Enhanced Optical and Antibacterial Activity of Hydrothermally Synthesized Cobalt-Doped Zinc Oxide Cylindrical Microcrystals
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
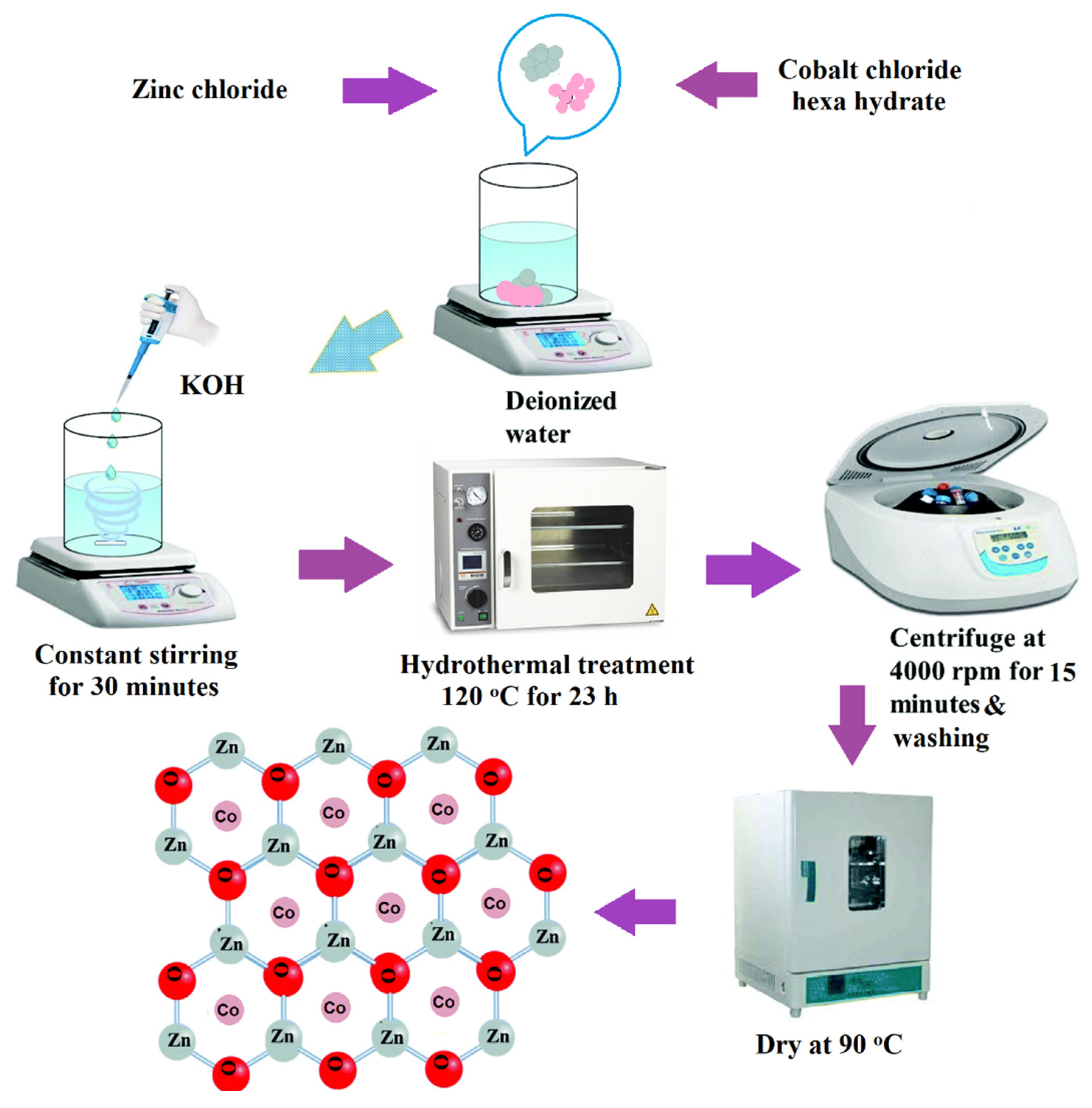
2.2. Synthesis of Co-Doped ZnO Microcrystals
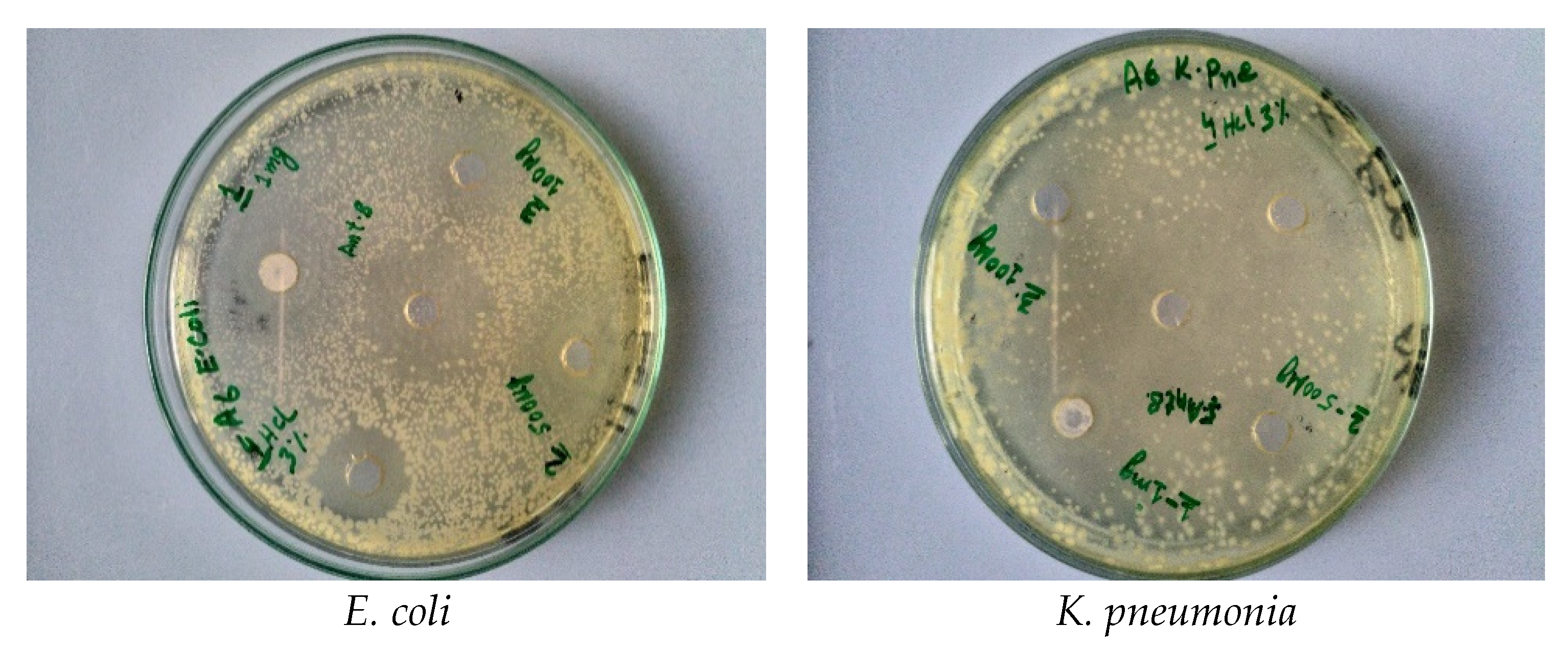
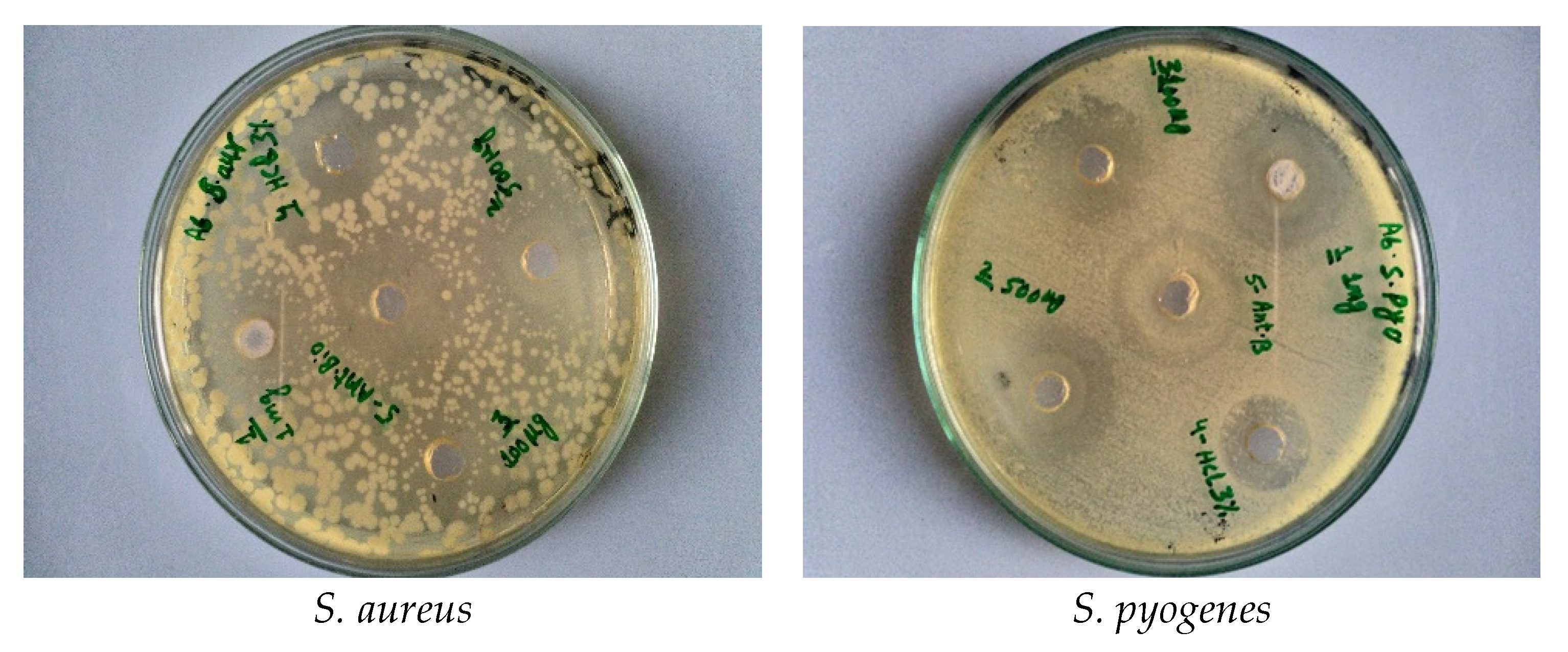
2.3. Screening of Antibacterial Activity
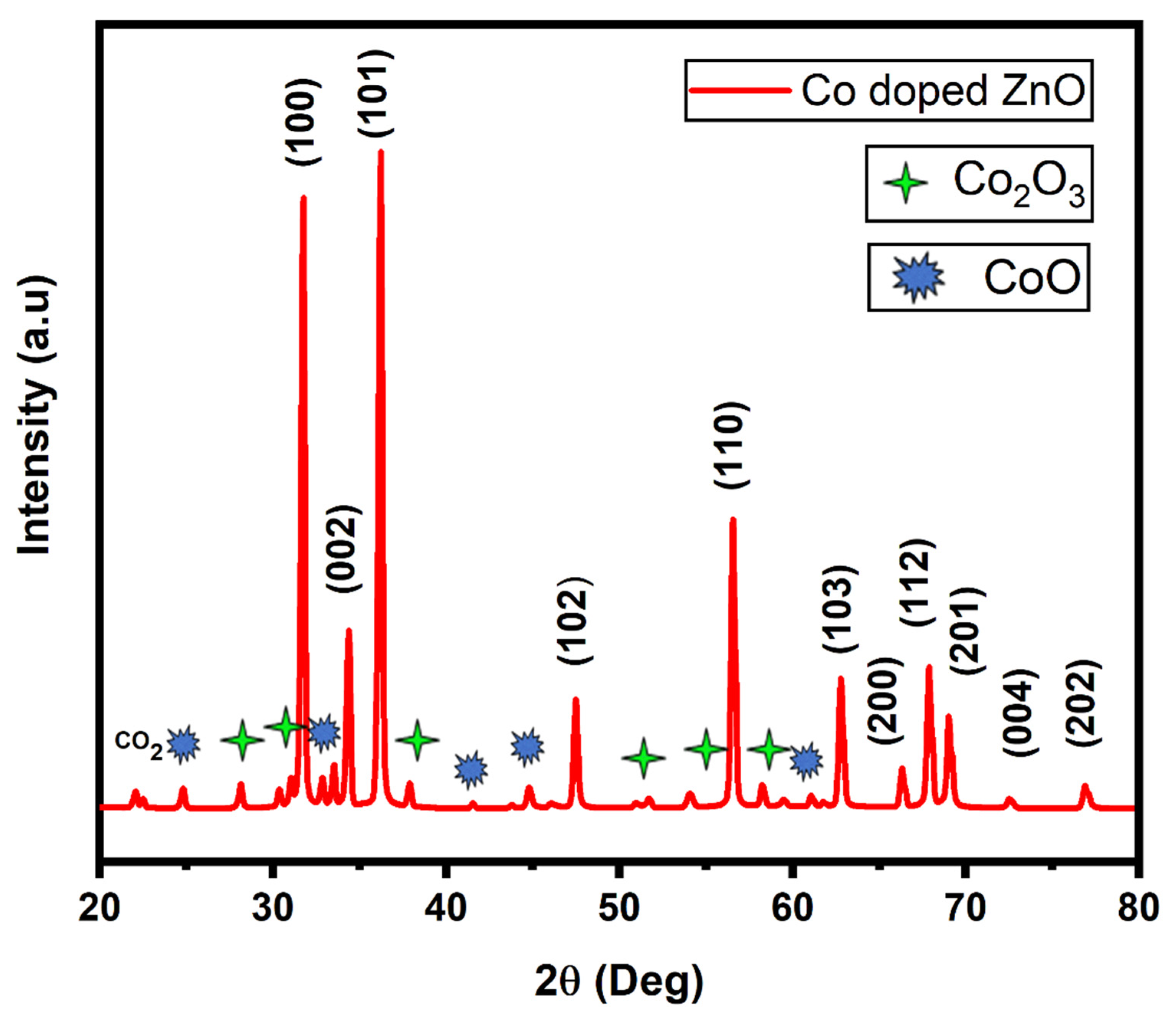
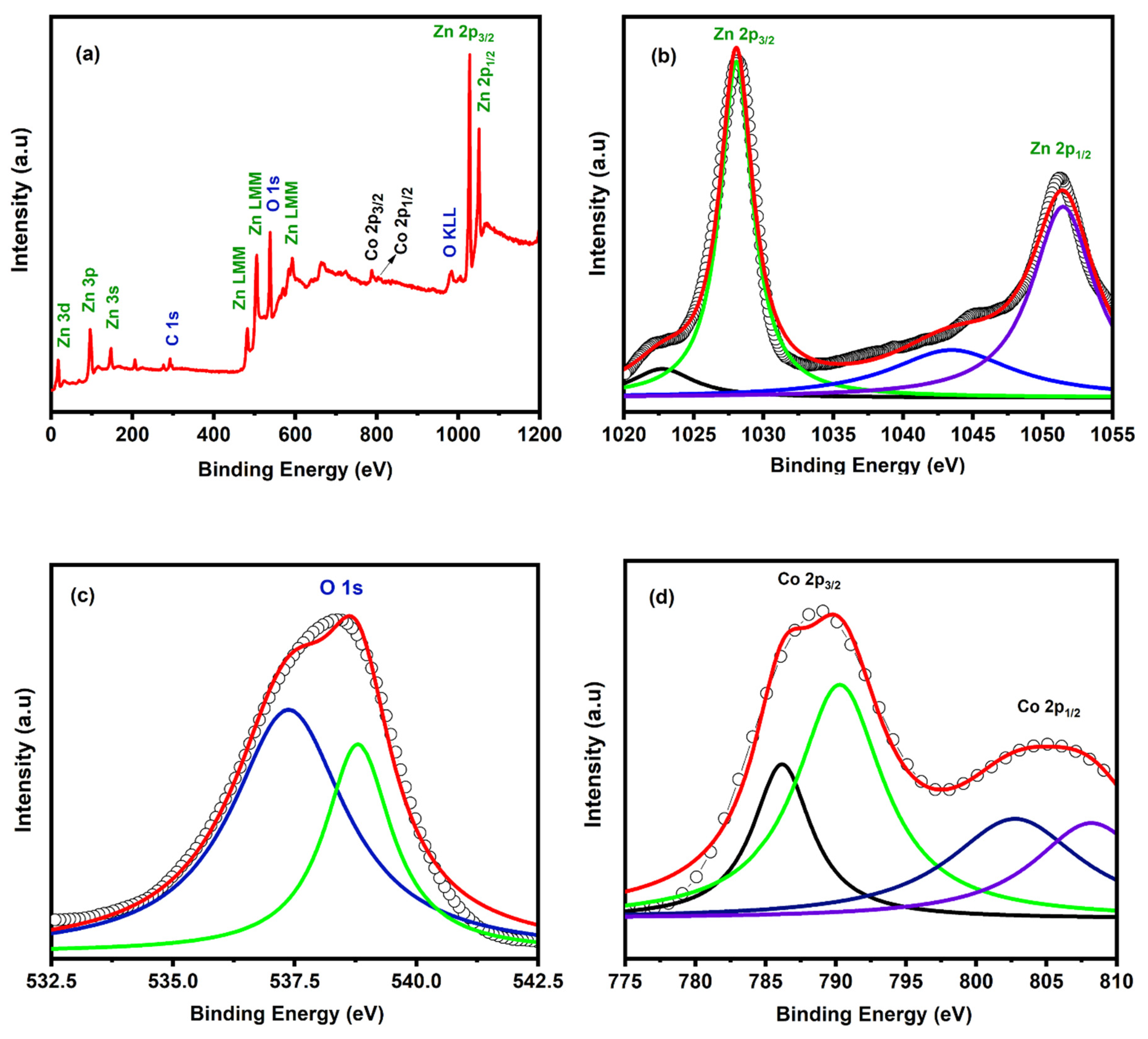
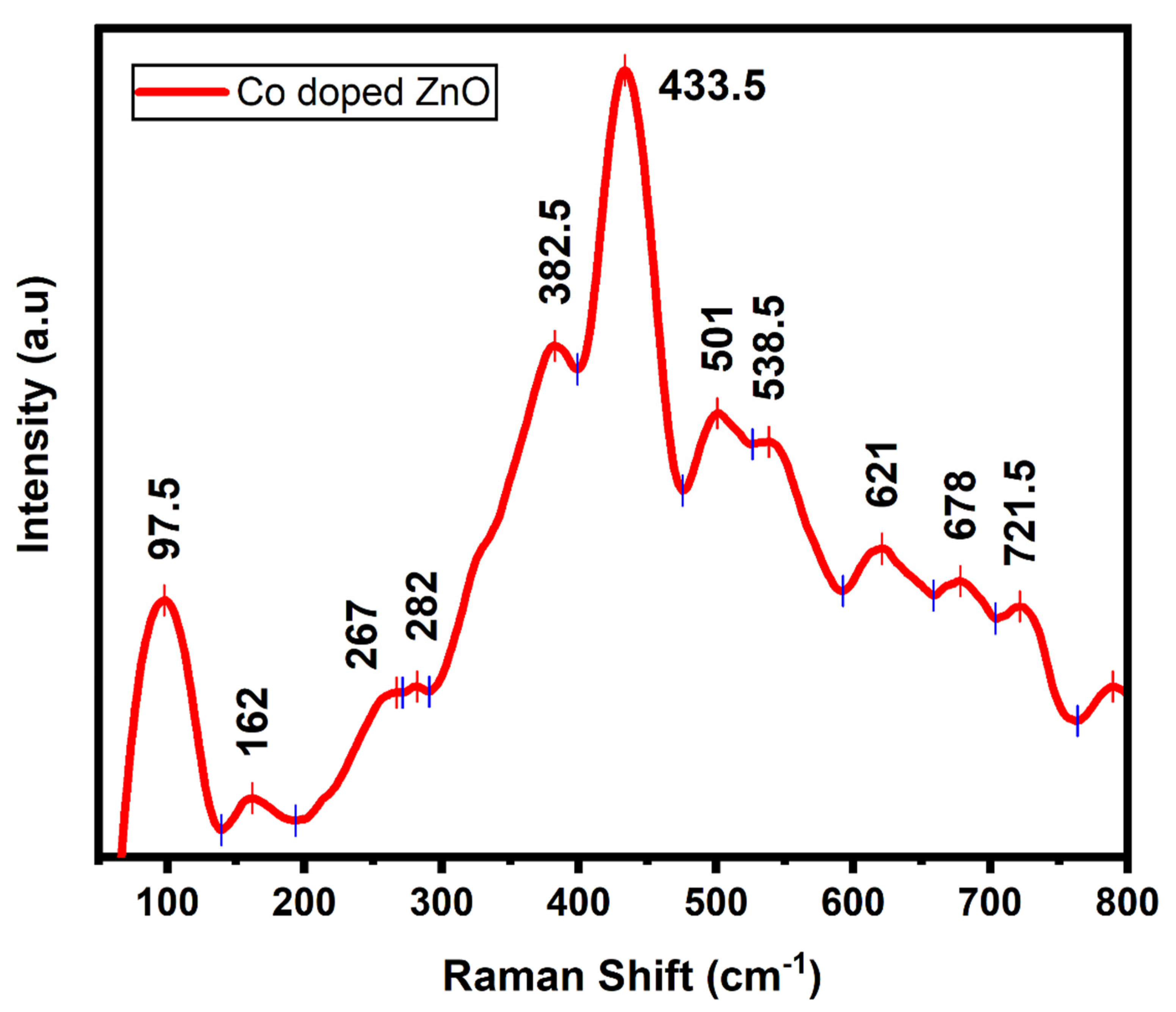
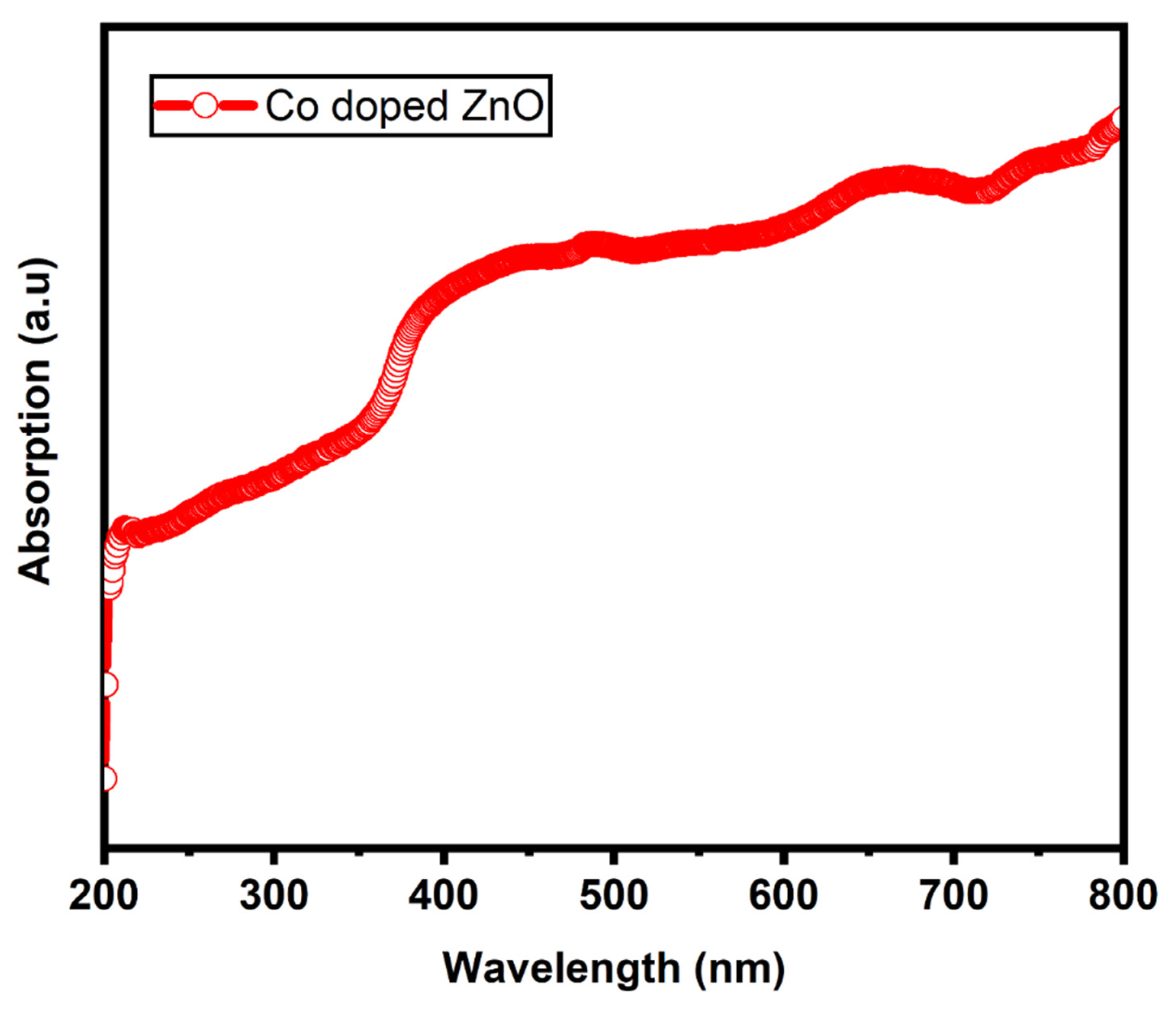
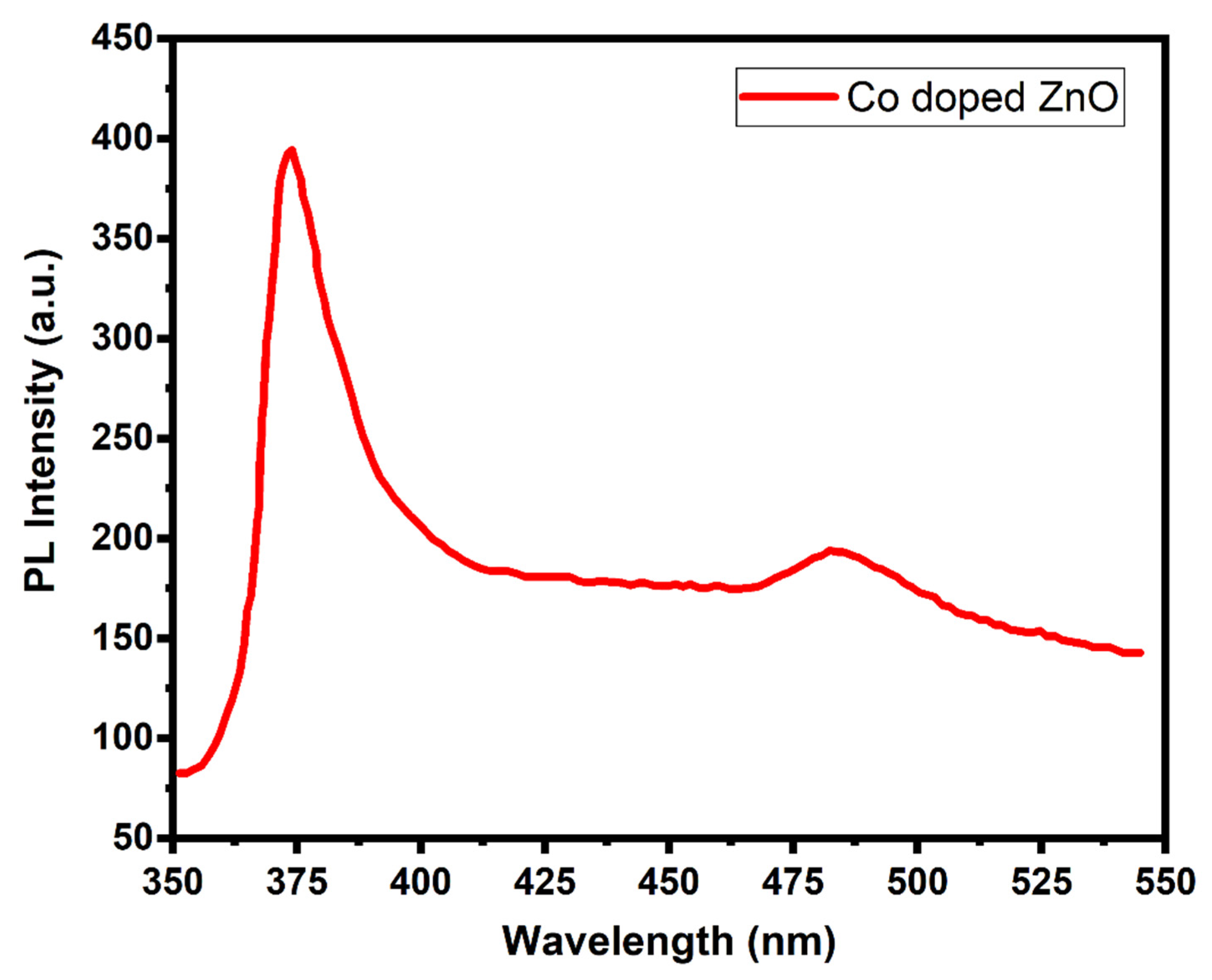
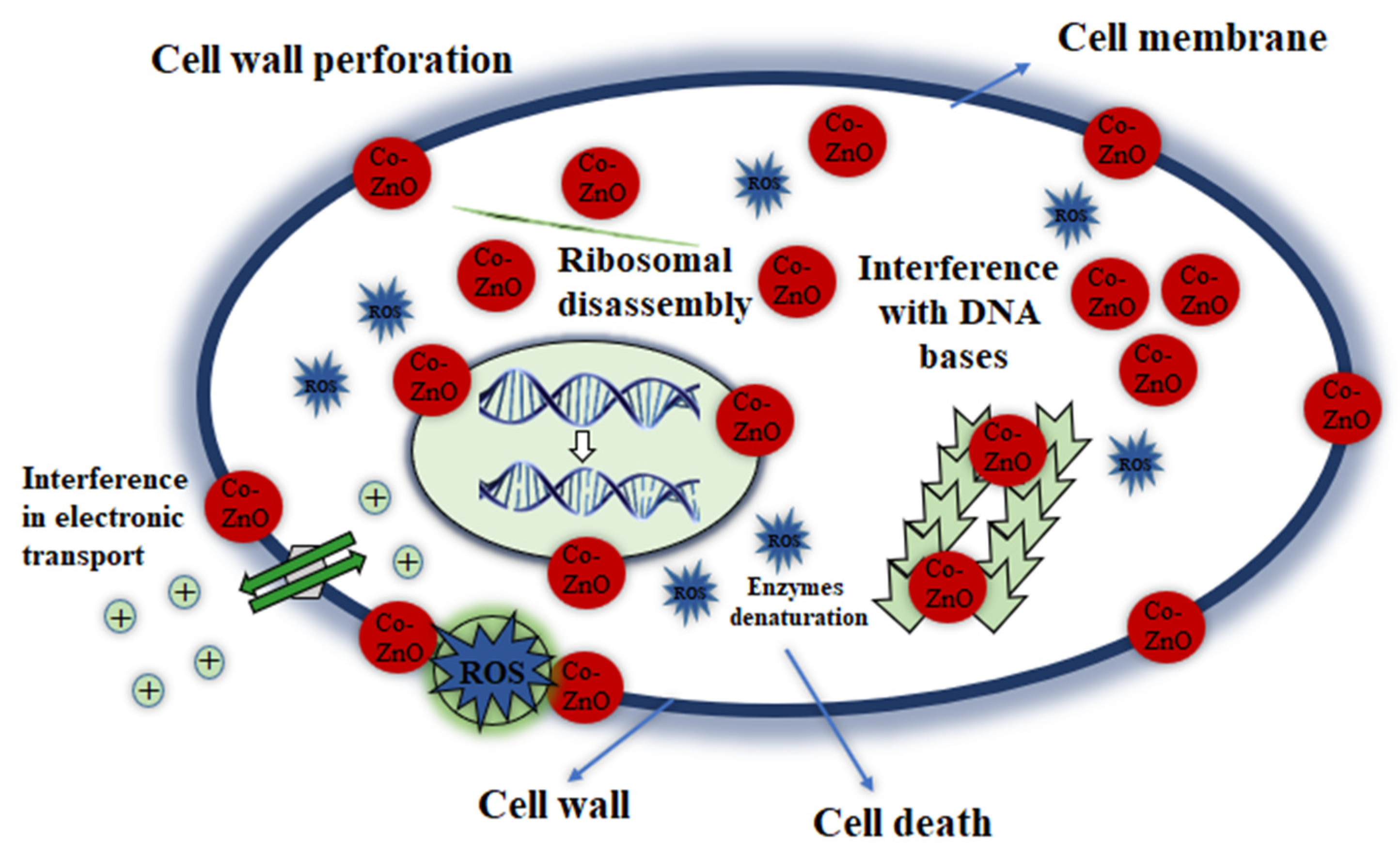
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bhushan, B. Fundamentals of Tribology and Bridging the Gap between the Macro-and Micro/Nanoscales; Springer: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Khalid, P.A.; Ahmad, A.I.; Alharthi, S.; Muhammad, M.U.; Khandaker, M.; Rehman, M.R.I.; Faruque, I.U.; Din, M.A.; Alotaibi, K.A. Structural, optical and antibacterial efficacy of pure and zinc-doped copper oxide against pathogenic bacteria. Nanomaterials 2021, 11, 451. [Google Scholar] [CrossRef]
- Meulenkamp, E.A. Synthesis and growth of ZnO nanoparticles. J. Phys. Chem. B 1998, 102, 5566–5572. [Google Scholar] [CrossRef]
- Özgür, Ü.; Alivov, Y.I.; Teke, C.L.A.; Reshchikov, M.A.; Doğan, S.; Avrutin, V.; Cho, S.-J.; Morkoç, H. A comprehensive review of ZnO materials and devices. J. Appl. Phys. 2005, 98, 11. [Google Scholar] [CrossRef]
- Ahmad, P.; Khandaker, M.U.; Amin, Y.M.; Muhammad, N. Synthesis and characterization of boron nitride microtubes. Mater. Express 2015, 5, 249–254. [Google Scholar] [CrossRef]
- Ozgür, Ü.; Hofstetter, D.; Morkoç, H. ZnO devices and applications: A review of current status and future prospects. Proc. IEEE 2010, 98, 1255–1268. [Google Scholar] [CrossRef]
- Necib, K.; Touam, T.; Chelouche, A.; Ouarez, L.; Djouadi, D.; Boudine, B. Investigation of the effects of thickness on physical properties of AZO sol-gel films for photonic device applications. J. Alloys Compd. 2018, 735, 2236–2246. [Google Scholar] [CrossRef]
- Srivastava, A.; Kumar, N.; Misra, K.P.; Khare, S. Enhancement of band gap of ZnO nanocrystalline films at a faster rate using Sr dopant. Electron. Mater. Lett. 2014, 10, 703–711. [Google Scholar] [CrossRef]
- Atif, M.; Younas, U.; Khalid, W.; Ahmed, Z.; Ali, M.; Nadeem, Z. Impedance spectroscopy, ferroelectric and optical properties of cobalt-doped Zn1−xCoxO nanoparticles. J. Mater. Sci. 2020, 31, 1–9. [Google Scholar]
- Chattopadhyay, S.; Misra, K.P.; Agarwala, A.; Rao, A.; Babu, P. Correlated quartic variation of band gap and NBE energy in sol-gel derived Zn1−xCoxO nanoparticles. Mater. Chem. Phys. 2019, 227, 236–241. [Google Scholar] [CrossRef]
