Commercial Bone Grafts Claimed as an Alternative to Autografts: Current Trends for Clinical Applications in Orthopaedics
Abstract
1. Introduction
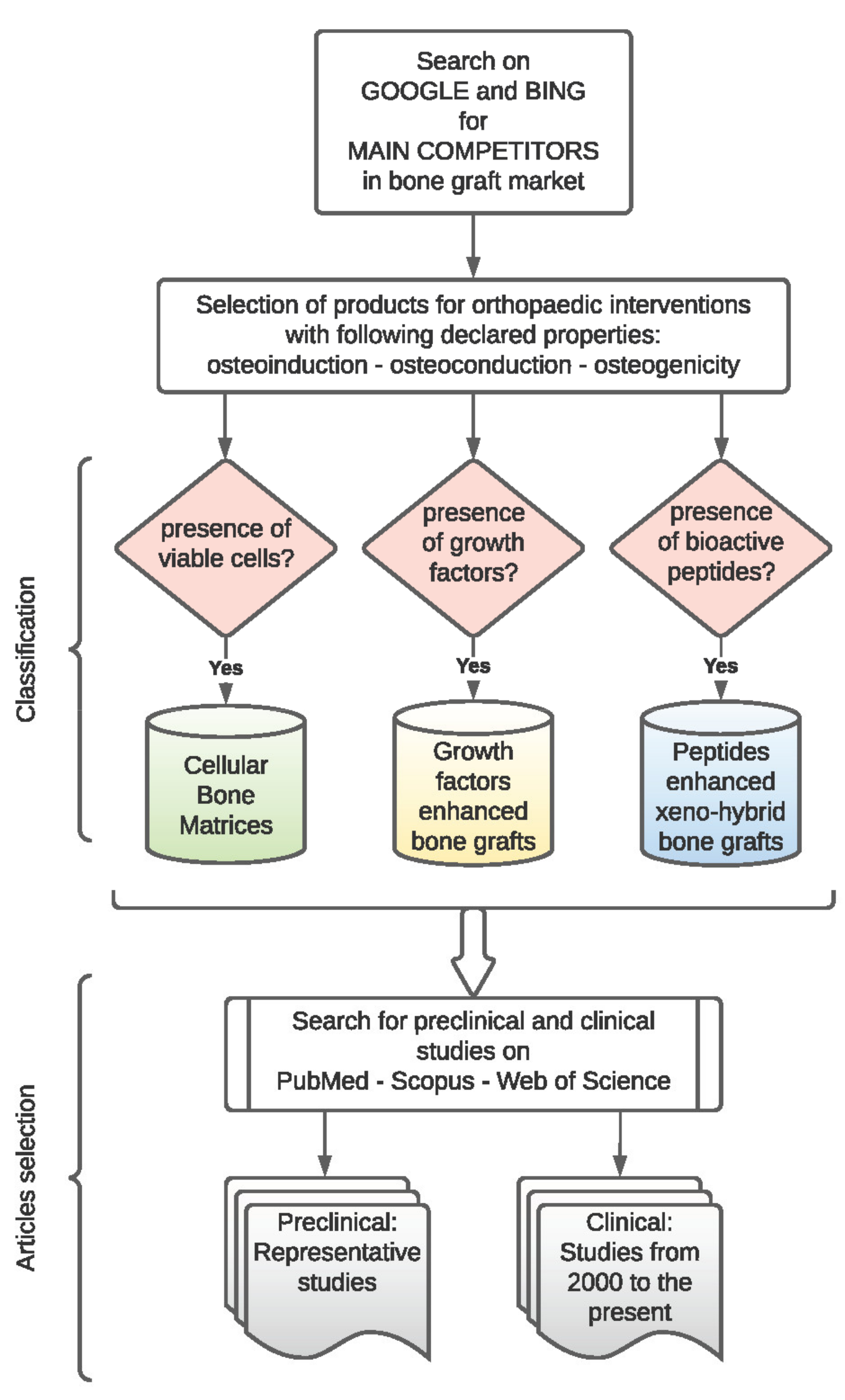
2. Materials and Methods
3. Results
3.1. Cellular Bone Matrices
3.1.1. Cell Type, Cell Amount, and Post-Thaw Cell Viability
3.1.2. Bone Tissue Processing, Components, and Formulations
3.1.3. Bone Cryoprotectant Agents
3.2. Growth Factor Enhanced Bone Grafts
3.3. Peptide Enhanced Xeno-Hybrid Bone Grafts
4. Discussion
| Commercial Name/Manufacturer | Cell Type/Amount/Viability Post-Thaw | Composition | Formulations | Cryoprotectant Agent | Clinical Indications | Clinical Studies [Ref.] or NCT |
|---|---|---|---|---|---|---|
| Osteocel® Plus Osteocel® PRO Nuvasive, San Diego, CA, USA | MSCs and osteoprogenitor cells. >250,000 cells/cc > 70% | Cryopreserved viable cancellous matrix. Ground demineralised bone matrix. | Particulate Putty | DMSO | Spine, orthopaedics, oral and maxillofacial applications. | [46,47,48,49,50,51] |
| Trinity EVOLUTION® Trinity ELITE® Orthofix Medical Inc., Lewisville, TX, USA | MSCs and osteoprogenitor cells. >750,000 cells/cc ≥70% | Cancellous bone. Demineralised bone particulates or Cancellous bone. Demineralised bone fibres. | Putty | DMSO | Treatment of musculoskeletal defects. | [52,53,54,55] |
| Via® Vivex Biologics Inc., Miami, FL, USA | Bone-derived cells. >150,000 cells/cc >80% | 100–300 µM demineralised cortical bone. Mineralised cortical and cancellous bone (Via® Graft). Cortical shavings, crushed cancellous chips (Via® Form). | Particulate Paste | ViaCoat™ DMSO-free cryoprotectant | Spine, upper extremity, foot and ankle, oral and maxillofacial, and orthopedic oncology. | [56] |
| ViviGen® ViviGen®Formable DePuy Synthes, Raynham, MA, USA | Osteoblasts, osteocytes, and bone lining cells. >16,000 cells/cc ≥96% | Corticocancellous chips. Demineralised bone particulate or fibres. | Particulate Putty | Proprietary cryopreservation medium | Fusion, non-union, and malunion for foot/ankle, long bone, and craniomaxillofacial trauma and reconstruction in patients with compromised biology. | [57,58,59,60] |
| BIO4™ Stryker, Kalamazoo, MI, USA | MSCs osteoprogenitor and osteoblasts. >600,000 cells/cc >70% | Native matrix. Endogenous Osteoinductive and angiogenic growth factors. | Putty | Minimal amount of a proprietary cryopreservation medium | Treatment of musculoskeletal defects. | NCT03077204 |
| PrimaGen® Zimmer Biomet, Warsaw, IN, USA | MSCs, osteoprogenitorcells, pre-osteoblasts >750,000 cells/cc >70% | Cancellous bone. Demineralised cortical bone. | Putty | N/A | Treatment of musculoskeletal defects. | NCT02182843 |
| Map3® RTI Surgical, Alachua, FL, USA | MAPC-class cells. >50,000 cells/cc n.a. | Cortical cancellous bone chips. Demineralised bone matrix. | Strips Chips | N/A | Small joint repair, filling bone defects. | n.a. |
| V92™ V92-FC ™ 28, Englewood, CO, USA | Bone-derived cells. >150,000 cells/cc >80% | Cortical cancellous bone particulate. Demineralisedbone matrix. Bone gel (only in V92-FC™) | Microparticulate Paste | DMSO-free cryoprotectant | Orthopaedic and spine applications. | n.a. |
| SCYLLA™ SCYLLA™-F Chamber Spine. King of Prussia, PA, USA | Bone-derived cells. >150,000 cells/cc >80% | Cortical cancellous bone particulate. DBM and bone mixture gel (only in SCYLLA™-F). | Microparticulate Paste | DMSO-free cryoprotectant | Treatment of musculoskeletal defects. | n.a. |
| Magnus Royal Biologics, Hackensack, NJ, USA | Cell population with MSC and pluripotent cell markers >150,000 cells/cc >80% | Cortical shavings, crushed cancellous chips, and demineralised cortical bone microparticulate scaffold blend with bone gel mixture. | Paste | DMSO-free cryoprotectant | Fusion, midfoot arthrodesis. | n.a. |
| CeLLogix Omnia Medical, Morgantown, WV, USA | Bone-derived cells. >150,000 cells/cc >80% | Cortical–cancellous bone particulate. DBM | Microparticulate | DMSO-free cryoprotectant | Treatment of musculoskeletal defects. | n.a. |
