Electrophoretic Deposition of Biocompatible and Bioactive Hydroxyapatite-Based Coatings on Titanium
Abstract
:1. Introduction
2. Engineering Implant Surfaces to Prevent Microbial Adhesion and Infection
- Poly(lactide-co-glycolide) (PLGA)—PLGA in combination with the natural polymer chitosan, applied as a stent coating, can reduce platelet adhesion [35], while the combination of PLGA with HAP and the antibiotic atorvastatin can be applicable in bone tissue engineering as injectable PLGA micro-particulate system [36].
- Poly(glycolic acid) (PGA)—in combination with hydroxyapatite, osteoblast differentiation and mineralized bone matrix formation can be increased after implantation of PGA/HAP composites [37].
- Polyether ether ketone (PEEK)—regardless of the fact that PEEK is a bioinert material, its excellent mechanical and chemical properties in addition to the fact that it does not induce positive/negative body reaction, make it a widely used material as a bone substitute, e.g., in dental implants, etc. Three-component coatings (PEEK, hydroxyapatite and chitosan) have efficient antibacterial properties against the microorganisms E. coli as well as S. aureus [38].
- Poly(lactic acid) (PLA)—biocompatible polymer often used because of its good biodegradability, high elastic modulus as well as the fact that the final degradation products of PLA are water and carbon dioxide, non-toxic or carcinogenic. Additionally, PLA has higher elastic modulus than natural cancellous bone [39]. Mechanical characteristics of composite coatings, e.g., elongation at break, substantially increased in hydroxyapatite nanorod network in the poly(lactic acid) [40].
- Poly-L-lactic acid (PLLA)—homogenous dispersion of hydroxyapatite nanoparticles and microcrystalline cellulose in a PLLA matrix led to the formation of nanocomposite material that is, according to the composition, structure and mechanical characteristics, comparable with the trabecular bone [41].
- Poly-caprolactone (PCL)—can be used as a drug delivery device and biomaterial for regenerative medicine. PCL can be employed as the binding agent for PCL-hydroxyapatite scaffold preparation. Poly-caprolactone/hydroxyapatite scaffold with heparin sulfate showed positive effects on the differentiation of osteoblasts, accelerating the repair of biological bone defects [42].
- Poly(vinyl alcohol) (PVA)—is known as a flexible, biocompatible, and biodegradable polymer, having a high tensile strength. PVA has the ability to form composites with chitosan, leading to formation of nanofibrous polymer matrix for hydroxyapatite incorporation, mimicking native extracellular matrix for bone tissue repairing. It was demonstrated that the nanofibrous scaffold of PVA/chitosan/hydroxyapatite provides versatile surface for attachment and proliferation of the osteoblast cells [43].
- Polymethyl methacrylate (PMMA) is an acrylic material that is widely used in dentistry due to its high tensile and flexural modulus. Addition of hydroxyapatite into PMMA leads to formation of composite material with improved characteristics, e.g., the density, and tensile and flexural modulus of PMMA increased significantly after incorporation of hydroxyapatite [44].
- Natural polymers, such as collagen, fibrinogen, hyaluronic acid, elastin, alginate, chondroitin sulfate, lignin, and chitosan can be used to obtain materials for hard and soft tissue repair [45]. The efficiency of the natural polymers’ application in tissue engineering is reflected in the possibilities to be recognized by host cells, due to the specific amino-acid sequences in their structure [46]. Additionally, the polymers are able to promote tissue healing and integration, through the induction of biochemical signals that trigger cell migration, proliferation, and differentiation [47]. Collagen sponge material, for example, can provide cell growth and promote the nutrients absorption. In clinical practice, the application of the collagen sponge culture system combined with the Rotary Cell Culture System is a promising tool for tissue engineering [48]. Fibrinogen is the major plasma glycoprotein coagulation factor. Its role is to facilitate the adhesion, spreading and aggregation of the cells. Addition of fibrinogen to the hydroxyapatite nanoparticles improves the cell adhesion [49]. The two predominant proteins in the body, responsible for modulating biological and mechanical properties of tissues, are elastin and collagen [50]. Elastin, found in connective tissue, is a bioactive protein, very often used in tissue engineering. A composite of elastin and collagen, can be applicable in vascular tissue engineering, e.g., tubular polymer scaffolds. Since the blood vessels are predominantly fibrous, composed of collagen and elastin, the combination of these two polymers with polyurethane, was employed in the production of fibrous scaffolds. The obtained scaffold was hydrophilic and cell viability was proven for 72 h [51]. The natural ionic mucopolysaccharide with linear structure hyaluronic acid, possesses multiple active groups (carboxyl, hydroxyl and amino groups) that can be further chemically modified, expanding the applications of hyaluronic acid in the biomedical field [52]. Oxidized hyaluronic acid, due to its lower cytotoxicity, was investigated as a potential substitute of glutaraldehyde, as a fixation component in the abdominal wall repair surgery [53]. Low toxicity, degradability in the physiologic conditions, high affinity to calcium ions, and hydrophilic nature, make alginate as potent candidate material in tissue engineering [54]. Microencapsulation of hydroxyapatite in scaffolds obtained by the combination of alginate and gelatin, results in a promising material to induce osteogenic differentiation and to improve cell proliferation [55]. Chondroitin sulfate is known to facilitate cell proliferation and for having antithrombogenic activity. By combination of chondroitin sulfate and collagen, vascular scaffold with antithrombosis and endothelialization function can be obtained [56]. One of the most widely used natural polymers, chitosan (CS), possesses all the main characteristics required for biomedical application as a drug carrier, or a component of the biocompatible coating or repair of hard and soft tissues [57]. The most valuable characteristic of CS, as a biomaterial approved by Food and Drug Administration [58,59] are: biocompatibility [60], biodegradability [61], antibacterial activity [62,63], and high potential in drug delivery systems [64,65]. The tissue response upon implantation is reduced to a minimum due to CS presence, thanks to its hydrophilic surface that improves cell adhesion, differentiation and proliferation [66]. Additionally, it was shown that, during in vivo investigation, the degradation of CS occurs under the influence of some proteases (mostly lysozymes), leading to formation of non-harmful oligosaccharides [67]. As a drug carrier in orthopedic implants, chitosan can be employed in local drug administration of antibiotics. In that way, it is possible to preserve the therapeutic effect of antibiotic with lower administered dose. It was shown that gentamicin release from the composite hydroxyapatite/chitosan/gentamicin coating included the initial burst-release effect, meaning that more than 50% of the total amount of pre-loaded antibiotic was released during the first 7 days. An additional advantage of chitosan presence in composite coating can be observed through the slower release of gentamicin during the next 14 days. Prolonged gentamicin release from the hydroxyapatite/chitosan/gentamicin composite coatings makes these coatings great candidates for potential application in the treatment of orthopedic infections [68]. Composite coatings based on chitosan and hydroxyapatite show excellent biocompatibility, which was confirmed through the formation of new hydroxyapatite layer after only 7 days of immersion in simulated body fluid (SBF) [68,69]. Above everything mentioned before, chitosan has very high position in the field of biomaterials production due to its exceptional properties of easy film forming ability [57,70]. Chitosan, as a cationic polysaccharide, can be used in the electrophoretic deposition process (EPD), one of the most attractive techniques for bioactive coatings production [68,69,71,72,73,74,75,76,77].
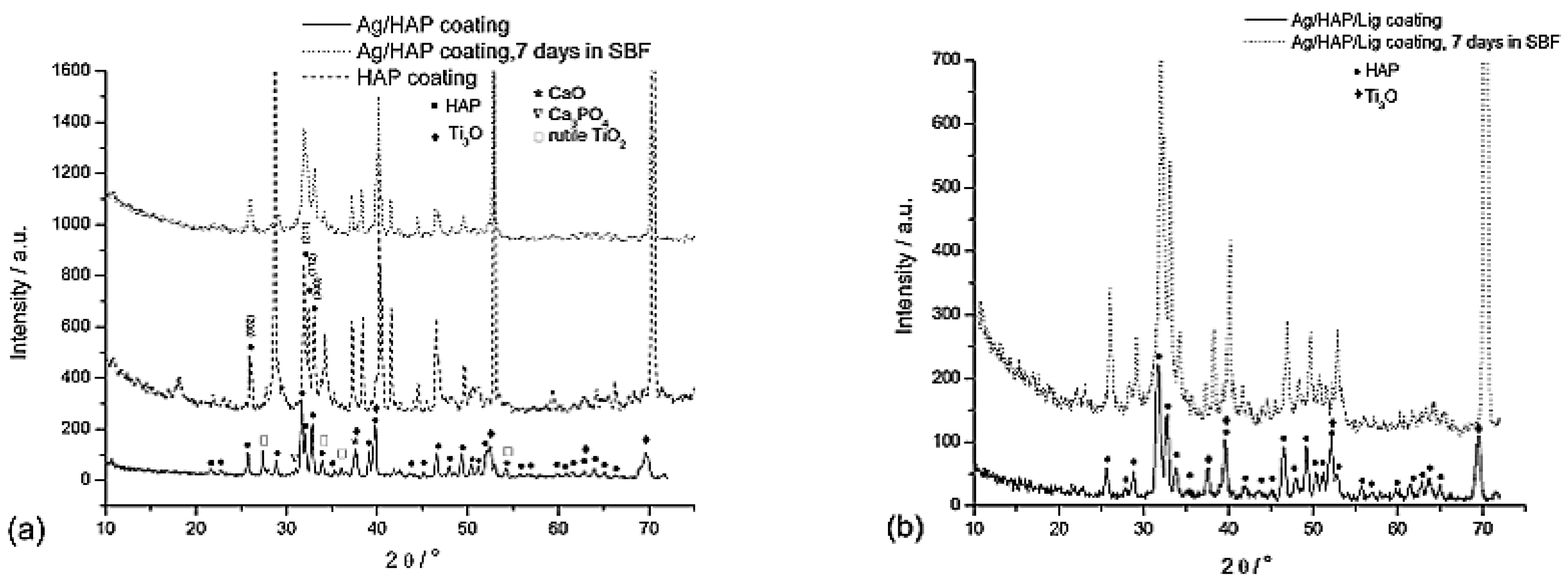
- Lignin (Lig) is a polyphenolic polymer, originated from the various natural sources [78,79,80]. According to the different processing methods used during production, several different types of lignin can be distinguished, e.g., kraft lignin, organosolv lignin, soda lignins [81]. The existence of different lignin types has limited its application in the biomedical fields. Despite these drawbacks, lignin possesses some unique properties, making it suitable for biomedical application, e.g., antioxidant, antibacterial, thermal stability, anti-ultraviolet protection, antigenotoxic, anticarcinogenic, antimutagenic and biocompatibility [80,82]. Organosolv lignin represents the purest type of lignin, extracted from natural sources (hardwood, softwood, plant crop) by solvent precipitation [73]. Lignin found the application as a drug nanocarrier [83,84], as well as in preparation of bioactive composite coatings [73,74,75] and tissue scaffolds [85]. Composite coatings based on lignin and hydroxyapatite with the addition of antimicrobial agent, such as silver, show excellent biocompatibility after SBF immersion. Namely, layer of newly grown hydroxyapatite can be obtained after 7 days of immersion [73].
