Auxetic Structures for Tissue Engineering Scaffolds and Biomedical Devices
Abstract
:1. Introduction
2. Definition, Design, and Fabrication of Auxetic Structures
2.1. Definition of Auxetic Property
2.2. Design of Auxetic Structures
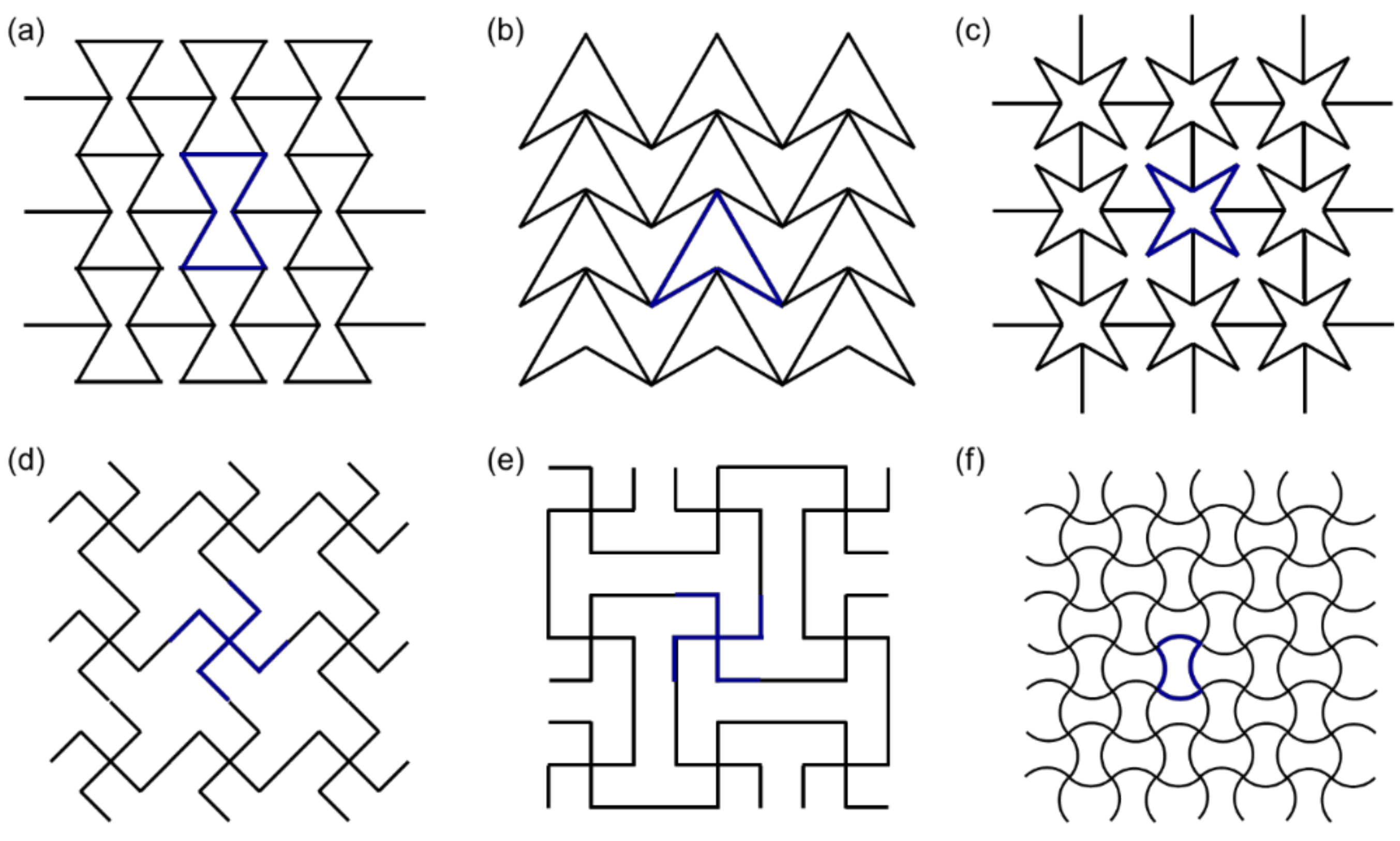
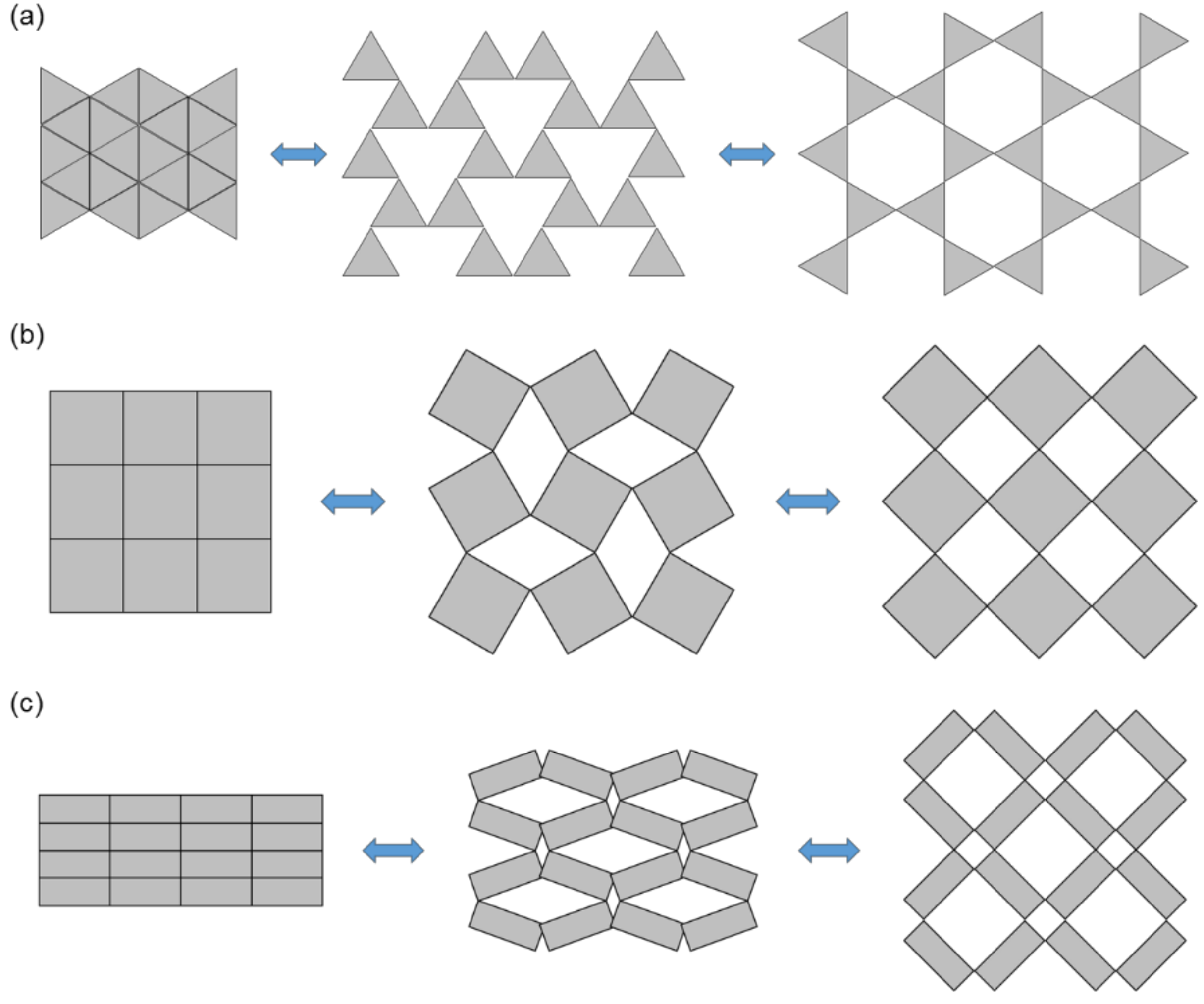
2.2.1. Re-Entrant Unit Cells
2.2.2. Chiral Unit Cells
2.2.3. Rotating Unit Cells
2.3. Fabrication of Auxetic Structures
2.3.1. Fabrication Using Additive Manufacturing
2.3.2. Fabrication Using Soft Lithography
2.3.3. Fabrication Using Machining Technology
2.3.4. Fabrication Using Compressed Foam
2.3.5. Fabrication Using Textile Technology
3. Auxetic Structures as a Tissue Engineering Scaffold
3.1. Additive Manufacturing-Based Auxetic Scaffold
3.2. Machining-Based Auxetic Scaffold
3.3. Auxetic Foam Scaffold
3.4. Auxetic Textile Scaffold
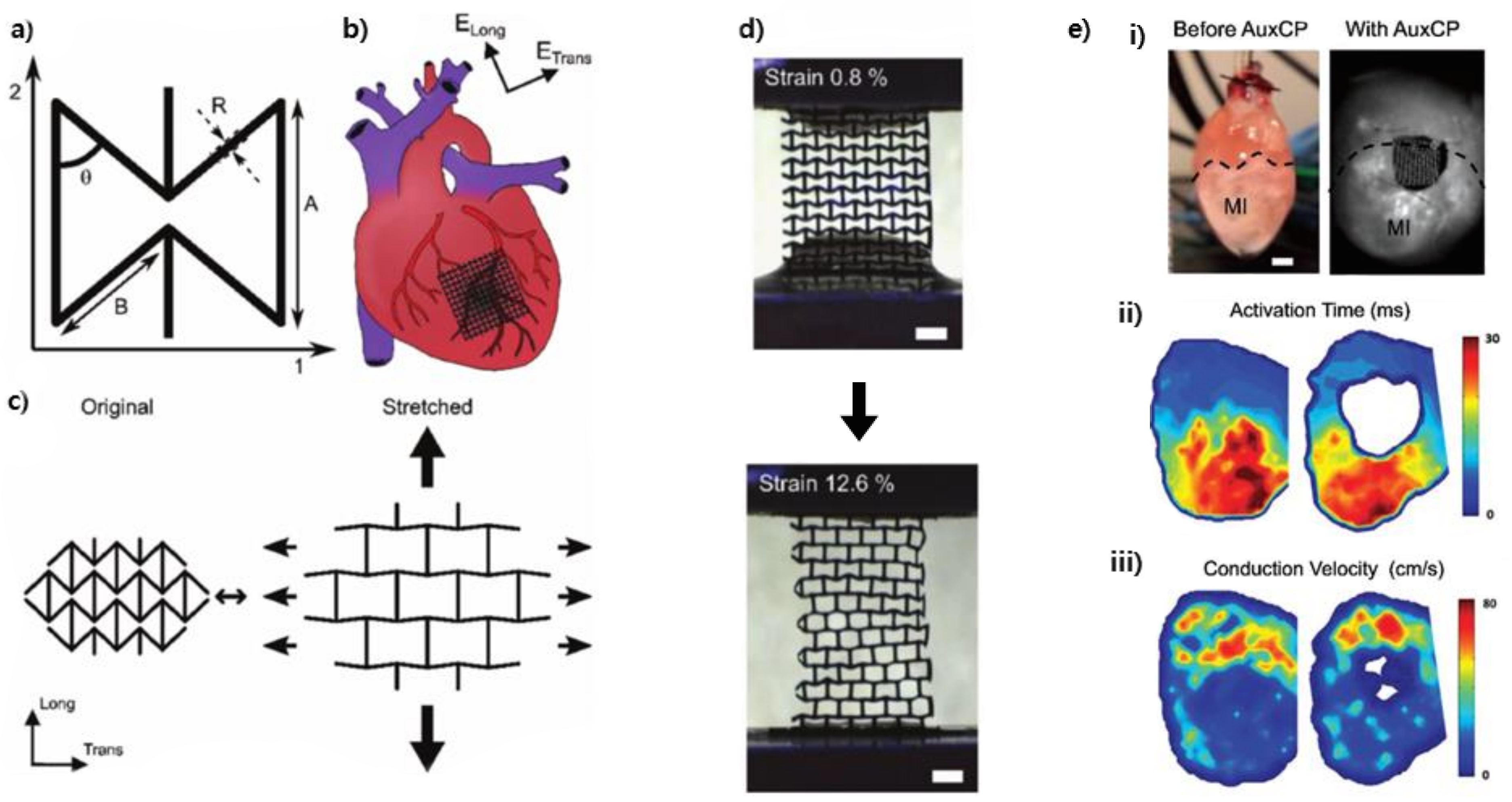
3.5. Auxetic Biomedical Devices
4. Prospect of Auxetic Structures
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Park, Y.J.; Kim, J.K. The effect of negative Poisson’s ratio polyurethane scaffolds for articular cartilage tissue engineering applications. Adv. Mater. Sci. Eng. 2013, 2013, 853289. [Google Scholar] [CrossRef] [Green Version]
- Ghasemi-Mobarakeh, L.; Prabhakaran, M.P.; Morshed, M.; Nasr-Esfahani, M.H.; Ramakrishna, S. Electrospun poly(epsilon-caprolactone)/gelatin nanofibrous scaffolds for nerve tissue engineering. Biomaterials 2008, 29, 4532–4539. [Google Scholar] [CrossRef] [PubMed]
- Shuai, C.; Yang, W.; Feng, P.; Peng, S.; Pan, H. Accelerated degradation of HAP/PLLA bone scaffold by PGA blending facilitates bioactivity and osteoconductivity. Bioact. Mater. 2021, 6, 490–502. [Google Scholar] [CrossRef]
- Cho, Y.S.; Gwak, S.J.; Cho, Y.S. Fabrication of polycaprolactone/nano hydroxyapatite (PCL/nHA) 3D scaffold with enhanced in vitro cell response via design for additive manufacturing (DfAM). Polymers 2021, 13, 1394. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.W. 3D nanoprinting technologies for tissue engineering applications. J. Nanomater. 2015, 2015, 213521. [Google Scholar] [CrossRef]
- Lakes, R.S. Foam structures with a negative Poisson’s ratio. Science 1987, 235, 1038–1040. [Google Scholar] [CrossRef]
- Evans, K.E. Design of doubly-curved sandwich panels with honeycomb cores. Compos. Struct. 1991, 17, 95–111. [Google Scholar] [CrossRef]
- He, C.B.; Liu, P.W.; McMullan, P.J.; Griffin, A.C. Toward molecular auxetics: Main chain liquid crystalline polymers consisting of laterally attached para-quaterphenyls. Phys. Status Solidi B 2005, 242, 576–584. [Google Scholar] [CrossRef]
- Liu, Y.; Hu, H. A review on auxetic structures and polymeric materials. Sci. Res. Essays 2010, 5, 1052–1063. [Google Scholar]
- Zadpoor, A.A. Mechanical meta-materials. Mater. Horiz. 2016, 3, 365–462. [Google Scholar] [CrossRef] [Green Version]