| Manufacturer | Commercial Name | Active Molecule | Carrier | Formulations | Clinical Indications | Clinical Studies [Ref.] or NCT |
|---|---|---|---|---|---|---|
| Medtronic Spinal and Biologics, Memphis, TN, USA | INFUSE® | rhBMP-2 | ACS | Vial + sponge | Spinal fusion procedures. Treatment of open tibial fractures with an intramedullary (IM) nail fixation. Sinus floor and alveolar ridge augmentations. | [66,67,68,69,70,71,72,73,74,75,76,86] |
| Lynch Biologics, Franklin, TN, USA | Gem 21S® | rhPDGF-BB | β-TCP | Vial + granules | Periodontal defects. | n.a. |
| Wright Medical Group N.V., Memphis, TN, USA | Augment® | rhPDGF-BB | β-TCP | Vial + granules | Arthrodesis (i.e., fusion procedures) of the ankle and/or hindfoot. | [81,82,84] |
| Bioventus LLC, Durham, NC, USA | OsteoAMP® | AMP | n.a. | Granules, putty, or sponge | Cervical/lumbar spine fusion. | [86,87] NCT02225444 |
| Manufacturer | Commercial Name | Peptide | Composition | Formulations | Clinical Indications | Clinical Studies [Ref.] or NCT |
|---|---|---|---|---|---|---|
| IBI, Mezzo-Vico Vira, Switzerland | SmartBone® | RGD | Bovine bone matrix. Micrometric thin poly(l-lactic-co-ε-caprolactone). | Microchips Blocks, plates, wedges | Dental and orthopaedic indications. | [89,90,91,92,93,94] |
| Cerapedics, Westminster, CO, USA | i-FACTOR® | P-15 | Anorganic bovine bone matrix. Inert biocompatible hydrogel. | Putty Strips (not in the USA) | Bone filling defects in the spine and extremities. | [101,102,103,104,105,106] |
| Manufacturer | Commercial Name | FDA Injunction | Status |
|---|---|---|---|
| Osiris Therapeutics, Columbia, MD, USA | Ovation® | The manufacturing process alters the original relevant characteristics of the tissue. The product is dependent upon the metabolic activity of living cells for their primary function and is not intended for autologous use or allogeneic use in a first- or second-degree relative [45]. | Withdrawn. It was transitioned to Ovation OS and currently available as BIO4™ (distributed by Stryker). |
| RTI Surgical, Alachua, FL, USA | Map3® | The processing does not meet the definition of minimal manipulation for cells or nonstructural tissues [109]. | Withdrawn. |
| MedtronicSpinal and Biologics, Memphis, TN, USA | INFUSE® | FDA warning was issued not to use in the anterior cervical spine due to inflammation causing severe dysphagia and a potential increase in mortality [73]. | Available for use in interbody spine fusion, fresh tibial fractures, and oral maxillofacial bone grafting procedures. |
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Majidinia, M.; Sadeghpour, A.; Yousefi, B. The roles of signaling pathways in bone repair and regeneration. J. Cell Physiol. 2018, 233, 2937–2948. [Google Scholar] [CrossRef] [PubMed]
- Wiese, A.; Pape, H.C. Bone Defects Caused by High-energy Injuries, Bone Loss, Infected Nonunions, and Nonunions. Orthop. Clin. N. Am. 2010, 41, 1–4. [Google Scholar] [CrossRef]
- Kacarevic, Z.P.; Rider, P.; Alkildani, S.; Retnasingh, S.; Pejakić, M.; Schnettler, R.; Gosau, M.; Smeets, R.; Jung, O.; Barbeck, M. An introduction to bone tissue engineering. Int. J. Artif. Organs 2020, 43, 69–86. [Google Scholar] [CrossRef] [PubMed]
- Wubneh, A.; Tsekoura, E.K.; Ayranci, C.; Uludağ, H. Current state of fabrication technologies and materials for bone tissue engineering. Acta Biomater. 2018, 80, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Yeung, K.W. Bone grafts and biomaterials substitutes for bone defect repair: A review. Bioact. Mater. 2017, 2, 224–247. [Google Scholar] [CrossRef] [PubMed]
- Graziani, G.; Govoni, M.; Vivarelli, L.; Boi, M.; de Carolis, M.; Bianchi, M.; Sassoni, E.; Bignozzi, M.C.; Carnevale, G.; Marmi, F.; et al. A Comprehensive Microstructural and Compositional Characterization of Allogenic and Xenogenic Bone: Application to Bone Grafts and Nanostructured Biomimetic Coatings. Coatings 2020, 10, 522. [Google Scholar] [CrossRef]
