Enhancing Thermal and Mechanical Properties of Ramie Fiber via Impregnation by Lignin-Based Polyurethane Resin
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Isolation and Fractionation of Lignin
2.3. Lignin Characterization
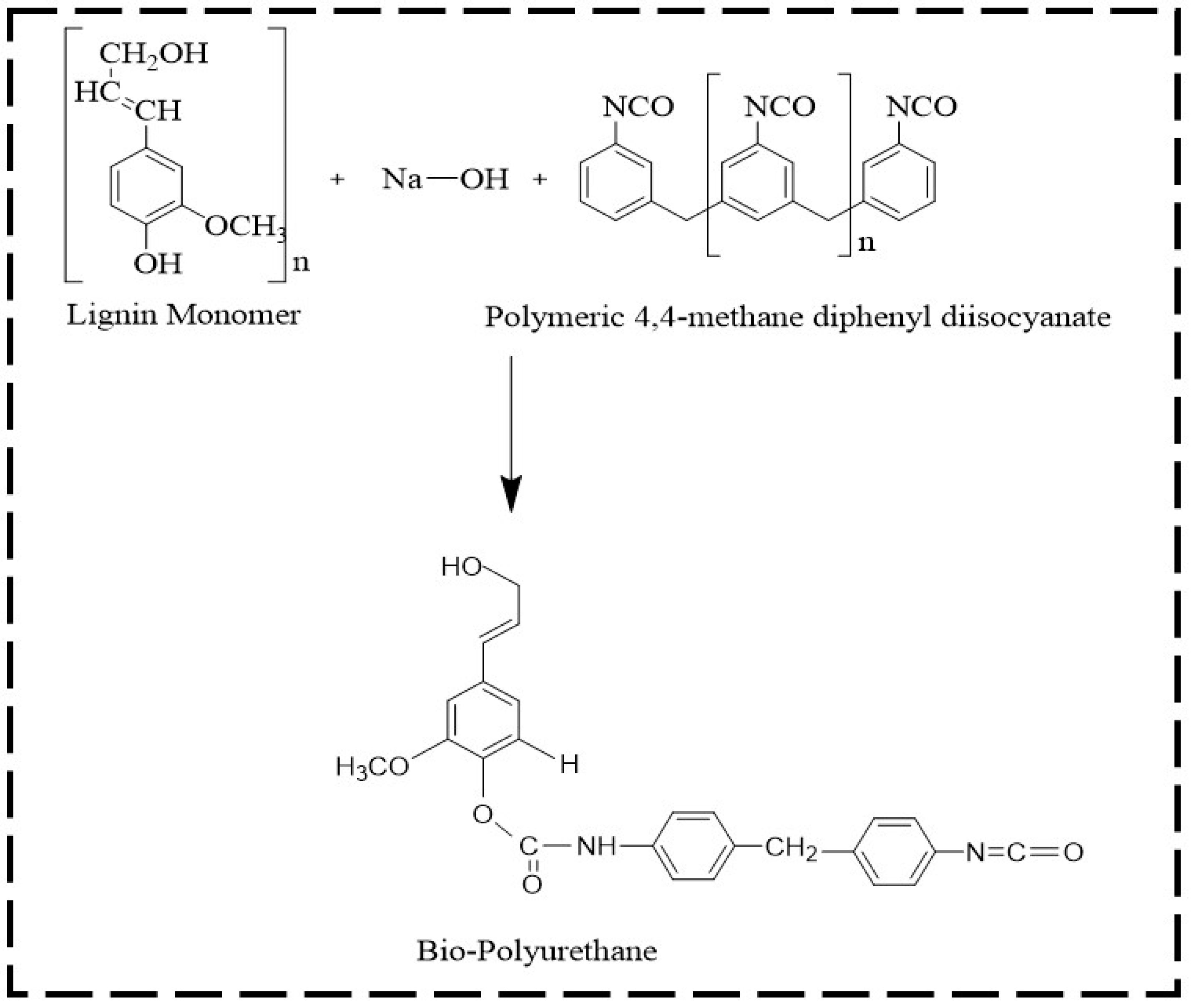
2.4. Preparation of Bio-Polyurethane Resin
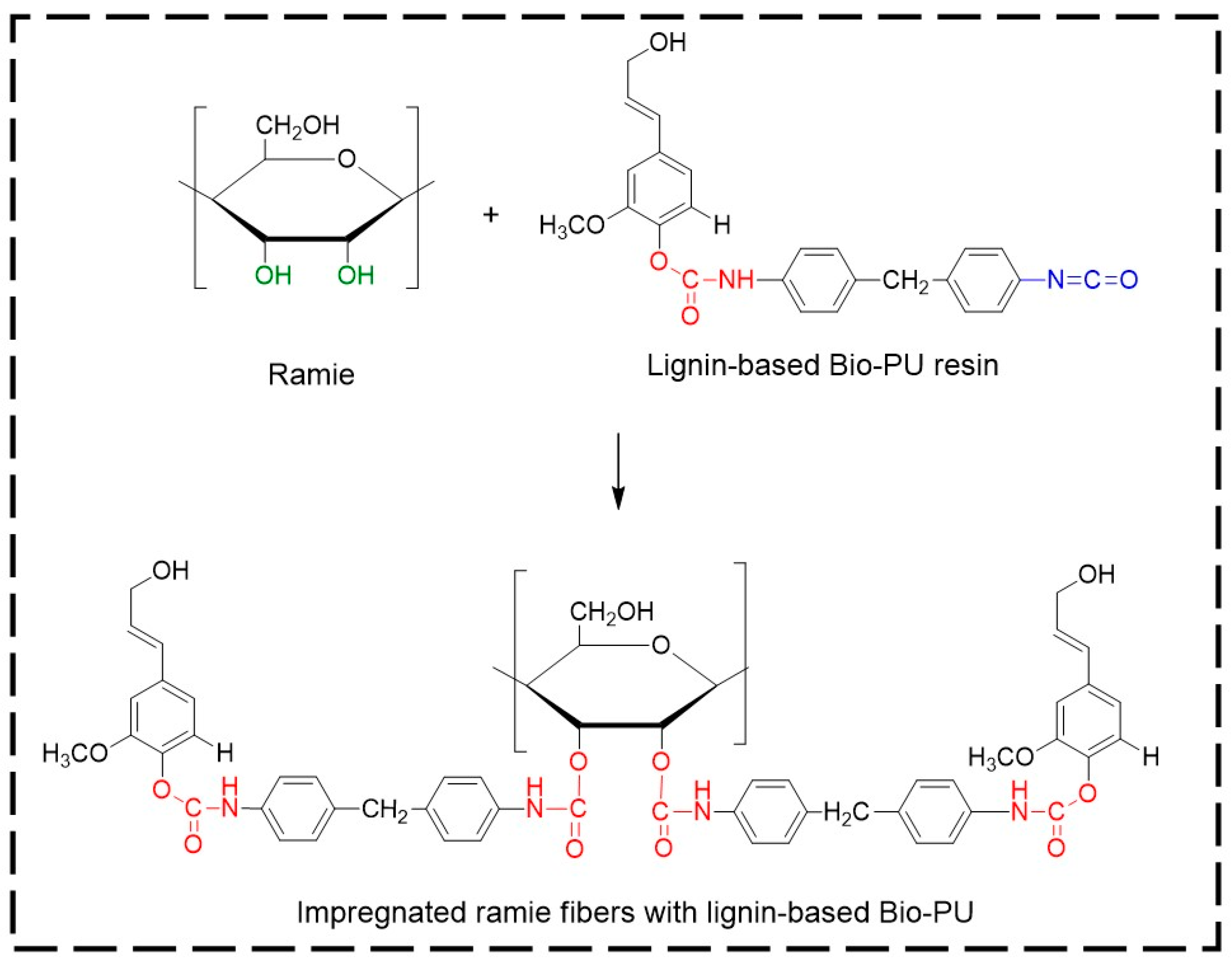
2.5. Impregnation of Ramie Fiber with Bio-PU
2.6. Characterization of Lignin-Based Bio-PU Resin
2.7. Evaluation of Ramie Fibers Properties
3. Results and Discussion
3.1. Basic Properties of Isolated Lignin
| Analysis Parameters | Value | References |
|---|---|---|
| Moisture Content of Black Liquor (%) | 27.81 ± 1.11 | 10.00 [48] |
| Solid Content of Black Liquor (%) | 76.79 ± 0.64 | 65.00–85.00 [49,50] |
| pH of Black Liquor | 12.14 ± 0.02 | 12.00–13.00 [51,52] |
| Yield of Lignin (%) | 35.88 ± 1.81 | 45.76 [38] |
| Moisture Content of Lignin (%) | 5.07 ± 0.71 | 8.05 [46] |
| Ash Content of Lignin (%) | 0.31 ± 0.19 | 8.25–19.19 [38,47] |
| Acid-insoluble Lignin (AIL) (%) | 82.54 ± 0.96 | 53.08 [38] |
| Acid-soluble Lignin (ASL) (%) | 12.77 ± 0.67 | 7.26 [38] |
| Purity Levels of Lignin (%) | 95.32 ± 0.61 | 60.34 [38] |
3.2. Characterizations of Lignin
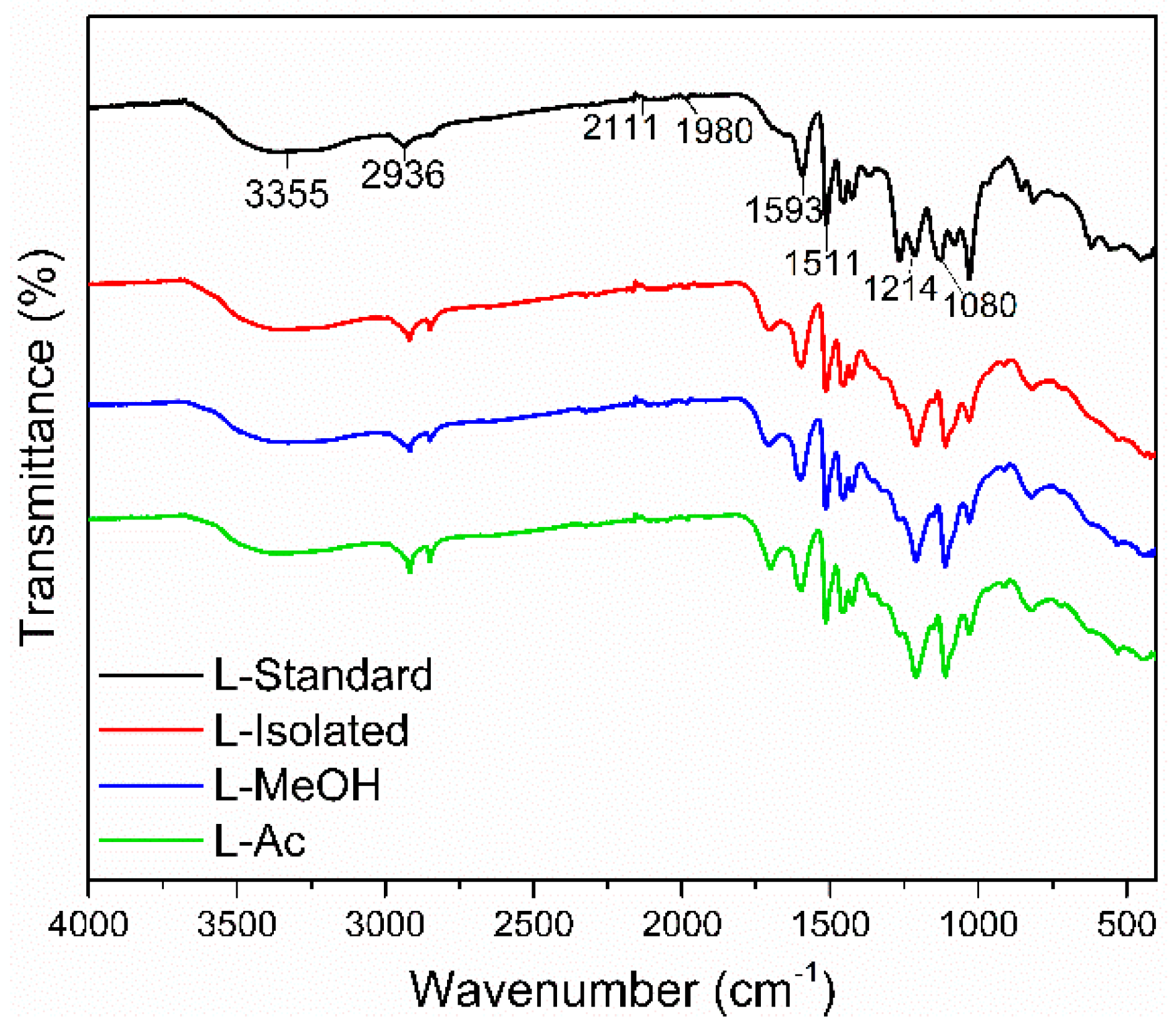
3.2.1. FTIR Analysis
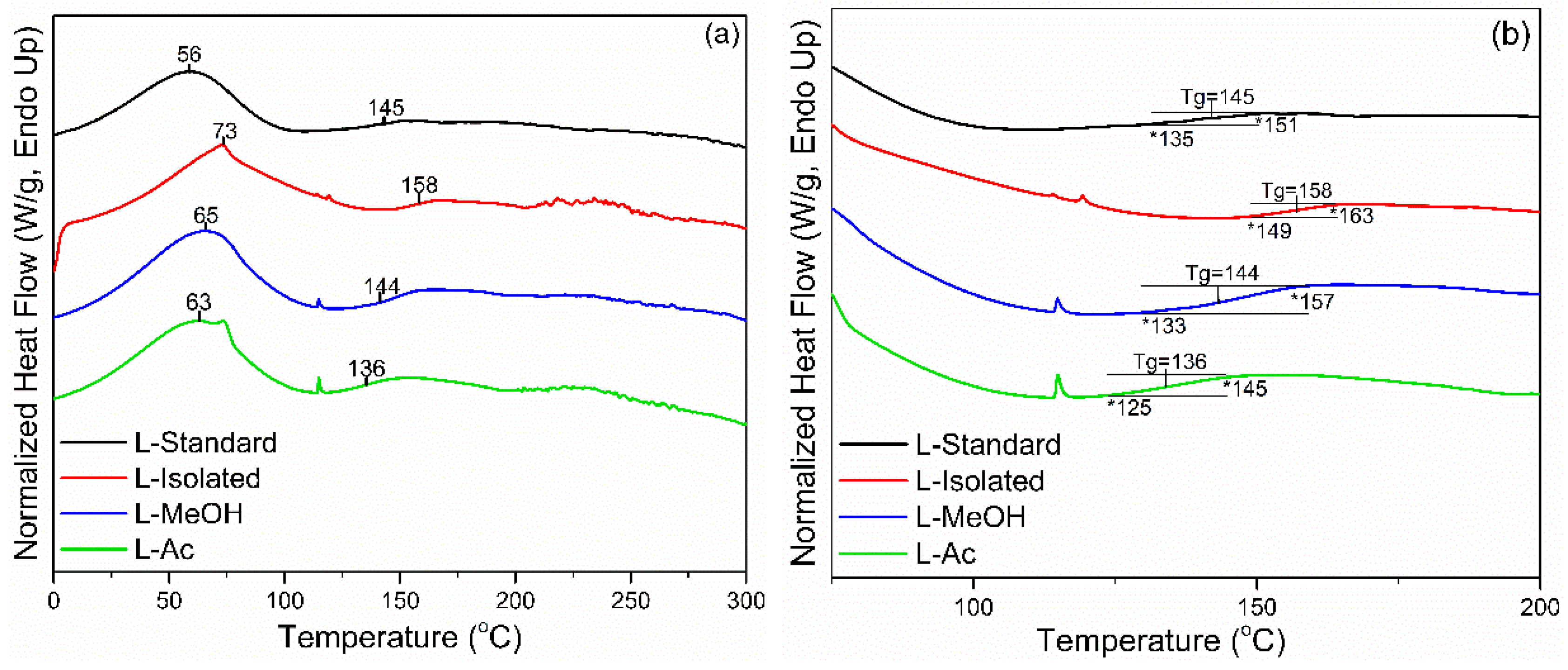
3.2.2. DSC Analysis
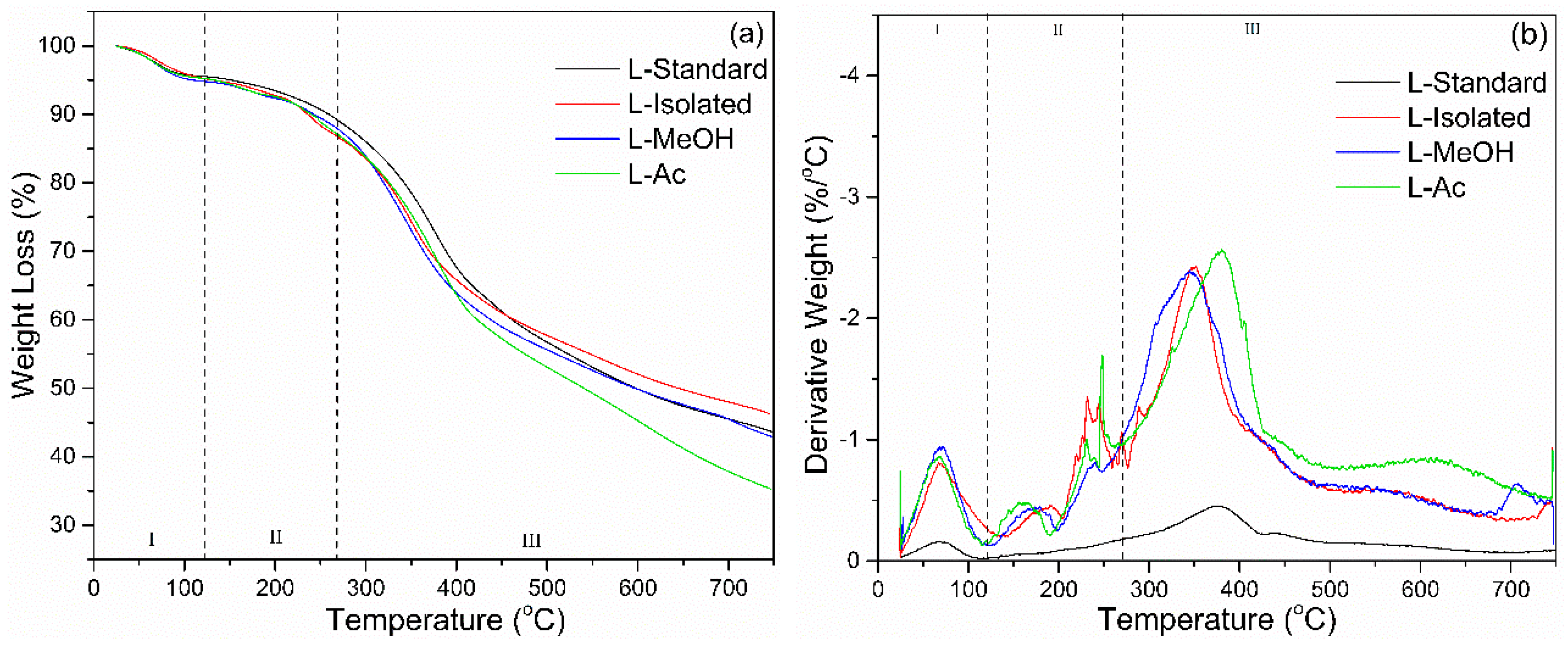
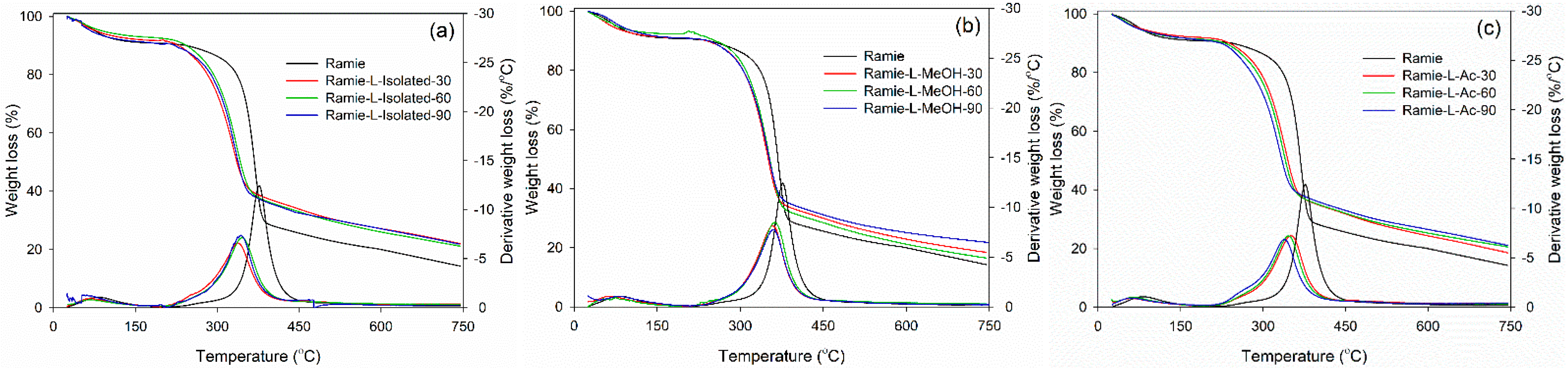
3.2.3. TGA Analysis
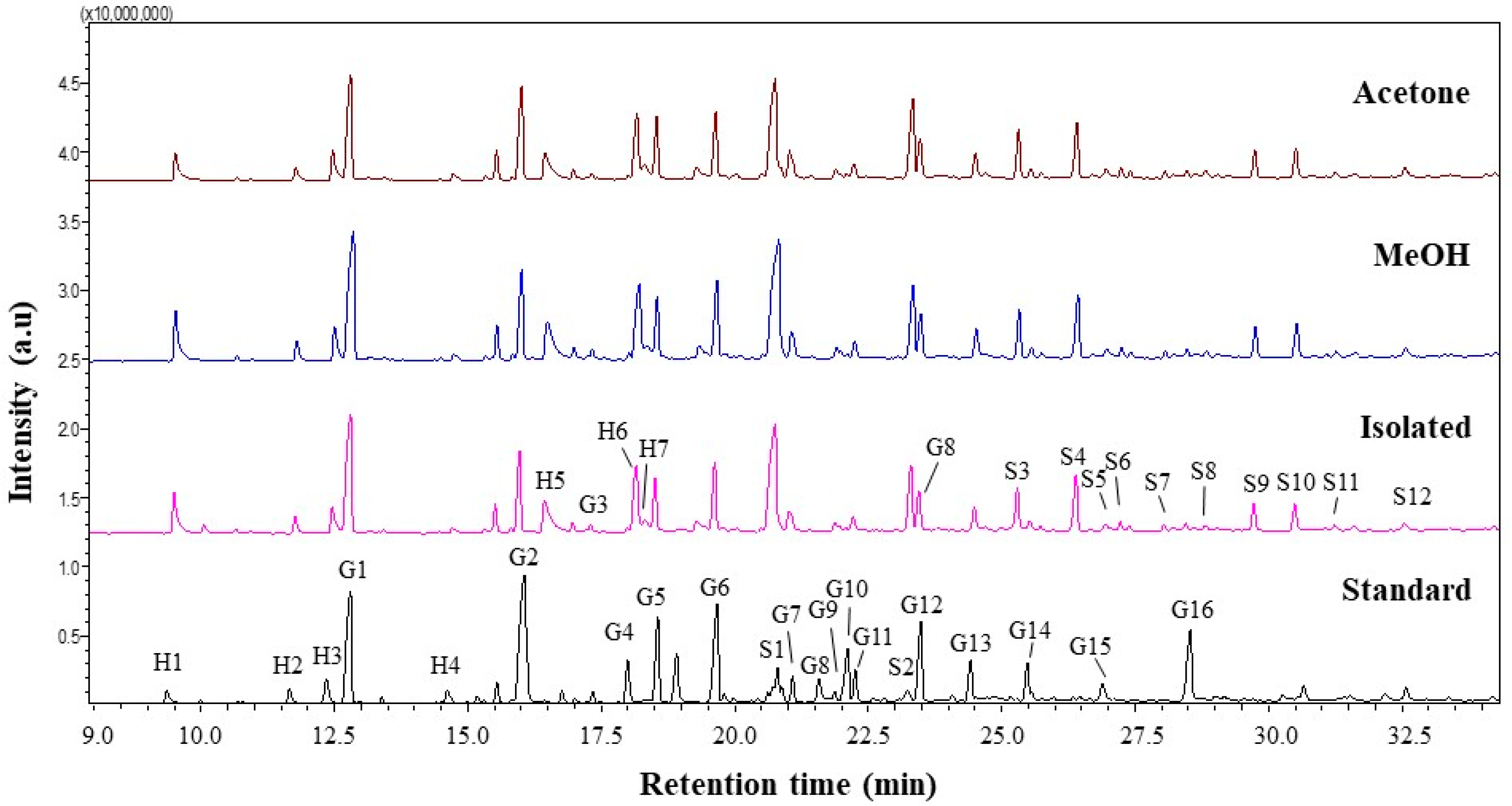
3.2.4. Py-GCMS Analysis
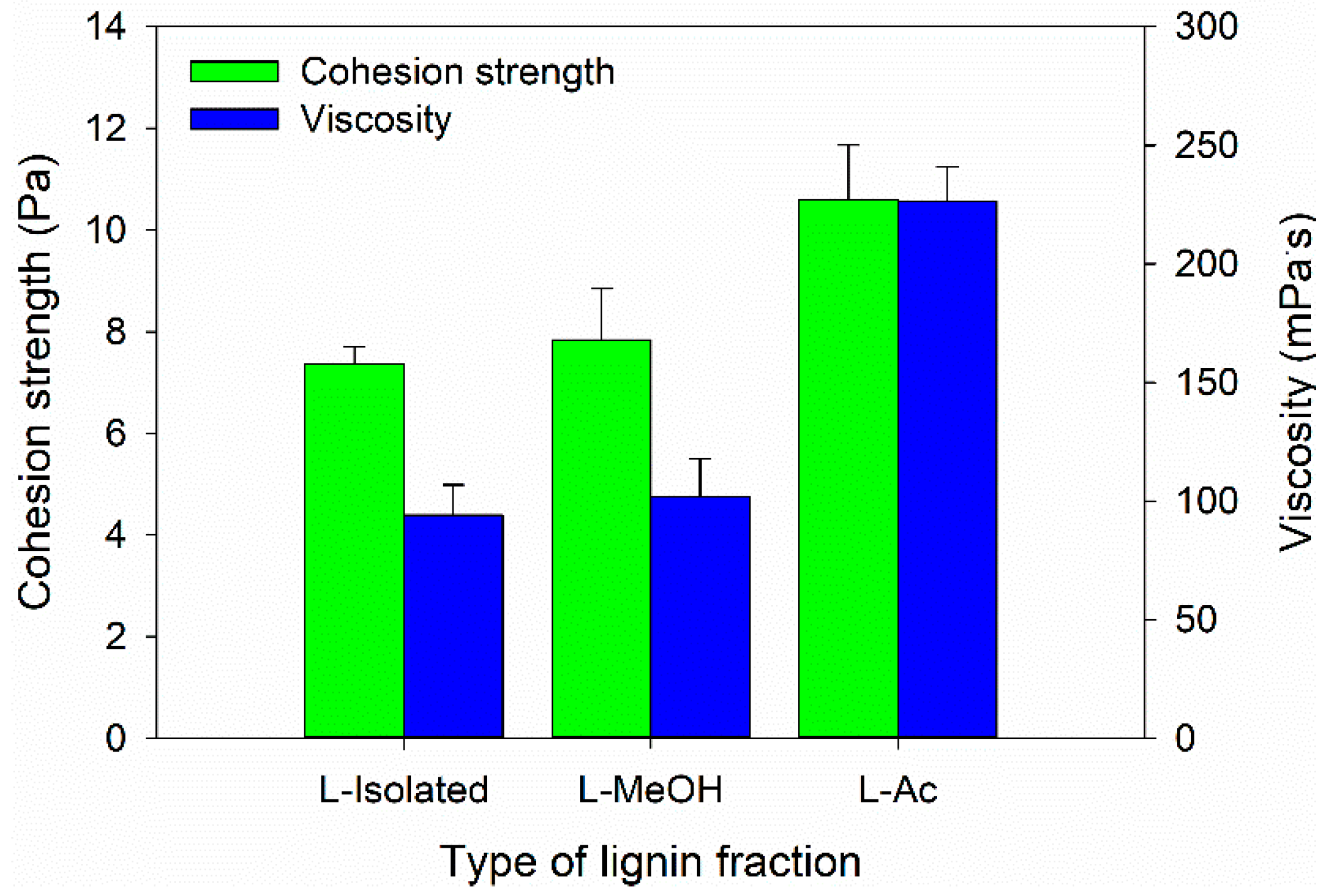
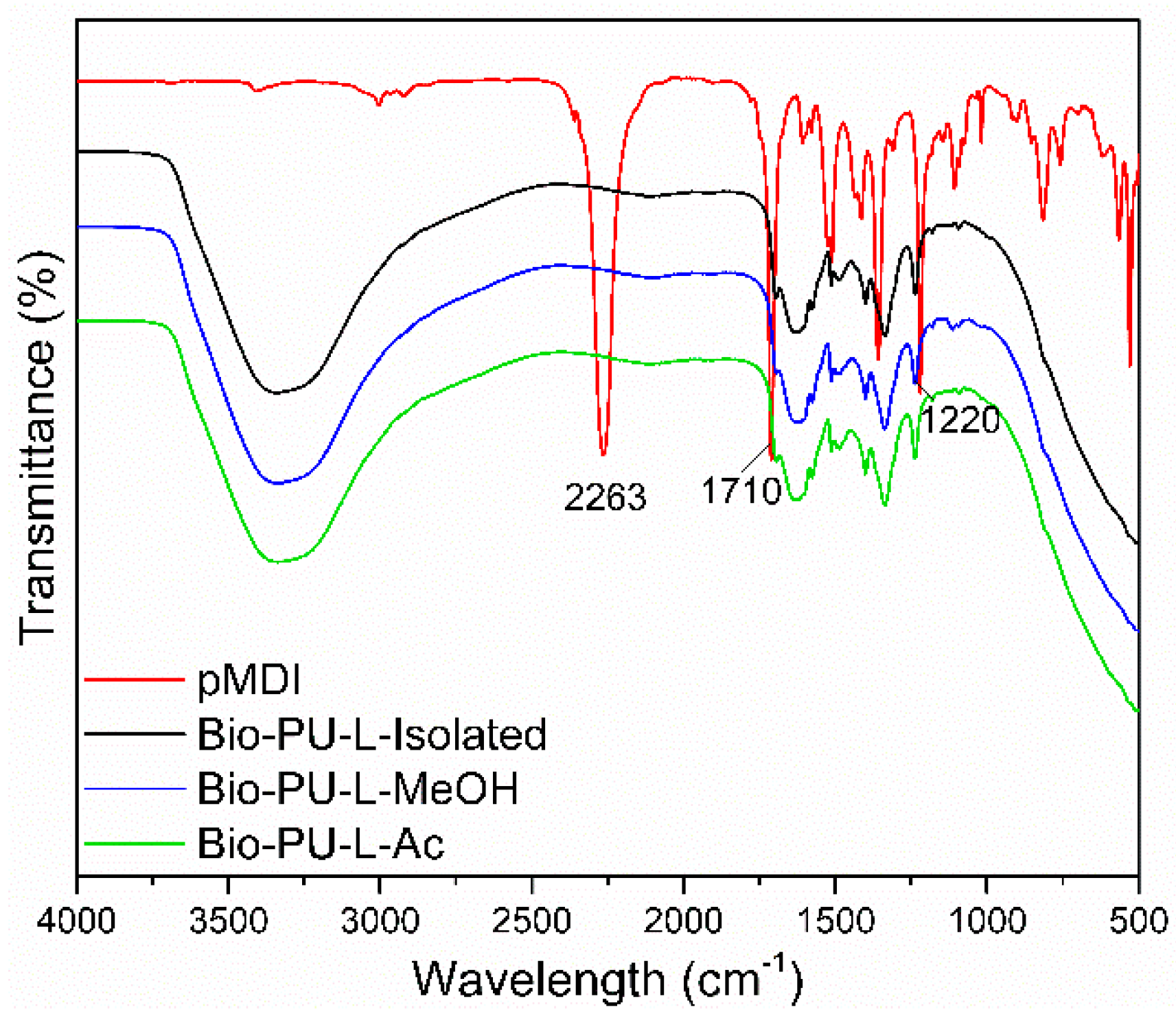
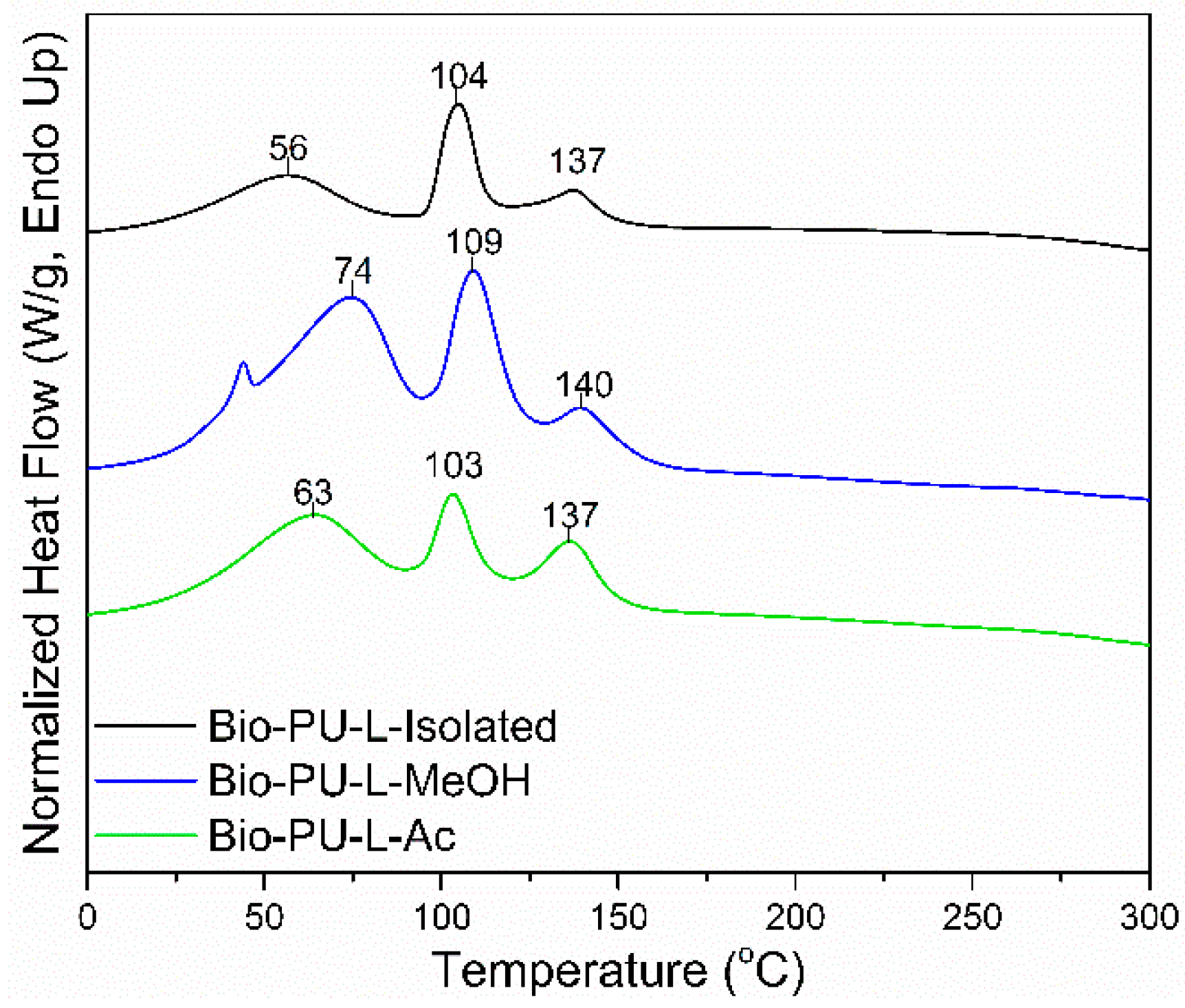
3.3. Properties of Bio-Polyurethane Resin
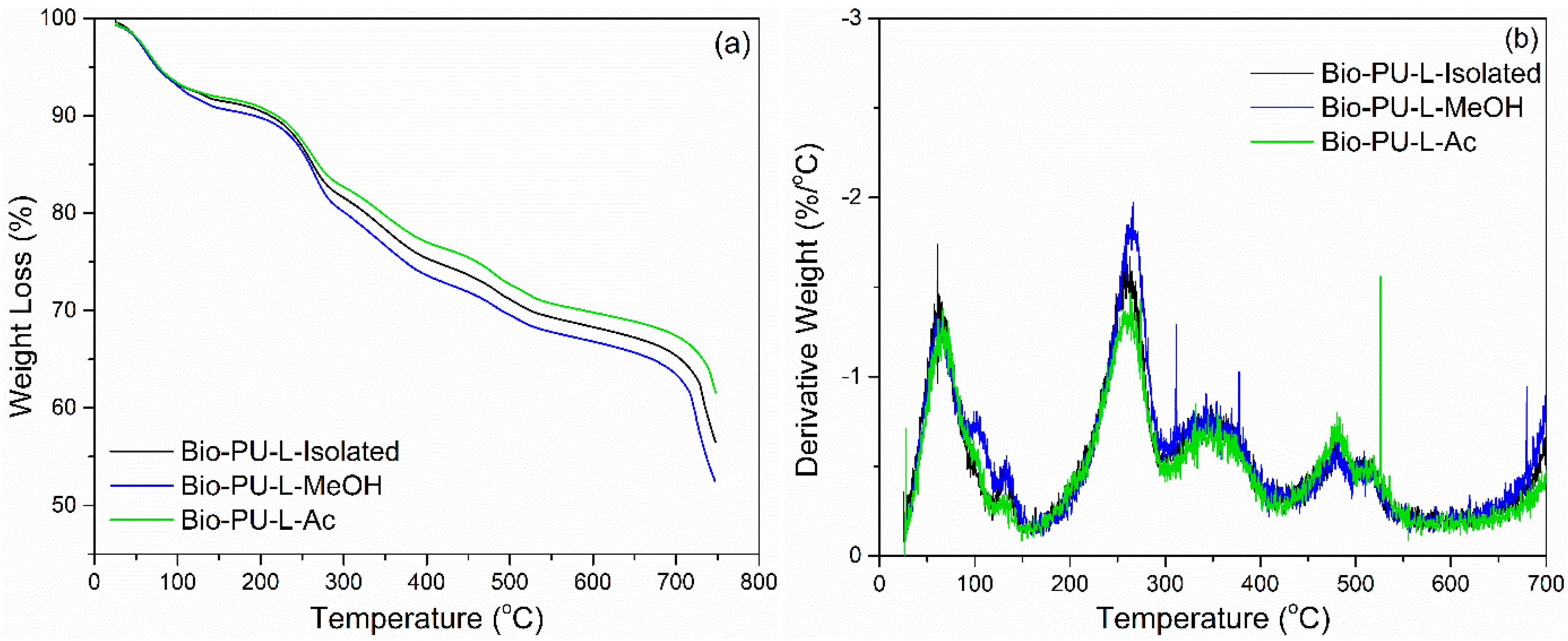
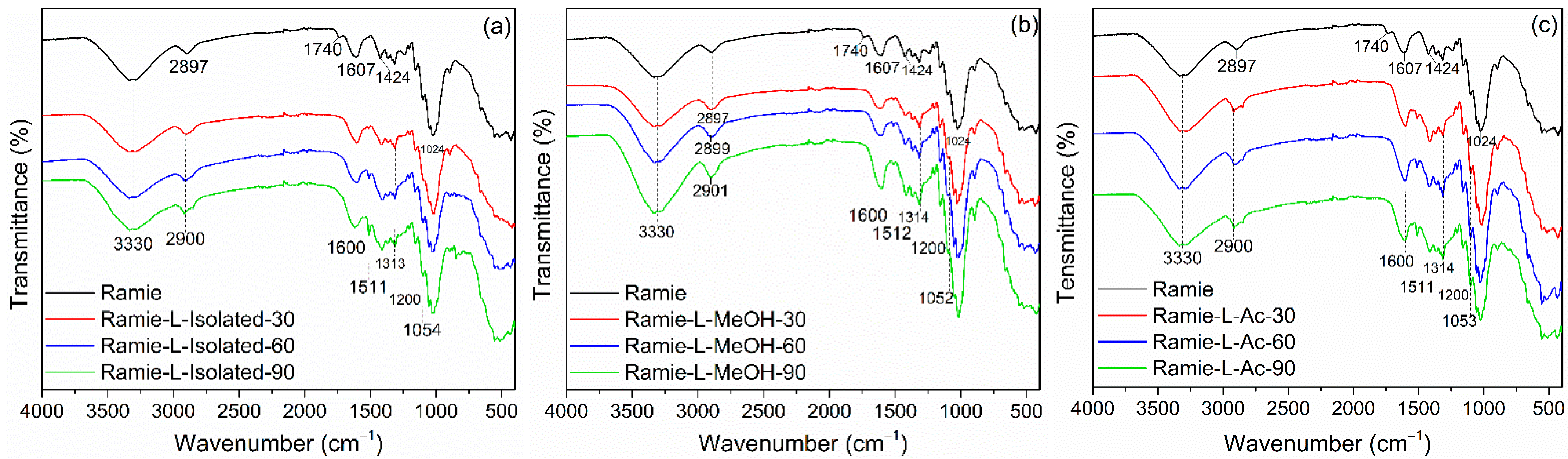