- Mardling, P.; Alderson, A.; Jordan-Mahy, N.; Lyn Le Maitre, C. The use of auxetic materials in tissue engineering. Biomater. Sci. 2020, 8, 2074. [Google Scholar] [CrossRef] [PubMed]
- Ahn, C.B.; Kim, J.H.; Lee, J.H.; Park, K.Y.; Son, K.H.; Lee, J.W. Development of multi-layer tubular vascular scaffold to enhance compliance by exhibiting a negative Poisson’s ratio. Int. J. Pr. Eng. Man.-GT 2021, 8, 841–853. [Google Scholar] [CrossRef]
- Kapnisi, M.; Mansfield, C.; Marijon, C.; Guex, A.G.; Perbellini, F.; Bardi, I.; Humphrey, E.J.; Puetzer, J.L.; Mawad, D.; Koutsogeorgis, D.C.; et al. Auxetic cardiac patches with tunable mechanical and conductive properties toward treating myocardial infarction. Adv. Funct. Mater. 2018, 28, 1800618. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sanami, M. Auxetic Materials for Biomedical Applications. Ph.D. Thesis, University of Bolton, Bolton, UK, 2015. [Google Scholar]
- Alderson, A.; Alderson, K.L.; Sanami, M. Bone Implant Comprising Auxetic Material. GB2495272A. Br. Patent Application No. 1116625.3, 2013. [Google Scholar]
- Yao, Y.; Wang, L.; Li, J.; Tian, S.; Zhang, M.; Fan, Y. A novel auxetic structure based bone screw design: Tensile mechanical characterization and pullout fixation strength evaluation. Mater. Des. 2020, 188, 108424. [Google Scholar] [CrossRef]
- Ghavidelnia, N.; Bodaghi, M.; Hedayati, R. Femur auxetic meta-implants with tuned micromotion distribution. Materials 2021, 14, 114. [Google Scholar] [CrossRef] [PubMed]
- Popov, E.P. Engineering Mechanics of Solid, 1st ed.; Prentice Hall: Hoboken, NJ, USA, 1990; pp. 82–83. [Google Scholar]
- Eisenstadt, M.M. Introduction to Mechanical Properties of Materials, 1st ed.; Macmillan: New York, NY, USA, 1971; p. 444. [Google Scholar]
- Walpole, L.J. Fourth-rank tensors of the thirty-two crystal classes: Multiplication tables. Proc. R. Soc. Lond. A Math. Phys. Eng. Sci. 1984, 391, 149–179. [Google Scholar]
- Rychlewski, J. On Hooke’s law. J. Appl. Math. Mech. 1984, 48, 303–314. [Google Scholar] [CrossRef]
- Wojciechowski, K.W. Poisson’s ratio of anisotropic systems. Comput. Methods Sci. Technol. 2005, 11, 73–79. [Google Scholar] [CrossRef]
- Wake, M.C.; Gupta, P.K.; Mikos, A.G. Fabrication of pliable biodegradable polymer foams to engineer soft tissues. Cell Transplant. 1996, 5, 465–473. [Google Scholar] [CrossRef] [PubMed]
- Gibson, L.J.; Ashby, M.F.; Schajer, G.S.; Robertson, C.I. The mechanics of two dimensional cellular materials. Proc. Lond. R. Soc. 1982, 382, 25–42. [Google Scholar]
- Prall, D.; Lakes, R.S. Properties of a chiral honeycomb with a Poisson’s ratio of −1. Int. J. Mech. Sci. 1997, 39, 305–314. [Google Scholar] [CrossRef]
- Grima, J.N.; Evans, K.E. Auxetic behaviour from rotating squares. J. Mater. Sci. Lett. 2000, 19, 1563–1565. [Google Scholar] [CrossRef]
- Larsen, U.D.; Sigmund, O.; Bouwstra, S. Design and fabrication of compliant mechanisms and material structures with negative Poisson’s ratio. J. Microelectromech. Syst. 1997, 6, 99–106. [Google Scholar] [CrossRef] [Green Version]