- Nair, M.G.; Nirmala, M.; Rekha, K.; Anukaliani, A. Structural, optical, photo catalytic and antibacterial activity of ZnO and Co doped ZnO nanoparticles. Mater. Lett. 2011, 65, 1797–1800. [Google Scholar] [CrossRef]
- Chithira, P.; John, T.T. Correlation among oxygen vacancy and doping concentration in controlling the properties of cobalt doped ZnO nanoparticles. J. Magn. Magn. Mater. 2020, 496, 165928. [Google Scholar] [CrossRef]
- Thool, G.S.; Singh, A.K.; Singh, R.; Gupta, A.; Susan, A.B.H. Facile synthesis of flat crystal ZnO thin films by solution growth method: A micro-structural investigation. J. Saudi Chem. Soc. 2014, 18, 712–721. [Google Scholar] [CrossRef]
- Shatnawi, M.; Alsmadi, A.; Bsoul, I.; Salameh, B.; Alna’Washi, G.; Al-Dweri, F.; El Akkad, F. Magnetic and optical properties of Co-doped ZnO nanocrystalline particles. J. Alloys Compd. 2016, 655, 244–252. [Google Scholar] [CrossRef]
- Sharma, D.; Jha, R. Transition metal (Co, Mn) co-doped ZnO nanoparticles: Effect on structural and optical properties. J. Alloys Compd. 2017, 698, 532–538. [Google Scholar] [CrossRef]
- Goel, S.; Kumar, B. A review on piezo-/ferro-electric properties of morphologically diverse ZnO nanostructures. J. Alloys Compd. 2020, 816, 152491. [Google Scholar] [CrossRef]
- Khalid, A.; Ahmad, P.; Alharthi, A.; Muhammad, S.; Khandaker, M.; Faruque, M.I.; Din, I.; Alotaibi, M. Unmodified titanium dioxide nanoparticles as a potential contrast agent in photon emission computed tomography. Crystals 2021, 11, 171. [Google Scholar] [CrossRef]
- Fukumura, T.; Jin, Z.; Ohtomo, A.; Koinuma, H.; Kawasaki, M. An oxide-diluted magnetic semiconductor: Mn-doped ZnO. Appl. Phys. Lett. 1999, 75, 3366–3368. [Google Scholar] [CrossRef]
- Dejene, F.; Onani, M.; Koao, L.; Wako, A.; Motloung, S.; Yihunie, M. Structure, morphology and optical properties of undoped and MN-doped ZnO(1−x)Sx nano-powders prepared by precipitation method. Physica B 2016, 480, 63–67. [Google Scholar] [CrossRef]
- Godavarthi, U.; Mote, V.; Reddy, M.R.; Nagaraju, P.; Kumar, Y.V.; Dasari, K.T.; Dasari, M.P. Precipitated cobalt doped ZnO nanoparticles with enhanced low temperature xylene sensing properties. Physica B 2019, 553, 151–160. [Google Scholar] [CrossRef]
- Arif, M.; Sanger, A.; Shkir, M.; Singh, A.; Katiyar, R. Influence of interparticle interaction on the structural, optical and magnetic properties of NiO nanoparticles. Physica B 2019, 552, 88–95. [Google Scholar] [CrossRef]
- Khalid, A.; Ahmad, P.; Alharthi, A.I.; Muhammad, S.; Khandaker, M.U.; Faruque, M.R.I.; Din, I.U.; Alotaibi, M.A.; Khan, A. Synergistic effects of Cu-doped ZnO nanoantibiotic against Gram-positive bacterial strains. PLoS ONE 2021, 16, e0251082. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, P.; Khandaker, M.U.; Muhammad, N.; Khan, G.; Rehman, F.; Khan, A.S.; Ullah, Z.; Khan, A.; Ali, H.; Ahmed, S.M.; et al. Fabrication of hexagonal boron nitride quantum dots via a facile bottom-up technique. Ceram. Int. 2019, 45, 22765–22768. [Google Scholar] [CrossRef]
