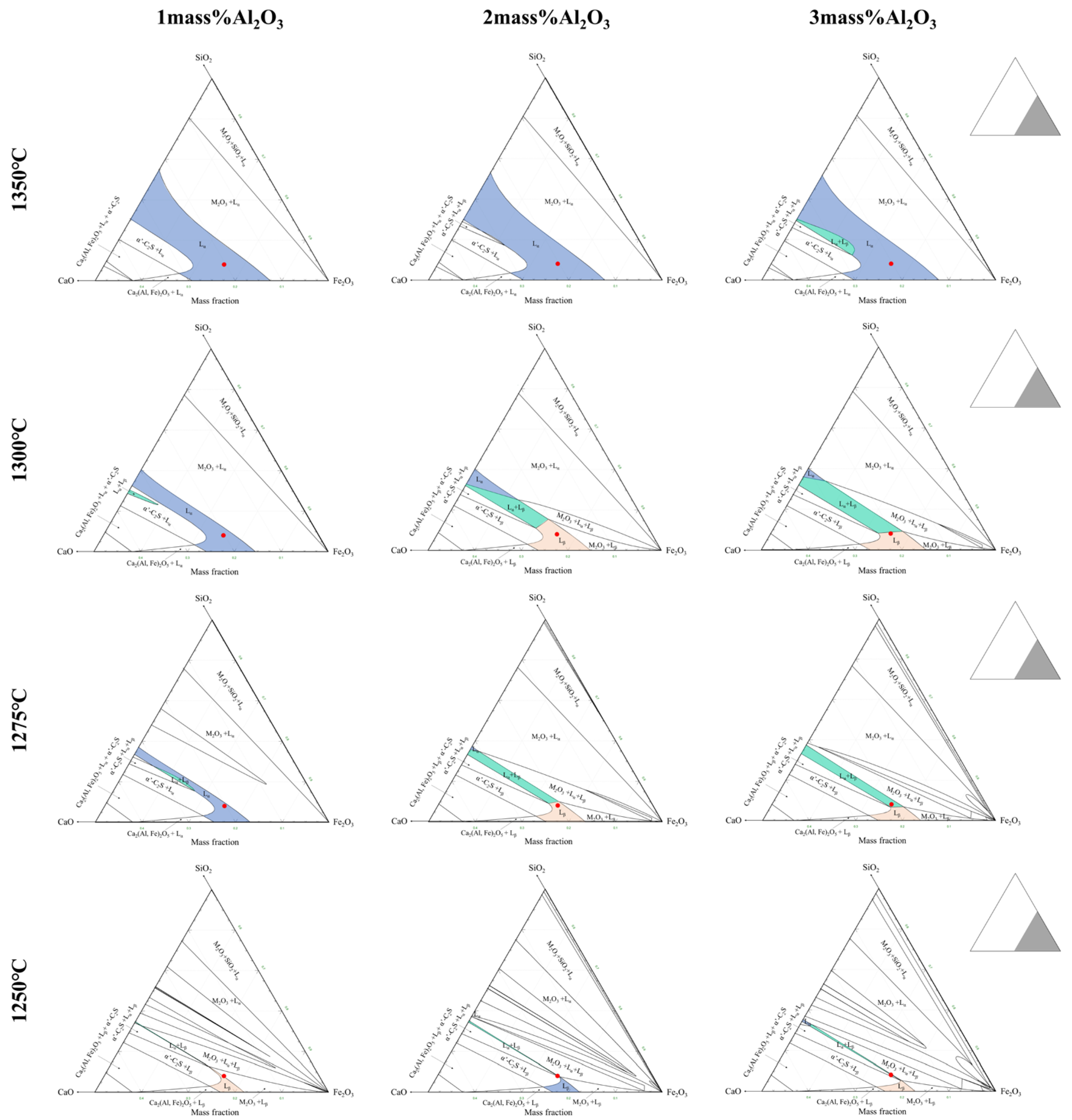
3.4. Discussion on Crystallization Mechanism
- η—solid-liquid mixing viscosity;
- η0—viscosity of pure liquid phase;
- c—a mass fraction of solid phase.