- Theocaris, P.S.; Stavroulakis, G.E.; Panagiotopoulos, P.D. Negative Poisson’s ratio in materials with a star-shaped microstructure. A numerical homogenization approach. Arch. Appl. Mech. 1997, 67, 274–286. [Google Scholar] [CrossRef]
- Grima, J.N.; Gatt, R.; Alderson, A.; Evans, K.E. On the potential of connected stars as auxetic systems. Mol. Simul. 2005, 31, 925–935. [Google Scholar] [CrossRef] [Green Version]
- Smith, C.W.; Grima, J.N.; Evans, K.E. A novel mechanism for generating auxetic behaviour in reticulated foams: Missing rib foam model. Acta Mater. 2000, 48, 4349–4356. [Google Scholar] [CrossRef]
- Gaspar, N.; Ren, X.J.; Smith, C.W.; Grima, J.N.; Evans, K.E. Novel honeycombs with auxetic behaviour. Acta Mater. 2005, 53, 2439–2445. [Google Scholar] [CrossRef]
- Dolla, W.J.S.; Fricke, B.A.; Becker, B.R. Structural and drug diffusion models of conventional and auxetic drug-eluting stents. J. Med. Devices 2007, 1, 47–55. [Google Scholar] [CrossRef]
- Grima, J.N.; Gatt, R.; Farrugia, P.S. On the properties of auxetic meta-tetrachiral structures. Phys. Status Solidi B 2008, 245, 511–520. [Google Scholar] [CrossRef]
- Fu, M.-H.; Zheng, B.-B.; Li, W.-H. A novel chiral three-dimensional material with negative Poisson’s ratio and the equivalent elastic parameters. Compos. Struct. 2017, 176, 442–448. [Google Scholar] [CrossRef]
- Grima, J.N.; Alderson, A.; Evans, K.E. Auxetic behaviour from rotating rigid units. Phys. Status Solidi B 2005, 242, 561–575. [Google Scholar] [CrossRef]
- Grima, J.N.; Evans, K.E. Auxetic behavior from rotating triangles. J. Mater. Sci. 2006, 41, 3193–3196. [Google Scholar] [CrossRef]
- Alderson, A.; Evans, K.E. Molecular origin of auxetic behavior in tetrahedral framework Silicates. Phys. Rev. Lett. 2002, 89, 225503. [Google Scholar] [CrossRef]
- Gatt, R.; Mizzi, L.; Azzopardi, J.I.; Azzopardi, K.M.; Attard, D.; Casha, A.; Briffa, J.; Grima, J.N. Hierarchical auxetic mechanical metamaterials. Sci. Rep. 2015, 5, 8395. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fozdar, D.Y.; Soman, P.; Lee, J.W.; Han, L.-H.; Chen, S. Three-dimensional polymer constructs exhibiting a tunable negative Poisson ratio. Adv. Funct. Mater. 2011, 21, 2712–2720. [Google Scholar] [CrossRef]
- Soman, P.; Lee, J.W.; Phadke, A.; Varghese, S.; Chen, S. Spatial tuning of negative and positive Poisson’s ratio in a multi-layer scaffold. Acta Biomater. 2012, 8, 2587–2594. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soman, P.; Fozdar, D.Y.; Lee, J.W.; Phadke, A.; Varghese, S.; Chen, S. A three-dimensional polymer scaffolding material exhibiting a zero Poisson’s ratio. Soft Matter 2012, 8, 4946–4951. [Google Scholar] [CrossRef] [Green Version]
