Spray Flame Synthesis (SFS) of Lithium Lanthanum Zirconate (LLZO) Solid Electrolyte
Abstract
:1. Introduction
2. Experimental Section
3. Characterization
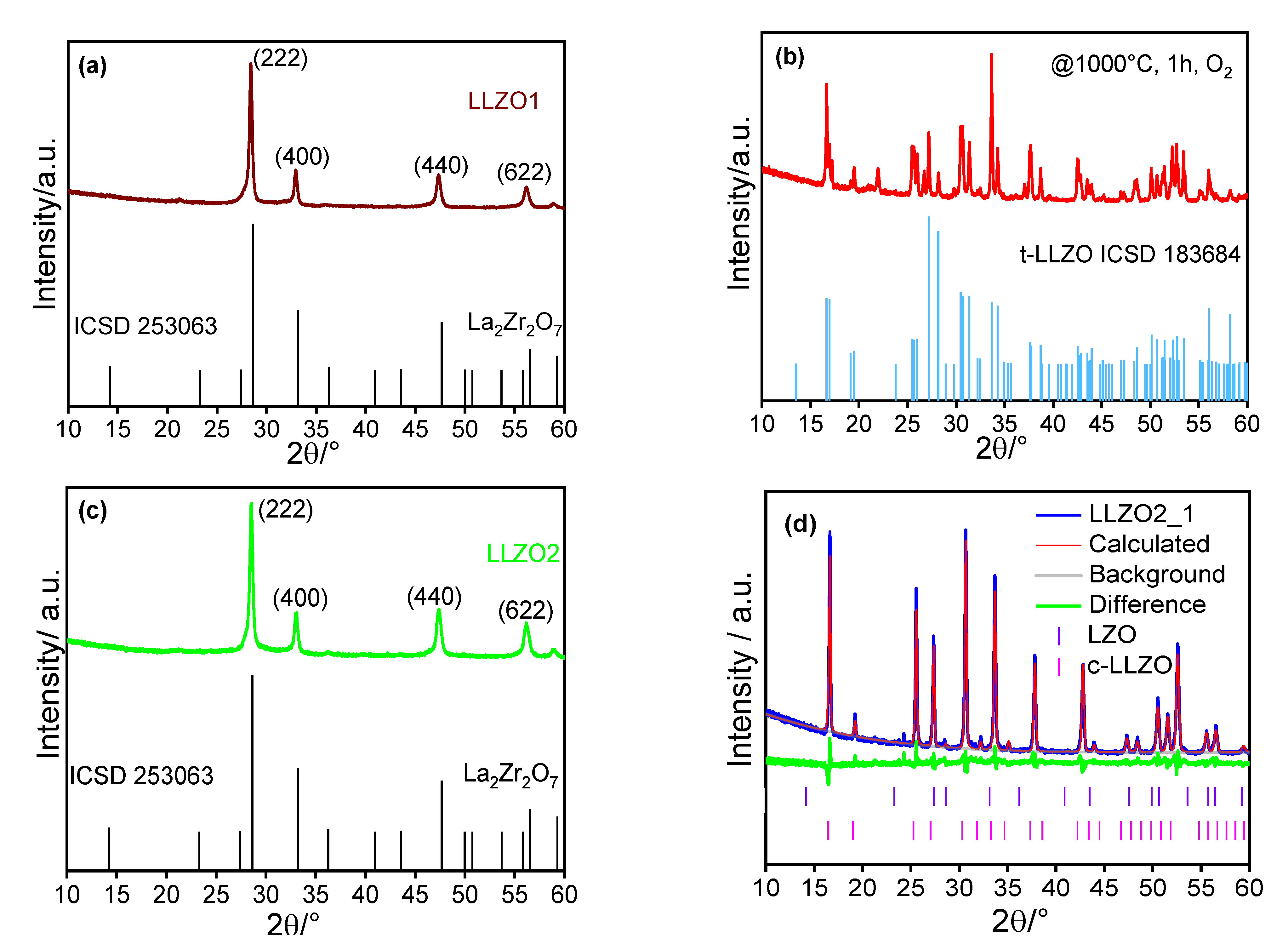
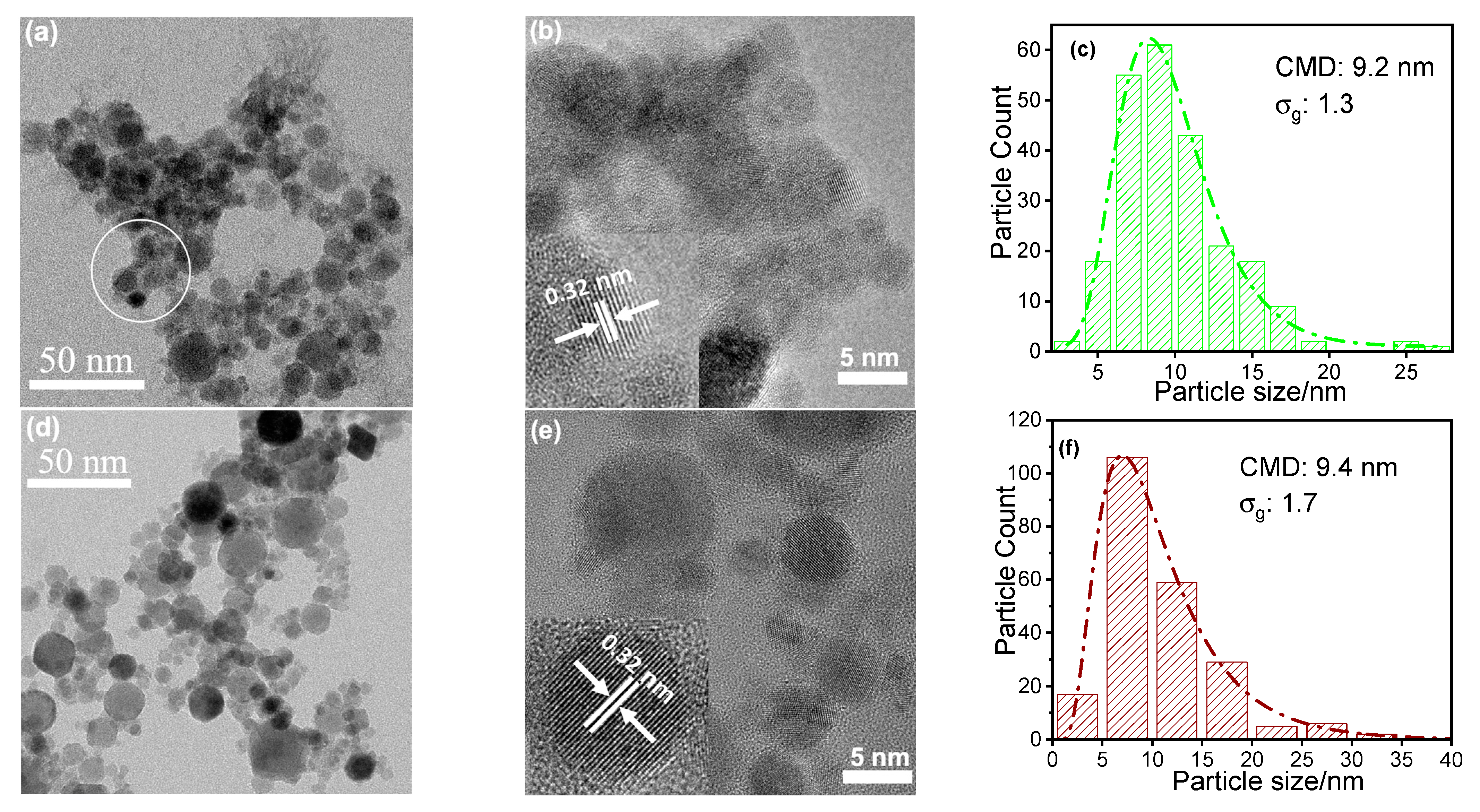
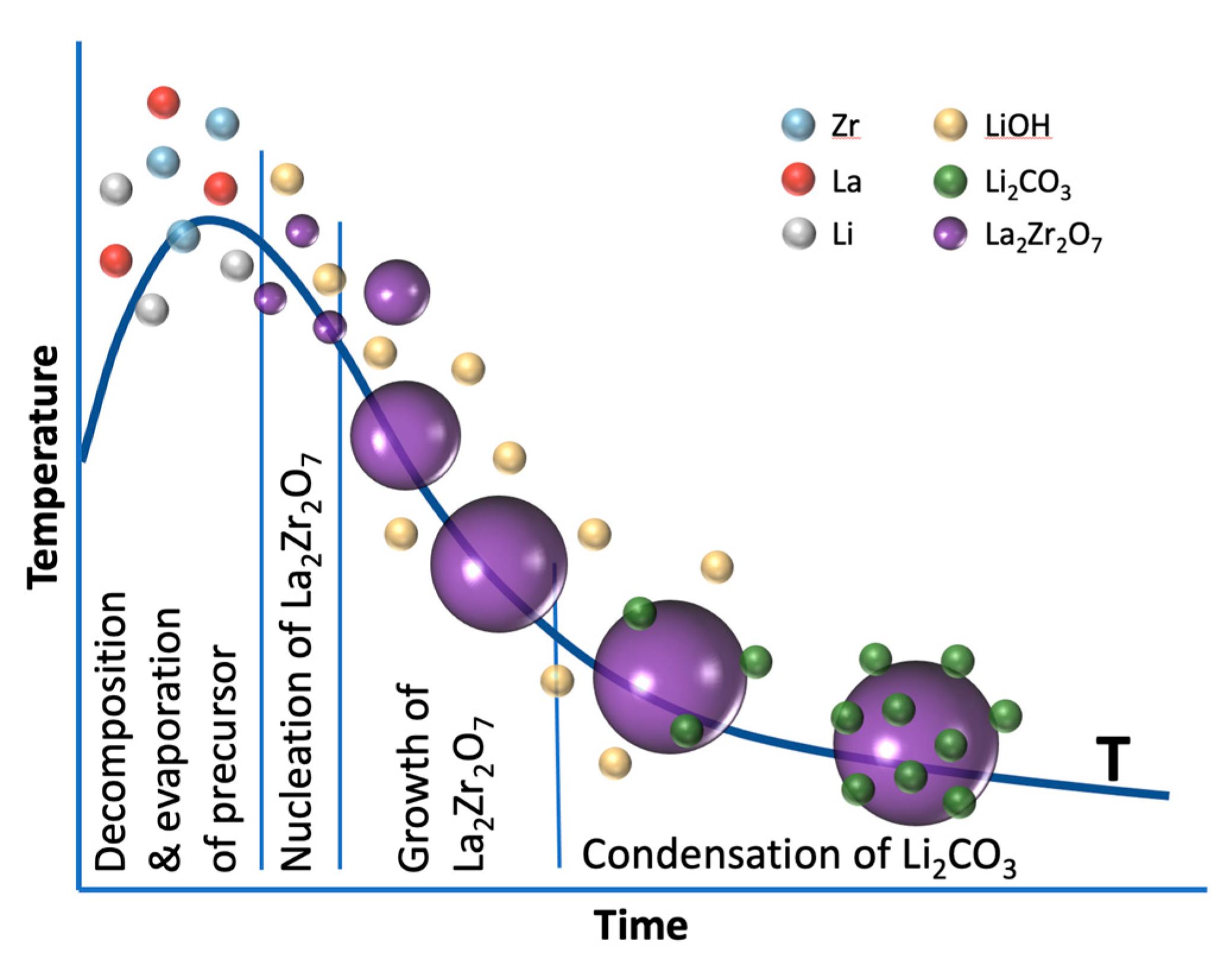
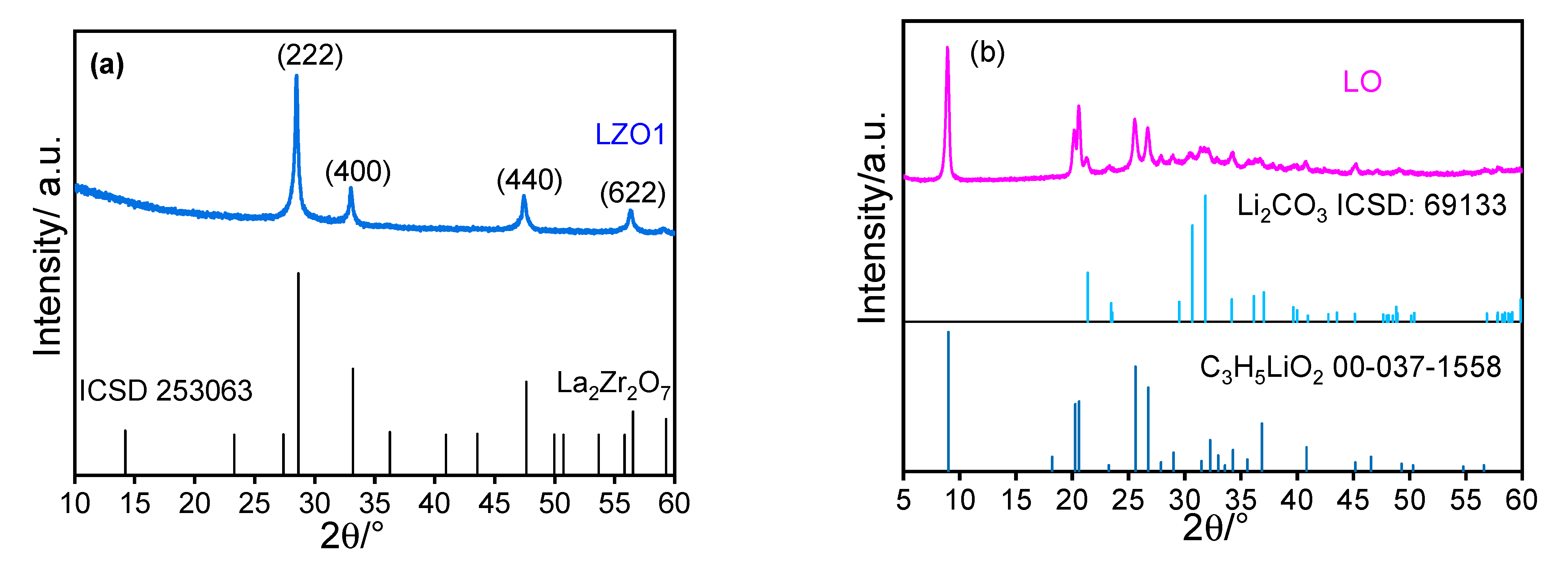
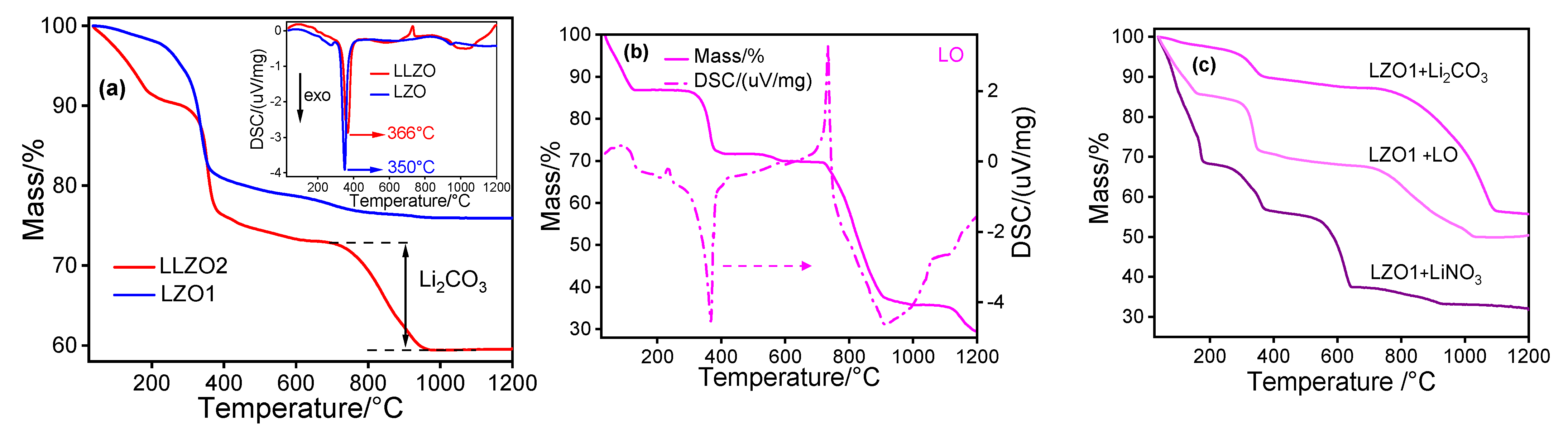
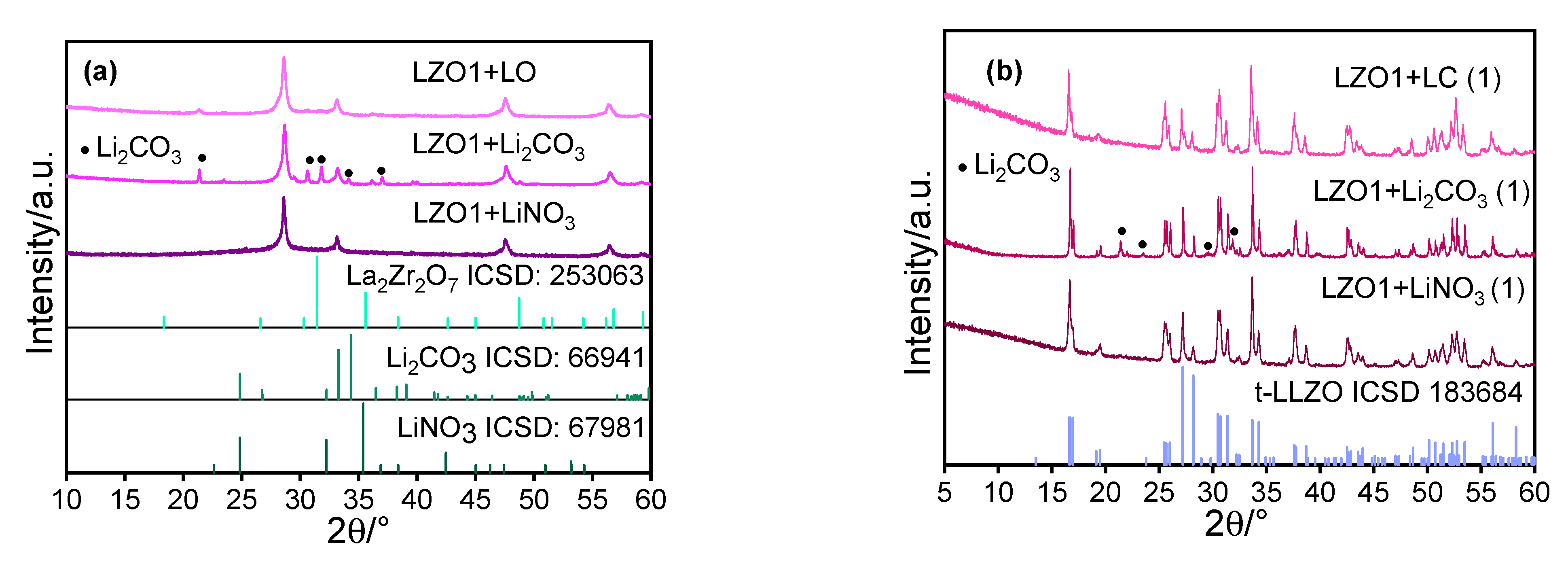
4. Results and Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Abada, S.; Marlair, G.; Lecocq, A.; Petit, M.; Sauvant-Moynot, V.; Huet, F. Safety focused modeling of lithium-ion batteries: A review. J. Power Sources 2016, 306, 178–192. [Google Scholar] [CrossRef]
- Ravdel, B.; Abraham, K.; Gitzendanner, R.; DiCarlo, J.; Lucht, B.; Campion, C. Thermal stability of lithium-ion battery electrolytes. J. Power Sources 2003, 119-121, 805–810. [Google Scholar] [CrossRef]
- Spotnitz, R.; Franklin, J. Abuse behavior of high-power, lithium-ion cells. J. Power Sources 2003, 113, 81–100. [Google Scholar] [CrossRef]
- Inoue, T.; Mukai, K. Are All-Solid-State Lithium-Ion Batteries Really Safe?–Verification by Differential Scanning Calorimetry with an All-Inclusive Microcell. ACS Appl. Mater. Interfaces 2017, 9, 1507–1515. [Google Scholar] [CrossRef] [PubMed]
- Takada, K.; Nakano, S.; Inada, T.; Kajiyama, A.; Kouguchi, M.; Sasaki, H.; Kondo, S.; Watanabe, M.; Murayama, M.; Kanno, R. Solid-state lithium batteries with sulfide-based solid electrolytes. Solid State Ionics 2004, 172, 425–436. [Google Scholar] [CrossRef]
- Judez, X.; Zhang, H.; Li, C.; Eshetu, G.G.; González-Marcos, J.A.; Armand, M.; Rodriguez-Martinez, L.M. Review—Solid Electrolytes for Safe and High Energy Density Lithium-Sulfur Batteries: Promises and Challenges. J. Electrochem. Soc. 2018, 165, A6008–A6016. [Google Scholar] [CrossRef]