4. Conclusions
- (1)
- Al2O3 has an important effect on the composition of the crystal phase of the Fe2O3-CaO-SiO2-Al2O3 system. Adding alumina promoted the crystallization of Fe2O3, γ-C2S, SFCA-I, and SFCA, while it inhibited the crystallization of C4F14 and SFC. However, the content of CF first decreased and then increased. This is mainly because of the formation of complex calcium ferrite and the transformation of SFCA-I to SFCA.
- (2)
- The crystallization sequence in Fe2O3-CaO-SiO2-Al2O3 melt under different Al2O3 content was investigated, where the corresponding crystalline order is (Fe2O3, C4F14) → CF → (SFC, γ-C2S), (Fe2O3, C4F14) → SFCA-I → CF → SFCA → γ-C2S, and (Fe2O3, SFCA-I) → CF → SFCA → γ-C2S under the Al2O3 content of 0 mass pct, 1.5 mass pct, and 3.0 mass pct respectively. It can be concluded that the C4F14 reacts with Si4+ and Al3+ in the melt to form SFCA-I (C4F14 + Si4+ + Al3+ → SFCA-I), and then SFCA-I reacts with Si4+ and Al3+ to form SFCA (SFCA-I + Si4+ + Al3+ → SFCA).
- (3)
- As the cooling rate increase, C4F14, SFCA-I, Fe2O3, β-C2S, and the amorphous phases are increased while CF and SFCA are reduced, and the crystal transformation from β-C2S to γ-C2S can be effectively inhibited. However, when the cooling rate was increased from 15 °C/s to 65 °C/s, C4F14 was found to crystallize before Fe2O3.
- (4)
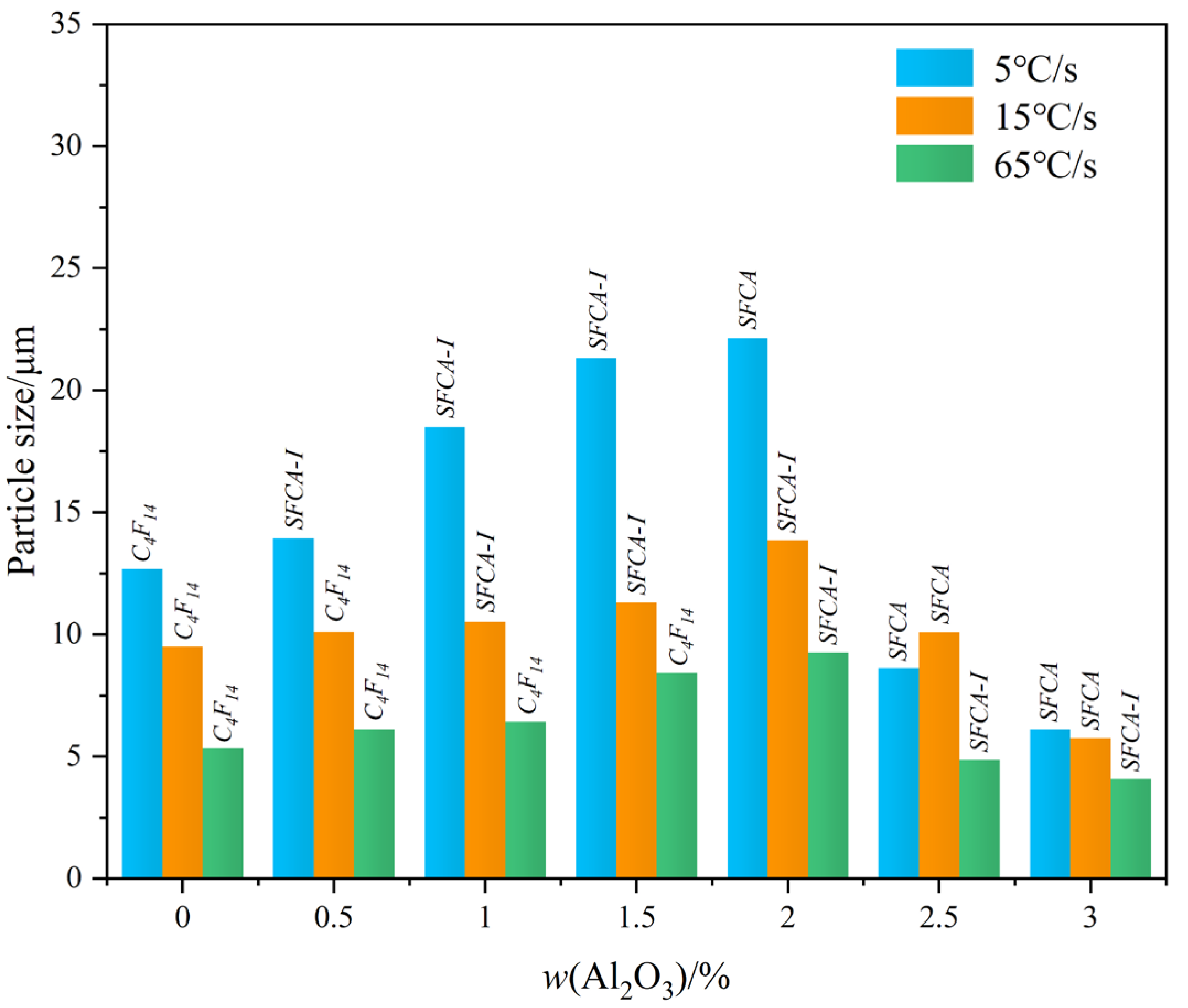
- The crystal size of complex calcium ferrite first increases and then decreases by an increase of Al2O3 content, and the order of crystal morphology evolved is from the strip, columnar to needle-shaped. When Al2O3 content reached or exceeded 2.5 mass pct, the viscosity of Fe2O3-CaO-SiO2-Al2O3 melt increased sharply, resulting in the decrease in the crystalline size of calcium ferrite.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Loo, C.E.; Leung, W. Factors Influencing the Bonding Phase Structure of Iron Ore Sinters. ISIJ Int. 2003, 43, 1393–1402. [Google Scholar] [CrossRef]
- Guo, H.; Guo, X.M. Effect of Aluminum Dissolved in Hematite on Formation of Calcium Ferrites at 1473 K. Metall. Mater. Trans. B 2018, 49, 1974–1984. [Google Scholar] [CrossRef]
- Yang, L.X.; Loo, C.E. Structure of Sinters Formed from Complex Chinese Iron Ores. ISIJ Int. 1997, 37, 449–457. [Google Scholar] [CrossRef][Green Version]
