Allogeneic Dentin Graft: A Review on Its Osteoinductivity and Antigenicity
Abstract
1. Introduction
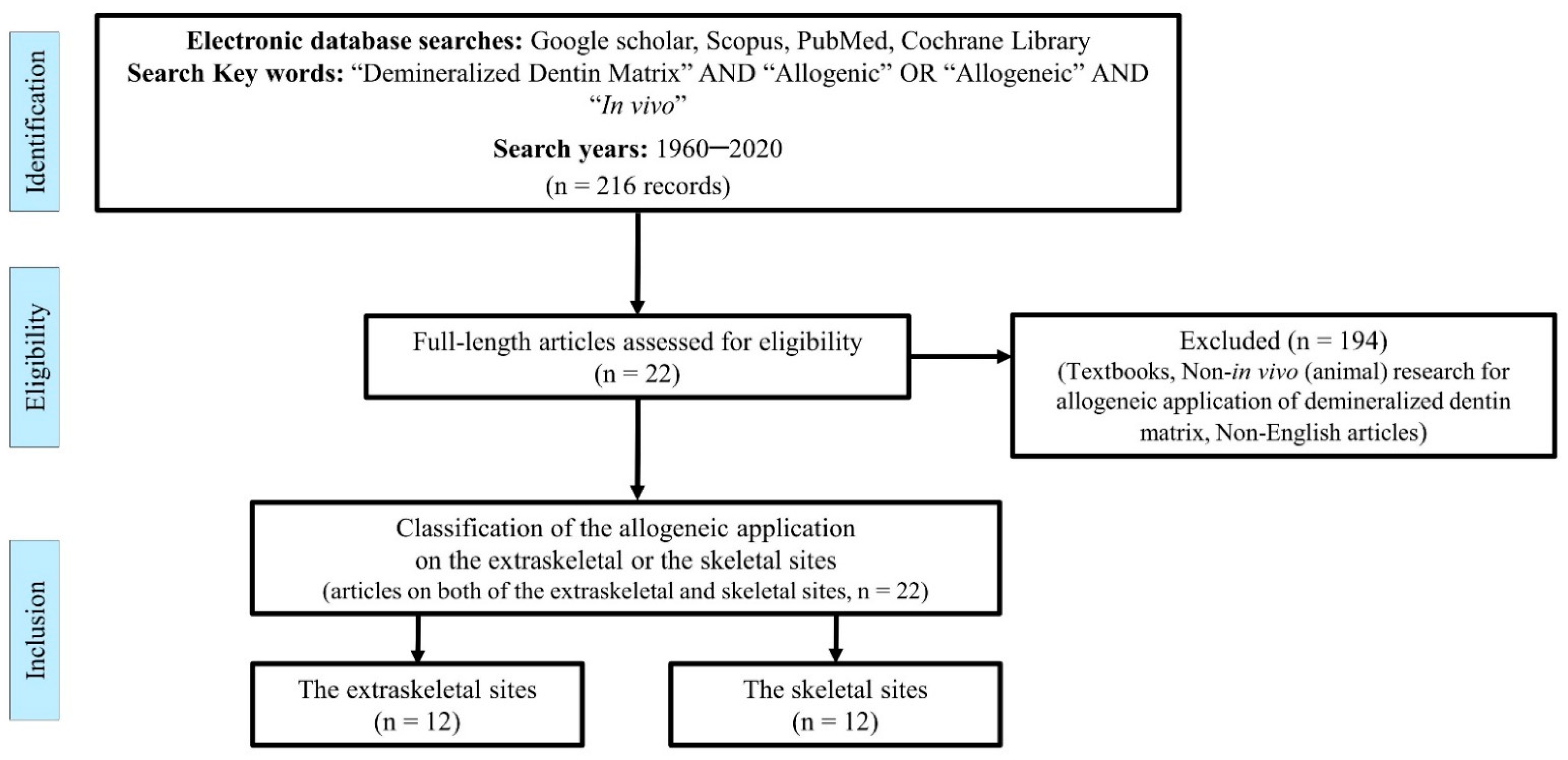
2. Methods
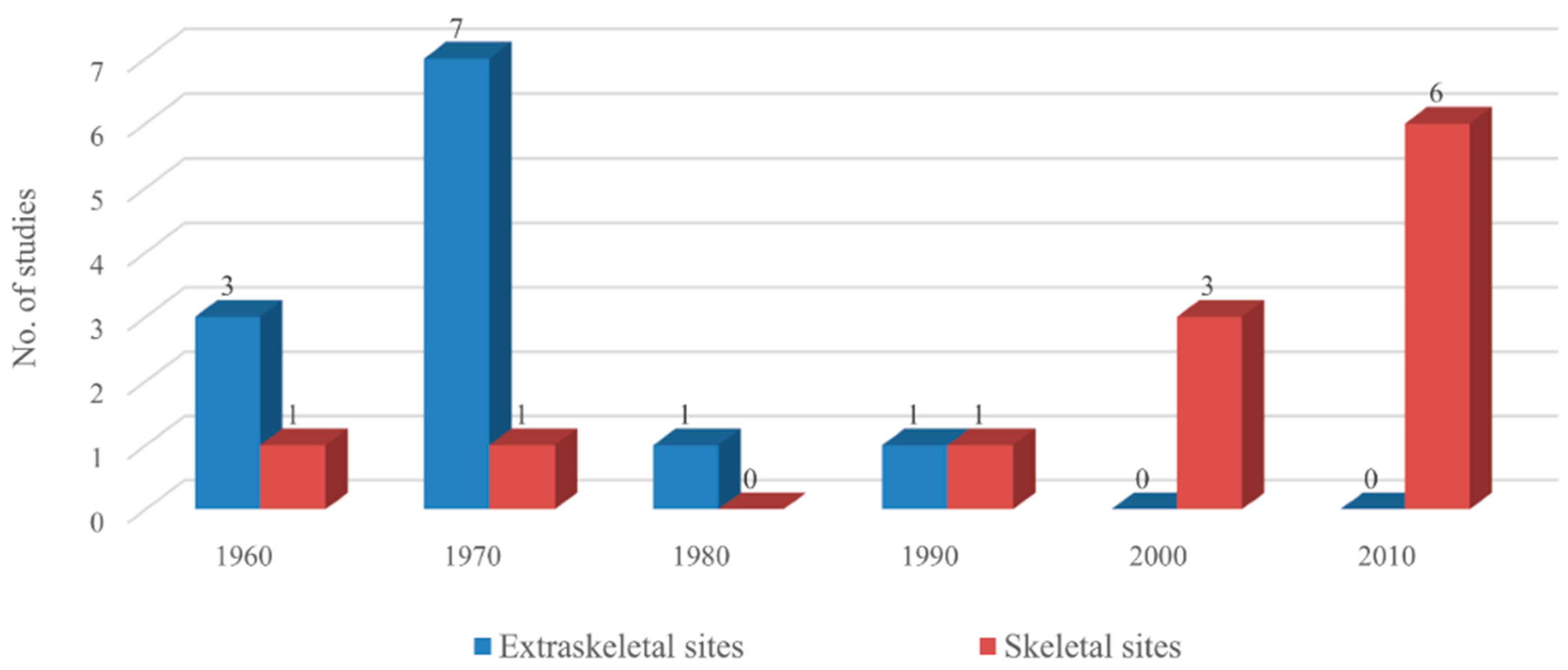
3. Results
4. Discussion
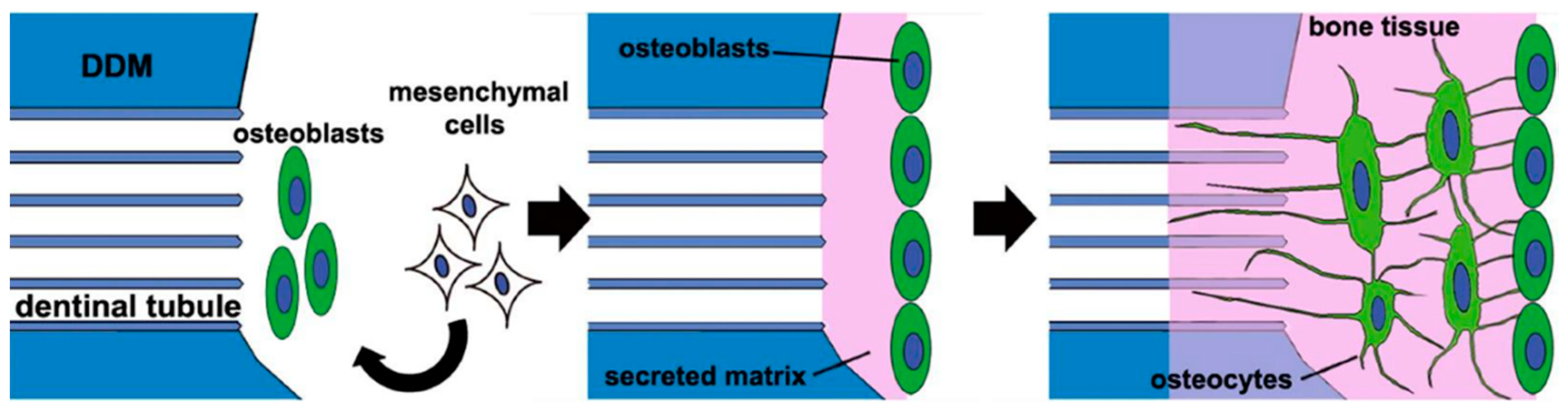
4.1. Osteoinductivity
4.2. Antigenicity
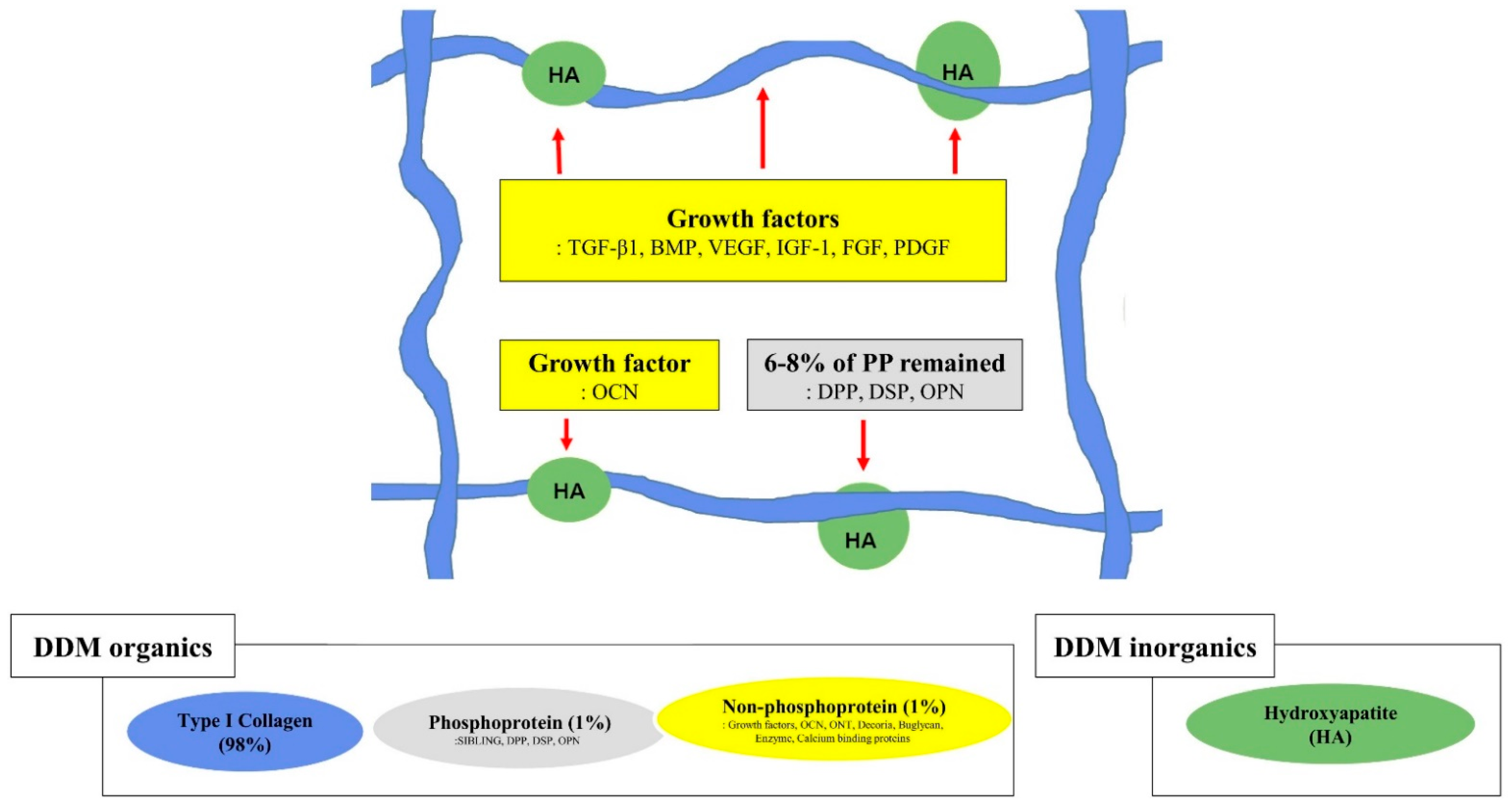
4.3. Demineralization of Dentin Matrix
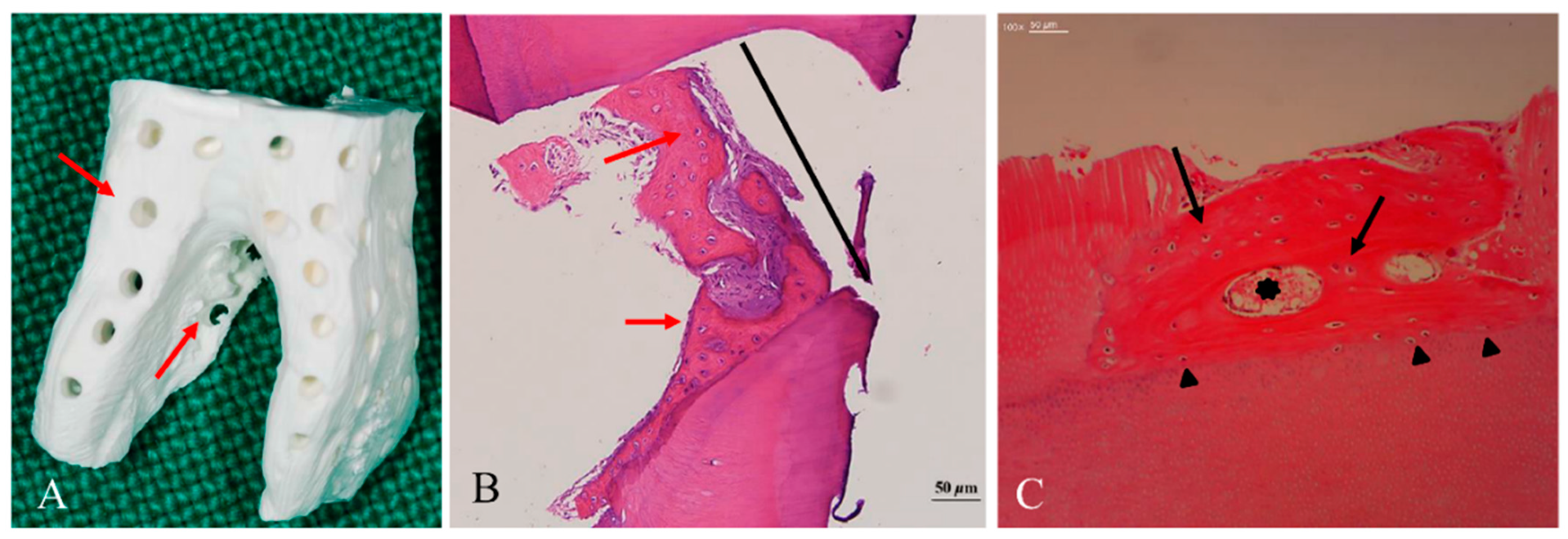
4.4. Geometry of Allo-DDM
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Avery, S.J.; Sadaghiani, L.; Sloan, A.J.; Waddington, R.J. Analysing the bioactive makeup of demineralised dentine matrix on bone marrow mesenchymal stem cells for enhanced bone repair. Eur. Cell Mater. 2017, 34, 1–14. [Google Scholar] [CrossRef]
- Urist, M.R.; Silverman, B.F.; Büring, K.; Dubuc, F.L.; Rosenberg, J.M. The bone induction principle. Clin. Orthop. Relat. Res. 1967, 53, 243–283. [Google Scholar] [CrossRef]
- Murata, M.; Okubo, N.; Shakya, M.; Kabir, M.; Yokozeki, K.; Zhu, B.; Ishikawa, M.; Kitamura, R.; Akazawa, T. Dentin Materials as Biological Scaffolds for Tissue Engineering. In Biomaterial-Supported Tissue Reconstruction or Regeneration; IntechOpen: London, UK, 2019; pp. 1–12. [Google Scholar] [CrossRef]
- Kim, Y.-K.; Um, I.-W.; Murata, M. Tooth Bank System for Bone Regeneration—Safety Report. J. Hard Tissue Biol. 2014, 23, 371–376. [Google Scholar] [CrossRef][Green Version]
- Murata, M. Bone Engineering Using Human Demineralized Dentin Matrix and Recombinant Human BMP-2. J. Hard Tissue Biol. 2005, 14, 80–81. [Google Scholar] [CrossRef]
- Kim, Y.-K.; Um, I.-W.; An, H.-J.; Kim, K.-W.; Hong, K.-S.; Murata, M. Effects of Demineralized Dentin Matrix Used as an rhBMP-2 Carrier for Bone Regeneration. J. Hard Tissue Biol. 2014, 23, 415–422. [Google Scholar] [CrossRef]
- Butler, W.T.; Mikulski, A.; Urist, M.R.; Bridges, G.; Uyeno, S. Noncollagenous proteins of a rat dentin matrix possessing bone morphogenetic activity. J. Dent. Res. 1977, 56, 228–232. [Google Scholar] [CrossRef]
- Kawai, T.; Urist, M.R. Bovine tooth-derived bone morphogenetic protein. J. Dent. Res. 1989, 68, 1069–1074. [Google Scholar] [CrossRef]
- Bessho, K.; Tagawa, T.; Murata, M. Purification of rabbit bone morphogenetic protein derived from bone, dentin, and wound tissue after tooth extraction. J. Oral. Maxillofac. Surg. 1990, 48, 162–169. [Google Scholar] [CrossRef]
- Bessho, K.; Tanaka, N.; Matsumoto, J.; Tagawa, T.; Murata, M. Human dentin-matrix-derived bone morphogenetic protein. J. Dent. Res. 1991, 70, 171–175. [Google Scholar] [CrossRef] [PubMed]
- Murata, M. Autogenous demineralized dentin matrix for maxillary sinus augmentation in humans: The first clinical report. J. Dent. Res. 2003, 82, B243. [Google Scholar]
- Gomes, M.F.; Abreu, P.P.; Morosolli, A.R.; Araújo, M.M.; Goulart, M. Densitometric analysis of the autogenous demineralized dentin matrix on the dental socket wound healing process in humans. Braz. Oral. Res. 2006, 20, 324–330. [Google Scholar] [CrossRef]
- Kim, Y.K.; Kim, S.G.; Byeon, J.H.; Lee, H.J.; Um, I.U.; Lim, S.C.; Kim, S.Y. Development of a novel bone grafting material using autogenous teeth. Oral. Surg. Oral. Med. Oral. Pathol. Oral. Radiol. Endod. 2010, 109, 496–503. [Google Scholar] [CrossRef]
- Minetti, E.; Giacometti, E.; Gambardella, U.; Contessi, M.; Ballini, A.; Marenzi, G.; Celko, M.; Mastrangelo, F. Alveolar Socket Preservation with Different Autologous Graft Materials: Preliminary Results of a Multicenter Pilot Study in Human. Materials 2020, 13, 1153. [Google Scholar] [CrossRef]