- Ahmed, S.M.; Kazi, S.N.; Khan, G.; Dahari, M.; Zubir, M.N.M.; Ahmad, P.; Montazer, E. Experimental investigation on momentum and drag reduction of Malaysian crop suspensions in closed conduit flow. IOP Conf. Ser. 2017, 210, 012065. [Google Scholar] [CrossRef]
- Ahmad, P.; Khandaker, M.U.; Amin, Y.M.; Muhammad, N.; Khan, G.; Khan, A.S.; Numan, A.; Rehman, M.A.; Ahmed, S.M.; Khan, A. Synthesis of hexagonal boron nitride fibers within two hour annealing at 500 °C and two hour growth duration at 1000 °C. Ceram. Int. 2016, 42, 14661–14666. [Google Scholar] [CrossRef]
- Shannon, R.D. Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Crystallogr. 1976, 32, 751–767. [Google Scholar] [CrossRef]
- Hammad, T.M.; Salem, J.K.; Harrison, R.G. Structure, optical properties and synthesis of Co-doped ZnO superstructures. Appl. Nanosci. 2013, 3, 133–139. [Google Scholar] [CrossRef]
- Straumal, B.; Baretzky, B.; Mazilkin, A.; Protasova, S.; Myatiev, A.; Straumal, P. Increase of Mn solubility with decreasing grain size in ZnO. J. Eur. Ceram. Soc. 2009, 29, 1963–1970. [Google Scholar] [CrossRef]
- Woo, H.-S.; Kwak, C.-H.; Chung, J.-H.; Lee, J.-H. Co-doped branched ZnO nanowires for ultraselective and sensitive detection of xylene. ACS Appl. Mater. Interfaces 2014, 6, 22553–22560. [Google Scholar] [CrossRef]
- Zhu, S.; Xu, L.; Yang, S.; Zhou, X.; Chen, X.; Dong, B.; Bai, X.; Lu, G.; Song, H. Cobalt-doped ZnO nanoparticles derived from zeolite imidazole frameworks: Synthesis, characterization, and application for the detection of an exhaled diabetes biomarker. J. Colloid Interface Sci. 2020, 569, 358–365. [Google Scholar] [CrossRef]
- Taskin, M.; Podder, J. Structural, optical and electrical properties of pure and Co-doped ZnO nano fiber thin films prepared by spray pyrolysis. Appl. Sci. Rep. 2014, 2, 107–113. [Google Scholar]
- Adeel, M.; Saeed, M.; Khan, I.; Muneer, M.; Akram, N. Synthesis and characterization of Co-ZnO and evaluation of its photocatalytic activity for photodegradation of methyl orange. ACS Omega 2021, 6, 1426–1435. [Google Scholar] [CrossRef] [PubMed]
- Fabbiyola, S.; Kennedy, L.J. Bandgap engineering in doped ZnO nanostructures for dye sensitized solar cell applications. J. Nanosci. Nanotechnol. 2019, 19, 2963–2970. [Google Scholar] [CrossRef] [PubMed]
- Ali, H.T.; Jacob, J.; Khalid, M.; Mahmood, K.; Yusuf, M.; Mehboob, K.; Ikram, S.; Ali, A.; Amin, N.; Ashar, A. Optimizing the structural, morphological and thermoelectric properties of zinc oxide by the modulation of cobalt doping concentration. J. Alloys Compd. 2021, 871, 159564. [Google Scholar] [CrossRef]
- Kumar, R.; Kumar, P. Co-Doped ZnO Nanoparticles Reinforcement in PVDF for 3D Printing of Magnetic Structures; Elsevier: Amsterdam, The Netherlands, 2020. [Google Scholar] [CrossRef]
