Chemo-Enzymatic Baeyer–Villiger Oxidation Facilitated with Lipases Immobilized in the Supported Ionic Liquid Phase
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.3. Synthetic Procedures
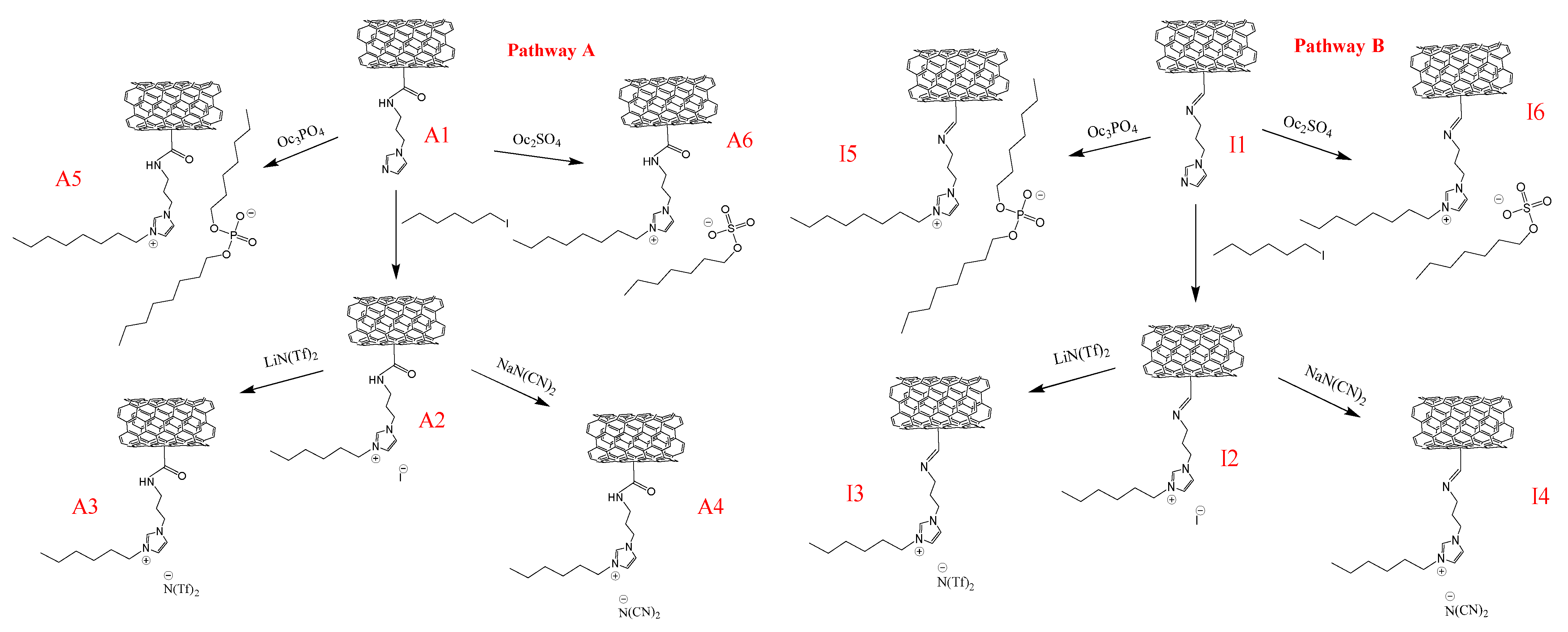
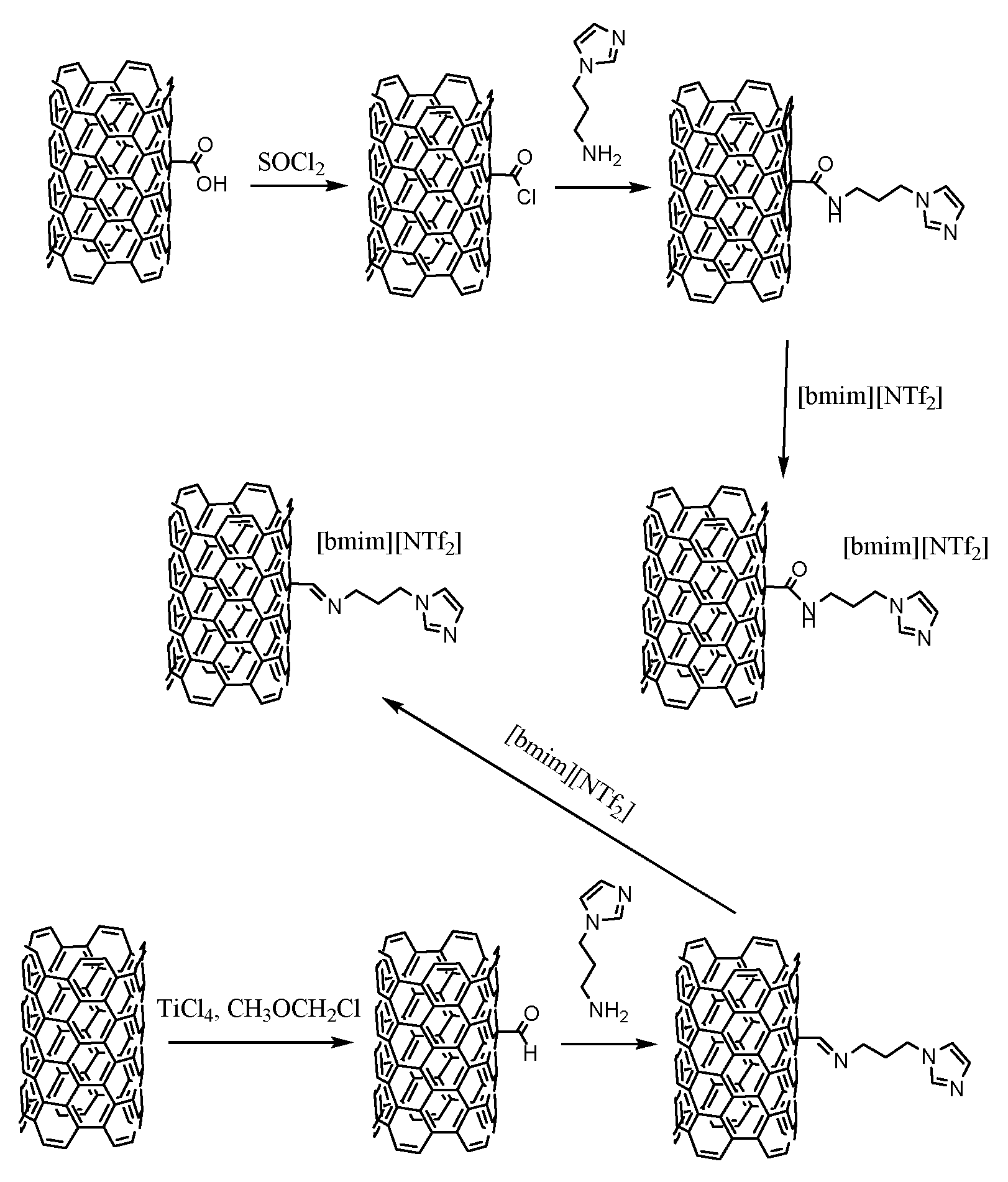
2.3.1. Synthesis of CNTs-CONH (A1)
2.3.2. Synthesis of CNTs-C=N (I1)
2.3.3. Synthesis of CNTs-CONH-HEX-I (A2) and CNTs-C=N-HEX-I (I2)
2.3.4. Synthesis of CNTs-CONH-HEX-N(Tf)2 (A3) and CNTs-C=N-HEX- N(Tf)2 (I3)
2.3.5. Synthesis of CNTs-CONH-HEX-N(CN)2 (A4) and CNTs-C=N-HEX- N(CN)2 (I4)
2.3.6. Synthesis of CNTs-CONH-Oc2PO4 (A5) or CNTs-C=N- Oc2PO4 (I5)
2.3.7. Synthesis of CNTs-CONH-OcSO4 (A6) or CNTs-C=N- OcSO4 (I6)
2.3.8. Synthesis of CNTs-CONH-[bmim][N(Tf)2] and CNTs-C=N-[bmim][N(Tf)2]
2.3.9. Immobilization of the Lipase
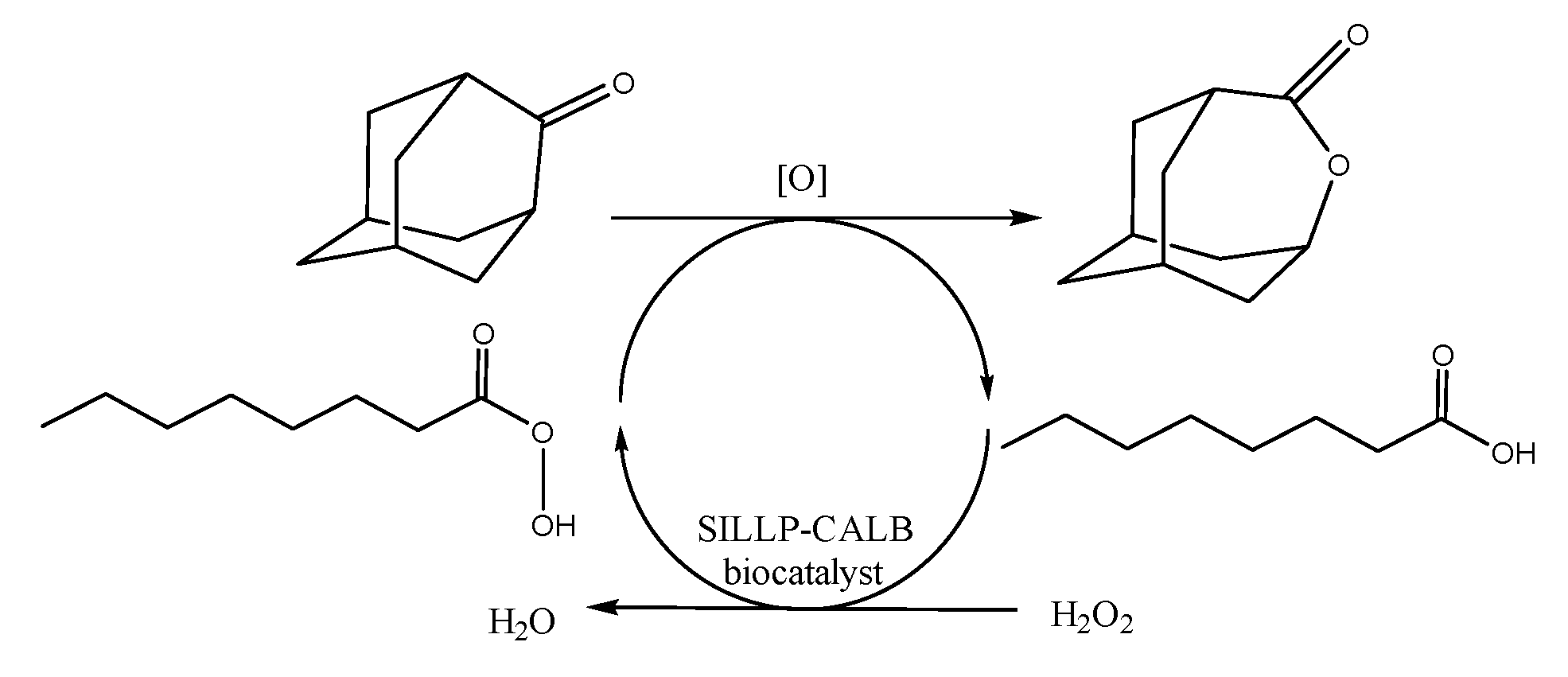
2.3.10. General Procedure of the Baeyer–Villiger Oxidation of 2-Adamantanone
2.3.11. Recycle of the Biocatalyst
3. Results and Discussion
3.1. Synthesis and Characterization of Biocatalysts
3.2. SILLP Biocatalysts in Chemo-Enzymatic Baeyer–Villiger Oxidation
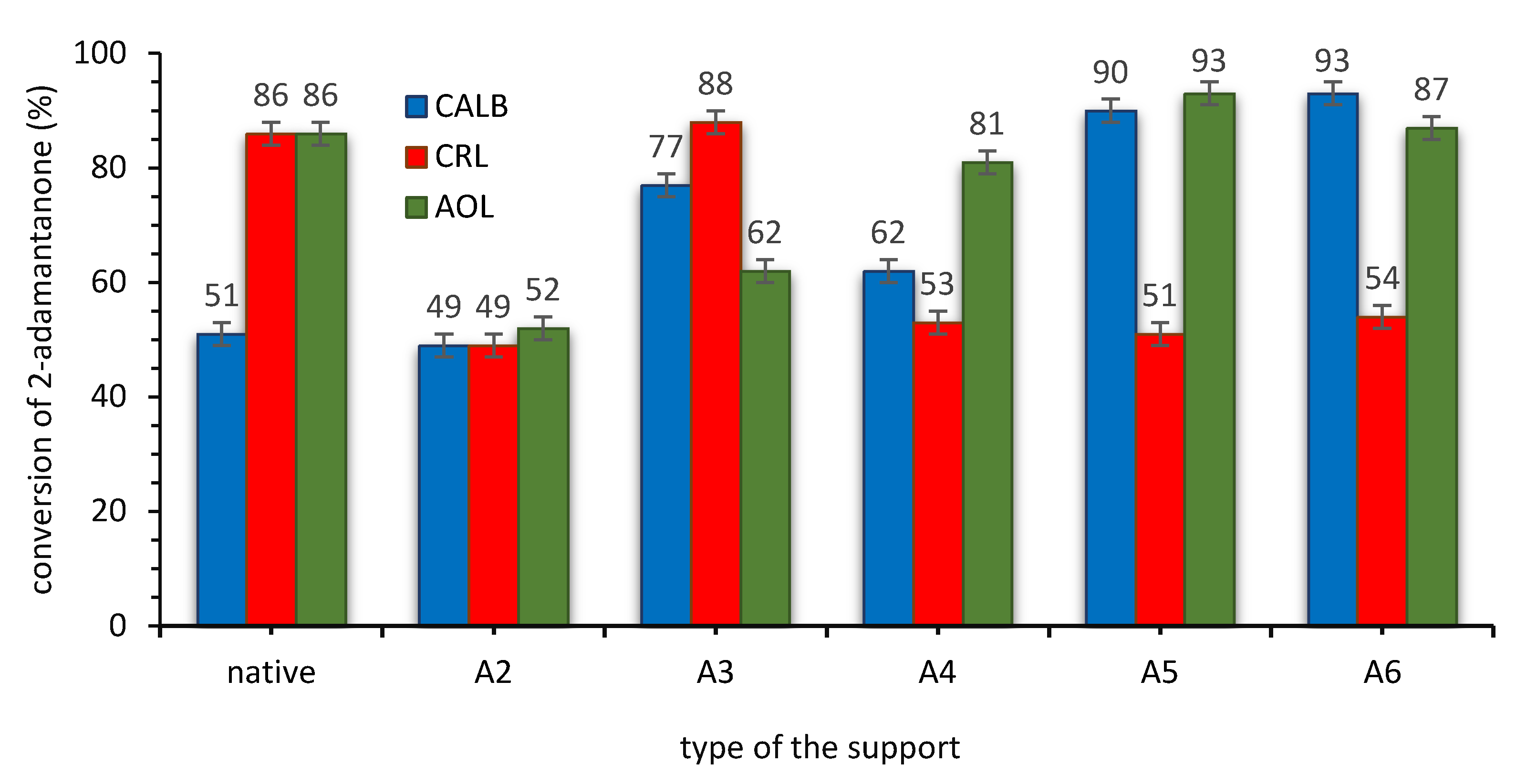
3.2.1. Influence of the Structure of SILLP Biocatalyst
3.2.2. Influence of the Amount of SILLP Biocatalyst
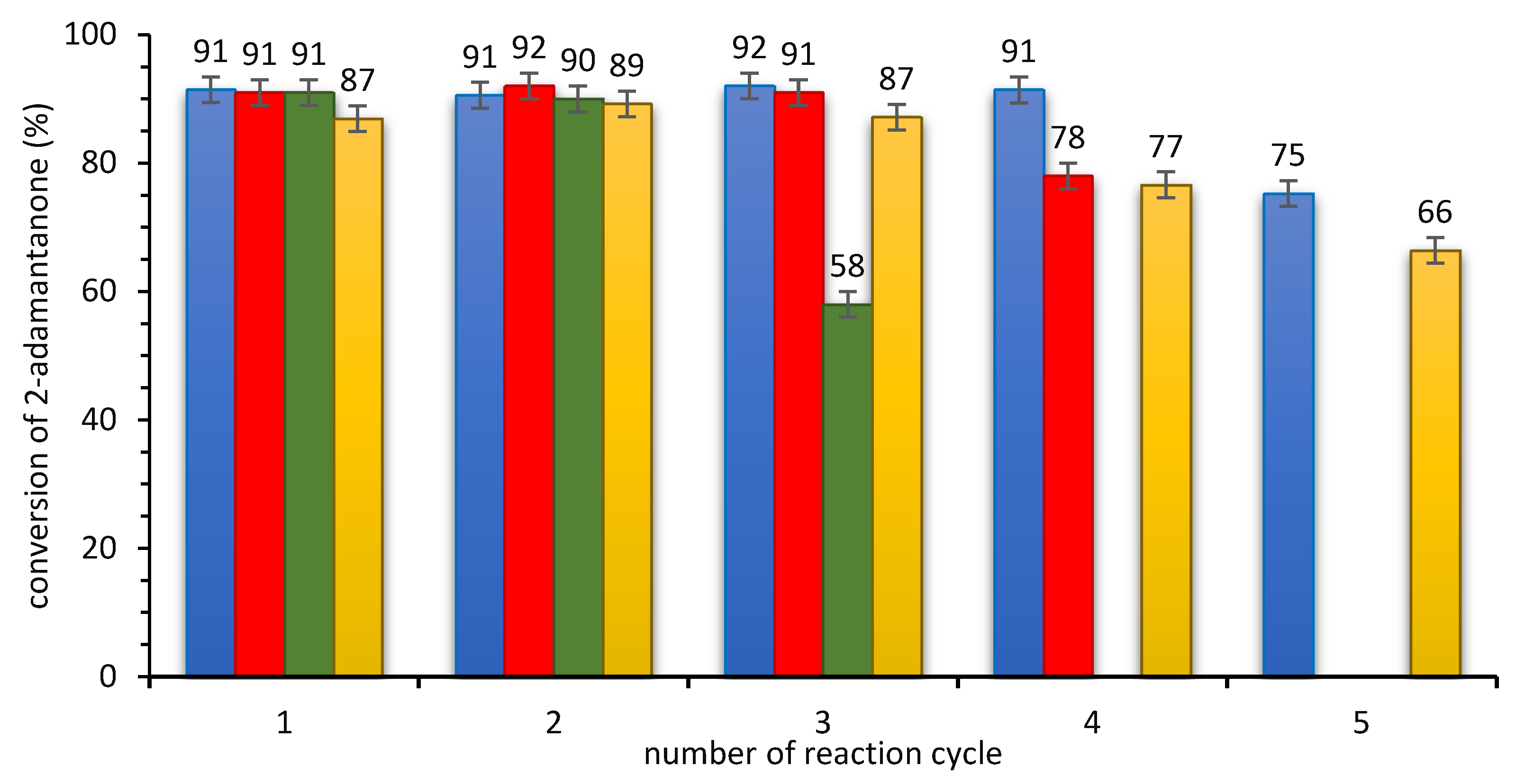
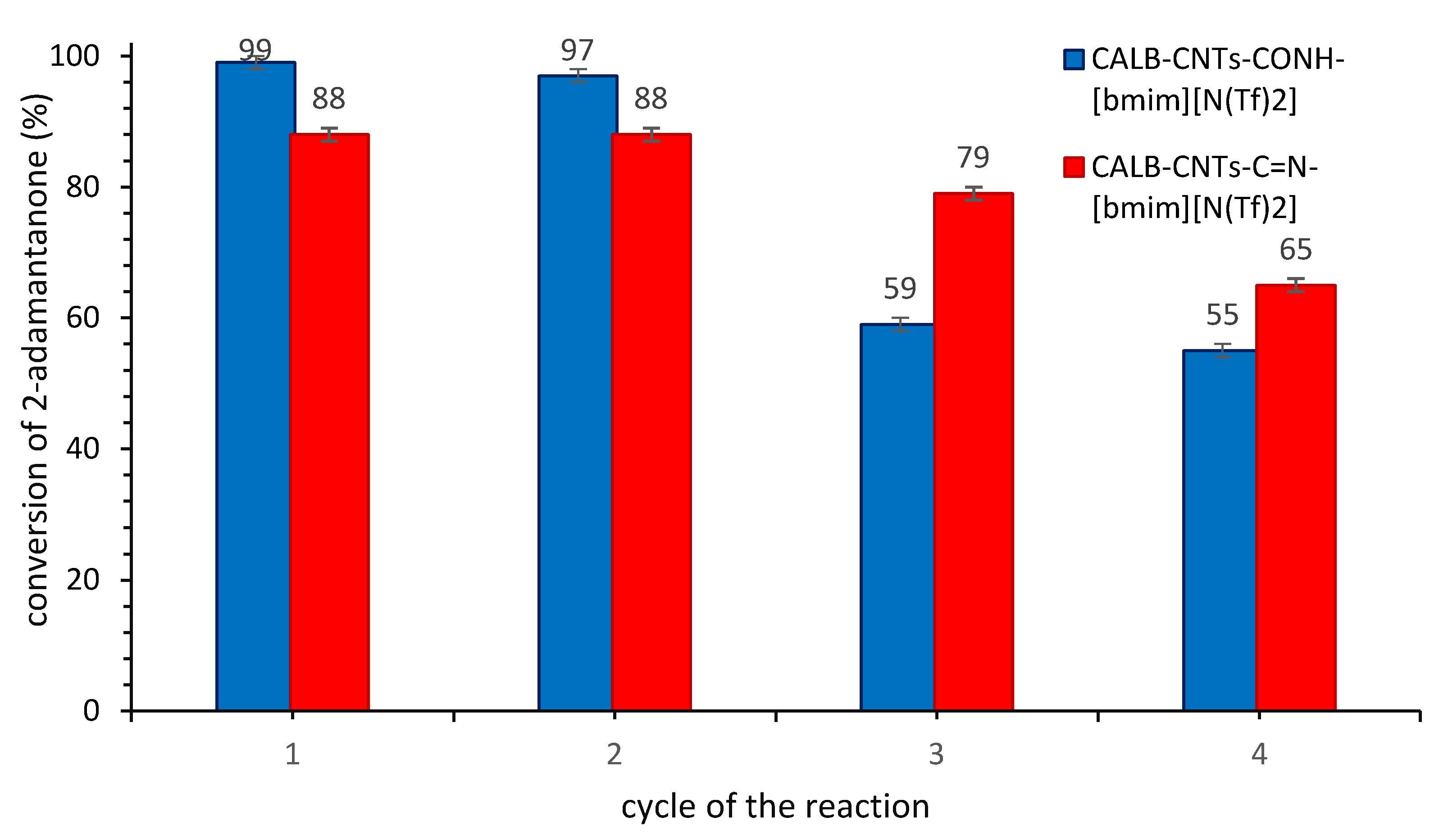
3.2.3. Stability of SILLP Biocatalyst
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sheldon, R.A.; Woodley, J.M. Role of biocatalysis in sustainable chemistry. Chem. Rev. 2018, 118, 801–838. [Google Scholar] [CrossRef]
- Chapman, J.; Ismail, A.E.; Dinu, C.Z. Industrial applications of enzymes: Recent advances, techniques, and outlooks. Catalysts 2018, 8, 238. [Google Scholar] [CrossRef] [Green Version]
- Basso, A.; Serban, S. Industrial applications of immobilized enzymes—A review. J. Mol. Catal. A Chem. 2019, 479, 110607. [Google Scholar] [CrossRef]
- Wu, S.; Bornscheuer, U.T.; Snajdrova, R.; Moore, J.C.; Baldenius, K. Biocatalysis: Enzymatic synthesis for industrial applications. Angew. Chem. 2021, 60, 88–119. [Google Scholar] [CrossRef]
