Physicochemical Properties of Biobutanol as an Advanced Biofuel
Abstract
1. Introduction
2. Materials and Methods
- Ex: x% (V/V) ethanol and (100–x)% (V/V) gasoline (e.g., E5).
- BUT x:x% (V/V) n–butanol and (100–x)% (V/V) gasoline (e.g., BUT 5).
- iBUT x:x% (V/V) i–butanol and (100–x)% (V/V) gasoline (e.g., iBUT 5).
- BUT x + MTBE y: x% (V/V) n–butanol, y% (V/V) methyl tert–butyl ether, and (100–x–y)% (V/V) gasoline (e.g., BUT 5 + MTBE 10).
- BUT x + ETBE y: x% (V/V) n–butanol, y% (V/V) ethyl tert-butyl ether, and (100–x–y)% (V/V) gasoline (e.g., BUT 5 + ETBE 10).
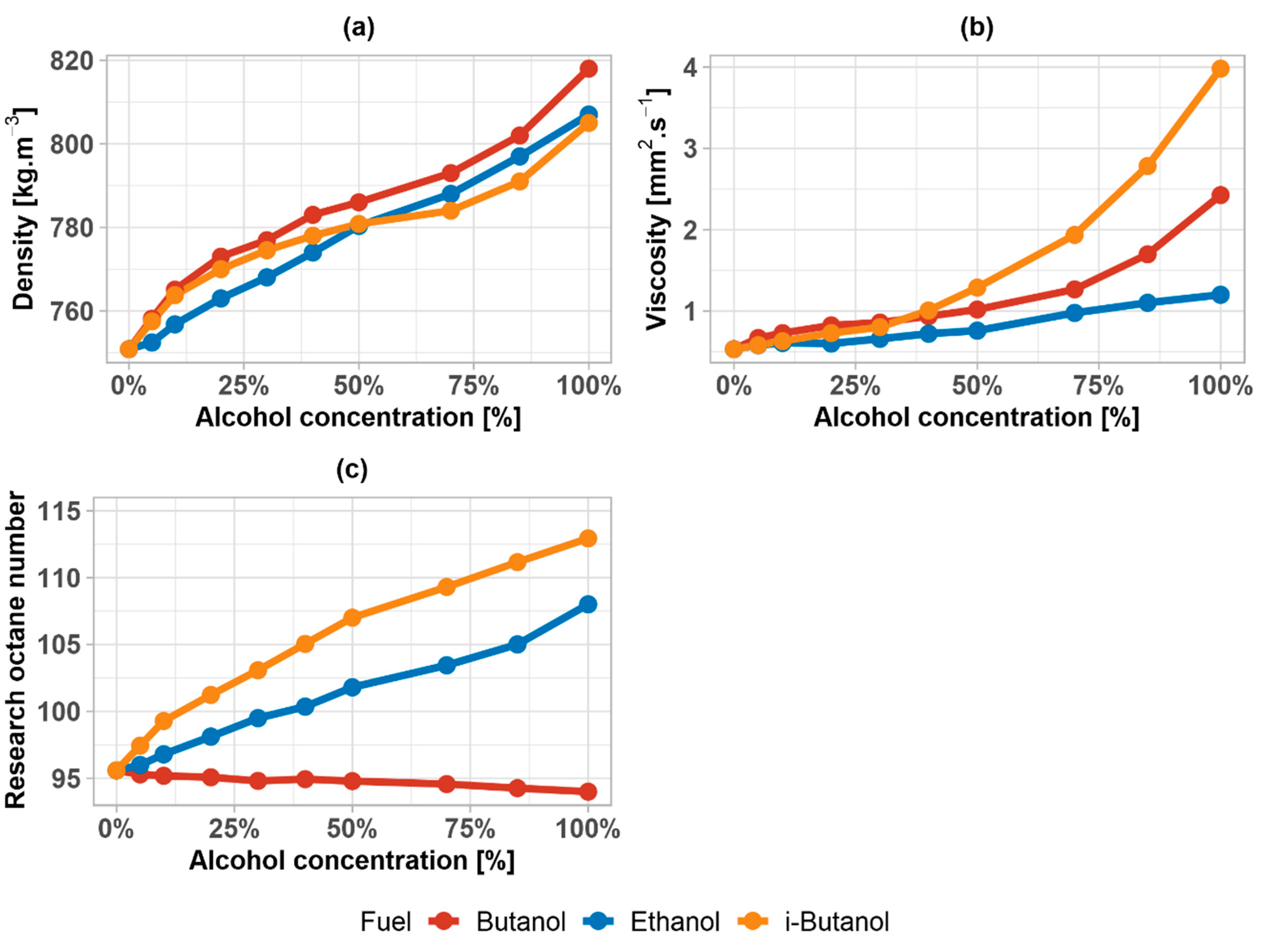
3. Results
3.1. Fuel Parameters
3.2. Distillation Properties
- Start of distillation:
- Ten percent point:
- Fifty percent point:
- End of distillation curve:
- End of distillation temperature:
- E0 (pure gasoline), E3, E5, E8, E10, E15, E20, and E25.