- Schnell, J.; Knörzer, H.; Imbsweiler, A.J.; Reinhart, G. Solid versus Liquid—A Bottom-Up Calculation Model to Analyze the Manufacturing Cost of Future High-Energy Batteries. Energy Technol. 2020, 8. [Google Scholar] [CrossRef]
- Kim, M.-J.; Park, J.-W.; Kim, B.G.; Lee, Y.-J.; Ha, Y.-C.; Lee, S.-M.; Baeg, K.-J. Facile fabrication of solution-processed solid-electrolytes for high-energy-density all-solid-state-batteries by enhanced interfacial contact. Sci. Rep. 2020, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.; Wang, S.; Lochala, J.; Desrochers, D.; Liu, B.; Zhang, W.; Yang, J.; Xiao, J. The role of the solid electrolyte interphase layer in preventing Li dendrite growth in solid-state batteries. Energy Environ. Sci. 2018, 11, 1803–1810. [Google Scholar] [CrossRef]
- Liu, Q.; Geng, Z.; Han, C.; Fu, Y.; Li, S.; He, Y.-B.; Kang, F.; Li, B. Challenges and perspectives of garnet solid electrolytes for all solid-state lithium batteries. J. Power Sources 2018, 389, 120–134. [Google Scholar] [CrossRef]
- Han, F.; Westover, A.; Yue, J.; Fan, X.; Wang, F.; Chi, M.; Leonard, D.N.; Dudney, N.J.; Wang, H.; Wang, C. High electronic conductivity as the origin of lithium dendrite formation within solid electrolytes. Nat. Energy 2019, 4, 187–196. [Google Scholar] [CrossRef]
- Seo, J.-H.; Nakaya, H.; Takeuchi, Y.; Fan, Z.; Hikosaka, H.; Rajagopalan, R.; Gomez, E.D.; Iwasaki, M.; Randall, C.A. Broad temperature dependence, high conductivity, and structure-property relations of cold sintering of LLZO-based composite electrolytes. J. Eur. Ceram. Soc. 2020, 40, 6241–6248. [Google Scholar] [CrossRef]
- Yu, S.; Schmidt, R.D.; Garcia-Mendez, R.; Herbert, E.G.; Dudney, N.J.; Wolfenstine, J.B.; Sakamoto, J.; Siegel, D. Elastic Properties of the Solid Electrolyte Li7La3Zr2O12 (LLZO). Chem. Mater. 2016, 28, 197–206. [Google Scholar] [CrossRef]
- Sastre, J.; Priebe, A.; Döbeli, M.; Michler, J.; Tiwari, A.N.; Romanyuk, Y.E. Lithium Garnet Li 7 La 3 Zr 2 O 12 Electrolyte for All-Solid-State Batteries: Closing the Gap between Bulk and Thin Film Li-Ion Conductivities. Adv. Mater. Interfaces 2020, 7. [Google Scholar] [CrossRef]