3. Engineering Bactericidal Surfaces by Incorporating Silver
3.1. Hydroxyapatite Coatings with Silver and Lignin
Silver Release
4. Designing Coatings with Long-Term Release of Antibiotics
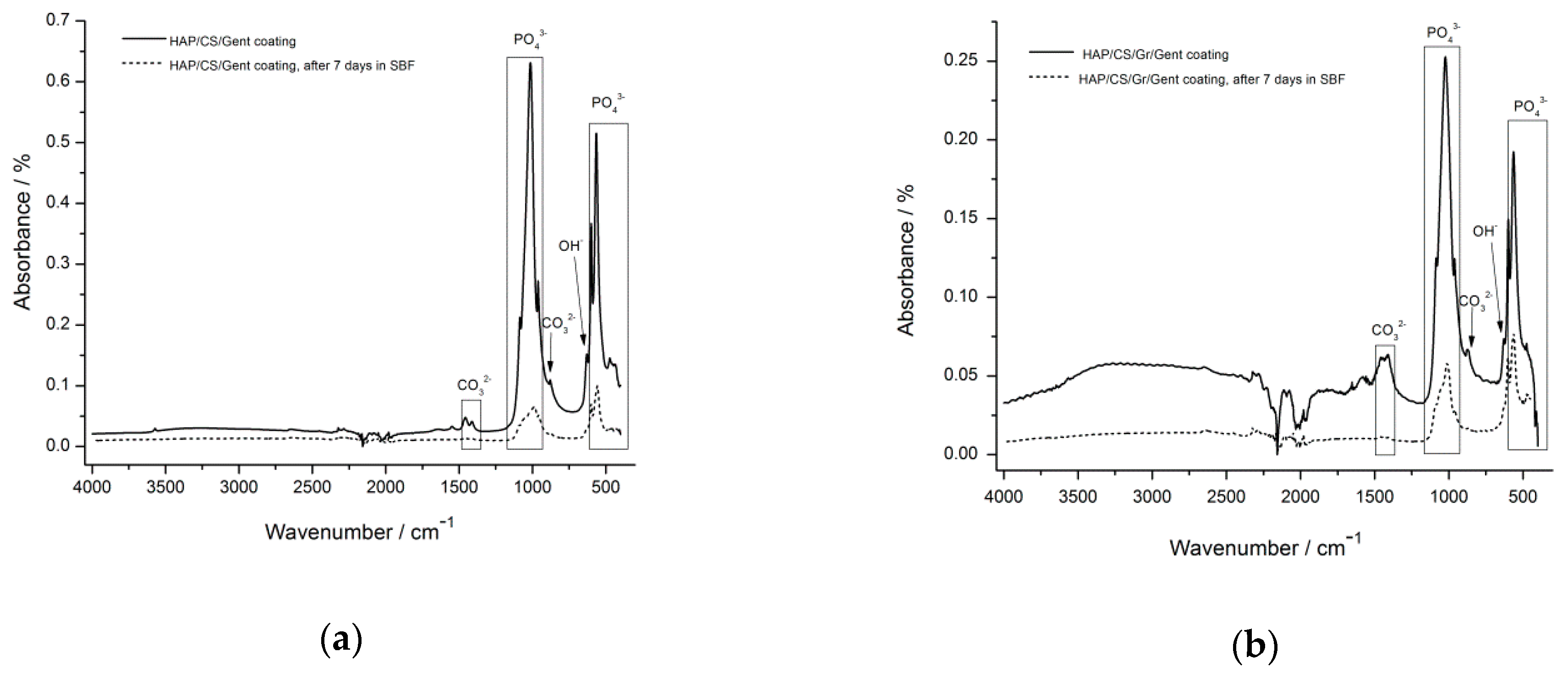
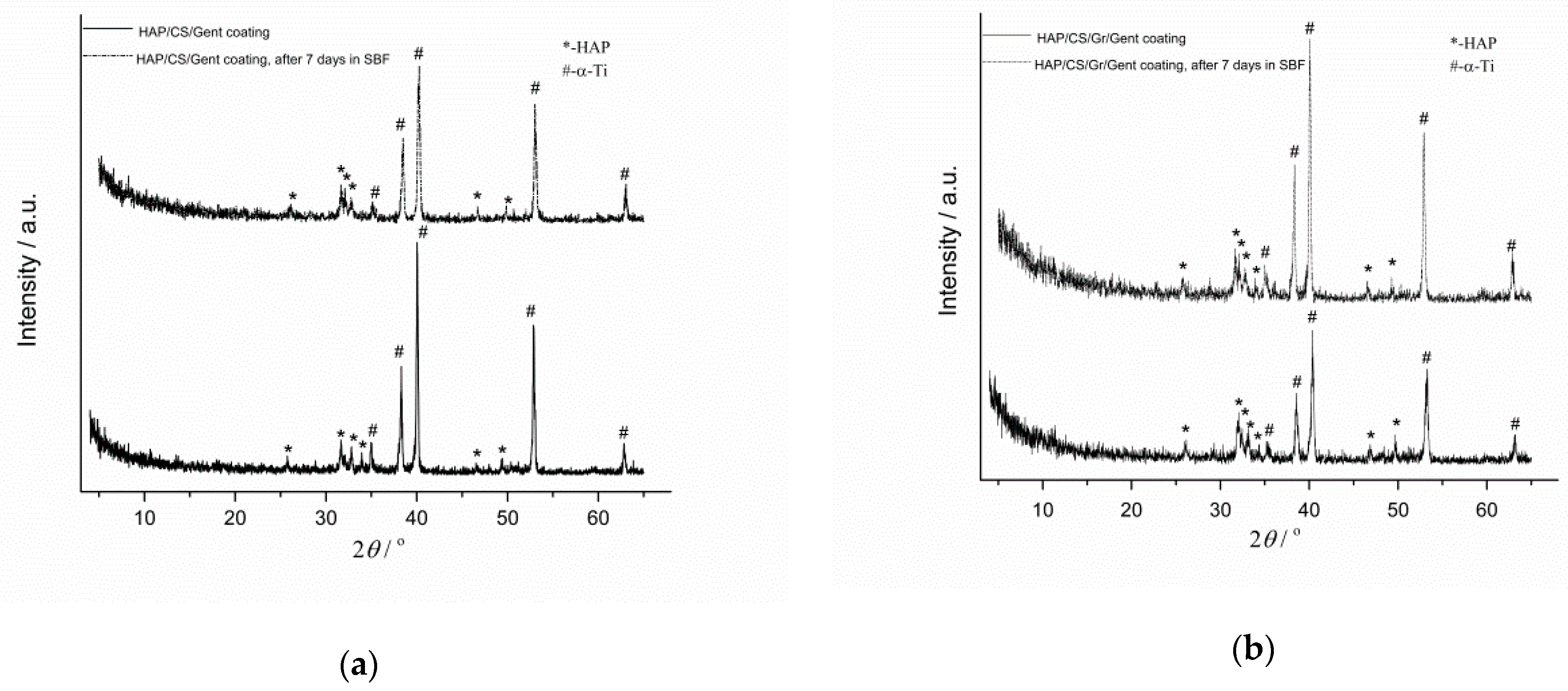
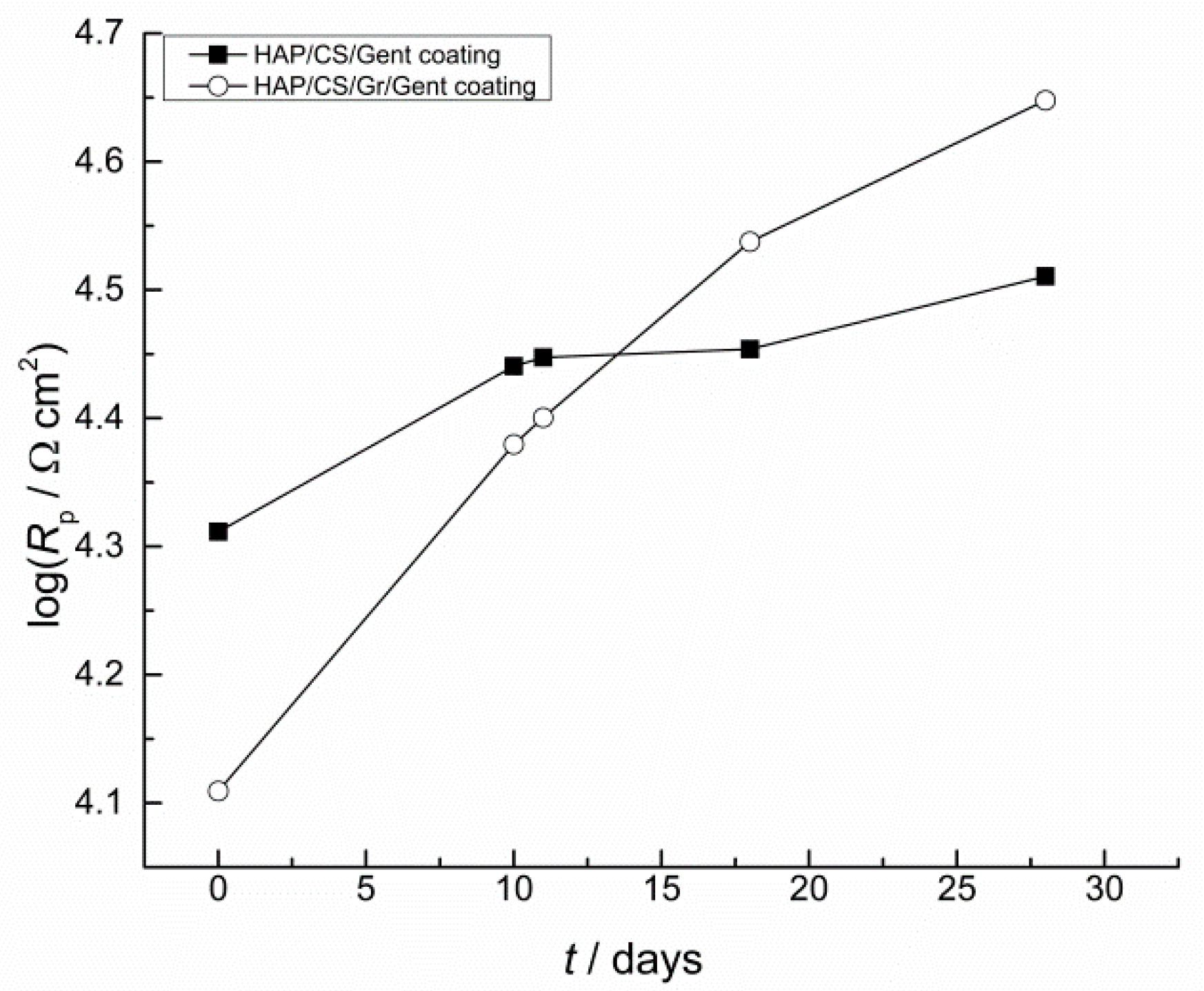
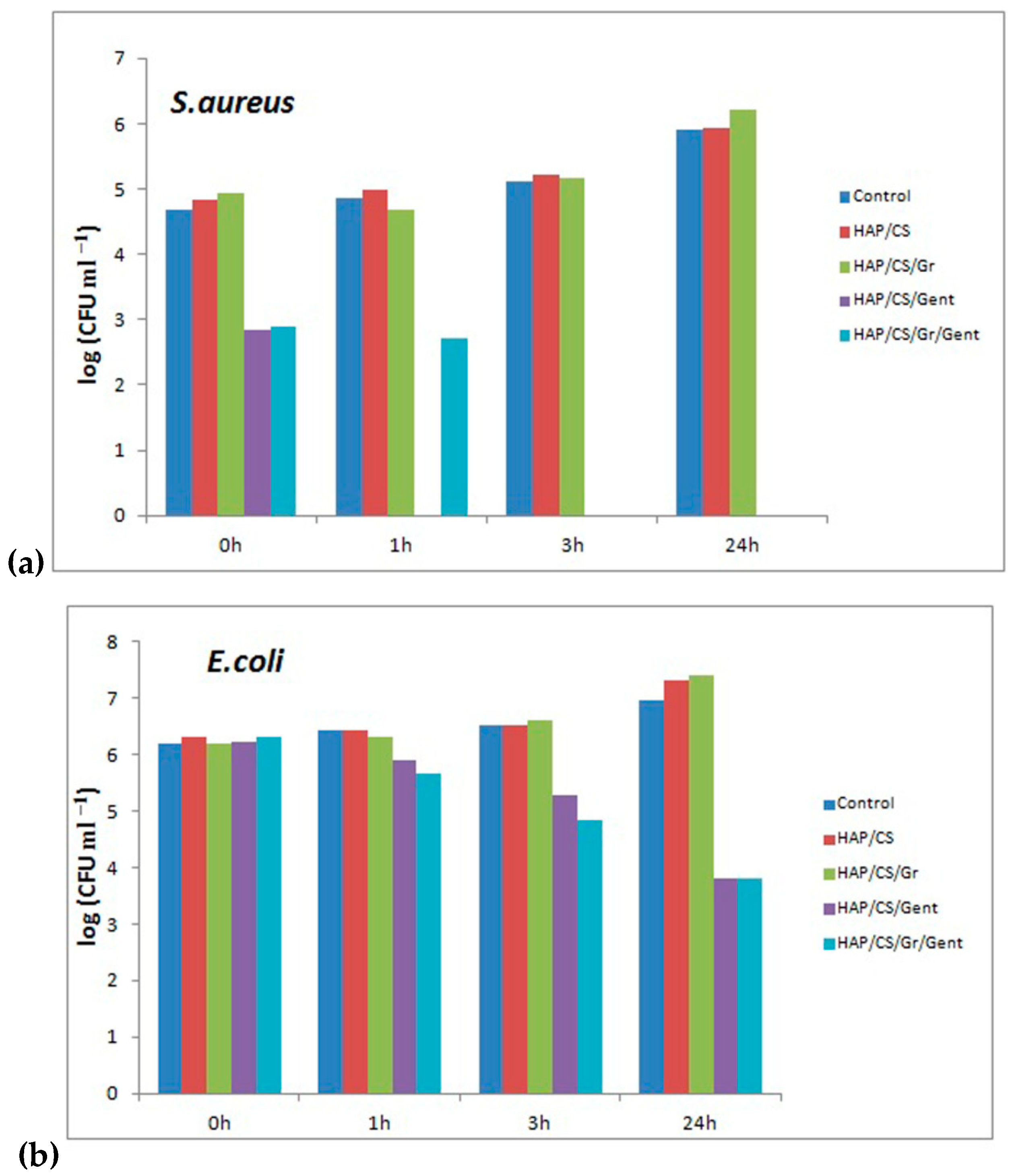
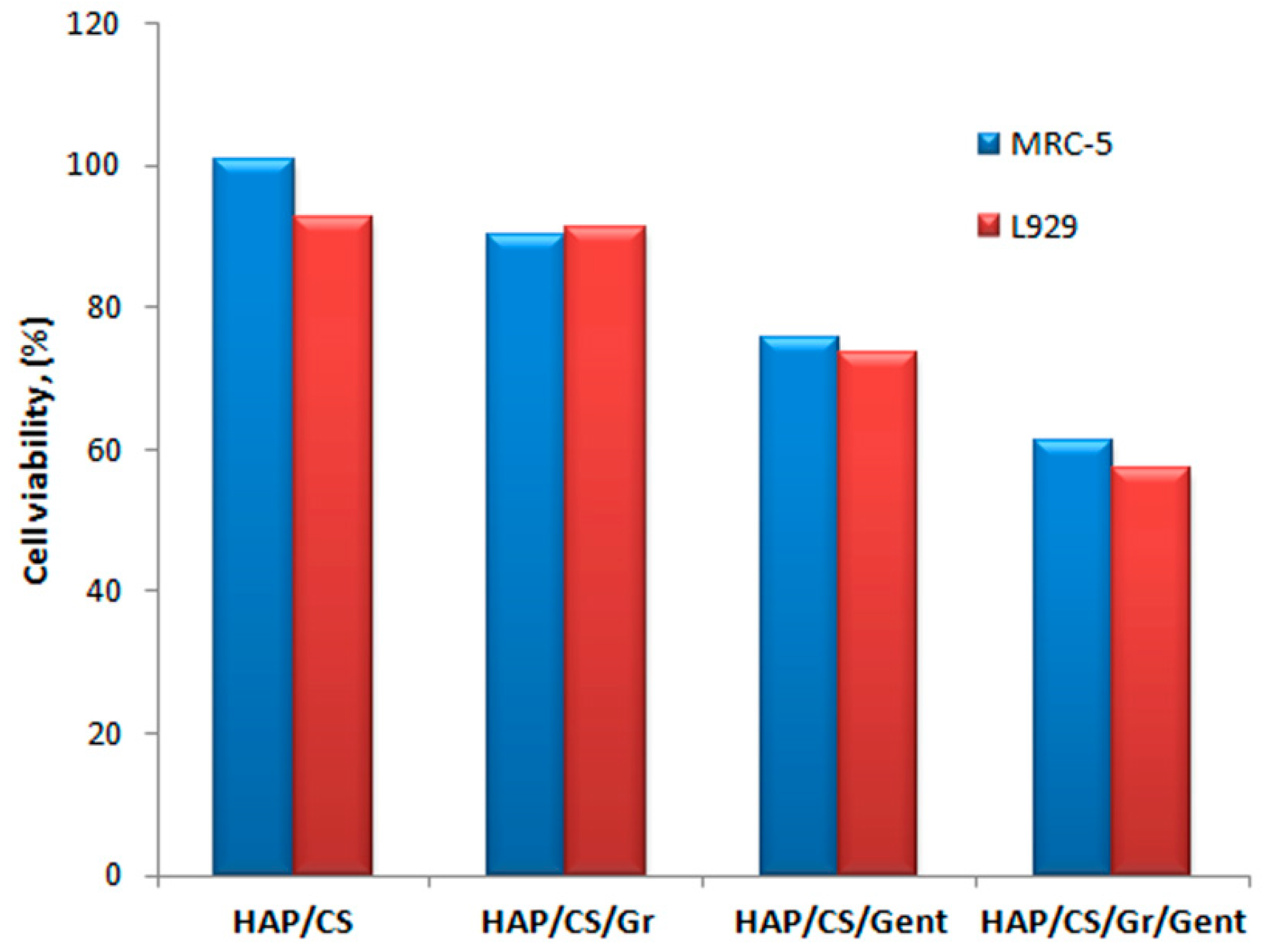
4.1. Hydroxyapatite Coatings with Gentamicin, Chitosan and Graphene
Gentamicin Release
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mas-Moruno, C.; Su, B.; Dalby, M.J. Multifunctional Coatings and Nanotopographies: Toward Cell Instructive and Antibacterial Implants. Adv. Healthc. Mater. 2019, 8, 1801103. [Google Scholar] [CrossRef]
- Hudecki, A.; Kiryczyński, G.; Łos, M.J. Biomaterials, definition, overview. In Stem Cells and Biomaterials for Regenerative Medicine; Elsevier: Amsterdam, The Netherlands, 2018; pp. 85–98. [Google Scholar] [CrossRef]
- Jin, W.; Chu, P.K. Orthopedic implants. In Encyclopedia of Biomedical Engineering; Elsevier: Amsterdam, The Netherlands, 2019; pp. 425–439. [Google Scholar] [CrossRef]
- dos Santos, G.A. The Importance of Metallic Materials as Biomaterials. Adv. Tissue Eng. Regen. Med. Open Access. 2017, 3, 300–302. [Google Scholar] [CrossRef] [Green Version]
- Nicholson, J.W. Titanium Alloys for Dental Implants: A Review. Prosthesis 2020, 2, 100–116. [Google Scholar] [CrossRef]
- Aroussi, D.; Aour, B.; Bouaziz, A.S. A Comparative Study of 316L Stainless Steel and a Titanium Alloy in an Aggressive Biological Medium, Eng. Technol. Appl. Sci. Res. 2019, 9, 5093–5098. [Google Scholar] [CrossRef]
- Vaicelyte, A.; Janssen, C.; le Borgne, M.; Grosgogeat, B. Cobalt–Chromium Dental Alloys: Metal Exposures, Toxicological Risks, CMR Classification, and EU Regulatory Framework. Crystals 2020, 10, 1151. [Google Scholar] [CrossRef]
- Hussein, M.A.; Mohammed, A.S.; Al-Aqeeli, N. Wear characteristics of metallic biomaterials: A review. Materials 2015, 8, 2749–2768. [Google Scholar] [CrossRef]
- Findik, F. Recent developments of metallic implants for biomedical applications. Period. Eng. Nat. Sci. 2020, 8, 33–57. [Google Scholar] [CrossRef]
- Haseeb, M.; Butt, M.F.; Altaf, T.; Muzaffar, K.; Gupta, A.; Jallu, A. Indications of implant removal: A study of 83 cases. Int. J. Health Sci. 2017, 11, 1. [Google Scholar]
- Noumbissi, S.; Scarano, A.; Gupta, S. A literature review study on atomic ions dissolution of titanium and its alloys in implant dentistry. Materials 2019, 12, 368. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manam, N.S.; Harun, W.S.W.; Shri, D.N.A.; Ghani, S.A.C.; Kurniawan, T.; Ismail, M.H.; Ibrahim, M.H.I. Study of corrosion in biocompatible metals for implants: A review. J. Alloys Compd. 2017, 701, 698–715. [Google Scholar] [CrossRef] [Green Version]
- Kattimani, V.S.; Kondaka, S.; Lingamaneni, K.P. Hydroxyapatite–-Past, Present, and Future in Bone Regeneration, Bone Tissue Regen. Insights 2016, 7, BTRI.S36138. [Google Scholar] [CrossRef] [Green Version]
- Arcos, D.; Vallet-Regí, M. Substituted hydroxyapatite coatings of bone implants. J. Mater. Chem. B 2020, 8, 1781–1800. [Google Scholar] [CrossRef] [PubMed]
- Awasthi, S.; Pandey, S.K.; Arunan, E.; Srivastava, C. A review on hydroxyapatite coatings for the biomedical applications: Experimental and theoretical perspectives. J. Mater. Chem. B 2021, 9, 228–249. [Google Scholar] [CrossRef] [PubMed]
- Ko, H.S.; Lee, S.; Jho, J.Y. Synthesis and modification of hydroxyapatite nanofiber for poly(Lactic acid) composites with enhanced mechanical strength and bioactivity. Nanomaterials 2021, 11, 213. [Google Scholar] [CrossRef]
- Fielding, G.A.; Roy, M.; Bandyopadhyay, A.; Bose, S. Antibacterial and biological characteristics of silver containing and strontium doped plasma sprayed hydroxyapatite coatings. Acta Biomater. 2012, 8, 3144–3152. [Google Scholar] [CrossRef] [Green Version]
- Li, M.; Mondrinos, M.J.; Chen, X.; Gandhi, M.R.; Ko, F.K.; Lelkes, P.I. Elastin Blends for Tissue Engineering Scaffolds. J. Biomed. Mater. Res. Part A 2006, 79, 963–973. [Google Scholar] [CrossRef]
- Kazemzadeh-Narbat, M.; Noordin, S.; Masri, B.A.; Garbuz, D.S.; Duncan, C.P.; Hancock, R.E.W.; Wang, R. Drug release and bone growth studies of antimicrobial peptide-loaded calcium phosphate coating on titanium. J. Biomed. Mater. Res.-Part B Appl. Biomater. 2012, 100, 1344–1352. [Google Scholar] [CrossRef]
- Pan, C.J.; Dong, Y.X.; Zhang, Y.Y.; Nie, Y.D.; Zhao, C.H.; Wang, Y.L. Enhancing the antibacterial activity of biomimetic HA coatings by incorporation of norvancomycin. J. Orthop. Sci. 2011, 16, 105–113. [Google Scholar] [CrossRef]
- Abdulghani, S.; Mitchell, G.R. Biomaterials for in situ tissue regeneration: A review. Biomolecules 2019, 9, 750. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raut, H.K.; Das, R.; Liu, Z.; Liu, X.; Ramakrishna, S. Biocompatibility of Biomaterials for Tissue Regeneration or Replacement. Biotechnol. J. 2020, 15, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Tang, G.; Tan, Z.; Zeng, W.; Wang, X.; Shi, C.; Liu, Y.; He, H.; Chen, R.; Ye, X. Recent Advances of Chitosan-Based Injectable Hydrogels for Bone and Dental Tissue Regeneration. Front. Bioeng. Biotechnol. 2020, 8, 1–15. [Google Scholar] [CrossRef]
