Evaluation of the Antimicrobial Protection of Pharmaceutical Kaolin and Talc Modified with Copper and Zinc
Abstract
1. Introduction
2. Material and Methods
2.1. Materials
2.2. Sample Preparation
2.2.1. Metal Enrichment of Clays
2.2.2. Preparation of Pastes
2.3. Characterization
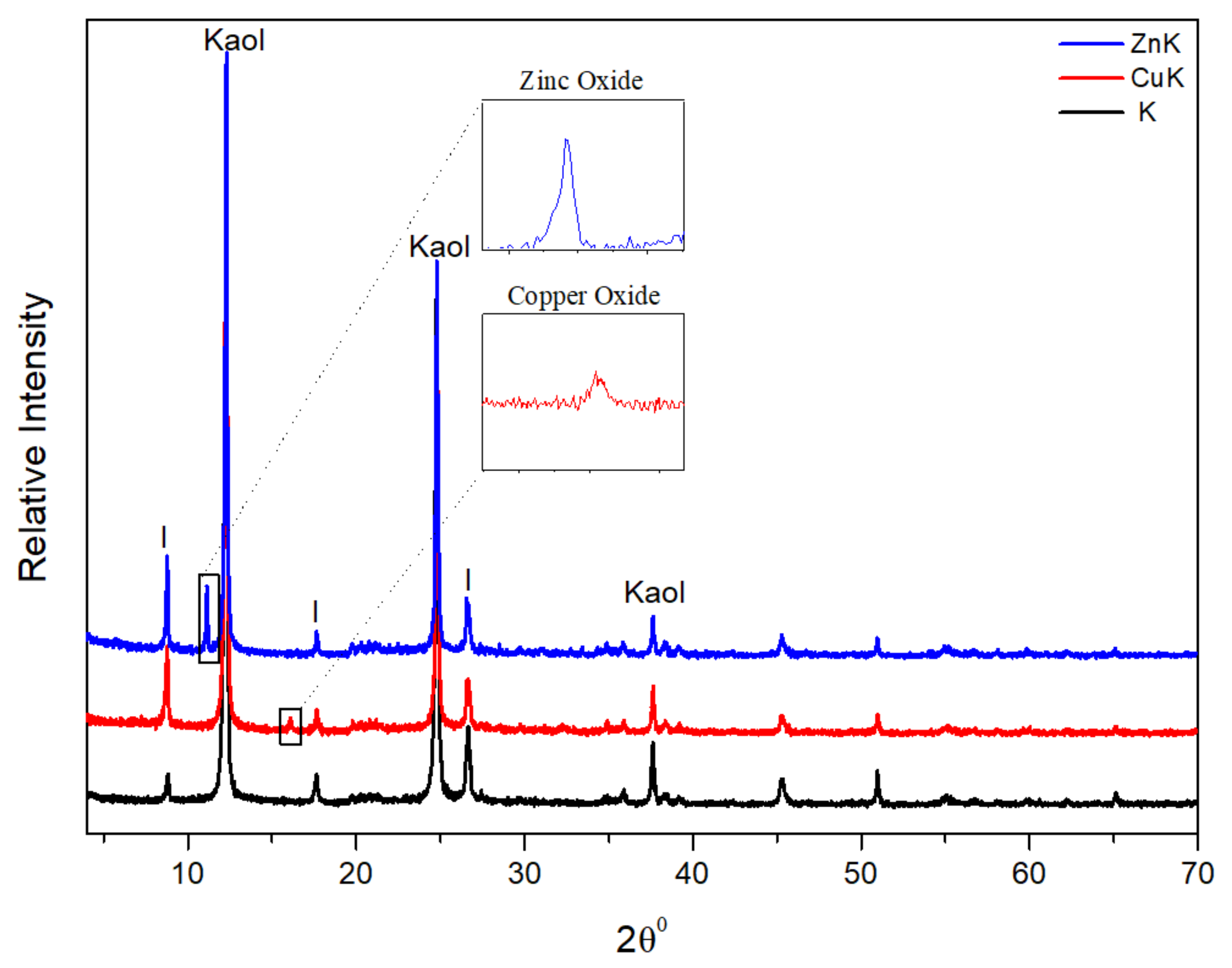
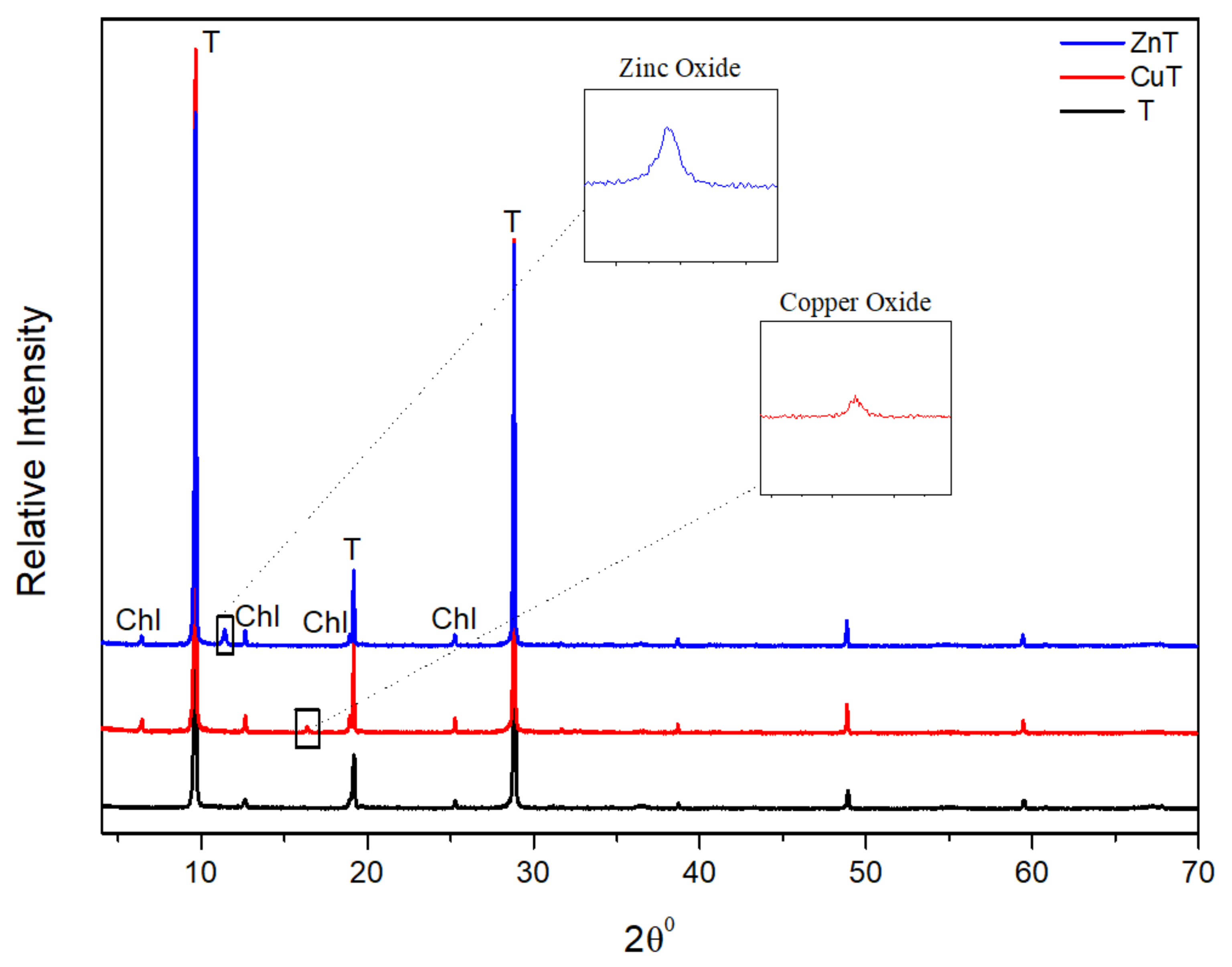
2.3.1. X-ray Diffraction Analysis
2.3.2. Scanning Electron Microscope-Energy Dispersive Spectroscopy
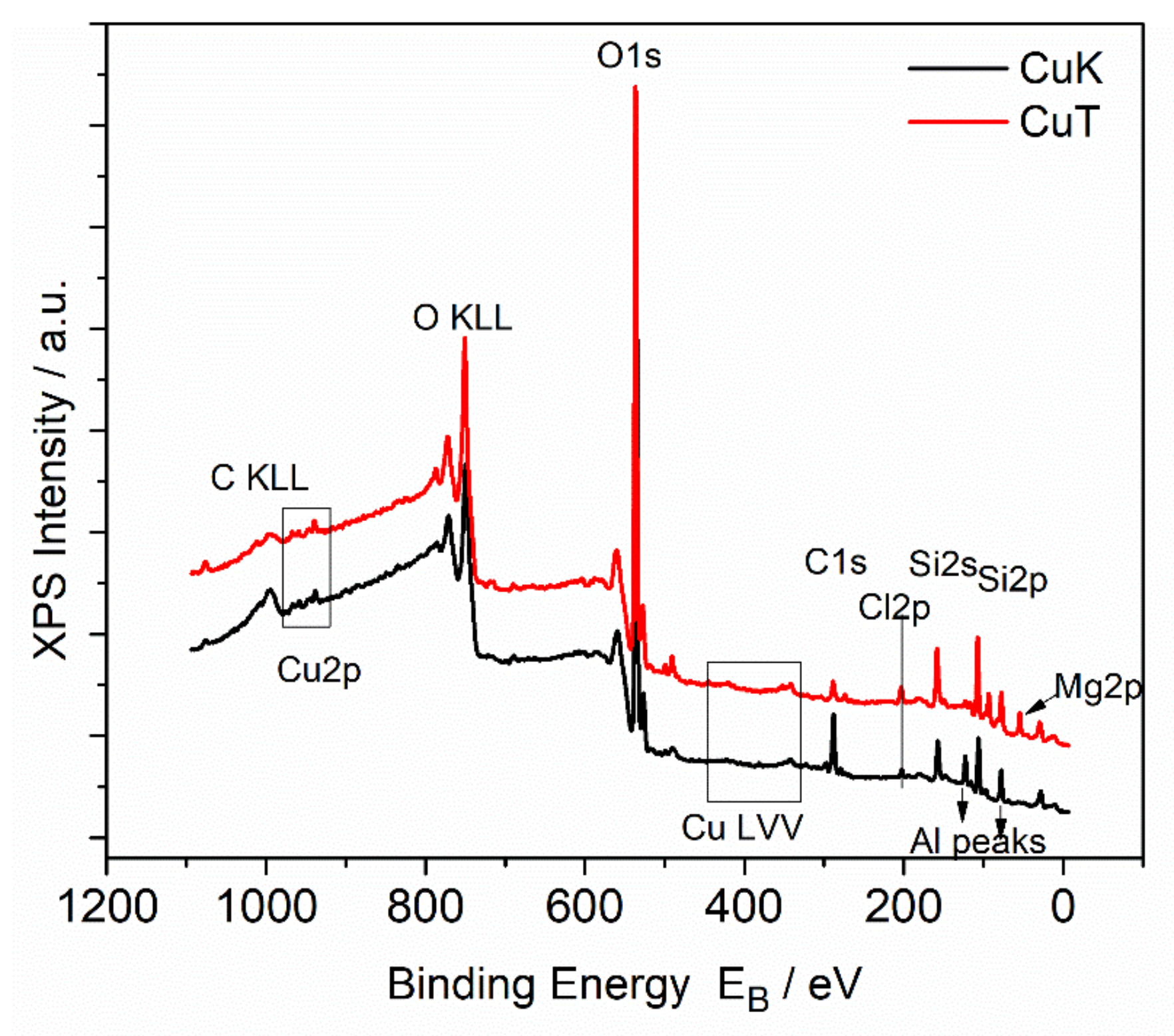
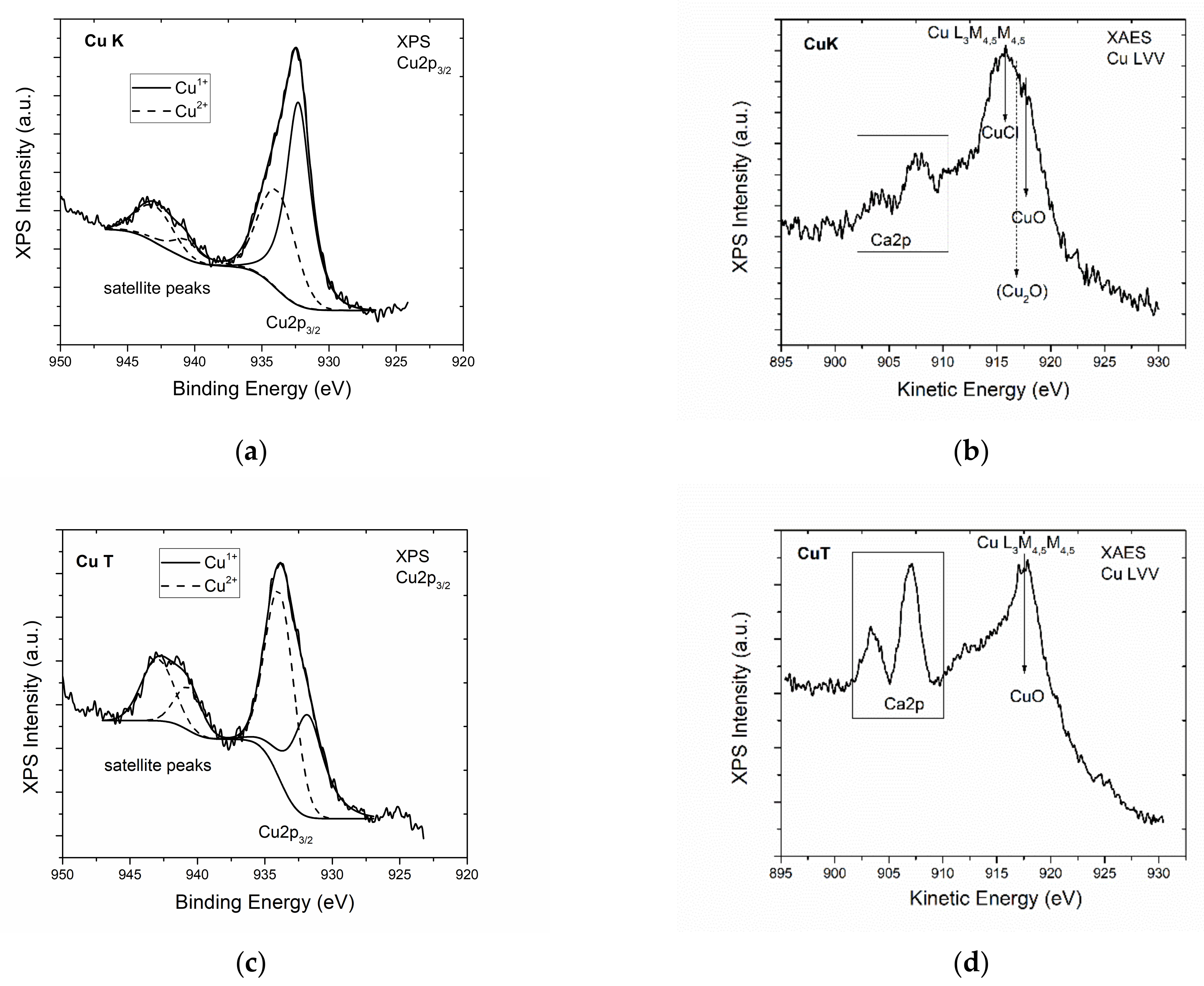
2.3.3. X-ray Photoelectron Spectroscopy
2.3.4. X-ray Fluorescence
2.4. Evaluation of Antimicrobial Protection
3. Results and Discussion
3.1. X-ray Diffraction Analysis
3.2. Scanning Electron Microscope (SEM)-Energy Dispersive Spectroscopy
3.3. X-ray Fluorescence Spectroscopy Analysis
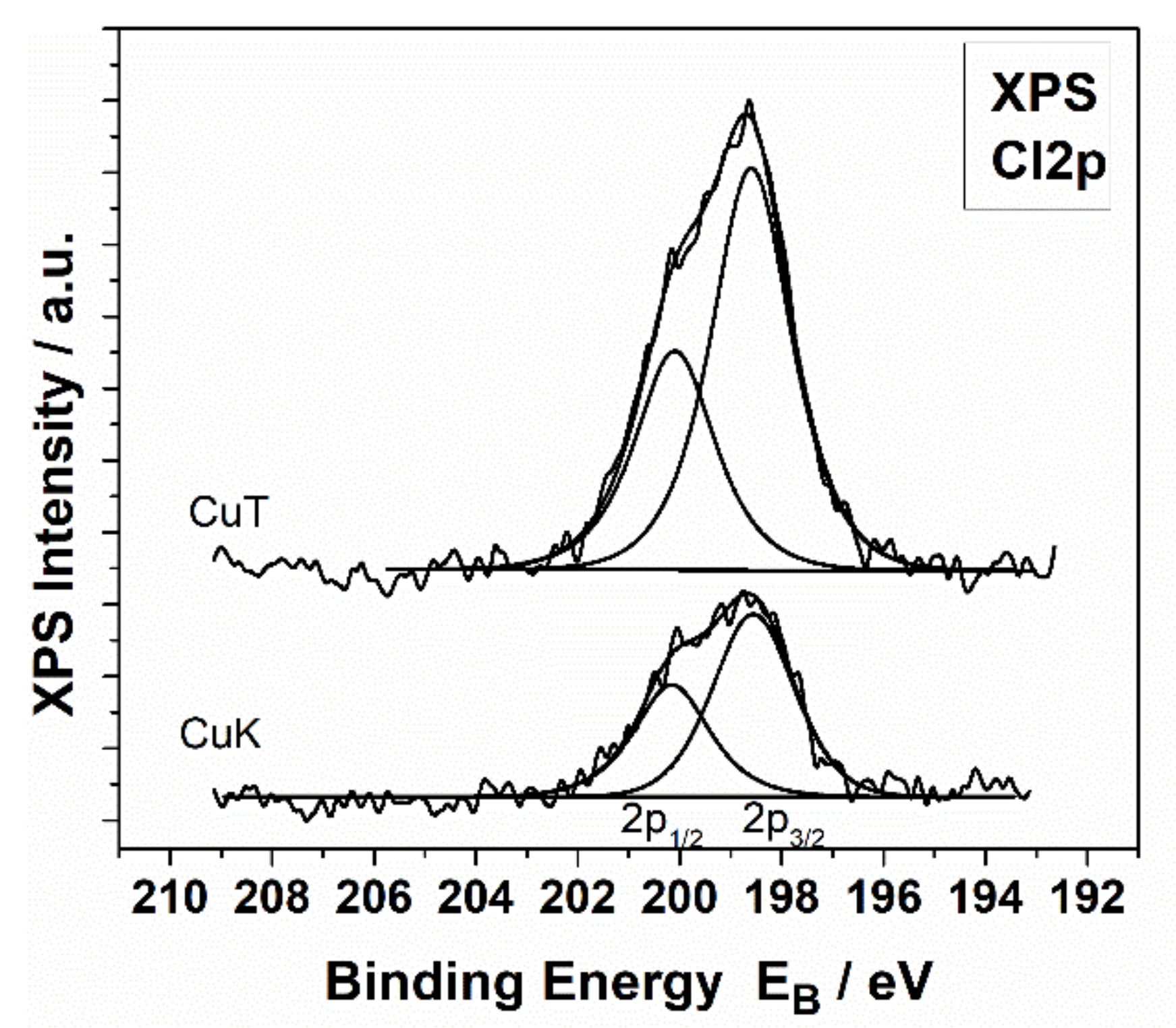
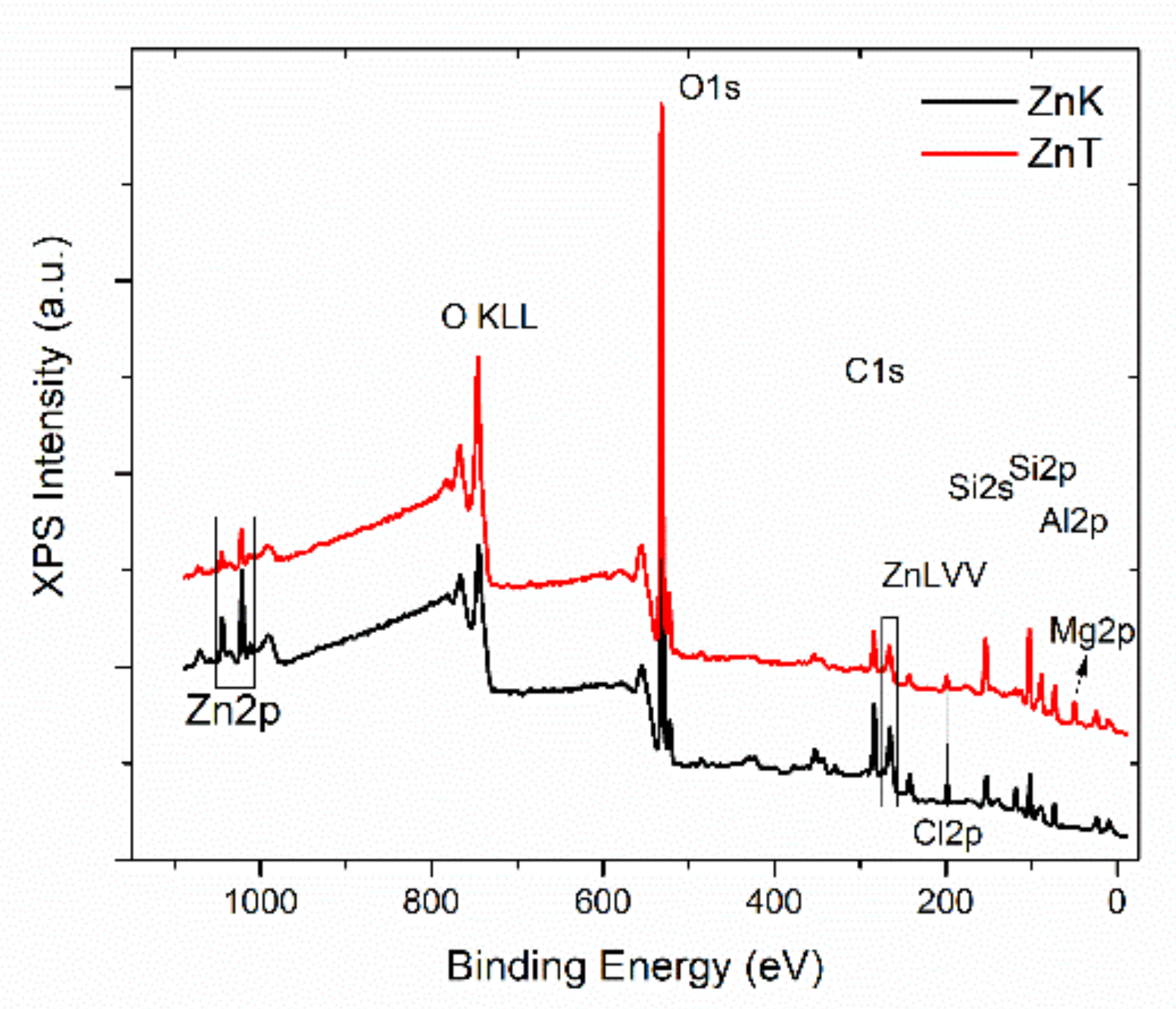
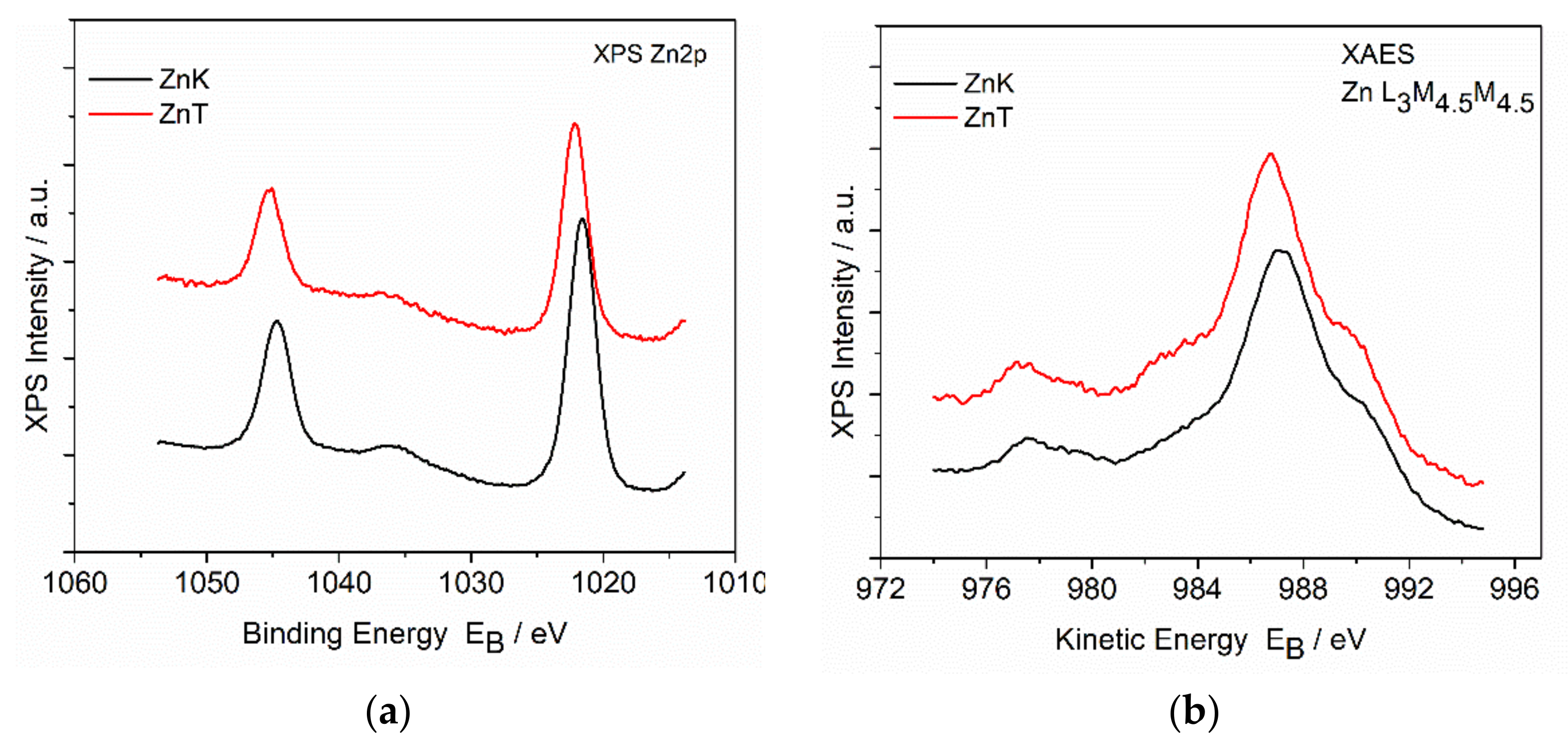
3.4. X-ray Photoelectron Spectroscopy
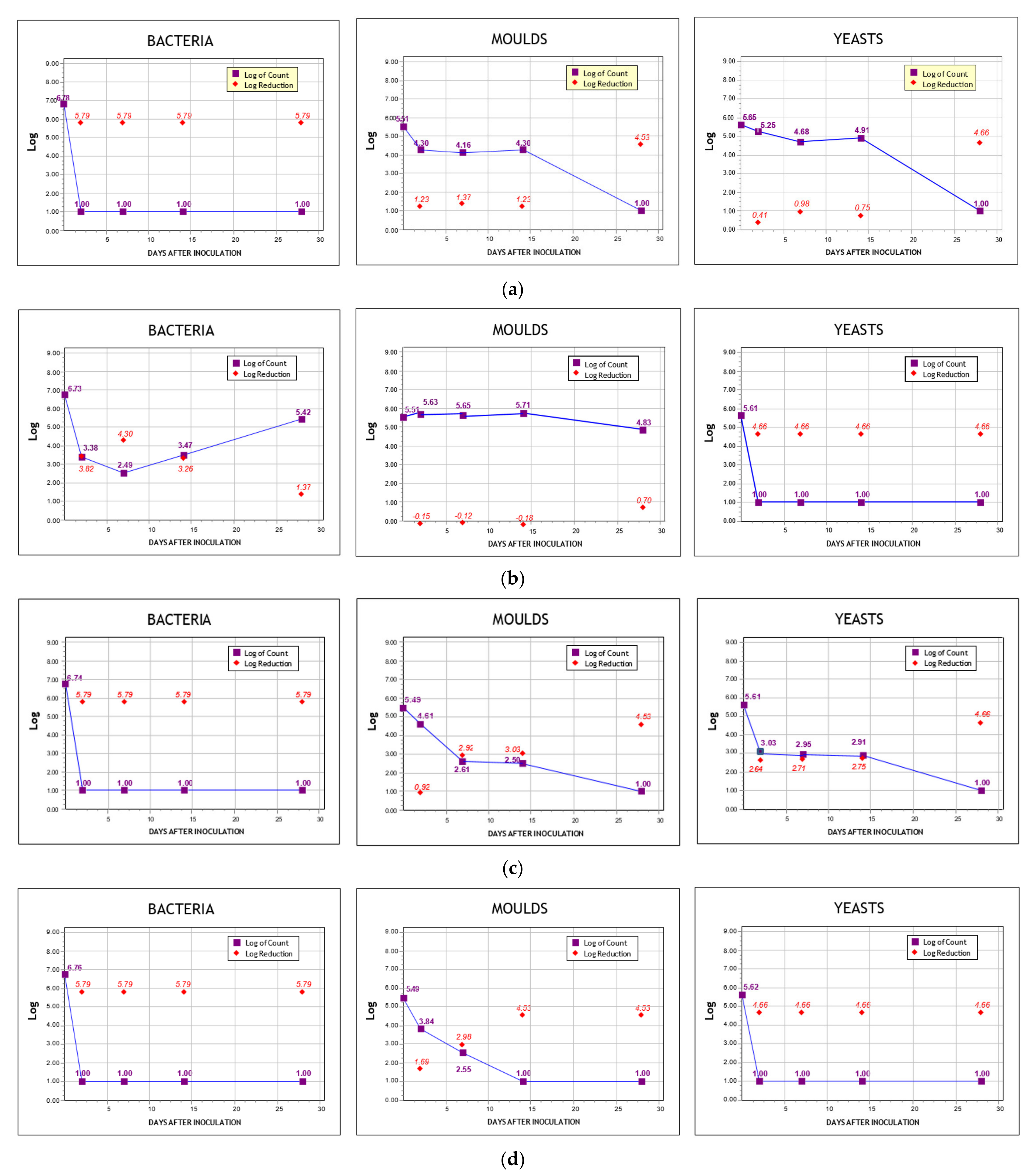
3.5. Challenge Test
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Carretero, M.; Gomes, C.; Tateo, F. Chapter 11.5 Clays and Human Health. Dev. Clay Sci. 2006, 1, 717–741. [Google Scholar]
- Lin, F.; Lee, Y.; Jian, C.; Wong, J.-M.; Shieh, M.-J.; Wang, C.-Y. A study of purified montmorillonite intercalated with 5-fluorouracil as drug carrier. Biomaterials 2002, 23, 1981–1987. [Google Scholar] [CrossRef]