- Cipolatti, P.; Valerio, A.; Henriques, R.O.; Moritz, D.E.; Ninow, J.L.; Freire, D.M.G.; Manoel, E.A.; Fernandez-Lafuente, R.; de Oliveira, D. Nanomaterials for biocatalyst immobilization–state of the art and future trends. RSC Adv. 2016, 6, 104675–104692. [Google Scholar] [CrossRef]
- Sheldon, R.A.; van Pelt, S. Enzyme immobilisation in biocatalysis: Why, what and how. Chem. Soc. Rev. 2013, 42, 6223–6235. [Google Scholar] [CrossRef] [Green Version]
- Stepankova, V.; Bidmanova, S.; Koudelakova, T.; Prokop, Z.; Chaloupkova, R.; Damborsky, J. Strategies for stabilization of enzymes in organic solvents. ACS Catal. 2013, 3, 2823–2836. [Google Scholar] [CrossRef]
- Zdarta, J.; Meyer, A.S.; Jesionowski, T.; Pinelo, M. A general overview of support materials for enzyme immobilization: Characteristics, properties, practical utility. Catalysts 2018, 8, 92. [Google Scholar] [CrossRef] [Green Version]
- Patila, M.; Chalmpes, N.; Dounousi, E.; Stamatis, H.; Gournis, D. Chapter Twelve-Use of functionalized carbon nanotubes for the development of robust nanobiocatalysts. In Methods in Enzymology; Elsevier: Amsterdam, The Netherlands, 2020; Volume 630, pp. 263–301. [Google Scholar] [CrossRef]
- Coleman, J.N.; Khan, U.; Blau, W.J.; Gun’ko, Y.K. Small but strong: A review of the mechanical properties of carbon nanotube–polymer composites. Carbon 2006, 44, 1624–1652. [Google Scholar] [CrossRef]
- Feng, W.; Li, P. Enzymes immobilized on carbon nanotubes. Biotechnol. Adv. 2011, 29, 889–895. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, C.; Ferreira, M.L.; Barbosa, O.; dos Santos, J.C.S.; Rodrigues, R.C.; Berenguer-Murcia, A.; Briand, L.A.; Fernandez-Lafuente, R. Novozym 435: The “perfect” lipase immobilized biocatalyst? Catal. Sci. Technol. 2019, 9, 2380–2420. [Google Scholar] [CrossRef] [Green Version]
- Cheng, C.; Jiang, T.; Wu, Y.; Cui, L.; Quin, S.; He, B. Elucidation of lid open and orientation of lipase activated in interfacial activation by amphiphilic environment. Int. J. Biol. Macromol. 2018, 119, 1211–1217. [Google Scholar] [CrossRef]
