Synthesis, Bottom up Assembly and Thermoelectric Properties of Sb-Doped PbS Nanocrystal Building Blocks
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of PbS Nanocrystals
2.3. Removal of the Capping Ligands
2.4. Structural and Chemical Characterization of PbS and Sb-Doped PbS NCs
2.5. Consolidation and Thermoelectric Characterization of PbS and Sb-Doped PbS NCs
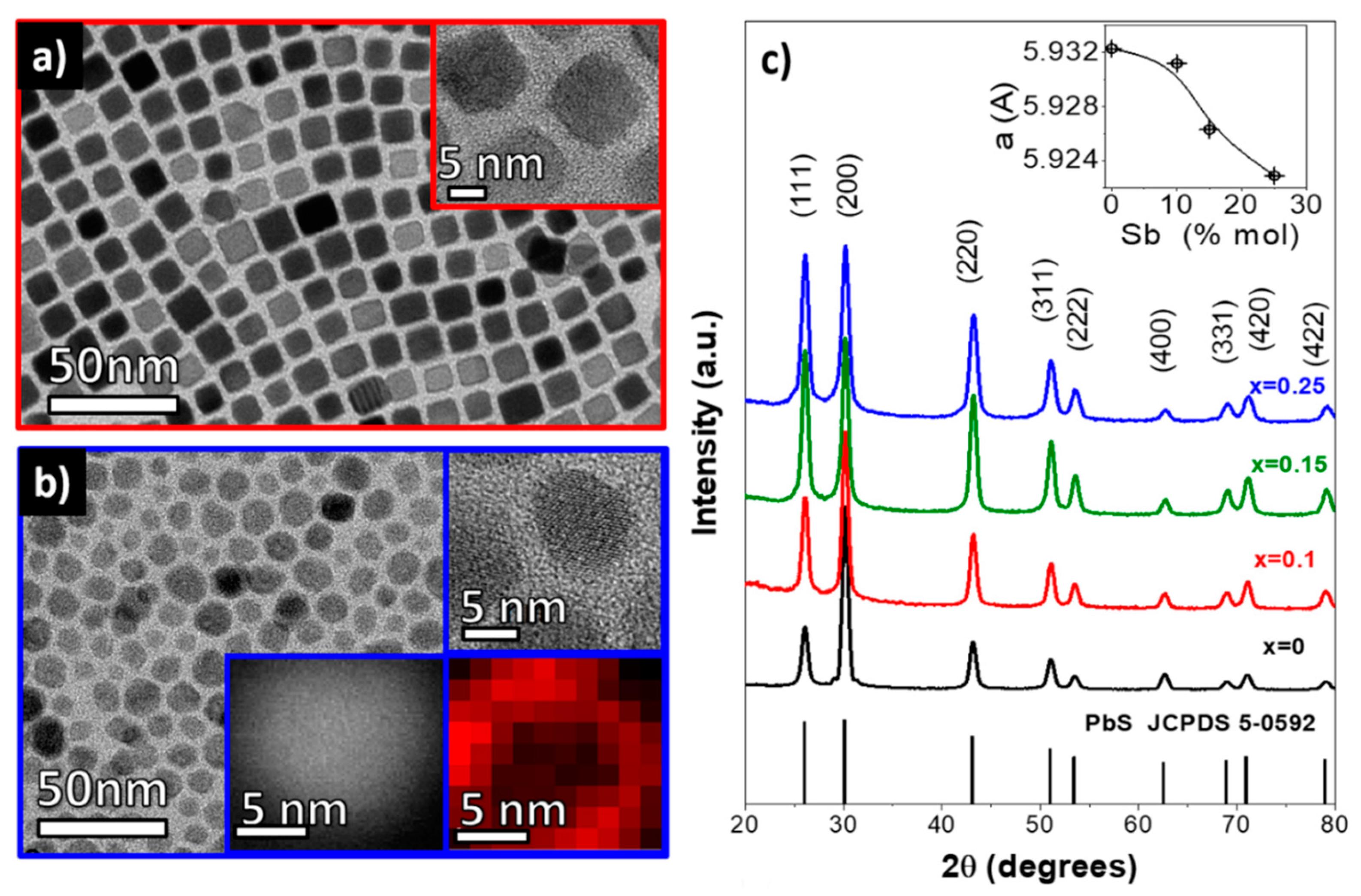
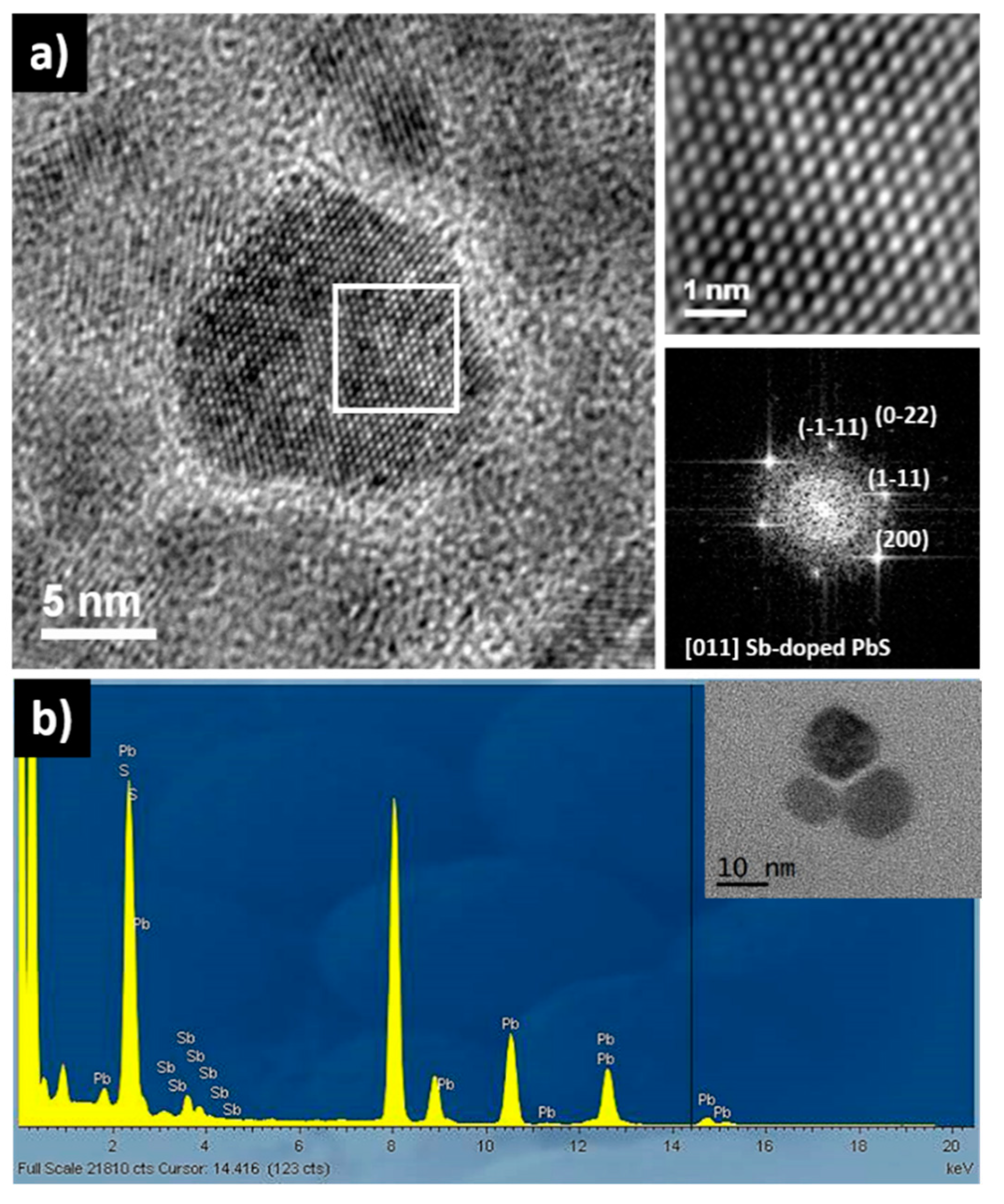
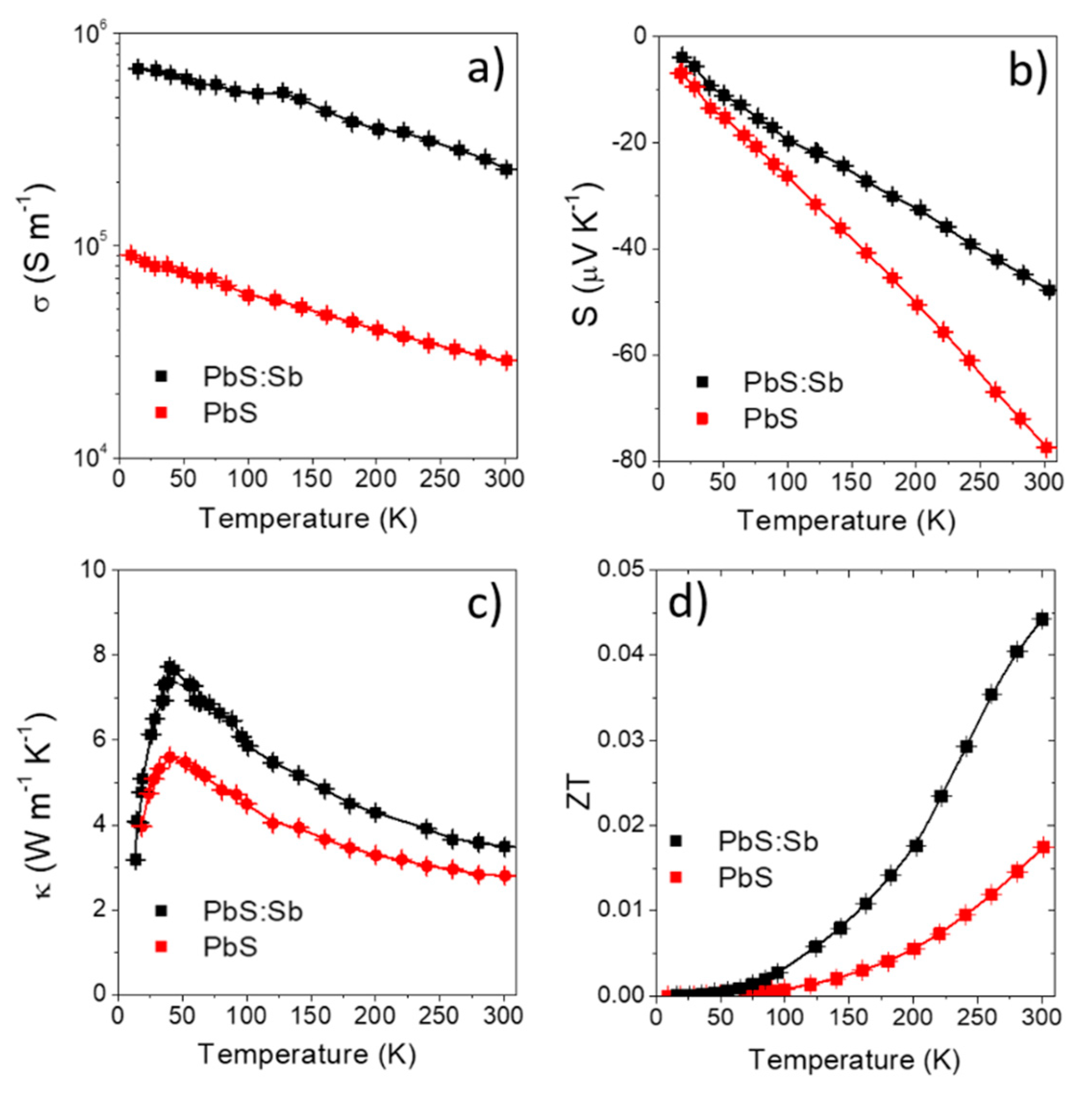
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Norris, D.J.; Efros, A.L.; Erwin, S.C. Doped nanocrystals. Science 2008, 319, 1776–1779. [Google Scholar] [CrossRef] [PubMed]
- Stavrinadis, A.; Konstantatos, G. Strategies for the controlled electronic doping of colloidal quantum dot solids. ChemPhysChem 2016, 17, 632–644. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, Y.; Lim, K.H.; Ibáñez, M.; Ortega, S.; Li, M.; David, J.; Martí-Sánchez, S.; Ng, K.M.; Arbiol, J.; et al. High thermoelectric performance in crystallographically textured n-Type Bi2Te3−xSex produced from asymmetric colloidal nanocrystals. ACS Nano 2018, 12, 7174–7184. [Google Scholar] [CrossRef]
- Liu, Y.; García, G.; Ortega, S.; Cadavid, D.; Palacios, P.; Lu, J.; Ibáñez, M.; Xi, L.; De Roo, J.; López, A.M.; et al. Solution-based synthesis and processing of Sn- and Bi-doped Cu3SbSe4 nanocrystals, nanomaterials and ring-shaped thermoelectric generators. J. Mater. Chem. A 2016, 5, 2592–2602. [Google Scholar] [CrossRef]
- Ibáñez, M.; Zamani, R.; Gorsse, S.; Fan, J.; Ortega, S.; Cadavid, D.; Morante, J.R.; Arbiol, J.; Cabot, A. Core–shell nanoparticles as building blocks for the bottom-up production of functional nano-composites: PbTe–PbS thermoelectric properties. ACS Nano 2013, 7, 2573–2586. [Google Scholar] [CrossRef]