- Durner, J.; Schrickel, K.; Watts, D.C.; Becker, M.; Hickel, R.; Draenert, M.E. An alternate methodology for studying diffusion and elution kinetics of dimethacrylate monomers through dentinal tubules. Dent. Mater. 2020, 36, 479–490. [Google Scholar] [CrossRef] [PubMed]
- Ma, P.; Wu, W.; Wei, Y.; Ren, L.; Lin, S.; Wu, J. Biomimetic gelatin/chitosan/polyvinyl alcohol/nano-hydroxyapatite scaffolds for bone tissue engineering. Mater. Des. 2021, 207, 109865. [Google Scholar] [CrossRef]
- Nouri, A.; Shirvan, A.R.; Li, Y.; Wen, C. Additive manufacturing of metallic and polymeric load-bearing biomaterials using laser powder bed fusion: A review. J. Mater. Sci. Technol. 2021, 94, 196–215. [Google Scholar] [CrossRef]
- Feczkó, T. Polymeric nanotherapeutics acting at special regions of body. J. Drug Deliv. Sci. Technol. 2021, 64. [Google Scholar] [CrossRef]
- Hu, Y.; Hu, S.; Zhang, S.; Dong, S.; Hu, J.; Kang, L.; Yang, X. A double-layer hydrogel based on alginate-carboxymethyl cellulose and synthetic polymer as sustained drug delivery system. Sci. Rep. 2021, 11, 1–14. [Google Scholar] [CrossRef]
- Prakash, J.; Prema, D.; Venkataprasanna, K.S.; Balagangadharan, K.; Selvamurugan, N.; Venkatasubbu, G.D. Nanocomposite chitosan film containing graphene oxide/hydroxyapatite/gold for bone tissue engineering. Int. J. Biol. Macromol. 2020, 154, 62–71. [Google Scholar] [CrossRef] [PubMed]
- Mitra, D.; Kang, E.T.; Neoh, K.G. Polymer-Based Coatings with Integrated Antifouling and Bactericidal Properties for Targeted Biomedical Applications. ACS Appl. Polym. Mater. 2021, 3, 2233–2263. [Google Scholar] [CrossRef]
- Graça, M.F.P.; de Melo-Diogo, D.; Correia, I.J.; Moreira, A.F. Electrospun asymmetric membranes as promising wound dressings: A review. Pharmaceutics 2021, 13, 183. [Google Scholar] [CrossRef]
- Camarena, D.E.M.; Matsuyama, L.S.A.S.; Maria-Engler, S.S.; Catalani, L.H. Development of epidermal equivalent from electrospun synthetic polymers for in vitro irritation/corrosion testing. Nanomaterials 2020, 10, 2528. [Google Scholar] [CrossRef]
- Dimopoulos, A.; Markatos, D.N.; Mitropoulou, A.; Panagiotopoulos, I.; Koletsis, E.; Mavrilas, D. A novel polymeric fibrous microstructured biodegradable small-caliber tubular scaffold for cardiovascular tissue engineering. J. Mater. Sci. Mater. Med. 2021, 32, 1–12. [Google Scholar] [CrossRef]
- Malik, S.; Sundarrajan, S.; Hussain, T.; Nazir, A.; Berto, F.; Ramakrishna, S. Electrospun biomimetic polymer nanofibers as vascular grafts. Mater. Design Process. Comm. 2020, e203. [Google Scholar] [CrossRef]
- Du, R.; Wang, Y.; Huang, Y.; Zhao, Y.; Zhang, D.; Du, D.; Zhang, Y.; Li, Z.; McGinty, S.; Pontrelli, G.; et al. Design and testing of hydrophobic core/hydrophilic shell nano/micro particles for drug-eluting stent coating. NPG Asia Mater. 2018, 10, 642–658. [Google Scholar] [CrossRef] [Green Version]
- Shokrolahi, F.; Khodabakhshi, K.; Shokrollahi, P.; Badiani, R.; Moghadam, Z.M. Atorvastatin loaded PLGA microspheres: Preparation, HAp coating, drug release and effect on osteogenic differentiation of ADMSCs. Int. J. Pharm. 2019, 565, 95–107. [Google Scholar] [CrossRef]
- Takahashi, M.; Yamaguchi, M.; Tanimoto, Y.; Yao-Umezawa, E.; Kasai, K. Biological evaluation of a prototype material made of polyglycolic acid and hydroxyapatite. J. Hard Tissue Biol. 2015, 24, 375–384. [Google Scholar] [CrossRef] [Green Version]
- Abdulkareem, M.H.; Abdalsalam, A.H.; Bohan, A.J. Influence of chitosan on the antibacterial activity of composite coating (PEEK/HAp) fabricated by electrophoretic deposition. Prog. Org. Coat. 2019, 130, 251–259. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Hoang, T.; Can, V.M.; Ho, A.S. In Vitro and in Vivo tests of PLA/d-HAp nanocomposite. Adv. Nat. Sci. Nanosci. Nanotechnol. 2017, 8, 045013. [Google Scholar] [CrossRef] [Green Version]
- Zhang, R.; Hu, H.; Liu, Y.; Tan, J.; Chen, W.; Ying, C.; Liu, Q.; Fu, X.; Hu, S.; Wong, C.P. Homogeneously dispersed composites of hydroxyapatite nanorods and poly(lactic acid) and their mechanical properties and crystallization behavior. Compos. Part A Appl. Sci. Manuf. 2020, 132, 105841. [Google Scholar] [CrossRef]
- Eftekhari, S.; el Sawi, I.; Bagheri, Z.S.; Turcotte, G.; Bougherara, H. Fabrication and characterization of novel biomimetic PLLA/cellulose/hydroxyapatite nanocomposite for bone repair applications. Mater. Sci. Eng. C 2014, 39, 120–125. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, R.; Chen, S.; Xu, Z.; Wang, Q.; Yuan, P.; Zhou, Y.; Zhang, Y.; Chen, J. Heparan sulfate loaded polycaprolactone-hydroxyapatite scaffolds with 3D printing for bone defect repair. Int. J. Biol. Macromol. 2020, 148, 153–162. [Google Scholar] [CrossRef]
- Januariyasa, I.K.; Ana, I.D.; Yusuf, Y. Nanofibrous poly(vinyl alcohol)/chitosan contained carbonated hydroxyapatite nanoparticles scaffold for bone tissue engineering. Mater. Sci. Eng. C 2020, 107, 110347. [Google Scholar] [CrossRef] [PubMed]