- Arana-Peña, S.; Riosa, N.S.; Carballares, D.; Gonçalves, L.R.B.; Fernandez-Lafuente, R. Immobilization of lipases via interfacial activation on hydrophobic supports: Production of biocatalysts libraries by altering the immobilization conditions. Catal. Today 2021, 362, 130–140. [Google Scholar] [CrossRef]
- Szelwicka, A.; Kolanowska, A.; Latos, P.; Jurczyk, S.; Boncel, S.; Chrobok, A. Carbon nanotube/PTFE as a hybrid platform for lipase B from Candida antarctica in transformation of α-angelica lactone into alkyl levulinates. Catal. Sci. Technol. 2020, 10, 3255–3264. [Google Scholar] [CrossRef]
- Markiton, M.; Boncel, S.; Janas, S.; Chrobok, A. Highly active nanobiocatalyst from lipase noncovalently immobilized on multiwalled carbon nanotubes for Baeyer–Villiger synthesis of lactones. ACS Sustain. Chem. Eng. 2017, 5, 1685–1691. [Google Scholar] [CrossRef]
- Szelwicka, A.; Zawadzki, P.; Sitko, M.; Boncel, S.; Czardybon, W.; Chrobok, A. Continuous flow chemo-enzymatic Baeyer–Villiger oxidation with superactive and extra-stable enzyme/carbon nanotube catalyst: An efficient upgrade from batch to flow. Org. Process Res. Dev. 2019, 23, 1386–1395. [Google Scholar] [CrossRef]
- Szelwicka, A.; Boncel, S.; Jurczyk, S.; Chrobok, A. Exceptionally active and reusable nanobiocatalyst comprising lipase non-covalently immobilized on multi-wall carbon nanotubes for the synthesis of diester plasticizers. Appl. Catal. A Gen. 2019, 574, 41–47. [Google Scholar] [CrossRef]
- Szelwicka, A.; Siewniak, A.; Kolanowska, A.; Boncel, S.; Chrobok, A. PTFE-carbon nanotubes and lipase B from Candida antarctica-long-lasting marriage for ultra-fast and fully selective synthesis of levulinate esters. Materials 2021, 14, 1518. [Google Scholar] [CrossRef]
- Szelwicka, A.; Erfurt, K.; Jurczyk, S.; Boncel, S.; Chrobok, A. Outperformance in acrylation: Supported D-glucose-based ionic liquid phase on MWCNTs for immobilized lipase B from candida antarctica as catalytic system. Materials 2021, 14, 3090. [Google Scholar] [CrossRef]
- Itoh, T. Ionic liquids as tool to improve enzymatic organic synthesis. Chem. Rev. 2017, 117, 10567–10607. [Google Scholar] [CrossRef]
