Comparison of Mechanical and Barrier Properties of Al2O3/TiO2/ZrO2 Layers in Oxide–Hydroxyapatite Sandwich Composite Coatings Deposited by Sol–Gel Method on Ti6Al7Nb Alloy
Abstract
1. Introduction
2. Materials and Methods
2.1. Coatings Preparation
2.2. Characterization of Coatings
- MAl—mass of Al ions in the solution [µg];
- Cp—measured ion concentration [µg/dm3];
- V—volume of the liquid in which the samples were submerged [dm3].
- Pp—mass of Al ions released per surface unit [µg/cm2];
- MAl—mass of Al ions in the solution [µg];
- M0—mass of Al ions in the relative solution (HBSS without sample) [µg];
- Sp—sample surface in contact with the liquid [cm2].
3. Results and Discussion
3.1. Morphology, Roughness, and Thickness of Coatings
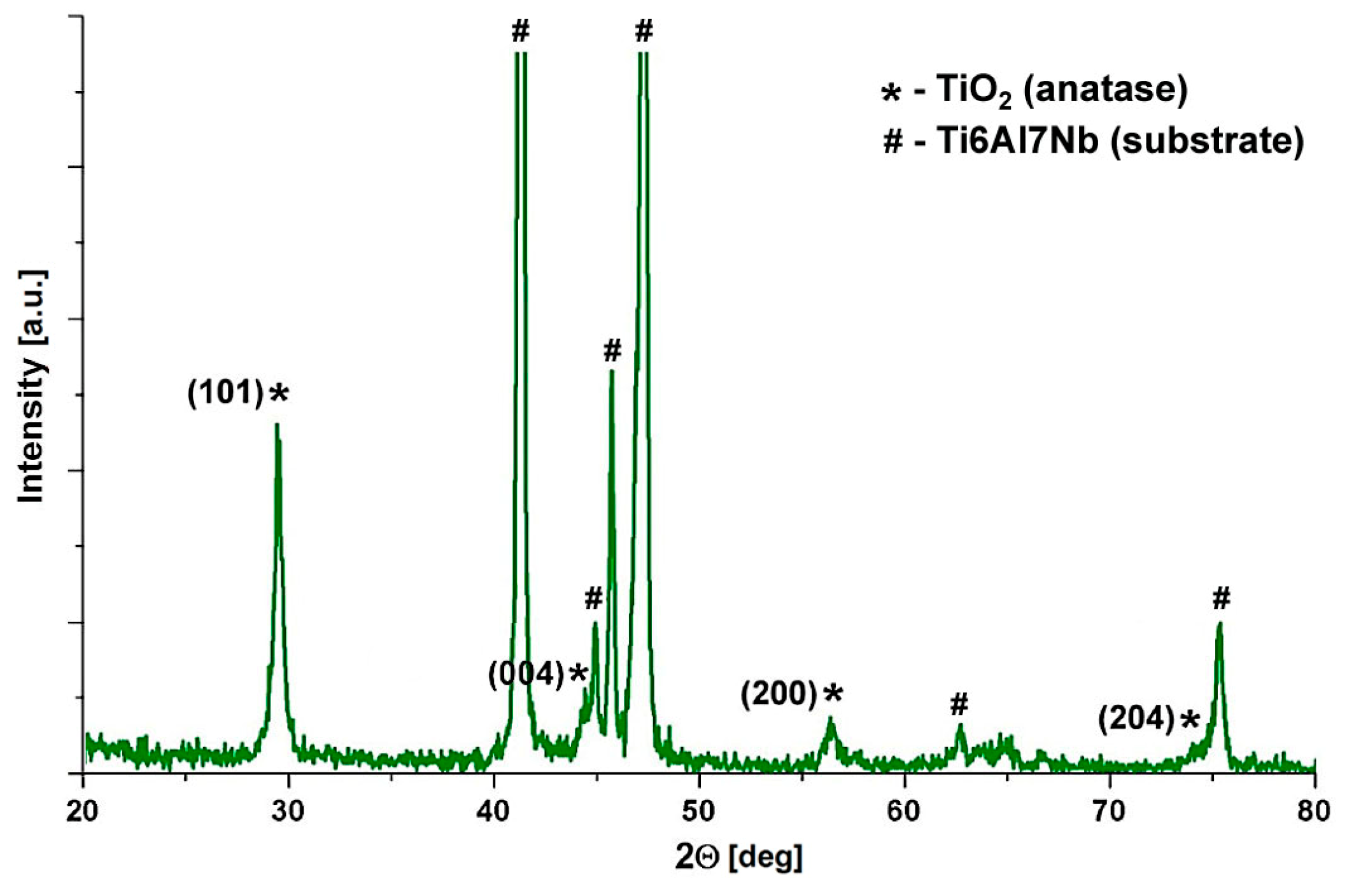
