Synthetic, Mesomorphic, and DFT Investigations of New Nematogenic Polar Naphthyl Benzoate Ester Derivatives
Abstract
1. Introduction
2. Experiment
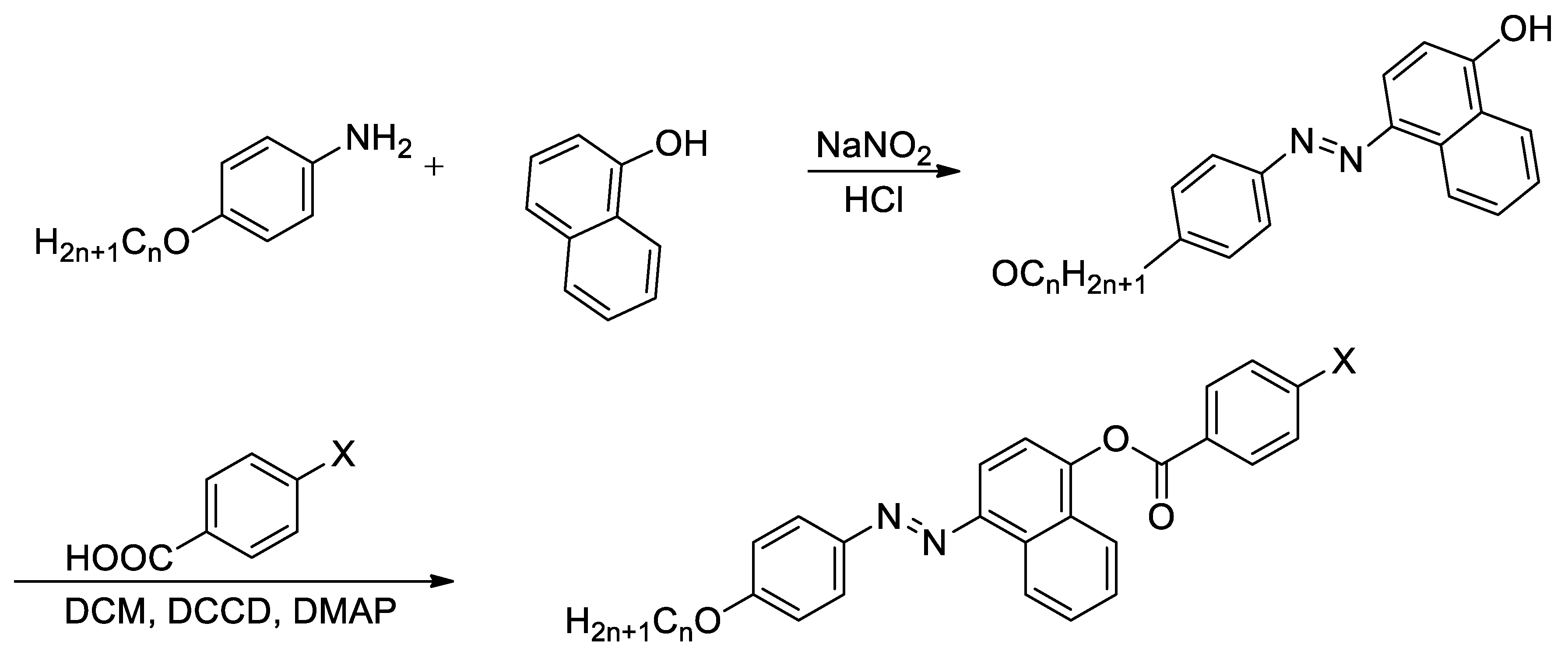
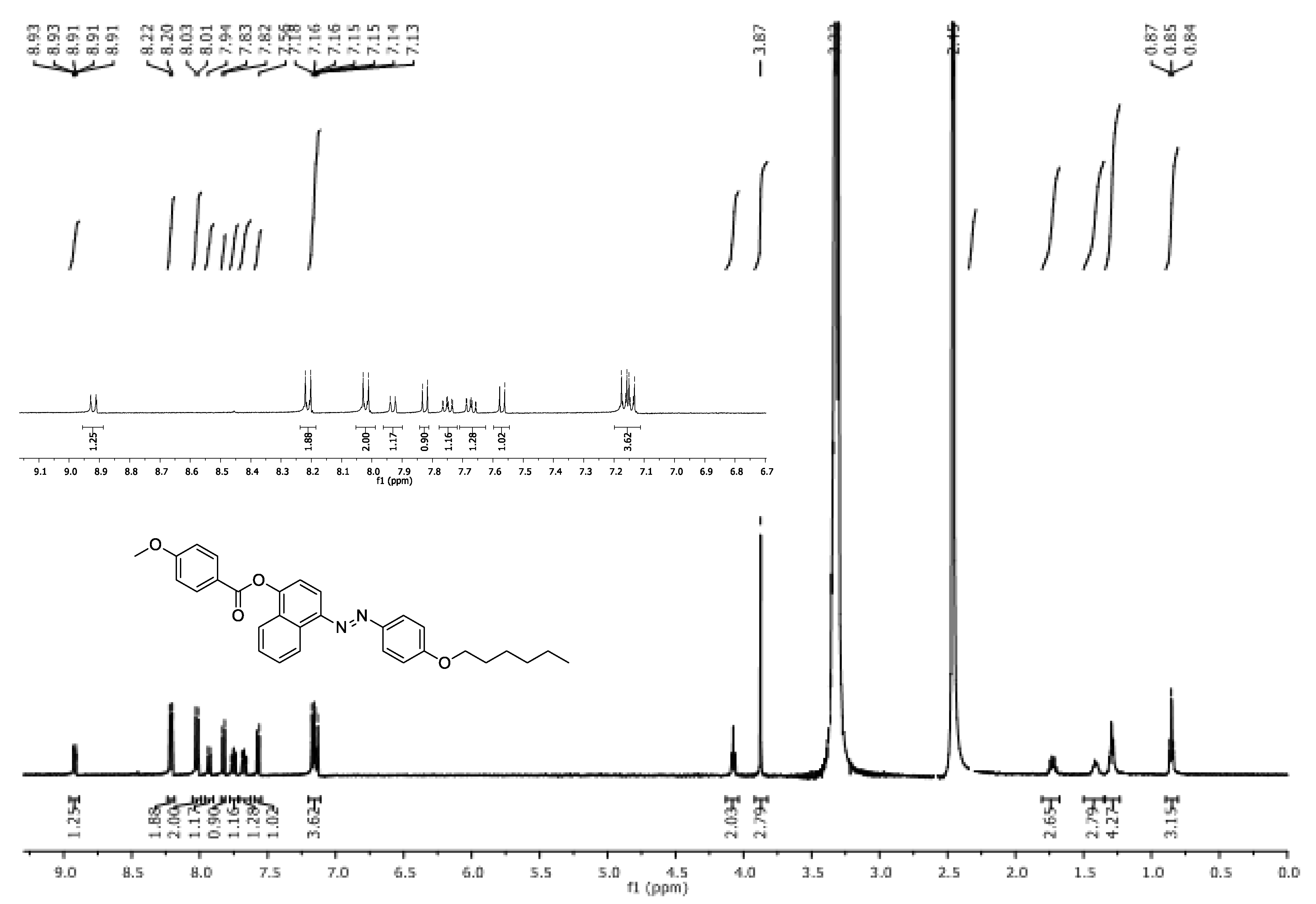
Synthesis
3. Results and Discussion
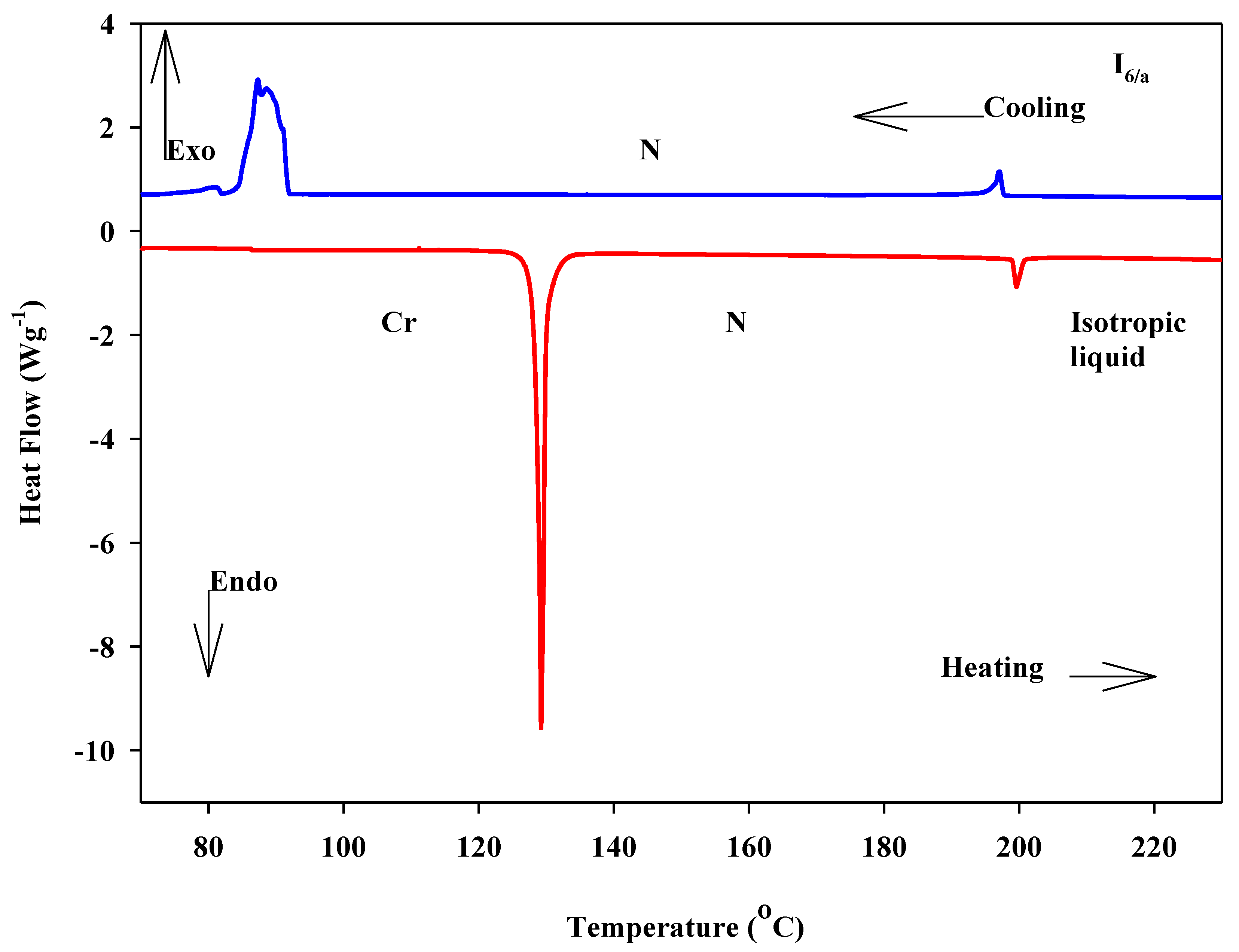
3.1. Mesomorphic and Optical Behaviour
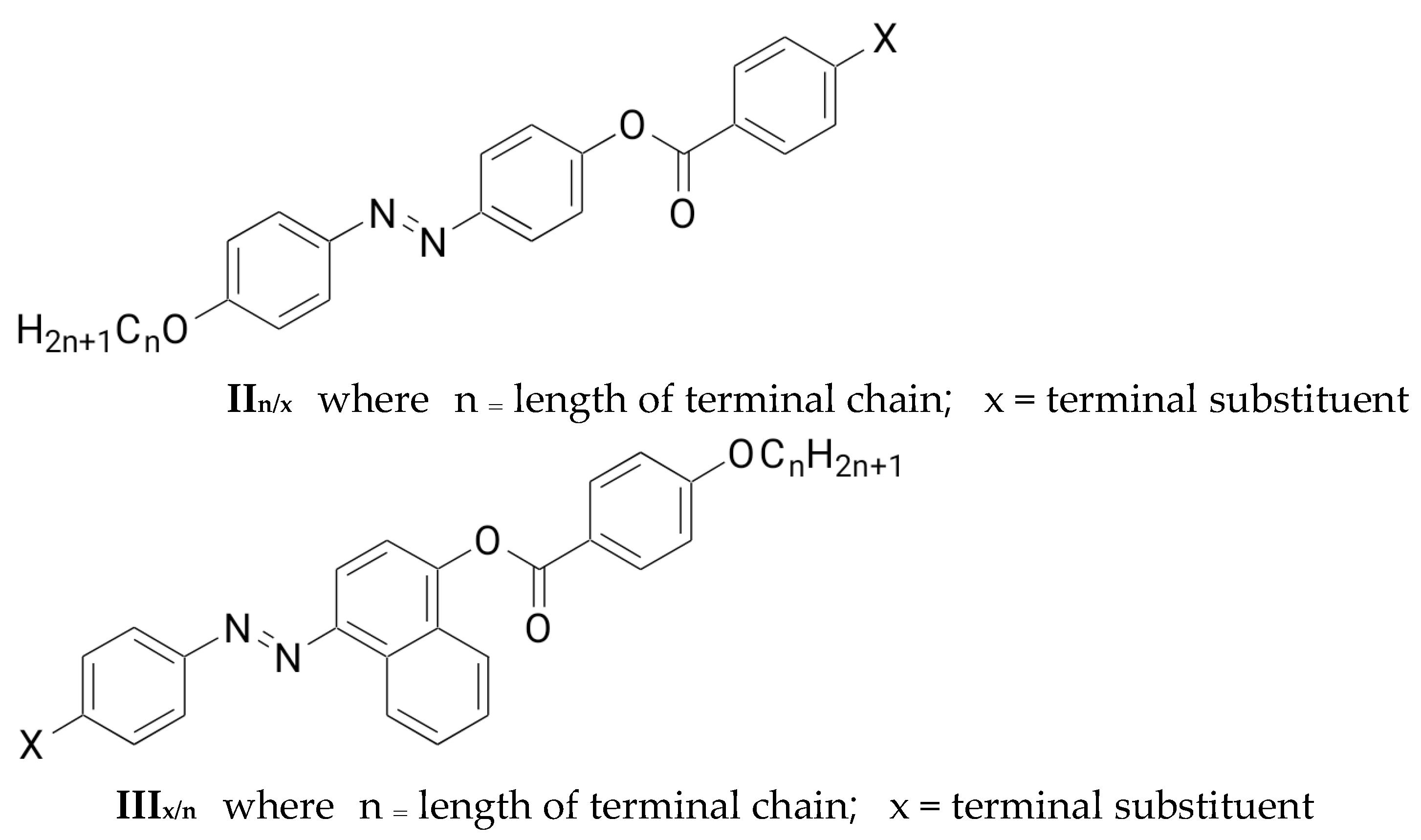
3.2. Comparison of the Investigated Derivatives (In/x) with Previously Prepared Series
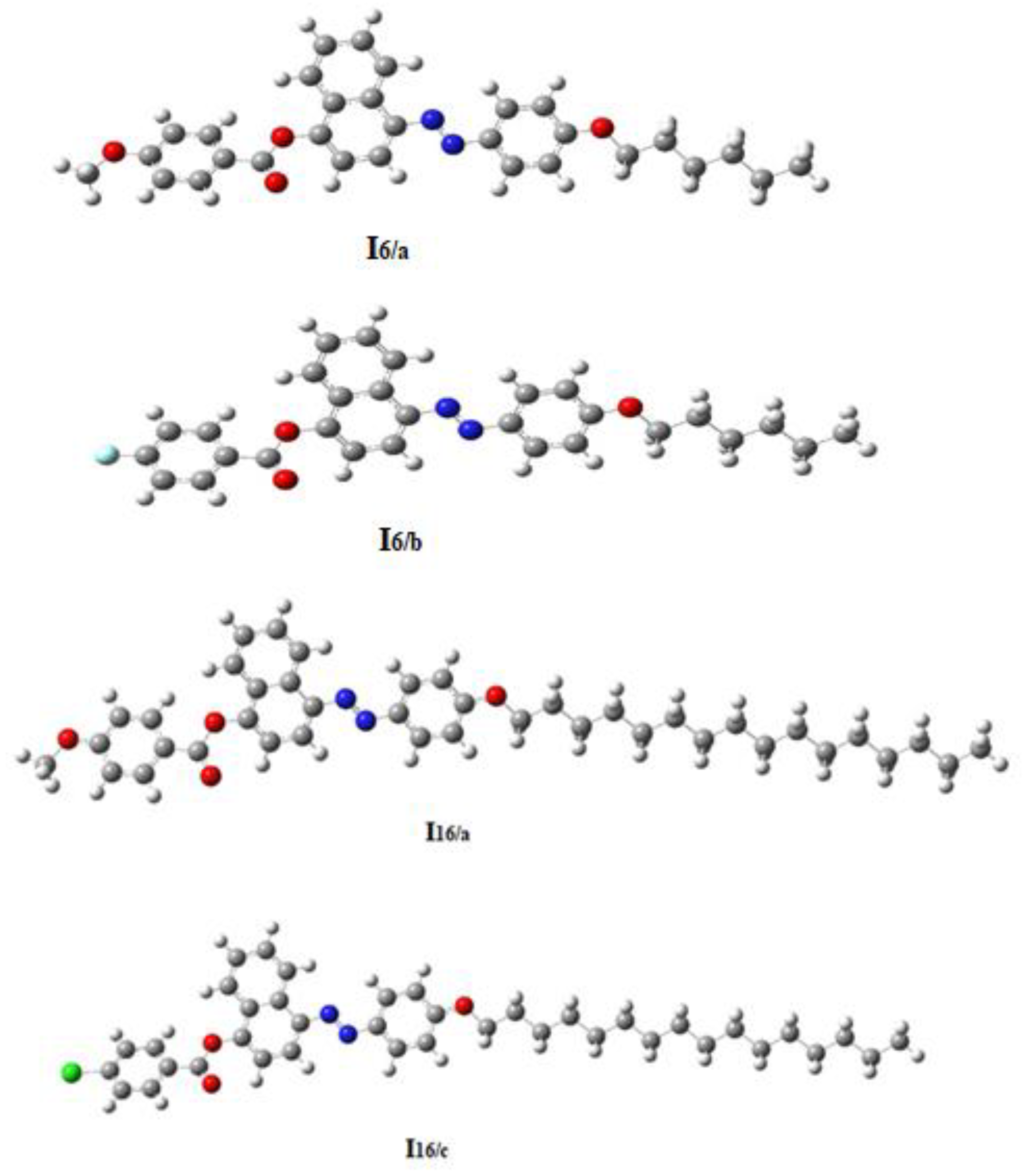
3.3. Geometrical Structures and DFT Investigations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Geelhaar, T.; Griesar, K.; Reckmann, B. 125 Years of Liquid Crystals-A Scientific Revolution in the Home. Angew. Chem. Int. Ed. 2013, 52, 8798–8809. [Google Scholar] [CrossRef]
- Luo, Z.; Peng, F.; Chen, H.; Hu, M.; Li, J.; An, Z.; Wu, S.-T. Fast-response liquid crystals for high image quality wearable displays. Opt. Mater. Express 2015, 5, 603–610. [Google Scholar] [CrossRef]
- Aleksandriiskii, V.; Novikov, I.; Kuvshinova, S.; Burmistrov, V.; Koifman, O. Dielectric, optical and orientational properties of liquid crystalline 4-alkyloxy-4′-cyanoazoxybenzenes and 4-alkyloxy-4′-cyanoazobenzenes. J. Mol. Liq. 2016, 223, 1270–1276. [Google Scholar] [CrossRef]
- Dave, J.S.; Bhatt, H.S. Synthesis of liquid crystals with lateral methyl group and study of their mesomorphic properties. Mol. Cryst. Liq. Cryst. 2012, 562, 1–9. [Google Scholar] [CrossRef]
- Thaker, B.T.; Kanojiya, J.B.; Tandel, R.S. Effects of Different Terminal Substituents on the Mesomorphic Behavior of Some Azo-Schiff Base and Azo-Ester-Based Liquid Crystals. Mol. Cryst. Liq. Cryst. 2010, 528, 120–137. [Google Scholar] [CrossRef]
- Dave, J.S.; Menon, M. Azomesogens with a heterocyclic moiety. Bull. Mater. Sci. 2000, 23, 237–238. [Google Scholar] [CrossRef]
- Vora, R.A.; Prajapati, A.K. Azomesogens with 1,2,4-trisubstituted benzene moiety. Bull. Mater. Sci. 2002, 25, 355–358. [Google Scholar] [CrossRef]
- Prajapati, A.K.; Pandya, H.M. Azomesogens with methoxyethyl tail: Synthesis and characterization. J. Chem. Sci. 2005, 117, 255–261. [Google Scholar] [CrossRef]
- Al-Hamdani, U.J.; Gassim, T.E.; Radhy, H.H. Synthesis and Characterization of Azo Compounds and Study of the Effect of Substituents on Their Liquid Crystalline Behavior. Molecules 2010, 15, 5620–5628. [Google Scholar] [CrossRef] [PubMed]