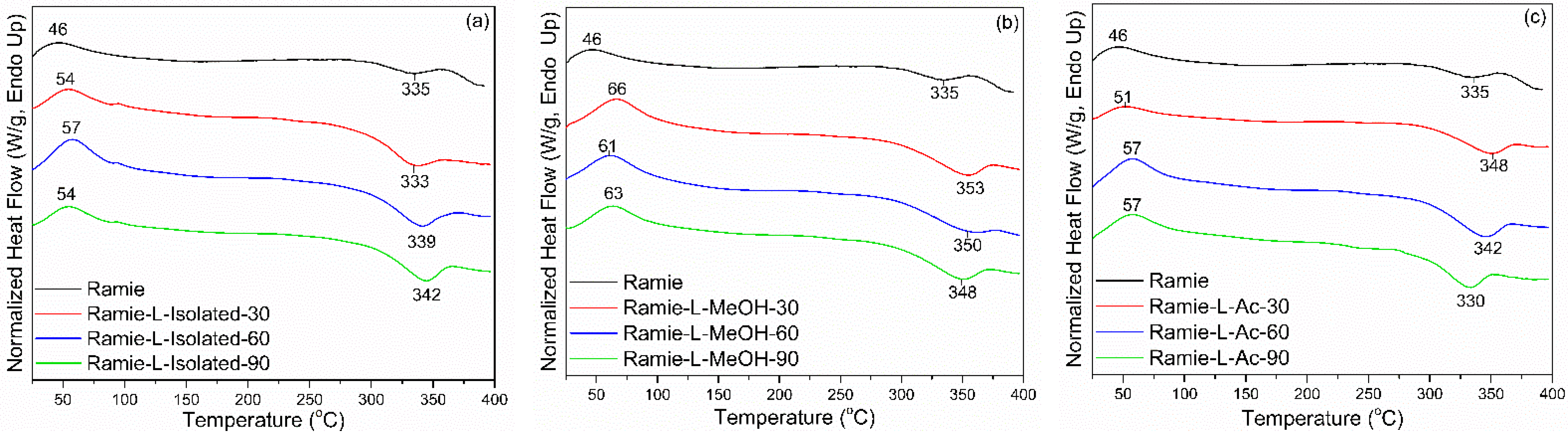
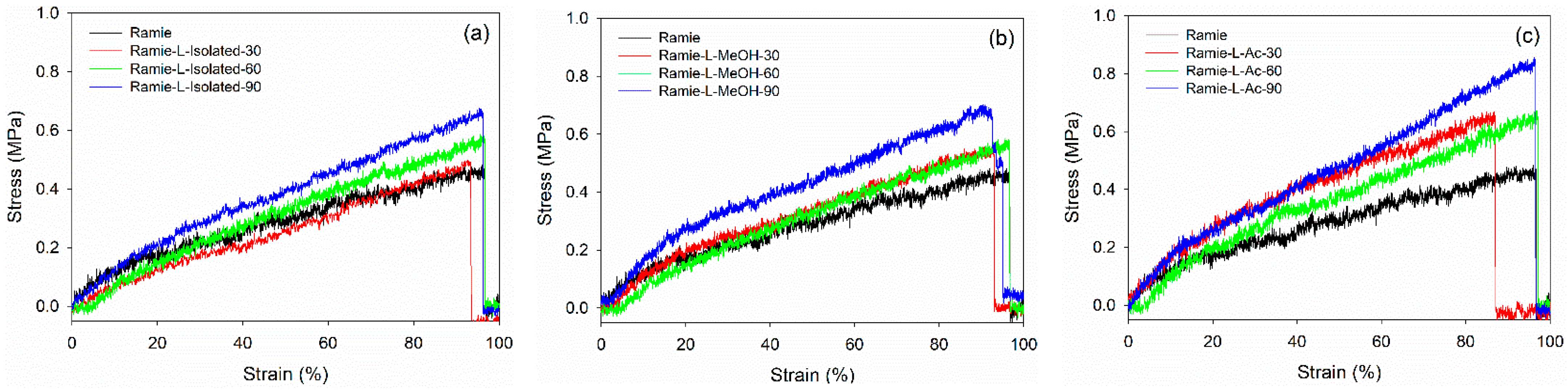
3.4. Properties of Ramie Fiber Impregnated with Bio-PU Resin
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hwang, J.Z.; Wang, S.C.; Chen, P.C.; Huang, C.Y.; Yeh, J.T.; Chen, K.N. A new UV-curable PU resin obtained through a nonisocyanate process and used as a hydrophilic textile treatment. J. Polym. Res. 2012, 19, 9900. [Google Scholar] [CrossRef]
- Zia, K.M.; Anjum, S.; Zuber, M.; Mujahid, M.; Jamil, T. Synthesis and molecular characterization of chitosan based polyurethane elastomers using aromatic diisocyanate. Int. J. Biol. Macromol. 2014, 66, 26–32. [Google Scholar] [CrossRef]
- Das, A.; Mahanwar, P. A brief discussion on advances in polyurethane applications. Adv. Ind. Eng. Polym. Res. 2020, 3, 93–101. [Google Scholar] [CrossRef]
- Chen, W.-J.; Wang, S.-C.; Chen, P.-C.; Chen, T.-W.; Chen, K.-N. Hybridization of aqueous PU/epoxy resin via a dual self-curing process. J. Appl. Polym. Sci. 2008, 110, 147–155. [Google Scholar] [CrossRef]
- Wang, S.-C.; Chen, P.-C.; Yeh, J.-T.; Chen, K.-N. Curing reaction of amino-terminated aqueous-based polyurethane dispersions with triglycidyl-containing compound. J. Appl. Polym. Sci. 2008, 110, 725–731. [Google Scholar] [CrossRef]
- Strakowska, A.; Członka, S.; Kairyte, A. Rigid polyurethane foams reinforced with poss-impregnated sugar beet pulp filler. Materials 2020, 13, 5493. [Google Scholar] [CrossRef]
- Uram, K.; Kurańska, M.; Andrzejewski, J.; Prociak, A. Rigid Polyurethane Foams Modified with Biochar. Materials 2021, 14, 5616. [Google Scholar] [CrossRef] [PubMed]
- Gama, N.V.; Ferreira, A.; Barros-Timmons, A. Polyurethane foams: Past, present, and future. Materials 2018, 11, 1841. [Google Scholar] [CrossRef] [Green Version]