- Zhao, H. Protein stabilization and enzyme activation in ionic liquids: Specific ion effects. J. Chem. Technol. Biotechnol. 2016, 91, 25–50. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sureshkumar, M.; Lee, C.K.J. Biocatalytic reactions in hydrophobic ionic liquids. J. Mol. Catal. B Enzym. 2009, 60, 1–12. [Google Scholar] [CrossRef]
- Lozano, P.; Alvarez, E.; Bernal, J.M.; Nieto, S.; Gomez, C.; Sanchez-Gomez, G. Ionic liquids for clean biocatalytic processes. Curr. Green Chem. 2017, 4, 116–129. [Google Scholar] [CrossRef]
- Lau, R.M.; Sorgedrager, M.J.; Carrea, G.; van Rantwijk, F.; Secundo, F.; Sheldon, R.A. Dissolution of Candida antarctica lipase B in ionic liquids: Effects on structure and activity. Green Chem. 2004, 6, 483–487. [Google Scholar] [CrossRef]
- Arce, A.; Earle, M.J.; Rodriguez, H.; Seddon, K.R.; Soto, A. Bis{(trifluoromethyl)sulfonyl}amide ionic liquids as solvents for the extraction of aromatic hydrocarbons from their mixtures with alkanes: Effect of the nature of the cation. Green Chem. 2009, 11, 365–372. [Google Scholar] [CrossRef]
- Brzeczek-Szafran, A.; Wieclawik, J.; Barteczko, N.; Szelwicka, A.; Byrne, E.L.; Kolanowska, A.; Swadzba-Kwasny, M.; Chrobok, A. Protic ionic liquids from di- or triamines: Even cheaper Brønsted acidic catalysts. Green Chem. 2021. [Google Scholar] [CrossRef]
- MacFarlane, D.; Tachikawa, N.; Forsyth, M.; Pringle, J.M.; Howlett, P.C.; Elliot, G.D.; Davis, J.H.; Watanabe, M.; Simon, P.; Angell, C.A. Energy applications of ionic liquids. Energy Environ. Sci. 2014, 7, 232–250. [Google Scholar] [CrossRef] [Green Version]
- Lu, C.; Chen, K.; Shi, G.; Lin, W.; Bai, H.; Li, H.; Tang, G.; Wang, C. Design and tuning of ionic liquid–based HNO donor through intramolecular hydrogen bond for efficient inhibition of tumor growth. Sci. Adv. 2020, 6, e7788. [Google Scholar] [CrossRef]
- Chen, K.; Shi, G.; Zhou, X.; Li, H.; Wang, C. Highly efficient nitric oxide capture by azole-based ionic liquids through multiple-site absorption. Angew. Chem. Int. Edit. 2016, 55, 14362–14366. [Google Scholar] [CrossRef]