- Yeap, G.Y.; Ha, S.T.; Lim, P.L.; Boey, P.L.; Ito, M.M.; Sanehisa, S.; Youhei, Y. Synthesis, physical and mesomorphic properties of Schiff’s base esters containing ortho-, meta-and para-substituents in benzylidene-4′-alkanoyloxyanilines. Liq. Cryst. 2006, 33, 205–211. [Google Scholar] [CrossRef]
- Bhatt, H.S.; Patel, P.D.; Dave, J.S. Study of Mixed Mesomorphism in Binary Systems of Azo-Ester Mesogens with Structurally Dissimilar Nonmesogenic as Well as Mesogenic Ester Homologues. Mol. Cryst. Liq. Cryst. 2013, 587, 80–91. [Google Scholar] [CrossRef]
- Naoum, M.M.; Fahmi, A.A.; Abaza, A.H.; Saad, G.R. Effect of exchange of terminal substituents on the mesophase behaviour of some azo/ester compounds. Liq. Cryst. 2014, 41, 1559–1568. [Google Scholar] [CrossRef]
- Ha, S.-T.; Ong, L.-K.; Wong, J.P.-W.; Yeap, G.-Y.; Lin, H.-C.; Ong, S.-T.; Koh, T.-M. Mesogenic Schiff’s base ether with dimethylamino end group. Phase Transit. 2009, 82, 387–397. [Google Scholar] [CrossRef]
- Ichimura, K. Photoalignment of Liquid-Crystal Systems. Chem. Rev. 2000, 100, 1847–1874. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, T. Photomodulation of liquid crystal orientations for photonic applications. J. Mater. Chem. 2003, 13, 2037–2057. [Google Scholar] [CrossRef]
- Ikeda, T.; Wu, Y. Photoinduced alignment behavior of polymer liquid crystals containing azobenzene moieties in the side chain. Pure Appl. Chem. 1999, 71, 2131–2136. [Google Scholar] [CrossRef][Green Version]
- Eich, M.; Wendorff, J.H.; Reck, B.; Ringsdorf, H. Reversible digital and holographic optical storage in polymeric liquid crystals. Die Makromol. Chem. Rapid Commun. 1987, 8, 59–63. [Google Scholar] [CrossRef]
- Eich, M.; Wendorff, J.H. Erasable holograms in polymeric liquid crystals. Die Makromol. Chem. Rapid Commun. 1987, 8, 467–471. [Google Scholar] [CrossRef]
- Anderle, K.; Birenheide, R.; Werner, M.J.A.; Wendorff, J.H. Molecular addressing? Studies on light-induced reorientation in liquid-crystalline side chain polymers. Liq. Cryst. 1991, 9, 691–699. [Google Scholar] [CrossRef]
- Kato, T.; Mizoshita, N.; Kishimoto, K. Functional Liquid-Crystalline Assemblies: Self-Organized Soft Materials. Angew. Chem. Int. Ed. 2006, 45, 38–68. [Google Scholar] [CrossRef]
- Ikeda, T.; Mamiya, J.-I.; Yu, Y. Photomechanics of Liquid-Crystalline Elastomers and Other Polymers. Angew. Chem. Int. Ed. 2007, 46, 506–528. [Google Scholar] [CrossRef]
- Kelly, S.M. Smectic liquid crystals. VIII. Some new laterally substituted smectic C compounds. Liq. Cryst. 1989, 5, 171–175. [Google Scholar] [CrossRef]