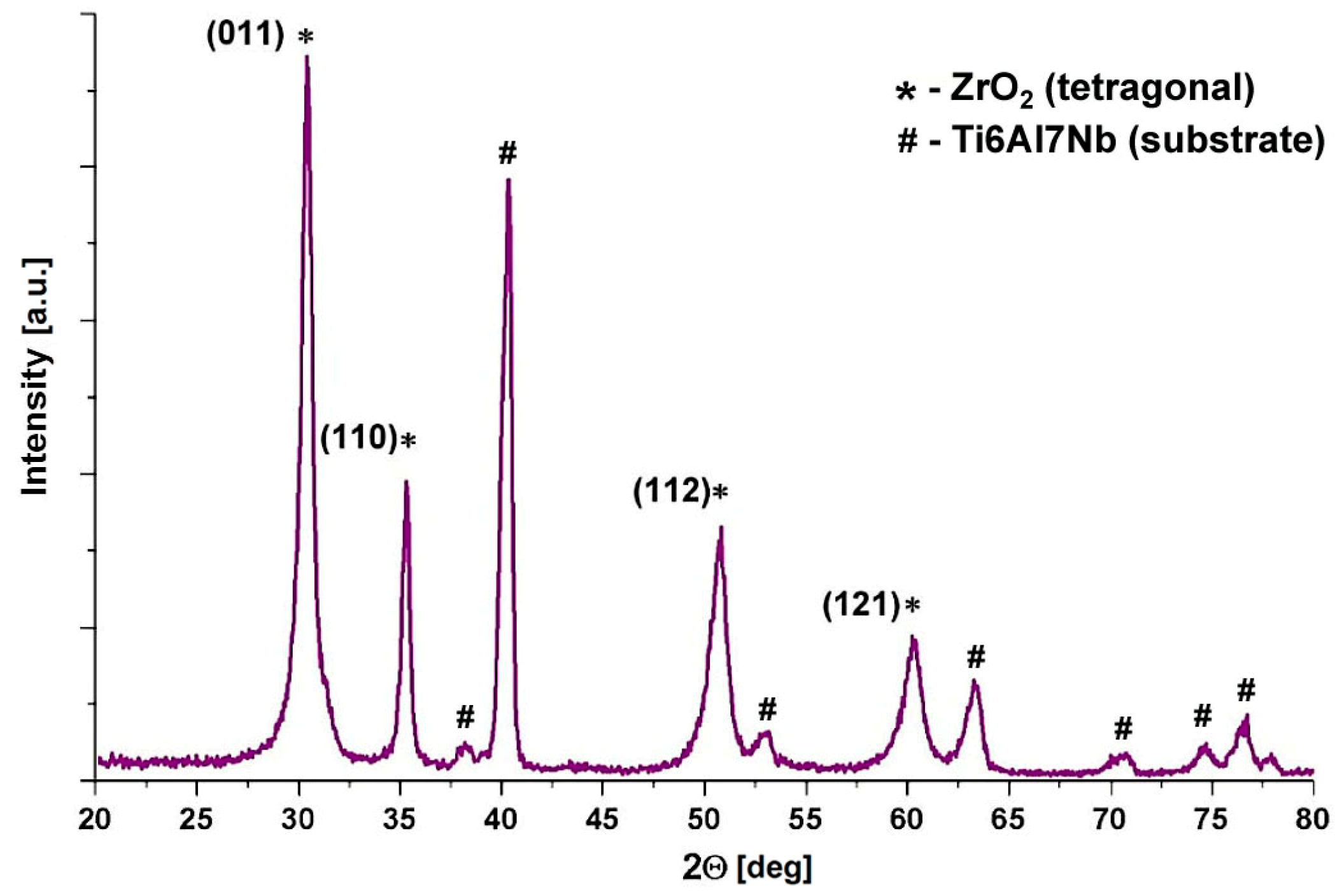
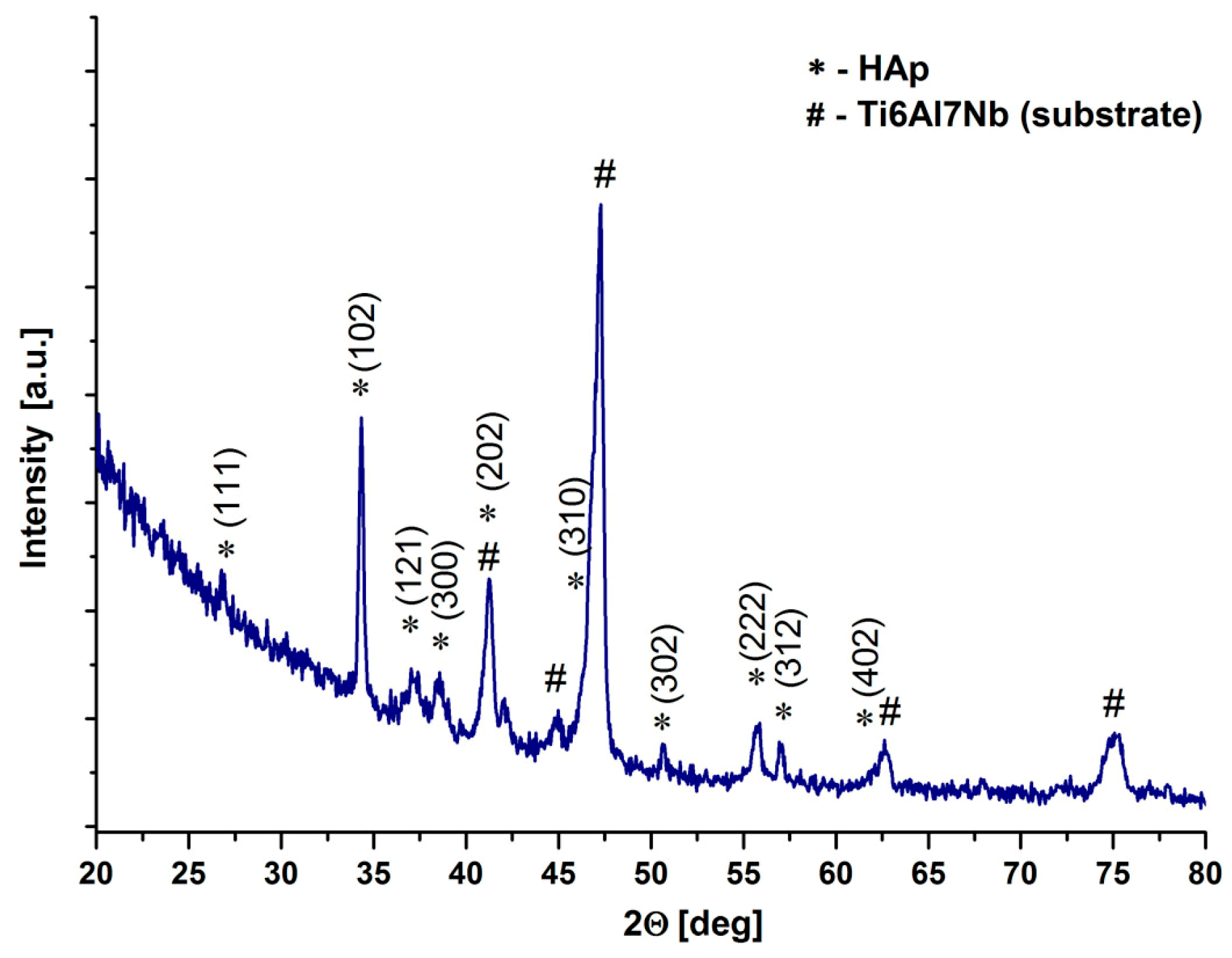
3.2. Phase Composition
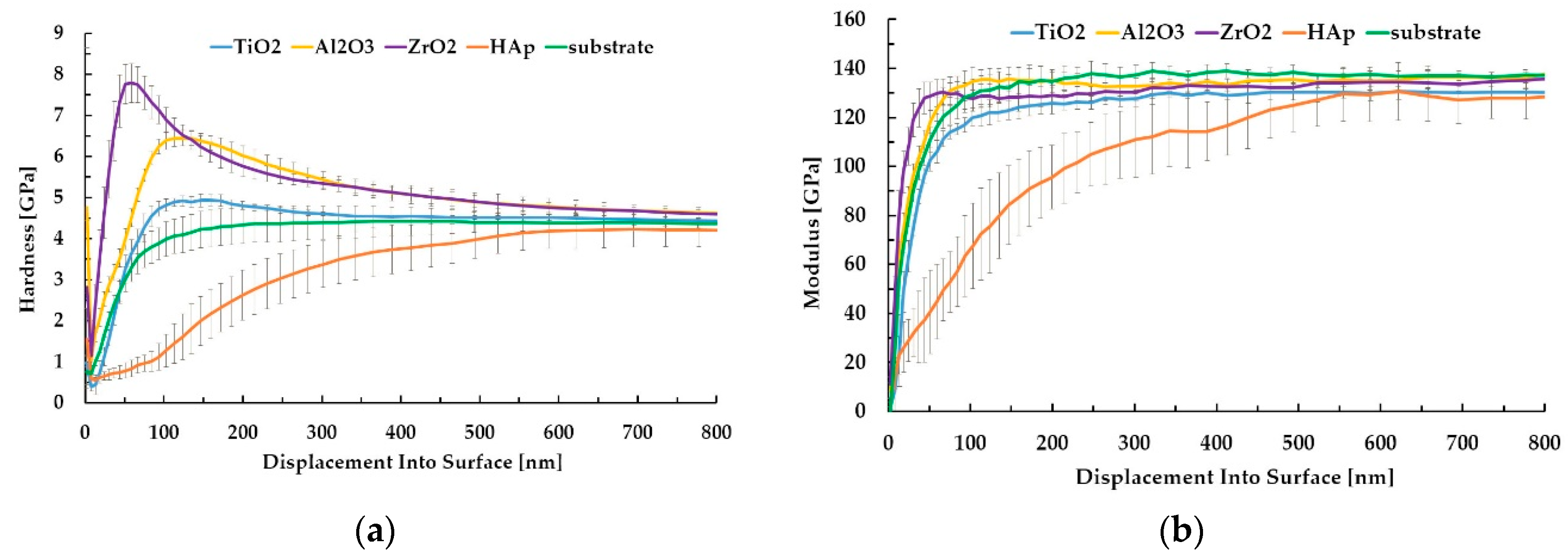
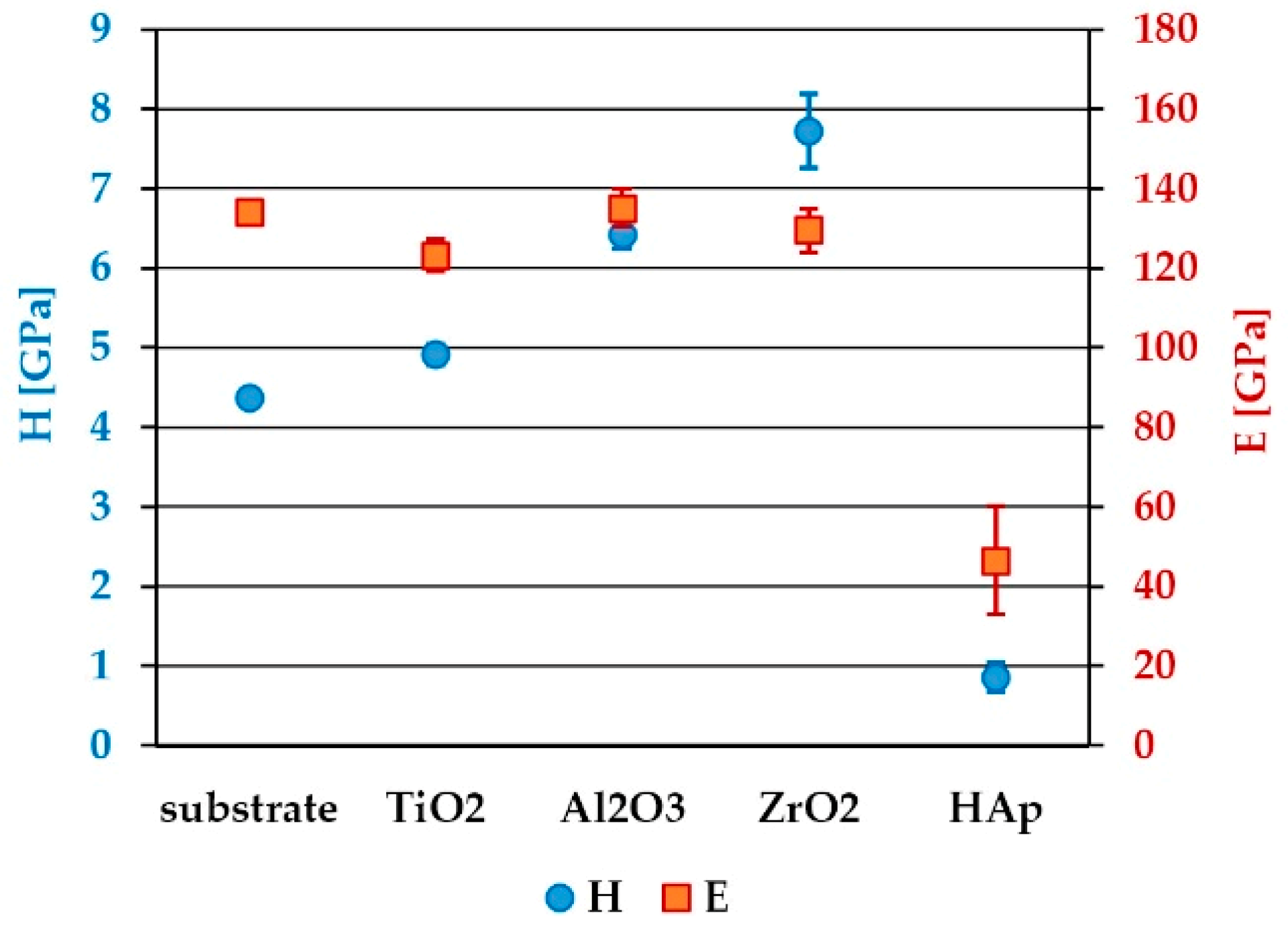
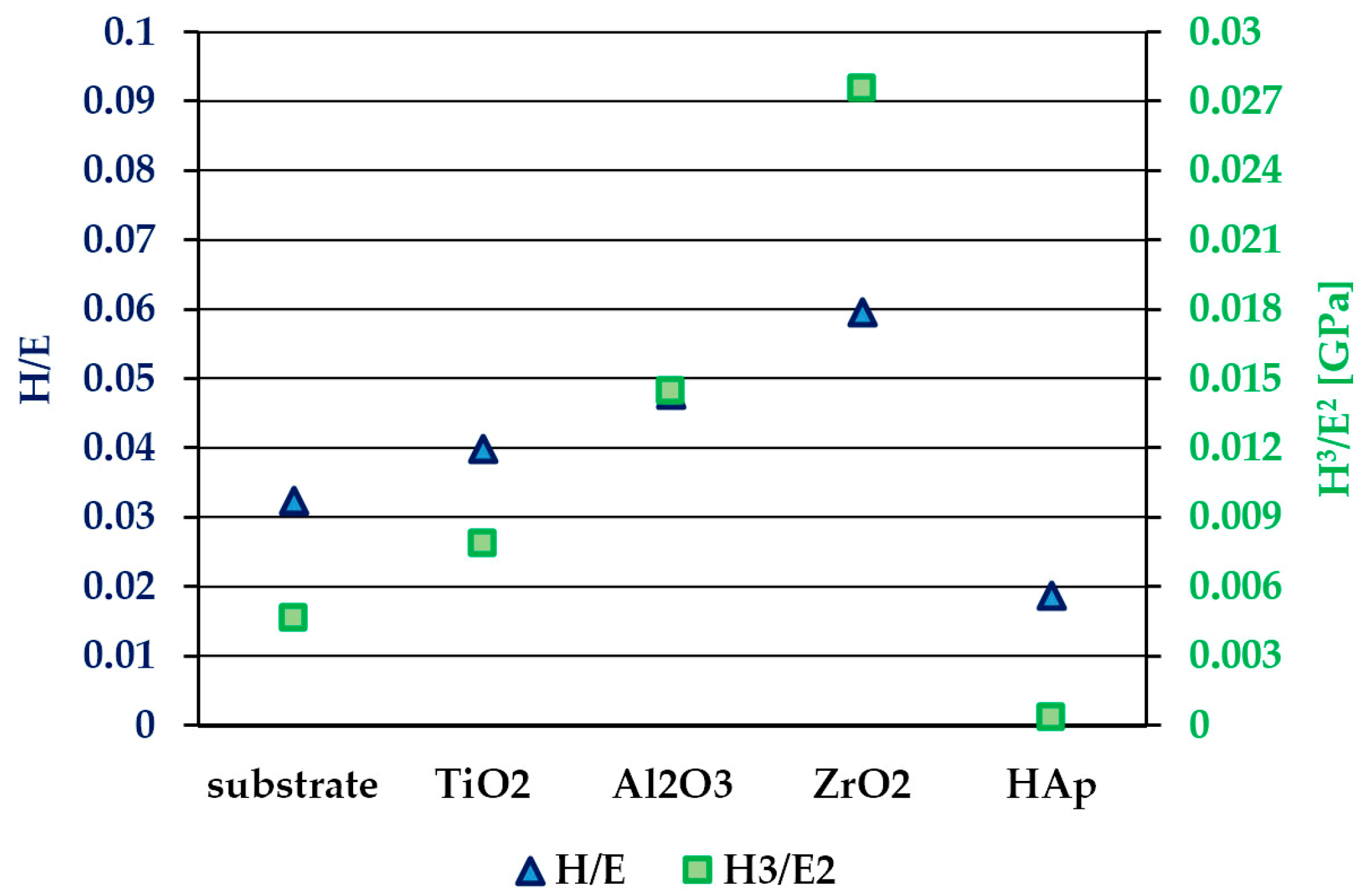
3.3. Hardness and Elastic Modulus
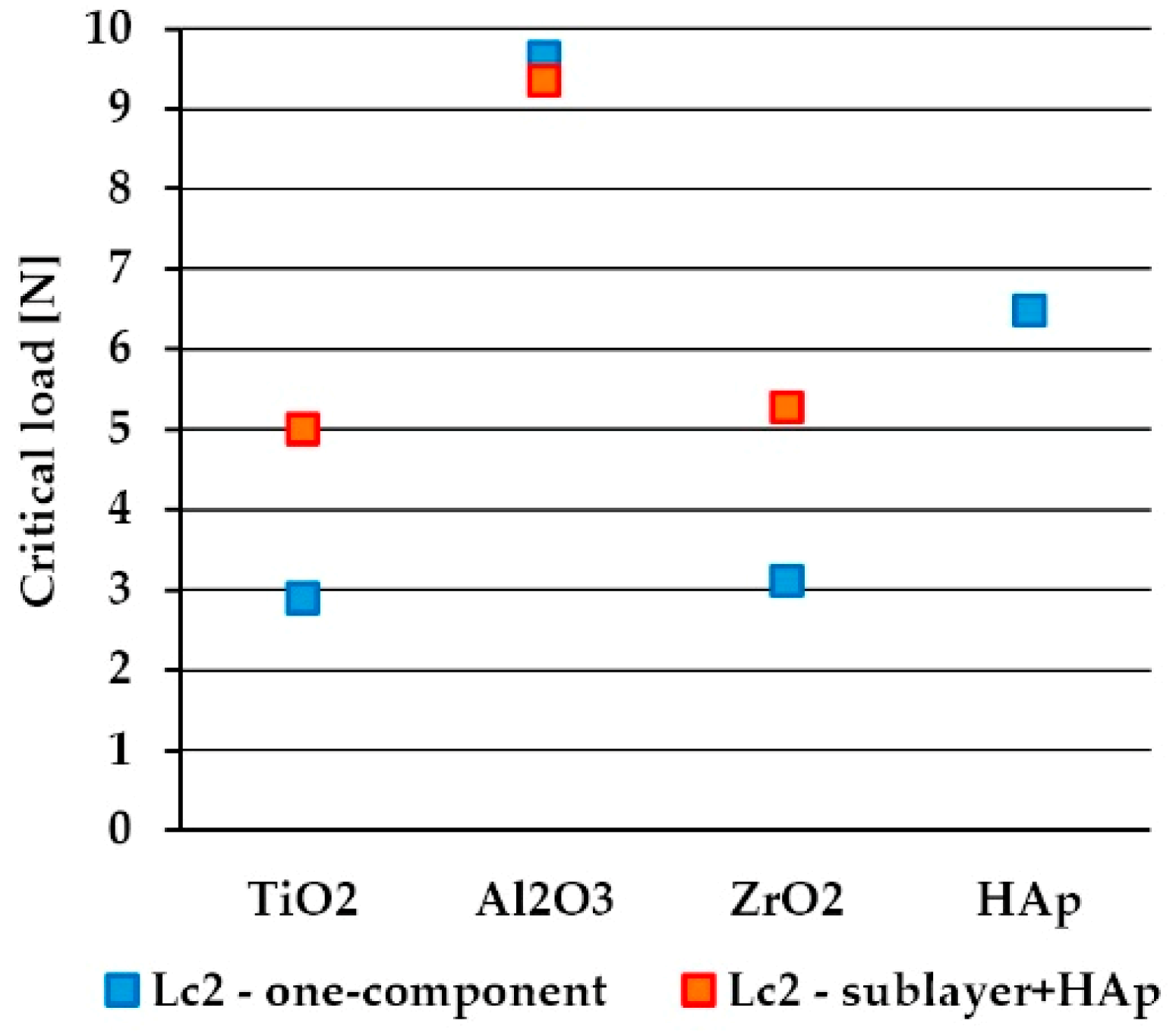
3.4. Scratch Adhesion
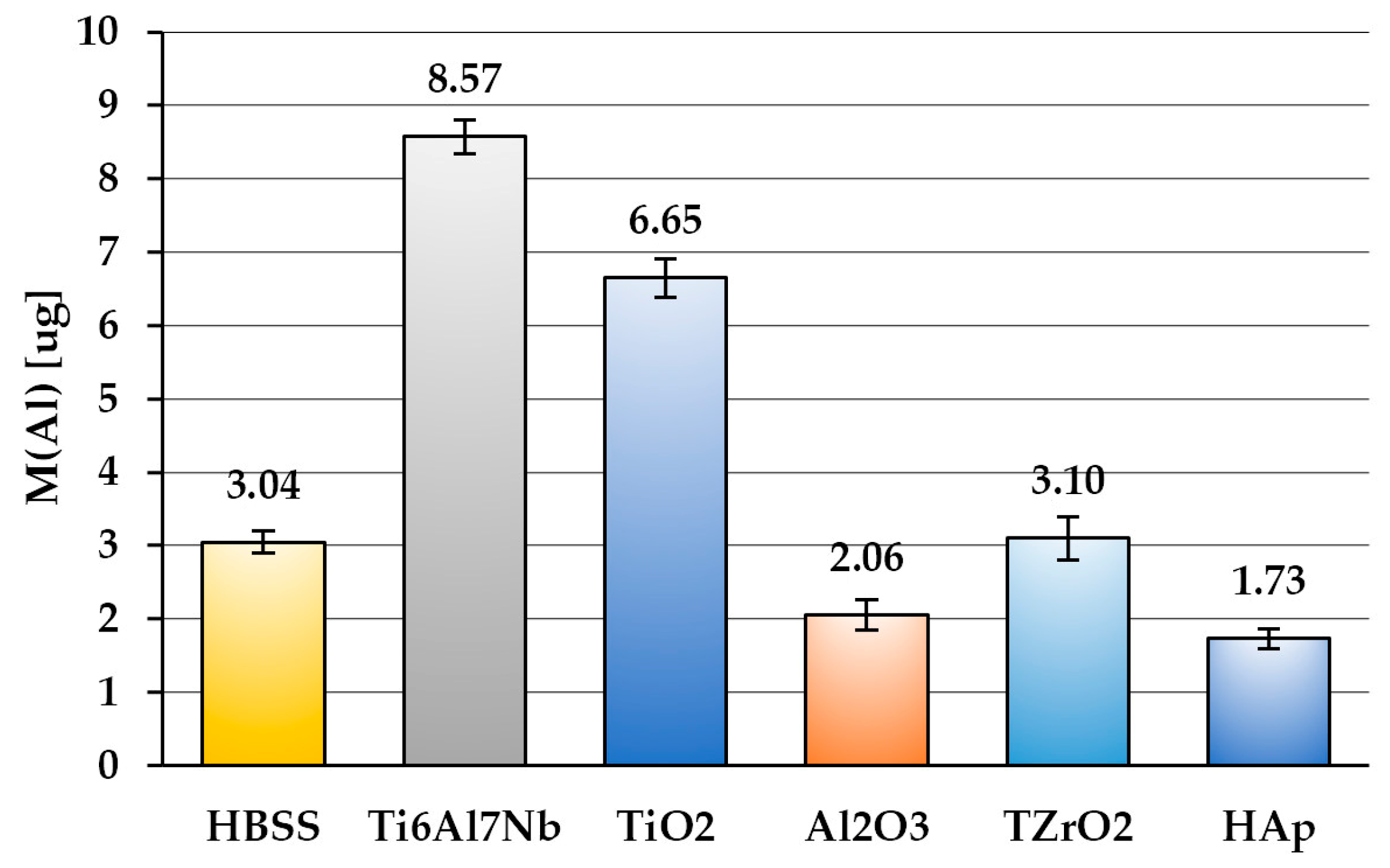
3.5. Barrier Properties
4. Summary and Conclusions
- elastic strain to failure and the toughness of the investigated oxide layers are mainly governed by the hardness parameter;
- no relation between the mechanical properties and scratch resistance of individual layers has been found;
- the best scratch resistance was observed for sandwich composite coatings with Al2O3 as a sublayer (Al2O3+HAp).