- Garcia-Verdugo, E.; Lozano, P.; Lui, S.V. Biocatalytic Processes Based on Supported Ionic Liquids. In Supported Ionic Liquids: Fundamentals and Applications; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2014; pp. 351–368. [Google Scholar] [CrossRef]
- Lai, J.Q.; Li, Z.; Lu, Y.H.; Yang, Z. Specific ion effects of ionic liquids on enzyme activity and stability. Green Chem. 2011, 13, 1860–1868. [Google Scholar] [CrossRef]
- Wan, X.; Xiang, X.; Tang, S.; Yu, D.; Huang, H.; Hu, Y. Immobilization of Candida antarctica lipase B on MWNTs modified by ionic liquids with different functional groups. Colloids Surf. B 2017, 160, 416–422. [Google Scholar] [CrossRef]
- Sandig, B.; Michalek, L.; Vlahovic, S.; Antonovici, M.; Hauer, B.; Buchmeiser, M.R. A monolithic hybrid cellulose-2.5-acetate/polymer bioreactor for biocatalysis under continuous liquid–liquid conditions using a supported ionic liquid phase. Chem. Eur. J. 2015, 21, 15835–15842. [Google Scholar] [CrossRef] [PubMed]
- Peris, E.; Porcar, R.; Burguete, M.I.; Garcia-Verdugo, E.; Luis, S.V. Supported ionic liquid-like phases (SILLPs) as immobilised catalysts for the multistep and multicatalytic continuous flow synthesis of chiral cyanohydrins. ChemCatChem 2019, 11, 1955–1962. [Google Scholar] [CrossRef]
- Wan, X.; Tang, S.; Xiang, X.; Huang, H.; Hu, Y. Immobilization of Candida antarctica Lipase B on functionalized ionic liquid modified MWNTs. Appl. Biochem. Biotechnol. 2017, 183, 807–819. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, K.; Berenguer-Murcia, A.; Rodrigues, R.C.; Fernandez-Lafuente, R. Hydrogen peroxide in biocatalysis. A dangerous liaison. Curr. Org. Chem. 2012, 16, 2652–2672. [Google Scholar] [CrossRef]
- Renz, M.; Meunier, B. 100 years of Baeyer–Villiger oxidations. Eur. J. Org. Chem. 1999, 4, 737–750. [Google Scholar] [CrossRef]
- Brink, G.; Arends, I.; Sheldon, R. The Baeyer−Villiger reaction: New developments toward greener procedures. Chem. Rev. 2004, 104, 4105–4123. [Google Scholar] [CrossRef]
- Rios, M.Y.; Salazar, E.; Olivo, H.F. Baeyer-Villiger oxidation of substituted cyclohexanones via lipase-mediated perhydrolysis utilizing urea-hydrogen peroxide in ethyl acetate. Green. Chem. 2007, 9, 459–462. [Google Scholar] [CrossRef]