- Kelly, S.M. Ferroelectric Liquid Crystals. Part 9. Laterally substituted phenyl benzoates incorporating a trans-1, 4-disubstituted cyclohexane ring. Helv. Chim. Acta 1989, 72, 594–607. [Google Scholar] [CrossRef]
- Kelly, S.M.; Buchecker, R.; Fromm, H.; Schadt, M. Ferroelectric liquid crystals part I. The synthesis and liquid crystal transition temperatures of some new smectic c materials. Ferroelectrics 1988, 85, 385–392. [Google Scholar] [CrossRef]
- Leenhouts, F.; Fuenfschilling, J.; Buchecker, R.; Kelly, S.M. Ferroelectric smectic C liquid crystal mixtures: Relationship between molecular structure, ferroelectricity and electro-optic response. Liq. Cryst. 1989, 5, 1179–1186. [Google Scholar] [CrossRef]
- Ahmed, H.; Khushaim, M.S. Nematogenic Laterally Substituted Supramolecular H-Bonded Complexes Based on Flexible Core. Crystals 2020, 10, 878. [Google Scholar] [CrossRef]
- Blatch, A.E.; Luckhurst, G.R. The liquid crystal properties of symmetric and non-symmetric dimers based on the azobenzene mesogenic group. Liq. Cryst. 2000, 27, 775–787. [Google Scholar] [CrossRef]
- Prasad, V. Liquid crystalline compounds with V-shaped molecular structures: Synthesis and characterization of new azo compounds. Liq. Cryst. 2001, 28, 145–150. [Google Scholar] [CrossRef]
- Prajapati, A.K.; Varia, M.C. H-shaped symmetrical twin liquid crystalline compounds with polar-terminal substituents. Liq. Cryst. 2013, 40, 1151–1158. [Google Scholar] [CrossRef]
- Podruczna, M.; Hofmańska, A.; Niezgoda, I.; Pociecha, D.; Galewski, Z. Influence of terminal groups on liquid-crystalline polymorphism of selected azobenzene derivatives. Liq. Cryst. 2013, 41, 113–125. [Google Scholar] [CrossRef]
- Nessim, R.I.; Naoum, M.M.; Mohamed, S.Z.; Nessim, M.I. Effect of molecular structure on the phase behaviour of some liquid crystalline compounds and their mixtures XIII. 4-(4-Substituted phenylazo)phenyl 4-alkoxybenzoates. Liq. Cryst. 2004, 31, 649–654. [Google Scholar] [CrossRef]
- Naoum, M.M.; Fahmi, A.A.; Alaasar, M.A. Supramolecular Hydrogen-Bonded Liquid Crystals Formed from 4-(4′-Pyridylazophenyl)-4″-alkoxy Benzoates and 4-Substituted Benzoic Acids. Mol. Cryst. Liq. Cryst. 2008, 487, 74–91. [Google Scholar] [CrossRef]
- Thaker, B.; Kanojiya, J. Mesomorphic properties of liquid crystalline compounds with biphenyl moiety containing azo-ester, azo-cinnamate central linkages and different terminal group. Liq. Cryst. 2011, 38, 1035–1055. [Google Scholar] [CrossRef]
