Calcium Carbonate Cement: A Carbon Capture, Utilization, and Storage (CCUS) Technique
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Materials
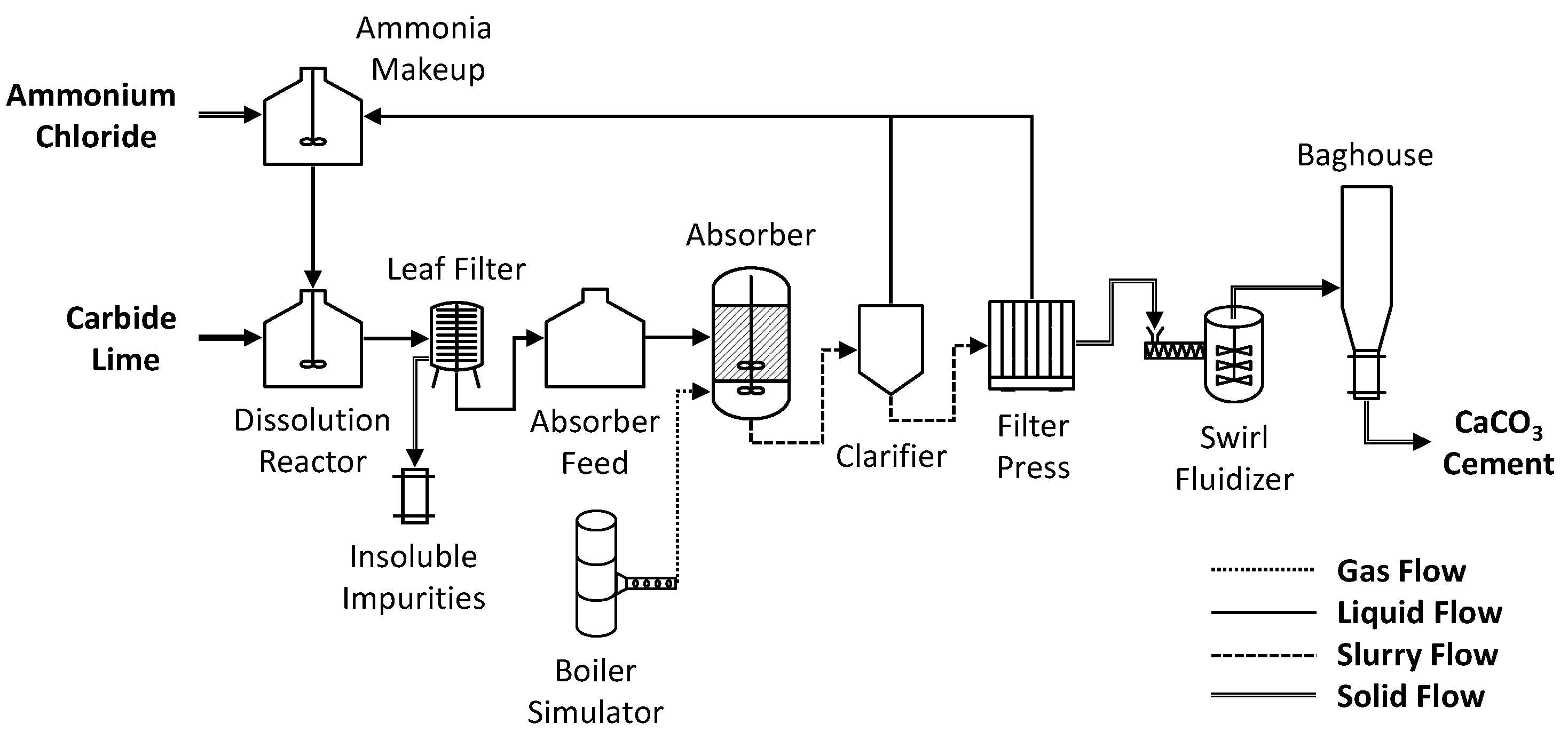
2.2. Aqueous Mineralization Process
2.3. Analysis and Testing
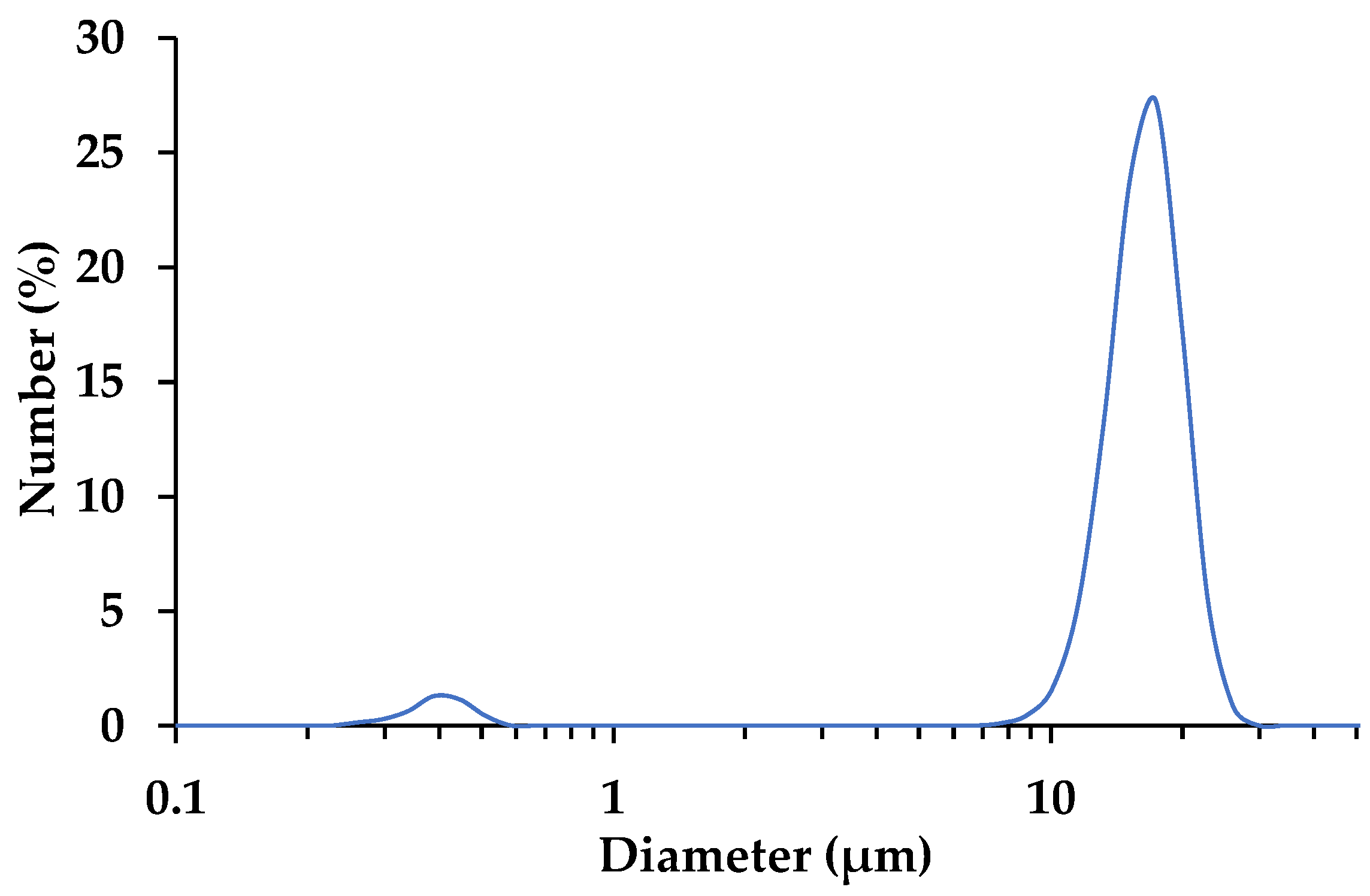
2.3.1. Laser Particle Size Distribution
2.3.2. X-ray Florescence
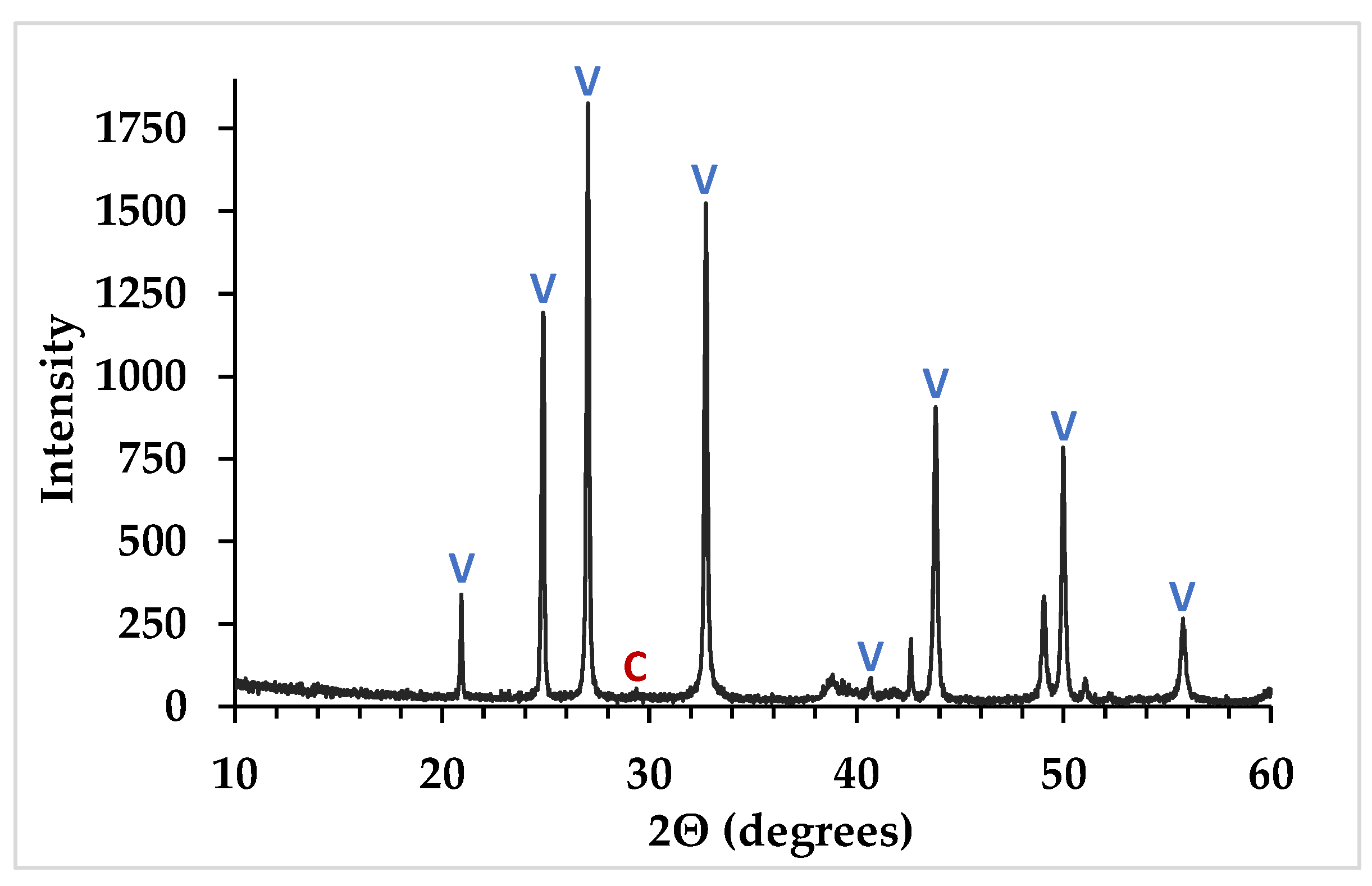
2.3.3. X-ray Diffraction
2.3.4. Scanning Electron Microscopy
2.3.5. Compressive Strength
3. Results
3.1. CaCO3 Cement Characteristics
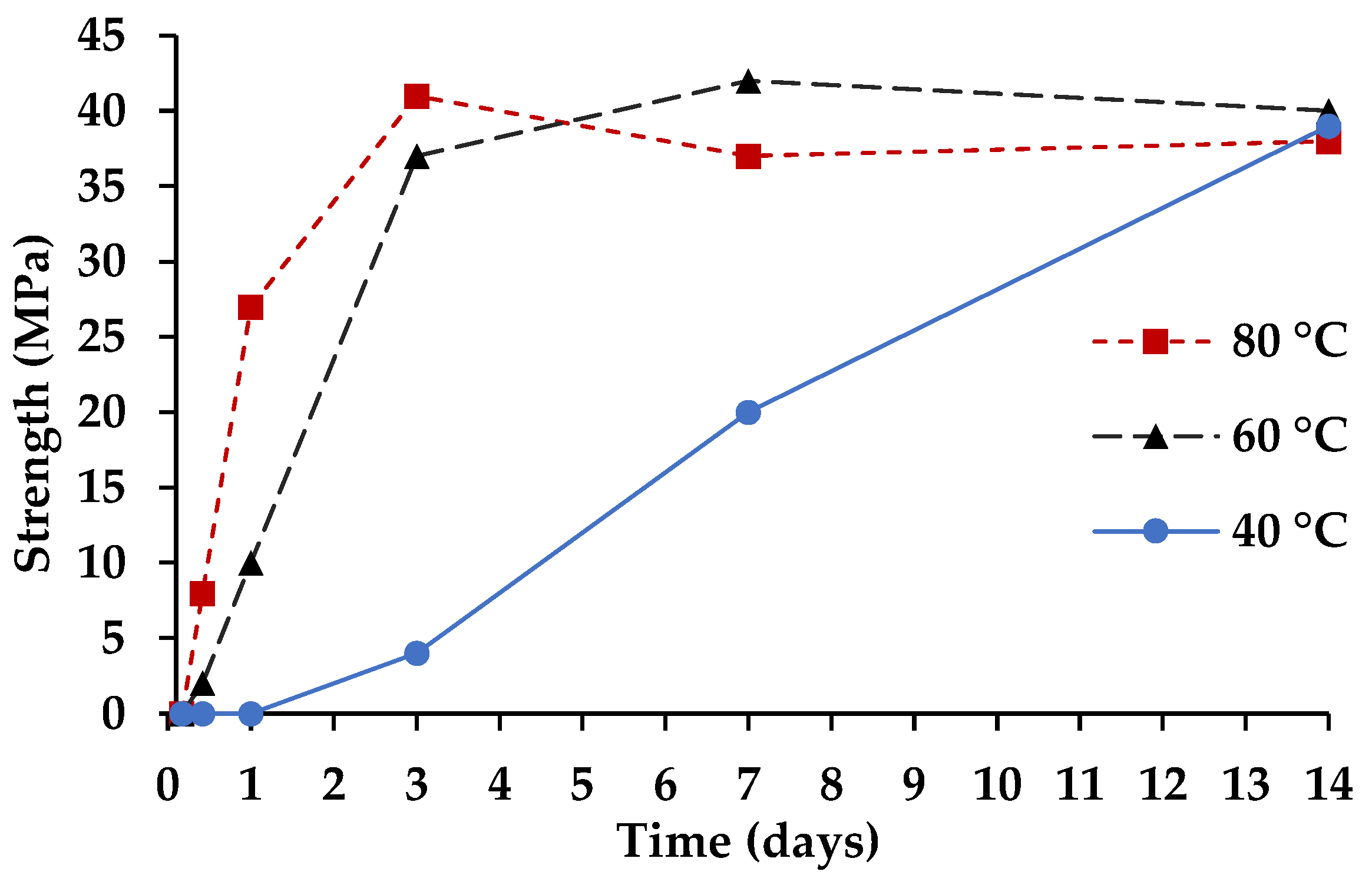
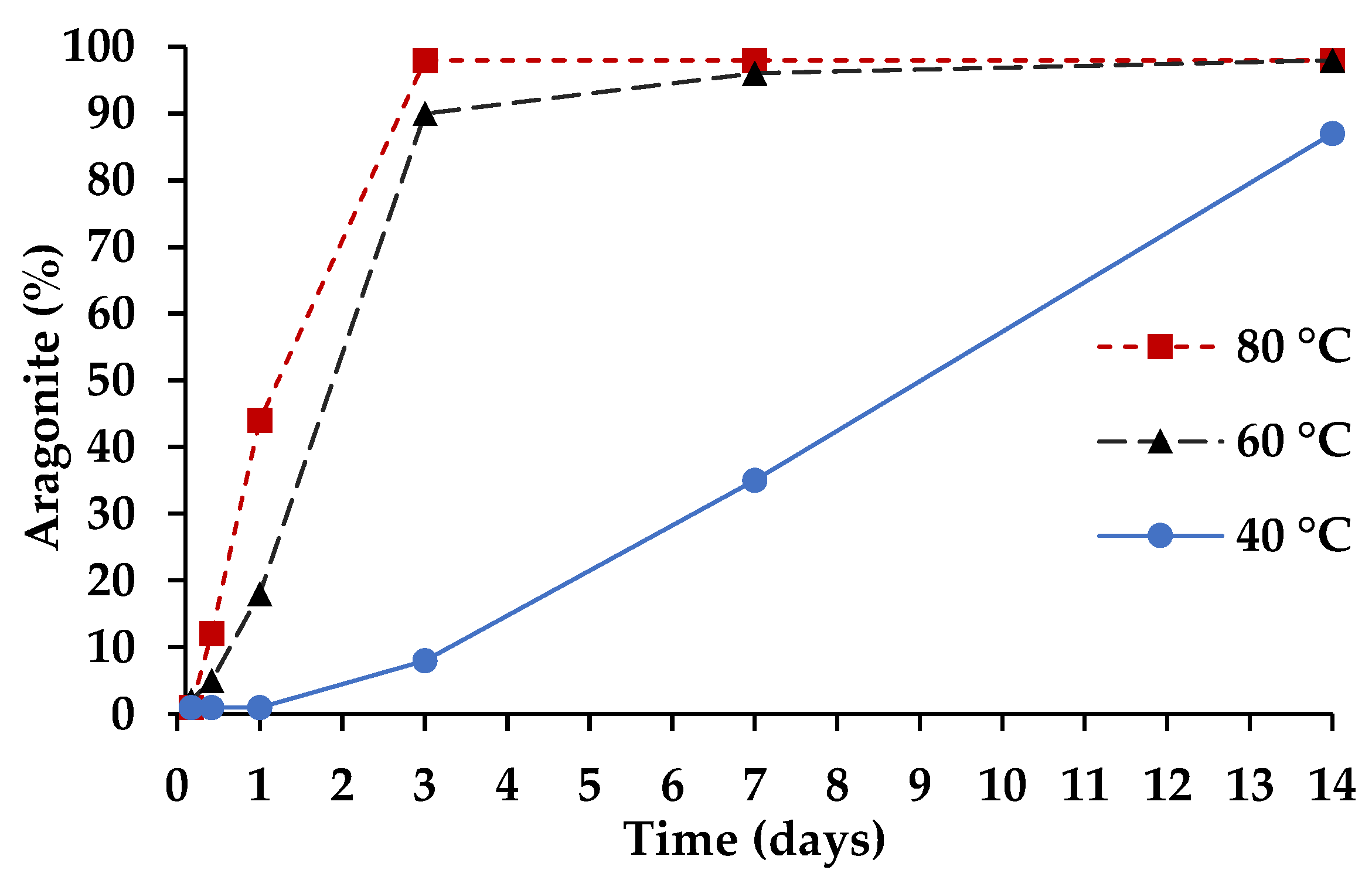
3.2. Cementitious Reaction
4. Discussion
4.1. Raw Materials Availability
4.2. Building Material Applications
4.3. CaCO3 Cement Manufacturing Energy and Carbon Footprint
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- IPCC. Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; IPCC: Geneva, Switzerland, 2014; p. 151. [Google Scholar]
- Akashi, O.; Hanaoka, T.; Matsuoka, Y.; Kainuma, M. A projection for global CO2 emissions from the industrial sector through 2030 based on activity level and technology changes. Energy 2011, 36, 1855–1867. [Google Scholar] [CrossRef]