Author Contributions
Funding
Conflicts of Interest
References
- Ribas, R.G.; Schatkoski, V.M.; do Amaral Montanheiro, T.L.; de Menezes, B.R.C.; Stegemann, C.; Leite, D.M.G.; Thim, G.P. Current advances in bone tissue engineering concerning ceramic and bioglass scaffolds: A review. In Ceram. Int.; 2019; Volume 45, pp. 21051–21061. [Google Scholar]
- Ibrahim, M.Z.; Sarhan, A.A.D.; Yusuf, F.; Hamdi, M. Biomedical materials and techniques to improve the tribological, mechanical and biomedical properties of orthopedic implants—A review article. J. Alloy Compd. 2017, 714, 636–667. [Google Scholar] [CrossRef]
- Su, Y.; Luo, C.; Zhang, Z.; Hermawan, H.; Zhu, D.; Huang, J.; Liang, Y.; Li, G.; Ren, L. Bioinspired surface functionalization of metallic biomaterials. J. Mech. Behav. Biomed. Mater. 2018, 77, 90–105. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Li, P.; Feng, H.; Zhang, X.; Chu, P. Engineering and functionalization of biomaterials via surface modification. J. Mater. Chem. B 2015, 3, 2024–2042. [Google Scholar] [CrossRef]
- Xiao, M.; Chen, Y.M.; Biao, M.N.; Zhang, X.D.; Yang, B.C. Bio-functionalization of biomedical metals. Mater. Sci. Eng. C 2017, 70, 1057–1070. [Google Scholar] [CrossRef] [PubMed]
- Szcześ, A.; Hołysz, L.; Chibowski, E. Synthesis of hydroxyapatite for biomedical applications. Adv. Colloid Interface Sci. 2017, 249, 321–330. [Google Scholar] [CrossRef]
- Denry, I.; Kuhn, L.T. Design and characterization of calcium phosphate ceramic scaffolds for bone tissue engineering. Dent. Mater. 2016, 32, 43–53. [Google Scholar] [CrossRef]
- Su, Y.; Cockerill, I.; Zheng, Y.; Tang, L.; Qin, Y.-X.; Zhu, D. Biofunctionalization of metallic implants by calcium phosphate coatings. Bioact. Mater. 2019, 4, 196–206. [Google Scholar] [CrossRef]
- Surmenev, R.A.; Surmeneva, M.A.; Ivanova, A.A. Significance of calcium phosphate coatings for the enhancement of new bone osteogenesis—A review. Acta Biomater. 2014, 10, 557–579. [Google Scholar] [CrossRef]
- Harun, W.S.W.; Asri, R.I.M.; Alias, J.; Zulkifli, F.H.; Kadirgama, K.; Ghani, S.A.C.; Shariffuddin, J.H.M. A comprehensive review of hydroxyapatite-based coatings adhesion on metallic biomaterials. Ceram. Int. 2018, 44, 1250–1268. [Google Scholar] [CrossRef]
- Chambard, M.; Marsan, O.; Charvillat, C.; Grossin, D.; Fort, P.; Rey, C.; Gitzhofer, F.; Bertrand, G. Effect of the deposition route on the microstructure of plasma-sprayed hydroxyapatite coatings. Surf. Coat. Technol. 2019, 371, 68–77. [Google Scholar] [CrossRef]
- Kumari, R.; Majumdar, J.D. Microstructure and surface mechanical properties of plasma spray deposited and post spray heat treated hydroxyapatite (HA) based composite coating on titanium alloy (Ti-6Al-4V) substrate. Mater. Charact. 2017, 131, 12–20. [Google Scholar] [CrossRef]
- Popescu-Pelin, G.; Sima, F.; Sima, L.E.; Mihailescu, C.N.; Luculescu, C.; Iordache, I.; Socol, M.; Socol, G.; Mihailescu, I.N. Hydroxyapatite thin films grown by pulsed laser deposition and matrix assisted pulsed laser evaporation: Comparative study. Appl. Surf. Sci. 2017, 418, 580–588. [Google Scholar] [CrossRef]
- Hidalgo-Robatto, B.M.; López-Álvarez, M.; Azevedo, A.S.; Dorado, J.; Serra, J.; Azevedo, N.F.; González, P. Pulsed laser deposition of copper and zinc doped hydroxyapatite coatings for biomedical applications. Surf. Coat. Technol. 2018, 333, 168–177. [Google Scholar] [CrossRef]
- Ivanova, A.A.; Surmeneva, M.A.; Tyurin, A.I.; Surmenev, R.A. Correlation between structural and mechanical properties of RF magnetron sputter deposited hydroxyapatite coating. Mater. Charact. 2018, 142, 261–269. [Google Scholar] [CrossRef]
- Qadir, M.; Li, Y.; Wen, C. Ion-substituted calcium phosphate coatings by physical vapor deposition magnetron sputtering for biomedical applications: A review. Acta Biomater. 2019, 89, 14–32. [Google Scholar] [CrossRef] [PubMed]
- Mardali, M.; SalimiJazi, H.R.; Karimzadeh, F.; Luthringer, B.; Blawert, C.; Labbaf, S. Comparative study on microstructure and corrosion behavior of nanostructured hydroxyapatite coatings deposited by high velocity oxygen fuel and flame spraying on AZ61 magnesium based substrates. Appl. Surf. Sci. 2019, 465, 614–624. [Google Scholar] [CrossRef]
- Vilardell, A.M.; Cinca, N.; Cano, I.G.; Concustell, A.; Dosta, S.; Guilemany, J.M.; Estradé, S.; Ruiz-Caridad, A.; Peiró, F. Dense nanostructured calcium phosphate coating on titanium by cold spray. J. Eur. Ceram. Soc. 2017, 37, 1747–1755. [Google Scholar] [CrossRef]