- Park, T.J.; Min, S.K.; Kim, H. Estimation of Reducibility of Sinter with Continuous Measurement of Reduction Degradation Test. Met. Mater. Int. 2020, 26, 532–540. [Google Scholar] [CrossRef]
- Lu, L.; Holmes, R.J.; Manuel, J.R. Effects of Alumina on Sintering Performance of Hematite Iron Ores. ISIJ Int. 2007, 47, 349–358. [Google Scholar] [CrossRef]
- Nicol, S.; Chen, J.; Pownceby, M.I.; Webster, N.A.S. A Review of the Chemistry, Structure and Formation Conditions of Silico-Ferrite of Calcium and Aluminum (‘SFCA’) Phases. ISIJ Int. 2018, 58, 2157–2172. [Google Scholar] [CrossRef]
- Sato, S.; Kawaguuchi, T.; Ichidate, M.; Yoshinaga, M. Mineral Forming and Its Composition Model for Iron Ore Sinter. Tetsu-to-Hagane 1987, 73, 964. [Google Scholar] [CrossRef]
- Bristow, N.J.; Loo, C.E. Sintering Properties of Iron Ore Mixes Containing Titanium. ISIJ Int. 1992, 32, 819–828. [Google Scholar] [CrossRef]
- Nyembwe, A.M.; Garbers-Craig, A.M. Study of iron ore sinter when fine ore is replaced with coarse ore, using infrared furnace and sinter pot tests. Ironmak. Steelmak. 2014, 41, 173–181. [Google Scholar] [CrossRef]
- Wang, Z.; Pinson, D.; Chew, S.; Monaghan, B.J.; Rogers, H.; Zhang, G.Q. Mineral Phase Formation and Zinc Removal during Sintering of Filter Cake Wastes. ISIJ Int. 2016, 56, 505–512. [Google Scholar] [CrossRef]
- Scarlett, N.V.Y.; Pownceby, M.I.; Madsen, I.C.; Christensen, A.N. Reaction sequences in the formation of silico-ferrites of calcium and aluminum in iron ore sinter. Metall. Mater. Trans. B 2004, 35, 929–936. [Google Scholar] [CrossRef]
- Guo, X.M. The Formation and Mineralogy of Calcium Ferrite in Sintering Process; Metallurgical Industry Press: Beijing, China, 1999; pp. 143–146. [Google Scholar]
- Patrick, T.R.C.; Pownceby, M.I. Stability of silico-ferrite of calcium and aluminum (SFCA) in air-solid solution limits between 1240 °C and 1390 °C and phase relationships within the Fe2O3-CaO-Al2O3-SiO2 (FCAS) system. Metall. Mater. Trans. B 2002, 33, 79–89. [Google Scholar] [CrossRef]
- Webster, N.A.S.; Pownceby, M.I.; Madsen, I.C.; Kimpton, J.A. Silico-ferrite of Calcium and Aluminum (SFCA) Iron Ore Sinter Bonding Phases: New Insights into Their Formation During Heating and Cooling. Metall. Mater. Trans. B 2012, 43, 1344–1357. [Google Scholar] [CrossRef]