- Vaure, L.; Liu, Y.; Cadavid, D.; Agnese, F.; Aldakov, D.; Pouget, S.; Cabot, A.; Reiss, P.; Chenevier, P. Doping and surface effects of CuFeS2 nanocrystals used in thermoelectric nanocomposites. ChemNanoMat 2018, 4, 982–991. [Google Scholar] [CrossRef]
- Zebarjadi, M.; Joshi, G.; Zhu, G.; Yu, B.; Minnich, A.; Lan, Y.; Wang, X.; Dresselhaus, M.; Ren, Z.; Chen, G. Power factor enhancement by modulation doping in bulk nanocomposites. Nano Lett. 2011, 11, 2225–2230. [Google Scholar] [CrossRef]
- Nolas, G.S.; Sharp, J.; Goldsmid, J. Thermoelectrics: Basic Principles and New Materials Developments; Springer Series in Materials Science; Springer: Berlin/Heidelberg, Germany, 2001; ISBN 978-3-540-41245-8. [Google Scholar]
- Zevalkink, A.; Smiadak, D.M.; Blackburn, J.L.; Ferguson, A.J.; Chabinyc, M.L.; Delaire, O.; Wang, J.; Kovnir, K.; Martin, J.; Schelhas, L.T.; et al. A practical field guide to thermoelectrics: Fundamentals, synthesis, and characterization. Appl. Phys. Rev. 2018, 5, 021303. [Google Scholar] [CrossRef]
- Snyder, G.J.; Toberer, E.S. Complex thermoelectric materials. Nat. Mater. 2008, 7, 105–114. [Google Scholar] [CrossRef]
- Ortega, S.; Ibáñez, M.; Liu, Y.; Zhang, Y.; Kovalenko, M.V.; Cadavid, D.; Cabot, A. Bottom-up engineering of thermoelectric nanomaterials and devices from solution-processed nanoparticle building blocks. Chem. Soc. Rev. 2017, 46, 3510–3528. [Google Scholar] [CrossRef] [PubMed]
- Biswas, K.; He, J.; Blum, I.D.; Wu, C.-I.; Hogan, T.P.; Seidman, D.N.; Dravid, V.P.; Kanatzidis, M.G. High-performance bulk thermoelectrics with all-scale hierarchical architectures. Nature 2012, 489, 414–418. [Google Scholar] [CrossRef] [PubMed]
- Ibáñez, M.; Luo, Z.; Genç, A.; Piveteau, L.; Ortega, S.; Cadavid, D.; Dobrozhan, O.; Liu, Y.; Nachtegaal, M.; Zebarjadi, M.; et al. High-performance thermoelectric nanocomposites from nano-crystal building blocks. Nat. Commun. 2016, 7, 10766. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, Y.; Ortega, S.; Ibáñez, M.; Lim, K.H.; Grau-Carbonell, A.; Martí-Sánchez, S.; Ng, K.M.; Arbiol, J.; Kovalenko, M.V.; et al. Crystallographically textured nanomaterials produced from the liquid phase sintering of BixSb2−xTe3 nanocrystal building blocks. Nano Lett. 2018, 18, 2557–2563. [Google Scholar] [CrossRef] [PubMed]