- Zeng, X.; Li, M.; El-Hady, D.A.; Alshitari, W.; Al-Bogami, A.S.; Lu, J.; Amine, K. Commercialization of Lithium Battery Technologies for Electric Vehicles. Adv. Energy Mater. 2019, 9, 1900161. [Google Scholar] [CrossRef]
- Thangadurai, V.; Kaack, H.; Weppner, W.J.F. Novel Fast Lithium Ion Conduction in Garnet-Type Li5La3M2O12(M = Nb, Ta). J. Am. Ceram. Soc. 2003, 86, 437–440. [Google Scholar] [CrossRef]
- Murugan, R.; Thangadurai, V.; Weppner, W. Fast Lithium Ion Conduction in Garnet-Type Li7La3Zr2O12. Angew. Chem. Int. Ed. 2007, 46, 7778–7781. [Google Scholar] [CrossRef]
- Thompson, T.; Wolfenstine, J.; Allen, J.; Johannes, M.; Huq, A.; David, I.N.; Sakamoto, J. Tetragonal vs. cubic phase stability in Al – free Ta doped Li7La3Zr2O12 (LLZO). J. Mater. Chem. A 2014, 2, 13431–13436. [Google Scholar] [CrossRef]
- Geiger, C.A.; Alekseev, E.; Lazic, B.; Fisch, M.; Armbruster, T.; Langner, R.; Fechtelkord, M.; Kim, N.; Pettke, T.; Weppner, W. Crystal Chemistry and Stability of “Li7La3Zr2O12” Garnet: A Fast Lithium-Ion Conductor. Inorg. Chem. 2011, 50, 1089–1097. [Google Scholar] [CrossRef] [PubMed]
- Geng, H.; Chen, K.; Yi, D.; Mei, A.; Huang, M.; Lin, Y.; Nan, C. Formation Mechanism of Garnet-Like Li7La3Zr2O12 Powder Prepared by Solid State Reaction. Rare Met. Mater. Eng. 2016, 45, 612–616. [Google Scholar] [CrossRef]
- Paolella, A.; Zhu, W.; Bertoni, G.; Savoie, S.; Feng, Z.; Demers, H.; Gariepy, V.; Girard, G.; Rivard, E.; Delaporte, N.; et al. Discovering the Influence of Lithium Loss on Garnet Li7La3Zr2O12 Electrolyte Phase Stability. ACS Appl. Energy Mater. 2020, 3, 3415–3424. [Google Scholar] [CrossRef]