- Kurańska, M.; Pinto, J.A.; Salach, K.; Barreiro, M.F.; Prociak, A. Synthesis of thermal insulating polyurethane foams from lignin and rapeseed based polyols: A comparative study. Ind. Crops Prod. 2020, 143, 111882. [Google Scholar] [CrossRef]
- Hu, S.; Luo, X.; Li, Y. Polyols and polyurethanes from the liquefaction of lignocellulosic biomass. ChemSusChem 2014, 7, 66–72. [Google Scholar] [CrossRef]
- Petrovic, Z.S. Polyurethanes from vegetable oils. Polym. Rev. 2008, 48, 109–155. [Google Scholar] [CrossRef]
- Pfister, D.P.; Xia, Y.; Larock, R.C. Recent advances in vegetable oil-based polyurethanes. ChemSusChem 2011, 4, 703–717. [Google Scholar] [CrossRef] [PubMed]
- Lligadas, G.; Ronda, J.C.; Galiá, M.; Cádiz, V. Plant oils as platform chemicals for polyurethane synthesis: Current state-of-the-art. Biomacromolecules 2010, 11, 2825–2835. [Google Scholar] [CrossRef] [PubMed]
- Ge, X.; Chang, C.; Zhang, L.; Cui, S.; Luo, X.; Hu, S.; Qin, Y.; Li, Y. Conversion of Lignocellulosic Biomass into Platform Chemicals for Biobased Polyurethane Application, 1st ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2018; Volume 3. [Google Scholar]
- Thébault, M.; Pizzi, A.; Essawy, H.A.; Barhoum, A.; Van Assche, G. Isocyanate free condensed tannin-based polyurethanes. Eur. Polym. J. 2015, 67, 513–526. [Google Scholar] [CrossRef]
- Aristri, M.A.; Lubis, M.A.R.; Iswanto, A.H.; Fatriasari, W.; Sari, R.K.; Antov, P.; Gajtanska, M.; Papadopoulos, A.N.; Pizzi, A. Bio-Based Polyurethane Resins Derived from Tannin: Source, Synthesis, Characterisation, and Application. Forests 2021, 12, 1516. [Google Scholar] [CrossRef]
- Watanabe, M.; Kanaguri, Y.; Smith, R.L. Hydrothermal separation of lignin from bark of Japanese cedar. J. Supercrit. Fluids 2018, 133, 696–703. [Google Scholar] [CrossRef]