- Cadavid, D.; Ibáñez, M.; Gorsse, S.; López, A.M.; Solano, E.; Morante, J.R.; Cabot, A. Bottom-up processing of thermoelectric nanocomposites from colloidal nanocrystal building blocks: The case of Ag2Te–PbTe. J. Nanopart. Res. 2012, 14, 1328. [Google Scholar] [CrossRef]
- Cadavid, D.; Ibáñez, M.; Shavel, A.; Durá, O.J.; de la Torre, M.A.L.; Cabot, A. Organic ligand displacement by metal salts to enhance nanoparticle functionality: Thermoelectric properties of Ag2Te. J. Mater. Chem. A 2013, 1, 4864–4870. [Google Scholar] [CrossRef]
- Datta, A.; Nolas, G.S. Synthesis and characterization of nanocrystalline FeSb2 for thermoelectric applications. Eur. J. Inorg. Chem. 2011, 2012, 55–58. [Google Scholar] [CrossRef]
- Wei, K.; Nolas, G.S. Synthesis and characterization of nanostructured stannite Cu2ZnSnSe4 and Ag2ZnSnSe4 for thermoelectric applications. ACS Appl. Mater. Interfaces 2015, 7, 9752–9757. [Google Scholar] [CrossRef] [PubMed]
- Ibáñez, M.; Zamani, R.R.; Li, W.; Cadavid, D.; Gorsse, S.; Katcho, N.A.; Shavel, A.; López, A.M.; Morante, J.R.; Arbiol, J.; et al. Crystallographic control at the nanoscale to enhance functionality: Polytypic Cu2GeSe3 nanoparticles as thermoelectric materials. Chem. Mater. 2012, 24, 4615–4622. [Google Scholar] [CrossRef]
- Ge, Z.-H.; Wei, K.; Lewis, H.; Martin, J.; Nolas, G.S. Bottom-up processing and low temperature transport properties of polycrystalline SnSe. J. Solid State Chem. 2015, 225, 354–358. [Google Scholar] [CrossRef]