- Shin, D.O.; Oh, K.; Kim, K.M.; Park, K.-Y.; Lee, B.; Lee, Y.-G.; Kang, K. Synergistic multi-doping effects on the Li7La3Zr2O12 solid electrolyte for fast lithium ion conduction. Sci. Rep. 2015, 5, 18053. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weller, J.M.; Whetten, J.A.; Chan, C.K. Synthesis of Fine Cubic Li7La3Zr2O12 Powders in Molten LiCl–KCl Eutectic and Facile Densification by Reversal of Li+/H+ Exchange. ACS Appl. Energy Mater. 2018, 1, 552–560. [Google Scholar] [CrossRef]
- Yang, T.; Gordon, Z.D.; Li, Y.; Chan, C.K. Nanostructured Garnet-Type Solid Electrolytes for Lithium Batteries: Electrospinning Synthesis of Li7La3Zr2O12 Nanowires and Particle Size-Dependent Phase Transformation. J. Phys. Chem. C 2015, 119, 14947–14953. [Google Scholar] [CrossRef]
- Larraz, G.; Orera, A.; Sanjuán, M.L. Cubic phases of garnet-type Li7La3Zr2O12: The role of hydration. J. Mater. Chem. A 2013, 1, 11419–11428. [Google Scholar] [CrossRef] [Green Version]
- Daza, F.A.G.; Bonilla, M.R.; Llordés, A.; Carrasco, J.; Akhmatskaya, E. Atomistic Insight into Ion Transport and Conductivity in Ga/Al-Substituted Li7La3Zr2O12 Solid Electrolytes. ACS Appl. Mater. Interfaces 2018, 11, 753–765. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rangasamy, E.; Wolfenstine, J.; Sakamoto, J. The role of Al and Li concentration on the formation of cubic garnet solid electrolyte of nominal composition Li7La3Zr2O12. Solid State Ionics 2012, 206, 28–32. [Google Scholar] [CrossRef]
- Rettenwander, D.; Geiger, C.A.; Amthauer, G. Synthesis and Crystal Chemistry of the Fast Li-Ion Conductor Li7La3Zr2O12Doped with Fe. Inorg. Chem. 2013, 52, 8005–8009. [Google Scholar] [CrossRef]
- Pfenninger, R.; Struzik, M.; Garbayo, I.; Stilp, E.; Rupp, J.L.M. A low ride on processing temperature for fast lithium conduction in garnet solid-state battery films. Nat. Energy 2019, 4, 475–483. [Google Scholar] [CrossRef]
- Rosero-Navarro, N.C.; Yamashita, T.; Miura, A.; Higuchi, M.; Tadanaga, K. Effect of Sintering Additives on Relative Density and Li-ion Conductivity of Nb-Doped Li 7 La 3 ZrO 12 Solid Electrolyte. J. Am. Ceram. Soc. 2016, 100, 276–285. [Google Scholar] [CrossRef]