- Einhorn, T.A.; Gerstenfeld, L.C. Fracture healing: Mechanisms and interventions. Nat. Rev. Rheumatol. 2015, 11, 45–54. [Google Scholar] [CrossRef]
- Grageda, E. Platelet-Rich Plasma and Bone Graft Materials: A Review and a Standardized Research Protocol. Implant Dent. 2004, 13, 301–309. [Google Scholar] [CrossRef]
- Falacho, R.; Palma, P.; Marques, J.; Figueiredo, M.; Caramelo, F.; Dias, I.; Viegas, C.; Guerra, F. Collagenated Porcine Heterologous Bone Grafts: Histomorphometric Evaluation of Bone Formation Using Different Physical Forms in a Rabbit Cancellous Bone Model. Molecules 2021, 26, 1339. [Google Scholar] [CrossRef]
- Kumar, Y.C.; Nalini, K.B.; Menon, J.; Patro, D.K.; Banerji, B.H. Calcium sulfate as bone graft substitute in the treatment of osseous bone defects, a prospective study. J. Clin. Diagn. Res. 2013, 7, 2926–2928. [Google Scholar] [CrossRef]
- Bohner, M.; Santoni, B.L.G.; Dobelin, N. β-tricalcium phosphate for bone substitution: Synthesis and properties. Acta Biomater 2020, 113, 23–41. [Google Scholar] [CrossRef]
- Välimäki, V.-V.; Aro, H.T. Molecular basis for action of bioactive glasses as bone graft substitute. Scand. J. Surg. 2006, 95, 95–102. [Google Scholar] [CrossRef]
- Aslankoohi, N.; Mondal, D.; Rizkalla, A.S.; Mequanint, K. Bone Repair and Regenerative Biomaterials: Towards Recapitulating the Microenvironment. Polymers 2019, 11, 1437. [Google Scholar] [CrossRef]
- Palma, P.J.; Ramos, J.; Martins, J.B.; Diogenes, A.; Figueiredo, M.H.; Ferreira, P.; Viegas, C.; Santos, J.M. Histologic Evaluation of Regenerative Endodontic Procedures with the Use of Chitosan Scaffolds in Immature Dog Teeth with Apical Periodontitis. J. Endod. 2017, 43, 1279–1287. [Google Scholar] [CrossRef] [PubMed]
- Evans, C.H. Barriers to the Clinical Translation of Orthopedic Tissue Engineering. Tissue Eng. Part B Rev. 2011, 17, 437–441. [Google Scholar] [CrossRef] [PubMed]
- Skovrlj, B.; Guzman, J.Z.; Al Maaieh, M.; Cho, S.K.; Iatridis, J.C.; Qureshi, S.A. Cellular bone matrices: Viable stem cell-containing bone graft substitutes. Spine J. 2014, 14, 2763–2772. [Google Scholar] [CrossRef] [PubMed]
- Abedi, A.; Formanek, B.; Russell, N.; Vizesi, F.; Boden, S.D.; Wang, J.C.; Buser, Z. Examination of the role of cells in commercially available cellular allografts in spine fusion: An In Vivo animal study. J. Bone Jt. Surg. Am. 2020, 102, e135. [Google Scholar] [CrossRef]
- Martinez, S.A.; Walker, T. Bone Grafts. Veter Clin. North Am. Small Anim. Pr. 1999, 29, 1207–1219. [Google Scholar] [CrossRef]
- Zhu, H.; Blahnová, V.H.; Perale, G.; Xiao, J.; Betge, F.; Boniolo, F.; Filová, E.; Lyngstadaas, S.P.; Haugen, H.J. Xeno-Hybrid Bone Graft Releasing Biomimetic Proteins Promotes Osteogenic Differentiation of hMSCs. Front. Cell Dev. Biol. 2020, 8, 619111. [Google Scholar] [CrossRef]
- Wright, J.G.; Swiontkowski, M.F.; Heckman, J.D. Introducing Levels of Evidence to the Journal. J. Bone Jt. Surg. Am. 2003, 85, 1–3. [Google Scholar] [CrossRef]
- Méndez-Ferrer, S.; Michurina, T.V.; Ferraro, F.; Mazloom, A.R.; MacArthur, B.D.; Lira, S.A.; Scadden, D.T.; Ma’Ayan, A.; Enikolopov, G.N.; Frenette, P.S. Mesenchymal and haematopoietic stem cells form a unique bone marrow niche. Nature 2010, 466, 829–834. [Google Scholar] [CrossRef] [PubMed]
- Khan, R.S.; Newsome, P.N. A Comparison of Phenotypic and Functional Properties of Mesenchymal Stromal Cells and Multipotent Adult Progenitor Cells. Front. Immunol. 2019, 10, 1952. [Google Scholar] [CrossRef] [PubMed]
- Tolar, J.; O’Shaughnessy, M.J.; Panoskaltsis-Mortari, A.; McElmurry, R.T.; Bell, S.; Riddle, M.; McIvor, R.S.; Yant, S.R.; Kay, M.A.; Krause, D.; et al. Host factors that impact the biodistribution and persistence of multipotent adult progenitor cells. Blood 2006, 107, 4182–4188. [Google Scholar] [CrossRef]