- Datta, A.; Nolas, G.S. Nanostructuring and porosity in anisotropic thermoelectric materials prepared by bottom-up processing. In Thermoelectric Nanomaterials: Materials Design and Applications; Springer Series in Materials Science; Koumoto, K., Mori, T., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 177–191. ISBN 978-3-642-37537-8. [Google Scholar]
- Ibáñez, M.; Cadavid, D.; Zamani, R.; García-Castelló, N.; Izquierdo-Roca, V.; Li, W.; Fairbrother, A.; Prades, J.D.; Shavel, A.; Arbiol, J.; et al. Composition control and thermoelectric properties of quaternary chalcogenide nanocrystals: The case of stannite Cu2CdSnSe4. Chem. Mater. 2012, 24, 562–570. [Google Scholar] [CrossRef]
- Ibáñez, M.; Zamani, R.; LaLonde, A.; Cadavid, D.; Li, W.; Shavel, A.; Arbiol, J.; Morante, J.R.; Gorsse, S.; Snyder, G.J.; et al. Cu2ZnGeSe4 nanocrystals: Synthesis and thermoelectric properties. J. Am. Chem. Soc. 2012, 134, 4060–4063. [Google Scholar] [CrossRef]
- Ortega, S.; Ibáñez, M.; Cadavid, D.; Cabot, A. Bottom-up processing of PbTe-PbS thermoelectric nanocomposites. Int. J. Nanotechnol. 2014, 11, 955–970. [Google Scholar] [CrossRef]
- Cadavid, D.; Ortega, S.; Illera, S.; Liu, Y.; Ibáñez, M.; Shavel, A.; Zhang, Y.; Li, M.; López, A.M.; Noriega, G.; et al. Influence of the ligand stripping on the transport properties of nanoparticle-based PbSe nanomaterials. ACS Appl. Energy Mater. 2020, 3, 2120–2129. [Google Scholar] [CrossRef]
- Martin, J.; Wang, L.; Chen, L.; Nolas, G.S. Enhanced Seebeck coefficient through energy-barrier scattering in PbTe nanocomposites. Phys. Rev. B 2009, 79, 115311. [Google Scholar] [CrossRef]
- Martin, J.; Nolas, G.S.; Zhang, W.; Chen, L. PbTe nanocomposites synthesized from PbTe nanocrystals. Appl. Phys. Lett. 2007, 90, 222112. [Google Scholar] [CrossRef]
- Wei, K.; Martin, J.; Nolas, G.S. Synthesis, SPS processing and low temperature transport properties of polycrystalline FeSb2 with nano-scale grains. Mater. Lett. 2014, 122, 289–291. [Google Scholar] [CrossRef]