- Aldabib, J.M.; Ishak, Z.A.M. Effect of hydroxyapatite filler concentration on mechanical properties of poly (methyl methacrylate) denture base. SN Appl. Sci. 2020, 2, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Joyce, K.; Fabra, G.T.; Bozkurt, Y.; Pandit, A. Bioactive potential of natural biomaterials: Identification, retention and assessment of biological properties. Signal Transduct. Target. Ther. 2021, 6, 1–28. [Google Scholar] [CrossRef]
- Chun, H.J.; Park, K.; Kim, C.H.; Khang, G. (Eds.) Novel Biomaterials for Regenerative Medicine; Springer: Singapore, 2018. [Google Scholar] [CrossRef]
- Faulk, D.M.; Badylak, S.F. Regenerative Medicine Applications in Organ Transplantation; Academic Press: Cambridge, MA, USA, 2014. [Google Scholar]
- Cui, Y.; Yin, Y.; Zou, Y.; Zhao, Y.; Han, J.; Xu, B.; Chen, B.; Xiao, Z.; Song, H.; Shi, Y.; et al. The Rotary Cell Culture System increases NTRK3 expression and promotes neuronal differentiation and migratory ability of neural stem cells cultured on collagen sponge. Stem Cell Res. Ther. 2021, 12, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Galindo, T.G.P.; Yamada, I.; Yamada, S.; Tagaya, M. Studies on preparation of surfactant-assisted elliptical hydroxyapatite nanoparticles and their protein-interactive ability. Mater. Chem. Phys. 2019, 221, 367–376. [Google Scholar] [CrossRef]
- Miranda-Nieves, D.; Chaikof, E.L. Collagen and Elastin Biomaterials for the Fabrication of Engineered Living Tissues. ACS Biomater. Sci. Eng. 2017, 3, 694–711. [Google Scholar] [CrossRef]
- Rodrigues, I.C.P.; Pereira, K.D.; Woigt, L.F.; Jardini, A.L.; Luchessi, A.D.; Lopes, É.S.N.; Webster, T.J.; Gabriel, L.P. A novel technique to produce tubular scaffolds based on collagen and elastin. Artif. Organs 2021, 45, E113–E122. [Google Scholar] [CrossRef]
- Oh, E.J.; Kang, S.W.; Kim, B.S.; Jiang, G.; Cho, I.H.; Hahn, S.K. Control of the molecular degradation of hyaluronic acid hydrogels for tissue augmentation. J. Biomed. Mater. Res.-Part A 2008, 86, 685–693. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, Y.; Peng, X.; Yu, X.; Cheng, C.; Yu, X. Feasibility study of oxidized hyaluronic acid cross-linking acellular bovine pericardium with potential application for abdominal wall repair. Int. J. Biol. Macromol. 2021, 184, 831–842. [Google Scholar] [CrossRef]
- Salatin, S.; Jelvehgari, M. Natural polysaccharide based nanoparticles for drug/gene delivery. Pharm. Sci. 2017, 23, 84–94. [Google Scholar] [CrossRef] [Green Version]
- Alipour, M.; Firouzi, N.; Aghazadeh, Z.; Samiei, M.; Montazersaheb, S.; Khoshfetrat, A.B.; Aghazadeh, M. The osteogenic differentiation of human dental pulp stem cells in alginate-gelatin/Nano-hydroxyapatite microcapsules. BMC Biotechnol. 2021, 21, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Jia, W.; Li, M.; Liu, L.; Zhou, H.; Liu, X.; Gu, G.; Xiao, M.; Chen, Z. Fabrication and assessment of chondroitin sulfate-modified collagen nanofibers for small-diameter vascular tissue engineering applications. Carbohydr. Polym. 2021, 257, 117573. [Google Scholar] [CrossRef] [PubMed]
- Avcu, E.; Baştan, F.E.; Abdullah, H.Z.; Rehman, M.A.U.; Avcu, Y.Y.; Boccaccini, A.R. Electrophoretic deposition of chitosan-based composite coatings for biomedical applications: A review. Prog. Mater. Sci. 2019, 103, 69–108. [Google Scholar] [CrossRef]
- Nivethaa, E.A.K.; Martin, C.A.; Frank-Kamenetskaya, O.V.; Kalkura, S.N. Chitosan and chitosan based nanocomposites for applications as a drug delivery carrier: A review. In Processes and Phenomena on the Boundary between Biogenic and Abiogenic Nature; Frank-Kamenetskaya, O., Vlasov, D.Y., Panova, E.G., Lessovaia, S.N., Eds.; Springer: Berlin/Heidelberg, Germany, 2020; pp. 23–37. [Google Scholar] [CrossRef]
- Prasanthi, N.L.; Roy, H.; Jyothi, N.; Vajrapriya, V.S. A Brief Review on Chitosan and Application in Biomedical Field. Am. J. Pharm. Tech. Res. 2016, 6, 41–51. [Google Scholar]
- Li, W.W.; Wang, H.Y.; Zhang, Y.Q. A novel chitosan hydrogel membrane by an improved electrophoretic deposition and its characteristics in vitro and in vivo. Mater. Sci. Eng. C 2017, 74, 287–297. [Google Scholar] [CrossRef]
- Islam, N.; Dmour, I.; Taha, M.O. Degradability of chitosan micro/nanoparticles for pulmonary drug delivery. Heliyon 2019, 5, e01684. [Google Scholar] [CrossRef] [Green Version]
- Wu, Y.; Yan, C.; Wang, Y.; Gao, C.; Liu, Y. Biomimetic structure of chitosan reinforced epoxy natural rubber with self-healed, recyclable and antimicrobial ability. Int. J. Biol. Macromol. 2021, 184, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Afrasiabi, S.; Bahador, A.; Partoazar, A. Combinatorial therapy of chitosan hydrogel-based zinc oxide nanocomposite attenuates the virulence of Streptococcus mutans. BMC Microbiol. 2021, 21, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Gorantla, S.; Dabholkar, N.; Sharma, S.; Rapalli, V.K.; Alexander, A.; Singhvi, G. Chitosan-based microneedles as a potential platform for drug delivery through the skin: Trends and regulatory aspects. Int. J. Biol. Macromol. 2021, 184, 438–453. [Google Scholar] [CrossRef] [PubMed]
- Gulati, N.; Dua, K.; Dureja, H. Role of chitosan based nanomedicines in the treatment of chronic respiratory diseases. Int. J. Biol. Macromol. 2021, 185, 20–30. [Google Scholar] [CrossRef]