- Damtoft, J.; Lukasik, J.; Herfort, D.; Sorrentino, D.; Gartner, E. Sustainable development and climate change initiatives. Cem. Concr. Res. 2008, 38, 115–127. [Google Scholar] [CrossRef]
- Gartner, E. Industrially interesting approaches to “low-CO2” cements. Cem. Concr. Res. 2004, 34, 1489–1498. [Google Scholar] [CrossRef]
- Gartner, E.; Hirao, H. A review of alternative approaches to the reduction of CO2 emissions associated with the manufacture of the binder phase in concrete. Cem. Concr. Res. 2015, 78, 126–142. [Google Scholar] [CrossRef]
- Hargis, C.W.; Kirchheim, A.P.; Monteiro, P.J.M.; Gartner, E.M. Early age hydration of calcium sulfoaluminate (synthetic ye’elimite, C4A3$) in the presence of gypsum and varying amounts of calcium hydroxide. Cem. Concr. Res. 2013, 48, 105–115. [Google Scholar] [CrossRef]
- Hargis, C.W.; Telesca, A.; Monteiro, P.J. Calcium sulfoaluminate (Ye’elimite) hydration in the presence of gypsum, calcite, and vaterite. Cem. Concr. Res. 2014, 65, 15–20. [Google Scholar] [CrossRef]