- Bartmański, M.; Pawłowski, Ł.; Strugała, G.; Mielewczyk-Gryń, A.; Zieliński, A. Properties of Nanohydroxyapatite Coatings Doped with Nanocopper, Obtained by Electrophoretic Deposition on Ti13Zr13Nb Alloy. Materials 2019, 12, 3741. [Google Scholar] [CrossRef]
- Strąkowska, P.; Beutner, R.; Gnyba, M.; Zielinski, A.; Scharnweber, D. Electrochemically assisted deposition of hydroxyapatite on Ti6Al4V substrates covered by CVD diamond films—Coating characterization and first cell biological results. Mater. Sci. Eng. C 2016, 59, 624–635. [Google Scholar] [CrossRef]
- Asri, R.I.M.; Harun, W.S.W.; Hassan, M.A.; Ghani, S.A.C.; Buyong, Z. A review of hydroxyapatite-based coating techniques: Sol–gel and electrochemical depositions on biocompatible metals. J. Mech. Behav. Biomed. Mater. 2016, 57, 95–108. [Google Scholar] [CrossRef]
- Domínguez-Trujillo, C.; Peón, E.; Chicardi, E.; Pérez, H.; Rodríguez-Ortiz, J.A.; Pavón, J.J.; García-Couce, J.; Galván, J.C.; García-Moreno, F.; Torres, Y. Sol-gel deposition of hydroxyapatite coatings on porous titanium for biomedical applications. Surf. Coat. Technol. 2018, 333, 158–162. [Google Scholar] [CrossRef]
- Mohseni, E.; Zalnezhad, E.; Bushroa, A.R. Comparative investigation on the adhesion of hydroxyapatite coating on Ti–6Al–4V implant: A review paper. Int. J. Adhes. Adhes. 2014, 48, 238–257. [Google Scholar] [CrossRef]
- Kaur, G.; Kumar, V.; Baino, F.; Mauro, J.C.; Pickrell, G.; Evans, I.; Bretcanu, O. Mechanical properties of bioactive glasses, ceramics, glass-ceramics and composites: State-of-the-art review and future challenges. Mater. Sci. Eng. C 2019, 104, 109895. [Google Scholar] [CrossRef] [PubMed]
- Chien, C.S.; Liu, C.W.; Kuo, T.Y.; Wu, C.C.; Hong, T.F. Bioactivity of fluorapatite/alumina composite coatings deposited on Ti6Al4V substrates by laser cladding. Appl. Phys. A 2016, 122, 303. [Google Scholar] [CrossRef]
- Wang, Z.C.; Ni, Y.J.; Huang, J.C. Fabrication and Characterization of HAp/Al2O3 Composite Coating on Titanium Substrate. In Proceedings of the 2008 2nd International Conference on Bioinformatics and Biomedical Engineering, Shanghai, China, 16–18 May 2008; pp. 1526–1530. [Google Scholar]
- Wang, X.; Liu, F.; Song, Y. Enhanced corrosion resistance and in vitro bioactivity of NiTi alloys modified with hydroxyapatite-containing Al2O3 coatings. Surf. Coat. Technol. 2018, 344, 288–294. [Google Scholar] [CrossRef]
- Singh, A.; Singh, G.; Chawla, V. Influence of post coating heat treatment on microstructural, mechanical and electrochemical corrosion behaviour of vacuum plasma sprayed reinforced hydroxyapatite coatings. J. Mech. Behav. Biomed. Mater. 2018, 85, 20–36. [Google Scholar] [CrossRef] [PubMed]
- Shojaee, P.; Afshar, A. Effects of zirconia content on characteristics and corrosion behavior of hydroxyapatite/ZrO2 biocomposite coatings codeposited by electrodeposition. Surf. Coat. Technol. 2015, 262, 166–172. [Google Scholar] [CrossRef]
- Sidane, D.; Khireddine, H.; Yala, S.; Ziani, S.; Bir, F.; Chicot, D. Morphological and Mechanical Properties of Hydroxyapatite Bilayer Coatings Deposited on 316L SS by Sol–Gel Method. Metall. Mater. Trans. B 2015, 46, 2340–2347. [Google Scholar] [CrossRef]
- Walia, B.; Sarao, T.P.S.; Grewal, J. Corrosion studies of thermal sprayed HA and HA/Al2O3-TiO2 bond coating on 316L stainless steel. Trends Biomater. Artif. Organs 2015, 29, 245–252. [Google Scholar]
- Zieliński, A.; Antoniuk, P.; Krzysztofowicz, K. Nanotubular oxide layers and hydroxyapatite coatings on ‘Ti–13Zr–13Nb’ alloy. Surf. Eng. 2014, 30, 643–649. [Google Scholar] [CrossRef]
- Wang, M.-J.; Chao, S.-C.; Yen, S.-K. Electrolytic calcium phosphate/zirconia composite coating on AZ91D magnesium alloy for enhancing corrosion resistance and bioactivity. Corros. Sci. 2016, 104, 47–60. [Google Scholar] [CrossRef]
- Bermúdez-Reyes, B.; Contreras-Garcia, M.E.; Almaral-Sánchez, J.L.; Espitia Cabrera, M.; Espinoza-Beltrán, F.J. Chemical anchorage of Hydroxyapatite on 316LSS using a ZrO2 interlayer for orthopedic prosthesis applications. Superf. Y Vacio 2012, 25, 150–156. [Google Scholar]
- Kaur, M.; Singh, K. Review on titanium and titanium based alloys as biomaterials for orthopaedic applications. Mater. Sci. Eng. C 2019, 102, 844–862. [Google Scholar] [CrossRef] [PubMed]
- Owens, G.J.; Singh, R.K.; Foroutan, F.; Alqaysi, M.; Han, C.-M.; Mahapatra, C.; Kim, H.-W.; Knowles, J.C. Sol–gel based materials for biomedical applications. Prog. Mater. Sci. 2016, 77, 1–79. [Google Scholar] [CrossRef]