- Skoda, D.; Urbanek, P.; Sevcik, J.; Münster, L.; Nadazdy, V.; Cullen, D.A.; Bazant, P.; Antos, J.; Kuritka, I. Colloidal cobalt-doped ZnO nanoparticles by microwave-assisted synthesis and their utilization in thin composite layers with MEH-PPV as an electroluminescent material for polymer light emitting diodes. Org. Electron. 2018, 59, 337–348. [Google Scholar] [CrossRef]
- Zang, W.; Li, P.; Fu, Y.; Xing, L.; Xue, X. Hydrothermal synthesis of Co-ZnO nanowire array and its application as piezo-driven self-powered humidity sensor with high sensitivity and repeatability. RSC Adv. 2015, 5, 84343–84349. [Google Scholar] [CrossRef]
- Vijayaprasath, G.; Murugan, R.; Narayanan, J.S.; Dharuman, V.; Ravi, G.; Hayakawa, Y. Glucose sensing behavior of cobalt doped ZnO nanoparticles synthesized by co-precipitation method. J. Mater. Sci. Mater. Electron. 2015, 26, 4988–4996. [Google Scholar] [CrossRef]
- Kołodziejczak-Radzimska, A.; Jesionowski, T. Zinc oxide—From synthesis to application: A review. Materials 2014, 7, 2833–2881. [Google Scholar] [CrossRef]
- Kumaresan, S.; Vallalperuman, K.; Sathishkumar, S.; Karthik, M.; Siva-Karthik, P. Synthesis and systematic investigations of Al and Cu-doped ZnO nanoparticles and its structural, optical and photo-catalytic properties. J. Mater. Sci. 2017, 28, 9199–9205. [Google Scholar] [CrossRef]
- Lu, Y.; Lin, Y.; Wang, D.; Wang, L.; Xie, T.; Jiang, T. A high performance cobalt-doped ZnO visible light photocatalyst and its photogenerated charge transfer properties. Nano Res. 2011, 4, 1144–1152. [Google Scholar] [CrossRef]
- Kalpana, S.; Krishnan, S.; Senthil, T.; Elangovan, S. Cobalt doped Zinc oxide nanoparticles for photocatalytic applications. J. Ovonic Res. 2017, 13, 263–269. [Google Scholar]
- Wang, X.L.; Luan, C.Y.; Shao, Q.; Pruna, A.; Leung, C.W.; Lortz, R.; Zapien, J.A.; Ruotolo, A. Effect of the magnetic order on the room-temperature band-gap of Mn-doped ZnO thin films. Appl. Phys. Lett. 2013, 102, 102112. [Google Scholar] [CrossRef]
- Srinet, G.; Varshney, P.; Kumar, R.; Sajal, V.; Kulriya, P.; Knobel, M.; Sharma, S. Structural, optical and magnetic properties of Zn1-xCoxO prepared by the sol-gel route. Ceram. Int. 2013, 39, 6077–6085. [Google Scholar] [CrossRef]
- Gandhi, V.; Ganesan, R.; Syedahamed, H.H.A.; Thaiyan, M. Effect of cobalt doping on structural, optical, and magnetic properties of ZnO nanoparticles synthesized by coprecipitation method. J. Phys. Chem. C 2014, 118, 9715–9725. [Google Scholar] [CrossRef]
- Djerdj, I.; Jagličić, Z.; Arčon, D.; Niederberger, M. Co-doped ZnO nanoparticles: Minireview. Nanoscale 2010, 2, 1096–1104. [Google Scholar] [CrossRef]
- Wojnarowicz, J.; Chudoba, T.; Lojkowski, W. A review of microwave synthesis of zinc oxide nanomaterials: Reactants, process parameters and morphologies. Nanomaterials 2020, 10, 1086. [Google Scholar] [CrossRef] [PubMed]