- Viseras, C.; Carazo, E.; Borrego-Sánchez, A.; García-Villén, F.; Sánchez-Espejo, R.; Cerezo, P.; Aguzzi, C. Clay Minerals in Skin Drug Delivery. Clays Clay Miner. 2019, 67, 59–71. [Google Scholar] [CrossRef]
- Carretero, M. Clay minerals and their beneficial effects upon human health. A review. Appl. Clay Sci. 2002, 21, 155–163. [Google Scholar] [CrossRef]
- Bergaya, F.; Lagaly, G. Chapter 1 General Introduction: Clays, Clay Minerals, and Clay Science. Dev. Clay Sci. 2006, 1, 1–18. [Google Scholar]
- Duffin, C.J.; Moody, R.T.J.; Gardner-Thorpe, C. A History of Geology and Medicine; The Geological Society: London, UK, 2013; p. 375. [Google Scholar]
- Bech, J. Les Terres Medicinals. Discurs per Reial Academia de Farmàcia de Barcelona; Reial Acadèmia de farmàcia de Barcelona—CIRIT (Generalitat de Catalunya): Barcelona, Spain, 1987; p. 105. [Google Scholar]
- Bolger, R. Industrial minerals in pharmaceuticals. Ind. Miner. 1995, 8, 52–63. [Google Scholar]
- Cornejo, J. Las arcillas en formulaciones farmacéuticas. In Conferencias de IXy X Reuniones de la Sociedad Española de Arcillas, Madrid, Spain, March 1988; Galan, E., Ortega, M., Eds.; Universidad de Granada: Granada, Spain, 1990; pp. 51–68. [Google Scholar]
- Del Pozo, A. Farmacia Galenica Especial. Tomo 2; Romargraf: Barcelona, Spain, 1978; p. 399. [Google Scholar]
- Del Pozo, A. Farmacia Galenica Especial. Tomo 3; Romargraf: Barcelona, Spain, 1979; p. 452. [Google Scholar]
- Galan, E.; Liso, M.J.; Forteza, M. Minerales utilizados en la industria farmaceútica. Boletín Soc. Española Mineral. 1985, 8, 369–378. [Google Scholar]