- Allen, J.; Wolfenstine, J.; Rangasamy, E.; Sakamoto, J. Effect of substitution (Ta, Al, Ga) on the conductivity of Li7La3Zr2O12. J. Power Sources 2012, 206, 315–319. [Google Scholar] [CrossRef]
- Lin, C.; Tang, Y.; Song, J.; Han, L.; Yu, J.; Lu, A. Sr- and Nb-co-doped Li7La3Zr2O12 solid electrolyte with Al2O3 addition towards high ionic conductivity. Appl. Phys. A 2018, 124, 439. [Google Scholar] [CrossRef]
- Chen, F.; Li, J.; Zhang, Y.; Yang, D.; Shen, Q.; Zhang, L. Effect of Mo6+ Substitution on Microstructure and Lithium Ionic Conductivity of Garnet-Type Li7La3Zr2O12 Solid Electrolytes by Field Assisted Sintering Technology. In Proceedings of the International Conference on Martensitic Transformations, Chicago, IL, USA, 9–14 July 2017; pp. 115–123. [Google Scholar]
- Ramakumar, S.; Satyanarayana, L.; Manorama, S.V.; Murugan, R. Structure and Li+ dynamics of Sb-doped Li7La3Zr2O12 fast lithium ion conductors. Phys. Chem. Chem. Phys. 2013, 15, 11327. [Google Scholar] [CrossRef]
- Schwanz, D.K.; Villa, A.; Balasubramanian, M.; Helfrecht, B.; Marinero, E.E. Bi aliovalent substitution in Li7La3Zr2O12 garnets: Structural and ionic conductivity effects. AIP Adv. 2020, 10, 035204. [Google Scholar] [CrossRef] [Green Version]
- Wagner, R.; Rettenwander, D.; Redhammer, G.J.; Tippelt, G.; Sabathi, G.; Musso, M.E.; Stanje, B.; Wilkening, M.; Suard, E.; Amthauer, G. Synthesis, Crystal Structure, and Stability of Cubic Li7–xLa3Zr2–xBixO12. Inorg. Chem. 2016, 55, 12211–12219. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hanc, E.; Zając, W.; Molenda, J. Synthesis procedure and effect of Nd, Ca and Nb doping on structure and electrical conductivity of Li7La3Zr2O12 garnets. Solid State Ionics 2014, 262, 617–621. [Google Scholar] [CrossRef]