- Leyssens, L.; Vinck, B.; van der Straeten, C.; Wuyts, F.; Maes, L. Cobalt toxicity in humans—A review of the potential sources and systemic health effects. Toxicology 2017, 387, 43–56. [Google Scholar] [CrossRef]
- Tvermoes, B.E.; Paustenbach, D.J.; Kerger, B.D.; Finley, B.L.; Unice, K.M. Review of cobalt toxicokinetics following oral dosing: Implications for health risk assessments and metal-on-metal hip implant patients. Crit. Rev. Toxicol. 2015, 45, 367–387. [Google Scholar] [CrossRef]
- Unice, K.M.; Kerger, B.D.; Paustenbach, D.J.; Finley, B.L.; Tvermoes, B.E. Refined biokinetic model for humans exposed to cobalt dietary supplements and other sources of systemic cobalt exposure. Chem. Biol. Interact. 2014, 216, 53–74. [Google Scholar] [CrossRef]
- Kaphle, A.; Reed, T.; Apblett, A.; Hari, P. Doping efficiency in cobalt-doped ZnO nanostructured materials. J. Nanomater. 2019, 2019, 1–13. [Google Scholar] [CrossRef]
- Rajendar, V.; Dayakar, T.; Shobhan, K.; Srikanth, I.; Rao, K.V. Systematic approach on the fabrication of Co doped ZnO semiconducting nanoparticles by mixture of fuel approach for Antibacterial applications. Superlattices Microstruct. 2014, 75, 551–563. [Google Scholar] [CrossRef]
- Narasimman, S.; Balakrishnan, L.; Alex, Z.C. Fiber optic magnetic field sensor using Co doped ZnO nanorods as cladding. RSC Adv. 2018, 8, 18243–18251. [Google Scholar] [CrossRef]
- Rana, S.B.; Singh, R.P.P.; Arya, S. Structural, optical, magnetic and antibacterial study of pure and cobalt doped ZnO nanoparticles. J. Mater. Sci. Mater. Electron. 2016, 28, 2660–2672. [Google Scholar] [CrossRef]
- Godavarthi, U.; Mote, V.; Dasari, M. Role of cobalt doping on the electrical conductivity of ZnO nanoparticles. J. Asian Ceram. Soc. 2017, 5, 391–396. [Google Scholar] [CrossRef]
- Kayani, Z.N.; Shah, I.; Zulfiqar, B.; Riaz, S.; Naseem, S.; Sabah, A. Structural, optical and magnetic properties of nanocrystalline co-doped ZnO thin films grown by sol-gel. Zeitschrift für Naturforschung A 2017, 73, 13–21. [Google Scholar] [CrossRef]
- Kaphle, A.; Echeverria, E.; Mcllroy, D.N.; Roberts, K.; Hari, P. Thermo-optical properties of cobalt-doped zinc oxide (ZnO) nanorods. J. Nanosci. Nanotechnol. 2019, 19, 3893–3904. [Google Scholar] [CrossRef]
- National Committee for Clinical Laboratory Standards; Barry, A.L. Methods for Determining Bactericidal Activity of Antimicrobial Agents: Approved Guideline; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 1999. [Google Scholar]
- Wojnarowicz, J.; Kusnieruk, S.; Chudoba, T.; Gierlotka, S.; Lojkowski, W.; Knoff, W.; Lukasiewicz, M.I.; Witkowski, B.S.; Wolska, A.; Klepka, M.T.; et al. Paramagnetism of cobalt-doped ZnO nanoparticles obtained by microwave solvothermal synthesis. Beilstein J. Nanotechnol. 2015, 6, 1957–1969. [Google Scholar] [CrossRef]