- Zhang, W.; Soman, P.; Meggs, K.; Qu, X.; Chen, S. Tuning the Poisson’s ratio of biomaterials for investigating cellular response. Adv. Funct. Mater. 2013, 23, 3226–3232. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Easey, N.; Chuprynyuk, D.; Wan Musa, W.M.S.; Bangs, A.; Dobah, Y.; Shterenlikht, A.; Scarpa, F. Dome-shape auxetic cellular metamaterials: Manufacturing, modeling, and testing. Front. Mater. 2019, 6, 86. [Google Scholar] [CrossRef] [Green Version]
- Wu, Z.; Zhao, J.; Wu, W.; Wang, P.; Wang, B.; Li, G.; Zhang, S. Radial compressive property and the proof-of-concept study for realizing self-expansion of 3D printing polylactic acid vascular stents with negative Poisson’s ratio structure. Materials 2018, 11, 1357. [Google Scholar] [CrossRef] [Green Version]
- Ebrahimi, H.; Mousanezhad, D.; Nayeb-Hashemi, H.; Norato, J.; Vaziri, A. 3D cellular metamaterials with planar anti-chiral topology anti-chiral topology. Mater. Des. 2018, 145, 226–231. [Google Scholar] [CrossRef]
- Bückmann, T.N.; Stenger, T.; Kadic, M.; Kaschke, J.; Frölich, A.; Kennerknecht, T.; Eberl, C.; Thiel, M.; Wegener, M. Tailored 3D mechanical metamaterials made by dip-in direct-laser-writing optical lithography. Adv. Mater. 2012, 24, 2710–2714. [Google Scholar] [CrossRef] [PubMed]
- Hengsbach, S.; Lantada, A.D. Direct laser writing of auxetic structures: Present capabilities and challenges. Smart Mater. Struct. 2014, 23, 085033. [Google Scholar] [CrossRef]
- Yuan, S.; Shen, F.; Bai, J.; Chua, C.K.; Wei, J.; Zhou, K. 3D soft auxetic lattice structures fabricated by selective laser sintering: TPU powder evaluation and process optimization. Mater. Des. 2017, 120, 317–327. [Google Scholar] [CrossRef]
- Remennikov, A.; Kalubadanage, D.; Ngo, T.; Mendis, P.; Alici, G.; Whittaker, A. Development and performance evaluation of large-scale auxetic protective systems for localized impulsive loads. Int. J. Protect. Struct. 2019, 10, 390–417. [Google Scholar] [CrossRef]
- Park, Y.; Vella, G.; Loh, K.J. Bio-inspired active skins for surface morphing. Sci. Rep. 2019, 9, 18609. [Google Scholar] [CrossRef] [PubMed]
- Attar, M.; Aydin, S.S.; Arabaci, A.; Mutlu, I. Mechanical meta-material-based polymer skin graft production by rapid prototyping and replica method. Rapid Prototyp. J. 2021, 27, 278–287. [Google Scholar] [CrossRef]
- Xu, B.; Arias, F.; Brittain, S.T.; Zhao, X.-M.; Grzybowski, B.; Torquato, S.; Whitesides, G.M. Making negative Poisson’s ratio microstructures by soft lithography. Adv. Mater. 1999, 11, 1186–1189. [Google Scholar] [CrossRef]
- Muslija, A.; Lantada, A.D. Deep reactive ion etching of auxetic structures: Present capabilities and challenges. Smart Mater. Struct. 2014, 23, 087001. [Google Scholar] [CrossRef] [Green Version]
- Lantada, A.D.; Muslija, A.; García-Ruíz, J.P. Auxetic tissue engineering scaffolds with nanometric features and resonances in the megahertz range. Smart Mater. Struct. 2015, 24, 055013. [Google Scholar] [CrossRef]