- Ankrum, J.; Ong, J.F.; Karp, J.M. Mesenchymal stem cells: Immune evasive, not immune privileged. Nat. Biotechnol. 2014, 32, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Corradetti, B.; Taraballi, F.; Powell, S.; Sung, D.; Minardi, S.; Ferrari, M.; Weiner, B.K.; Tasciotti, E. Osteoprogenitor Cells from Bone Marrow and Cortical Bone: Understanding How the Environment Affects Their Fate. Stem Cells Dev. 2015, 24, 1112–1123. [Google Scholar] [CrossRef]
- Franz-Odendaal, T.A.; Hall, B.K.; Witten, P.E. Buried alive: How osteoblasts become osteocytes. Dev. Dyn. 2006, 235, 176–190. [Google Scholar] [CrossRef]
- Yu, H.; Shen, G.; Wei, F. Effect of Cryopreservation on the Immunogenicity of Osteoblasts. Transplant. Proc. 2007, 39, 3030–3031. [Google Scholar] [CrossRef]
- Cornell, C.N.; Lane, J.M. Current Understanding of Osteoconduction in Bone Regeneration. Clin. Orthop. Relat. Res. 1998, 355, S267–S273. [Google Scholar] [CrossRef]
- Gruskin, E.; Doll, B.A.; Futrell, F.W.; Schmitz, J.P.; Hollinger, J.O. Demineralized bone matrix in bone repair: History and use. Adv. Drug Deliv. Rev. 2012, 64, 1063–1077. [Google Scholar] [CrossRef]
- Asghar, W.; El Assal, R.; Shafiee, H.; Anchan, R.M.; Demirci, U. Preserving human cells for regenerative, reproductive, and transfusion medicine. Biotechnol. J. 2014, 9, 895–903. [Google Scholar] [CrossRef]
- Best, B.P. Cryoprotectant Toxicity: Facts, Issues, and Questions. Rejuvenation Res. 2015, 18, 422–436. [Google Scholar] [CrossRef] [PubMed]
- Notman, R.; Noro, M.; O’Malley, B.; Anwar, J. Molecular Basis for Dimethylsulfoxide (DMSO) Action on Lipid Membranes. J. Am. Chem. Soc. 2006, 128, 13982–13983. [Google Scholar] [CrossRef] [PubMed]
- Hsu, E.L.; Stock, S.R. Growth Factors, Carrier Materials, and Bone Repair. Bone Regul. Osteoporos. Ther. 2020, 262, 121–156. [Google Scholar]
- Siddiqui, J.A.; Partridge, N.C. Physiological Bone Remodeling: Systemic Regulation and Growth Factor Involvement. Physiology 2016, 31, 233–245. [Google Scholar] [CrossRef] [PubMed]
- Nickel, J.; Mueller, T.D. Specification of BMP Signaling. Cells 2019, 8, 1579. [Google Scholar] [CrossRef]
- Chaudhary, L.; Hofmeister, A.; Hruska, K. Differential growth factor control of bone formation through osteoprogenitor differentiation. Bone 2004, 34, 402–411. [Google Scholar] [CrossRef]
- Rossi, F.; Santoro, M.; Perale, G. Polymeric scaffolds as stem cell carriers in bone repair. J. Tissue Eng. Regen. Med. 2015, 9, 1093–1119. [Google Scholar] [CrossRef]
- Bellis, S.L. Advantages of RGD peptides for directing cell association with biomaterials. Biomaterials 2011, 32, 4205–4210. [Google Scholar] [CrossRef] [PubMed]
- Bhatnagar, R.S.; Qian, J.J.; Wedrychowska, A.; Sadeghi, M.; Wu, Y.M.; Smith, N. Design of Biomimetic Habitats for Tissue Engineering with P-15, a Synthetic Peptide Analogue of Collagen. Tissue Eng. 1999, 5, 53–65. [Google Scholar] [CrossRef] [PubMed]
- De Grado, G.F.; Keller, L.; Idoux-Gillet, Y.; Wagner, Q.; Musset, A.-M.; Benkirane-Jessel, N.; Bornert, F.; Offner, D. Bone substitutes: A review of their characteristics, clinical use, and perspectives for large bone defects management. J. Tissue Eng. 2018, 9. [Google Scholar] [CrossRef]
- Campana, V.; Milano, G.; Pagano, E.D.; Barba, M.; Cicione, C.; Salonna, G.; Lattanzi, W.; Logroscino, G. Bone substitutes in orthopaedic surgery: From basic science to clinical practice. J. Mater. Sci. Mater. Med. 2014, 25, 2445–2461. [Google Scholar] [CrossRef] [PubMed]
- Abjornson, C.; Brecevich, A.; Callanan, T.; Dowe, C.; Cammisa, F.P.; Lorio, M.P. ISASS Recommendations and Coverage Criteria for Bone Graft Substitutes used in Spinal Surgery. Int. J. Spine Surg. 2018, 12, 757–771. [Google Scholar] [CrossRef] [PubMed]
- Venuti, N.; Mohan, V. Spinal fusion with mesenchymal stem cell bone matrices: A review on product comparison. Spine Res. 2019, 5, 1–9. [Google Scholar]
- FDA. Regulatory Considerations for Human Cells, Tissues, and Cellular and Tissue-Based Products: Minimal Manipulation and Homologous Use. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/regulatory-considerations-human-cells-tissues-and-cellular-and-tissue-based-products-minimal (accessed on 7 June 2021).