- Monteiro, P.J.; Clodic, L.; Battocchio, F.; Kanitpanyacharoen, W.; Chae, S.R.; Ha, J.; Wenk, H.-R. Incorporating carbon sequestration materials in civil infrastructure: A micro and nano-structural analysis. Cem. Concr. Compos. 2013, 40, 14–20. [Google Scholar] [CrossRef]
- Hargis, C.W. Advances in Sustainable Cements, Doctoral Dissertation; University of California: Berkeley, CA, USA, 2013. [Google Scholar]
- Oelkers, E.H.; Gislason, S.R.; Matter, J. Mineral Carbonation of CO2. Elements 2008, 4, 333–337. [Google Scholar] [CrossRef]
- Freas, R.C.; Hayden, J.S.; Pryor, C.A., Jr. Limestone and dolomite in Industrial Minerals & Rocks: Commodities, Markets, and Uses, 7th ed.; Society of Mining, Metallurgy and Exploration: Littleton, CO, USA, 2006. [Google Scholar]
- Morse, J.W.; Arvidson, R.S.; Lüttge, A. Calcium carbonate formation and dissolution. Chem. Rev. 2007, 107, 342–381. [Google Scholar] [CrossRef]
- Addadi, L.; Raz, S.; Weiner, S. Taking advantage of disorder: Amorphous calcium carbonate and its roles in biomineralization. Adv. Mater. 2003, 15, 959–970. [Google Scholar] [CrossRef]