- Li, P.; Zhu, H.; Huang, D. Autogenous DDM versus Bio-Oss granules in GBR for immediate implantation in periodontal postextraction sites: A prospective clinical study. Clin. Implant. Dent. Relat. Res. 2018, 20, 923–928. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.K.; Lee, J.H.; Um, I.W.; Cho, W.J. Guided Bone Regeneration Using Demineralized Dentin Matrix: Long-Term Follow-Up. J. Oral. Maxillofac. Surg. 2016, 74, 515.e1–515.e9. [Google Scholar] [CrossRef] [PubMed]
- Minetti, E.; Palermo, A.; Contessi, M.; Gambardella, U.; Schmitz, J.; Giacometti, E.; Celko, M.; Trisi, P. Autologous tooth graft for maxillary sinus augmentation: A multicenter clinical study. Int. J. Growth Factors Stem. Cells Dent. 2019, 2, 45–51. [Google Scholar] [CrossRef]
- Del Canto-Díaz, A.; de Elío-Oliveros, J.; Del Canto-Díaz, M.; Alobera-Gracia, M.A.; Del Canto-Pingarrón, M.; Martínez-González, J.M. Use of autologous tooth-derived graft material in the post-extraction dental socket. Pilot study. Med. Oral. Patol. Oral. Y Cir. Bucal 2019, 24, e53–e60. [Google Scholar] [CrossRef] [PubMed]
- Tanoue, R.; Ohta, K.; Miyazono, Y.; Iwanaga, J.; Koba, A.; Natori, T.; Iwamoto, O.; Nakamura, K.I.; Kusukawa, J. Three-dimensional ultrastructural analysis of the interface between an implanted demineralised dentin matrix and the surrounding newly formed bone. Sci. Rep. 2018, 8, 2858. [Google Scholar] [CrossRef] [PubMed]
- Urist, M.R. Bone: Formation by autoinduction. Science 1965, 150, 893–899. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Powers, R.M., Jr.; Wolfinbarger, L., Jr. Effect(s) of the demineralization process on the osteoinductivity of demineralized bone matrix. J. Periodontol. 1997, 68, 1085–1092. [Google Scholar] [CrossRef]
- Glowacki, J. A review of osteoinductive testing methods and sterilization processes for demineralized bone. Cell Tissue Bank 2005, 6, 3–12. [Google Scholar] [CrossRef]
- Bang, G.; Urist, M.R. Bone induction in excavation chambers in matrix of decalcified dentin. Arch. Surg. 1967, 94, 781–789. [Google Scholar] [CrossRef]
- Yeomans, J.D.; Urist, M.R. Bone induction by decalcified dentine implanted into oral, osseous and muscle tissues. Arch. Oral. Biol. 1967, 12, 999–1008. [Google Scholar] [CrossRef]
- Urist, M.R.; Dowell, T.A.; Hay, P.H.; Strates, B.S. Inductive substrates for bone formation. Clin. Orthop. Relat. Res. 1968, 59, 59–96. [Google Scholar] [CrossRef] [PubMed]
- Huggins, C.B.; Urist, M.R. Dentin matrix transformation: Rapid induction of alkaline phosphatase and cartilage. Science 1970, 167, 896–898. [Google Scholar] [CrossRef] [PubMed]
- Huggins, C.; Wiseman, S.; Reddi, A.H. Transformation of fibroblasts by allogeneic and xenogeneic transplants of demineralized tooth and bone. J. Exp. Med. 1970, 132, 1250–1258. [Google Scholar] [CrossRef]
- Bang, G. Induction of heterotopic bone formation by demineralized dentin in guinea pigs: Antigenicity of the dentin matrix. J. Oral. Pathol. 1972, 1, 172–185. [Google Scholar] [CrossRef] [PubMed]
- Reddi, A.H.; Huggins, C.B. Influence of geometry of transplanted tooth and bone on transformation of fibroblasts. Proc. Soc. Exp. Biol. Med. 1973, 143, 634–637. [Google Scholar] [CrossRef]
- Bang, G. Induction of heterotopic bone formation by demineralized dentin: An experimental model in guinea pigs. Scand. J. Dent. Res. 1973, 81, 240–250. [Google Scholar] [CrossRef] [PubMed]
- Linden, G.J. Bone induction in implants of decalcified bone and dentine. J. Anat. 1975, 119, 359–367. [Google Scholar]
- Nilsen, R. Electron microscopy of induced heterotopic bone formation in guinea pigs. Arch. Oral. Biol. 1977, 22, 485–493. [Google Scholar] [CrossRef]
- Inoue, T.; Deporter, D.A.; Melcher, A.H. Induction of chondrogenesis in muscle, skin, bone marrow, and periodontal ligament by demineralized dentin and bone matrix in vivo and in vitro. J. Dent. Res. 1986, 65, 12–22. [Google Scholar] [CrossRef] [PubMed]