- Kovalenko, M.V.; Manna, L.; Cabot, A.; Hens, Z.; Talapin, D.V.; Kagan, C.R.; Klimov, V.I.; Rogach, A.L.; Reiss, P.; Milliron, D.J.; et al. Prospects of nanoscience with nanocrystals. ACS Nano 2015, 9, 1012–1057. [Google Scholar] [CrossRef]
- Capitani, C.; Pinchetti, V.; Gariano, G.; Santiago-González, B.; Santambrogio, C.; Campione, M.; Prato, M.; Brescia, R.; Camellini, A.; Bellato, F.; et al. Quantized electronic doping towards atomically controlled “charge-engineered” semiconductor nanocrystals. Nano Lett. 2019, 19, 1307–1317. [Google Scholar] [CrossRef]
- Robin, A.; Livache, C.; Ithurria, S.; Lacaze, E.; Dubertret, M.N.B.; Lhuillier, E. Surface control of doping in self-doped nanocrystals. ACS Appl. Mater. Interfaces 2016, 8, 27122–27128. [Google Scholar] [CrossRef] [PubMed]
- Ibáñez, M.; Korkosz, R.J.; Luo, Z.; Riba, P.; Cadavid, D.; Ortega, S.; Cabot, A.; Kanatzidis, M.G. Electron doping in bottom-up engineered thermoelectric nanomaterials through HCl-mediated ligand displacement. J. Am. Chem. Soc. 2015, 137, 4046–4049. [Google Scholar] [CrossRef]
- Liu, Y.; Cadavid, D.; Ibáñez, M.; Roo, J.D.; Ortega, S.; Dobrozhan, O.; Kovalenko, M.V.; Cabot, A. Colloidal AgSbSe2 nanocrystals: Surface analysis, electronic doping and processing into thermoelectric nanomaterials. J. Mater. Chem. C 2016, 4, 4756–4762. [Google Scholar] [CrossRef]
- Johnsen, S.; He, J.; Androulakis, J.; Dravid, V.P.; Todorov, I.; Chung, D.Y.; Kanatzidis, M.G. Nanostructures boost the thermoelectric performance of PbS. J. Am. Chem. Soc. 2011, 133, 3460–3470. [Google Scholar] [CrossRef]
- Androulakis, J.; Todorov, I.; He, J.; Chung, D.-Y.; Dravid, V.; Kanatzidis, M. Thermoelectrics from abundant chemical elements: High-performance nanostructured PbSe–PbS. J. Am. Chem. Soc. 2011, 133, 10920–10927. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.-D.; Lo, S.-H.; He, J.; Li, H.; Biswas, K.; Androulakis, J.; Wu, C.-I.; Hogan, T.P.; Chung, D.