- Hu, J.; Gao, F.; Zhao, Z.; Sang, S.; Li, P.; Zhang, W.; Zhou, X.; Chen, Y. Synthesis and characterization of Cobalt-doped ZnO microstructures for methane gas sensing. Appl. Surf. Sci. 2016, 363, 181–188. [Google Scholar] [CrossRef]
- Singhal, A.; Achary, S.N.; Manjanna, J.; Chatterjee, S.; Ayyub, P.; Tyagi, A.K. Chemical synthesis and structural and magnetic properties of dispersible cobalt- and nickel-doped ZnO nanocrystals. J. Phys. Chem. C 2010, 114, 3422–3430. [Google Scholar] [CrossRef]
- Taher, F.A.; Abdeltwab, E. Shape-controlled synthesis of nanostructured Co-doped ZnO thin films and their magnetic properties. CrystEngComm 2018, 20, 5844–5856. [Google Scholar] [CrossRef]
- Pal, B.; Giri, P.K. High temperature ferromagnetism and optical properties of Co doped ZnO nanoparticles. J. Appl. Phys. 2010, 108, 084322. [Google Scholar] [CrossRef]
- Bundesmann, C.; Ashkenov, N.; Schubert, M.; Spemann, D.; Butz, T.; Kaidashev, E.M.; Lorenz, M.; Grundmann, M. Raman scattering in ZnO thin films doped with Fe, Sb, Al, Ga, and Li. Appl. Phys. Lett. 2003, 83, 1974–1976. [Google Scholar] [CrossRef]
- Manjón, F.J.; Marí, B.; Serrano, J.; Romero, A.H. Silent Raman modes in zinc oxide and related nitrides. J. Appl. Phys. 2005, 97, 053516. [Google Scholar] [CrossRef]
- Chanda, A.; Gupta, S.; Vasundhara, M.; Joshi, S.R.; Mutta, G.R.; Singh, J. Study of structural, optical and magnetic properties of cobalt doped ZnO nanorods. RSC Adv. 2017, 7, 50527–50536. [Google Scholar] [CrossRef]
- Duan, L.B.; Rao, G.H.; Wang, Y.C.; Yu, J.; Wang, T. Magnetization and Raman scattering studies of (Co, Mn) codoped ZnO nanoparticles. J. Appl. Phys. 2008, 104, 013909. [Google Scholar] [CrossRef]
- Ghosh, J.; Ghosh, R.; Giri, P. Tuning the visible photoluminescence in Al doped ZnO thin film and its application in label-free glucose detection. Sens. Actuators B 2018, 254, 681–689. [Google Scholar] [CrossRef]
- Çakıcı, T.; Sarıtaş, S.; Muğlu, G.M.; Kundakçı, M.; Yildirim, M. Magnetic, optical and structural characterization of ZnO: Co; ZnO: Fe thin films. AIP Conf. Proc. 2017, 1833, 020094. [Google Scholar]
- Lim, J.; Shin, K.; Kim, H.W.; Lee, C. Photoluminescence studies of ZnO thin films grown by atomic layer epitaxy. J. Lumin. 2004, 109, 181–185. [Google Scholar] [CrossRef]
- Azmi, R.; Oh, S.-H.; Jang, S.-Y. High-efficiency colloidal quantum dot photovoltaic devices using chemically modified heterojunctions. ACS Energy Lett. 2016, 1, 100–106. [Google Scholar] [CrossRef]
- Baiqi, W.; Xudong, S.; Qiang, F.; Iqbal, J.; Yan, L.; Honggang, F.; Dapeng, Y. Photoluminescence properties of Co-doped ZnO nanorods array fabricated by the solution method. Phys. E 2009, 41, 413–417. [Google Scholar] [CrossRef]
- Valgas, C.; Souza, S.M.D.; Smânia, E.F.; Smânia, A., Jr. Screening methods to determine antibacterial activity of natural products. Braz. J. Microbiol. 2007, 38, 369–380. [Google Scholar] [CrossRef]