- Debnath, S.; Mohiuddin, G.; Turlapati, S.; Begum, N.; Sarkar, D.D.; Rao, V.N. Nematic phases in achiral unsymmetrical four-ring bent-core azo compounds possessing strongly polar cyano and nitro moieties as end substituents: Synthesis and characterization. Dyes Pigments 2013, 99, 447–455. [Google Scholar] [CrossRef]
- Chauhan, B.; Doshi, A.A.; Doshi, A. Synthesis and Study of Novel Liquid Crystalline Homologous Series: 4-(4′-n-Alkoxy Benzoyloxy)-3-Methoxy Phenyl azo-3″, 4″Dichlorobenzenes. Mol. Cryst. Liq. Cryst. 2013, 570, 84–91. [Google Scholar] [CrossRef]
- Ahmed, H.A.; Naoum, M.M.; Saad, G.R. Effect of alkoxy-chain length proportionation on the mesophase behaviour of terminally di-substituted phenylazo phenyl benzoates. Liq. Cryst. 2013, 40, 914–921. [Google Scholar] [CrossRef]
- Jadeja, U.H.; Patel, R.B. Study of mesomorphism dependence on molecular flexibility of an azoester series containing a napthyl unit. Mol. Cryst. Liq. Cryst. 2016, 637, 10–18. [Google Scholar] [CrossRef]
- Naoum, M.M.; Fahmi, A.A.; Ahmed, N.H.; Saad, G.R. The effect of inversion of the ester group on the mesophase behaviour of some azo/ester compounds. Liq. Cryst. 2015, 42, 1298–1308. [Google Scholar] [CrossRef]
- Patel, D.H.; Doshi, A.A.; Prajapati, H.R. Synthesis of novel azoester homologous series of liquid crystalline behavior and the study of mesomorphism dependence on lateral substitution of middle phenyl ring. Mol. Cryst. Liq. Cryst. 2016, 624, 69–76. [Google Scholar] [CrossRef]
- Karim, R.; Sheikh, R.K.; Yahya, R.; Salleh, N.M.; Lo, K.M.; Mahmud, H.E. The effect of terminal substituents on crystal structure, mesophase behaviour and optical property of azo-ester linked materials. Liq. Cryst. 2016, 43, 1862–1874. [Google Scholar] [CrossRef]
- Al-Mutabagani, L.A.; Alshabanah, L.A.; Naoum, M.M.; Hagar, M.; Ahmed, H.A. Experimental and Computational Approaches of Newly Polymorphic Supramolecular H-Bonded Liquid Crystal Complexes. Front. Chem. 2020, 8, 8. [Google Scholar] [CrossRef] [PubMed]
- Khushaim, M.S.; Alalawy, H.H.; Naoum, M.M.; Ahmed, H.A. Experimental and computational simulations of nematogenic liquid crystals based on cinnamic acid in pure and mixed state. Liq. Cryst. 2021. [Google Scholar] [CrossRef]
- El-Atawy, M.A.; Naoum, M.M.; Al-Zahrani, S.A.; Ahmed, H.A. New Nitro-Laterally Substituted Azomethine Derivatives; Synthesis, Mesomorphic and Computational Characterizations. Molecules 2021, 26, 1927. [Google Scholar] [CrossRef] [PubMed]