-Y.; Dravid, V.P.; et al. High performance thermoelectrics from earth-abundant materials: Enhanced figure of merit in PbS by second phase nanostructures. J. Am. Chem. Soc. 2011, 133, 20476–20487. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.-D.; He, J.; Wu, C.-I.; Hogan, T.P.; Zhou, X.; Uher, C.; Dravid, V.P.; Kanatzidis, M.G. Thermoelectrics with Earth abundant elements: High performance p-type PbS nanostructured with SrS and CaS. J. Am. Chem. Soc. 2012, 134, 7902–7912. [Google Scholar] [CrossRef]
- Zhao, L.-D.; He, J.; Hao, S.; Wu, C.-I.; Hogan, T.P.; Wolverton, C.; Dravid, V.P.; Kanatzidis, M.G. Raising the thermoelectric performance of p-type PbS with endotaxial nanostructuring and valence-band offset engineering using CdS and ZnS. J. Am. Chem. Soc. 2012, 134, 16327–16336. [Google Scholar] [CrossRef]
- Liu, Y.; Cadavid, D.; Ibáñez, M.; Ortega, S.; Marti-Sanchez, S.; Dobrozhan, O.; Kovalenko, M.V.; Arbiol, J.; Cabot, A. Thermoelectric properties of semiconductor-metal composites produced by particle blending. APL Mater. 2016, 4, 104813. [Google Scholar] [CrossRef]
- Wang, Q.; Fang, Y.; Yin, H.; Li, J. Inhomogenous doping induced the imperfect self-assembly of nanocrystals for the synthesis of porous AgPb10BiTe12 nanosheets and their thermoelectric transport properties. Chem. Commun. 2014, 51, 1594–1596. [Google Scholar] [CrossRef]
- Wang, Q.; Fang, Y.; Zhou, J.; Zhang, G.; Yang, L.; Huo, H. AgPbmSbTem+2 microspheres comprised of self-assembled nanoparticles driven by inhomogenous co-doping synergetic induced dipoles and their thermoelectric and electrochemical Li-storage properties. Chem. Eng. J. 2016, 304, 271–281. [Google Scholar] [CrossRef]
- Tan, G.; Stoumpos, C.C.; Wang, S.; Bailey, T.P.; Zhao, L.-D.; Uher, C.; Kanatzidis, M.G. Subtle roles of Sb and S in regulating the thermoelectric properties of n-type PbTe to high performance. Adv. Energy Mater. 2017, 7, 1700099. [Google Scholar] [CrossRef]