- Gámiz, E.; Linares, J.; Delgado, R. Assessment of two Spanish bentonites for pharmaceutical uses. Appl. Clay Sci. 1992, 6, 359–368. [Google Scholar] [CrossRef]
- Lopez Galindo, A.; Viseras, C. Pharmaceutical applications of fibrous clays (sepiolite and palygorskite) from some circummediterranean deposits. In 1st Latin American Clay Conference. Funchal, Portugal, 17–22 September 2000; Gomes, C.S.F., Ed.; Associacao Portuguesa de Argilas (APA): Funchal, Portugal, 2000; pp. 258–270. [Google Scholar]
- Veniale, F. Applicazioni e utilizzazioni medico-sanitarie di materialiargillosi (naturali emodificati). In Argille e MineralidelleArgille. Guidaalla Definizione di Caratteristiche e Proprieta‘ per gliUsiIndustriali. Corso di Formazione; Morandi, N., Dondi, M., Eds.; Gruppo Ital. AIPEA: Rimini, Italy, 1997; pp. 205–239. [Google Scholar]
- Viseras, C.; Lopez-Galindo, A. Pharmaceutical applications of some spanish clays (sepiolite, palygorskite, bentonite): Some preformulation studies. Appl. Clay Sci. 1999, 14, 69–82. [Google Scholar] [CrossRef]
- López-Galindo, A.; Viseras, C.; Cerezo, P. Compositional, technical and safety specifications of clays to be used as pharmaceutical and cosmetic products. Appl. Clay Sci. 2007, 36, 51–63. [Google Scholar] [CrossRef]
- Dixon, J.B.; Weed, S.B.; Parpitt, R.L. Minerals in Soil Environments. Soil Sci. 1990, 150, 562. [Google Scholar] [CrossRef][Green Version]
- Grim, R.E. Clay Mineralogy. Soil Sci. 1953, 76, 317. [Google Scholar] [CrossRef]