- Venkatesan, J.; Lowe, B.; Pallela, R.; Kim, S.-K. Chitosan-Based Polysaccharide Biomaterials. Polysaccharides 2014, 1–13. [Google Scholar] [CrossRef]
- Croisier, F.; Jérôme, C. Chitosan-based biomaterials for tissue engineering. Eur. Polym. J. 2013, 49, 780–792. [Google Scholar] [CrossRef] [Green Version]
- Stevanović, M.; Djošić, M.; Janković, A.; Nešović, K.; Kojić, V.; Stojanović, J.; Grujić, S.; Bujagić, M.I.; Rhee, .K.Y.; Mišković-Stanković, V. Assessing the Bioactivity of Gentamicin-Preloaded Hydroxyapatite/Chitosan Composite Coating on Titanium Substrate. ACS Omega 2020, 5, 15433–15445. [Google Scholar] [CrossRef] [PubMed]
- Ðošić, M.; Eraković, S.; Janković, A.; Vukašinović-Sekulić, M.; Matić, I.Z.; Stojanović, J.; Rhee, .K.Y.; Mišković-Stanković, V.; Park, S.J. In vitro investigation of electrophoretically deposited bioactive hydroxyapatite/chitosan coatings reinforced by graphene. J. Ind. Eng. Chem. 2017, 47, 336–347. [Google Scholar] [CrossRef]
- D’Almeida, M.; Attik, N.; Amalric, J.; Brunon, C.; Renaud, F.; Abouelleil, H.; Toury, B.; Grosgogeat, B. Chitosan coating as an antibacterial surface for biomedical applications. PLoS ONE 2017, 12, e0189537. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stevanović, M.; Djošić, M.; Janković, A.; Kojić, V.; Vukašinović-Sekulić, M.; Stojanović, J.; Odović, J.; Sakač, M.C.; Rhee, .K.Y.; Mišković-Stanković, V. Antibacterial graphene-based hydroxyapatite/chitosan coating with gentamicin for potential applications in bone tissue engineering. J. Biomed. Mater. Res.-Part A 2020, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Stevanović, M.; Đošić, M.; Janković, A.; Kojić, V.; Vukašinović-Sekulić, M.; Stojanović, J.; Odović, J.; Sakač, M.C.; Rhee, .K.Y.; Mišković-Stanković, V. Gentamicin-Loaded Bioactive Hydroxyapatite/Chitosan Composite Coating Electrodeposited on Titanium. ACS Biomater. Sci. Eng. 2018, 4, 3994–4007. [Google Scholar] [CrossRef]
- Erakovic, S.; Jankovic, A.; Tsui, G.C.P.P.; Tang, C.-Y.Y.; Miskovic-Stankovic, V.; Stevanovic, T. Novel Bioactive Antimicrobial Lignin Containing Coatings on Titanium Obtained by Electrophoretic Deposition. Int. J. Mol. Sci. 2014, 15, 12294–12322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Erakovic, S.; Jankovic, A.; Veljovic, D.; Palcevskis, E.; Mitric, M.; Stevanovic, T.; Janaćković, D.; Mišković-Stanković, V. Corrosion Stability and Bioactivity in Simulated Body Fluid of Silver/Hydroxyapatite and Silver/Hydroxyapatite/Lignin Coatings on Titanium Obtained by Electrophoretic Deposition. J. Phys. Chem. B 2013, 117, 1633–1643. [Google Scholar] [CrossRef] [PubMed]
- Erakovic, S.; Veljovic, D.; Diouf, P.N.; Stevanovic, T.; Mitric, M.; Milonjic, S.; Mišković-Stanković, V. Electrophoretic Deposition of Biocomposite Lignin/Hydroxyapatite Coatings on Titanium Electrophoretic Deposition of Biocomposite Lignin/Hydroxyapatite Coatings on Titanium. Int. J. Chem. React. Eng. 2009, 7, A62. [Google Scholar] [CrossRef]
- Eraković, S.; Janković, A.; Matić, I.Z.; Juranić, Z.D.; Vukašinović-Sekulić, M.; Stevanović, T.; Mišković-Stanković, V. Investigation of silver impact on hydroxyapatite/lignin coatings electrodeposited on titanium. Mater. Chem. Phys. 2013, 142, 521–530. [Google Scholar] [CrossRef]
- Janković, A.; Eraković, S.; Vukašinović-Sekulić, M.; Mišković-Stanković, V.; Park, S.J.; Rhee, K.Y. Graphene-based antibacterial composite coatings electrodeposited on titanium for biomedical applications. Prog. Org. Coat. 2015, 83, 1–10. [Google Scholar] [CrossRef]
- Rosova, E.; Smirnova, N.; Dresvyanina, E.; Smirnova, V.; Vlasova, E.; Ivan’kova, E.; Sokolova, M.; Maslennikova, T.; Malafeev, K.; Kolbe, K.; et al. Biocomposite materials based on chitosan and lignin: Preparation and characterization. Cosmetics 2021, 8, 24. [Google Scholar] [CrossRef]
- Baurhoo, B.; Ruiz-Feria, C.A.; Zhao, X. Purified lignin: Nutritional and health impacts on farm animals-A review. Anim. Feed Sci. Technol. 2008, 144, 175–184. [Google Scholar] [CrossRef]
- Sugiarto, S.; Leow, Y.; Tan, C.L.; Wang, G.; Kai, D. How far is Lignin from being a biomedical material? Bioact. Mater. 2021. [Google Scholar] [CrossRef]
- Witzler, M.; Alzagameem, A.; Bergs, M.; Khaldi-Hansen, B.; Klein, S.E.; Hielscher, D.; Kamm, B.; Kreyenschmidt, J.; Tobiasch, E.; Schulze, M. Lignin-derived biomaterials for drug release and tissue engineering. Molecules 2018, 23, 1885. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vinardell, M.P.; Mitjans, M. Lignins and their derivatives with beneficial effects on human health. Int. J. Mol. Sci. 2017, 18, 1219. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alqahtani, M.S.; Alqahtani, A.; Kazi, M.; Ahmad, M.Z.; Alahmari, A.; Alsenaidy, M.A.; Syed, R. Wound-healing potential of curcumin loaded lignin nanoparticles. J. Drug Deliv. Sci. Technol. 2020, 60, 102020. [Google Scholar] [CrossRef]
- Dai, L.; Zhu, W.; Liu, R.; Si, C. Lignin-Containing Self-Nanoemulsifying Drug Delivery System for Enhance Stability and Oral Absorption of trans-Resveratrol. Part. Part. Syst. Charact. 2018, 35, 1700447. [Google Scholar] [CrossRef]
