Biomaterials for Drugs Nose–Brain Transport: A New Therapeutic Approach for Neurological Diseases
Abstract
1. Introduction
2. Nasal Cavity: Anatomy and Physiology
2.1. Nose-To-Brain Delivery Pathways
2.1.1. The Olfactory Pathway
2.1.2. The Trigeminal Pathway
3. Nose-To-Brain Delivery: Limitations
4. Stimuli-Responsive Hydrogels
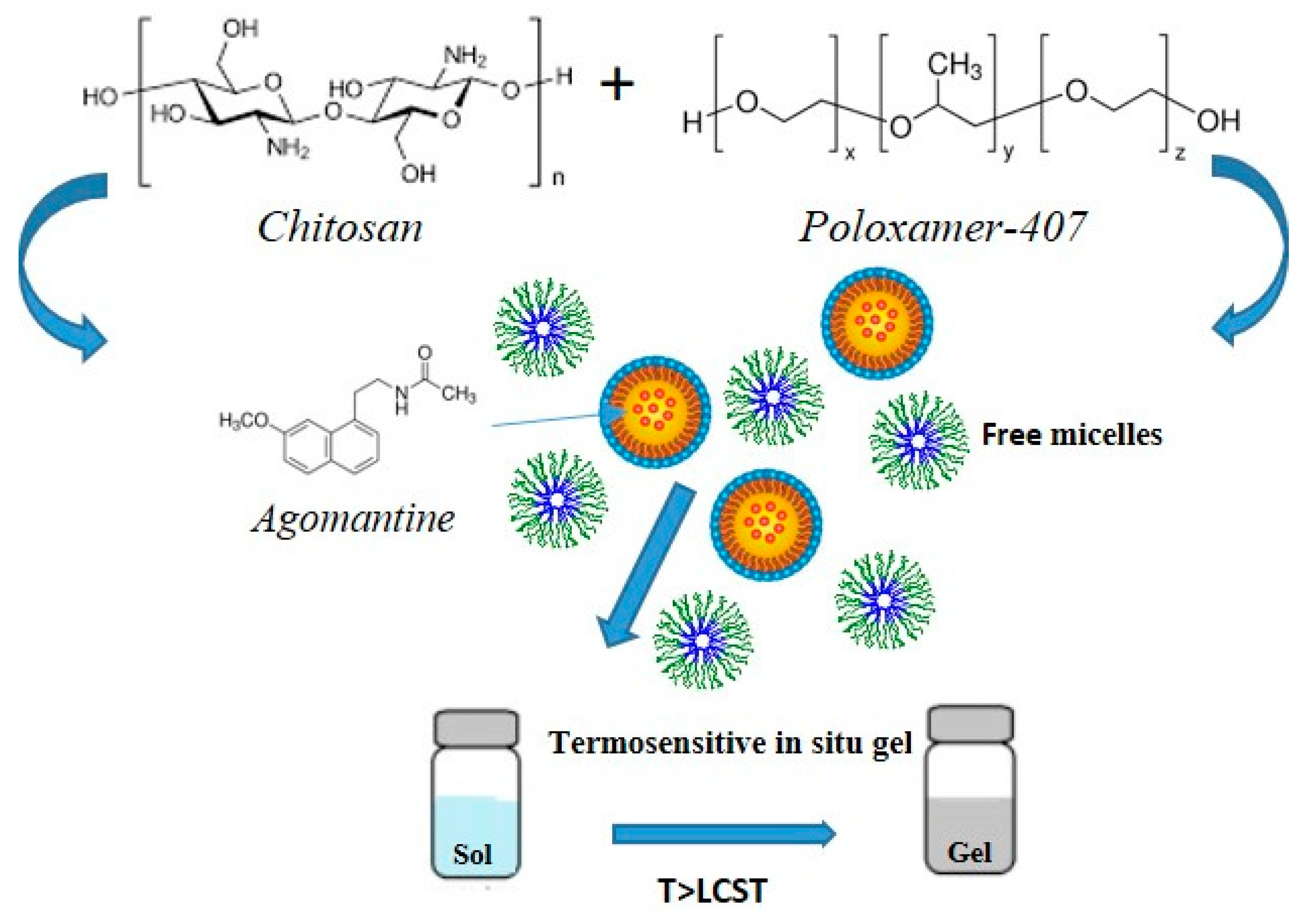
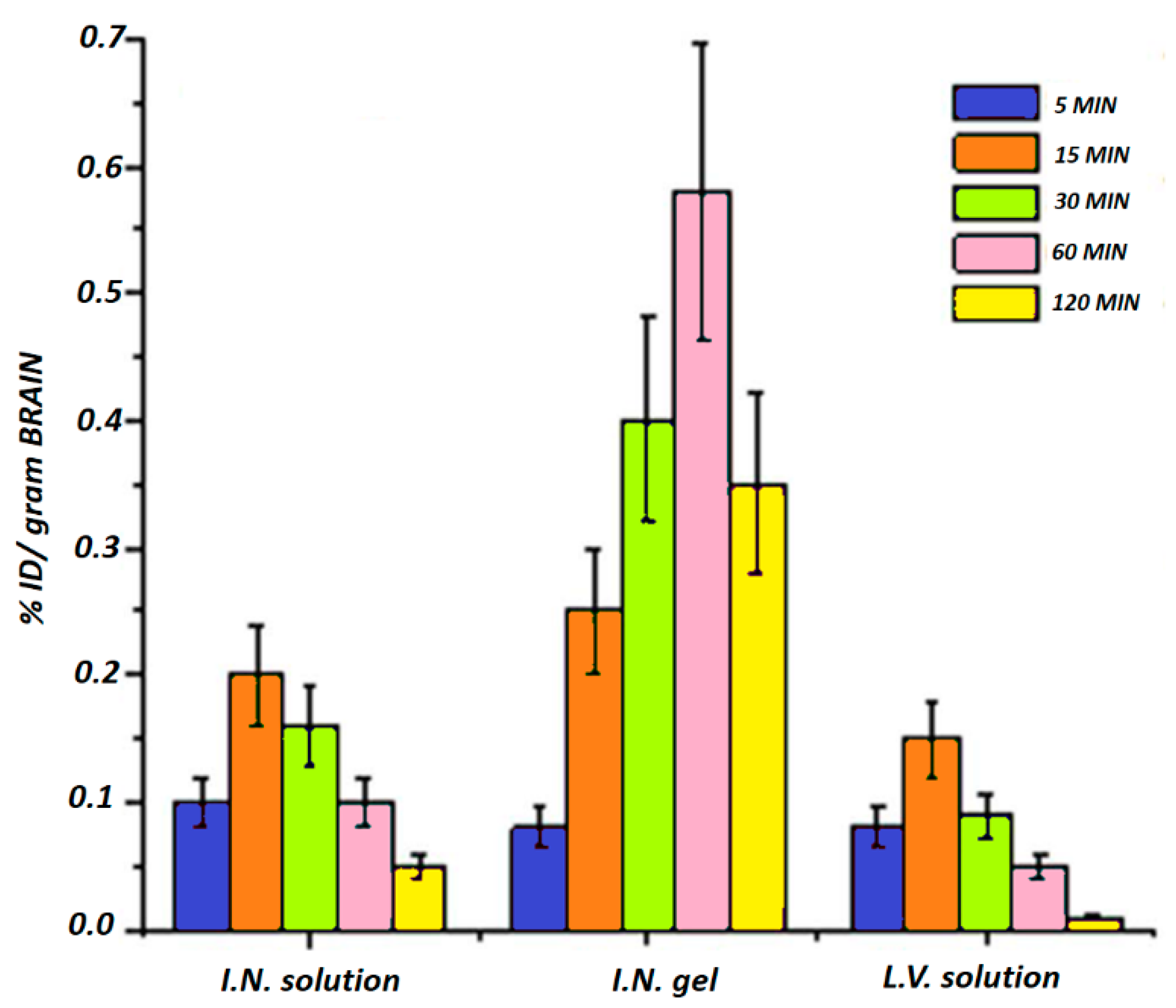
4.1. Stimuli-Responsive Hydrogel for the Treatment of Alzheimer’s Disease
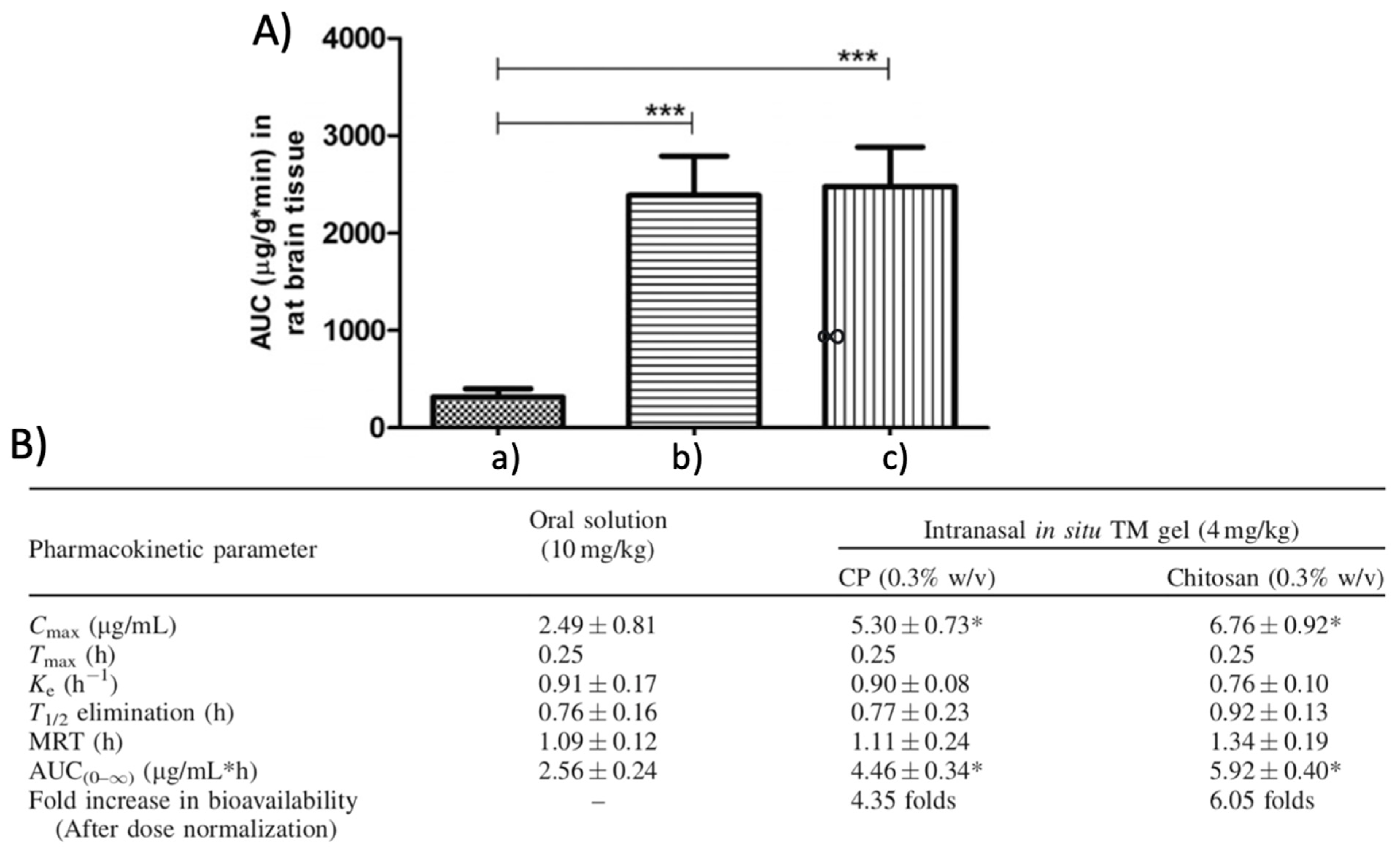
4.2. Stimuli-Responsive Hydrogels for the Treatment of Parkinson’s Disease
4.3. Stimuli-Responsive Hydrogels for the Treatment of Depression
4.4. Stimuli-Responsive Hydrogels for the Treatment of Schizophrenia
4.5. Stimuli-Responsive Hydrogels for the Treatment of Brain Injury
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Feigin, V.L.; Nichols, E.; Alam, T.; Bannick, M.S.; Beghi, E.; Blake, N.; Culpepper, W.J.; Dorsey, E.R.; Elbaz, A.; Ellenbogen, R.G.; et al. Global, regional, and national burden of neurological disorders, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019, 18, 459–480. [Google Scholar] [CrossRef]
- Feigin, V.L.; Vos, T.; Nichols, E.; Owolabi, M.O.; Carroll, W.M.; Dichgans, M.; Deuschl, G.; Parmar, P.; Brainin, M.; Murray, C. The global burden of neurological disorders: Translating evidence into policy. Lancet Neurol. 2020, 19, 255–265. [Google Scholar] [CrossRef]
- Carroll, W.M. The global burden of neurological disorders. Lancet Neurol. 2019, 18, 418–419. [Google Scholar] [CrossRef]
- Neurological Disorders, Public Health Challenges. Available online: https://www.who.int/mental_health/neurology/neurological_disorders_report_web.pdf (accessed on 31 December 2006.).
- Simonato, M.; Bennett, J.; Boulis, N.M.; Castro, M.G.; Fink, D.J.; Goins, W.F.; Glorioso, J.C. Progress in gene therapy for neurological disorders. Nat. Rev. Neurol. 2013, 9, 277–291. [Google Scholar] [CrossRef] [PubMed]
- Kanwar, J.R.; Sriramoju, B.; Kanwar, R.K. Neurological disorders and therapeutics targeted to surmount the blood–brain barrier. Int. J. Nanomed. 2012, 7, 3259–3278. [Google Scholar] [CrossRef]
- Agrawal, M.; Saraf, S.; Saraf, S.; Dubey, S.K.; Puri, A.; Gupta, U.; Alexander, A. Stimu-li-responsive In situ gelling system for nose-to-brain drug delivery. J. Control. Release 2020, 327, 235–265. [Google Scholar] [CrossRef] [PubMed]
- Bourganis, V.; Kammona, O.; Alexopoulos, A.; Kiparissides, C. Recent advances in carrier mediated nose-to-brain delivery of pharmaceutics. Eur. J. Pharm. Biopharm. 2018, 128, 337–362. [Google Scholar] [CrossRef]
- Aderibigbe, B.A. In situ-based gels for nose to brain delivery for the treatment of neurological dis-eases. Pharmaceutics 2018, 10, 40. [Google Scholar] [CrossRef]