- Chavez, G.; Hatti-Kaul, R.; Sheldon, R.A.; Mamo, G. Baeyer-Villiger oxidation with peracid generated in situ by CALB-CLEA catalyzed perhydrolysis. J. Mol. Catal. B Enzym. 2013, 89, 67–72. [Google Scholar] [CrossRef]
- Chavez, G.; Rasmussen, J.; Janssen, M.; Mamo, G.; Hatti-Kaul, R.; Sheldon, R.A. Baeyer–Villiger oxidation of cyclohexanone in aqueous medium with in situ generation of peracid catalyzed by perhydrolase CLEA. Top. Catal. 2014, 57, 349–355. [Google Scholar] [CrossRef]
- Drożdż, A.; Chrobok, A.; Baj, S.; Szymańska, K.; Mrowiec-Białoń, J.; Jarzębski, A. The chemo-enzymatic Baeyer–Villiger oxidation of cyclic ketones with an efficient silica-supported lipase as a biocatalyst. Appl. Catal. A Gen. 2013, 467, 163–170. [Google Scholar] [CrossRef]
- Drożdż, A.; Hanefeld, U.; Szymańska, K.; Jarzębski, A.; Chrobok, A. A robust chemo-enzymatic lactone synthesis using acyltransferase from Mycobacterium smegmatis. Catal. Commun. 2016, 81, 37–40. [Google Scholar] [CrossRef]
- Kotlewska, J.; Rantwijk, F.; Sheldon, R.A.; Arends, I.W.C.E. Epoxidation and Baeyer–Villiger oxidation using hydrogen peroxide and a lipase dissolved in ionic liquids. Green. Chem. 2011, 13, 2154–2160. [Google Scholar] [CrossRef]
- Drożdż, A.; Erfurt, K.; Bielas, R.; Chrobok, A. Chemo-enzymatic Baeyer–Villiger oxidation in the presence of Candida antarctica lipase B and ionic liquids. New J. Chem. 2015, 39, 1315–1321. [Google Scholar] [CrossRef]
- Drożdż, A.; Chrobok, A. Chemo-enzymatic Baeyer-Villiger oxidation of 4-methylcyclohexanone via kinetic resolution of racemic carboxylic acids: Direct access to enantioenriched lactone. Chem. Commun. 2016, 52, 1230–1233. [Google Scholar] [CrossRef]
- Kolanowska, A.; Kuziel, A.; Li, Y.; Jurczyk, S.; Boncel, S. Rieche formylation of carbon nanotubes–one-step and versatile functionalization route. RSC Adv. 2017, 7, 51374–51381. [Google Scholar] [CrossRef] [Green Version]
- Talotta, C.; Gaeta, C.; Pierro, T.; Neri, P. Sequence stereoisomerism in calixarene-based pseudo[3]rotaxanes. Org. Lett. 2011, 13, 2098–2101. [Google Scholar] [CrossRef] [PubMed]
