In Situ Scanning Electron Microscopy Observation of Crack Initiation and Propagation in Hydroxide Films Formed by Steam Coating on Aluminum-Alloy Sheets
Abstract
1. Introduction
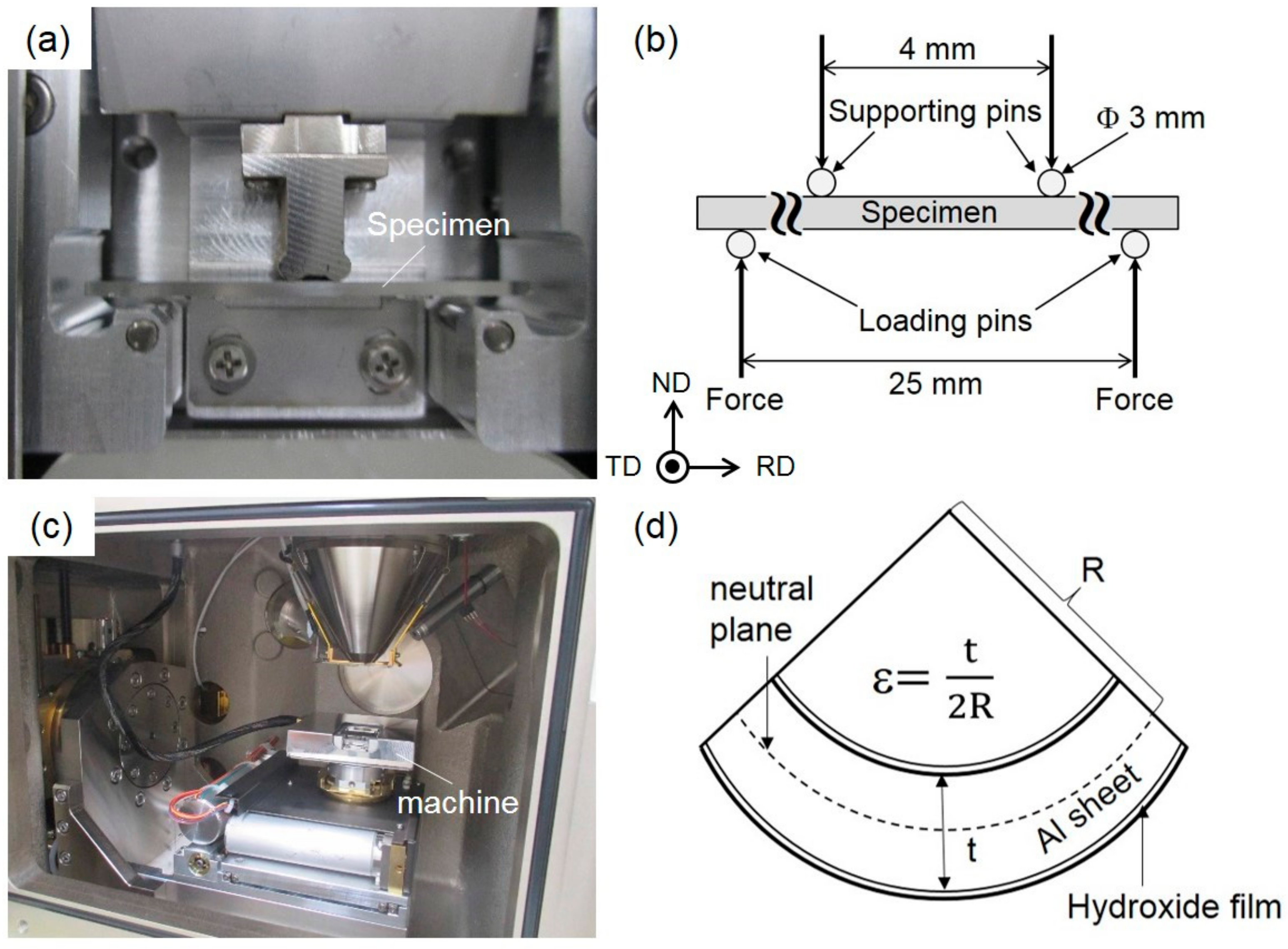
2. Experimental Procedure
3. Results
3.1. Microstructure Observation of Al Substrate and Hydroxide Film
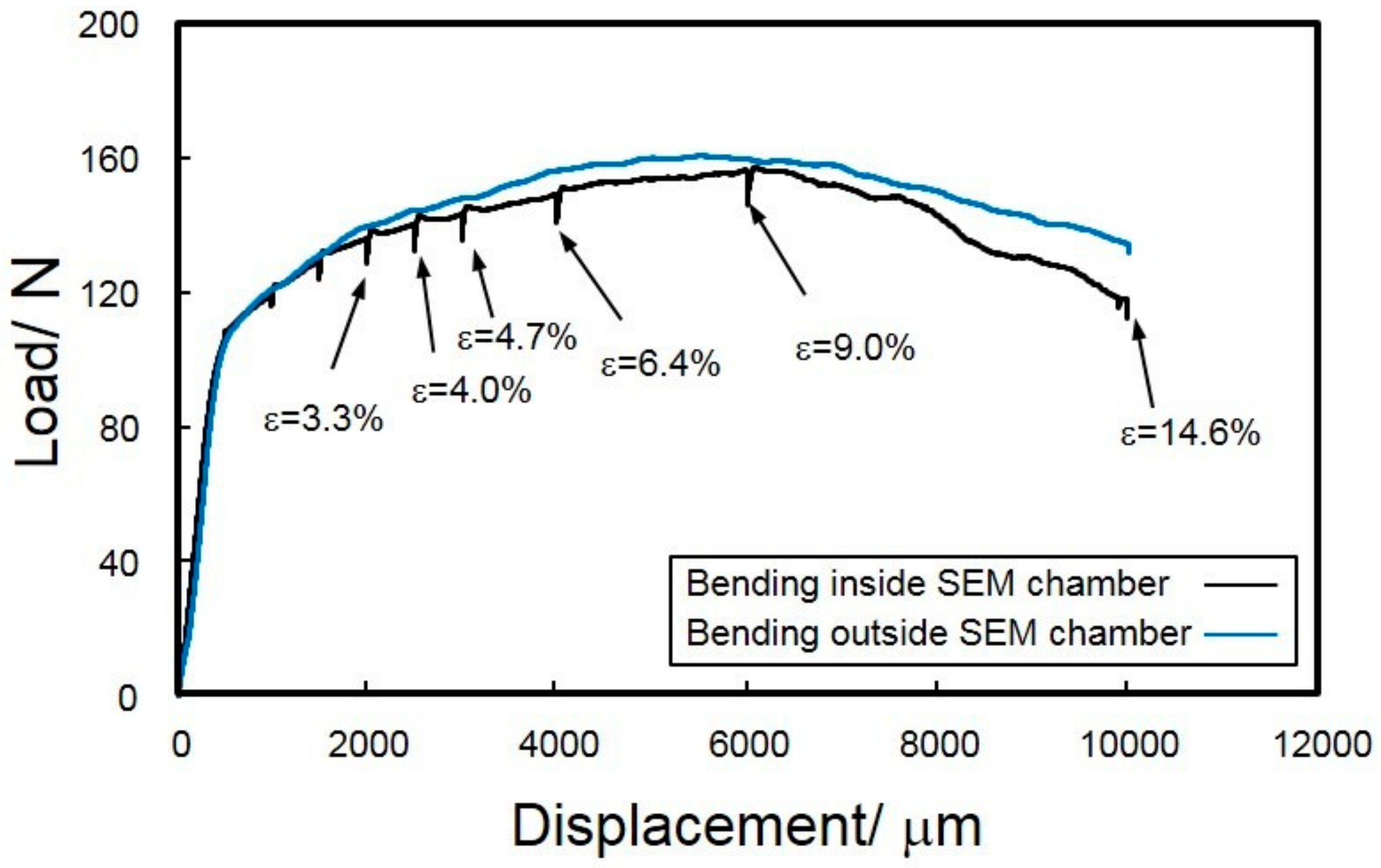
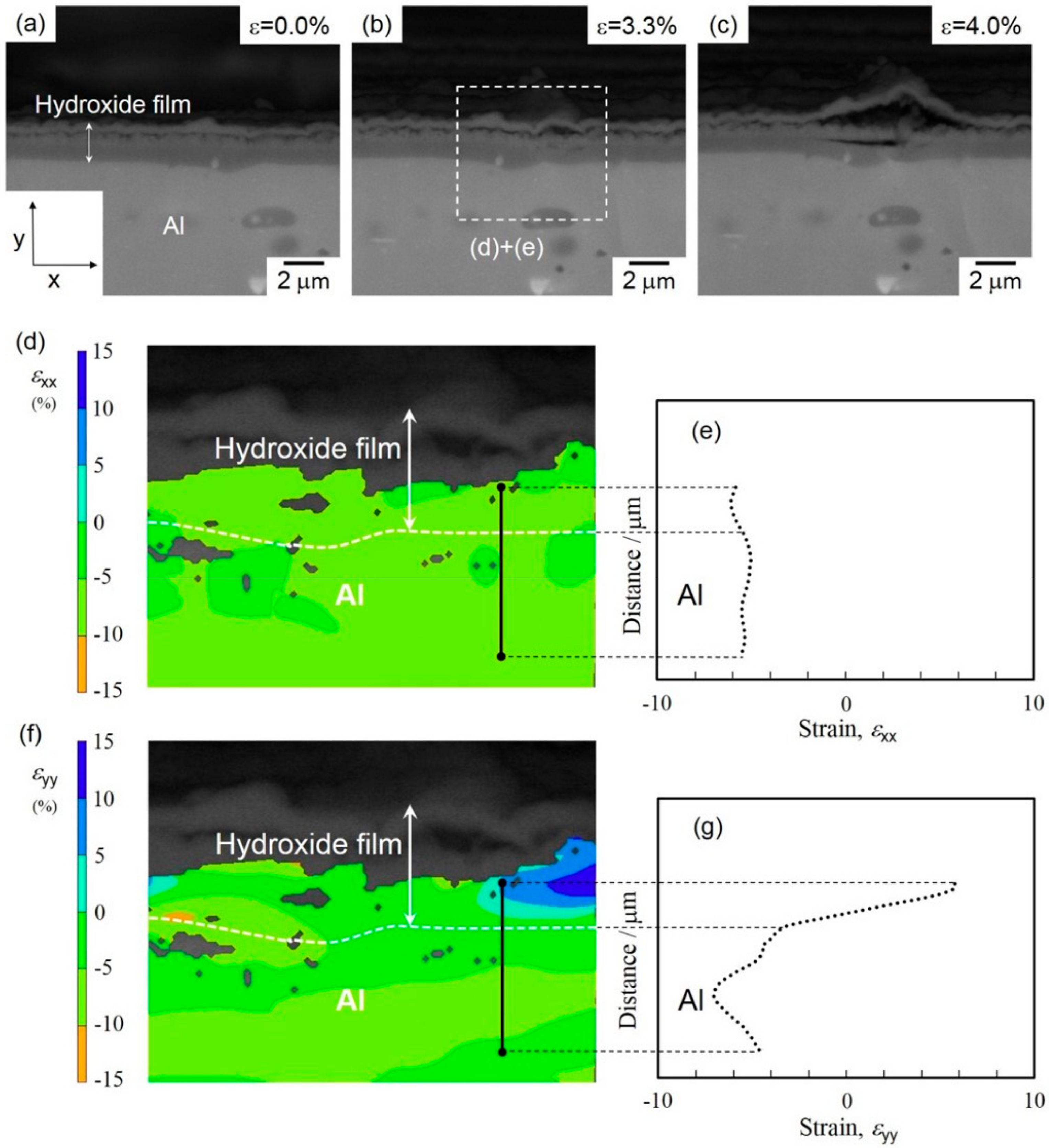
3.2. In Situ Observation of Crack Initiation and Propagation of Hydroxide Film
4. Discussion
5. Summary
Author Contributions
Funding
Conflicts of Interest
References
- Mazzolani, F.M. 3D aluminium structures. Thin-Walled Struct. 2012, 61, 258–266. [Google Scholar] [CrossRef]
- Ozturk, F.; Sisman, A.; Toros, S.; Kilic, S.; Picu, R.C. Influence of aging treatment on mechanical properties of 6061 aluminum alloy. Mater. Des. 2010, 31, 972–975. [Google Scholar] [CrossRef]