- Bhullar, S.K.; Rana, D.; Lekesiz, H.; Bedeloglu, A.C.; Ko, J.; Cho, Y.; Aytac, Z.; Uyar, T.; Jun, M.; Ramalingam, M. Design and fabrication of auxetic PCL nanofiber membranes for biomedical applications. Mater. Sci. Eng. C 2017, 81, 334–340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burns, S. Negative Poisson’s ratio materials. Science 1987, 238, 551. [Google Scholar] [CrossRef] [PubMed]
- Chen, J. A Biodegradable Knitted Cardiac Patch for Myocardium Regeneration Using Cardiosphere-Derived Cells (CDCs). Master’s Thesis, NC State University, Raleigh, NC, USA, 2015. [Google Scholar]
- Zhang, F. Studies of Tissue-Engineered Vascular Grafts Fabricated from Electro-Chemically Aligned Collagen Yarns and Electrospun Collagen Nanofibers. Master’s Thesis, NC State University, Raleigh, NC, USA, 2018. [Google Scholar]
- He, T. A Study of Three Dimensional Warp Knits for Novel Applications as Tissue Engineering Scaffolds. Master’s Thesis, NC State University, Raleigh, NC, USA, 2011. [Google Scholar]
- Gajjar, C.R.; King, M.W. Resorbable Fiber-Forming Polymers for Biotextile Applications, 1st ed.; Springer: Cham, Switzerland, 2014. [Google Scholar]
- Wang, Z.; Hu, H. Tensile and forming properties of auxetic warp-knitted spacer fabrics. Text. Res. J. 2016, 87, 1925–1937. [Google Scholar] [CrossRef]
- Ge, Z.; Hu, H. A theoretical analysis of deformation behavior of an innovative 3D auxetic textile structure. J. Text. Inst. 2015, 106, 101–109. [Google Scholar] [CrossRef]
- Ahmed, H.I.; Umair, M.; Nawab, Y.; Hamdani, S.T.A. Development of 3D auxetic structures using para-aramid and ultra-high molecular weight polyethylene yarns. J. Text. Inst. 2020, 112, 1417–1427. [Google Scholar] [CrossRef]
- Warner, J.J.; Gillies, A.R.; Hwang, H.H.; Zhang, H.; Lieber, R.L.; Chen, S. 3D-printed biomaterials with regional auxetic properties. J. Mech. Behav. Biomed. Mater. 2017, 76, 145–152. [Google Scholar] [CrossRef]
- Lee, J.W.; Soman, P.; Park, J.H.; Chen, S.; Cho, D.-W. A tubular biomaterial construct exhibiting a negative Poisson’s ratio. PLoS ONE 2016, 11, e1055681. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Xie, C.; Gao, Q.; Zhou, X.; Li, G.; Du, J.; He, Y. Fabrication of multi-scale and tunable auxetic scaffolds for tissue engineering. Mater. Des. 2021, 197, 109277. [Google Scholar] [CrossRef]
- Flamourakis, G.; Spanos, I.; Vangelatos, Z.; Manganas, P.; Papadimitriou, L.; Grigoropoulos, C.; Ranella, A.; Farsari, M. Laser-made 3D auxetic metamaterial scaffolds for tissue engineering applications. Macromol. Mater. Eng. 2020, 305, 2000238. [Google Scholar] [CrossRef]
- Ahn, C.B.; Son, K.H.; Yu, Y.S.; Kim, T.H.; Lee, J.I.; Lee, J.W. Development of a flexible 3D printed scaffold with a cell-adhesive surface for artificial trachea. Biomed. Mater. 2019, 14, 055001. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.S.; Ahn, C.B.; Son, K.H.; Lee, J.W. Motility improvement of biomimetic trachea scaffold via hybrid 3D-bioprinting technology. Polymers 2021, 13, 971. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.J.; Lee, J.J.; Lee, J.B.; Sung, H.