- Kozlovskaya, L.; Abou-Kaoud, M.; Stepensky, D. Quantitative analysis of drug delivery to the brain via nasal route. J. Control. Release 2014, 189, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Pandit, R.; Chen, L.; Götz, J. The blood-brain barrier: Physiology and strategies for drug delivery. Adv. Drug Deliv. Rev. 2020, 165, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Obermeier, B.; Daneman, R.; Ransohoff, R.M. Development, maintenance and disruption of the blood-brain barrier. Nat. Med. 2013, 19, 1584–1596. [Google Scholar] [CrossRef]
- Abbott, N.J.; Rönnbäck, L.; Hansson, E. Astrocyte–endothelial interactions at the blood–brain bar-rier. Nat. Rev. Neurosci. 2006, 7, 41. [Google Scholar] [CrossRef] [PubMed]
- Banks, W.A. From blood–brain barrier to blood–brain interface: New opportunities for CNS drug de-livery. Nat. Rev. Drug Discov. 2016, 15, 275. [Google Scholar] [CrossRef]
- Banks, A.W. Characteristics of compounds that cross the blood-brain barrier. BMC Neurol. 2009, 9, 3. [Google Scholar] [CrossRef] [PubMed]
- Pardridge, W.M. The blood-brain barrier: Bottleneck in brain drug development. NeuroRx 2005, 2, 3–14. [Google Scholar] [CrossRef]
- Wang, Z.; Xiong, G.; Tsang, W.C.; Schätzlein, A.G.; Uchegbu, I.F. Nose-to-Brain Delivery. J. Pharm. Exp. 2019, 370, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Mittal, D.; Ali, A.; Shadab; Baboota, S.; Sahni, J.K.; Ali, J. Insights into direct nose to brain delivery: Current status and future perspective. Drug Deliv. 2013, 21, 75–86. [Google Scholar] [CrossRef] [PubMed]
- Pardeshi, C.V.; Belgamwar, V.S. Direct nose to brain drug delivery via integrated nerve pathways bypassing the blood–brain barrier: An excellent platform for brain targeting. Expert Opin. Drug Deliv. 2013, 10, 957–972. [Google Scholar] [CrossRef]
- Erdő, F.; Bors, L.A.; Farkas, D.; Bajza, Á.; Gizurarson, S. Evaluation of intranasal delivery route of drug administration for brain targeting. Brain Res. Bull. 2018, 143, 155–170. [Google Scholar] [CrossRef] [PubMed]
- Grassin-Delyle, S.; Buenestado, A.; Naline, E.; Faisy, C.; Blouquit-Laye, S.; Couderc, L.J.; Devillier, P. Intranasal drug delivery: An efficient and non-invasive route for systemic administration: Focus on opioids. Pharmachol. Ther. 2012, 134, 366–379. [Google Scholar] [CrossRef] [PubMed]
- Gizurarson, S. Anatomical and Histological Factors Affecting Intranasal Drug and Vaccine Delivery. Curr. Drug Deliv. 2012, 9, 566–582. [Google Scholar] [CrossRef] [PubMed]
- Gänger, S.; Schindowski, K. Tailoring Formulations for Intranasal Nose-to-Brain Delivery: A Review on Architecture, Physico-Chemical Characteristics and Mucociliary Clearance of the Nasal Olfactory Mucosa. Pharmaceutics 2018, 10, 116. [Google Scholar] [CrossRef] [PubMed]
- Crowe, T.P.; Greenlee, M.H.W.; Kanthasamy, A.G.; Hsu, W.H. Mechanism of intranasal drug de-livery directly to the brain. Life Sci. 2018, 195, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Watelet, J.B.; Cauwenberge, P.V. Applied anatomy and physiology of the nose and paranasal si-nuses. Allergy 1999, 54, 14–25. [Google Scholar] [CrossRef] [PubMed]
- Ugwoke, I.M.; Agu, R.U.; Verbeke, N.; Kinget, R. Nasal mucoadhesive drug delivery: Background, applications, trends and future perspectives. Adv. Drug Deliv. Rev. 2005, 57, 1640–1665. [Google Scholar] [CrossRef] [PubMed]