- Tudor, E.M.; Barbu, M.C.; Petutschnigg, A.; Réh, R.; Krišťák, Ľ. Analysis of larch-bark capacity for formaldehyde removal in wood adhesives. Int. J. Environ. Res. Public Health 2020, 17, 764. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cateto, C.A.; Barreiro, M.F.; Rodrigues, A.E. Monitoring of lignin-based polyurethane synthesis by FTIR-ATR. Ind. Crops Prod. 2008, 27, 168–174. [Google Scholar] [CrossRef] [Green Version]
- Bajwa, D.S.; Pourhashem, G.; Ullah, A.H.; Bajwa, S.G. A concise review of current lignin production, applications, products and their environment impact. Ind. Crops Prod. 2019, 139, 111526. [Google Scholar] [CrossRef]
- Mandlekar, N.; Cayla, A.; Rault, F.; Giraud, S.; Salaün, F.; Malucelli, G.; Guan, J.-P. An Overview on the Use of Lignin and Its Derivatives in Fire Retardant Polymer Systems. In Lignin—Trends and Applications; InTech: London, UK, 2018; pp. 207–231. [Google Scholar]
- Zhang, H.; Bai, Y.; Yu, B.; Liu, X.; Chen, F. A practicable process for lignin color reduction: Fractionation of lignin using methanol/water as a solvent. Green Chem. 2017, 19, 5152–5162. [Google Scholar] [CrossRef]
- Evtuguin, D.V.; Andreolety, J.P.; Gandini, A. Polyurethanes based on oxygen-organosolv lignin. Eur. Polym. J. 1998, 34, 1163–1169. [Google Scholar] [CrossRef]
- Aristri, M.A.; Lubis, M.A.R.; Yadav, S.M.; Antov, P.; Papadopoulos, A.N.; Pizzi, A.; Fatriasari, W.; Ismayati, M.; Iswanto, A.H. Recent Developments in Lignin- and Tannin-Based Non-Isocyanate Polyurethane Resins for Wood Adhesives—A Review. Appl. Sci. 2021, 11, 4242. [Google Scholar] [CrossRef]
- Thring, R.W.; Vanderlaan, M.N.; Griffin, S.L. Polyurethanes from Alcell® lignin. Biomass Bioenergy 1997, 13, 125–132. [Google Scholar] [CrossRef]