- BUT 0 (pure gasoline), BUT 3, BUT 5, BUT 8, BUT 10, BUT 15, BUT 20, and BUT 25.
- iBUT 0 (pure gasoline), iBUT 3, iBUT 5, iBUT 8, iBUT 10, iBUT 15, BUT 20, and iBUT 25.
3.3. Vapour Pressure
3.4. Water Stability
3.5. Gas Chromatography Analysis
4. Discussion
5. Conclusions
- Butanol has a higher kinematic viscosity value than gasoline and ethanol. The viscosity of the mixture increases with the addition of butanol.
- The octane number of mixtures is mainly influenced by the octane numbers of the individual alcohols. Butanol reduces the octane number in gasoline, but the decrease is not so significant.
- Ethanol with gasoline creates an azeotrope, which increases the vapor pressure of formed mixture. Such a phenomenon does not occur with butanol.
- Ethers can have a positive effect on the increase of vapor pressure. It was verified that ETBE has more favorable effects than MTBE.
- Ethanol mainly affects the first half of the distillation curve and especially the temperature T50. Butanol mainly affects the second half of the distillation curve, which is also reflected in the values of E100 and E150.
- The solubility of water in butanol and ethanol mixtures is similar. However, butanol is more stable at low temperatures. Water is only slightly soluble in MTBE and ETBE.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ucal, M.; Xydis, G. Multidirectional Relationship between Energy Resources, Climate Changes and Sustainable Development: Technoeconomic Analysis. Sustain. Cities Soc. 2020, 60, 102210. [Google Scholar] [CrossRef]
- Southworth, K. Corporate Voluntary Action: A Valuable but Incomplete Solution to Climate Change and Energy Security Challenges. Policy Soc. 2009, 27, 329–350. [Google Scholar] [CrossRef]
- Peterka, B.; Pexa, M.; Mader, D.; Kotek, M. Comparison of Exhaust Emissions and Fuel Consumption of Small Combustion Engine of Portable Generator Operated on Petrol and Biobutanol. Agronomy Res. 2017, 15, 1162–1169. [Google Scholar]
- Elfasakhany, A. Investigations on Performance and Pollutant Emissions of Spark-Ignition Engines Fueled with n-Butanol–, Isobutanol–, Ethanol–, Methanol–, and Acetone–Gasoline Blends: A Comparative Study. Renew. Sustain. Energy Rev. 2017, 71, 404–413. [Google Scholar] [CrossRef]
- Elfasakhany, A. Experimental Study on Emissions and Performance of an Internal Combustion Engine Fueled with Gasoline and Gasoline/n-Butanol Blends. Energy Convers. Manag. 2014, 88, 277–283. [Google Scholar] [CrossRef]
- Zhao, L.; Wang, D.; Qi, W. Particulate Matter (PM) Emissions and Performance of Bio-Butanol-Methanol-Gasoline Blends Coupled with Air Dilution in SI Engines. J. Aerosol Sci. 2020, 145, 105546. [Google Scholar] [CrossRef]
- Feng, R.; Fu, J.; Yang, J.; Wang, Y.; Li, Y.; Deng, B.; Liu, J.; Zhang, D. Combustion and Emissions Study on Motorcycle Engine Fueled with Butanol-Gasoline Blend. Renew. Energ. 2015, 81, 113–122. [Google Scholar] [CrossRef]
- Qureshi, N.; Meagher, M.M.; Huang, J.; Hutkins, R.W. Acetone Butanol Ethanol (ABE) Recovery by Pervaporation Using Silicalite–Silicone Composite Membrane from Fed-Batch Reactor of Clostridium Acetobutylicum. J. Membr. Sci. 2001, 187, 93–102. [Google Scholar] [CrossRef]
- Shah, R. A Review on Generation of Liquid Bio-Fuels from Renewable Resources for Energy Security of India. Indira Manage Rev 2018, 12, 66–75. [Google Scholar]
- Timilsina, G. Potential Future Impacts of Increased Biofuels Use. Available online: https://blogs.worldbank.org/developmenttalk/potential-future-impacts-of-increased-biofuels-use (accessed on 1 January 2021).