- Lochhead, J.J.; Thorne, R.G. Intranasal delivery of biologics to the central nervous system. Adv. Drug Deliv. Rev. 2012, 64, 614–628. [Google Scholar] [CrossRef]
- Arora, P.; Sharma, S.; Garg, S. Permeability issues in nasal drug delivery. Drug Discov. Today 2002, 7, 967–975. [Google Scholar] [CrossRef]
- Illum, L. Transport of drugs from the nasal cavity to the central nervous system. Eur. J. Pharm. Sci. 2000, 11, 1–18. [Google Scholar] [CrossRef]
- Veronesi, M.C.; Alhamami, M.; Miedema, S.B.; Yun, Y.; Ruiz-Cardozo, M.; Vannier, M.W. Imaging of intranasal drug delivery to the brain. Am. J. Nucl. Med. Mol. Imaging. 2020, 10, 1–31. [Google Scholar] [PubMed]
- Dhuria, S.V.; Hanson, L.R.; Frey II, W.H. Intranasal delivery to the central nervous system: Mechanisms and experimental considerations. J. Pharm. Sci. 2010, 99, 1654–1673. [Google Scholar] [CrossRef]
- Balin, B.J.; Broadwell, R.D.; Salcman, M.; El-Kalliny, M. Avenues for entry of peripherally admin-istered protein to the central nervous system in mouse, rat, and squirrel monkey. J. Comp. Neurol. 1986, 251, 260–280. [Google Scholar] [CrossRef] [PubMed]
- Altner, H.; Altner-Kolnberger, I. Freeze-fracture and tracer experiments on the permeability of the zonulae occludentes in the olfactory mucosa of vertebrates. Cell Tissue Res. 1974, 154, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Keller, L.A.; Merkel, O.; Popp, A. Intranasal drug delivery: Opportunities and toxicologic challenges during drug development. Drug Deliv. Transl. Res. 2021, 1, 1–23. [Google Scholar]
- Thorne, R.G.; Pronk, G.J.; Padmanabhan, V.; Frey Ii, W.H. Delivery of insulin-like growth factor-I to the rat brain and spinal cord along olfactory and trigeminal pathways following intranasal administra-tion. Neuroscience 2004, 127, 481–496. [Google Scholar] [CrossRef] [PubMed]
- Thorne, R.G.; Frey, W.H. Delivery of neurotrophic factors to the central nervous system. Clin. Pharmacokinet. 2001, 40, 907–946. [Google Scholar] [CrossRef]
- Luzzati, V.; Benoit, E.; Charpentier, G.; Vachette, P. X-ray Scattering Study of Pike Olfactory Nerve: Elastic, Thermodynamic and Physiological Properties of the Axonal Membrane. J. Mol. Biol. 2004, 343, 199–212. [Google Scholar] [CrossRef]
- Van Riel, D.; Leijten, L.M.; Verdijk, R.M.; Kessel, G.C.; van der Vries, E.; van Rossum, A.M.; Kuiken, T. Evidence for influenza virus CNS invasion along the olfactory route in an immunocom-promised infant. J. Infect. Dis. 2014, 210, 419–423. [Google Scholar] [CrossRef] [PubMed]
- Kristensson, K.; Olsson, Y. Uptake of exogenous proteins in mouse olfactory cells. Acta Neuropathol. 1971, 19, 145–154. [Google Scholar] [CrossRef]
- Broadwell, R.D.; Balin, B.J. Endocytic and exocytic pathways of the neuronal secretory process and trans synaptic transfer of wheat germ agglutinin-horseradish peroxidase in vivo. J. Comp. Neurol. 1985, 242, 632–650. [Google Scholar] [CrossRef] [PubMed]