- Kobi, S.; Amardeep, A.; Vyas, A.; Bhargava, P.P.; Mukhopadhyay, P.A. Al and Mg Co-Doping Towards Development of Air-Stable and Li-Ion Conducting Li-La-Zirconate Based Solid Electrolyte Exhibiting Low Electrode/Electrolyte Interfacial Resistance. J. Electrochem. Soc. 2020, 167, 120519. [Google Scholar] [CrossRef]
- Xia, W.; Xu, B.; Duan, H.; Guo, Y.; Kang, H.; Li, H.; Liu, H. Ionic Conductivity and Air Stability of Al-Doped Li7La3Zr2O12 Sintered in Alumina and Pt Crucibles. ACS Appl. Mater. Interfaces 2016, 8, 5335–5342. [Google Scholar] [CrossRef] [PubMed]
- Kubicek, M.; Wachter-Welzl, A.; Rettenwander, D.; Wagner, R.; Berendts, S.; Uecker, R.; Amthauer, G.; Hutter, H.; Fleig, J. Oxygen Vacancies in Fast Lithium-Ion Conducting Garnets. Chem. Mater. 2017, 29, 7189–7196. [Google Scholar] [CrossRef]
- Kun, R.; Langer, F.; Piane, M.D.; Ohno, S.; Zeier, W.G.; Gockeln, M.; Ciacchi, L.C.; Busse, M.; Fekete, I. Structural and Computational Assessment of the Influence of Wet-Chemical Post-Processing of the Al-Substituted Cubic Li7La3Zr2O12. ACS Appl. Mater. Interfaces 2018, 10, 37188–37197. [Google Scholar] [CrossRef]
- Wagner, R.; Redhammer, G.J.; Rettenwander, D.; Senyshyn, A.; Schmidt, W.; Wilkening, M.; Amthauer, G. Crystal Structure of Garnet-Related Li-Ion Conductor Li7–3xGaxLa3Zr2O12: Fast Li-Ion Conduction Caused by a Different Cubic Modification? Chem. Mater. 2016, 28, 1861–1871. [Google Scholar] [CrossRef]
- Li, J.; Liu, Z.; Ma, W.; Dong, H.; Zhang, K.; Wang, R. Low-temperature synthesis of cubic phase Li7La3Zr2O12 via sol-gel and ball milling induced phase transition. J. Power Sources 2019, 412, 189–196. [Google Scholar] [CrossRef]
- Zheng, D.; Zhang, J.; Wang, Q.; Pan, X.; Wang, J.; Guan, W.; Yang, J. Synthesis and characterization of Li7La3Zr2O12 via a novel solid-liquid route. Solid State Ionics 2020, 345, 115179. [Google Scholar] [CrossRef]
- Strobel, R.; Pratsinis, S.E. Flame aerosol synthesis of smart nanostructured materials. J. Mater. Chem. 2007, 17, 4743–4756. [Google Scholar] [CrossRef]