- McBride, M.B. Environmental Chemistry of Soils; Oxford University Press: New York, NY, USA, 1994. [Google Scholar]
- Sumner, M.E. Handbook of Soil Science; CRC Press: Washington, DC, USA, 2000. [Google Scholar]
- Wilson, M.J. Sheet Silicates-Clay Minerals, 2nd ed.; The Geological Society: London, UK, 2013; p. 724. [Google Scholar]
- Zhang, Y.; Long, M.; Huang, P.; Yang, H.; Chang, S.; Hu, Y.; Tang, A.; Mao, L. Intercalated 2D nanoclay for emerging drug delivery in cancer therapy. Nano Res. 2017, 10, 2633–2643. [Google Scholar] [CrossRef]
- Zhang, Y.; Long, M.; Huang, P.; Yang, H.; Chang, S.; Hu, Y.; Tang, A.; Mao, L. Emerging integrated nanoclay-facilitated drug delivery system for papillary thyroid cancer therapy. Sci. Rep. 2016, 6, 33335. [Google Scholar] [CrossRef] [PubMed]
- Schiffenbauer, M.; Stotzky, G. Adsorption of coliphages T1 and T7 to clay minerals. Appl. Environ. Microbiol. 1982, 43, 590–596. [Google Scholar] [CrossRef] [PubMed]
- Lipson, S.M.; Stotzky, G. Adsorption of reovirus to clay minerals: Effects of cation-exchange capacity, cation saturation, and surface area. Appl. Environ. Microbiol. 1983, 46, 673–682. [Google Scholar] [CrossRef]
- Steel, R.F.; Anderson, W. The interaction between kaolinite and Staphylococcus aureus. J. Pharm. Pharmacol. 1972, 24, 129. [Google Scholar]
- Wallace, W.E., Jr.; Headley, L.C.; Weber, K.C. Dipalmitoyl lecithin surfactant adsorption by kaolin dust in vitro. J. Colloid Interface Sci. 1975, 51, 535–537. [Google Scholar] [CrossRef]