- Bórawski, P.; Bełdycka-Bórawska, A.; Szymańska, E.J.; Jankowski, K.J.; Dubis, B.; Dunn, J.W. Development of Renewable Energy Sources Market and Biofuels in The European Union. J. Clean. Prod. 2019, 228, 467–484. [Google Scholar] [CrossRef]
- Kolesinska, B.; Fraczyk, J.; Binczarski, M.; Modelska, M.; Berlowska, J.; Dziugan, P.; Antolak, H.; Kaminski, Z.J.; Witonska, I.A.; Kregiel, D. Butanol Synthesis Routes for Biofuel Production: Trends and Perspectives. Materials 2019, 12, 350. [Google Scholar] [CrossRef] [PubMed]
- Ziolkowska, J.R. Biofuels technologies. In Biofuels for a More Sustainable Future; Elsevier: Amsterdam, The Netherland, 2020; pp. 1–19. ISBN 978-0-12-815581-3. [Google Scholar]
- Saini, J.K.; Saini, R.; Tewari, L. Lignocellulosic Agriculture Wastes as Biomass Feedstocks for Second-Generation Bioethanol Production: Concepts and Recent Developments. 3 Biotech 2015, 5, 337–353. [Google Scholar] [CrossRef]
- Chen, H. Biotechnology of Lignocellulose: Theory and Practice; Springer Netherlands: Dordrecht, The Netherland, 2014; ISBN 978-94-007-6897-0. [Google Scholar]
- Bajpai, P. Structure of Lignocellulosic Biomass. In Pretreatment of Lignocellulosic Biomass for Biofuel Production; SpringerBriefs in Molecular Science; Springer Singapore: Singapore, 2016; pp. 7–12. ISBN 978-981-10-0686-9. [Google Scholar]
- Sharma, B.; Larroche, C.; Dussap, C.-G. Comprehensive Assessment of 2G Bioethanol Production. Bioresour. Technol. 2020, 313, 123630. [Google Scholar] [CrossRef]
- Minteer, S.D. 11 - Biochemical production of other bioalcohols: biomethanol, biopropanol, bioglycerol, and bioethylene glycol. In Handbook of Biofuels Production; Luque, R., Campelo, J., Clark, J., Eds.; Woodhead Publishing Series in Energy; Woodhead Publishing: Cambridge, UK, 2011; pp. 258–265. ISBN 978-1-84569-679-5. [Google Scholar]
- Kim, T.-W.; Kim, M.-J.; Chae, H.-J.; Ha, K.-S.; Kim, C.-U. Ordered Mesoporous Carbon Supported Uniform Rhodium Nanoparticles as Catalysts for Higher Alcohol Synthesis from Syngas. Fuel 2015, 160, 393–403. [Google Scholar] [CrossRef]
- Slaa, J.C.; van Ommen, J.G.; Ross, J.R.H. The Synthesis of Higher Alcohols Using Modified Cu/ZnO/Al2O3 Catalysts. Catalysis Today 1992, 15, 129–148. [Google Scholar] [CrossRef]
- Zhang, H.; Chu, W.; Xu, H.; Zhou, J. Plasma-Assisted Preparation of Fe–Cu Bimetal Catalyst for Higher Alcohols Synthesis from Carbon Monoxide Hydrogenation. Fuel 2010, 89, 3127–3131. [Google Scholar] [CrossRef]
- Aslam, W.; Ahmed, M.H.; Qui, T.; Konarova, M. Syngas to Higher Alcohols Synthesis over 3D Printed KMoCo/ZSM5 Monolith. Chem. Eng. J. Adv. 2020, 3, 100024. [Google Scholar] [CrossRef]
- Luk, H.T.; Mondelli, C.; Ferré, D.C.; Stewart, J.A.; Pérez-Ramírez, J. Status and Prospects in Higher Alcohols Synthesis from Syngas. Chem. Soc. Rev. 2017, 46, 1358–1426. [Google Scholar] [CrossRef] [PubMed]
- Worden, R.M.; Grethlein, A.J.; Jain, M.K.; Datta, R. Production of Butanol and Ethanol from Synthesis Gas via Fermentation. Fuel 1991, 70, 615–619. [Google Scholar] [CrossRef]
- Phillips, J.R.; Clausen, E.C.; Gaddy, J.L. Synthesis Gas as Substrate for the Biological Production of Fuels and Chemicals. Appl. Biochem. Biotechnol. 1994, 45–46, 145–157. [Google Scholar] [CrossRef]
- Vega, J.L.; Clausen, E.C.; Gaddy, J.L. Design of Bioreactors for Coal Synthesis Gas Fermentations. Resour. Conserv. Recycl. 1990, 3, 149–160. [Google Scholar] [CrossRef]
- Wilkins, M.R.; Atiyeh, H.K. Microbial Production of Ethanol from Carbon Monoxide. Curr. Opin. Biotechnol. 2011, 22, 326–330. [Google Scholar] [CrossRef] [PubMed]
- Benevenuti, C.; Botelho, A.; Ribeiro, R.; Branco, M.; Pereira, A.; Vieira, A.C.; Ferreira, T.; Amaral, P. Experimental Design to Improve Cell Growth and Ethanol Production in Syngas Fermentation by Clostridium Carboxidivorans. Catalysts 2020, 10, 59. [Google Scholar] [CrossRef]
- Liberato, V.; Benevenuti, C.; Coelho, F.; Botelho, A.; Amaral, P.; Pereira, N.; Ferreira, T. Clostridium Sp. as Bio-Catalyst for Fuels and Chemicals Production in a Biorefinery Context. Catalysts 2019, 9, 962. [Google Scholar] [CrossRef]
- Henstra, A.M.; Sipma, J.; Rinzema, A.; Stams, A.J.M. Microbiology of Synthesis Gas Fermentation for Biofuel Production. Curr. Opin. Biotechnol. 2007, 18, 200–206. [Google Scholar] [CrossRef]
- Munasinghe, P.C.; Khanal, S.K. Biomass-Derived Syngas Fermentation into Biofuels: Opportunities and Challenges. Bioresour. Technol. 2010, 101, 5013–5022. [Google Scholar] [CrossRef]
- Tirado-Acevedo, O.; Chinn, M.S.; Grunden, A.M. Chapter 2—Production of Biofuels from Synthesis Gas Using Microbial Catalysts. In Advances in Applied Microbiology; Academic Press: Cambridge, MA, USA, 2010; Volume 70, pp. 57–92. [Google Scholar]