- Domínguez de María, P.; Sánchez-Montero, J.M.; Sinisterra, J.V.; Alcántara, A.R. Understanding Candida rugosa lipases: An overview. Biotechnol. Adv. 2006, 24, 180–196. [Google Scholar] [CrossRef] [PubMed]
- Grochulski, P.; Li, Y.; Schrag, J.D.; Cygler, M. Two conformational states of Candida rugosa lipase. Protein Sci. 1994, 3, 82–91. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lan, D.; Zhao, G.; Holzmann, N.; Yuan, S.; Wang, J.; Wang, Y. Structure-guided rational design of a mono- and diacylglycerol lipase from Aspergillus Oryzae: A single residue mutant increases the hydrolysis ability. J. Agric. Food Chem. 2021, 69, 5344–5352. [Google Scholar] [CrossRef]
- Uppenberg, J.; Hansen, M.T.; Patkar, S.; Jones, T.A. The sequence, crystal structure determination and refinement of two crystal forms of lipase B from Candida antarctica. Structure 1994, 2, 293–308. [Google Scholar] [CrossRef] [Green Version]
- Fisher, S.J.; Blakeley, M.P.; Cianci, M.; McSweeney, S.; Helliwell, J.R. Protonation-state determination in proteins using high-resolution X-ray crystallography: Effects of resolution and completeness. Acta Crystallogr. Sect. D Biol. Crystallogr. 2012, 68, 800–809. [Google Scholar] [CrossRef] [PubMed]
- Okur, H.I.; Hladilkova, J.; Rembert, K.B.; Cho, Y.; Heyda, J.; Dzubiella, J.; Cremer, P.S.; Jungwirth, P. Beyond the Hofmeister series: Ion-specific effects on proteins and their biological functions. J. Phys. Chem. B 2017, 121, 1997–2014. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z. Hofmeister effects: An explanation for the impact of ionic liquids on biocatalysis. J. Biot. 2009, 144, 12–22. [Google Scholar] [CrossRef] [PubMed]





| Material | Content of Functional Groups (wt.%) * | Content of CALB (wt.%) * | Content of CRL (wt.%) * | Content of AOL (wt.%) * |
|---|---|---|---|---|
| CNTs-COOH | 1.4 | N/A | N/A | N/A |
| CNTs-CONH (A1) | 5.8 | N/A | N/A | N/A |
| CNTs-CONH-HEX-I (A2) | 6.4 | 2.7 | 7.9 | 5.0 |
| CNTs-CONH-HEX-N(Tf)2 (A3) | 9.1 | 2.8 | 1.1 | 6.7 |
| CNTs-CONH-HEX-N(CN)2 (A4) | 7.0 | 2.3 | 4.7 | 5.3 |