- Raghupathi, K.R.; Koodali, R.T. Manna, size-dependent bacterial growth inhibition and mechanism of antibacterial activity of zinc oxide nanoparticles. Langmuir 2011, 27, 4020–4028. [Google Scholar] [CrossRef]
- Alfadda, A.A.; Sallam, R.M. Reactive oxygen species in health and disease. J. Biomed. Biotechnol. 2012, 2012, 1–14. [Google Scholar] [CrossRef]
- Lee, S.H.; Gupta, M.; Bang, J.B.; Bae, H.; Sung, H.-J. Current progress in reactive oxygen species (ROS)-responsive materials for biomedical applications. Adv. Heal. Mater. 2013, 2, 908–915. [Google Scholar] [CrossRef]
- Khalid, A.; Ahmad, P.; Alharthi, A.I.; Mohammad, S.; Khandaker, M.U.; Faruque, M.R.I.; Din, U.; Alotaibi, M.A. A practical method for incorporation of Fe (III) in Titania matrix for photocatalytic applications. Mater. Res. Express 2021. [Google Scholar] [CrossRef]
- Semeraro, P.; Bettini, S.; Sawalha, S.; Pal, S.; Licciulli, A.; Marzo, F.; Lovergine, N.; Valli, L.; Giancane, G. Photocatalytic degradation of tetracycline by ZnO/γ-Fe2O3 paramagnetic nanocomposite material. Nanomaterials 2020, 10, 1458. [Google Scholar] [CrossRef]
- Bettini, S.; Pagano, R.; Semeraro, P.; Ottolini, M.; Salvatore, L.; Marzo, F.; Lovergine, N.; Giancane, G.; Valli, L. SiO 2—Coated ZnO nanoflakes decorated with Ag nanoparticles for photocatalytic water oxidation. Chem. A Eur. J. 2019, 25, 14123. [Google Scholar] [CrossRef]
- Fang, M.; Chen, J.-H.; Xu, X.-L.; Yang, P.-H.; Hildebrand, H.F. Antibacterial activities of inorganic agents on six bacteria associated with oral infections by two susceptibility tests. Int. J. Antimicrob. Agents 2006, 27, 513–517. [Google Scholar] [CrossRef]
- Hameed, A.S.H.; Karthikeyan, C.; Sasikumar, S.; Kumar, V.S.; Kumaresan, S.; Ravi, G. Impact of alkaline metal ions Mg2+, Ca2+, Sr2+ and Ba2+ on the structural, optical, thermal and antibacterial properties of ZnO nanoparticles prepared by the co-precipitation method. J. Mater. Chem. B 2013, 1, 5950–5962. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Jiang, Y.; Ding, Y.; Daskalakis, N.; Jeuken, L.; Povey, M.; O’Neill, A.J.; York, D.W. Mechanistic investigation into antibacterial behaviour of suspensions of ZnO nanoparticles against E. coli. J. Nanopart. Res. 2010, 12, 1625–1636. [Google Scholar] [CrossRef]
- Cowan, S.T. Cowan and Steel’s Manual for the Identification of Medical Bacteria; Cambridge University Press: Cambridge, UK, 2004. [Google Scholar]

| S. No/Reference | Precursors | Temperature/Time | Technique | Morphology | Product | Confirmation | Year |
|---|---|---|---|---|---|---|---|
| [52] | Zn(NO3)2 6H2O, Co(NO3)2 6H2O | 100 °C/16 h | Sol-gel combustion | Granular surface | Co-doped-ZnO | FE-SEM * | 2014 |
| [14] | ZnO, CoO | 250 rpm/12 h | Ball milling | Nano-particles | Co-doped-ZnO | SEM ** | 2016 |