- Pinholt, E.M.; Bang, G.; Haanaes, H.R. Alveolar ridge augmentation by osteoinduction in rats. Scand. J. Dent. Res. 1990, 98, 434–441. [Google Scholar] [CrossRef] [PubMed]
- Bang, G.; Nordenram, Å.; Anneroth, G. Allogenic demineralized dentin implants in jaw defects of Java monkeys. Int. J. Oral. Surg. 1972, 1, 126–136. [Google Scholar] [CrossRef]
- Carvalho, V.A.; Tosello Dde, O.; Salgado, M.A.; Gomes, M.F. Histomorphometric analysis of homogenous demineralized dentin matrix as osteopromotive material in rabbit mandibles. Int. J. Oral. Maxillofac. Implant. 2004, 19, 679–686. [Google Scholar]
- Gomes, M.F.; Banzi, E.C.; Destro, M.F.; Lavinicki, V.; Goulart, M. Homogenous demineralized dentin matrix for application in cranioplasty of rabbits with alloxan-induced diabetes: Histomorphometric analysis. Int. J. Oral. Maxillofac. Implant. 2007, 22, 939–947. [Google Scholar]
- Gomes, M.F.; Destro, M.F.; Banzi, E.C.; Vieira, E.M.; Morosolli, A.R.; Goulart, M. Optical density of bone repair after implantation of homogenous demineralized dentin matrix in diabetic rabbits. Braz. Oral. Res. 2008, 22, 275–280. [Google Scholar] [CrossRef]
- Bormann, K.H.; Suarez-Cunqueiro, M.M.; Sinikovic, B.; Kampmann, A.; von See, C.; Tavassol, F.; Binger, T.; Winkler, M.; Gellrich, N.C.; Rücker, M. Dentin as a suitable bone substitute comparable to ß-TCP—An experimental study in mice. Microvasc. Res. 2012, 84, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Bakhshalian, N.; Hooshmand, S.; Campbell, S.C.; Kim, J.S.; Brummel-Smith, K.; Arjmandi, B.H. Biocompatibility and microstructural analysis of osteopromotive property of allogenic demineralized dentin matrix. Int. J. Oral. Maxillofac. Implant. 2013, 28, 1655–1662. [Google Scholar] [CrossRef] [PubMed]
- Bakhshalian, N.; Jalayer, T.; Shahoon, H.; Arjmandi, B.H.; Azimi, H.R. Osteopromotive property of allogenic demineralized dentin matrix: A pilot study. J. West. Soc. Periodontol. Periodontal Abstr. 2013, 61, 35–38. [Google Scholar]
- Gomes, M.F.; Valva, V.N.; Vieira, E.M.; Giannasi, L.C.; Salgado, M.A.; Vilela-Goulart, M.G. Homogenous demineralized dentin matrix and platelet-rich plasma for bone tissue engineering in cranioplasty of diabetic rabbits: Biochemical, radiographic, and histological analysis. Int. J. Oral. Maxillofac. Surg. 2016, 45, 255–266. [Google Scholar] [CrossRef] [PubMed]
- Um, I.-W.; Kim, Y.-K.; Jun, S.-H.; Kim, M.-Y.; Cui, N. Demineralized Dentin Matrix as a Carrier of Recombinant Human Bone Morphogenetic Proteins: In vivo Study. J. Hard Tissue Biol. 2018, 27, 219–226. [Google Scholar] [CrossRef]
- Al-Namnam, N.; Shanmuhasuntharam, P.; Ha, K.O.; Siar, C.H. Processed allogenic dentine as a scaffold for bone healing: An in vivo study. Aust. J. Basic Appl. Sci. 2010, 4, 5932–5940. [Google Scholar]
- Um, I.W.; Ku, J.K.; Kim, Y.K.; Lee, B.K.; Leem, D.H. Histological Review of Demineralized Dentin Matrix as a Carrier of rhBMP-2. Tissue Eng. Part. B Rev. 2020, 26, 284–293. [Google Scholar] [CrossRef] [PubMed]
- Dubuc, F.L.; Urist, M.R. The accessibility of the bone induction principle in surface-decalcified bone implants. Clin. Orthop. Relat. Res. 1967, 55, 217–223. [Google Scholar] [PubMed]
- Urist, M.R.; Iwata, H.; Strates, B.S. Bone morphogenetic protein and proteinase in the guinea pig. Clin. Orthop. Relat. Res. 1972, 85, 275–290. [Google Scholar] [CrossRef]
- Masaru, M. Collagen biology for bone regenerative surgery. J. Korean Assoc. Oral. Maxillofac. Surg. 2012, 38, 321–325. [Google Scholar] [CrossRef]
- Fuentes, G.C.; Newgren, J. Physiology and clinical pathology of laboratory new zealand white rabbits housed individually and in groups. J. Am. Assoc. Lab. Anim. Sci. 2008, 47, 35–38. [Google Scholar]