- Al-Mutabagani, L.; Alshabanah, L.; Ahmed, H.; Alalawy, H.; Al Alwani, M. Synthesis, Mesomorphic and Computational Characterizations of Nematogenic Schiff Base Derivatives in Pure and Mixed State. Molecules 2021, 26, 2038. [Google Scholar] [CrossRef]
- Ahmed, H.A.; El-atawy, M.A. Synthesis, Mesomorphic and Geometrical approaches of New non-symmetrical System based on central Naphthalene moiety. Liq. Cryst. 2021. [Google Scholar] [CrossRef]
- Pytlarczyk, M.; Dmochowska, E.; Czerwiński, M.; Herman, J. Effect of lateral substitution by chlorine and fluorine atoms of 4-alkyl-p-terphenyls on mesomorphic behaviour. J. Mol. Liq. 2019, 292, 111379. [Google Scholar] [CrossRef]
- Sundaram, S.; Jayaprakasam, R.; Dhandapani, M.; Senthil, T.; Vijayakumar, V. Theoretical (DFT) and experimental studies on multiple hydrogen bonded liquid crystals comprising between aliphatic and aromatic acids. J. Mol. Liq. 2017, 243, 14–21. [Google Scholar] [CrossRef]
- Praveen, P.L.; Ojha, D.P. Computational analysis of molecular properties and spectral characteristics of cyano-containing liquid crystals: Role of alkyl chains. Phys. Rev. E 2011, 83, 51710. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.F.; Senior, S.; Elatawy, M.A.K.; El-Sadany, S.K.; Hamed, E.A. DFT calculations of 2,4,6-trinitrophenylbenzoate derivatives: Structure, ground state properties and spectral properties. J. Mol. Struct. 2011, 1006, 303–311. [Google Scholar] [CrossRef]
- El-Atawy, M.A.; Hegazi, A.H.; Al Khalaf, M.; Amer, A. The structure elucidation of the isomeric mixture of 3-[L-threo-2,3,4-tri -hydroxy-1-(phenyl-hydrazono)butyl] quinoxalin-2(1H)-one in dimethyl sulfoxide solution revisited: Experimental and theoretical study. Struct. Chem. 2020, 31, 1065–1072. [Google Scholar] [CrossRef]
- Omar, A.Z.; Mahmoud, M.N.; El-Sadany, S.K.; Hamed, E.A.; El-Atawy, M.A. A combined experimental and DFT investigation of mono azo thiobarbituric acid based chalcone disperse dyes. Dyes Pigment. 2021, 185, 108887. [Google Scholar] [CrossRef]
- El-Atawy, M.A.; Omar, A.Z.; Hagar, M.; Shashira, E.M. Transalkylidation reaction: Green, catalyst-free synthesis of thiosemicarbazones and solving the NMR conflict between their acyclic structure and intramolecular cycloaddition products. Green Chem. Lett. Rev. 2019, 12, 364–376. [Google Scholar] [CrossRef]
- Kohout, M.; Svoboda, J.; Novotná, V.; Pociecha, D. Non-symmetrical bent-shaped liquid crystals based on a laterally substituted naphthalene central core with four ester groups. Liq. Cryst. 2011, 38, 1099–1110. [Google Scholar] [CrossRef]