- Adamis, Z.; Timar, M. Investigations of the effect of quartz, aluminum silicates and colliery dusts on peritoneal macrophages in vitro. In The In Vitro Effects of Mineral Dusts; Brown, R.C., Gormley, I.P., Chamberlain, M., Davies, R., Eds.; Academic Press: London, UK, 1980; pp. 13–18. [Google Scholar]
- Carretero, M.I.; Pozo, M. Clay and non-clay minerals in the pharmaceutical industry. Appl. Clay Sci. 2009, 46, 73–80. [Google Scholar] [CrossRef]
- Moraes, J.D.D.; Bertolino, S.R.A.; Cuffini, S.L.; Ducart, D.F.; Bretzke, P.E.; Leonardi, G.R. Clay minerals: Properties and applications to dermocosmetic products and perspectives of natural raw materials for therapeutic purposes—A review. Int. J. Pharm. 2017, 534, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Carretero, M.; Gomes, C.; Tateo, F. Clays, Drugs, and Human Health. Dev. Clay Sci. 2013, 5, 711–764. [Google Scholar]
- Awad, M.E.; López-Galindo, A.; Setti, M.; El-Rahmany, M.M.; Iborra, C.V. Kaolinite in pharmaceutics and biomedicine. Int. J. Pharm. 2017, 533, 34–48. [Google Scholar] [CrossRef] [PubMed]
- Schliemann, S.; Petri, M.; Elsner, P. Preventing irritant contact dermatitis with protective creams: Influence of the application dose. Contact Dermat. 2013, 70, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Intekom, Pharmaceutical Information about Caloplast Kaolin Poultice (Medicinal Informative Website, South African Electronic Package). Available online: http://home.intekom.com/pharm/allied/caloplst.html/ (accessed on 19 September 2017).