- Abubackar, H.N.; Veiga, M.C.; Kennes, C. Carbon Monoxide Fermentation to Ethanol by Clostridium Autoethanogenum in a Bioreactor with No Accumulation of Acetic Acid. Bioresour. Technol. 2015, 186, 122–127. [Google Scholar] [CrossRef]
- Daniell, J.; Köpke, M.; Simpson, S.D. Commercial Biomass Syngas Fermentation. Energies 2012, 5, 5372–5417. [Google Scholar] [CrossRef]
- Liu, K.; Atiyeh, H.K.; Tanner, R.S.; Wilkins, M.R.; Huhnke, R.L. Fermentative Production of Ethanol from Syngas Using Novel Moderately Alkaliphilic Strains of Alkalibaculum Bacchi. Bioresour. Technol. 2012, 104, 336–341. [Google Scholar] [CrossRef]
- van Steen, E.; Claeys, M. Fischer-Tropsch Catalysts for the Biomass-to-Liquid (BTL)-Process. Chem. Eng. Technol. 2008, 31, 655–666. [Google Scholar] [CrossRef]
- Xu, D.; Tree, D.R.; Lewis, R.S. The Effects of Syngas Impurities on Syngas Fermentation to Liquid Fuels. Biomass Bioenergy 2011, 35, 2690–2696. [Google Scholar] [CrossRef]
- Shapovalov, O.I.; Ashkinazi, L.A. Biobutanol: Biofuel of Second Generation. Russ. J. Appl. Chem. 2008, 81, 2232–2236. [Google Scholar] [CrossRef]
- Dukulis, I.; Pirs, V.; Jesko, Z.; Birkavs, A.; Birzietis, G. Development of Methods for Testing Automobiles Operating on Biofuels. In Proceedings of the 8th International Scientific Conference on Project Management in the Baltic Countries, Riga, Latvia, 25–26 April 2019; University of Latvia: Jelgava, Latvia, 2009; p. 8. [Google Scholar]
- Šebor, G.; Pospíšil, M.; Maxa, D. Využití kapalných biopaliv pro pohon motorových vozidel. Available online: https://biom.cz/cz/knihovna/vyuziti-kapalnych-biopaliv-pro-pohon-motorovych-vozidel (accessed on 31 July 2020).
- Mužíková, Z.; Káňa, J.; Pospíšil, M.; Šebor, G. Fyzikálně-chemické vlastnoti butanol-benzínových směsí. Chemické listy 2009, 106, 7. [Google Scholar]
- Demirbas, A. Competitive Liquid Biofuels from Biomass. Appl. Energy 2011, 88, 17–28. [Google Scholar] [CrossRef]
- Atmanlı, A.; Yüksel, B.; Ileri, E. Experimental Investigation of the Effect of Diesel–Cotton Oil–n-Butanol Ternary Blends on Phase Stability, Engine Performance and Exhaust Emission Parameters in a Diesel Engine. Fuel 2013, 109, 503–511. [Google Scholar]
- Atmanlı, A.; Ileri, E.; Yuksel, B.; Yilmaz, N. Extensive Analyses of Diesel–Vegetable Oil–n-Butanol Ternary Blends in a Diesel Engine. Appl. Energy 2015, 145, 155–162. [Google Scholar] [CrossRef]
- Atmanlı, A.; Ileri, E.; Yüksel, B. Effects of Higher Ratios of N-Butanol Addition to Diesel–Vegetable Oil Blends on Performance and Exhaust Emissions of a Diesel Engine. J. Energy Inst. 2015, 88, 209–220. [Google Scholar] [CrossRef]
- Dernotte, J.; Mounaim-Rousselle, C.; Halter, F.; Seers, P. Evaluation of Butanol–Gasoline Blends in a Port Fuel-Injection, Spark-Ignition Engine. Oil Gas Sci. Technol. Rev. IFP 2010, 65, 345–351. [Google Scholar] [CrossRef]
- Rakopoulos, D.C.; Rakopoulos, C.D.; Giakoumis, E.G.; Dimaratos, A.M.; Kyritsis, D.C. Effects of Butanol–Diesel Fuel Blends on the Performance and Emissions of a High-Speed DI Diesel Engine. Energy Convers. Manag. 2010, 51, 1989–1997. [Google Scholar] [CrossRef]
- Mack, J.H.; Schuler, D.; Butt, R.H.; Dibble, R.W. Experimental Investigation of Butanol Isomer Combustion in Homogeneous Charge Compression Ignition (HCCI) Engines. Appl. Energy 2016, 165, 612–626. [Google Scholar] [CrossRef]
- Higashide, W.; Li, Y.; Yang, Y.; Liao, J.C. Metabolic Engineering of Clostridium Cellulolyticum for Production of Isobutanol from Cellulose. Appl. Environ. Microbiol. 2011, 77, 2727–2733. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Kim, J.K.; Ahn, J.-O.; Song, Y.-H.; Shin, C.-S.; Park, Y.-C.; Kim, K.H. Isobutanol Production from Empty Fruit Bunches. Renew. Energy 2020, 157, 1124–1130. [Google Scholar] [CrossRef]
- Jung, H.-M.; Lee, J.Y.; Lee, J.-H.; Oh, M.-K. Improved Production of Isobutanol in Pervaporation-Coupled Bioreactor Using Sugarcane Bagasse Hydrolysate in Engineered Enterobacter Aerogenes. Bioresour. Technol. 2018, 259, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Mariano, A.P.; Tomasella, R.C.; Martino, C.D.; Morais, E.B.; Filho, R.M.; Seleghim, M.H.R.; Contiero, J.; Tornisielo, S.M.T.; de Angelis, D.D. Aerobic Biodegradation of Butanol and Diesel Oil Blends. Afr. J. Biotechnol. 2010, 9, 7094–7101. [Google Scholar] [CrossRef]
- Huzir, N.M.; Aziz, M.M.A.; Ismail, S.B.; Abdullah, B.; Mahmood, N.A.N.; Umor, N.A.; Syed Muhammad, S.A.F. Agro-Industrial Waste to Biobutanol Production: Eco-Friendly Biofuels for next Generation. Renew. Sustain. Energy Rev. 2018, 94, 476–485. [Google Scholar] [CrossRef]
- FuelsEurope Statistical Report 2018; FuelsEurope: Brussels, Belgium, 2018.