- Sultan, A.A.; Fahmi, A.A.; Saad, G.R.; Naoum, M.M. Effect of orientation of lateral fluorine atom on the mesophase behaviour of azo/ester molecules with terminal naphthyl group. Liq. Cryst. 2019, 46, 2322–2333. [Google Scholar] [CrossRef]
- Kozmík, V.; Kuchař, M.; Svoboda, J.; Novotná, V.; Glogarová, M.; Baumeister, U.; Diele, S.; Pelzl, G. Laterally substituted naphthalene-2,7-diol-based bent-shaped liquid crystals. Liq. Cryst. 2005, 32, 1151–1160. [Google Scholar] [CrossRef]
- Svoboda, J.; Novotná, V.; Kozmík, V.; Glogarová, M.; Weissflog, W.; Diele, S.; Pelzl, G. A novel type of banana liquid crystals based on 1-substituted naphthalene-2,7-diol cores. J. Mater. Chem. 2003, 13, 2104–2110. [Google Scholar] [CrossRef]
- Mohammady, S.Z.; Nessim, R.I.; Shehab, O.R.; Naoum, M.M. Effect of steric factors on mesomorphic stability I. 4-(4-Substituted phenylazo)-1-naphthyl 4-alkoxybenzoates. Liq. Cryst. 2005, 32, 477–482. [Google Scholar] [CrossRef]
- Naoum, M.M.; Metwally, N.H.; Eltawab, M.M.A.; Ahmed, H.A. Polarity and steric effect of the lateral substituent on the mesophase behaviour of some newly prepared liquid crystals. Liq. Cryst. 2015, 42, 1351–1369. [Google Scholar] [CrossRef]
- Demus, D.; Goodby, J.W.; Gray, G.W.; Spiess, H.W.; Vill, V. Handbook of Liquid Crystals, Volume 2A: Low Molecular Weight Liquid Crystals I: Calamitic Liquid Crystals; John Wiley & Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- Hird, M.; Toyne, K.J.; Gray, G.W.; McDonnell, D.G.; Sage, I.C. The relationship between molecular structure and mesomorphic properties of 2,2′- and 3,2′-difluoroterphenyls synthesized by palladium-catalysed cross-couplings. Liq. Cryst. 1995, 18, 1–11. [Google Scholar] [CrossRef]
- Sugiura, H.; Saklrai, Y.; Masuda, Y.; Takeda, H.; Kusabayashi, S.; Takenaka, S. The substituent effect on mesomorphic properties of 4-octyloxyphenyl 4-(4-R-3-nitrobenzoyloxy)benzoates and 4-(4-octyloxybenzoyloxy)phenyl 4-R-3-nitrobenzoates. Liq. Cryst. 1991, 9, 441–450. [Google Scholar] [CrossRef]
- Masuda, Y.; Sakurai, Y.; Sugiura, H.; Miyake, S.; Takenaka, S.; Kusabayashi, S. The effect of a lateral substituent on the mesomorphic properties of 4-cyanophenyl 4-(4-alkyloxybenzoyloxy)benzoates. Liq. Cryst. 1991, 10, 623–634. [Google Scholar] [CrossRef]
- Dabrowski, R.; Bezborodov, V.S.; Lapanik, V.J.; Dziaduszek, J.; Czuprynski, K. Mesomorphic properties of phenyl 4-(5-alkyl-1,3,2-dioxaborin-2-yl) benzoates Influence of terminal and lateral substitution. Liq. Cryst. 1995, 18, 213–218. [Google Scholar] [CrossRef]