- De Lorenzo, A.D. The olfactory neuron and the blood-brain barrier. In Ciba Foundation Symposium-Internal Secretions of the Pancreas (Colloquia on Endocrinology); John Wiley Sons, Ltd.: Chichester, UK, 1970; pp. 151–176. [Google Scholar]
- Haberly, L.B.; Price, J.L. The axonal projection patterns of the mitral and tufted cells of the olfac-tory bulb in the rat. Brain Res. 1977, 129, 152–157. [Google Scholar] [CrossRef]
- Illum, L. Is nose-to-brain transport of drugs in man a reality? J. Pharm. Pharm. 2010, 56, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Nonaka, N.; Farr, S.A.; Kageyama, H.; Shioda, S.; Banks, W.A. Delivery of Galanin-Like Peptide to the Brain: Targeting with Intranasal Delivery and Cyclodextrins. J. Pharm. Exp. 2008, 325, 513–519. [Google Scholar] [CrossRef] [PubMed]
- Charlton, S.T.; Whetstone, J.; Fayinka, S.T.; Read, K.D.; Illum, L.; Davis, S.S. Evaluation of Direct Transport Pathways of Glycine Receptor Antagonists and an Angiotensin Antagonist from the Nasal Cavity to the Central Nervous System in the Rat Model. Pharm. Res. 2008, 25, 1531–1543. [Google Scholar] [CrossRef] [PubMed]
- Hashizume, R.; Ozawa, T.; Gryaznov, S.M.; Bollen, A.W.; Lamborn, K.R.; Frey, I.W.H.; Deen, D.F. New therapeutic approach for brain tumors: Intranasal delivery of telomerase inhibitor GRN163. Neuro Oncol. 2008, 10, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Clerico, D.; To, W.; Lanza, D. Anatomy of the Human Nasal Passages. In Handbook of Olfaction and Gustation; Apple Academic Press: Palm Bay, FL, USA, 2003; pp. 1–16. [Google Scholar]
- Schaefer, M.L.; Bottger, B.; Silver, W.L.; Finger, T.E. Trigeminal collaterals in the nasal epithelium and olfactory bulb: A potential route for direct modulation of olfactory information by trigeminal stimuli. J. Comp. Neurol. 2002, 444, 221–226. [Google Scholar] [CrossRef] [PubMed]
- Johnson, N.J.; Hanson, L.R.; Frey, I.W.H. Trigeminal Pathways Deliver a Low Molecular Weight Drug from the Nose to the Brain and Orofacial Structures. Mol. Pharm. 2010, 7, 884–893. [Google Scholar] [CrossRef]
- Thorne, R.G.; Hanson, L.R.; Ross, T.M.; Tung, D.; Frey, W.H., II. Delivery of interferon-beta to the monkey nervous system following intranasal administration. Neuroscience 2008, 152, 785–797. [Google Scholar] [CrossRef]
- Hallschmid, M. Intranasal Insulin for Alzheimer’s Disease. CNS Drugs 2021, 35, 21–37. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.-P.; Liu, H.-J.; Cheng, S.-M.; Wang, Z.-L.; Cheng, X.; Yu, H.-X.; Liu, X.-F. Direct transport of VEGF from the nasal cavity to brain. Neurosci. Lett. 2009, 449, 108–111. [Google Scholar] [CrossRef]
- Kanazawa, T.; Kaneko, M.; Niide, T.; Akiyama, F.; Kakizaki, S.; Ibaraki, H.; Seta, Y. Enhancement of nose-to-brain delivery of hydrophilic macromolecules with stearate-or polyethylene glycol-modified argi-nine-rich peptide. Int. J. Pharm. 2017, 530, 195–200. [Google Scholar] [CrossRef]