- Wright, J.D.; Sommerdijk, N.A.J.M. Sol-Gel Materials: Chemistry and Applications; CRC Press: London, UK, 2000; ISBN 978-90-5699-326-9. [Google Scholar]
- Dehghanghadikolaei, A.; Ansary, J.; Ghoreishi, R. Sol-gel process applications: A mini-review. Proc. Nat. Res. Soc. 2018, 2, 02008. [Google Scholar] [CrossRef]
- Miszczak, S.; Pietrzyk, B.; Gawronski, Z. Protective properties of composite oxide coatings deposited by sol-gel method. Powder Metall. Met. Ceram. 2011, 50, 295–300. [Google Scholar] [CrossRef]
- Zydorczyk, A.; Pietrzyk, B.; Szymanowski, H.; Miszczak, S. Comparison of structure and morphology of hydroxyapatite films obtained by sol-gel and pe RFCVD method. Inz. Mater. 2010, 31, 770–772. [Google Scholar]
- Kwon, S.J.; Park, J.-G. Dewetting of a Sol–Gel-derived Thin Film. Langmuir 2006, 22, 3895–3898. [Google Scholar] [CrossRef]
- Oliver, W.C.; Pharr, G.M. An improved technique for determining hardness and elastic modulus using load and displacement sensing indentation experiments. J. Mater. Res. 1992, 7, 1564–1583. [Google Scholar] [CrossRef]
- Miszczak, S.; Pietrzyk, B. Anatase–rutile transformation of TiO2 sol–gel coatings deposited on different substrates. Ceram. Int. 2015, 41, 7461–7465. [Google Scholar] [CrossRef]
- Zu, G.; Shen, J.; Wei, X.; Ni, X.; Zhang, Z.; Wang, J.; Liu, G. Preparation and characterization of monolithic alumina aerogels. J. Non Cryst. Solids 2011, 357, 2903–2906. [Google Scholar] [CrossRef]
- Pietrzyk, B.; Miszczak, S.; Szymanowski, H.; Kucharski, D. Plasma enhanced aerosol–gel deposition of Al2O3 coatings. J. Eur. Ceram. Soc. 2013, 33, 2341–2346. [Google Scholar] [CrossRef]
- Miszczak, S.; Pietrzyk, B.; Kucharski, D. Zirconia coatings deposited by novel plasma-enhanced aerosol–gel method. Phys. Status Solidi A 2016, 213, 1109–1114. [Google Scholar] [CrossRef]
- Di Girolamo, G.; Blasi, C.; Pagnotta, L.; Schioppa, M. Phase evolution and thermophysical properties of plasma sprayed thick zirconia coatings after annealing. Ceram. Int. 2010, 36, 2273–2280. [Google Scholar] [CrossRef]
- Shimizu, H.; Konagai, S.; Ikeda, M.; Nishide, T. Characterization of Sol–Gel Derived and Crystallized ZrO2Thin Films. Jpn. J. Appl. Phys. 2009, 48, 101101. [Google Scholar] [CrossRef]
- Fu, T.; Wen, C.S.; Lu, J.; Zhou, Y.M.; Ma, S.G.; Dong, B.H.; Liu, B.G. Sol-gel derived TiO2 coating on plasma nitrided 316L stainless steel. Vacuum 2012, 86, 1402–1407. [Google Scholar] [CrossRef]
- Borrero-López, O.; Hoffman, M.; Bendavid, A.; Martin, P.J. Mechanical properties and scratch resistance of filtered-arc-deposited titanium oxide thin films on glass. Thin Solid Film. 2011, 519, 7925–7931. [Google Scholar] [CrossRef]
- Modes, T.; Scheffel, B.; Metzner, C.; Zywitzki, O.; Reinhold, E. Structure and properties of titanium oxide layers deposited by reactive plasma activated electron beam evaporation. Surf. Coat. Technol. 2005, 200, 306–309. [Google Scholar] [CrossRef]
- Lucca, D.A.; Klopfstein, M.J.; Ghisleni, R.; Gude, A.; Mehner, A.; Datchary, W. Investigation of Sol-Gel Derived Zr02 Thin Films by Nanoindentation. CIRP Ann. 2004, 53, 475–478. [Google Scholar] [CrossRef]
- Anast, M.; Bell, J.M.; Bell, T.J.; Ben-Nissan, B. Precision ultra-microhardness measurements of sol-gel-derived zirconia thin films. J. Mater. Sci. Lett. 1992, 11, 1483–1485. [Google Scholar] [CrossRef]
- Leyland, A.; Matthews, A. On the significance of the H/E ratio in wear control: A nanocomposite coating approach to optimised tribological behaviour. Wear 2000, 246, 1–11. [Google Scholar] [CrossRef]
- Musil, J.; Kunc, F.; Zeman, H.; Poláková, H. Relationships between hardness, Young’s modulus and elastic recovery in hard nanocomposite coatings. Surf. Coat. Technol. 2002, 154, 304–313. [Google Scholar] [CrossRef]
- Musil, J. Hard and superhard nanocomposite coatings. Surf. Coat. Technol. 2000, 125, 322–330. [Google Scholar] [CrossRef]
- Galvan, D.; Pei, Y.T.; De Hosson, J.T.M. Deformation and failure mechanism of nano-composite coatings under nano-indentation. Surf. Coat. Technol. 2006, 200, 6718–6726. [Google Scholar] [CrossRef]
- Voevodin, A.A.; Zabinski, J.S. Load-adaptive crystalline–amorphous nanocomposites. J. Mater. Sci. 1998, 33, 319–327. [Google Scholar] [CrossRef]
- Daniel, J.; Souček, P.; Grossman, J.; Zábranský, L.; Bernátová, K.; Buršíková, V.; Fořt, T.; Vašina, P.; Sobota, J. Adhesion and dynamic impact wear of nanocomposite TiC-based coatings prepared by DCMS and HiPIMS. Int. J. Refract. Met. Hard Mater. 2020, 86, 105123. [Google Scholar] [CrossRef]