- Eurostat Supply, Transformation and Consumption of Renewable Energies. Available online: http://r6d.cn/bbj0v (accessed on 4 October 2020).
- EN 228:2008 - Automotive Fuels - Unleaded Petrol - Requirements and Test Methods; European Committee for Standardization: Brussels, Belgium, 2008.
- The European Parliament. Directive 2009/30/EC of the European Parliament and of the Council of 23 April 2009. Off. J. Eur. Union 2009, L 140, 88–133. [Google Scholar]
- The European Union Next Generation Bio-Butanol | ButaNexT Project | H2020. Available online: https://cordis.europa.eu/project/id/640462 (accessed on 3 January 2021).
- Pugazhendhi, A.; Mathimani, T.; Varjani, S.; Rene, E.R.; Kumar, G.; Kim, S.-H.; Ponnusamy, V.K.; Yoon, J.-J. Biobutanol as a Promising Liquid Fuel for the Future - Recent Updates and Perspectives. Fuel 2019, 253, 637–646. [Google Scholar] [CrossRef]
- Jin, C.; Yao, M.; Liu, H.; Lee, C.F.; Ji, J. Progress in the Production and Application of N-Butanol as a Biofuel. Renew. Sustain. Energy Rev. 2011, 15, 4080–4106. [Google Scholar] [CrossRef]
- Bankar, S.B.; Survase, S.A.; Ojamo, H.; Granström, T. Biobutanol: The Outlook of an Academic and Industrialist. RSC Adv. 2013, 3, 24734. [Google Scholar] [CrossRef]
- He, Z.; Liu, G.; Li, Z.; Jiang, C.; Qian, Y.; Lu, X. Comparison of Four Butanol Isomers Blended with Diesel on Particulate Matter Emissions in a Common Rail Diesel Engine. J. Aerosol Sci. 2019, 137, 105434. [Google Scholar] [CrossRef]
- Anderson, J.E.; Kramer, U.; Mueller, S.A.; Wallington, T.J. Octane Numbers of Ethanol− and Methanol−Gasoline Blends Estimated from Molar Concentrations. Energy Fuels 2010, 24, 6576–6585. [Google Scholar] [CrossRef]
- EN 15376:2014 - Automotive Fuels - Ethanol as a Blending Component for Petrol - Requirements and Test Methods; European Committee for Standardization: Brussels, Belgium, 2014.
- Ardrey, R.E. Liquid Chromatography-Mass Spectrometry: An Introduction; Analytical techniques in the sciences; J. Wiley: New York, NY, USA, 2003; ISBN 978-0-471-49799-8. [Google Scholar]
- ISO/IEC Guide 98-3:2008 Uncertainty of Measurement — Part 3: Guide to the Expression of Uncertainty in Measurement (GUM:1995); International Organization for Standardization: Geneva, Switzland, 2008.
- ISO 3675:1998 – Crude Petroleum and Liquid Petroleum Products – Laboratory Determination of Density – Hydrometer Method, 3rd ed.; International Organization for Standardization: Geneva, Switzland, 1998.
- ISO 3104:1994 – Petroleum Products – Transparent and Opaque Liquids – Determination of Kinematic Viscosity and Calculation of Dynamic Viscosity; International Organization for Standardization: Geneva, Switzland, 1994.
- ISO 5164:2014 – Petroleum Products – Determination of Knock Characteristics of Motor Fuels – Research Method; International Organization for Standardization: Geneva, Switzland, 2014.
- ISO 3007:1999 – Petroleum Products and Crude Petroleum – Determination of Vapour Pressure – Reid Method; International Organization for Standardization: Geneva, Switzland, 1999.