- Fontaine, M.-L.; Combes, C.; Sillam, T.; Dechambre, G.; Rey, C. New Calcium Carbonate-Based Cements for Bone Reconstruction. Key Eng. Mater. 2005, 284–286, 105–108. [Google Scholar] [CrossRef]
- Combes, C.; Miao, B.; Bareille, R.; Rey, C. Preparation, physical–chemical characterisation and cytocompatibility of calcium carbonate cements. Biomaterials 2006, 27, 1945–1954. [Google Scholar] [CrossRef] [PubMed]
- Combes, C.; Tadier, S.; Galliard, H.; Fullana, S.G.; Charvillat, C.; Rey, C.; Auzely-Velty, R.; El Kissi, N. Rheological properties of calcium carbonate self-setting injectable paste. Acta Biomater. 2010, 6, 920–927. [Google Scholar] [CrossRef] [PubMed]
- Devenney, M.; Fernandez, M.; Chen, I.; Calas, G. Methods and Systems for Utilizing Carbide Lime. U.S. Patent 9,902,652 B2, 31 July 2013. [Google Scholar]
- Eloneva, S.; Teir, S.; Salminen, J.; Fogelholm, C.-J.; Zevenhoven, R. Steel Converter Slag as a Raw Material for Precipitation of Pure Calcium Carbonate. Ind. Eng. Chem. Res. 2008, 47, 7104–7111. [Google Scholar] [CrossRef]
- Lee, M.-G.; Kang, D.; Yoo, Y.; Jo, H.; Song, H.-J.; Park, J. Continuous and Simultaneous CO2 Absorption, Calcium Extraction, and Production of Calcium Carbonate Using Ammonium Nitrate. Ind. Eng. Chem. Res. 2016, 55, 11795–11800. [Google Scholar] [CrossRef]