- FDA. Osiris Therapeutics, Inc.—Untitled letter. Available online: https://www.fda.gov/vaccines-blood-biologics/enforcement-actions-cber/osiris-therapeutics-inc-untitled-letter (accessed on 7 June 2021).
- Hollawell, S.M. Allograft Cellular Bone Matrix as an Alternative to Autograft in Hindfoot and Ankle Fusion Procedures. J. Foot Ankle Surg. 2012, 51, 222–225. [Google Scholar] [CrossRef] [PubMed]
- Eastlack, R.K.; Garfin, S.R.; Brown, C.R.; Meyer, S.C. Osteocel Plus cellular allograft in anterior cervical discectomy and fusion: Evaluation of clinical and radiographic outcomes from a prospective multicenter study. Spine (Phila Pa 1976) 2014, 39, E1331–E1337. [Google Scholar] [CrossRef] [PubMed]
- Ammerman, J.M.; Libricz, J.; Ammerman, M.D. The role of Osteocel Plus as a fusion substrate in minimally invasive instrumented transforaminal lumbar interbody fusion. Clin. Neurol. Neurosurg. 2013, 115, 991–994. [Google Scholar] [CrossRef]
- Kerr, E.J., III; Jawahar, A.; Wooten, T.; Kay, S.; Cavanaugh, D.A.; Nunley, P.D. The use of osteo-conductive stem-cells allograft in lumbar interbody fusion procedures: An alternative to recombinant human bone morphogenetic protein. J. Surg. Orthop. Adv. 2011, 20, 193–197. [Google Scholar]
- Tohmeh, A.G.; Watson, B.; Tohmeh, M.; Zielinski, X.J. Allograft Cellular Bone Matrix in Extreme Lateral Interbody Fusion: Preliminary Radiographic and Clinical Outcomes. Sci. World J. 2012, 2012, 1–8. [Google Scholar] [CrossRef]
- Whitt, J.; Al-Sabbagh, M.; Dawson, D.; Shehata, E.; Housley-Smith, M.; Tezanos, A.; Kutkut, A. Efficacy of stem cell allograft in maxillary sinus bone regeneration: A randomized controlled clinical and blinded histomorphometric study. Int. J. Implant Dent. 2020, 6, 25. [Google Scholar] [CrossRef]
- Peppers, T.A.; Bullard, D.E.; Vanichkachorn, J.S.; Stanley, S.K.; Arnold, P.M.; Waldorff, E.I.; Hahn, R.; Atkinson, B.L.; Ryaby, J.T.; Linovitz, R.J. Prospective clinical and radiographic evaluation of an allogeneic bone matrix containing stem cells (Trinity Evolution® Viable Cellular Bone Matrix) in patients undergoing two-level anterior cervical discectomy and fusion. J. Orthop. Surg. Res. 2017, 12, 67. [Google Scholar] [CrossRef]
- Vanichkachorn, J.; Peppers, T.; Bullard, D.; Stanley, S.K.; Linovitz, R.J.; Ryaby, J.T. A prospective clinical and radiographic 12-month outcome study of patients undergoing single-level anterior cervical discectomy and fusion for symptomatic cervical degenerative disc disease utilizing a novel viable allogeneic, cancellous, bone matrix (trinity evolution) with a comparison to historical controls. Eur. Spine J. 2016, 25, 2233–2238. [Google Scholar] [PubMed]
- Musante, D.B.; Firtha, M.E.; Atkinson, B.L.; Hahn, R.; Ryaby, J.T.; Linovitz, R.J. Clinical evaluation of an allogeneic bone matrix containing viable osteogenic cells in patients undergoing one- and two-level posterolateral lumbar arthrodesis with decompressive laminectomy. J. Orthop. Surg. Res. 2016, 11, 63. [Google Scholar] [CrossRef] [PubMed]
- Loveland, J.; Waldorff, E.; He, D.; Atkinson, B.L. A retrospective clinical comparison of two allogeneic bone matrices containing viable osteogenic cells in patients undergoing foot and/or ankle arthrodesis. J. Stem Cell Res. Ther. 2017, 7, 1–7. [Google Scholar] [CrossRef]
- Tally, W.C.; Temple, H.T.; Subhawong, T.Y.; Ganey, T. Transforaminal lumbar interbody fusion with viable allograft: 75 consecutive cases at 12-month follow-up. Int. J. Spine Surg. 2018, 12, 76–84. [Google Scholar] [PubMed]
- Divi, S.N.; Mikhael, M.M. Use of allogenic mesenchymal cellular bone matrix in anterior and posterior cervical spinal fusion: A case series of 21 patients. Asian Spine J. 2017, 11, 454–462. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.F.; McLean, J.B.; Jones, S.M.; Moore, M.A.; Nicholson, M.D.; Dorsch, K.A. Multilevel instrumented posterolateral lumbar spine fusion with an allogeneic cellular bone graft. J. Orthop. Surg. Res. 2019, 14, 372. [Google Scholar] [CrossRef] [PubMed]