- Antov, P.; Savov, V.; Trichkov, N.; Krišťák, Ľ.; Réh, R.; Papadopoulos, A.N.; Taghiyari, H.R.; Pizzi, A.; Kunecová, D.; Pachikova, M. Properties of high-density fiberboard bonded with urea–formaldehyde resin and ammonium lignosulfonate as a bio-based additive. Polymers 2021, 13, 2775. [Google Scholar] [CrossRef]
- Alzagameem, A.; El Khaldi-Hansen, B.; Büchner, D.; Larkins, M.; Kamm, B.; Witzleben, S.; Schulze, M. Lignocellulosic biomass as source for lignin-based environmentally benign antioxidants. Molecules 2018, 23, 2664. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Réh, R.; Krišťák, Ľ.; Sedliačik, J.; Bekhta, P.; Božiková, M.; Kunecová, D.; Vozárová, V.; Tudor, E.M.; Antov, P.; Savov, V. Utilization of birch bark as an eco-friendly filler in urea-formaldehyde adhesives for plywood manufacturing. Polymers 2021, 13, 511. [Google Scholar] [CrossRef]
- Bachtiar, E.V.; Kurkowiak, K.; Yan, L.; Kasal, B.; Kolb, T. Thermal stability, fire performance, and mechanical properties of natural fibre fabric-reinforced polymer composites with different fire retardants. Polymers 2019, 11, 699. [Google Scholar] [CrossRef] [Green Version]
- Twite-Kabamba, E.; Mechraoui, A.; Rodrigue, D. Rheological properties of polypropylene/hemp fiber composites. Polym. Compos. 2009, 30, 1401–1407. [Google Scholar] [CrossRef]
- Rehman, M.; Gang, D.; Liu, Q.; Chen, Y.; Wang, B.; Peng, D.; Liu, L. Ramie, a multipurpose crop: Potential applications, constraints and improvement strategies. Ind. Crops Prod. 2019, 137, 300–307. [Google Scholar] [CrossRef]
- Mulyawan, A.S.; Sana, A.W.; Kaelani, Z. Identifikasi Sifat Fisik Dan Sifat Termal Serat-Serat Selulosa Untuk Pembuatan Komposit. Arena Tekst. 2015, 30, 75–82. [Google Scholar] [CrossRef]