- Chung, E.P.; Cotter, J.D.; Prakapenka, A.V.; Cook, R.L.; DiPerna, D.M.; Sirianni, R.W. Targeting small molecule delivery to the brain and spinal cord via intranasal administration of rabies virus glycopro-tein (RVG29)-modified PLGA nanoparticles. Pharmaceutics 2020, 12, 93. [Google Scholar] [CrossRef]
- Wu, H.; Hu, K.; Jiang, X. From nose to brain: Understanding transport capacity and transport rate of drugs. Expert Opin. Drug Deliv. 2008, 5, 1159–1168. [Google Scholar] [CrossRef] [PubMed]
- Kaur, P.; Garg, T.; Rath, G.; Goyal, A.K. In situ nasal gel drug delivery: A novel approach for brain targeting through the mucosal membrane. Artif. Cellsnanomed. Biotechnol. 2015, 44, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Bustamante-Marin, X.M.; Ostrowski, L.E. Cilia and Mucociliary Clearance. Cold Spring Harb. Perspect. Biol. 2017, 9, a028241. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, M.; Kumar, M.; Pathak, K. A review on mucoadhesive polymer used in nasal drug de-livery system. J. Adv. Pharm. Technol. Res. 2011, 2, 215. [Google Scholar] [CrossRef] [PubMed]
- Illum, L. Nasal drug delivery: New developments and strategies. Drug Discov. Today 2002, 7, 1184–1189. [Google Scholar] [CrossRef]
- Karmakar, A.; Zhang, Q.; Zhang, Y. Neurotoxicity of nanoscale materials. J. Food Drug Anal. 2014, 22, 147–160. [Google Scholar] [CrossRef] [PubMed]
- Davis, S.S.; Illum, L. Absorption Enhancers for Nasal Drug Delivery. Clin. Pharm. 2003, 42, 1107–1128. [Google Scholar] [CrossRef] [PubMed]
- Gomez, D.; Martinez, J.A.; Hanson, L.R.; Frey, W.H., 2nd; Toth, C.C. Intranasal treatment of neu-rodegenerative diseases and stroke. Front. Biosci. 2012, 4, 74–89. [Google Scholar] [CrossRef]
- Hoffman, A.S. Hydrogels for biomedical applications. Adv. Drug Deliv. Rev. 2012, 64, 18–23. [Google Scholar] [CrossRef]
- Trombino, S.; Servidio, C.; Curcio, F.; Cassano, R. Strategies for hyaluronic acid-based hydrogel de-sign in drug delivery. Pharmaceutics 2019, 11, 407. [Google Scholar] [CrossRef]
- Karavasili, C.; Fatouros, D.G. Smart materials: In situ gel-forming systems for nasal delivery. Drug Discov. Today 2016, 21, 157–166. [Google Scholar] [CrossRef]
- Singh, R.M.; Kumar, A.; Pathak, K. Mucoadhesive in situ nasal gelling drug delivery systems for modulated drug delivery. Expert Opin. Drug Deliv. 2013, 10, 115–130. [Google Scholar] [CrossRef] [PubMed]
- Narayanaswamy, R.; Torchilin, V.P. Hydrogels and Their Applications in Targeted Drug Delivery. Molecules 2019, 24, 603. [Google Scholar] [CrossRef]
- Echeverria, C.; Fernandes, S.N.; Godinho, M.H.; Borges, J.P.; Soares, P.I.P. Functional Stimuli-Responsive Gels: Hydrogels and Microgels. Gels 2018, 4, 54. [Google Scholar] [CrossRef]
- Giuliano, E.; Paolino, D.; Fresta, M.; Cosco, D. Mucosal applications of poloxamer 407-based hy-drogels: An overview. Pharmaceutics 2018, 10, 159. [Google Scholar] [CrossRef]
- Abdeltawab, H.; Svirskis, D.; Sharma, M. Formulation strategies to modulate drug release from poloxamer based in situ gelling systems. Expert Opin. Drug Deliv. 2020, 17, 495–509. [Google Scholar] [CrossRef]