- Koirala, R.; Pratsinis, S.E.; Baiker, A. Synthesis of catalytic materials in flames: Opportunities and challenges. Chem. Soc. Rev. 2016, 45, 3053–3068. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, S.; Ren, Y.; Biswas, P.; Tse, S.D. Flame aerosol synthesis of nanostructured materials and functional devices: Processing, modeling, and diagnostics. Prog. Energy Combust. Sci. 2016, 55, 1–59. [Google Scholar] [CrossRef] [Green Version]
- Yi, E.; Wang, W.; Kieffer, J.; Laine, R.M. Flame made nanoparticles permit processing of dense, flexible, Li+ conducting ceramic electrolyte thin films of cubic-Li7La3Zr2O12 (c-LLZO). J. Mater. Chem. A 2016, 4, 12947–12954. [Google Scholar] [CrossRef]
- Karasulu, B.; Emge, S.P.; Groh, M.F.; Grey, C.P.; Morris, A.J. Al/Ga-Doped Li7La3Zr2O12 Garnets as Li-Ion Solid-State Battery Electrolytes: Atomistic Insights into Local Coordination Environments and Their Influence on 17O, 27Al, and 71Ga NMR Spectra. J. Am. Chem. Soc. 2020, 142, 3132–3148. [Google Scholar] [CrossRef] [Green Version]
- Schneider, F.; Suleiman, S.; Menser, J.; Borukhovich, E.; Wlokas, I.; Kempf, A.; Wiggers, H.; Schulz, C. SpraySyn—A standardized burner configuration for nanoparticle synthesis in spray flames. Rev. Sci. Instruments 2019, 90, 085108. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Kumar, R.; Roller, J.; Maric, R. Synthesis and Characterization of Nano-crystalline La2Zr2O7 Film by Reactive Spray Deposition Technology for Application in Thermal Barrier Coatings. MRS Adv. 2017, 2, 1519–1525. [Google Scholar] [CrossRef]
- Djenadic, R.; Botros, M.; Benel, C.; Clemens, O.; Indris, S.; Choudhary, A.; Bergfeldt, T.; Hahn, H. Nebulized spray pyrolysis of Al-doped Li7La3Zr2O12 solid electrolyte for battery applications. Solid State Ionics 2014, 263, 49–56. [Google Scholar] [CrossRef]