-J.; Shin, J.-W.; Shin, J.W.; Wu, Y.; Kim, J.K. MG-63 cells proliferation following various types of mechanical stimulation on cells by auxetic hybrid scaffolds. Biomater. Res. 2016, 20, 32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choi, H.J.; Lee, J.J.; Park, Y.J.; Shin, J.-W.; Sung, H.-J.; Shin, J.W.; Wu, Y.; Kim, J.K. MG-63 osteoblast-like cell proliferation on auxetic PLGA scaffold with mechanical stimulation for bone tissue regeneration. Biomater. Res. 2016, 20, 33. [Google Scholar] [CrossRef] [Green Version]
- Kaspar, D.; Seidl, W.; Neidlinger-Wilke, C.; Beck, A.; Claes, L.; Ignatius, A. Proliferation of human-derived osteoblast-like cells depends on the cycle number and frequency of uniaxial strain. J. Biomech. 2002, 35, 873–880. [Google Scholar] [CrossRef]
- Song, L.; Ahmed, M.F.; Li, Y.; Zeng, C.; Li, Y. Vascular differentiation from pluripotent stem cells in 3-D auxetic scaffolds. J. Tissue Eng. Regener. Med. 2018, 12, 1679–1689. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Li, Y.; Song, L.; Zeng, C.; Li, Y. Pluripotent stem cell expansion and neural differentiation in 3-D scaffolds of tunable Poisson’s ratio. Acta Biomater. 2017, 49, 192–203. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deshpande, M.V.; West, A.J.; Bernacki, S.H.; Luan, K.; King, M.W. Poly(e-caprolactone) resorbable auxetic designed knitted scaffolds for craniofacial skeletal muscle regeneration. Bioengineering 2020, 7, 134. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Hassanin, H.; Attallah, M.M.; Adkins, N.J.E.; Essa, K. The development of TiNi-based negative Poisson’s ratio structure using selective laser melting. Acta Mater. 2016, 105, 75–83. [Google Scholar] [CrossRef] [Green Version]
- Burriesci, G.; Bergamasco, G. Annuloplasty Prosthesis with an Auxetic Structure. EP1803420B1. Eur. Patent Application 05425926.2, 2009. [Google Scholar]
- Scarpa, F. Auxetic materials for bioprostheses. IEEE Signal Process. Mag. 2008, 25, 126–128. [Google Scholar] [CrossRef]
- Martz, E.O.; Lakes, R.S.; Goel, V.K.; Park, J.B. Design of an artificial intervertebral disc exhibiting a negative poisson’s ratio. Cell. Polym. 2005, 24, 127–138. [Google Scholar] [CrossRef]
- Wang, Y.; Arabnejad, S.; Tanzer, M.; Pasini, D. Hip implant design with three-dimensional porous architecture of optimized graded density. J. Mech. Des. 2018, 140, 111406. [Google Scholar] [CrossRef] [Green Version]
- Fraldi, M.; Esposito, L.; Perrella, G.; Cutolo, A.; Cowin, S.C. Topological optimization in hip prosthesis design. Biomech. Model. Mechanobiol. 2010, 9, 389–402. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Wang, Y.; Yan, J.; Liu, T.; Wang, S. Topology optimization of total femur structure: Application of parameterized level set method under geometric constraints. J. Mech. Des. 2016, 138, 011402. [Google Scholar] [CrossRef]