- Balakrishnan, P.; Park, E.-K.; Song, C.-K.; Ko, H.-J.; Hahn, T.-W.; Song, K.-W.; Cho, H.-J. Carbopol-Incorporated Thermoreversible Gel for Intranasal Drug Delivery. Molecules 2015, 20, 4124–4135. [Google Scholar] [CrossRef] [PubMed]
- Ur-Rehman, T.; Tavelin, S.; Gröbner, G. Chitosan in situ gelation for improved drug loading and retention in poloxamer 407 gels. Int. J. Pharm. 2011, 409, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Gull, A.; Aqil, M.; Ansari, M.D.; Sultana, Y. Poloxamer-407 thickened lipid colloidal system of agomelatine for brain targeting: Characterization, brain pharmacokinetic study and behavioral study on Wistar rats. Colloids Surf. B Biointerfaces 2019, 181, 426–436. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Zuo, Z.; Cheung, C.K.C.; Leung, S.S.Y. Updates on thermosensitive hydrogel for nasal, ocular and cutaneous delivery. Int. J. Pharm. 2019, 559, 86–101. [Google Scholar] [CrossRef]
- Dalvi, A.V.; Ravi, P.R.; Uppuluri, C.T.; Mahajan, R.R.; Katke, S.V.; Deshpande, V.S. Thermosensi-tive nasal in situ gelling systems of rufinamide formulated using modified tamarind seed xyloglucan for di-rect nose-to-brain delivery: Design, physical characterization, and in vivo evaluation. J. Pharm. Investig. 2021, 51, 1–13. [Google Scholar] [CrossRef]
- Wang, X.; Liu, G.; Ma, J.; Guo, S.; Gao, L.; Jia, Y.; Zhang, Q. In situ gel-forming system: An attrac-tive alternative for nasal drug delivery. Crit. Rev. Ther. Drug Carr. Syst. 2013, 30, 411–434. [Google Scholar] [CrossRef] [PubMed]
- Sherje, A.P.; Londhe, V. Development and evaluation of pH-responsive cyclodextrin-based in situ gel of paliperidone for intranasal delivery. AAPS Pharmscitech 2018, 19, 384–394. [Google Scholar] [CrossRef] [PubMed]
- Grasdalen, H.; Smidsrød, O. Gelation of gellan gum. Carbohydr. Polym. 1987, 7, 371–393. [Google Scholar] [CrossRef]
- Jelkmann, M.; Leichner, C.; Zaichik, S.; Laffleur, F.; Bernkop-Schnürch, A. A gellan gum derivative as in-situ gelling cationic polymer for nasal drug delivery. Int. J. Biol. Macromol. 2020, 158, 1037–1046. [Google Scholar] [CrossRef] [PubMed]
- Long, J.M.; Holtzman, D.M. Alzheimer disease: An update on pathobiology and treatment strate-gies. Cell 2019, 179, 312–339. [Google Scholar] [CrossRef] [PubMed]
- Van der Kant, R.; Goldstein, L.S.; Ossenkoppele, R. Amyloid-β-independent regulators of tau pa-thology in Alzheimer disease. Nat. Rev. Neurosci. 2020, 21, 21–35. [Google Scholar] [CrossRef]
- Leng, F.; Edison, P. Neuroinflammation and microglial activation in Alzheimer disease: Where do we go from here? Nat. Rev. Neurol. 2021, 17, 157–172. [Google Scholar] [CrossRef]
- Abouhussein, D.M.; Khattab, A.; Bayoumi, N.A.; Mahmoud, A.F.; Sakr, T.M. Brain targeted ri-vastigmine mucoadhesive thermosensitive in situ gel: Optimization, in vitro evaluation, radiolabeling, in vivo pharmacokinetics and biodistribution. J. Drug Deliv. Sci. Technol. 2018, 43, 129–140. [Google Scholar] [CrossRef]