- Urist, M.R.; Mikulski, A.; Boyd, S.D. A chemosterilized antigen-extracted autodigested alloimplant for bone banks. Arch. Surg. 1975, 110, 416–428. [Google Scholar] [CrossRef]
- Horowitz, M.C.; Friedlaender, G.E. Induction of specific T-cell responsiveness to allogeneic bone. J. Bone Jt. Surg. Am. 1991, 73, 1157–1168. [Google Scholar] [CrossRef]
- Mikulski, A.J.; Urist, M.R. An antigenic antimorphogenetic bone hydrophobic glycopeptide (AHG). Prep. Biochem. 1975, 5, 21–37. [Google Scholar] [CrossRef]
- Russell, J.L.; Block, J.E. Clinical utility of demineralized bone matrix for osseous defects, arthrodesis, and reconstruction: Impact of processing techniques and study methodology. Orthopedics 1999, 22, 524–531, quiz 532–533. [Google Scholar]
- Um, I.W.; Ku, J.K.; Lee, B.K.; Yun, P.Y.; Lee, J.K.; Nam, J.H. Postulated release profile of recombinant human bone morphogenetic protein-2 (rhBMP-2) from demineralized dentin matrix. J. Korean Assoc. Oral Maxillofac. Surg. 2019, 45, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Murata, M.; Akazawa, T.; Mitsugi, M.; Um, I.W.; Kim, K.W.; Kim, Y.K. Human dentin as novel biomaterial for bone regeneration. In Pignatello R, 1st ed.; InTech: New York, NY, USA, 2011; pp. 127–140. [Google Scholar]
- Ike, M.; Urist, M.R. Recycled dentin root matrix for a carrier of recombinant human bone morphogenetic protein. J. Oral. Implant. 1998, 24, 124–132. [Google Scholar] [CrossRef]
- Koga, T.; Minamizato, T.; Kawai, Y.; Miura, K.-I.; I, T.; Nakatani, Y.; Sumita, Y.; Asahina, I. Bone Regeneration Using Dentin Matrix Depends on the Degree of Demineralization and Particle Size. PLoS ONE 2016, 11, e0147235. [Google Scholar] [CrossRef] [PubMed]
- Um, I.W.; Kim, Y.K.; Mitsugi, M. Demineralized dentin matrix scaffolds for alveolar bone engineering. J. Indian Prosthodont. Soc. 2017, 17, 120–127. [Google Scholar] [CrossRef]
- Rijal, G.; Shin, H.I. Human tooth-derived biomaterial as a graft substitute for hard tissue regeneration. Regen. Med. 2017, 12, 263–273. [Google Scholar] [CrossRef]
- Friedenstein, A.J.; Chailakhyan, R.K.; Gerasimov, U.V. Bone marrow osteogenic stem cells: In vitro cultivation and transplantation in diffusion chambers. Cell Tissue Kinet. 1987, 20, 263–272. [Google Scholar] [CrossRef]
- Owen, M. Marrow stromal stem cells. J. Cell Sci. Suppl. 1988, 10, 63–76. [Google Scholar] [CrossRef]
- Folkman, J.; Greenspan, H.P. Influence of geometry on control of cell growth. Biochim. Et Biophys. Acta (BBA) Rev. Cancer 1975, 417, 211–236. [Google Scholar] [CrossRef]
- Kim, Y.K.; Pang, K.M.; Yun, P.Y.; Leem, D.H.; Um, I.W. Long-term follow-up of autogenous tooth bone graft blocks with dental implants. Clin. Case Rep. 2017, 5, 108–118. [Google Scholar] [CrossRef]
- Moon, Y.S.; Sohn, D.S.; Kim, G.; Park, I. Comparative Histomorphometric Evaluation of Bone Regeneration with Different Preparations of Xenogeneic Tooth Block Bone. Int. J. Oral. Maxillofac. Implant. 2019, 34, 1413–1422. [Google Scholar] [CrossRef]
- Kabir, M.A.; Murata, M.; Akazawa, T.; Kusano, K.; Yamada, K.; Ito, M. Evaluation of perforated demineralized dentin scaffold on bone regeneration in critical-size sheep iliac defects. Clin. Oral. Implant. Res. 2017, 28, e227–e235. [Google Scholar] [CrossRef]
- Glowacki, J.; Altobelli, D.; Mulliken, J.B. Fate of mineralized and demineralized osseous implants in cranial defects. Calcif. Tissue Int. 1981, 33, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Dozza, B.; Lesci, I.G.; Duchi, S.; Della Bella, E.; Martini, L.; Salamanna, F.; Falconi, M.; Cinotti, S.; Fini, M.; Lucarelli, E.; et al. When size matters: Differences in demineralized bone matrix particles affect collagen structure, mesenchymal stem cell behavior, and osteogenic potential. J. Biomed. Mater. Res. A 2017, 105, 1019–1033. [Google Scholar] [CrossRef] [PubMed]