- Yan, L.; Chouw, N.; Jayaraman, K. Flax fibre and its composites—A review. Compos. Part B Eng. 2014, 56, 296–317. [Google Scholar] [CrossRef]
- Yuan, J.M.; Feng, Y.R.; He, L.P. Effect of thermal treatment on properties of ramie fibers. Polym. Degrad. Stab. 2016, 133, 303–311. [Google Scholar] [CrossRef]
- Kalia, S.; Sheoran, R. Modification of ramie fibers using microwaveassisted grafting and cellulase enzyme-assisted biopolishing: A comparative study of morphology, thermal stability, and crystallinity. Int. J. Polym. Anal. Charact. 2011, 16, 307–318. [Google Scholar] [CrossRef]
- Liu, X.; Dai, G. Impregnation of thermoplastic resin in jute fiber mat. Front. Chem. Eng. China 2008, 2, 145–149. [Google Scholar] [CrossRef]
- Xia, C.; Zhang, S.; Shi, S.Q.; Cai, L.; Huang, J. Property enhancement of kenaf fiber reinforced composites by in situ aluminum hydroxide impregnation. Ind. Crop. Prod. 2016, 79, 131–136. [Google Scholar] [CrossRef]
- Hermiati, E.; Risanto, L.; Lubis, M.A.R.; Laksana, R.P.B.; Dewi, A.R. Chemical characterization of lignin from kraft pulping black liquor of Acacia mangium. AIP Conf. Proc. 2017, 1803, 020005. [Google Scholar] [CrossRef]
- Ponnuchamy, V.; Gordobil, O.; Diaz, R.H.; Sandak, A.; Sandak, J. Fractionation of lignin using organic solvents: A combined experimental and theoretical study. Int. J. Biol. Macromol. 2021, 168, 792–805. [Google Scholar] [CrossRef] [PubMed]
- TAPPI. T211 Ash in Wood, Pulp, Paper and Paperboard: Combustion at 525 °C; TAPPI Standard Test Methods; TAPPI: Peachtree Corners, GA, USA, 2007; Volume T211 om-02, p. 5. [Google Scholar]
- Templeton, D.; Ehrman, T. Determination of Acid-Insoluble Lignin in Biomass—LAP-003; NREL—National Renewable Energy Laboratory: Golden, CO, USA, 1995; p. 14.