- Udrea, I.; Capat, C.; Olaru, E.A.; Isopescu, R.; Mihai, M.; Mateescu, C.D.; Bradu, C. Vaterite Synthesis via Gas–Liquid Route under Controlled pH Conditions. Ind. Eng. Chem. Res. 2012, 51, 8185–8193. [Google Scholar] [CrossRef]
- Zeebe, R.E.; Wolf-Gladrow, D. CO2 in Seawater: Equilibrium, Kinetics, Isotopes; Elsevier Science: Amsterdam, The Netherlands, 2001; Volume 65. [Google Scholar]
- Fernandez, M.; Chen, I.; Lee, P.T.; Ginder-Vogel, M. Methods and Compositions Using Calcium Carbonate and Stabilizer. U.S. Patent 9,139,472 B2, 22 September 2015. [Google Scholar]
- Asia Market Information & Development Company. Chinese Markets for Calcium Carbide; Asia Market Information & Development Company: Rockville, MD, USA, 2012. [Google Scholar]
- Lutterotti, L.; Matthies, S.; Wenk, H.-R. MAUD: A friendly Java program for material analysis using diffraction. IUCR Newsl. CPD 1999, 21, 14–15. [Google Scholar]
- ASTM. C109/C109M-13: Standard Test Method for Compressive Strength of Hydraulic Cement Mortars (Using 2-in. or [50-mm] Cube Specimens); ASTM: West Conshohocken, PA, USA, 2013; p. 10. [Google Scholar]