- Eivazzadeh-Keihan, R.; Aliabadi, H.A.M.; Radinekiyan, F.; Sobhani, M.; Khalili, F.; Maleki, A.; Mahdavi, M.; Shalan, A.E. Investigation of the biological activity, mechanical properties and wound healing application of a novel scaffold based on lignin-agarose hydrogel and silk fibroin embedded zinc chromite nanoparticles. RSC Adv. 2021, 11, 17914–17923. [Google Scholar] [CrossRef]
- Ordikhani, F.; Farani, M.R.; Dehghani, M.; Tamjid, E.; Simchi, A. Physicochemical and biological properties of electrodeposited graphene oxide/chitosan films with drug-eluting capacity. Carbon 2015, 84, 91–102. [Google Scholar] [CrossRef]
- Chen, Q.; Cabanas-Polo, S.; Ding, Y.P.; Boccaccini, A.R. Bioactive Glass-Biopolymer Multilayer Coatings Fabricated by Electrophoretic Deposition Combined with Layer-by-Layer Assembly. Key Eng. Mater. 2015, 654, 170–175. [Google Scholar] [CrossRef]
- Choi, H.; Schulte, A.; Müller, M.; Park, M.; Jo, S.; Schönherr, H. Drug Release from Thermo-Responsive Polymer Brush Coatings to Control Bacterial Colonization and Biofilm Growth on Titanium Implants. Adv. Healthc. Mater. 2021, 10, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Geuli, O.; Metoki, N.; Zada, T.; Reches, M.; Eliaz, N.; Mandler, D. Synthesis, coating, and drug-release of hydroxyapatite nanoparticles loaded with antibiotics. J. Mater. Chem. B 2017, 5, 7819–7830. [Google Scholar] [CrossRef]
- Stigter, M.; Bezemer, J.; de Groot, K.; Layrolle, P. Incorporation of different antibiotics into carbonated hydroxyapatite coatings on titanium implants, release and antibiotic efficacy. J. Control. Release 2004, 99, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Patel, K.D.; Singh, R.K.; Lee, E.J.; Han, C.M.; Won, J.E.; Knowles, J.C.; Kim, H.W. Tailoring solubility and drug release from electrophoretic deposited chitosan-gelatin films on titanium. Surf. Coat. Technol. 2014, 242, 232–236. [Google Scholar] [CrossRef]
- Jugowiec, D.; Łukaszczyk, A.; Cieniek, Ł.; Kowalski, K.; Rumian, Ł.; Pietryga, K.; Kot, M.; Pamuła, E.; Moskalewicz, T. Influence of the electrophoretic deposition route on the microstructure and properties of nano-hydroxyapatite/chitosan coatings on the Ti-13Nb-13Zr alloy. Surf. Coat. Technol. 2017, 324, 64–79. [Google Scholar] [CrossRef]
- Bakhshandeh, S.; Yavari, S.A. Electrophoretic deposition: A versatile tool against biomaterial associated infections. J. Mater. Chem. B 2018, 6, 1128–1148. [Google Scholar] [CrossRef]
- Asgari, N.; Rajabi, M. Enhancement of mechanical properties of hydroxyapatite coating prepared by electrophoretic deposition method. Int. J. Appl. Ceram. Technol. 2021, 18, 147–153. [Google Scholar] [CrossRef]
- Saadati, A.; Hesarikia, H.; Nourani, M.R.; Taheri, R.A. Electrophoretic deposition of hydroxyapatite coating on biodegradable Mg–4Zn–4Sn–0.6Ca–0.5Mn alloy. Surf. Eng. 2020, 36, 908–918. [Google Scholar] [CrossRef]
- LASKA, A. Parameters of the Electrophoretic Deposition Process and Its Influence on the Morphology of Hydroxyapatite Coatings. Rev. Inżynieria Mater. 2020, 1, 20–25. [Google Scholar] [CrossRef]
- Sorkhi, L.; Farrokhi-Rad, M.; Shahrabi, T. Electrophoretic Deposition of Hydroxyapatite–Chitosan–Titania on Stainless Steel 316 L. Surfaces 2019, 2, 458–467. [Google Scholar] [CrossRef] [Green Version]
- Sotniczuk, A.; Heise, S.; Topolski, K.; Garbacz, H.; Boccaccini, A.R. Chitosan/bioactive glass coatings as a protective layer against corrosion of nanocrystalline titanium under simulated inflammation. Mater. Lett. 2020, 264, 127284. [Google Scholar] [CrossRef]
- Nawrotek, K.; Grams, J. Understanding electrodeposition of chitosan–hydroxyapatite structures for regeneration of tubular-shaped tissues and organs. Materials 2021, 14, 1288. [Google Scholar] [CrossRef]
- Djošić, M.S.; Mitrić, M.; Mišković-Stankovic, V.B. The porosity and roughness of electrodeposited calcium phosphate coatings in simulated body fluid. J. Serbian Chem. Soc. 2015, 80, 237–251. [Google Scholar] [CrossRef]
- Janković, A.; Eraković, S.; Mitrić, M.; Matić, I.Z.; Juranić, Z.D.; Tsui, G.C.P.; Tang, C.Y.; Mišković-Stanković, V.; Rhee, K.Y.; Park, S.J. Bioactive hydroxyapatite/graphene composite coating and its corrosion stability in simulated body fluid. J. Alloys Compd. 2015, 624, 148–157. [Google Scholar] [CrossRef]
- Meng, Q.; Yan, J.; Wu, R.; Liu, H.; Sun, Y.; Wu, N.; Xiang, J.; Zheng, L.; Zhang, J.; Han, B. Sustainable production of benzene from lignin. Nat. Commun. 2021, 12, 4534. [Google Scholar] [CrossRef]
- Gosselink, R.J.A.; Abächerli, A.; Semke, H.; Malherbe, R.; Käuper, P.; Nadif, A.; van Dam, J.E.G. Analytical protocols for characterisation of sulphur-free lignin. Ind. Crops Prod. 2004, 19, 271–281. [Google Scholar] [CrossRef]
- Janković, A.; Eraković, S.; Dindune, A.; Veljović, D.; Stevanović, T.; Janaćković, D.; Mišković-Stanković, V. Electrochemical impedance spectroscopy of a silver-doped hydroxyapatite coating in simulated body fluid used as a corrosive agent. J. Serb. Chem. Soc. 2012, 77, 1609–1623. [Google Scholar] [CrossRef]
- Berzina-Cimdina, L.; Borodajenko, N. Research of Calcium Phosphates Using Fourier Transform Infrared Spectroscopy. Infrared Spectrosc.–Mater. Sci. Eng. Technol. 2012, 123–148. [Google Scholar] [CrossRef] [Green Version]