- Webster, N.A.S.; Churchill, J.G.; Tufaile, F.; Pownceby, M.I.; Manuel, J.R.; Kimpton, J.A. Fundamentals of Silico-Ferrite of Calcium and Aluminium (SFCA) and SFCA-I Iron Ore Sinter Bonding Phase Formation: Effects of Titanomagnetite-based Ironsand and Titanium Addition. ISIJ Int. 2016, 56, 1715–1722. [Google Scholar] [CrossRef]
- Hsieh, L.H.; Whiteman, J.A. Effect of Raw Material Composition on the Mineral Phases in Lime-fluxed Iron Ore Sinter. ISIJ Int. 1993, 33, 462–473. [Google Scholar] [CrossRef]
- Cai, B.; Watanabe, T.; Kamijo, C.; Susa, M.; Hayashi, M. Comparison between Reducibilities of Columnar Silico-ferrite of Calcium and Aluminum (SFCA) Covered with Slag and Acicular SFCA with Fine Pores. ISIJ Int. 2018, 58, 642–651. [Google Scholar] [CrossRef]
- Webster, N.A.S.; O’Dea, D.P.; Ellis, B.G.; Pownceby, M.I. Effects of Gibbsite, Kaolinite and Al-rich Goethite as Alumina Sources on Silico-Ferrite of Calcium and Aluminium (SFCA) and SFCA-I Iron Ore Sinter Bonding Phase Formation. ISIJ Int. 2017, 57, 41–47. [Google Scholar] [CrossRef]
- Liles, D.C.; Villiers, J.P.R.; Kahlenberg, V. Refinement of iron ore sinter phases: A silico-ferrite of calcium and aluminium (SFCA) and an Al-free SFC, and the effect on phase quantification by X-ray diffraction. Mineral. Petrol. 2016, 110, 141–147. [Google Scholar] [CrossRef]
- Ding, C.Y.; Lv, X.W.; Chen, Y.; Bai, C.G. Crystallization Kinetics of 2CaO·Fe2O3 and CaO·Fe2O3 in the CaO–Fe2O3 System. ISIJ Int. 2016, 56, 1157–1163. [Google Scholar] [CrossRef]
- Yang, D.W.; Wang, W.; Li, J.X.; Xu, R.S.; Wang, X.Z.; Wang, G. Effect and Mechanism of Alumina on the Morphology and Mechanical Properties of Calcium Ferrite. Metall. Mater. Trans. B 2020, 51, 776–785. [Google Scholar] [CrossRef]
- Park, T.J.; Choi, J.S.; Min., D.J. Influence of Al2O3 Content and Cooling Rate on Crystallization in Fe2O3–CaO–SiO2–Al2O3 Systems. Met. Mater. Int. 2021. [Google Scholar] [CrossRef]
- Fan, X.H. Principle and Technology of Optimum Blending of Iron Ore Sintering; Metallurgical Industry Press: Beijing, China, 2013; pp. 62–65. [Google Scholar]