- Shahrdar, C.; McLean, J.; Gianulis, E.; Softic, D.; Qin, X.; Moore, M.A.; Chen, J. Clinical outcome and explant histology after using a cellular bone allograft in two-stage total hip arthroplasty. J. Orthop. Surg. Res. 2020, 15, 16. [Google Scholar] [CrossRef]
- Moran, T.E.; Sequeira, S.; Cooper, M.T.; Park, J. A retrospective analysis of outcomes from foot and ankle arthrodesis and open reduction and internal fixation using cellular bone allograft augmentation. Foot Ankle Spec. 2020. [Google Scholar] [CrossRef]
- Tortelli, F.; Tasso, R.; Loiacono, F.; Cancedda, R. The development of tissue-engineered bone of different origin through endochondral and intramembranous ossification following the implantation of mesenchymal stem cells and osteoblasts in a murine model. Biomaterials 2010, 31, 242–249. [Google Scholar] [CrossRef]
- Nakasaki, M.; Yoshioka, K.; Miyamoto, Y.; Sasaki, T.; Yoshikawa, H.; Itoh, K. IGF-I secreted by osteoblasts acts as a potent chemotactic factor for osteoblasts. Bone 2008, 43, 869–879. [Google Scholar] [CrossRef]
- Lin, C.; Zhang, N.; Waldorff, E.I.; Punsalan, P.; Wang, D.; Semler, E.; Ryaby, J.T.; Yoo, J.; Johnstone, B. Comparing cellular bone matrices for posterolateral spinal fusion in a rat model. JOR Spine 2020, 3, e1084. [Google Scholar] [CrossRef] [PubMed]
- Lord, E.L.; Cohen, J.R.; Buser, Z.; Meisel, H.J.; Brodke, D.S.; Yoon, S.T.; Youssef, J.A.; Wang, J.C.; Park, J.B. Trends, costs, and complications of anterior cervical discectomy and fusion with and without bone morphogenetic protein in the united states medicare population. Global Spine J. 2017, 7, 603–608. [Google Scholar] [CrossRef]
- McKay, B.; Sandhu, H.S. Use of recombinant human bone morphogenetic protein-2 in spinal fusion applications. Spine 2002, 27, S66–S85. [Google Scholar] [CrossRef] [PubMed]
- Boden, S.D.; Zdeblick, T.A.; Sandhu, H.S.; Heim, S.E. The use of rhBMP-2 in interbody fusion cages. Definitive evidence of osteoinduction in humans: A preliminary report. Spine 2000, 25, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Govender, S.; Csimma, C.; Genant, H.K.; Valentin-Opran, A.; Amit, Y.; Arbel, R.; Aro, H.; Atar, D.; Bishay, M.; Borner, M.G.; et al. Recombinant human bone morphogenetic protein-2 for treatment of open tibial fractures: A prospective, controlled, randomized study of four hundred and fifty patients. J. Bone Jt. Surg. Am. 2002, 84, 2123–2134. [Google Scholar] [CrossRef] [PubMed]
- Cahill, K.S.; Chi, J.H.; Day, A.; Claus, E.B. Prevalence, complications, and hospital charges associated with use of bone-morphogenetic proteins in spinal fusion procedures. JAMA 2009, 302, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Baskin, D.S.; Ryan, P.; Sonntag, V.; Westmark, R.; Widmayer, M.A. A prospective, randomized, controlled cervical fusion study using recombinant human bone morphogenetic protein-2 with the CORNERSTONE-SRTM allograft ring and the ATLANTISTM anterior cervical plate. Spine 2003, 28, 1219–1224. [Google Scholar] [CrossRef] [PubMed]
- Burkus, J.K.; Heim, S.E.; Gornet, M.F.; Zdeblick, T.A. Is INFUSE bone graft superior to autograft bone? An integrated analysis of clinical trials using the LT-CAGE lumbar tapered fusion device. J. Spinal Disord. Tech. 2003, 16, 113–122. [Google Scholar] [CrossRef]
- Haid, R.W., Jr.; Branch, C.L., Jr.; Alexander, J.T.; Burkus, J.K. Posterior lumbar interbody fusion using recombinant human bone morphogenetic protein type 2 with cylindrical interbody cages. Spine J. 2004, 4, 527–538. [Google Scholar] [CrossRef]
- Lykissas, M.; Gkiatas, I. Use of recombinant human bone morphogenetic protein-2 in spine surgery. World J. Orthop. 2017, 8, 531–535. [Google Scholar] [CrossRef]
- Hustedt, J.W.; Blizzard, D.J. The controversy surrounding bone morphogenetic proteins in the spine: A review of current research. Yale J. Biol. Med. 2014, 87, 549–561. [Google Scholar]
- Medtronic Announces New Clinical Trial to Study Infuse Bone Graft in TLIF Spine Procedures. Available online: https://newsroom.medtronic.com/news-releases/news-release-details/medtronic-announces-new-clinical-trial-study-infuse-bone-graft/ (accessed on 27 April 2021).