- Ehrman, T. Determination of Acid-Soluble Lignin in Biomass—LAP-004; NREL—National Renewable Energy Laboratory: Golden, CO, USA, 1996; Volume LAP-004, p. 8.
- Serrano, L.; Esakkimuthu, E.S.; Marlin, N.; Brochier-Salon, M.C.; Mortha, G.; Bertaud, F. Fast, Easy, and Economical Quantification of Lignin Phenolic Hydroxyl Groups: Comparison with Classical Techniques. Energy Fuels 2018, 32, 5969–5977. [Google Scholar] [CrossRef]
- Aristri, M.A.; Lubis, M.A.R.; Laksana, R.P.B.; Fatriasari, W.; Ismayati, M.; Wulandari, A.P.; Ridho, M.R. Bio-Polyurethane Resins Derived from Liquid Fractions of Lignin for the Modification of Ramie Fibers. J. Sylva Lestari 2021, 9, 223–238. [Google Scholar] [CrossRef]
- ASTM. ASTM D 3379–75 Standard Test Method for Tensile Strength and Young’s Modulus for High-Modulus Single-Filament Materials; ASTM International: Washington, DC, USA, 2000; Volume 75, pp. 1–5. [Google Scholar]
- Sameni, J.; Krigstin, S.; Derval dos Santos, R.; Leao, A.; Sain, M. Thermal characteristics of lignin residue from industrial processes. BioResources 2014, 9, 725–737. [Google Scholar] [CrossRef] [Green Version]
- Lubis, M.A.R.; Dewi, A.R.; Risanto, L.; Zaini, L.H.; Hermiati, E. Isolation and Characterization of Lignin from Alkaline Pretreatment Black Liquor of Oil Palm Empty Fruit Bunch and Sugarcane Bagasse. In Proceedings of the ASEAN COSAT 2014, Bogor, Indonesia, 18–19 August 2014; pp. 483–491. [Google Scholar]
- Gordobil, O.; Herrera, R.; Poohphajai, F.; Sandak, J.; Sandak, A. Impact of drying process on kraft lignin: Lignin-water interaction mechanism study by 2D NIR correlation spectroscopy. J. Mater. Res. Technol. 2021, 12, 159–169. [Google Scholar] [CrossRef]
- Cardoso, M.; de Oliveira, É.D.; Passos, M.L. Chemical composition and physical properties of black liquors and their effects on liquor recovery operation in Brazilian pulp mills. Fuel 2009, 88, 756–763. [Google Scholar] [CrossRef]
- Nikolskaya, E.; Janhunen, P.; Haapalainen, M.; Hiltunen, Y. Solids Content of Black Liquor Measured by Online. Appl. Sci. 2019, 9, 2169. [Google Scholar] [CrossRef] [Green Version]
- Yotwadee, H.; Duangduen, A.; Viboon, S. Lignin isolation from black liquor for wastewater quality improvement and bio-material recovery. Int. J. Environ. Sci. Dev. 2020, 11, 365–371. [Google Scholar] [CrossRef]
- Jusuf, P.G.; Purwono, S.; Tawfiequrahman, A. Permodelan Ekstraksi Lignin Mentah dari Black Liquor dengan Metode Asidifikasi pada pH Rendah. In Proceedings of the Seminar Nasional Teknik Kimia “Kejuangan”, Jakarta, Indonesia, 25 April 2019; pp. 1–5. [Google Scholar]
- Sadeghifar, H.; Sadeghifar, H.; Ragauskas, A.; Ragauskas, A.; Ragauskas, A.; Ragauskas, A. Perspective on Technical Lignin Fractionation. ACS Sustain. Chem. Eng. 2020, 8, 8086–8101. [Google Scholar] [CrossRef]
- Sameni, J.; Krigstin, S.; Sain, M. Solubility of Lignin and Acetylated Lignin in Organic Solvents. BioResources 2017, 12, 1548–1565. [Google Scholar] [CrossRef]
- Kubo, S.; Kadla, J.F. Hydrogen bonding in lignin: A fourier transform infrared model compound study. Biomacromolecules 2005, 6, 2815–2821. [Google Scholar] [CrossRef]
- Li, H.; McDonald, A.G. Fractionation and characterization of industrial lignins. Ind. Crops Prod. 2014, 62, 67–76. [Google Scholar] [CrossRef]
- Buranov, A.U.; Ross, K.A.; Mazza, G. Isolation and characterization of lignins extracted from flax shives using pressurized aqueous ethanol. Bioresour. Technol. 2010, 101, 7446–7455. [Google Scholar] [CrossRef]
- Lucejko, J.J.; Tamburini, D.; Modugno, F.; Ribechini, E.; Colombini, M.P. Analytical pyrolysis and mass spectrometry to characterise lignin in archaeological wood. Appl. Sci. 2021, 11, 240. [Google Scholar] [CrossRef]
- Gogoi, R.; Alam, M.; Khandal, R. Effect of increasing NCO/OH molar ratio on the physicomechanical and thermal properties of isocyanate terminated polyurethane prepolymer. Int. J. Basic Appl. Sci. 2014, 3, 118–123. [Google Scholar] [CrossRef] [Green Version]
- Restasari, A.; Ardianingsih, R.; Abdillah, L.H.; Hartaya, K. Effects of Toluene Diisocyanate’s Chemical Structure on Polyurethane’s Viscosity and Mechanical Properties for Propellant. In Proceedings of the International Seminar on Aerospace Science and Technology, Kuta, Indonesia, 27–29 October 2015; pp. 59–67. [Google Scholar]
- Gharib, J.; Pang, S.; Holland, D. Synthesis and characterisation of polyurethane made from pyrolysis bio-oil of pine wood. Eur. Polym. J. 2020, 133, 109725. [Google Scholar] [CrossRef]
- Lubis, M.A.R.; Park, B.D.; Lee, S.M. Microencapsulation of polymeric isocyanate for the modification of urea-formaldehyde resins. Int. J. Adhes. Adhes. 2020, 100, 102599. [Google Scholar] [CrossRef]
- Luo, S.; Gao, L.; Guo, W. Effect of incorporation of lignin as bio-polyol on the performance of rigid lightweight wood–polyurethane composite foams. J. Wood Sci. 2020, 66, 23. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.; Zhang, H.; Zhu, Z.; Fu, S. High-Value Utilization of Hydroxymethylated Lignin in Polyurethane Adhesives; Elsevier B.V.: Amsterdam, The Netherlands, 2020; Volume 152, ISBN 1343036402. [Google Scholar]
- Amado, J.C.Q. Thermal Resistance Properties of Polyurethanes and Its Composites. In Thermosoftening Plastics; InTech: London, UK, 2019; pp. 1–13. [Google Scholar]
- Wang, Y.-Y.; Wyman, C.E.; Cai, C.M.; Ragauskas, A.J. Lignin-Based Polyurethanes from Unmodified Kraft Lignin Fractionated by Sequential Precipitation. ACS Appl. Polym. Mater. 2019, 1, 1672–1679. [Google Scholar] [CrossRef] [Green Version]
- Tavares, L.; Stilhano, C.R.; Boas, V. Bio-Based polyurethane prepared from Kraft lignin and modified castor oil. Express Polym. Lett. 2016, 10, 927–940. [Google Scholar] [CrossRef]
- Trovati, G.; Sanches, E.A.; Neto, S.C.; Mascarenhas, Y.P.; Chierice, G.O. Characterization of Polyurethane Resins by FTIR, TGA and XRD. J. Appl. Polym. Sci. 2009, 115, 263–268. [Google Scholar] [CrossRef]