- Kleinwechter, H.; Janzen, C.; Knipping, J.; Wiggers, H.; Roth, P. Formation and properties of ZnO nano-particles from gas phase synthesis processes. J. Mater. Sci. 2002, 37, 4349–4360. [Google Scholar] [CrossRef]
- Alkan, B.; Cychy, S.; Varhade, S.; Muhler, M.; Schulz, C.; Schuhmann, W.; Wiggers, H.; Andronescu, C. Spray-Flame-Synthesized LaCo 1− x Fe x O 3 Perovskite Nanoparticles as Electrocatalysts for Water and Ethanol Oxidation. ChemElectroChem 2019, 6, 4266–4274. [Google Scholar] [CrossRef] [Green Version]
- Timoshevskii, A.N.; Ktalkherman, M.G.; Emel’Kin, V.A.; Pozdnyakov, B.A.; Zamyatin, A.P. High-temperature decomposition of lithium carbonate at atmospheric pressure. High Temp. 2008, 46, 414–421. [Google Scholar] [CrossRef]
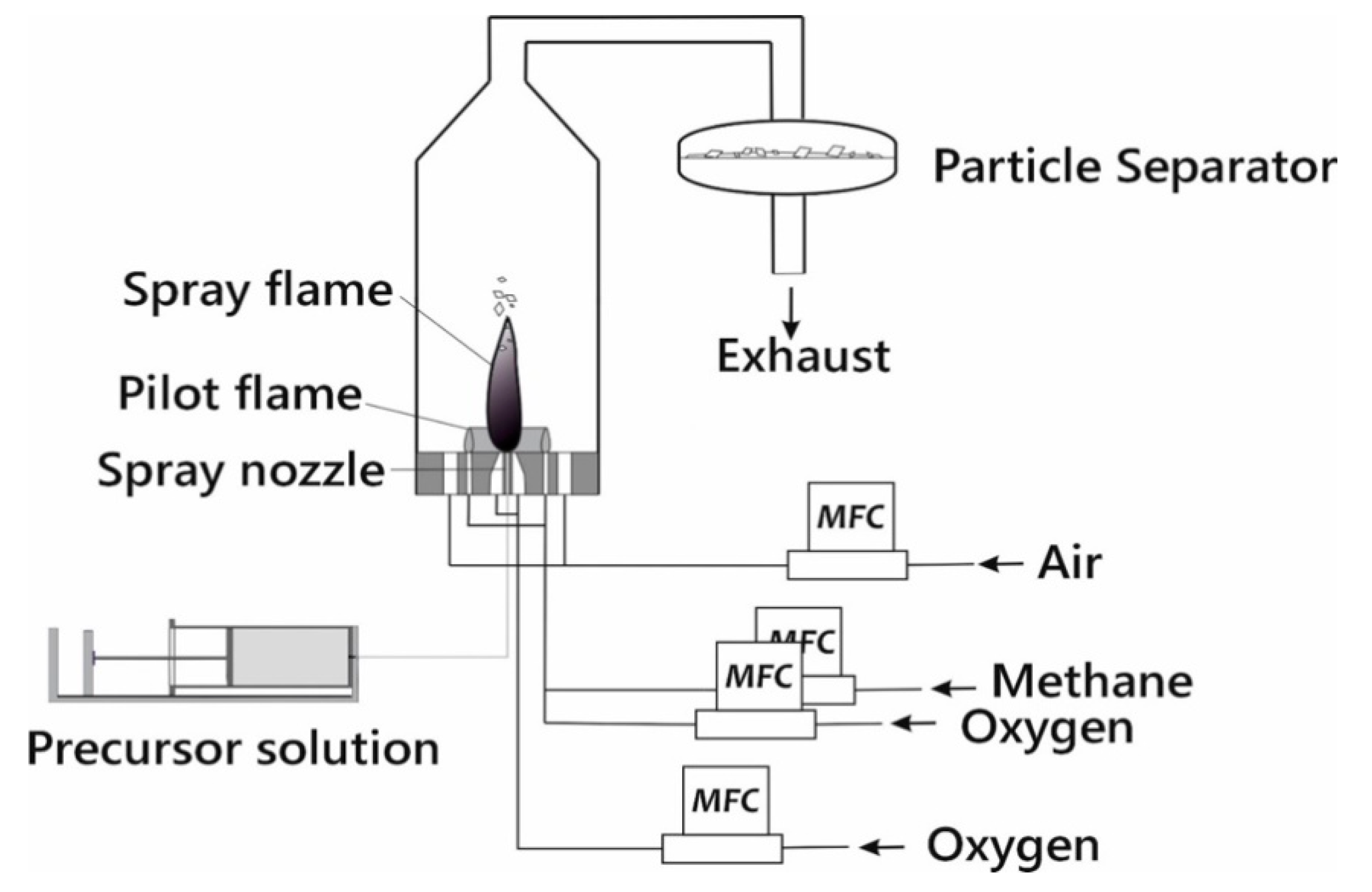
| Nomenclature | Product Name | Precursor | Solvent | |||
|---|---|---|---|---|---|---|
| LiNO3 | La(OAC)3 | Al(NO3)3 | Zr-Propoxide | Propanol + propionic acid 1:1 by vol | ||
| SolLLZO1 | LLZO1 | ✓ | ✓ | ⅹ | ✓ | ✓ |
| SolLLZO2 | LLZO2 | (50%excess) ✓ | ✓ | ✓ | ✓ | ✓ |
| SolLZO | LZO | ⅹ | ✓ | ✓ | ✓ | ✓ |
| SolLO | LO | ✓ | ⅹ | ⅹ | ⅹ | ✓ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ali, M.Y.; Orthner, H.; Wiggers, H. Spray Flame Synthesis (SFS) of Lithium Lanthanum Zirconate (LLZO) Solid Electrolyte. Materials 2021, 14, 3472. https://doi.org/10.3390/ma14133472
Ali MY, Orthner H, Wiggers H. Spray Flame Synthesis (SFS) of Lithium Lanthanum Zirconate (LLZO) Solid Electrolyte. Materials. 2021; 14(13):3472. https://doi.org/10.3390/ma14133472
Chicago/Turabian StyleAli, Md Yusuf, Hans Orthner, and Hartmut Wiggers. 2021. "Spray Flame Synthesis (SFS) of Lithium Lanthanum Zirconate (LLZO) Solid Electrolyte" Materials 14, no. 13: 3472. https://doi.org/10.3390/ma14133472
APA StyleAli, M. Y., Orthner, H., & Wiggers, H. (2021). Spray Flame Synthesis (SFS) of Lithium Lanthanum Zirconate (LLZO) Solid Electrolyte. Materials, 14(13), 3472. https://doi.org/10.3390/ma14133472