- James, A.W.; LaChaud, G.; Shen, J.; Asatrian, G.; Nguyen, V.; Zhang, X.; Ting, K.; Soo, C. A review of the clinical side effects of bone morphogenetic protein-2. Tissue Eng. Part B Rev. 2016, 22, 284–297. [Google Scholar] [CrossRef]
- Liu, S.; Wang, Y.; Liang, Z.; Zhou, M.; Chen, C. Comparative clinical effectiveness and safety of bone morphogenetic protein versus autologous iliac crest bone graft in lumbar fusion: A meta-analysis and systematic review. Spine 2020, 45, E729–E741. [Google Scholar] [CrossRef] [PubMed]
- Al-Zube, L.; Breitbart, E.A.; O’Connor, J.P.; Parsons, J.R.; Bradica, G.; Hart, C.E.; Lin, S.S. Recombinant human platelet-derived growth factor BB (rhPDGF-BB) and β-tricalcium phosphate/collagen matrix enhance fracture healing in a diabetic rat model. J. Orthop. Res. 2009, 27, 1074–1081. [Google Scholar] [CrossRef]
- Hollinger, J.O.; Onikepe, A.O.; MacKrell, J.; Einhorn, T.; Bradica, G.; Lynch, S.; Hart, C.E. Accelerated fracture healing in the geriatric, osteoporotic rat with recombinant human platelet-derived growth factor-BB and an injectable β-tricalcium phosphate/collagen matrix. J. Orthop. Res. 2008, 26, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Ojima, Y.; Mizuno, M.; Kuboki, Y.; Komori, T. In Vitro effect of platelet-derived growth factor-BB on collagen synthesis and proliferation of human periodontal ligament cells. Oral Dis. 2003, 9, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Vikjaer, D.; Blom, S.; Hjorting-Hansen, E.; Pinholt, E.M. Effect of platelet-derived growth factor-BB on bone formation in calvarial defects: An experimental study in rabbits. Eur. J. Oral Sci. 1997, 105, 59–66. [Google Scholar] [CrossRef]
- Daniels, T.R.; Younger, A.S.; Penner, M.J.; Wing, K.J.; Le, I.L.; Russell, I.S.; Lalonde, K.A.; Evangelista, P.T.; Quiton, J.D.; Glazebrook, M.; et al. Prospective randomized controlled trial of hindfoot and ankle fusions treated with rhPDGF-BB in combination with a β-TCP-collagen matrix. Foot Ankle Int. 2015, 36, 739–748. [Google Scholar] [CrossRef]
- Di Giovanni, C.W.; Lin, S.S.; Baumhauer, J.F.; Daniels, T.; Younger, A.; Glazebrook, M.; Anderson, J.; Anderson, R.; Evangelista, P.; Lynch, S.E.; et al. Recombinant human platelet-derived growth factor-BB and β-tricalcium phosphate (rhPDGF-BB/β-TCP): An alternative to autogenous bone graft. J. Bone Jt. Surg. Am. 2013, 95, 1184–1192. [Google Scholar] [CrossRef]
- Nevins, M.; Camelo, M.; Nevins, M.L.; Schenk, R.K.; Lynch, S.E. Periodontal regeneration in humans using recombinant human platelet-derived growth factor-BB (rhPDGF-BB) and allogenic bone. J. Periodontol. 2003, 74, 1282–1292. [Google Scholar] [CrossRef] [PubMed]
- Solchaga, L.A.; Hee, C.K.; Roach, S.; Snel, L.B. Safety of recombinant human platelet-derived growth factor-BB in Augment® Bone Graft. J. Tissue Eng. 2012, 3. [Google Scholar] [CrossRef]
- Baldo, B.A. Side effects of cytokines approved for therapy. Drug Saf. 2014, 37, 921–943. [Google Scholar] [CrossRef] [PubMed]
- Roh, J.S.; Yeung, C.A.; Field, J.S.; McClellan, R.T. Allogeneic morphogenetic protein vs. recombinant human bone morphogenetic protein-2 in lumbar interbody fusion procedures: A radiographic and economic analysis. J. Orthop. Surg. Res. 2013, 8, 49. [Google Scholar] [CrossRef]
- Yeung, C.; Field, J.; Roh, J. Clinical validation of allogeneic morphogenetic protein: Donor intervariability, terminal irradiation and age of product is not clinically relevant. J. Spine 2013, 3, 1–7. [Google Scholar]
- Bullock, G.; Atkinson, J.; Gentile, P.; Hatton, P.; Miller, C. Osteogenic peptides and attachment methods determine tissue regeneration in modified bone graft substitutes. J. Funct. Biomater. 2021, 12, 22. [Google Scholar] [CrossRef] [PubMed]
- D’Alessandro, D.; Perale, G.; Milazzo, M.; Moscato, S.; Stefanini, C.; Pertici, G.; Danti, S. Bovine bone matrix/poly(l-lactic-co-epsilon-caprolactone)/gelatin hybrid scaffold (SmartBone®) for maxillary sinus augmentation: A histologic study on bone regeneration. Int. J. Pharm. 2017, 523, 534–544. [Google Scholar] [CrossRef]
- Grecchi, F.; Perale, G.; Candotto, V.; Busato, A.; Pascali, M.; Carinci, F. Reconstruction of the zygomatic bone with SmartBone®: Case report. J. Biol. Regul. Homeost. Agents 2015, 29, 42–47. [Google Scholar] [PubMed]
- Spinato, S.; Galindo-Moreno, P.; Bernardello, F.; Zaffe, D. Minimum abutment height to eliminate bone loss: Influence of implant neck design and platform switching. Int. J. Oral Maxillofac. Implant. 2018, 33, 405–411. [Google Scholar] [CrossRef]
- Facciuto, E.; Grottoli, C.F.; Mattarocci, M.; Illiano, F.; Compagno, M.; Ferracini, R.; Perale, G. Three-dimensional craniofacial bone reconstruction with SmartBone on demand. J. Craniofac. Surg. 2019, 30, 739–741. [Google Scholar] [CrossRef]
- Boffano, M.; Ratto, N.; Conti, A.; Pellegrino, P.; Rossi, L.; Perale, G.; Piana, R. A preliminary study on the mechanical reliability and regeneration capability of artificial bone grafts in oncologic cases, with and without osteosynthesis. J. Clin. Med. 2020, 9, 1388. [Google Scholar] [CrossRef]
- Ferracini, R.; Bistolfi, A.; Garibaldi, R.; Furfaro, V.; Battista, A.; Perale, G. Composite xenohybrid bovine bone-derived scaffold as bone substitute for the treatment of tibial plateau fractures. Appl. Sci. 2019, 9, 2675. [Google Scholar] [CrossRef]