- Sun, R.; Li, M.; Lu, Y.; Wang, A. Immersion behavior of hydroxyapatite (HA) powders before and after sintering. Mater. Charact. 2006, 56, 250–254. [Google Scholar] [CrossRef]
- Barinov, S.M.; Rau, J.V.; Cesaro, S.N.; Ďurišin, J.; Fadeeva, I.V.; Ferro, D.; Medvecky, L.; Trionfetti, G. Carbonate release from carbonated hydroxyapatite in the wide temperature rage. J. Mater. Sci. Mater. Med. 2006, 17, 597–604. [Google Scholar] [CrossRef] [PubMed]
- Ren, F.; Ding, Y.; Leng, Y. Infrared spectroscopic characterization of carbonated apatite: A combined experimental and computational study. J. Biomed. Mater. Res.-Part A 2014, 102, 496–505. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.W.; Khor, K.A.; Cheang, P. Bone-like apatite layer formation on hydroxyapatite prepared by spark plasma sintering (SPS). Biomaterials 2004, 25, 4127–4134. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Xi, X.; Li, J.; Cai, K. Comparison of crystal structure between carbonated hydroxyapatite and natural bone apatite with theoretical calculation. Asian J. Chem. 2013, 25, 3673–3678. [Google Scholar] [CrossRef]
- Kim, H.M.; Himeno, T.; Kokubo, T.; Nakamura, T. Process and kinetics of bonelike apatite formation on sintered hydroxyapatite in a simulated body fluid. Biomaterials 2005, 26, 4366–4373. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Lee, D.Y.; Oh, K.T.; Lee, Y.K.; Kim, K.M.; Kim, K.N. Bioactivity of calcium phosphate coatings prepared by electrodeposition in a modified simulated body fluid. Mater. Lett. 2006, 60, 2573–2577. [Google Scholar] [CrossRef]
- Mokabber, T.; Cao, H.T.; Norouzi, N.; van Rijn, P.; Pei, Y.T. Antimicrobial Electrodeposited Silver-Containing Calcium Phosphate Coatings, ACS Appl. Mater. Interfaces 2020, 12, 5531–5541. [Google Scholar] [CrossRef]
- Jamuna-Thevi, K.; Bakar, S.A.; Ibrahim, S.; Shahab, N.; Toff, M.R.M. Quantification of silver ion release, in vitro cytotoxicity and antibacterial properties of nanostuctured Ag doped TiO2coatings on stainless steel deposited by RF magnetron sputtering. Vacuum 2011, 86, 235–241. [Google Scholar] [CrossRef]
- Greulich, C.; Diendorf, J.; Simon, T.; Eggeler, G.; Epple, M.; Köller, M. Uptake and intracellular distribution of silver nanoparticles in human mesenchymal stem cells. Acta Biomater. 2011, 7, 347–354. [Google Scholar] [CrossRef]
- Sjögren, G.; Sletten, G.; Dahl, J.E. Cytotoxicity of dental alloys, metals, and ceramics assessed by Millipore filter, agar overlay, and MTT tests. J. Prosthet. Dent. 2000, 84, 229–236. [Google Scholar] [CrossRef]
- Raj, R.M.; Priya, P.; Raj, V. Gentamicin-loaded ceramic-biopolymer dual layer coatings on the Ti with improved bioactive and corrosion resistance properties for orthopedic applications. J. Mech. Behav. Biomed. Mater. 2018, 82, 299–309. [Google Scholar] [CrossRef]
- Boelch, S.P.; Jordan, M.C.; Arnholdt, J.; Steinert, A.F.; Rudert, M.; Luedemann, M. Antibiotic elution and compressive strength of gentamicin/vancomycin loaded bone cements are considerably influenced by immersion fluid volume. J. Mater. Sci. Mater. Med. 2019, 30. [Google Scholar] [CrossRef]
- Ji, X.J.; Gao, L.; Liu, J.C.; Jiang, R.Z.; Sun, F.Y.; Cui, L.Y.; Li, S.-Q.; Zhi, K.-Q.; Zeng, R.-C.; Wang, Z.-L. Corrosion resistance and antibacterial activity of hydroxyapatite coating induced by ciprofloxacin-loaded polymeric multilayers on magnesium alloy. Prog. Org. Coat. 2019, 135, 465–474. [Google Scholar] [CrossRef]
- Draghi, L.; Preda, V.; Moscatelli, M.; Santin, M.; Chiesa, R. Gentamicin-Loaded TiO2 Nanotubes as Improved Antimicrobial Surfaces for Orthopedic Implants. Front. Mater. 2020, 7, 1–7. [Google Scholar] [CrossRef]
- Grohmann, S.; Menne, M.; Hesse, D.; Bischoff, S.; Schiffner, R.; Diefenbeck, M.; Liefeith, K. Biomimetic multilayer coatings deliver gentamicin and reduce implant-related osteomyelitis in rats. Biomed. Tech. 2019, 64, 383–395. [Google Scholar] [CrossRef] [PubMed]
- Oshima, S.; Sato, T.; Honda, M.; Suetsugu, Y.; Ozeki, K.; Kikuchi, M. Fabrication of gentamicin-loaded hydroxyapatite/collagen bone-like nanocomposite for anti-infection bone void fillers. Int. J. Mol. Sci. 2020, 21, 551. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thompson, K.; Petkov, S.; Zeiter, S.; Sprecher, C.M.; Richards, R.G.; Moriarty, T.F.; Eijer, H. Intraoperative loading of calcium phosphate-coated implants with gentamicin prevents experimental Staphylococcus aureus infection in vivo. PLoS ONE 2019, 14, e0210402. [Google Scholar] [CrossRef] [PubMed]
- Li, T.T.; Ling, L.; Lin, M.C.; Peng, H.K.; Ren, H.T.; Lou, C.W.; Lin, J.H. Recent advances in multifunctional hydroxyapatite coating by electrochemical deposition. J. Mater. Sci. 2020, 55, 6352–6374. [Google Scholar] [CrossRef]
- Zhang, S.; Cheng, X.; Shi, J.; Pang, J.; Wang, Z.; Shi, W.; Liu, F.; Ji, B. Electrochemical deposition of calcium phosphate/chitosan/gentamicin on a titanium alloy for bone tissue healing. Int. J. Electrochem. Sci. 2018, 13, 4046–4054. [Google Scholar] [CrossRef]