- Bieger, R.; Ignatius, A.; Decking, R.; Claes, L.; Reichel, H.; Dürselen, L. Primary stability and strain distribution of cementless hip stems as a function of implant design. Clin. Biomech. 2012, 27, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-M.; Tsai, W.-C.; Lin, S.-C.; Tseng, C.-S. A cementless, proximally fixed anatomic femoral stem induces high micromotion with nontraumatic femoral avascular necrosis: A finite element study. J. Orthop. Transl. 2014, 2, 149–156. [Google Scholar] [CrossRef] [Green Version]
- Dirrenberger, J.; Forest, S.; Jeulin, D. Effective elastic properties of auxetic microstructures: Anisotropy and structural applications. Int. J. Mech. Mater. Des. 2013, 9, 21–33. [Google Scholar] [CrossRef]
- Kolken, H.M.A.; Janbaz, S.; Leeflang, S.M.A.; Lietaert, K.; Weinans, H.H.; Zadpoor, A.A. Rationally designed meta-implants: A combination of auxetic and conventional meta-biomaterials. Mater. Horiz. 2018, 5, 28–35. [Google Scholar] [CrossRef] [Green Version]
- Zadpoor, A.A. Mechanical performance of additively manufactured meta-biomaterials. Acta Biomater. 2019, 85, 41–59. [Google Scholar] [CrossRef]
- Panico, M.; Langella, C.; Santulli, C. Development of a biomedical neckbrace through tailored auxetic shapes. Ital. J. Sci. Eng. 2017, 1, 105–117. [Google Scholar] [CrossRef]
- Ali, M.N.; Rehman, I.U. An Auxetic structure configured as oesophageal stent with potential to be used for palliative treatment of oesophageal cancer; development and in vitro mechanical analysis. J. Mater. Sci. Mater. Med. 2011, 22, 2573–2581. [Google Scholar] [CrossRef] [PubMed]
- Bhullar, S.K.; Ko, J.; Cho, Y.; Jun, M.B.G. Fabrication and characterization of non-woven auxetic polymer stent. Polym.-Plast. Technol. 2015, 54, 1553–1559. [Google Scholar] [CrossRef]
- Tan, T.W.; Douglas, G.R.; Bond, T.; Phani, A.S. Compliance and longitudinal strain of cardiovascular stents: Influence of cell geometry. J. Med. Devices 2011, 5, 041002. [Google Scholar] [CrossRef]
- Gatt, R.; Caruana-Gauci, R.; Attard, D.; Casha, A.R.; Wolak, W.; Dudek, K.; Mizzi, L.; Grima, J.N. On the properties of real finitesized planar and tubular stent-like auxetic structures. Phys. Status Solidi B 2014, 251, 321–327. [Google Scholar] [CrossRef]




| Author | Fabrication Technology | Specific Fabrication Method | Material | Cell Type | Biological Effect |
| Soman et al. [40] (2012) | Additive manufacturing | Dynamic optical projection stereolithography | PEGDA | HMSC (human bone marrow) | Grew on scaffold |
| Soman et al. [41] (2012) | Dynamic optical projection stereolithography | PEGDA | HMSC (human bone marrow) | Grew on scaffold | |
| Zhang et al. [42] (2013) | Two-photon stereolithography | PEGDA | 10T1/2 (mouse embryonic fibroblast) | Unable to divide | |
| Warner et al. [64] (2017) | Dynamic optical projection stereolithography | Polyurethane | 10T1/2 (Mouse embryonic fibroblast) C2C12 (mouse myoblast) | Grew on scaffold | |