- Cara, S.; Carcangiu, G.; Padalino, G.; Palomba, M.; Tamanini, M. The bentonites in pelotherapy: Chemical, mineralogical and technological properties of materials from Sardinia deposits (Italy). Appl. Clay Sci. 2000, 16, 117–124. [Google Scholar] [CrossRef]
- Cara, S.; Carcangiu, G.; Padalino, G.; Palomba, M.; Tamanini, M. The bentonites in pelotherapy: Thermal properties of clay pastes from Sardinia (Italy). Appl. Clay Sci. 2000, 16, 125–132. [Google Scholar] [CrossRef]
- Etich, W.K.N.; Mwangi, E.M.; Kiptoo, J.; Digo, C.A.; Ombito, J. In vitro determination of sun protection factor on clays used for cosmetic purposes in Kenya. Chem. Mater. Res. 2014, 6, 25–30. [Google Scholar]
- Lacto® Calamine Informative and Commercial Website of Dermatological Products. 2016. Available online: http://www.lactocalamine.in/ (accessed on 28 February 2021).
- Carretero, M.I.; Pozo, M. Clay and non-clay minerals in the pharmaceutical and cosmetic industries Part II. Active ingredients. Appl. Clay Sci. 2010, 47, 171–181. [Google Scholar] [CrossRef]
- Cervini-Silva, J.; Camacho, A.N.; Palacios, E.; Del Angel, P.; Pentrak, M.; Pentrakova, L.; Kaufhold, S.; Ufer, K.; Ramírez-Apan, M.T.; Gómez-Vidales, V.; et al. Anti-inflammatory, antibacterial, and cytotoxic activity by natural matrices of nano-iron(hydr)oxide/halloysite. Appl. Clay Sci. 2016, 120, 101–110. [Google Scholar] [CrossRef]
- Karthikeyan, P.; Mitu, L.; Pandian, K.; Anbarasu, G.; Rajavel, R. Electrochemical deposition of a Zn–HNT/p(EDOT–co–EDOP) nanocomposite on LN SS for anti–bacterial and anti–corrosive applications. New J. Chem. 2017, 41, 4758–4762. [Google Scholar] [CrossRef]
- Stavitskaya, A.; Shakhbazova, C.; Cherednichenko, Y.; Nigamatzyanova, L.; Fakhrullina, G.; Khaertdinov, N.; Kuralbayeva, G.; Filimonova, A.; Vinokurov, V.; Fakhrullin, R. Antibacterial properties and in vivo studies of tannic acid-stabilized silver–halloysite nanomaterials. Clay Miner. 2020, 55, 112–119. [Google Scholar] [CrossRef]
- Lansdown, A.B. Silver in Health Care: Antimicrobial Effects and Safety in Use. Biofunctional Text. Skin 2006, 33, 17–34. [Google Scholar]
- Hassen, A.; Saidi, N.; Cherif, M.; Boudabous, A. Effects of heavy metals on Pseudomonas aeruginosa and Bacillus thuringiensis. Bioresour. Technol. 1998, 65, 73–82. [Google Scholar] [CrossRef]
- Harrison, J.J.; Ceri, H.; Stremick, C.A.; Turner, R.J. Biofilm susceptibility to metal toxicity. Environ. Microbiol. 2004, 6, 1220–1227. [Google Scholar] [CrossRef] [PubMed]
- Borkow, G.; Gabbay, J.; Zatcoff, R.C. Could chronic wounds not heal due to too low local copper levels? Med. Hypotheses 2008, 70, 610–613. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.; Han, G.; Zhang, Y.; Pan, Y.; Li, X.; Xia, Y.; Wu, Y. Antifungal Activity and Cytotoxicity of Zinc, Calcium, or Copper Alginate Fibers. Biol. Trace Element Res. 2012, 148, 415–419. [Google Scholar] [CrossRef]
- Strodtbeck, F. Physiology of wound healing. Newborn Infant Nurs. Rev. 2001, 1, 43–52. [Google Scholar] [CrossRef]
- Rougier, A.; Humbert, P. Clinical efficacy on epidermal wound healing of topically applied madecassoside associated with copper/zinc/manganese salts. J. Am. Acad. Dermatol. 2008, 58, AB144. [Google Scholar]
- Abendrot, M.; Chęcińska, L.; Kusz, J.; Lisowska, K.; Zawadzka, K.; Felczak, A.; Kalinowska-Lis, U. Zinc(II) Complexes with Amino Acids for Potential Use in Dermatology: Synthesis, Crystal Structures, and Antibacterial Activity. Molecules 2020, 25, 951. [Google Scholar] [CrossRef]
- Abendrot, M.; Kalinowska-Lis, U. Zinc-containing compounds for personal care applications. Int. J. Cosmet. Sci. 2018, 40, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Pasquet, J.; Chevalier, Y.; Pelletier, J.; Couval, E.; Bouvier, D.; Bolzinger, M.-A. The contribution of zinc ions to the antimicrobial activity of zinc oxide. Colloids Surfaces A Physicochem. Eng. Asp. 2014, 457, 263–274. [Google Scholar] [CrossRef]
- Li, M.; Nan, L.; Liang, C.; Sun, Z.; Yang, L.; Yang, K. Antibacterial behavior and related mechanisms of martensitic Cu-bearing stainless steel evaluated by a mixed infection model of Escherichia coli and Staphylococcus aureus in vitro. J. Mater. Sci. Technol. 2021, 62, 139–147. [Google Scholar] [CrossRef]
- Zhou, Y.; Xia, M.; Ye, Y.; Hu, C. Antimicrobial ability of Cu-montmorillonite. Appl. Clay Sci. 2004, 27, 215–218. [Google Scholar] [CrossRef]
- Hu, C.-H.; Xia, M.-S. Adsorption and antibacterial effect of copper-exchanged montmorillonite on Escherichia coli K88. Appl. Clay Sci. 2006, 31, 180–184. [Google Scholar] [CrossRef]
- Tong, G.; Yulong, M.; Peng, G.; Zirong, X. Antibacterial effects of the Cu(II)-exchanged montmorillonite on Escherichia coli K88 and Salmonella choleraesuis. Veter. Microbiol. 2005, 105, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Martsouka, F.; Papagiannopoulos, K.; Gianni, E.; Papoulis, D.; Hatziantoniou, S. Antibacterial activity of pharmaceutical kaolin modified with copper. In Proceedings of the International Multidisciplinary Scientific GeoConference Surveying Geology and Mining Ecology Management (SGEM), Albena, Bulgaria, 6 July 2019; Volume 19, pp. 27–33. [Google Scholar]
- EDQM. European Pharmacopoeia, 10th ed.; Council of Europe: Strasbourg, France, 2020. [Google Scholar]
- Bekiari, V.; Panagopoulos, G.; Papoulis, D.; Panagiotaras, D. Use of halloysite nanotubes to reduce ammonium concentration in water and wastewaters. Mater. Res. Innov. 2016, 21, 313–319. [Google Scholar] [CrossRef]
- DIFFRAC plus EVA12® Software (Bruker-AXS). Available online: https://www.bruker.com/content/bruker/int/en/products-and-solutions/diffractometers-and-scattering-systems/x-ray-diffractometers/diffrac-suite-software/diffrac-eva.html (accessed on 28 February 2021).