- ASTM. C305-14: Standard Practice for Mechanical Mixing of Hydraulic Cement Pastes and Mortars of Plastic Consistency; ASTM: West Conshohocken, PA, USA, 2014; p. 3. [Google Scholar]
- Gehrke, N.; Cölfen, H.; Pinna, N.; Antonietti, A.M.; Nassif, N. Superstructures of Calcium Carbonate Crystals by Oriented Attachment. Cryst. Growth Des. 2005, 5, 1317–1319. [Google Scholar] [CrossRef]
- Hu, Q.; Zhang, J.; Teng, H.; Becker, U. Growth process and crystallographic properties of ammonia-induced vaterite. Am. Miner. 2012, 97, 1437–1445. [Google Scholar] [CrossRef]
- Trushina, D.; Bukreeva, T.V.; Kovalchuk, M.V.; Antipina, M.N. CaCO3 vaterite microparticles for biomedical and personal care applications. Mater. Sci. Eng. C 2014, 45, 644–658. [Google Scholar] [CrossRef]
- Popławski, D.; Hoffmann, J.; Hoffmann, K. Effect of carbonate minerals on the thermal stability of fertilisers containing ammonium nitrate. J. Therm. Anal. Calorim. 2016, 124, 1561–1574. [Google Scholar] [CrossRef]