- Mrówka-Nowotnik, G. Influence of chemical composition variation and heat treatment on microstructure and mechanical properties of 6xxx alloys. Arch. Mater. Sci. Eng. 2010, 46, 98–107. [Google Scholar]
- Altenpohl, D.G.; Kaufman, J.G. Aluminum: Technology, Applications and Environment: A Profile of a Modern Metal; The Aluminum Association, Inc.: Washington, DC, USA, 1998. [Google Scholar]
- De Miera, M.S.; Curioni, M.; Skeldon, P.; Thompson, G.E. Modelling the anodizing behavior of aluminium alloys in sulphuric acid through alloy analogues. Corros. Sci. 2008, 50, 3410–3415. [Google Scholar] [CrossRef]
- Metroke, T.L.; Kachurina, O.; Knobbe, E.T. Spectroscopic and corrosion resistance characterization of GLYMO–TEOS Ormosil coatings for aluminum alloy corrosion inhibition. Prog. Org. Coat. 2002, 44, 295–305. [Google Scholar] [CrossRef]
- Ishizaki, T.; Chiba, S.; Watanabe, K.; Suzuki, H. Corrosion resistance of Mg–Al layered double hydroxide container-containing magnesium hydroxide films formed directly on magnesium alloy by chemical-free steam coating. J. Mater. Chem. A 2013, 1, 8968–8977. [Google Scholar] [CrossRef]
- Ishizaki, T.; Chiba, S.; Suzuki, H. In Situ Formation of Anticorrosive Mg–Al Layered Double Hydroxide-Containing Magnesium Hydroxide Film on Magnesium Alloy by Steam Coating. ECS Electronchem. Lett. 2013, 2, C15–C17. [Google Scholar] [CrossRef]
- Nakamura, K.; Tsunakawa, M.; Shimada, Y.; Serizawa, A.; Ishizaki, T. Formation mechanism of Mg-Al layered double hydroxide-containing magnesium hydroxide films prepared on Ca-added flame-resistant magnesium alloy by steam coating. Surf. Coat. Technol. 2017, 328, 436–443. [Google Scholar] [CrossRef]
- Serizawa, A.; Oda, T.; Watanabe, K.; Mori, K.; Yokomizo, T.; Ishizaki, T. Formation of anticorrosive film for suppressing pitting corrosion on Al-Mg-Si alloy by steam Coating. Coating 2018, 8, 23. [Google Scholar] [CrossRef]
- Takata, N.; Li, H.; Kobashi, M.; Shimada, Y.; Serizawa, A.; Ishizaki, T. Transmission electron microscopic observation of layered double hydroxide films formed on aluminum alloys prepared by steam coating process. Tetsu-to-Hagane 2019, 105, 177–182. [Google Scholar] [CrossRef]
- Li, H.; Takata, N.; Kobashi, M.; Serizawa, A. Microstructure and crack behavior of hydroxide films formed on aluminum-alloy sheets prepared by steam coating. Mat. Sci. Eng. A 2019, 764, 138247. [Google Scholar] [CrossRef]
- ASM Handbook; ASM International: Cleveland, OH, USA, 1991; Volume 4, p. 846.