- Chen, T.; Zhang, K.; Wang, H.; Su, W.; Mehmood, F.; Wang, T.; Zhai, J.; Wang, X.; Huo, T.; Wang, C. The high thermoelectric performance of slightly Sb doped PbTe alloys. J. Mater. Chem. C 2020, 8, 1679–1685. [Google Scholar] [CrossRef]
- Martin, J.; Nolas, G.S. Apparatus for the measurement of electrical resistivity, Seebeck coefficient, and thermal conductivity of thermoelectric materials between 300 K and 12 K. Rev. Sci. Instrum. 2016, 87, 015105. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.; Erickson, S.; Nolas, G.S.; Alboni, P.; Tritt, T.M.; Yang, J. Structural and transport properties of Ba8Ga16SixGe30−x clathrates. J. Appl. Phys. 2006, 99, 044903. [Google Scholar] [CrossRef]
- Van Oversteeg, C.H.M.; Oropeza, F.E.; Hofmann, J.P.; Hensen, E.J.; De Jongh, P.E.; Donega, C.D.M. Water-dispersible copper sulfide nanocrystals via ligand exchange of 1-dodecanethiol. Chem. Mater. 2019, 31, 541–552. [Google Scholar] [CrossRef]
- Nag, A.; Kovalenko, M.V.; Lee, J.-S.; Liu, W.; Spokoyny, B.; Talapin, D.V. Metal-free inorganic ligands for colloidal nanocrystals: S2−, HS−, Se2−, HSe−, Te2−, HTe−, TeS32−, OH−, and NH2− as surface ligands. J. Am. Chem. Soc. 2011, 133, 10612–10620. [Google Scholar] [CrossRef]
- Brown, P.R.; Kim, D.; Lunt, R.R.; Zhao, N.; Bawendi, M.G.; Grossman, J.C.; Bulović, V. Energy level modification in lead sulfide quantum dot thin films through ligand exchange. ACS Nano 2014, 8, 5863–5872. [Google Scholar] [CrossRef]
- Knight, K.S. Low temperature thermoelastic properties of galena in a simple, self-consistent, two-term Debye model. Phys. Chem. Miner. 2014, 42, 235–242. [Google Scholar] [CrossRef]
- Tritt, T.M. Thermal Conductivity: Theory, Properties, and Applications; Kluwer Academic/Plenum Publishers: New York, NY, USA, 2004; ISBN 978-0-306-48327-1. [Google Scholar]
- Goldsmid, H.; Penn, A. Boundary scattering of phonons in solid solutions. Phys. Lett. A 1968, 27, 523–524. [Google Scholar] [CrossRef]