- Nam, J.W.; Kim, M.Y.; Han, S.J. Cranial bone regeneration according to different particle sizes and densities of demineralized dentin matrix in the rabbit model. Maxillofac. Plast Reconstr. Surg. 2016, 38, 27. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Guo, W.; Yang, B.; Guo, L.; Sheng, L.; Chen, G.; Li, Y.; Zou, Q.; Xie, D.; An, X.; et al. Human treated dentin matrix as a natural scaffold for complete human dentin tissue regeneration. Biomaterials 2011, 32, 4525–4538. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.K.; Kim, S.G.; Yun, P.Y.; Yeo, I.S.; Jin, S.C.; Oh, J.S.; Kim, H.J.; Yu, S.K.; Lee, S.Y.; Kim, J.S.; et al. Autogenous teeth used for bone grafting: A comparison with traditional grafting materials. Oral. Surg. Oral. Med. Oral. Pathol. Oral. Radiol. 2014, 117, e39–e45. [Google Scholar] [CrossRef]
| Year (Author) [Ref.] | Theme | Donor (Tooth) | Geometry (Dentin) | Processing Method | Implant Site/Time | Key Findings |
|---|---|---|---|---|---|---|
| 1967 (Bang & Urist) [23] | Osteoinduction | Rabbit (Mature molar) | Whole root dentin | Complete demineralization (0.6 N HCl) | Abdominis muscle/12 weeks | Demineralized dentin (DDM): Similar to the matrix of bone, induces bone formation at 4 weeks proportional to the volume of the original implant. |
| Rat (Mature molar) | Un-demineralization | Abdominis muscle/12 weeks | Undemineralized dentin (MDM): Does not begin to be resorbed until 8–12 weeks later than demineralized dentine. | |||
| 1967 (Yeomans & Urist) [24] | Osteoinduction | Young New Zealand rabbit (Dentin) | Grinding dentin 1.0 mL | Complete demineralization (0.6 N HCl) | Rectus abdominis muscle/4, 8, 12 weeks | DDM: Rapidly resorbed more than bone, induces bone in 100% of implants within 4 weeks (3H-glycine autoradiograph: Positive evidence of translocation of 3H-glycine from the donor tissue to the cells of the host bed. Figure 2 in text) |
| Un-demineralization | Rectus abdominis muscle/4, 8, 12 weeks | Undemineralized dentin (MDM) induces bone in 75% of implants, but only after a latent period of 8–12 weeks. | ||||
| 1968 (Urist et al.) [25] | Inductive substrate antigenicity (HCl vs. EDTA) | Rabbit (Dentin) | Grinding dentin 1.0 mL | Demineralization (0.6 N HCl) | Abdominis muscle/4–12 weeks | Inductive substrate such that new bone formation is the best at 0.6 N HCl and 150 mmol NaCl, and is derived from the extracellular and not cytoplasmic proteins. Antigenicity: The immune response is lowered, and the inductive activity appears earlier with lyophilized and irradiated 60Co at the dose of 0.2 Mrads or less. |
| Rat (Dentin) | Demineralization (0.25 M EDTA) | EDTA: The inductive activity had been eliminated only from the exposed surface. Bone: The inductive activity was completely destroyed, Dentin: Persisted inductive activity for a longer time. | ||||
| 1970 (Huggins & Urist) [26] | ALP activity | Rat (Incisor) | Whole root dentin | Complete demineralization (0.5 N HCl, 1 mL/mg) | Rat abdominis muscle/1 year | Alkaline phosphatase activity (ALP) within 24 h: Reached maximum on day 7. Matrix transformation: Cartilage appeared within 5 days; bone and bone marrow formed within 14 days. The induced cartilage disappeared within 5 weeks. Transforming effects in postnatal life are comparable to embryonic induction because of highly cross-linked, exceptional resistance to the deleterious action of strong acid of dentin matrix. |
| 1970 (Huggins et al.) [27] | Transformation of fibroblasts | Rat (Incisor) Mice (Incisor) | Dentin powder (pooling) 200–400 µm (10–15 mg) | Complete demineralization (0.5 N HCl) | Subcutaneous pockets/9–21 days | DDM: Active transformation. An early and intense attraction for fibroblasts of the subcutaneous tissue. |