- Kuba, T.; Endo, N.; Okunishi, E.; Suzuki, T. FIB preparation and STEM observation of specified area from two direction. Microsc. Microanal. 2005, 11, 852–853. [Google Scholar] [CrossRef]
- Sutton, M.A.; Cheng, M.; Peters, W.H.; Chao, Y.J.; McNeill, S.R. Application of an optimized digital correlation method to planar deformation analysis. Image Vis. Comput. 1986, 4, 143–150. [Google Scholar] [CrossRef]
- Li, Z.; Limodin, N.; Tandejaoui, A.A.; Quaegebeur, P.; Witz, J.-F.; Balloy, D. Influence of Fe content on the damage mechanism in A319 aluminum alloy: Tensile tests and digital image correlation. Eng. Fract. Mech. 2017, 183, 94–108. [Google Scholar] [CrossRef]
- Barlock, J.G.; Mondolfo, L.F. Structure of some aluminium-iron-magnesium- manganese-silicon alloys. Z. Metallkd. 1975, 66, 605–611. [Google Scholar]
- Romming, C.; Hansen, V.; Gjonnes, J. Crystal structure of beta-Al4.5FeSi. Acta Crystallogr. Sect. B 1994, 50B, 307–312. [Google Scholar] [CrossRef]
- Seo, Y.I.; Hong, K.H.; Kim, S.H.; Chang, D.; Lee, K.H.; Kim, Y.D. Removal of bacterial pathogen from wastewater using Al filter with Ag-containing nanocomposite film by in situ dispersion involving polyol process. J. Hazard. Mater. 2010, 256, 4434–4437. [Google Scholar]
- Seo, Y.I.; Hong, K.H.; Kim, S.H.; Chang, D.; Lee, K.H.; Kim, Y.D. Phosphorus removal from wastewater by ionic exchange using a surface-modified Al alloy filter. J. Ind. Eng. Chem. 2013, 19, 744–747. [Google Scholar] [CrossRef]
- Goldberg, S.; Davis, J.A.; Hem, J.D. The surface chemistry of aluminum oxides and hydroxides. In The Environmental Chemistry of Aluminum; Sposito, G., Ed.; Lewis Publishers: New York, NY, USA, 1996. [Google Scholar]









© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, H.; Takata, N.; Kobashi, M.; Serizawa, A. In Situ Scanning Electron Microscopy Observation of Crack Initiation and Propagation in Hydroxide Films Formed by Steam Coating on Aluminum-Alloy Sheets. Materials 2020, 13, 1238. https://doi.org/10.3390/ma13051238
Li H, Takata N, Kobashi M, Serizawa A. In Situ Scanning Electron Microscopy Observation of Crack Initiation and Propagation in Hydroxide Films Formed by Steam Coating on Aluminum-Alloy Sheets. Materials. 2020; 13(5):1238. https://doi.org/10.3390/ma13051238
Chicago/Turabian StyleLi, Hongmei, Naoki Takata, Makoto Kobashi, and Ai Serizawa. 2020. "In Situ Scanning Electron Microscopy Observation of Crack Initiation and Propagation in Hydroxide Films Formed by Steam Coating on Aluminum-Alloy Sheets" Materials 13, no. 5: 1238. https://doi.org/10.3390/ma13051238
APA StyleLi, H., Takata, N., Kobashi, M., & Serizawa, A. (2020). In Situ Scanning Electron Microscopy Observation of Crack Initiation and Propagation in Hydroxide Films Formed by Steam Coating on Aluminum-Alloy Sheets. Materials, 13(5), 1238. https://doi.org/10.3390/ma13051238