- Adachi, T.; Saitoh, H.; Yamamura, Y.; Hishida, M.; Ueda, M.; Ito, S.; Saito, K. Universality of Molten State of Alkyl Chain in Liquid-Crystalline Mesophases: Smectic E Phase of 6-Alkyl-2-phenylazulene. Bull. Chem. Soc. Jpn. 2013, 86, 1022–1027. [Google Scholar] [CrossRef]
- Yamamura, Y.; Murakoshi, T.; Iwagaki, S.; Osiecka, N.; Saitoh, H.; Hishida, M.; Saito, K. Structure and molecular packing in smectic B Cr and A d phases of Schiff base liquid crystal compounds through the analyses of layer spacing, entropy and crystal structure. Phys. Chem. Chem. Phys. 2017, 19, 19434–19441. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.-C.; Lu, Z.; Henderson, P.A.; Achard, M.F.; Mahmood, W.A.K.; Yeap, G.-Y.; Imrie, C.T. Cholesteryl-based liquid crystal dimers containing a sulfur–sulfur link in the flexible spacer. Liq. Cryst. 2012, 39, 259–268. [Google Scholar] [CrossRef]
- Attard, G.S.; Imrie, C.T.; Karasz, F.E. Low molar mass liquid-crystalline glasses: Preparation and properties of the alpha-(4-cyanobiphenyl-4′-oxy)-.omega.-(1-pyreniminebenzylidene-4′-oxy)alkanes. Chem. Mater. 1992, 4, 1246–1253. [Google Scholar] [CrossRef]
- Attard, G.S.; Imrie, C.T. Liquid-crystalline and glass-forming dimers derived from 1-aminopyrene. Liq. Cryst. 1992, 11, 785–789. [Google Scholar] [CrossRef]
- Imrie, C.T.; Karasz, F.E.; Attard, G.S. Comparison of the mesogenic properties of monomeric, dimeric, and side-chain polymeric liquid crystals. Macromolecules 1993, 26, 545–550. [Google Scholar] [CrossRef]
- Donaldson, T.; Staesche, H.; Lu, Z.; Henderson, P.; Achard, M.; Imrie, C. Symmetric and non-symmetric chiral liquid crystal dimers. Liq. Cryst. 2010, 37, 1097–1110. [Google Scholar] [CrossRef]
- Henderson, P.A.; Imrie, C.T. Methylene-linked liquid crystal dimers and the twist-bend nematic phase. Liq. Cryst. 2011, 38, 1407–1414. [Google Scholar] [CrossRef]
- Nafee, S.S.; Hagar, M.; Ahmed, H.A.; Alhaddad, O.; El-Shishtawy, R.M.; Raffah, B.M. New two rings Schiff base liquid crystals; ball mill synthesis, mesomorphic, Hammett and DFT studies. J. Mol. Liq. 2020, 299, 112161. [Google Scholar] [CrossRef]
- Popoola, S.A.; Almohammedi, A.R.; Haruna, K. Spectroscopic and DFT evaluation of the positional effect of amino group on the properties of aminobenzenesulphonic acid: Solvents interactions. Chem. Pap. 2021, 75, 2775–2789. [Google Scholar] [CrossRef]