| Un-demineralization | MDM: Does not induce ALP, cartilage, or bone. | |||||
| Guinea pig (Incisor) | Dentin powder (Pooling) 200–400 µm (10–15 mg) | Complete demineralization (0.5 N HCl) | Subcutaneous pockets/9–21 days | DDM: No bone induction in guinea pig Transformation of fibroblast: Rat, mouse > guinea pig | ||
| Un-demineralization | MDM: Does not induce alkaline phosphate, cartilage, or bone. | |||||
| 1972 (Bang) [28] | Antigenicity | Guinea pig (Incisor) | Root dentin pieces 2 × 2 × 1 mm | Demineralization (0.2 N HCl) | Abdominis muscle/4–12 weeks | Antigenicity: Histological examination 1. Demineralized and undemineralized dentin could evoke an immune reaction. 2. The first set of Allo-DDM induces bone formation in high percentages of cases. 3. Induction was reduced in 2nd set of grafts 4. No difference was observed in bone-inducing capacity between DDM and lyophilized DDM. |
| Guinea pig (Molar) | Pieces 2 × 2 × 1 mm | Un-demineralization | Osteoinduction is prevented in the 2nd set of implants in MDM. | |||
| 1973 (Reddi & Huggins) [29] | Role of geometry | Rat (Incisor) | Whole tooth root | Demineralization (0.5 N HCl) | Subcutaneous tissue/7–35 days | Cartilage–chondrolysis–osteogenesis–ossicle with hemopoietic marrow. Figure 3a,b in Text |
| Whole tooth root | Un-demineralization | Bone and cartilage: Not observed. Finding of fibrosis: Consistent with hypoxic environment of pulp chamber. Figure 3c in Text | ||||
| 1973 (Bang) [30] | Osteoinduction (HCl vs. EDTA) | Guinea pig (Molar) | Molar dentin 1/2 root 6–7 × 3 × 1.5 mm, 30 mm3, 30 mg | Demineralization (0.6 N HCl, 0.5 M EDTA) | Abdominis muscle/4–12 weeks | HCl, DDM: 13 out of 30 implants induce bone formation EDTA, DDM: 12 out of 30 implants induce bone formation |
| Un-demineralization | MDM: Bone induction process is retarded, and the yield of new bone is low. | |||||
| Rat (Molar, Incisor) | Whole molar tooth bud | Abdominis muscle/4–12 weeks | Tooth bud: No induction | |||
| Incisor dentin 6–7 × 3 × 1.5 mm, 30 mm3, 30 mg | Demineralization (0.6 N HCl, 0.5 M EDTA) | HCl DDM: 1 out of 12 implants induce bone formation EDTA DDM: 3 out of 12 implants induce bone formation No distinct differences in the bone-inducing capacity of HCl and EDTA demineralization. | ||||
| Incisor dentin 6–7 × 3 × 1.5 mm, 30 mm3, 30 mg | Un-demineralization | 3 out of 24 MDM: Osteoinduction after 12 weeks, bone induction process was retarded, and the yield of new bone was low | ||||
| 1975 (Linden) [31] | Osteoinduction cell sequences | Ash-Wistar rat (Incisor) | Whole root dentin block | Demineralization (0.6 N HCl) | Abdominis muscles/40 days | Cell Sequences in bone induction Qualitative trends may be listed as: (1) inflammation; (2) formation of vascularized connective tissue; (3) erosion; (4) recalcification; and (5) bone formation. |
| 1977 (Nilsen) [32] | Cell reaction | Guinea pig (Molar) | Root dentin Piece 6–7 × 3 × 1.5 mm | Demineralization (0.2 M HCl) | Abdominal muscles/22 days | Induce osteoid formation: Resorption of DDM is a prerequisite for osteoid formation (1) Matrix resorptive reaction mediated by monocytes, macrophages, and dentinoclasts. (2) Fibroblastic reaction as an unspecific capsulation process. (3) Osteoblastic reaction with osteoid matrix formation |
| 1986 (Inoue et al.) [33] | Chondrogenesis in the muscle, skin, periodontal ligament, and bone marrow | Wistar rat (Incisor) | Dentin rolls One incisor-one implant (not pool) | Demineralization (0.6 N HCl) | Abdominal muscles pouch/21 days | Cartilage is formed at 7 days Rectus abdominis muscle (cartilage induce) > Chest subcutaneous tissue (cartilage induce) > Periodontal ligament (deposits of cartilage were not seen) |
| Subcutaneous pocket in the chest/21 days | Cartilage is first found at 10 days | |||||