- ASTM D6422-99 - Test Method for Water Tolerance (Phase Separation) of Gasoline-Alcohol Blends; ASTM International: West Conshohocken, PA, USA, 1999.
- ISO 3405:2011 – Petroleum Products – Determination of Distillation Characteristics at Atmospheric Pressure; International Organization for Standardization: Geneva, Switzland, 2011.
- ISO 22854:2016 - Liquid Petroleum Products - Determination of Hydrocarbon Types and Oxygenates in Automotive-Motor Gasoline and in Ethanol (E85) Automotive Fuel - Multidimensional Gas Chromatography Method; International Organization for Standardization: Geneva, Switzland, 2016.
- BS EN 15293:2018 - Automotive Fuels. Automotive Ethanol (E85) Fuel. Requirements and Test Methods; British Standards Institution: London, UK, 2018.
- Sarathy, S.M.; Vranckx, S.; Yasunaga, K.; Mehl, M.; Oßwald, P.; Metcalfe, W.K.; Westbrook, C.K.; Pitz, W.J.; Kohse-Höinghaus, K.; Fernandes, R.X.; et al. A Comprehensive Chemical Kinetic Combustion Model for the Four Butanol Isomers. Combust. Flame 2012, 159, 2028–2055. [Google Scholar] [CrossRef]
- Shahbakhti, M.; Ghazimirsaied, A.; Audet, A.; Koch, C.R. Combustion Characteristics of Butanol / n-Heptane Blend Fuels in an HCCI Engine. In Proceedings of the 2010 Combustion Institute−Canadian SectionSpring Technical Meeting, Ottawa, ON, Canada, 9–12 May 2010; Carleton University of Ottawa: Ottawa, ON, Canada, 2010. [Google Scholar]
- Mariano, A.P.; Tomasella, R.C.; Di Martino, C.; Filho, R.M.; Seleghim, M.H.R.; Contiero, J.; de Angelis, D.D. Aerobic Biodegradation of Butanol and Gasoline Blends. Biomass Bioenergy 2009, 33, 1175–1181. [Google Scholar] [CrossRef]
- Zoldy, M.; Hollo, A.; Thernesz, A. Butanol as a Diesel Extender Option for Internal Combustion Engines; SAE Technical Paper No. 2010-01–0481; SAE: Warrendale, PA, USA, 2010; p. 8. [Google Scholar]
- da Silva, R.; Cataluña, R.; de Menezes, E.W.; Samios, D.; Piatnicki, C.M.S. Effect of Additives on the Antiknock Properties and Reid Vapor Pressure of Gasoline. Fuel 2005, 84, 951–959. [Google Scholar] [CrossRef]
- Kokkinos, N.C.; Nikolaou, N.; Psaroudakis, N.; Mertis, K.; Mitkidou, S.; Mitropoulos, A.C. Two-Step Conversion of LLCN Olefins to Strong Anti-Knocking Alcohol Mixtures Catalysed by Rh, Ru/TPPTS Complexes in Aqueous Media. Catal. Today 2015, 247, 132–138. [Google Scholar] [CrossRef]
- Demirbas, A.; Balubaid, M.A.; Basahel, A.M.; Ahmad, W.; Sheikh, M.H. Octane Rating of Gasoline and Octane Booster Additives. Pet. Sci. Technol. 2015, 33, 1190–1197. [Google Scholar] [CrossRef]
- Stratiev, D.; Kirilov, K. Opportunities for Gasoline Octane Increase by Use of Iron Containing Octane Booster. Pet. Coal 2009, 51, 244–248. [Google Scholar]
- Corrubia, J.A.; Capece, J.M.; Cernansky, N.P.; Miller, D.L.; Durrett, R.P.; Najt, P.M. RON and MON Chemical Kinetic Modeling Derived Correlations with Ignition Delay Time for Gasoline and Octane Boosting Additives. Combust. Flame 2020, 219, 359–372. [Google Scholar] [CrossRef]
- Lapuerta, M.; Ballesteros, R.; Barba, J. Strategies to Introduce N-Butanol in Gasoline Blends. Sustainability 2017, 9, 589. [Google Scholar] [CrossRef]
- Aghahossein Shirazi, S.; Abdollahipoor, B.; Martinson, J.; Windom, B.; Foust, T.D.; Reardon, K.F. Effects of Dual-Alcohol Gasoline Blends on Physiochemical Properties and Volatility Behavior. Fuel 2019, 252, 542–552. [Google Scholar] [CrossRef]
- Andersen, V.F.; Anderson, J.E.; Wallington, T.J.; Mueller, S.A.; Nielsen, O.J. Distillation Curves for Alcohol−Gasoline Blends. Energy Fuels 2010, 24, 2683–2691. [Google Scholar] [CrossRef]