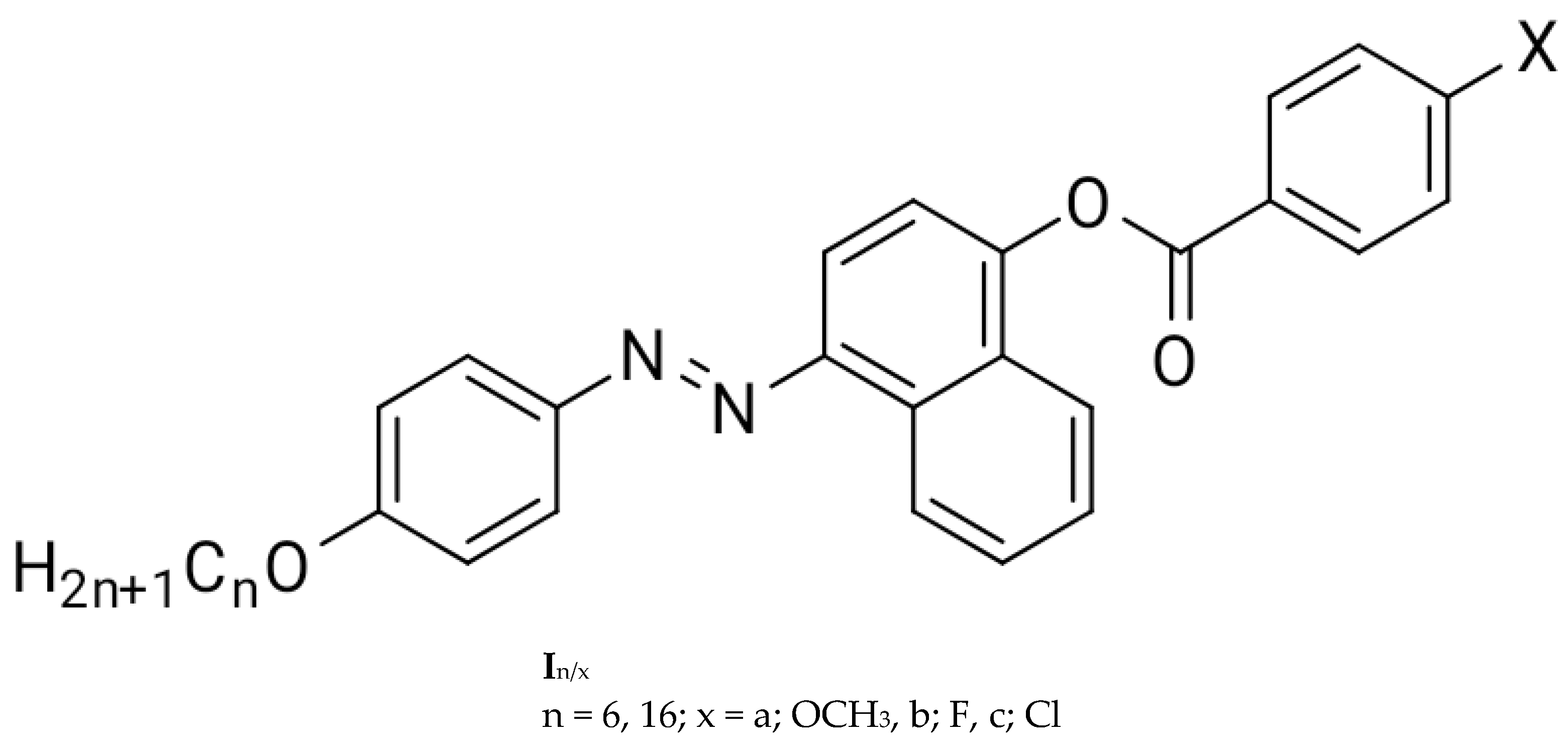

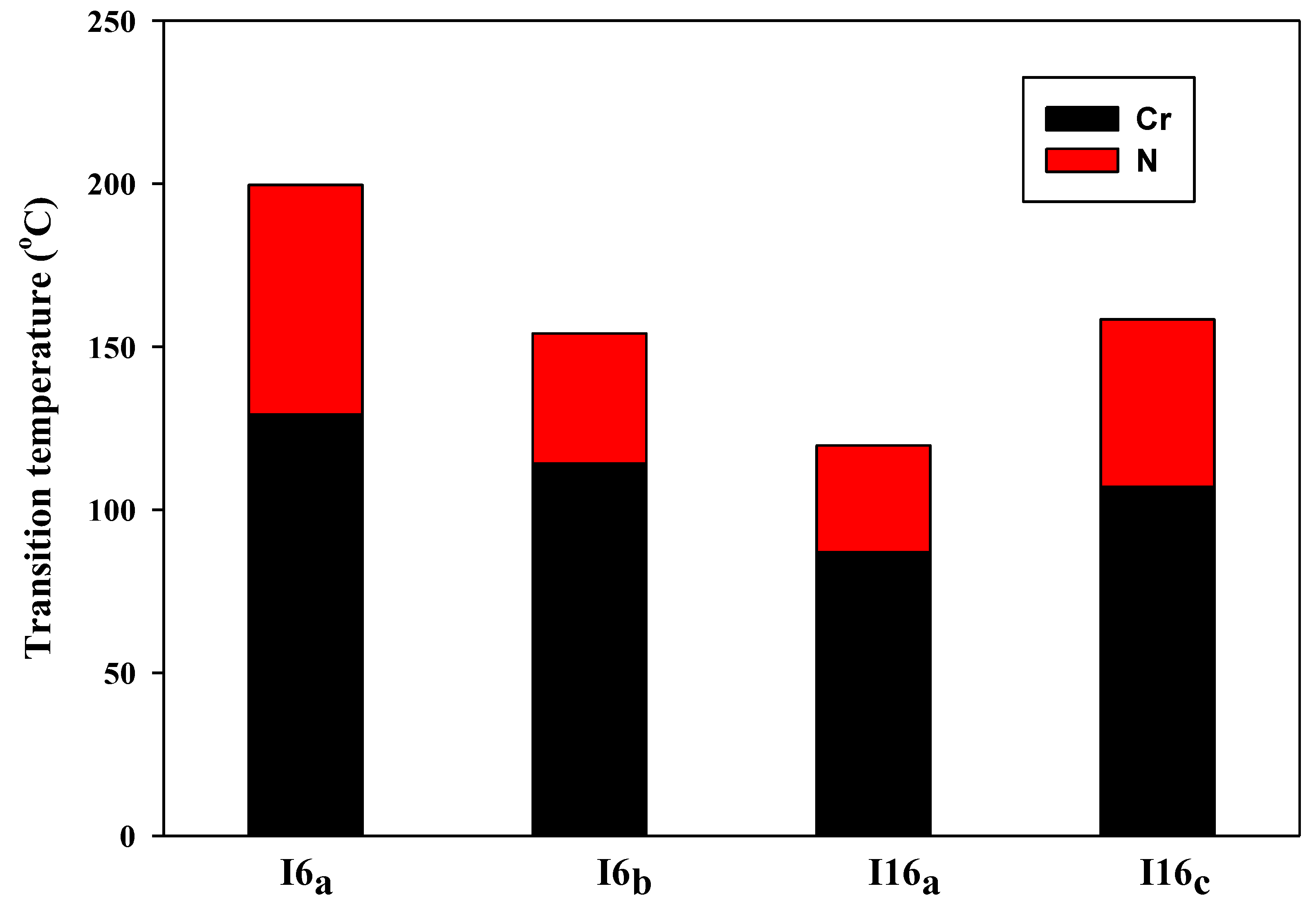
| Compound. | X | TCrystal–Nematic | ΔHCrystal–Nematic | TNematic–Iso | ΔHNematic–Iso | ΔSNematic–Iso/R | ΔT |
|---|---|---|---|---|---|---|---|
| I6/a | –OCH3 | 129.2 | 45.78 | 199.6 | 1.93 | 0.49 | 70.4 |
| I6/b | –F | 114.1 | 39.35 | 154.1 | 1.19 | 0.34 | 40.0 |
| I16/a | –OCH3 | 87.00 | 36.38 | 119.7 | 2.36 | 0.72 | 32.7 |
| I16/c | –Cl | 107.1 | 36.96 | 158.4 | 2.30 | 0.64 | 51.3 |
| Comp. | ZPE (Kcal/Mol) | Thermal Energy (Kcal/Mol) | Enthalpy (Kcal/Mol) | Gibbs Free Energy (Kcal/Mol) | Entropy (Cal Mol/k) |
|---|---|---|---|---|---|
| I6/a | 339.142 | 359.971 | 360.563 | 294.367 | 222.023 |
| I6/b | 313.606 | 333.312 | 333.905 | 270.090 | 214.037 |
| I16/a | 518.170 | 547.563 | 548.155 | 460.300 | 294.671 |
| I16/c | 491.790 | 520.310 | 520.903 | 434.582 | 289.519 |
| Comp. | Total Energy (Hartree) | EHOMO (ev) | EluMO (ev) | ∆E (ev) | Dipole Moment (Debye) | IE (ev) | EA (ev) | Polarizability Bohr3 |
|---|---|---|---|---|---|---|---|---|
| I6/a | −1570.995 | –5.632 | –2.439 | 3.193 | 1.7620 | 5.632 | 2.439 | 451.01 |
| I6/b | −1555.767 | −5.774 | −2.582 | 3.192 | 3.3197 | 5.774 | 2.582 | 424.69 |
| I16/a | −1963.817 | –5.627 | –2.439 | 3.188 | 1.6794 | 5.627 | 2.439 | 571.88 |
| I16/c | −2308.947 | −5.791 | −2.614 | 3.177 | 3.7771 | 5.791 | 2.614 | 561.58 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Zahrani, S.A.; Ahmed, H.A.; El-atawy, M.A.; Abu Al-Ola, K.A.; Omar, A.Z. Synthetic, Mesomorphic, and DFT Investigations of New Nematogenic Polar Naphthyl Benzoate Ester Derivatives. Materials 2021, 14, 2587. https://doi.org/10.3390/ma14102587
Al-Zahrani SA, Ahmed HA, El-atawy MA, Abu Al-Ola KA, Omar AZ. Synthetic, Mesomorphic, and DFT Investigations of New Nematogenic Polar Naphthyl Benzoate Ester Derivatives. Materials. 2021; 14(10):2587. https://doi.org/10.3390/ma14102587
Chicago/Turabian StyleAl-Zahrani, Salma A., Hoda A. Ahmed, Mohamed A. El-atawy, Khulood A. Abu Al-Ola, and Alaa Z. Omar. 2021. "Synthetic, Mesomorphic, and DFT Investigations of New Nematogenic Polar Naphthyl Benzoate Ester Derivatives" Materials 14, no. 10: 2587. https://doi.org/10.3390/ma14102587
APA StyleAl-Zahrani, S. A., Ahmed, H. A., El-atawy, M. A., Abu Al-Ola, K. A., & Omar, A. Z. (2021). Synthetic, Mesomorphic, and DFT Investigations of New Nematogenic Polar Naphthyl Benzoate Ester Derivatives. Materials, 14(10), 2587. https://doi.org/10.3390/ma14102587