- Novarini, E.; Sukardan, M.D. Potensi Serat Rami (Boehmeria Nivea S. Gaud) Sebagai Bahan Baku Industri Tekstil Dan Produk Tekstil Dan Tekstil Teknik. Arena Tekst. 2015, 30, 113–122. [Google Scholar] [CrossRef]
- Bevitori, A.B.; da Silva, I.L.A.; Rohen, L.A.; Margem, F.M.; de Moraes, Y.M.; Monteiro, S.N. Evaluation of Ramie Fibers Component by Infrared Spectroscopy. In Proceedings of the 21st CBECIMAT—Congresso Brasileiro de Engenharia e Ciência dos Materiais, Cuiabá, Brazil, 9–13 November 2014; pp. 2109–2116. [Google Scholar]
- Simonassi, N.T.; Pereira, A.C.; Monteiro, S.N.; Muylaert, F.; De Deus, J.F.; Fontes, C.M.; Drelich, J.; Vermelha, P.; De Janeiro, R.; California, P. Reinforcement of Polyester with Renewable Ramie Fibers. Mater. Res. 2017, 20, 51–59. [Google Scholar] [CrossRef] [Green Version]
- Kandimalla, R.; Kalita, S.; Choudhury, B.; Devi, D.; Kalita, D.; Kalita, K.; Dash, S.; Kotoky, J. Fiber from ramie plant (Boehmeria nivea): A novel suture biomaterial. Mater. Sci. Eng. C 2016, 62, 816–822. [Google Scholar] [CrossRef]
- Shahinur, S.; Hasan, M.; Ahsan, Q.; Haider, J. Effect of Chemical Treatment on Thermal Properties of Jute Fiber Used in Polymer Composites. J. Compos. Sci. 2020, 4, 132. [Google Scholar] [CrossRef]
- Shahinur, S.; Hasan, M.; Ahsan, Q.; Saha, D.K.; Islam, M.S. Characterization on the Properties of Jute Fiber at Different Portions. Int. J. Polym. Sci. 2015, 2015, 262348. [Google Scholar] [CrossRef] [Green Version]
- Tomczak, F.; Satyanarayana, K.G.; Sydenstricker, T.H.D. Studies on lignocellulosic fibers of Brazil: Part III—Morphology fibers and properties of Brazilian curaua. Compos. Part. A 2007, 38, 2227–2236. [Google Scholar] [CrossRef]
- Bevitori, A.B.; Margem, F.M.; Carreiro, R.S.; Monteiro, S.N.; Calado, V. Thermal Caracterization Behavior of Epoxy Composites Reinforced Ramie Fibers. In Proceedings of the 67th ABM International Congress, Rio de Janeiro, Brazil, 31 July–3 August 2012; pp. 473–480. [Google Scholar]
- Charlet, K.; Jernot, J.P.; Breard, J.; Gomina, M. Scattering of morphological and mechanical properties of flax fibres. Ind. Crops Prod. 2010, 32, 220–224. [Google Scholar] [CrossRef]
- Le Duigou, A.; Bourmaud, A.; Balnois, E.; Davies, P.; Baley, C. Improving the interfacial properties between flax fibres and PLLA by a water fibre treatment and drying cycle. Ind. Crops Prod. 2012, 39, 31–39. [Google Scholar] [CrossRef]


| Type of Lignin | Yield of Fractionated Lignin (%) | Total OH Group |
|---|---|---|
| L-Standard | - | 8.109 |
| L-Isolated | - | 7.968 |
| L-MeOH | 71.25 | 7.399 |
| L-Ac | 70.57 | 7.645 |
| Type of Lignin | TWL10% (°C) | TWL25% (°C) | TWL50% (°C) | WL (%) | Residue (%) |
|---|---|---|---|---|---|
| L-standard | 259 | 366 | 597 | 56.49 | 43.51 |
| L-isolated | 235 | 347 | 644 | 53.75 | 46.25 |
| L-MeOH | 242 | 341 | 596 | 57.06 | 42.94 |
| L-Ac | 239 | 351 | 540 | 64.71 | 35.29 |
| No | RT (min) | Pyrolysis Product | Origin | Lignin-Isolated | Fractionated Lignin | |
|---|---|---|---|---|---|---|
| MeOH | Ac | |||||
| 1 | 9.50 | Phenol | H | 3.93 | 4.17 | 2.85 |
| 2 | 11.76 | Phenol. 2-methyl- | H | 1.22 | 1.35 | 1.35 |
| 3 | 12.46 | Phenol. 4 methyl | H | 2.7 | 2.9 | 3.13 |
| 4 | 12.80 | Guaiacol | G | 14.24 | 14.87 | 10.86 |
| 5 | 15.97 | Guaiacol. 4-methyl | G | 6.79 | 6.66 | 8.7 |
| 6 | 16.44 | Catechol | H | 4.65 | 5.04 | 4.07 |
| 7 | 18.16 | Catechol. 3-methoxy | H | 7.28 | 6.58 | 6.72 |
| 8 | 18.30 | Catechol. 4 methyl | H | 1.44 | 0.2 | 1.84 |
| 9 | 19.62 | Guaiacol. 4-vinyl | G | 5.58 | 5.61 | 5.23 |
| 10 | 20.76 | Syringol | S | 16.77 | 17.5 | 14.38 |
| 11 | 23.30 | Syringol-4-methyl | S | 6.24 | 6.3 | 8.36 |
| LH (hydroxypenhyl) | 21.22 | 20.24 | 19.96 | |||
| LG (guaiacyl) | 39.84 | 39.94 | 39.51 | |||
| LS (syringyl) | 38.52 | 39.44 | 39.9 | |||
| S/G | 0.9669 | 0.9875 | 1.0099 | |||
| S/G/H | 0.0456 | 0.0488 | 0.0506 | |||
| Type | Weight Gain (%) | ||
|---|---|---|---|
| 30 min | 60 min | 90 min | |
| Ramie L-Isolated | 12.38 ± 1.69 | 15.17 ± 0.36 | 15.93 ± 2.43 |
| Ramie L-MEOH | 6.25 ± 1.10 | 7.21 ± 3.17 | 8.62 ± 1.09 |
| Ramie L-Ac | 6.68 ± 0.74 | 8.26 ± 0.06 | 9.07 ± 0.62 |
| Type | Impregnation Time (min) | Modulus of Elasticity (GPa) | Std. Dev | Tensile Strength (MPa) | Std. Dev |
|---|---|---|---|---|---|
| Ramie | - | 10.45 | 0.2402 | 397.72 | 58.26 |
| Ramie-L-isolated | 30 | 15.23 | 0.4169 | 441.19 | 226.37 |
| 60 | 20.35 | 0.6712 | 497.22 | 91.62 | |
| 90 | 31.10 | 0.6705 | 574.11 | 133.97 | |
| Ramie-L-MeOH | 30 | 13.06 | 0.4446 | 447.06 | 58.59 |
| 60 | 11.98 | 0.2780 | 406.71 | 175.64 | |
| 90 | 17.86 | 0.5629 | 461.32 | 106.13 | |
| Ramie-L-Ac | 30 | 16.78 | 0.2554 | 523.38 | 80.12 |
| 60 | 19.81 | 0.2923 | 547.66 | 91.37 | |
| 90 | 21.99 | 0.5288 | 577.61 | 68.87 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Handika, S.O.; Lubis, M.A.R.; Sari, R.K.; Laksana, R.P.B.; Antov, P.; Savov, V.; Gajtanska, M.; Iswanto, A.H. Enhancing Thermal and Mechanical Properties of Ramie Fiber via Impregnation by Lignin-Based Polyurethane Resin. Materials 2021, 14, 6850. https://doi.org/10.3390/ma14226850
Handika SO, Lubis MAR, Sari RK, Laksana RPB, Antov P, Savov V, Gajtanska M, Iswanto AH. Enhancing Thermal and Mechanical Properties of Ramie Fiber via Impregnation by Lignin-Based Polyurethane Resin. Materials. 2021; 14(22):6850. https://doi.org/10.3390/ma14226850
Chicago/Turabian StyleHandika, Sucia Okta, Muhammad Adly Rahandi Lubis, Rita Kartika Sari, Raden Permana Budi Laksana, Petar Antov, Viktor Savov, Milada Gajtanska, and Apri Heri Iswanto. 2021. "Enhancing Thermal and Mechanical Properties of Ramie Fiber via Impregnation by Lignin-Based Polyurethane Resin" Materials 14, no. 22: 6850. https://doi.org/10.3390/ma14226850
APA StyleHandika, S. O., Lubis, M. A. R., Sari, R. K., Laksana, R. P. B., Antov, P., Savov, V., Gajtanska, M., & Iswanto, A. H. (2021). Enhancing Thermal and Mechanical Properties of Ramie Fiber via Impregnation by Lignin-Based Polyurethane Resin. Materials, 14(22), 6850. https://doi.org/10.3390/ma14226850