- Bischoff, J.L. Kinetics of calcite nucleation: Magnesium ion inhibition and ionic strength catalysis. J. Geophys. Res. Space Phys. 1968, 73, 3315–3322. [Google Scholar] [CrossRef]
- Gutjahr, A.; Dabringhaus, H.; Lacmann, R. Studies of the growth and dissolution kinetics of the CaCO3 polymorphs calcite and aragonite II. The influence of divalent cation additives on the growth and dissolution rates. J. Cryst. Growth 1996, 158, 310–315. [Google Scholar] [CrossRef]
- Bischoff, J.L. Catalysis, inhibition, and the calcite-aragonite problem; II. The vaterite-aragonite transformation. Am. J. Sci. 1968, 266, 80–90. [Google Scholar] [CrossRef]
- Nielsen, M.H.; Aloni, S.; De Yoreo, J.J. In situ TEM imaging of CaCO3 nucleation reveals coexistence of direct and indirect pathways. Science 2014, 345, 1158–1162. [Google Scholar] [CrossRef]
- Sun, B.; Liu, Y.; Nie, Z.; Gao, F.; Wang, Z.; Cui, S. Exergy-based resource consumption analysis of cement clinker production using natural mineral and using calcium carbide sludge (CCS) as raw material in China. Int. J. Life Cycle Assess. 2020, 25, 667–677. [Google Scholar] [CrossRef]
- USG Survey. Mineral Commodity Summaries: Iron and Steel Slag; USGS: Lakewood, CO, USA, 2020.