- Wang, B.; Zhao, W.; Zhang, X.H.; Hu, S.Y.; Guo, H.W.; Chu, M.S. Revealing the Softening-Melting Behaviors and Slag Characteristics of Vanadium-Titanium Magnetite Burden with Various MgO Addition. Minerals 2022, 12, 842. [Google Scholar] [CrossRef]
- Wang, Z.J.; Sun, Y.Q.; Sridhar, S.; Zhang, M.; Zhang, Z.T. Selective Crystallization Behavior of CaO-SiO2-Al2O3-MgO-FetO-P2O5 Steelmaking Slags Modified through P2O5 and Al2O3. Metall. Mater. Trans. B 2015, 46, 2246–2254. [Google Scholar] [CrossRef]
- Wang, Z.J.; Sun, Y.Q.; Sridrar, S.; Zhang, M.; Zhang, Z.T. Investigation on Viscosity and Nonisothermal Crystallization Behavior of P-Bearing Steelmaking Slags with Varying TiO2 Content. Metall. Mater. Trans. B 2017, 48, 527–537. [Google Scholar] [CrossRef]
- Wang, W.; Yang, D.W.; Ou-Yang, Z.L.; Xu, R.S.; Song, M.M. Effect of SiO2 on the Formation of Acicular Calcium Ferrite in Sinter. Metall. Mater. Trans. B 2019, 50, 678–687. [Google Scholar] [CrossRef]
- Xin, R.F.; Guo, X.M. Effect of SiO2 on Crystallization of Calcium Ferrites in Fe2O3–CaO–SiO2–Al2O3 System in Cooling Process. Metall. Mater. Trans. B 2022, 53, 1904–1919. [Google Scholar] [CrossRef]
- Chang, R.G.; Yan, Y.T.; Zhang, J.Y.; Zhu, Z.L.; Gu, J.H. Large-grain and smooth cesium doped CH3NH3PbI3 perovskite films by cesium iodide post-treatment for improved solar cells. Thin Solid Film. 2020, 712, 138279. [Google Scholar] [CrossRef]
- Roscoe, R. The viscosity of suspensions of rigid spheres. Bri. J. Appl. Phys. 1952, 3, 267. [Google Scholar] [CrossRef]







| Samples | Fe2O3 | CaO | SiO2 | Al2O3 | LT (°C) |
|---|---|---|---|---|---|
| No. 1 | 75.64 | 20.36 | 4 | 0 | 1261 |
| No. 2 | 75.45 | 20.31 | 3.99 | 0.25 | 1259 |
| No. 3 | 75.26 | 20.26 | 3.98 | 0.50 | 1257 |
| No.4 | 75.07 | 20.21 | 3.97 | 0.75 | 1256 |
| No.5 | 74.88 | 20.16 | 3.96 | 1.00 | 1254 |
| No.6 | 74.51 | 20.05 | 3.94 | 1.50 | 1250 |
| No.7 | 74.13 | 19.95 | 3.92 | 2.00 | 1256 |
| No.8 | 73.75 | 19.85 | 3.90 | 2.50 | 1276 |
| No.9 | 73.37 | 19.75 | 3.88 | 3.00 | 1293 |
| Sample No. | Marked Points | Crystal Phase | Elements (at Pct) | ||||
|---|---|---|---|---|---|---|---|
| Fe | Ca | Si | Al | O | |||
| 0mass% | P1 | C4F14 | 46.20 | 13.54 | 1.79 | 0.74 | 37.72 |
| P2 | CF | 39.38 | 21.93 | 1.38 | 0.94 | 36.37 | |