- Zhang, L.; Yang, S.; Wong, L.R.; Xie, H.; Ho, P.C.-L. In Vitro and In Vivo Comparison of Curcumin-Encapsulated Chitosan-Coated Poly(lactic-co-glycolic acid) Nanoparticles and Curcumin/Hydroxypropyl-β-Cyclodextrin Inclusion Complexes Administered Intranasally as Therapeutic Strategies for Alzheimer’s Disease. Mol. Pharm. 2020, 17, 4256–4269. [Google Scholar] [CrossRef]
- Vingtdeux, V.; Dreses-Werringloer, U.; Zhao, H.; Davies, P.; Marambaud, P. Therapeutic potential of resveratrol in Alzheimer’s disease. BMC Neurosci. 2008, 9, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Son, I.H.; Park, Y.H.; Lee, S.I.; Yang, H.D.; Moon, H.-I. Neuroprotective Activity of Triterpenoid Saponins from Platycodi radix Against Glutamate-induced Toxicity in Primary Cultured Rat Cortical Cells. Molecules 2007, 12, 1147–1152. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Li, R.; Zhu, S.; Ma, J.; Pang, L.; Ma, B.; Du, L.; Jin, Y. Nasal timosaponin BII dually sensitive in situ hydrogels for the prevention of Alzheimer’s disease induced by lipopolysaccharides. Int. J. Pharm. 2020, 578, 119115. [Google Scholar] [CrossRef]
- Poewe, W.; Seppi, K.; Tanner, C.M.; Halliday, G.M.; Brundin, P.; Volkmann, J.; Lang, A.E. Parkinson disease. Nat. Rev. Dis. Primers 2017, 3, 1–21. [Google Scholar] [CrossRef]
- Armstrong, M.J.; Okun, M.S. Diagnosis and treatment of Parkinson disease: A review. JAMA 2020, 323, 548–560. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, M.J.; Okun, M.S. Choosing a Parkinson Disease Treatment. JAMA 2020, 323, 1420. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffer, R. Gastrointestinal Dysfunction in Parkinson’s Disease. Parkinson’s Dis. 2004, 2, 107–116. [Google Scholar] [CrossRef]
- Guay, D.R. Rasagiline (TVP-1012): A new selective monoamine oxidase inhibitor for Parkinson’s disease. Am. J. Geriatr. Pharmacother. 2006, 4, 330–346. [Google Scholar] [CrossRef] [PubMed]
- Ravi, P.R.; Aditya, N.; Patil, S.; Cherian, L. Nasal in-situ gels for delivery of rasagiline mesylate: Improvement in bioavailability and brain localization. Drug Deliv. 2015, 22, 903–910. [Google Scholar] [CrossRef] [PubMed]
- Waters, C. The Development of the Rotigotine Transdermal Patch: A Historical Perspective. Neurol. Clin. 2013, 31, S37–S50. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Yang, Z.; Liu, M.; Tao, Y.; Li, Z.; Wu, Z.; Gui, S. Facile nose-to-brain delivery of rotigo-tine-loaded polymer micelles thermosensitive hydrogels: In vitro characterization and in vivo behavior study. Int. J. Pharm. 2020, 577, 119046. [Google Scholar] [CrossRef] [PubMed]
- Otte, C.; Gold, S.M.; Penninx, B.W.; Pariante, C.M.; Etkin, A.; Fava, M.; Schatzberg, A.F. Major depressive disorder. Nat. Rev. Dis. Primers 2016, 2, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Kok, R.M.; Reynolds, C.F. Management of depression in older adults: A review. JAMA 2017, 317, 2114–2122. [Google Scholar] [CrossRef]
- Verduijn, J.; Milaneschi, Y.; Schoevers, R.A.; Van Hemert, A.M.; Beekman, A.T.F.; Penninx, B.W.J.H. Pathophysiology of major depressive disorder: Mechanisms involved in etiology are not associated with clinical progression. Transl. Psychiatry 2015, 5, e649. [Google Scholar] [CrossRef] [PubMed]