- Hestehave Pedersen, R.; Rasmussen, M.; Overgaard, S.; Ding, M. Effects of P-15 peptide coated hydroxyapatite on tibial defect repair In Vivo in normal and osteoporotic rats. Biomed. Res. Int. 2015, 2015, 253858. [Google Scholar] [CrossRef]
- Sherman, B.P.; Lindley, E.M.; Turner, A.S.; Seim, H.B., III; Benedict, J.; Burger, E.L.; Patel, V.V. Evaluation of ABM/P-15 versus autogenous bone in an ovine lumbar interbody fusion model. Eur. Spine J. 2010, 19, 2156–2163. [Google Scholar] [CrossRef] [PubMed]
- Thorwarth, M.; Schultze-Mosgau, S.; Wehrhan, F.; Srour, S.; Wiltfang, J.; Neukam, F.W.; Schlegel, K.A. Enhanced bone regeneration with a synthetic cell-binding peptide-In Vivo results. Biochem. Biophys. Res. Commun. 2005, 329, 789–795. [Google Scholar] [CrossRef]
- Vastardis, S.; Yukna, R.A.; Mayer, E.T.; Atkinson, B.L. Periodontal regeneration with peptide-enhanced anorganic bone matrix in particulate and putty form in dogs. J. Periodontol. 2005, 76, 1690–1696. [Google Scholar] [CrossRef]
- Ding, M.; Andreasen, C.M.; Dencker, M.L.; Jensen, A.E.; Theilgaard, N.; Overgaard, S. Efficacy of a small cell-binding peptide coated hydroxyapatite substitute on bone formation and implant fixation in sheep. J. Biomed. Mater. Res. A 2015, 103, 1357–1365. [Google Scholar] [CrossRef] [PubMed]
- Matos, S.; Guerra, F.; Krauser, J.T.; Figueiredo, H.; Marcelino, J.P.; Sanz, M. Evaluation of an anorganic bovine-derived mineral with P-15 hydrogel bone graft: Preliminary study in a rabbit cranial bone model. Clin. Oral Implant. Res. 2012, 23, 698–705. [Google Scholar] [CrossRef]
- Arnold, P.M.; Sasso, R.C.; Janssen, M.E.; Fehlings, M.G.; Heary, R.F.; Vaccaro, A.R.; Kopjar, B. i-Factor bone graft vs autograft in anterior cervical discectomy and fusion: 2-year follow-up of the randomized single-blinded food and drug administration investigational device exemption study. Neurosurgery 2018, 83, 377–384. [Google Scholar] [CrossRef]
- Arnold, P.M.; Sasso, R.C.; Janssen, M.E.; Fehlings, M.G.; Smucker, J.D.; Vaccaro, A.R.; Heary, R.F.; Patel, A.I.; Goulet, B.; Kalfas, I.H.; et al. Efficacy of i-Factor bone graft versus autograft in anterior cervical discectomy and fusion: Results of the prospective, randomized, single-blinded food and drug administration investigational device exemption study. Spine 2016, 41, 1075–1083. [Google Scholar] [CrossRef] [PubMed]
- Gomar, F.; Orozco, R.; Villar, J.L.; Arrizabalaga, F. P-15 small peptide bone graft substitute in the treatment of non-unions and delayed union. A pilot clinical trial. Int. Orthop. 2007, 31, 93–99. [Google Scholar] [CrossRef]
- Mobbs, R.J.; Maharaj, M.; Rao, P.J. Clinical outcomes and fusion rates following anterior lumbar interbody fusion with bone graft substitute i-FACTOR, an anorganic bone matrix/P-15 composite. J. Neurosurg. Spine 2014, 21, 867–876. [Google Scholar] [CrossRef]
- Arnold, P.M.; Vaccaro, A.R.; Sasso, R.C.; Goulet, B.; Fehlings, M.G.; Heary, R.F.; Janssen, M.E.; Kopjar, B. Two-year clinical and radiological outcomes in patients with diabetes undergoing single-level anterior cervical discectomy and fusion. Global Spine J. 2021, 11, 458–464. [Google Scholar] [CrossRef] [PubMed]
- Oxborrow, N.; Sundarapandian, R. Heterotopic ossification following use of i-Factor for spinal fusion in Mucopolysaccharidosis 1: A case report. J. Surg. Case Rep. 2018, 2018, rjy120. [Google Scholar] [CrossRef] [PubMed]
- SmartBonePep®. Available online: https://www.ibi-sa.com/on-going-projects/smartbone-pep/ (accessed on 27 April 2021).
- Perale, G.; Monjo, M.; Ramis, J.M.; Ovrebo, O.; Betge, F.; Lyngstadaas, P.; Haugen, H.J. Biomimetic biomolecules in next generation xeno-hybrid bone graft material show enhanced in vitro bone cells response. J. Clin. Med. 2019, 8, 2159. [Google Scholar] [CrossRef] [PubMed]
- FDA. map3 Chips Allograft—Untitled letter. Available online: https://www.fda.gov/vaccines-blood-biologics/enforcement-actions-cber/map3-chips-allograft-untitled-letter (accessed on 7 June 2021).
- Boanini, E.; Gazzano, M.; Bigi, A. Ionic substitutions in calcium phosphates synthesized at low temperature. Acta Biomater. 2010, 6, 1882–1894. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Govoni, M.; Vivarelli, L.; Mazzotta, A.; Stagni, C.; Maso, A.; Dallari, D. Commercial Bone Grafts Claimed as an Alternative to Autografts: Current Trends for Clinical Applications in Orthopaedics. Materials 2021, 14, 3290. https://doi.org/10.3390/ma14123290
Govoni M, Vivarelli L, Mazzotta A, Stagni C, Maso A, Dallari D. Commercial Bone Grafts Claimed as an Alternative to Autografts: Current Trends for Clinical Applications in Orthopaedics. Materials. 2021; 14(12):3290. https://doi.org/10.3390/ma14123290
Chicago/Turabian StyleGovoni, Marco, Leonardo Vivarelli, Alessandro Mazzotta, Cesare Stagni, Alessandra Maso, and Dante Dallari. 2021. "Commercial Bone Grafts Claimed as an Alternative to Autografts: Current Trends for Clinical Applications in Orthopaedics" Materials 14, no. 12: 3290. https://doi.org/10.3390/ma14123290
APA StyleGovoni, M., Vivarelli, L., Mazzotta, A., Stagni, C., Maso, A., & Dallari, D. (2021). Commercial Bone Grafts Claimed as an Alternative to Autografts: Current Trends for Clinical Applications in Orthopaedics. Materials, 14(12), 3290. https://doi.org/10.3390/ma14123290