- Aroke, U.; El-Nafaty, U. XRF, XRD and FTIR properties and characterization of HDTMA-Br surface modified organokaolinite clay. Int. J. Emerg. Technol. Adv. Eng. 2014, 4, 817–825. [Google Scholar]
- Jou, S.K.; Malek, N.A.N.N. Characterization and antibacterial activity of chlorhexidine loaded silver-kaolinite. Appl. Clay Sci. 2016, 127, 1–9. [Google Scholar] [CrossRef]
- Biesinger, M.C. Advanced analysis of copper X-ray photoelectron spectra. Surf. Interface Anal. 2017, 49, 1325–1334. [Google Scholar] [CrossRef]
- Krumpolec, R.; Homola, T.; Cameron, D.C.; Humlíček, J.; Caha, O.; Kuldová, K.; Zazpe, R.; Přikryl, J.; Macak, J.M. Structural and Optical Properties of Luminescent Copper(I) Chloride Thin Films Deposited by Sequentially Pulsed Chemical Vapour Deposition. Coatings 2018, 8, 369. [Google Scholar] [CrossRef]
- Martinvosshage, D.; Chowdari, B.V.R. XPS studies on (PEO)nLiClO4 and (PEO)nCu(CLO4)2 polymer electrolytes. J. Electrochem. Soc. 1995, 142, 1442–1446. [Google Scholar] [CrossRef]
- Winiarski, J.; Tylus, W.; Winiarska, K.; Szczygieł, I.; Szczygieł, B. XPS and FT-IR Characterization of Selected Synthetic Corrosion Products of Zinc Expected in Neutral Environment Containing Chloride Ions. J. Spectrosc. 2018, 2018, 2079278. [Google Scholar] [CrossRef]
- Dake, L.S.; Baer, D.R.; Zachara, J.M. Auger parameter measurements of zinc compounds relevant to zinc transport in the environment. Surf. Interface Anal. 1989, 14, 71–75. [Google Scholar] [CrossRef]
- Holešová, S.; Samlíková, M.; Pazdziora, E.; Valášková, M. Antibacterial activity of organomontmorillonites and organovermiculites prepared using chlorhexidine diacetate. Appl. Clay Sci. 2013, 83, 17–23. [Google Scholar] [CrossRef]
- Malachová, K.; Praus, P.; Rybková, Z.; Kozák, O. Antibacterial and antifungal activities of silver, copper and zinc montmorillonites. Appl. Clay Sci. 2011, 53, 642–645. [Google Scholar] [CrossRef]
- Song, J.; Li, Y.L.; Hu, C.H. Effects of copper-exchanged montmorillonite, as alternative to antibiotic, on diarrhea, intestinal permeability and proinflammatory cytokine of weanling pigs. Appl. Clay Sci. 2013, 52–55. [Google Scholar] [CrossRef]
- Özdemir, G.; Limoncu, M.H.; Yapar, S. The antibacterial effect of heavy metal and cetylpridinium-exchanged montmorillonites. Appl. Clay Sci. 2010, 48, 319–323. [Google Scholar] [CrossRef]
- Tan, S.-Z.; Zhang, K.-H.; Zhang, L.-L.; Xie, Y.-S.; Liu, Y.-L. Preparation and Characterization of the Antibacterial Zn2+ or/and Ce3+ Loaded Montmorillonites. Chin. J. Chem. 2008, 26, 865–869. [Google Scholar] [CrossRef]
- Herrera, P.; Burghardt, R.C.; Phillips, T.D. Adsorption of Salmonella enteritidis bycetylpyridinium-exchanged montmorillonite clays. Vet. Microbiol. 2000, 74, 259–272. [Google Scholar] [CrossRef]


| Test Organisms | |||||
|---|---|---|---|---|---|
| Gram-positive bacteria | St. aureus ATCC 6538 | St. epidermitis ATCC 12228 | Kokuria rhizophilia ATCC 9341 | ||
| Gram-negative bacteria | Enterobacteria | Pseudomonas | |||
| E. coli ATCC 8739 | Enterbac. Gergoviae ATCC 33028 | Pseudom. aeruginosa ATCC 9027 | Burkhold. cepacian In house | Pseudom. luteola ATCC 43330 | |
| Yeasts | Candida albicans ATCC 10231 | Molds | Aspergillus brasiliensis ATCC 16404 | Penicillium aurantiogriseum ATCC 16025 | |
| Criteria A | ||||
|---|---|---|---|---|
| Time | 2nd Day | 7th Day | 14th Day | 28th Day |
| Bacteria | 2 | 3 | - | NI |
| Yeasts | - | - | 2 | NI |
| Molds | - | - | 2 | NI |
| Criteria B | ||||
| Time | 2nd Day | 7th Day | 14th Day | 28th Day |
| Bacteria | - | - | 3 | NI |
| Yeasts | - | - | 1 | NI |
| Molds | - | - | 1 | NI |
| Route of Administration | Total Aerobic Microbial Count (cfu/g or cfu/mL) | Total Combined Yeasts/Molds Count (cfu/g or cfu/mL) | Specified Microorganism |
|---|---|---|---|
| Cutaneous use | 102 | 101 | Absence of Staphylococcus aureus (1 g or 1 mL) Absence of Pseudomonasaeruginosa (1 g or 1 mL) |
| Oxide | K | CuK | ZnK | T | CuT | ZnT |
|---|---|---|---|---|---|---|
| SiO2 | 44.58 | 42.79 | 45.52 | 64.71 | 60.62 | 56.21 |
| Al2O3 | 35.88 | 33.37 | 35.6 | 0.53 | 0.59 | 0.57 |
| Fe2O3 | 0.82 | 0.90 | 0.84 | 3.89 | 3.85 | 3.63 |
| MnO | 0.01 | 0.01 | 0.01 | 0.02 | 0.01 | 0.02 |
| MgO | 0.39 | 0.38 | 0.37 | 27.62 | 27.41 | 27.04 |
| CaO | 0.28 | 0.23 | 0.25 | 0.38 | 0.28 | 0.36 |
| Na2O | 0.15 | 0.13 | 0.47 | 0.07 | 0.08 | 0.29 |
| K2O | 2.48 | 2.40 | 2.51 | BDL | 0.00 | BDL |
| TiO2 | 0.03 | BDL | 0.03 | BDL | BDL | BDL |
| P2O5 | 0.12 | 0.10 | 0.1 | BDL | 0.01 | BDL |