| Periodontal ligament of the first molar/21 days | Cartilage was not seen | |||||
| Bone marrow in the femur/21 days | Woven bone is found at 10 days | |||||
| 1990 (Pinholt et al.) [34] | Osteoinduction | Male Wistar rat (Incisor) | 4 Granules1 × 1 × 1 mm | Demineralization (0.2 N HCl) | Abdominis muscle/4 weeks | Dentin induced new bone formation in 100% of implants No inflammatory or foreign body reactions were observed. |
| Year [Author] | Theme | Donor (Tooth) | Geometry (Dentin) | Processing Method | Implant Site/Time | Key Findings |
|---|---|---|---|---|---|---|
| 1967 (Yeomans & Urist) [24] | Osteoinduction | New Zealand rabbit | Grinding Dentin 1.0 mL | Complete demineralization (0.6 N HCl) | Mandibular drill hole (ø 5 mm)/4, 8, 12 weeks | DDM: Induce osteogenesis, not dentinogenesis. (1) Bone formed by extension of proliferating osteogenetic cells from the host bed and (2) bone formed by induction inside of dentine matrix are generally interwoven and continuous processes. The old DDM: Resorbed and partially refilled with new bone within 4 weeks, more slowly than bone; produced a separate and unattached ossicle inside the cavity. After 12 weeks, the end product is bone marrow and not solid bone tissue. |
| Extraction socket/4, 8, 12 weeks | DDM: Induce osteogenesis and not dentinogenesis. The processes of (1) The regeneration from host bed and (2) the osteoinductive new bone formation from the dentin matrix generally progressed separately. Bone induction is the same as implants in the abdominal wall. | |||||
| Un-demineralization | Mandibular drill hole (ø 5 mm)/4, 8, 12 weeks | MDM: New bone formation in 75% of implants only after a latent period of 8–12 weeks. Same results with (23, 24) in soft tissue | ||||
| Extraction socket/4, 8, 12 weeks | MDM: New bone formation in 75% of implants only after a latent period of 8–12 weeks. Same results with (23, 24) in soft tissue | |||||
| 1990 (Pinholt et al.) [34] | Osteoinduction | Male Wistar rat (Incisor) | Granules 1 × 1 × 1 mm | Demineralization (0.2 N HCl) | Premaxilla, alveolar ridge (subperiosteal)/ 4 weeks | All 10 DDMs: Induced new bone formation Tissue response: No inflammatory or foreign body reactions were observed |
| 1972 (Bang et al.) [35] | Osteoinduction Osteoconduction | 16 Java monkeys | Root dentin pieces 4 × 1 × 1 mm, | Demineralization (0.2 N HCl) | Mandibular defect (ø 7 mm)/1 week–1 year | DDM: Osteoinduction and osteoconduction in histologic study |
| 2004 (Carvalho et al.) [36] | Osteopromotion | 36 adult rabbits (Central incisor) | Slices, 8 mm thick (Consisting of enamel, dentin, and cementum) | Complete demineralization (0.6 N HCl) | Mandibular defect (ø 5 mm and 2 mm in depth)/Approx. 13 weeks (90 days) | DDM slices: Biocompatible, stimulated newly formed bone until 30 days after implantation; resorbed during the bone remodeling process. The volume of the newly formed bone is significantly greater in the dentin graft than in ungrafted negative controls with low antigenicity during 13 weeks in a histologic study |
| 2007 (Gomes et al.) [37] | Osteopromotion | 48 New Zealand rabbits. Central incisors | Slices, 8 mm in thickness (Consisting of enamel, dentin, and cementum) | Complete demineralization (0.6 N HCl) | Parietal defect (ø 8 mm)/Approx. 13 weeks (90 days) | DDM: Significantly greater bone density than the ungrafted controls with low antigenicity for 13 weeks |
| 2008 (Gomes et al.) [38] | Osteopromotion Optical density | 48 New Zealand rabbits (Central incisor) | Slices, 8 mm in thickness (Consisting of enamel, dentin, and cementum) | Complete demineralization (0.6 HCl) | Parietal defect (ø 8 mm)/Approx. 13 weeks (90 days) | DDM: Dentin shows significantly greater radio-opacity and better trabecular bone arrangement than the empty negative controls during 13 weeks in a radiological study |