| [54] | Zn(CH3COO)2·2H2O, Co(CH3COO)2·4H2O | Room temperature /3 h | Wet precipitation | Nano-particles | Co-doped-ZnO | SEM | 2017 |
| [55] | Zn(CH3COO)2·2H2O, Co(CH3COO)2·4H2O | 325 K/2 h | Co-Precipitation | Nano-particles | Co-doped-ZnO | SEM | 2017 |
| [56] | Zn(CH3COO)2·H2O, Co(NO3)2·6H2O | 60 °C/0.5 h | Sol-gel dip-coating | Clustered grains | Co-doped-ZnO | SEM | 2017 |
| [36] | Zn(OAc)2·2H2O, Co(II)(Acac)2 | 250 °C/0.25 h | Microwave-assisted polyol | Nano colloids | Co-doped-ZnO | SEM | 2018 |
| [57] | Zn(NO3)2·6H2O, Co(NO3)3·6H2O | 95 °C/6 h | Chemical bath deposition | Nano rods | Co-doped-ZnO | SEM | 2019 |
| Our Article | ZnCl2, CoCl3·6H2O | 120 °C/23 h | Hydro-thermal | Cylindrical microcrystals | Co-doped-ZnO | FE-SEM | 2021 |
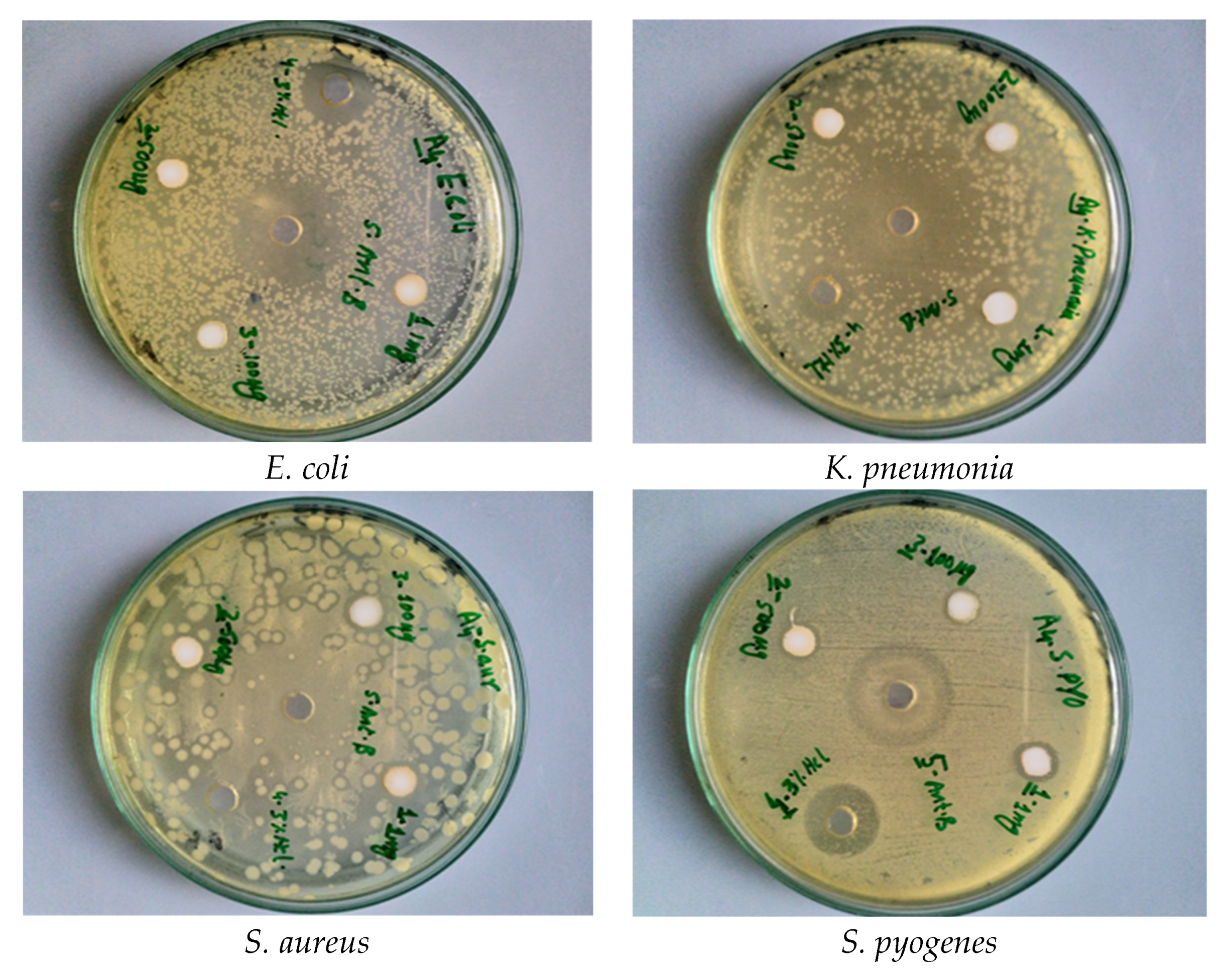
| Bacteria | ZnO | Co Doped ZnO | ||||||
|---|---|---|---|---|---|---|---|---|
| 100 µg/mL | 500 µg/mL | 1 mg/mL | 100 µg/mL | 500 µg/mL | 1 mg/mL | |||
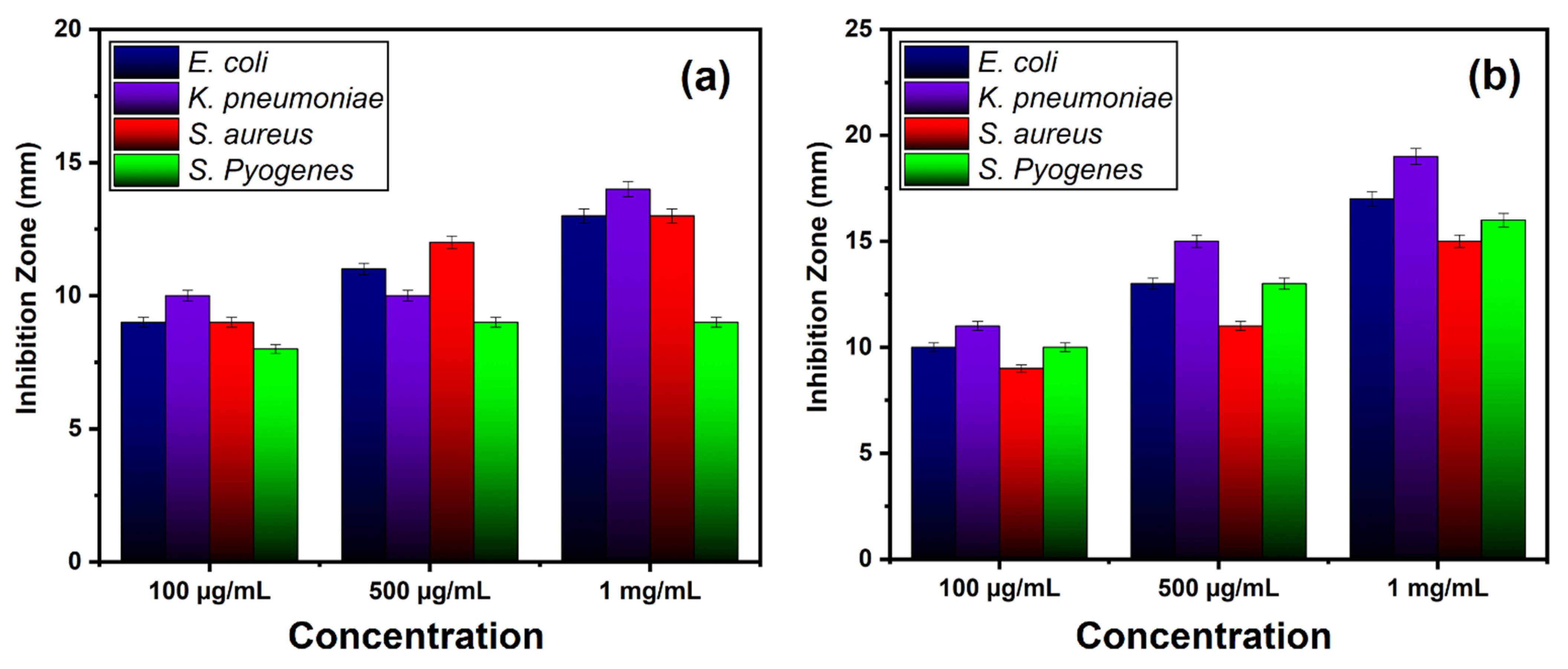
| Gram-negative | E. coli | Inhibition zone (mm) | 9 ± 0.18 | 11 ± 0.26 | 13 ± 0.26 | 10 ± 0.2 | 13 ± 0.26 | 17 ± 0.34 |
| K. pneumoniae | 10 ± 0.2 | 10 ± 0.21 | 14 ± 0.28 | 11 ± 0.22 | 15 ± 0.3 | 19 ± 0.38 | ||
| Gram-positive | S. aureus | 9 ± 0.19 | 12 ± 0.24 | 13 ± 0.26 | 9 ± 0.18 | 11 ± 0.22 | 15 ± 0.3 | |
| S. pyogenes | 8 ± 0.16 | 9 ± 0.18 | 9 ± 0.18 | 10 ± 0.2 | 13 ± 0.26 | 16 ± 0.32 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khalid, A.; Ahmad, P.; Alharthi, A.I.; Muhammad, S.; Khandaker, M.U.; Faruque, M.R.I.; Khan, A.; Din, I.U.; Alotaibi, M.A.; Alzimami, K.; et al. Enhanced Optical and Antibacterial Activity of Hydrothermally Synthesized Cobalt-Doped Zinc Oxide Cylindrical Microcrystals. Materials 2021, 14, 3223. https://doi.org/10.3390/ma14123223
Khalid A, Ahmad P, Alharthi AI, Muhammad S, Khandaker MU, Faruque MRI, Khan A, Din IU, Alotaibi MA, Alzimami K, et al. Enhanced Optical and Antibacterial Activity of Hydrothermally Synthesized Cobalt-Doped Zinc Oxide Cylindrical Microcrystals. Materials. 2021; 14(12):3223. https://doi.org/10.3390/ma14123223
Chicago/Turabian StyleKhalid, Awais, Pervaiz Ahmad, Abdulrahman I. Alharthi, Saleh Muhammad, Mayeen Uddin Khandaker, Mohammad Rashed Iqbal Faruque, Abdulhameed Khan, Israf Ud Din, Mshari A. Alotaibi, Khalid Alzimami, and et al. 2021. "Enhanced Optical and Antibacterial Activity of Hydrothermally Synthesized Cobalt-Doped Zinc Oxide Cylindrical Microcrystals" Materials 14, no. 12: 3223. https://doi.org/10.3390/ma14123223
APA StyleKhalid, A., Ahmad, P., Alharthi, A. I., Muhammad, S., Khandaker, M. U., Faruque, M. R. I., Khan, A., Din, I. U., Alotaibi, M. A., Alzimami, K., Alfuraih, A. A., & Bradley, D. A. (2021). Enhanced Optical and Antibacterial Activity of Hydrothermally Synthesized Cobalt-Doped Zinc Oxide Cylindrical Microcrystals. Materials, 14(12), 3223. https://doi.org/10.3390/ma14123223