- Aydemir, T.; Liverani, L.; Pastore, J.I.; Ceré, S.M.; Goldmann, W.H.; Boccaccini, A.R.; Ballarre, J. Functional behavior of chitosan/gelatin/silica-gentamicin coatings by electrophoretic deposition on surgical grade stainless steel. Mater. Sci. Eng. C 2020, 115, 111062. [Google Scholar] [CrossRef] [PubMed]
- Humayun, A.; Luo, Y.; Mills, D.K. Electrophoretic deposition of gentamicin-loaded znhnts-chitosan on titanium. Coatings 2020, 10, 944. [Google Scholar] [CrossRef]
- Ordikhani, F.; Zustiak, S.P.; Simchi, A. Surface Modifications of Titanium Implants by Multilayer Bioactive Coatings with Drug Delivery Potential: Antimicrobial, Biological, and Drug Release Studies. JOM 2016, 68, 1100–1108. [Google Scholar] [CrossRef]
- Pishbin, F.; Mouriño, V.; Flor, S.; Kreppel, S.; Salih, V.; Ryan, M.P.; Boccaccini, A.R. Electrophoretic Deposition of Gentamicin-Loaded Bioactive Glass/Chitosan Composite Coatings for Orthopaedic Implants. ACS Appl. Mater. Interfaces 2014, 6, 8796–8806. [Google Scholar] [CrossRef]
- Wu, L.Q.; Gadre, A.P.; Yi, H.; Kastantin, M.J.; Rubloff, G.W.; Bentley, W.E.; Payne, G.F.; Ghodssi, R. Voltage-dependent assembly of the polysaccharide chitosan onto an electrode surface. Langmuir 2002, 18, 8620–8625. [Google Scholar] [CrossRef]
- Cheng, Y.; Luo, X.; Betz, J.; Buckhout-White, S.; Bekdash, O.; Payne, G.F.; Bentley, W.E.; Rubloff, G.W. In Situ quantitative visualization and characterization of chitosan electrodeposition with paired sidewall electrodes. Soft Matter 2010, 6, 3177–3183. [Google Scholar] [CrossRef]
- Brangule, A.; Gross, K.A. Importance of FTIR Spectra Deconvolution for the Analysis of Amorphous Calcium Phosphates. IOP Conf. Ser. Mater. Sci. Eng. 2015, 77, 012027. [Google Scholar] [CrossRef] [Green Version]
- Rapacz-Kmita, A.; Stodolak-Zych, E.; Ziabka, M.; Rozycka, A.; Dudek, M. Instrumental characterization of the smectite clay-gentamicin hybrids. Bull. Mater. Sci. 2015, 38, 1069–1078. [Google Scholar] [CrossRef]
- Gebhardt, F.; Seuss, S.; Turhan, M.C.; Hornberger, H.; Virtanen, S.; Boccaccini, A.R. Characterization of electrophoretic chitosan coatings on stainless steel. Mater. Lett. 2012, 66, 302–304. [Google Scholar] [CrossRef]
- Cromme, P.; Zollfrank, C.; Müller, L.; Müller, F.A.; Greil, P. Biomimetic mineralisation of apatites on Ca2+activated cellulose templates. Mater. Sci. Eng. C 2007, 27, 1–7. [Google Scholar] [CrossRef]
- Bonadio, T.G.M.; Sato, F.; Medina, A.N.; Weinand, W.R.; Baesso, M.L.; Lima, W.M. Bioactivity and structural properties of nanostructured bulk composites containing Nb2O5 and natural hydroxyapatite. J. Appl. Phys. 2013, 113, 223505. [Google Scholar] [CrossRef]
- Szcześ, A.; Hołysz, L.; Chibowski, E. Synthesis of hydroxyapatite for biomedical applications. Adv. Colloid Interface Sci. 2017, 249, 321–330. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Huang, S.; Matinlinna, J.P.; Chen, Z.; Pan, H. Insight into biological apatite: Physiochemical properties and preparation approaches. BioMed Res. Int. 2013, 2013. [Google Scholar] [CrossRef] [PubMed] [Green Version]



| Coating | t/h | Rp/kΩ cm2 |
|---|---|---|
| Ag/HAP | 24 | 6.6 |
| 120 | 11.7 | |
| 240 | 12.6 | |
| Ag/HAP/Lig | 24 | 5.6 |
| 120 | 5.9 | |
| 240 | 6.3 |
| t/h | c (Ag+ Ion Released)/ppm |
|---|---|
| 1 | 0.455 |
| 4 | 0.550 |
| 24 | 0648 |
| 48 | 0.802 |
| 72 | 0.881 |
| 120 | 1.060 |
| 144 | 1.294 |
| 168 | 1.371 |
| 192 | 1.486 |
| 216 | 1.557 |
| 240 | 1.704 |
| S. aureus Colonies Incubated with Coating Material | ||||
|---|---|---|---|---|
| Sample | Control | Ag/HAP | Ag/HAP/Lig | |
| S. aureus [CFU mL−1] | Incubation period, h | |||
| 0 | 1.0 × 105 | 1.2 × 104 | 2.5 × 104 | |
| 1 | 3.0 × 104 | 1.62 × 103 | 2.0 × 103 | |
| 24 | 9.9 × 104 | 0 | 0 | |
| Survival of Peripheral Blood Mononuclear Cells (PBMC) | ||||
| Cell survival (S), % | 100% | 94.6 ± 4.2 | 89.4 ± 3.5 | |
| Classification | n/a | non-cytotoxic | non-cytotoxic | |
| PHA-Stimulated Peripheral Blood Mononuclear Cells (PBMC) | ||||
| Cell survival (S), % | 100% | 92.1 ± 5.0 | 83.8 ± 6.3 | |
| Classification | n/a | non-cytotoxic | non-cytotoxic | |
| Release Time (Days) | HAP/CS/Gent | HAP/CS/Gr/Gent |
|---|---|---|
| Mass of Released Gent (μg) | ||
| 1 | 2.43 | 7.65 |
| 7 | 9.40 | 18.13 |
| 21 | 11.80 | 20.63 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Djošić, M.; Janković, A.; Mišković-Stanković, V. Electrophoretic Deposition of Biocompatible and Bioactive Hydroxyapatite-Based Coatings on Titanium. Materials 2021, 14, 5391. https://doi.org/10.3390/ma14185391
Djošić M, Janković A, Mišković-Stanković V. Electrophoretic Deposition of Biocompatible and Bioactive Hydroxyapatite-Based Coatings on Titanium. Materials. 2021; 14(18):5391. https://doi.org/10.3390/ma14185391
Chicago/Turabian StyleDjošić, Marija, Ana Janković, and Vesna Mišković-Stanković. 2021. "Electrophoretic Deposition of Biocompatible and Bioactive Hydroxyapatite-Based Coatings on Titanium" Materials 14, no. 18: 5391. https://doi.org/10.3390/ma14185391
APA StyleDjošić, M., Janković, A., & Mišković-Stanković, V. (2021). Electrophoretic Deposition of Biocompatible and Bioactive Hydroxyapatite-Based Coatings on Titanium. Materials, 14(18), 5391. https://doi.org/10.3390/ma14185391