- Amine, M.; Awad, E.N.; Barakat, Y. Volatility Criteria of Isomerate-Enriched Gasoline-Ethanol Blends. Egypt. J. Pet. 2020, 29, 227–233. [Google Scholar] [CrossRef]
- Castillo-Hernández, P.; Mendoza-Domínguez, A.; Caballero-Mata, P. Analysis of Physicochemical Properties of Mexican Gasoline and Diesel Reformulated with Ethanol. Ingeniería, investigación y tecnología 2012, 13, 293–306. [Google Scholar] [CrossRef]
- Aleme, H.G.; Costa, L.M.; Barbeira, P.J.S. Determination of Ethanol and Specific Gravity in Gasoline by Distillation Curves and Multivariate Analysis. Talanta 2009, 78, 1422–1428. [Google Scholar] [CrossRef] [PubMed]
- Aleme, H.G.; Costa, L.M.; Barbeira, P.J.S. Determination of Gasoline Origin by Distillation Curves and Multivariate Analysis. Fuel 2008, 87, 3664–3668. [Google Scholar] [CrossRef]
- Kosychova, L.; Žukauskaitė, A.; Butkutė, R.; Sinicyna, A.; Narmontas, G.; Kryževičius, Ž.; Dodonova, N.; Jonaitytė, R.; Jotauta, G. Experimental Determination of Distillation Curves of Alcohols/Gasoline Blends as Bio-Fuel for SI Engines. In Proceedings of the 23rd International Scientific-Technical Conference on Transport, Road-Building, Agricultural, Hoisting and Hauling Andmilitary Technics And Technologies (Trans & Motauto ′15), Varna, Bulgaria, 24–27 June 2015; Scientific-Technical Union of Mechanical Engineering: Sofia, Bulgaria, 2015; Volume 1. [Google Scholar]
- Landera, A.; Mac Dowell, N.; George, A. Development of Robust Models for the Prediction of Reid Vapor Pressure (RVP) in Fuel Blends and Their Application to Oxygenated Biofuels Using the SAFT-γ Approach. Fuel 2021, 283, 118624. [Google Scholar] [CrossRef]
- Gaspar, D.J.; Phillips, S.D.; Polikarpov, E.; Albrecht, K.O.; Jones, S.B.; George, A.; Landera, A.; Santosa, D.M.; Howe, D.T.; Baldwin, A.G.; et al. Measuring and Predicting the Vapor Pressure of Gasoline Containing Oxygenates. Fuel 2019, 243, 630–644. [Google Scholar] [CrossRef]
- Mužíková, Z.; Pospíšil, M.; Šebor, G. Volatility and Phase Stability of Petrol Blends with Ethanol. Fuel 2009, 88, 1351–1356. [Google Scholar] [CrossRef]
- Kumar, S.; Singh, N.; Prasad, R. Anhydrous Ethanol: A Renewable Source of Energy. Renew. Sustain. Energy Rev. 2010, 14, 1830–1844. [Google Scholar] [CrossRef]
- Rodríguez-Antón, L.M.; Gutiérrez-Martín, F.; Hernández-Campos, M. Physical Properties of Gasoline-ETBE-Isobutanol (in Comparison with Ethanol) Ternary Blends and Their Impact on Regulatory Compliance. Energy 2019, 185, 68–76. [Google Scholar] [CrossRef]
- Tian, Z.; Zhen, X.; Wang, Y.; Liu, D.; Li, X. Combustion and Emission Characteristics of N-Butanol-Gasoline Blends in SI Direct Injection Gasoline Engine. Renew. Energy 2020, 146, 267–279. [Google Scholar] [CrossRef]
- Mužíková, Z.; Káňa, J.; Pospíšil, M.; Šebor, G. Physicochemical Properties of Butanol-Gasoline Blends. Chemické listy 2012, 106. [Google Scholar]
- Gramajo de Doz, M.B.; Bonatti, C.M.; Sólimo, H.N. Water Tolerance and Ethanol Concentration in Ethanol-Gasoline Fuels at Three Temperatures. Energy Fuels 2004, 18, 334–337. [Google Scholar] [CrossRef]
- Yoram Gerchman A Simple Rapid Gas-Chromatography Flame-Ionization-Detector (GC-FID) Method for the Determination of Ethanol from Fermentation Processes. Afr. J. Biotechnol. 2012, 11. [CrossRef]
- Lin, X.; Fan, J.; Wen, Q.; Li, R.; Jin, X.; Wu, J.; Qian, W.; Liu, D.; Xie, J.; Bai, J.; et al. Optimization and Validation of a GC–FID Method for the Determination of Acetone-Butanol-Ethanol Fermentation Products. J. Chromatogr. Sci. 2014, 52, 264–270. [Google Scholar] [CrossRef]









| Properties | Diesel | Gasoline | Ethanol | n–Butanol | s–Butanol | i–Butanol | t–Butanol |