| Properties | Charge Carrier Concentration cm−3 | Charge Carrier Mobility cm2/V s | |
|---|---|---|---|
| Sample | |||
| PbS | 6 × 1019 | 30 | |
| Pb0.9Sb0.1S | 9 × 1019 | 154 | |
| System | σ (S m−1 × 104) | S (µV K−1) | κ (W m−1 K−1) | ZT (300K) | Ref. |
|---|---|---|---|---|---|
| PbS + Sb | 23 | −48 | 3.48 | 0.045 | This Work |
| PbS + 4.6% Ag | 8 | −50 | 1.62 | 0.037 | [13] |
| PbTe0.1Se0.4S0.5-Cl | 1 | −75 | 1.25 | 0.013 | [32] |
| (PbSe)0.88(PbS)0.12 + 0.3% PbCl2 | 30 | −75 | 2.9 | 0.17 | [35] |
| Pb0.98Na0.02S | 12.6 | 82 | 2.3 | 0.11 | [37] |
| Pb0.975Na0.025S + 3.0% SrS | 7.5 | 80 | 1.6 | 0.09 | [38] |
| PbS + 4.9% Sn | 3.8 | −75 | 1.8 | 0.035 | [39] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cadavid, D.; Wei, K.; Liu, Y.; Zhang, Y.; Li, M.; Genç, A.; Berestok, T.; Ibáñez, M.; Shavel, A.; Nolas, G.S.; et al. Synthesis, Bottom up Assembly and Thermoelectric Properties of Sb-Doped PbS Nanocrystal Building Blocks. Materials 2021, 14, 853. https://doi.org/10.3390/ma14040853
Cadavid D, Wei K, Liu Y, Zhang Y, Li M, Genç A, Berestok T, Ibáñez M, Shavel A, Nolas GS, et al. Synthesis, Bottom up Assembly and Thermoelectric Properties of Sb-Doped PbS Nanocrystal Building Blocks. Materials. 2021; 14(4):853. https://doi.org/10.3390/ma14040853
Chicago/Turabian StyleCadavid, Doris, Kaya Wei, Yu Liu, Yu Zhang, Mengyao Li, Aziz Genç, Taisiia Berestok, Maria Ibáñez, Alexey Shavel, George S. Nolas, and et al. 2021. "Synthesis, Bottom up Assembly and Thermoelectric Properties of Sb-Doped PbS Nanocrystal Building Blocks" Materials 14, no. 4: 853. https://doi.org/10.3390/ma14040853
APA StyleCadavid, D., Wei, K., Liu, Y., Zhang, Y., Li, M., Genç, A., Berestok, T., Ibáñez, M., Shavel, A., Nolas, G. S., & Cabot, A. (2021). Synthesis, Bottom up Assembly and Thermoelectric Properties of Sb-Doped PbS Nanocrystal Building Blocks. Materials, 14(4), 853. https://doi.org/10.3390/ma14040853