| Lee et al. [65] (2016) | Micro-stereolithography | PEGDA | HTMSC (human turbinate mesenchymal stromal cell) | Proliferation increased on NPR | |
| Jin et al. [66] (2021) | Fused deposition modelling | poly(ε-caprolactone) | HUVEC (human umbilical vein endothelial cell) BMSC (bone marrow stem cell) | Adhered and grew on scaffold | |
| Ahn et al. [68] (2019) | Fused deposition modelling + electrospinning | Thermoplastic Polyurethane | HTMSC (human turbinate mesenchymal stromal cell) | Grew on scaffold | |
| Ahn et al. [12] (2019) | Additive manufacturing + electrospinning | Fused deposition modelling | poly(ε-caprolactone) | HUVEC (human umbilical vein endothelial cell) VSMC (vascular smooth muscle cell) | Made multi-layers |
| Yu et al. [69] (2021) | Fused deposition modelling + electrospinning | poly(ε-caprolactone) | - | Attached with the printed scaffold and hydrogel | |
| Muslija et al. [53] (2021) | Soft lithography | Deep reactive ion etching | Silicon | - | - |
| Lantada et al. [54] (2015) | Deep reactive ion etching | Silicon | HMSC (human mesenchymal stem cell) | Interacted at a sub-cellular level | |
| Kapnisi et al. [13] (2018) | Machining | Micro-ablation | Chitosan (polyaniline coating) | Neonatal rat ventricular myocytes and fibroblasts | Grew on scaffold (cytocompatibility) |
| Park et al. [1] (2013) | Foaming | Compressed foams | Polyurethane | Chondrocytes (primary from cartilage) | Proliferation increased |
| Choi et al. [70] (2016) | Solvent casting/salt leaching | HA/PLGA | MG-63 (human Osteoblast) | Proliferation increased | |
| Choi et al. [71] (2016) | Solvent casting/salt leaching | PLGA | MG-63 (human Osteoblast) | Proliferation increased | |
| Song et al. [73] (2018) | Heated foams | Polyurethane and polyester | ES-D3 (mouse embryonic stem cell) iPSK3 (human induced pluripotent stem cell) | Vascular differentiation increased | |
| Yan et al. [74] (2017) | Compressed and heated foams | Polyurethane | ES-D3 (mouse embryonic stem cell) iPSK3 (human induced pluripotent stem cell) | Neural differentiation increased | |
| Deshpande et al. [75] (2020) | Textile | Fabric knitting | poly(ε-caprolactone) | Human dermal fibroblasts | Cell metabolic activity increased |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, Y.; Son, K.H.; Lee, J.W. Auxetic Structures for Tissue Engineering Scaffolds and Biomedical Devices. Materials 2021, 14, 6821. https://doi.org/10.3390/ma14226821
Kim Y, Son KH, Lee JW. Auxetic Structures for Tissue Engineering Scaffolds and Biomedical Devices. Materials. 2021; 14(22):6821. https://doi.org/10.3390/ma14226821
Chicago/Turabian StyleKim, Yujin, Kuk Hui Son, and Jin Woo Lee. 2021. "Auxetic Structures for Tissue Engineering Scaffolds and Biomedical Devices" Materials 14, no. 22: 6821. https://doi.org/10.3390/ma14226821
APA StyleKim, Y., Son, K. H., & Lee, J. W. (2021). Auxetic Structures for Tissue Engineering Scaffolds and Biomedical Devices. Materials, 14(22), 6821. https://doi.org/10.3390/ma14226821