| P3 | γ-C2S | 0.61 | 40.07 | 20.33 | 0.24 | 38.75 | |
| P4 | Fe2O3 | 62.63 | 0.06 | 1.45 | 0.95 | 34.91 | |
| 1.0mass% | P2 | CF | 41.42 | 23.82 | 0.00 | 0.10 | 34.67 |
| P3 | γ-C2S | 0.48 | 41.01 | 19.91 | 0.31 | 38.29 | |
| P4 | Fe2O3 | 67.98 | 0.14 | 0.42 | 0.91 | 30.54 | |
| P5 | SFCA-I | 47.64 | 14.79 | 2.42 | 2.15 | 33.01 | |
| 1.5mass% | P2 | CF | 38.51 | 22.76 | 0.31 | 0.29 | 38.12 |
| P3 | γ-C2S | 0.86 | 37.95 | 17.98 | 0.26 | 42.96 | |
| P4 | Fe2O3 | 64.33 | 0.27 | 0.00 | 0.72 | 34.67 | |
| P5 | SFCA-I | 43.01 | 14.34 | 3.83 | 3.65 | 35.16 | |
| 2.0mass% | P2 | CF | 41.02 | 24.77 | 1.40 | 1.32 | 31.49 |
| P3 | γ-C2S | 0.64 | 42.53 | 21.15 | 0.35 | 35.33 | |
| P4 | Fe2O3 | 68.94 | 0.00 | 0.04 | 0.92 | 30.10 | |
| P5 | SFCA-I | 47.06 | 14.56 | 3.20 | 3.20 | 31.98 | |
| 2.5mass% | P2 | CF | 39.31 | 24.09 | 0.01 | 0.28 | 36.31 |
| P3 | γ-C2S | 0.35 | 39.08 | 19.78 | 0.16 | 40.63 | |
| P4 | Fe2O3 | 62.79 | 0.28 | 0.00 | 0.84 | 36.09 | |
| P6 | SFCA | 41.35 | 13.26 | 3.83 | 5.00 | 36.56 | |
| 3.0mass% | P2 | CF | 41.32 | 26.74 | 0.00 | 0.40 | 31.53 |
| P3 | γ-C2S | 0.94 | 45.13 | 17.11 | 0.00 | 36.82 | |
| P6 | SFCA | 42.86 | 13.37 | 4.17 | 7.19 | 32.41 | |
| Sample No. | Marked Points | Phase | Elements | ||||
|---|---|---|---|---|---|---|---|
| Fe | Ca | Si | Al | O | |||
| 0mass% | P1 | Fe2O3 | 68.63 | 0.49 | 1.36 | 0 | 28.94 |
| P2 | C4F14 | 51.13 | 14.23 | 1.16 | 0 | 33.48 | |
| P3 | Slag phase | 35.81 | 22.88 | 5.90 | 0 | 35.41 | |
| 1.5mass% | P1 | C4F14 | 47.25 | 12.37 | 0.00 | 1.08 | 39.30 |
| P2 | SFCA-I | 44.04 | 12.68 | 1.08 | 2.20 | 39.99 | |
| P3 | Slag phase | 44.52 | 15.88 | 2.71 | 2.55 | 34.34 | |
| 3.0mass% | P1 | Fe2O3 | 54.96 | 0.62 | 0.00 | 1.10 | 43.32 |
| P2 | SFCA-I | 39.52 | 11.80 | 2.31 | 4.84 | 41.53 | |
| P3 | Slag phase | 36.85 | 15.91 | 3.18 | 4.12 | 39.95 | |
| Cooling Rate (°C/s) | Al2O3 (Mass Pct) | ||||||
|---|---|---|---|---|---|---|---|
| 0 | 0.5 | 1 | 1.5 | 2 | 2.5 | 3 | |
| 5 | CF | CF | CF | CF | CF | CF | CF |
| C4F14 | SFCA-I | SFCA-I | SFCA-I | SFCA | SFCA | SFCA | |
| Fe2O3 | Fe2O3 | ||||||
| β-C2S | |||||||
| 15 | CF | CF | CF | CF | CF | CF | CF |
| C4F14 | SFCA-I | SFCA-I | SFCA-I | SFCA-I | SFCA-I | SFCA-I | |