| LOI | 11.32 | 12.08 | 11.48 | 5.09 | 6.46 | 6.59 |
| Sum | 96.06 | 92.39 | 97.18 | 102.31 | 99.31 | 94.71 |
| Element | K | CuK | ZnK | T | CuT | ZnT |
|---|---|---|---|---|---|---|
| Sc | 10 | 1866 | 11 | 8 | 1638 | 9 |
| Cr | 7 | 6 | 8 | 67 | 135 | 77 |
| Co | BDL | BDL | BDL | BDL | BDL | BDL |
| Cu | BDL | 23467 | BDL | BDL | 20603 | BDL |
| Zn | 35 | 1916 | 23148 | 25 | 1715 | 47365 |
| Sr | 108 | 121 | 106 | 6 | 15 | 7 |
| Y | 74 | 93 | 58 | 36 | 48 | 30 |
| Ba | 218 | 8994 | 210 | 31 | 7717 | 44 |
| Hf | 7 | 26 | 6 | 5 | 24 | 6 |
| Pb | BDL | 77 | BDL | BDL | 55 | BDL |
| Sample | Cu2+ | Cu1+ (CuCl) |
|---|---|---|
| CuK | 49 | 51 |
| CuT | 86 | 14 |
| Paste | Parameter | Result (cfu/g) | Limits (cfu/g) |
|---|---|---|---|
| Kp | Total Aerobic Microbial Count | >3.0 × 103 | <1.0 × 102 |
| Molds & Yeasts | <10 | <10 | |
| Tp | Total Aerobic Microbial Count | >3.0 × 103 | <1.0 × 102 |
| Molds & Yeasts | <10 | <10 | |
| CuTp | Total Aerobic Microbial Count | >3.0 × 103 | <1.0 × 102 |
| Molds & Yeasts | 4.0 × 101 | <10 | |
| ZnKp | Total Aerobic Microbial Count | >3.0 × 103 | <1.0 × 102 |
| Molds & Yeasts | >3.0 × 102 | <10 |
| Paste | Parameter | Sterility Control | Inoculation | 0 Time | 2nd Day | 7th Day | 14th Day | 28th Day |
|---|---|---|---|---|---|---|---|---|
| CuKp | Bacteria | <10 | 6.2 × 106 | 6.1 × 106 | <100 | <10 | <10 | <10 |
| Molds | <10 | 3.4 × 105 | 3.3 × 105 | 2.0 × 104 | 1.5 × 104 | 2.0 × 104 | <10 | |
| Yeasts | <10 | 4.6 × 105 | 4.5 × 105 | 1.8 × 105 | 4.8 × 105 | 8.2 × 105 | <10 | |
| ZnTp | Bacteria | <10 | 6.2 × 106 | 5.4 × 106 | 2.4 × 103 | 3.1 × 102 | 3.0 × 103 | 2.6 × 105 |
| Molds | <10 | 3.4 × 105 | 3.3 × 105 | 4.8 × 105 | 4.5 × 105 | 5.2 × 105 | 6.8 × 104 | |
| Yeasts | <10 | 4.6 × 105 | 4.1 × 105 | <100 | <10 | <10 | <10 | |
| PHKp | Bacteria | <10 | 6.2 × 106 | 5.5 × 106 | <100 | <10 | <10 | <10 |
| Molds | <10 | 3.4 × 105 | 3.1 × 105 | 4.1 × 104 | 4.1 × 102 | 3.2 × 102 | <10 | |
| Yeasts | <10 | 4.6 × 105 | 4.1 × 105 | 1.1 × 103 | 9.0 × 102 | 8.2 × 102 | <10 | |
| PHTp | Bacteria | <10 | 6.2 × 106 | 5.7 × 106 | <100 | <10 | <10 | <10 |
| Molds | <10 | 3.4 × 105 | 3.1 × 105 | 6.9 × 103 | 3.6 × 102 | <10 | <10 | |
| Yeasts | <10 | 4.6 × 105 | 4.2 × 105 | <100 | <10 | <10 | <10 |
| Paste | Log Reduction | 2nd Day | 7th Day | 14th Day | 28th Day | Criterion Α | Criterion B | Test Result |
|---|---|---|---|---|---|---|---|---|
| CuKp | Bacteria | 6.79 | 6.79 | 6.79 | 6.79 | ✓ | Failed, does not meet the relevant A/B criteria | |
| Molds | 1.23 | 1.37 | 1.23 | 4.63 | ✓ | |||
| Yeasts | 0.41 | 0.95 | 0.75 | 4.66 | ||||
| ZnTp | Bacteria | 3.82 | 4.30 | 1.92 | 1.37 | Failed, does not meet the relevant A/B criteria | ||
| Molds | −0.15 | −0.12 | −0.18 | 0.70 | ||||
| Yeasts | 4.06 | 4.06 | 4.06 | 4.06 | ✓ | |||
| PHKp | Bacteria | 5.79 | 5.79 | 5.79 | 5.79 | ✓ | Satisfactory, meet the relevant A criteria | |
| Molds | 0.92 | 2.92 | 3.03 | 3.53 | ✓ | |||
| Yeasts | 2.64 | 2.71 | 2.75 | 4.66 | ✓ | |||
| PHTp | Bacteria | 5.79 | 5.79 | 5.79 | 5.79 | ✓ | Satisfactory, meet the relevant A criteria | |
| Molds | 1.69 | 2.06 | 4.55 | 4.55 | ✓ | |||
| Yeasts | 4.06 | 4.06 | 4.06 | 4.06 | ✓ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martsouka, F.; Papagiannopoulos, K.; Hatziantoniou, S.; Barlog, M.; Lagiopoulos, G.; Tekerlekopoulou, A.G.; Papoulis, D. Evaluation of the Antimicrobial Protection of Pharmaceutical Kaolin and Talc Modified with Copper and Zinc. Materials 2021, 14, 1173. https://doi.org/10.3390/ma14051173
Martsouka F, Papagiannopoulos K, Hatziantoniou S, Barlog M, Lagiopoulos G, Tekerlekopoulou AG, Papoulis D. Evaluation of the Antimicrobial Protection of Pharmaceutical Kaolin and Talc Modified with Copper and Zinc. Materials. 2021; 14(5):1173. https://doi.org/10.3390/ma14051173
Chicago/Turabian StyleMartsouka, Fotini, Konstantinos Papagiannopoulos, Sophia Hatziantoniou, Martin Barlog, Giorgos Lagiopoulos, Athanasia G. Tekerlekopoulou, and Dimitrios Papoulis. 2021. "Evaluation of the Antimicrobial Protection of Pharmaceutical Kaolin and Talc Modified with Copper and Zinc" Materials 14, no. 5: 1173. https://doi.org/10.3390/ma14051173
APA StyleMartsouka, F., Papagiannopoulos, K., Hatziantoniou, S., Barlog, M., Lagiopoulos, G., Tekerlekopoulou, A. G., & Papoulis, D. (2021). Evaluation of the Antimicrobial Protection of Pharmaceutical Kaolin and Talc Modified with Copper and Zinc. Materials, 14(5), 1173. https://doi.org/10.3390/ma14051173