- Ahmedzade, P.; Sengoz, B. Evaluation of steel slag coarse aggregate in hot mix asphalt concrete. J. Hazard. Mater. 2009, 165, 300–305. [Google Scholar] [CrossRef]
- Hewlett, P.C.; Soutsos, M.; Domone, P. Construction Materials: Their Nature and Behaviour, 5th ed.; Soutsos, M., Domone, P., Eds.; CRC Press: Boca Raton, FL, USA, 2017; ISBN 978-1498741101. [Google Scholar]
- EIA Carbon Dioxide Emissions Coefficients. Available online: https://www.eia.gov/environment/emissions/co2_vol_mass.php (accessed on 14 May 2021).
- EPA eGRID Data Explorer. Available online: https://www.epa.gov/egrid/data-explorer (accessed on 14 May 2021).
- Barcelo, L.; Kline, J.; Walenta, G.; Gartner, E. Cement and carbon emissions. Mater. Struct. 2013, 47, 1055–1065. [Google Scholar] [CrossRef]
- World Business Council for Sustainable Development. Cement Industry Energy and CO2 Performance, Getting the Numbers Right; CSI: New York, NY, USA, 2009. [Google Scholar]
- Pade, C.; Guimaraes, M. The CO2 uptake of concrete in a 100 year perspective. Cem. Concr. Res. 2007, 37, 1348–1356. [Google Scholar] [CrossRef]




| Oxide | Mass (%) |
|---|---|
| CaO | 70.0 |
| SiO2 | 2.4 |
| Al2O3 | 1.7 |
| MgO | 0.1 |
| Fe2O3 | 0.3 |
| SO3 | 0.7 |
| MnO | 0 |
| TiO2 | 0 |
| P2O5 | 0 |
| Cl | 0 |
| LOI 1 | 24.4 |
| Oxide | Weight (%) | Mineral | Weight (%) |
|---|---|---|---|
| CaO | 55.1 | Vaterite | 99.5 |
| SiO2 | 0 | Calcite | 0.5 |
| Al2O3 | 0 | ||
| MgO | 0.1 | ||
| Fe2O3 | 0 | ||
| SO3 | 0.4 | ||
| MnO | 0 | ||
| TiO2 | 0 | ||
| P2O5 | 0 | ||
| Cl | 0.2 | ||
| LOI 1 | 44.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hargis, C.W.; Chen, I.A.; Devenney, M.; Fernandez, M.J.; Gilliam, R.J.; Thatcher, R.P. Calcium Carbonate Cement: A Carbon Capture, Utilization, and Storage (CCUS) Technique. Materials 2021, 14, 2709. https://doi.org/10.3390/ma14112709
Hargis CW, Chen IA, Devenney M, Fernandez MJ, Gilliam RJ, Thatcher RP. Calcium Carbonate Cement: A Carbon Capture, Utilization, and Storage (CCUS) Technique. Materials. 2021; 14(11):2709. https://doi.org/10.3390/ma14112709
Chicago/Turabian StyleHargis, Craig W., Irvin A. Chen, Martin Devenney, Miguel J. Fernandez, Ryan J. Gilliam, and Ryan P. Thatcher. 2021. "Calcium Carbonate Cement: A Carbon Capture, Utilization, and Storage (CCUS) Technique" Materials 14, no. 11: 2709. https://doi.org/10.3390/ma14112709
APA StyleHargis, C. W., Chen, I. A., Devenney, M., Fernandez, M. J., Gilliam, R. J., & Thatcher, R. P. (2021). Calcium Carbonate Cement: A Carbon Capture, Utilization, and Storage (CCUS) Technique. Materials, 14(11), 2709. https://doi.org/10.3390/ma14112709