| 2010 (Al-Namnam et al.) [44] | Osteocompatibility Quantitative comparison of bone formation | 16 New Zealand white rabbits (Central incisor root) | Dentin particles 2–4 mm | Un-demineralization | Femoral defects (ø 5 mm)/12 weeks | MDM particle: No significant difference in new bone formation between autogenous bone graft, ungrafted sites, and MDM particles on histomorphometric analysis until 12 weeks. |
| 2012 (Bormann et al.) [39] | Inflammatory and neovascularization response | 24 isogenic mice (Mandibular central incisor) | Perforated (300 µm) dentin slices. 3 × 3 × 1 mm1 mm thick | Un-demineralization | Calvarial defect (36 mm2)/3 weeks (22 days) | Perforated MDM slice and ß-TCP scaffolds are similar to isogenic bone in terms of inflammatory and neovascularization response, highlighting their potential utility in the regeneration of bone defects. |
| 2013 (Bakhshalian et al.) [40] | Osteopromotion | 6 rabbits (Central incisor) | Dentin pieces 2 mm3 | Complete demineralization (0.6 N HCl) | Parietal defect (ø 8 mm, 0.5 mm in depth)/15–90 days | The amount of bone regeneration: Significantly higher in the DDM group than in the ungrafted group. |
| 2013 (Bakhshalian et al.) [41] | Osteopromotion Blood biomarkers | 24 New Zealand white rabbits (Mandibular incisor) | Powders 300 µm in pooling | Complete demineralization (0.6 N HCl) | Skull defect (ø 8 mm, 0.5 mm in depth)/15–90 days | DDM: Significantly increased bone mass and improved bone quality without causing an inflammatory reaction or infection. WBC count: Higher in the early stage, but lower in the later stage than that in the empty control. ALP: There was no difference in the plasma. |
| 2016 (Gomes et al.) [42] | Osteopromotion | 60 adult New Zealand rabbits (Central incisor) | Slices, 8 mm in thickness (Consisting of enamel, dentin, and cementum) | Complete demineralization (0.6 N HCl) | Parietal defect (ø 8 mm)/Approx. 13 weeks (90 days) | ALP: Significantly higher in the DDM group than in the empty control, empty diabetic, and DDM–PRP groups, confirming the findings of intense osteoblastic activity and increased bone mineralization. DDM promoted superior bone architectural microstructure in bone defects in diabetic rabbits because of its effective osteoinductive and osteoconductive activity, whereas PRP stimulated angiogenesis and red bone marrow formation. |
| 2018 (Um et al.) [43] | Osteopromotion | 6 rabbits (Dentin) | Powder 300–800 µm | Partial demineralization (0.6 N HCl) | Calvarial defect (ø 8 mm)/1–4 weeks | DDM: Osteoinductive and osteoconductive function in a histological study. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Um, I.-W.; Lee, J.-K.; Kim, J.-Y.; Kim, Y.-M.; Bakhshalian, N.; Jeong, Y.K.; Ku, J.-K. Allogeneic Dentin Graft: A Review on Its Osteoinductivity and Antigenicity. Materials 2021, 14, 1713. https://doi.org/10.3390/ma14071713
Um I-W, Lee J-K, Kim J-Y, Kim Y-M, Bakhshalian N, Jeong YK, Ku J-K. Allogeneic Dentin Graft: A Review on Its Osteoinductivity and Antigenicity. Materials. 2021; 14(7):1713. https://doi.org/10.3390/ma14071713
Chicago/Turabian StyleUm, In-Woong, Jeong-Keun Lee, Jun-Young Kim, Yu-Mi Kim, Neema Bakhshalian, Yeong Kon Jeong, and Jeong-Kui Ku. 2021. "Allogeneic Dentin Graft: A Review on Its Osteoinductivity and Antigenicity" Materials 14, no. 7: 1713. https://doi.org/10.3390/ma14071713
APA StyleUm, I.-W., Lee, J.-K., Kim, J.-Y., Kim, Y.-M., Bakhshalian, N., Jeong, Y. K., & Ku, J.-K. (2021). Allogeneic Dentin Graft: A Review on Its Osteoinductivity and Antigenicity. Materials, 14(7), 1713. https://doi.org/10.3390/ma14071713