|---|---|---|---|---|---|---|---|
| Molecular weight | 198.4 | 111.19 | 46.07 | 74.11 | 74.11 | 74.11 | 74.11 |
| Cetane number | 40–55 | 0–10 | 5–8 | 12.0 | 8.5 | 8.5 | 5.6 |
| Research octane number | 20–30 | 91–99 | 108 | 94 | 101 | 113 | 105 |
| Motor Octane Number | – | 81–89 | 89–103 | 78 | 91 | 94 | 89 |
| Density [g/mL] at 20 °C | 0.82–0.86 | 0.72–0.78 | 0.789 | 0.808 | 0.808 | 0.805 | 0.800 |
| Flash point [°C] | 65–88 | −45 to −38 | 14 | 35 | 34 | 24 | 28 |
| Water solubility at 25 °C [g/L] | 0.01–0.06 | 0.046–0.077 | ∞ | 73 | 185.1 | 89.4 | ∞ |
| Boiling point [°C] | 180–370 | 25–215 | 78.5 | 117.7 | 99.51 | 107.89 | 82.4 |
| Flammability [% (V/V)] | 1.5–7.6 | 0.6–8 | 3.3–19 | 1.4–11.2 | 1.7–9.8 | 1.7–10.9 | 2.3–8 |
| Reid vapor pressure [kPa] | 0.2–0.7 | 75 | 16.5 | 6 | 5.3 | 3.3 | 12.2 |
| Viscosity [mm2/s] at 25 °C | 1.9–4.1 | 0.4–0.8 | 1.07 | 2.63 | 3.1 | 4 | 4.31 |
| Energy density [MJ/L] | 35.86 | 32 | 25 | 29.2 | 29.06 | 29.00 | 28.48 |
| Parameter | Unit | BUT 5 | BUT 30 | BUT 50 | BUT 85 | BUT 100 |
|---|---|---|---|---|---|---|
| Density at 15 °C | kg∙m−3 | 736.16 | 781.5 | 786.01 | 802.95 | 813.57 |
| Start of distillation | °C | 34 | 33 | 35 | 40 | 117 |
| Evaporated vol. at 70 °C | % (V/V) | 36 | 32 | 22 | * | * |
| Evaporated vol. at 100 °C | % (V/V) | 57 | 55 | 45 | 12 | * |
| Evaporated vol. at 150 °C | % (V/V) | 89 | * | * | * | * |
| End of distillation | °C | 188 | 117 | 117 | 117 | 117 |
| Distillation residue | % (V/V) | 1.2 | 1.8 | 2.3 | 3.2 | 4.5 |
| Reid vapor pressure | kPa | 48.0 | 45.5 | 30.5 | 26.0 | 8.1 |
| Volatility index | – | 725.0 | 679 | 459 | ** | ** |
| Viscosity at 40 °C | mm2∙s−1 | 0.67 | 0.86 | 1.02 | 1.69 | 2.43 |
| Flash point | °C | ** | ** | ** | ** | 35 |
| Fuel | A | B | R2 | p-Value |
|---|---|---|---|---|
| BUT 3 | 114.5 | −22.5 | 0.966 | 1.90 × 10−3 |
| BUT 5 | 73.1 | −30.3 | 0.951 | 1.17 × 10−4 |
| BUT 8 | 151.3 | −53.1 | 0.993 | 1.02 × 10−5 |
| BUT 10 | 107.8 | −58.9 | 0.984 | 4.26 × 10−14 |
| E10 | 120.8 | −72.6 | 0.984 | 1.06 × 10−8 |
| BUT 5 + MTBE 10 | 315.5 | −80.8 | 0.755 | 6.96 × 10−3 |
| BUT 5 + ETBE 10 | 460.7 | −116.3 | 0.995 | 3.98 × 10−7 |
| BUT 10 + MTBE 5 | 123.8 | −75.5 | 0.930 | 2.83 × 10−4 |
| BUT 10 + MTBE 10 | 101.2 | −66.6 | 0.845 | 2.13 × 10−3 |
| Mixture | Measured Concentration of Alcohol [% (V/V)] | |
|---|---|---|
| BUT 5 | 6.08 | |
| BUT 10 | 9.50 | |
| BUT 30 | 33.88 | |
| BUT 85 | 82.92 | |
| iBUT 5 | 4.46 | |
| iBUT 10 | 9.76 | |
| iBUT 30 | 34.11 | |
| iBUT 85 | 83.12 | |
| BUT 5 + iBUT 5 | 6.73 | 5.11 |
| BUT 10 + iBUT 10 | 8.01 | 7.06 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Obergruber, M.; Hönig, V.; Procházka, P.; Kučerová, V.; Kotek, M.; Bouček, J.; Mařík, J. Physicochemical Properties of Biobutanol as an Advanced Biofuel. Materials 2021, 14, 914. https://doi.org/10.3390/ma14040914
Obergruber M, Hönig V, Procházka P, Kučerová V, Kotek M, Bouček J, Mařík J. Physicochemical Properties of Biobutanol as an Advanced Biofuel. Materials. 2021; 14(4):914. https://doi.org/10.3390/ma14040914
Chicago/Turabian StyleObergruber, Michal, Vladimír Hönig, Petr Procházka, Viera Kučerová, Martin Kotek, Jiří Bouček, and Jakub Mařík. 2021. "Physicochemical Properties of Biobutanol as an Advanced Biofuel" Materials 14, no. 4: 914. https://doi.org/10.3390/ma14040914
APA StyleObergruber, M., Hönig, V., Procházka, P., Kučerová, V., Kotek, M., Bouček, J., & Mařík, J. (2021). Physicochemical Properties of Biobutanol as an Advanced Biofuel. Materials, 14(4), 914. https://doi.org/10.3390/ma14040914