| CNTs-CONH-Oc2PO4 (A5) | 10.9 | 6.4 | 1.2 | 1.9 |
| CNTs-CONH-OcSO4 (A6) | 8.3 | 4.1 | 6.5 | 1.6 |
| CNTs-CHO | 0.7 | N/A | N/A | N/A |
| CNTs-C=N (I1) | 3.2 | N/A | N/A | N/A |
| CNTs-C=N-HEX-I (I2) | 4.3 | 5.9 | 3.4 | 7.8 |
| CNTs-C=N-HEX-N(Tf)2 (I3) | 7.5 | 0.6 | 9.2 | 4.1 |
| CNTs-C=N-HEX-N(CN)2 (I4) | 5.1 | 1.9 | 5.3 | 3.7 |
| CNTs-C=N-Oc2PO4 (I5) | 2.7 | 3.1 | 7.1 | 2.6 |
| CNTs-C=N-OcSO4 (I6) | 2.6 | 2.6 | 6.2 | 5.0 |
| Material | [bmim][N(Tf)2] Content (wt.%) | CALB Content (wt.%) |
|---|---|---|
| CNTs-CONH-[bmim][NTf2] (MR 1:0.1) | 0.4 | 2.8 |
| CNTs-CONH-[bmim][NTf2] (MR 1:0.3) | 1.2 | 4.5 |
| CNTs-CONH-[bmim][NTf2] (MR 1:0.5) | 2.2 | 8.0 |
| CNTs-CONH-[bmim][NTf2] (MR 1:0.8) | 3.2 | 18.6 |
| CNTs-CONH-[bmim][NTf2] (MR 1:1.1) | 3.4 | 19.2 |
| CNTs-C=N-[bmim][NTf2] (MR 1:0.1) | 0.1 | 5.1 |
| CNTs-C=N-[bmim][NTf2] (MR 1:0.3) | 0.7 | 5.7 |
| CNTs-C=N-[bmim][NTf2] (MR 1:0.5) | 1.1 | 6.7 |
| CNTs-C=N-[bmim][NTf2] (MR 1:0.8) | 2.2 | 15.6 |
| CNTs-C=N-[bmim][NTf2] (MR 1:1.1) | 2.8 | 17.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szelwicka, A.; Wolny, A.; Grymel, M.; Jurczyk, S.; Boncel, S.; Chrobok, A. Chemo-Enzymatic Baeyer–Villiger Oxidation Facilitated with Lipases Immobilized in the Supported Ionic Liquid Phase. Materials 2021, 14, 3443. https://doi.org/10.3390/ma14133443
Szelwicka A, Wolny A, Grymel M, Jurczyk S, Boncel S, Chrobok A. Chemo-Enzymatic Baeyer–Villiger Oxidation Facilitated with Lipases Immobilized in the Supported Ionic Liquid Phase. Materials. 2021; 14(13):3443. https://doi.org/10.3390/ma14133443
Chicago/Turabian StyleSzelwicka, Anna, Anna Wolny, Miroslawa Grymel, Sebastian Jurczyk, Slawomir Boncel, and Anna Chrobok. 2021. "Chemo-Enzymatic Baeyer–Villiger Oxidation Facilitated with Lipases Immobilized in the Supported Ionic Liquid Phase" Materials 14, no. 13: 3443. https://doi.org/10.3390/ma14133443
APA StyleSzelwicka, A., Wolny, A., Grymel, M., Jurczyk, S., Boncel, S., & Chrobok, A. (2021). Chemo-Enzymatic Baeyer–Villiger Oxidation Facilitated with Lipases Immobilized in the Supported Ionic Liquid Phase. Materials, 14(13), 3443. https://doi.org/10.3390/ma14133443