| Fe2O3 | Fe2O3 | Fe2O3 | Fe2O3 | Fe2O3 | Fe2O3 | Fe2O3 | |
| β-C2S | β-C2S | β-C2S | |||||
| 65 | CF | CF | CF | CF | CF | ||
| C4F14 | C4F14 | C4F14 | C4F14 | SFCA-I | SFCA-I | SFCA-I | |
| Fe2O3 | Fe2O3 | Fe2O3 | Fe2O3 | Fe2O3 | |||
| β-C2S | β-C2S | β-C2S | β-C2S | β-C2S | β-C2S | β-C2S | |
| Al2O3 (Mass Pct) | Marked Points | Phase | Elements (at Pct) | ||||
|---|---|---|---|---|---|---|---|
| Fe | Ca | Si | Al | O | |||
| 0 | P1 | C4F14 | 52.06 | 12.37 | 0.79 | 0 | 34.78 |
| P2 | CF | 40.74 | 19.59 | 3.96 | 0 | 35.72 | |
| P3 | Slag phase | 37.10 | 20.93 | 6.93 | 0 | 35.04 | |
| 1.0 | P2 | CF | 40.49 | 21.60 | 4.95 | 1.77 | 31.20 |
| P4 | SFCA-I | 48.28 | 14.24 | 2.09 | 0.92 | 34.48 | |
| 1.5 | P2 | CF | 42.16 | 19.75 | 4.89 | 2.06 | 31.14 |
| P4 | SFCA-I | 49.19 | 14.77 | 2.94 | 2.77 | 30.33 | |
| 2.0 | P2 | CF | 43.43 | 26.11 | 0 | 0.69 | 29.77 |
| P5 | SFCA | 47.35 | 14.22 | 3.40 | 4.42 | 30.62 | |
| 2.5 | P2 | CF | 39.39 | 21.58 | 4.51 | 3.94 | 30.58 |
| P5 | SFCA | 43.77 | 12.73 | 4.28 | 6.27 | 32.95 | |
| 3.0 | P2 | CF | 39.39 | 21.58 | 4.51 | 3.94 | 30.58 |
| P5 | SFCA | 40.10 | 13.17 | 4.26 | 6.25 | 36.22 | |
| Al2O3 Content (Mass Pct) | Primary Phase | Primary Crystallization Temperature (°C) |
|---|---|---|
| 0 | M2O3 | 1261 |
| 0.5 | M2O3 | 1258 |
| 1.0 | M2O3 | 1253 |
| 1.5 | M2O3 | 1250 |
| 2.0 | α’-C2S | 1251 |
| 2.5 | α’-C2S | 1252 |
| 3.0 | α’-C2S | 1253 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xin, R.-F.; Du, Y.; Guo, X.-M. Effect of Alumina on Crystallization Behavior of Calcium Ferrite in Fe2O3-CaO-SiO2-Al2O3 System. Materials 2022, 15, 5257. https://doi.org/10.3390/ma15155257
Xin R-F, Du Y, Guo X-M. Effect of Alumina on Crystallization Behavior of Calcium Ferrite in Fe2O3-CaO-SiO2-Al2O3 System. Materials. 2022; 15(15):5257. https://doi.org/10.3390/ma15155257
Chicago/Turabian StyleXin, Rui-Feng, Yu Du, and Xing-Min Guo. 2022. "Effect of Alumina on Crystallization Behavior of Calcium Ferrite in Fe2O3-CaO-SiO2-Al2O3 System" Materials 15, no. 15: 5257. https://doi.org/10.3390/ma15155257
APA StyleXin, R.-F., Du, Y., & Guo, X.-M. (2022). Effect of Alumina on Crystallization Behavior of Calcium Ferrite in Fe2O3-CaO-SiO2-Al2O3 System. Materials, 15(15), 5257. https://doi.org/10.3390/ma15155257