- Kupfer, D.J. The pharmacological management of depression. Dialog Clin. Neurosci. 2005, 7, 191–205. [Google Scholar]
- Clevenger, S.S.; Malhotra, D.; Dang, J.; VanLe, B.; Ishak, W.W. The role of selective serotonin reuptake inhibitors in preventing relapse of major depressive disorder. Adv. Psychopharmacol. 2018, 8, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Thakkar, H.; Vaghela, D.; Patel, B.P. Brain targeted intranasal in-situ gelling spray of paroxe-tine: Formulation, characterization and in-vivo evaluation. J. Drug Deliv. Sci. Technol. 2021, 62, 102317. [Google Scholar] [CrossRef]
- Bhandwalkar, M.J.; Avachat, A.M. Thermoreversible nasal in situ gel of venlafaxine hydro-chloride: Formulation, characterization, and pharmacodynamic evaluation. AAPS Pharmscitech 2013, 14, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Marder, S.R.; Cannon, T.D. Schizophrenia. N. Engl. J. Med. 2019, 381, 1753–1761. [Google Scholar] [CrossRef] [PubMed]
- Kahn, R.S.; Sommer, I.E.; Murray, R.M.; Meyer-Lindenberg, A.; Weinberger, D.R.; Cannon, T.D.; Insel, T.R. Schizophrenia. Nat. Rev. Dis. Primers 2016, 1, 15067. [Google Scholar] [CrossRef] [PubMed]
- Owen, M.J.; Sawa, A.; Mortensen B., P. Schizophrenia. Lancet 2016, 388, 86–97. [Google Scholar] [CrossRef]
- Majcher, M.J.; Babar, A.; Lofts, A.; Leung, A.; Li, X.; Abu-Hijleh, F.; Hoare, T. In situ-gelling starch nanoparticle (SNP)/O-carboxymethyl chitosan (CMCh) nanoparticle network hydrogels for the in-tranasal delivery of an antipsychotic peptide. J. Control. Release 2021, 330, 738–752. [Google Scholar] [CrossRef] [PubMed]
- Kuriakose, D.; Xiao, Z. Pathophysiology and Treatment of Stroke: Present Status and Future Perspectives. Int. J. Mol. Sci. 2020, 21, 7609. [Google Scholar] [CrossRef] [PubMed]
- Xie, H.; Li, L.; Sun, Y.; Wang, Y.; Gao, S.; Tian, Y.; Ma, X.; Guo, C.; Bo, F.; Zhang, L. An Available Strategy for Nasal Brain Transport of Nanocomposite Based on PAMAM Dendrimers via In Situ Gel. Nanomaterials 2019, 9, 147. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cassano, R.; Servidio, C.; Trombino, S. Biomaterials for Drugs Nose–Brain Transport: A New Therapeutic Approach for Neurological Diseases. Materials 2021, 14, 1802. https://doi.org/10.3390/ma14071802
Cassano R, Servidio C, Trombino S. Biomaterials for Drugs Nose–Brain Transport: A New Therapeutic Approach for Neurological Diseases. Materials. 2021; 14(7):1802. https://doi.org/10.3390/ma14071802
Chicago/Turabian StyleCassano, Roberta, Camilla Servidio, and Sonia Trombino. 2021. "Biomaterials for Drugs Nose–Brain Transport: A New Therapeutic Approach for Neurological Diseases" Materials 14, no. 7: 1802. https://doi.org/10.3390/ma14071802
APA StyleCassano, R., Servidio, C., & Trombino, S. (2021). Biomaterials for Drugs Nose–Brain Transport: A New Therapeutic Approach for Neurological Diseases. Materials, 14(7), 1802. https://doi.org/10.3390/ma14071802