- Beake, B.D.; Vishnyakov, V.M.; Harris, A.J. Relationship between mechanical properties of thin nitride-based films and their behaviour in nano-scratch tests. Tribol. Int. 2011, 44, 468–475. [Google Scholar] [CrossRef]
- Kalidindi, R.S.R.; Subasri, R. 5—Sol-gel nanocomposite hard coatings. In Anti-Abrasive Nanocoatings; Aliofkhazraei, M., Ed.; Woodhead Publishing: Shaston, UK, 2015; pp. 105–136. ISBN 978-0-85709-211-3. [Google Scholar]
- Rezende, A.B.; dos Santos, J.A.; Câmara, A.M.; do Carmo, J.D.; Houmard, M.; Rodrigues, R.A.; Campos Rubio, C.J. Characterization of Ceramics Coatings Processed by Sol-Gel for Cutting Tools. Coatings 2019, 9, 775. [Google Scholar] [CrossRef]
- Bemporad, E.; Sebastiani, M.; Staia, M.H.; Puchi Cabrera, E. Tribological studies on PVD/HVOF duplex coatings on Ti6Al4V substrate. Surf. Coat. Technol. 2008, 203, 566–571. [Google Scholar] [CrossRef]
- Lin, C.-K.; Hsu, C.-H.; Kung, S.-C. Effect of electroless nickel interlayer on wear behavior of CrN/ZrN multilayer films on Cu-alloyed ductile iron. Appl. Surf. Sci. 2013, 284, 59–65. [Google Scholar] [CrossRef]
- Kim, H.-K.; La, J.-H.; Kim, K.-S.; Lee, S.-Y. The effects of the H/E ratio of various Cr–N interlayers on the adhesion strength of CrZrN coatings on tungsten carbide substrates. Surf. Coat. Technol. 2015, 284, 230–234. [Google Scholar] [CrossRef]
- Reichert, J.; Binner, J.G.P. An evaluation of hydroxyapatite-based filters for removal of heavy metal ions from aqueous solutions. J. Mater. Sci. 1996, 31, 1231–1241. [Google Scholar] [CrossRef]
- Joshi, P.; Manocha, S. Kinetic and thermodynamic studies of the adsorption of copper ions on hydroxyapatite nanoparticles. Mater. Today: Proc. 2017, 4, 10455–10459. [Google Scholar] [CrossRef]
- Iconaru, L.S.; Motelica-Heino, M.; Guegan, R.; Beuran, M.; Costescu, A.; Predoi, D. Adsorption of Pb (II) Ions onto Hydroxyapatite Nanopowders in Aqueous Solutions. Materials 2018, 11, 2204. [Google Scholar] [CrossRef] [PubMed]
- Janusz, W.; Skwarek, E. Study of sorption processes of strontium on the synthetic hydroxyapatite. Adsorption 2016, 22, 697–706. [Google Scholar] [CrossRef]
- Flores-Cano, J.V.; Leyva-Ramos, R.; Carrasco-Marin, F.; Aragón-Piña, A.; Salazar-Rabago, J.J.; Leyva-Ramos, S. Adsorption mechanism of Chromium(III) from water solution on bone char: Effect of operating conditions. Adsorption 2016, 22, 297–308. [Google Scholar] [CrossRef]
- Okazaki, Y.; Gotoh, E. Comparison of metal release from various metallic biomaterials in vitro. Biomaterials 2005, 26, 11–21. [Google Scholar] [CrossRef]
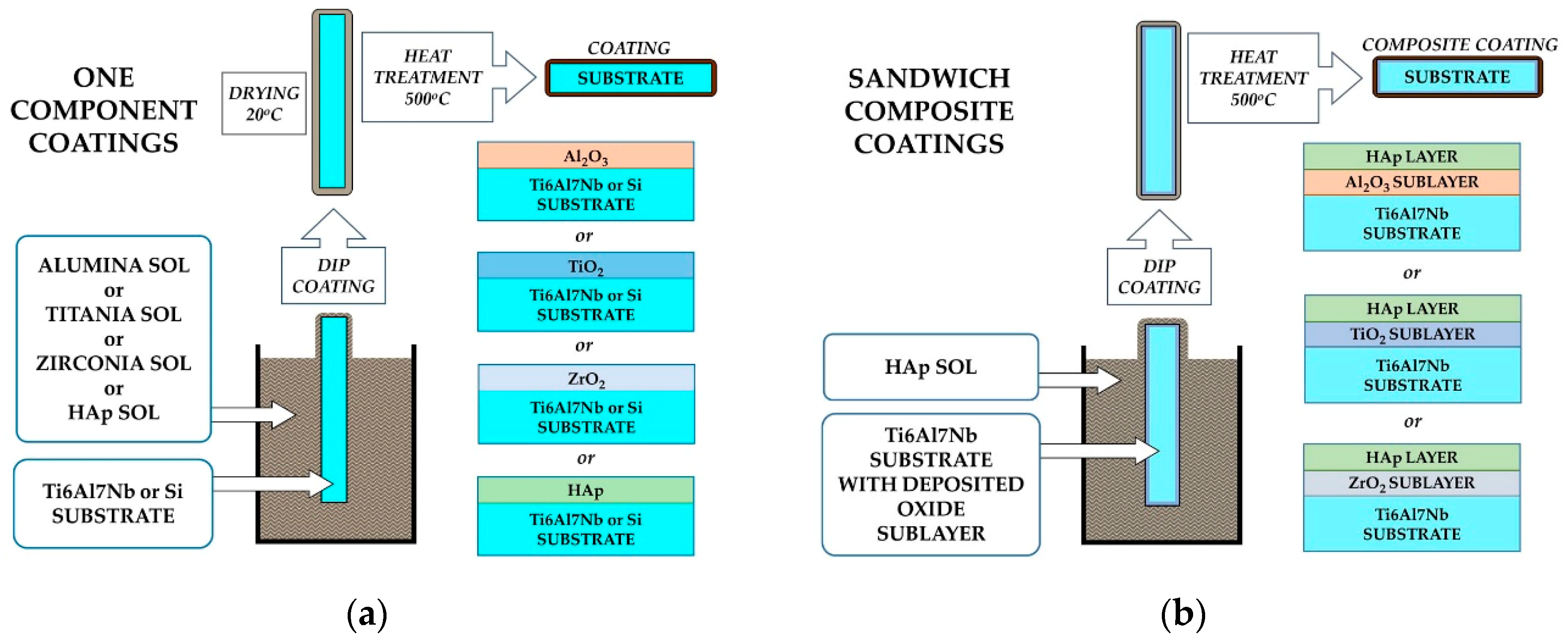


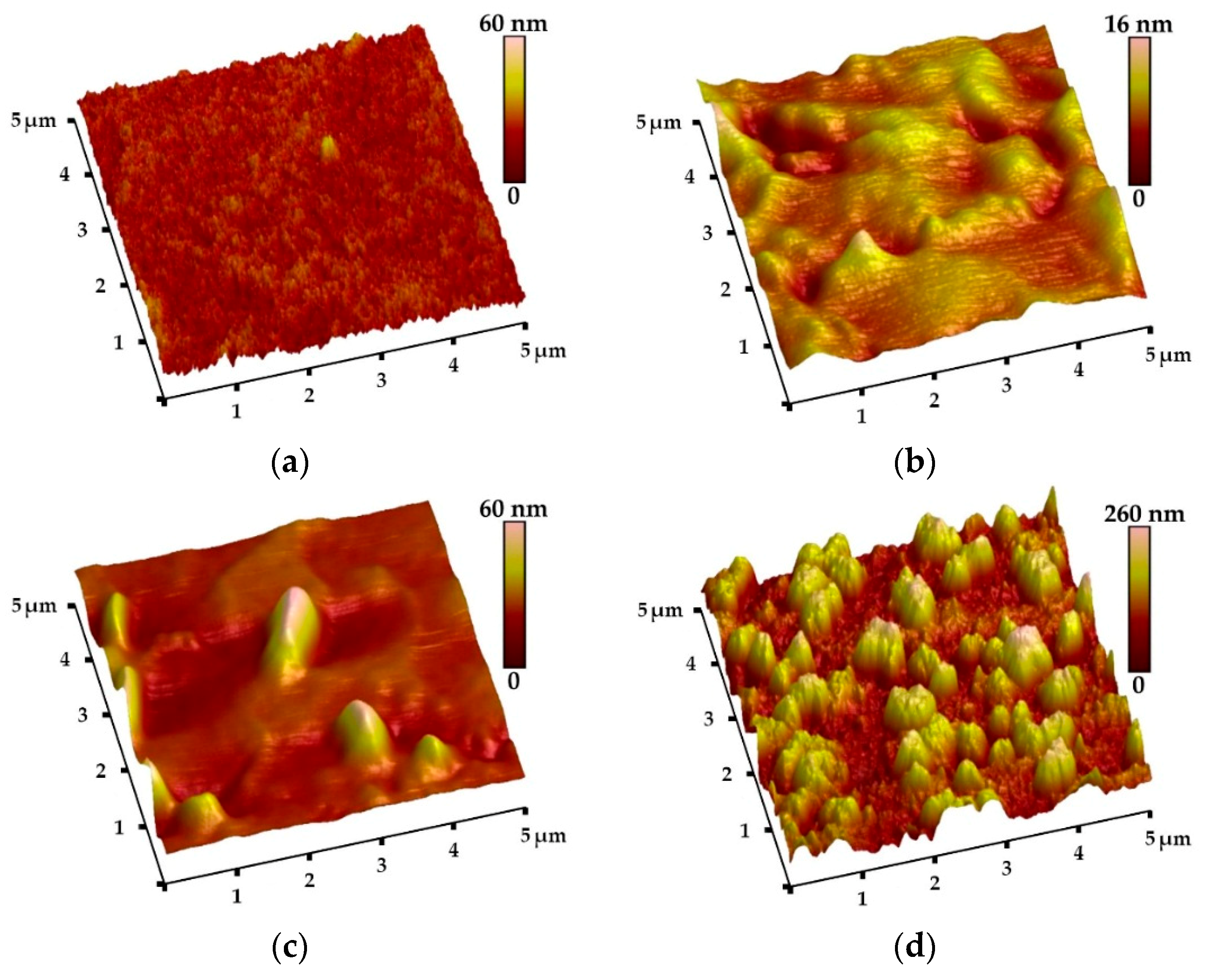

| Coating Type | Pulling Out Speed [mm/s] | Drying Time [Minutes] | Heat Treatment Temperature [°C] | Heat Treatment Time [Minutes] |
|---|---|---|---|---|
| TiO2 | 1.2 | 15 | 500 | 15 |
| Al2O3 | ||||
| ZrO2 | ||||
| HAp | 1.2 | − | 500 | 20 |
| Ti6Al7Nb | TiO2 | Al2O3 | ZrO2 | HAp | |
|---|---|---|---|---|---|
| Ra [nm] | 0.34 ± 0.10 | 2.27 ± 0.34 | 0.60 ± 0.27 | 1.45 ± 1.34 | 29.1 ± 7.82 |
| Rz [nm] | 2.37 ± 0.66 | 15.3 ± 2.96 | 2.85 ± 1.47 | 8.69 ± 7.74 | 131 ± 23.2 |
| TiO2 | Al2O3 | ZrO2 | TZrO2 | HAp | |
|---|---|---|---|---|---|
| Thickness [nm] | 81.49 ± 0.30 | 86.48 ± 0.46 | 70.93 ± 0.62 | 48.8 ± 0.11 | 175 ± 25 |
| Ti6Al7Nb | TiO2 | Al2O3 | TZrO2 | HAp | |
|---|---|---|---|---|---|
| Permeation of Al ions [µg/cm2] | 1.072 ± 0.044 | 0.708 ± 0.052 | 0 | 0.011 ± 0.023 | 0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pietrzyk, B.; Kucharski, D.; Kołodziejczyk, Ł.; Miszczak, S.; Fijalkowski, M. Comparison of Mechanical and Barrier Properties of Al2O3/TiO2/ZrO2 Layers in Oxide–Hydroxyapatite Sandwich Composite Coatings Deposited by Sol–Gel Method on Ti6Al7Nb Alloy. Materials 2020, 13, 502. https://doi.org/10.3390/ma13030502
Pietrzyk B, Kucharski D, Kołodziejczyk Ł, Miszczak S, Fijalkowski M. Comparison of Mechanical and Barrier Properties of Al2O3/TiO2/ZrO2 Layers in Oxide–Hydroxyapatite Sandwich Composite Coatings Deposited by Sol–Gel Method on Ti6Al7Nb Alloy. Materials. 2020; 13(3):502. https://doi.org/10.3390/ma13030502
Chicago/Turabian StylePietrzyk, Bożena, Daniel Kucharski, Łukasz Kołodziejczyk, Sebastian Miszczak, and Mateusz Fijalkowski. 2020. "Comparison of Mechanical and Barrier Properties of Al2O3/TiO2/ZrO2 Layers in Oxide–Hydroxyapatite Sandwich Composite Coatings Deposited by Sol–Gel Method on Ti6Al7Nb Alloy" Materials 13, no. 3: 502. https://doi.org/10.3390/ma13030502
APA StylePietrzyk, B., Kucharski, D., Kołodziejczyk, Ł., Miszczak, S., & Fijalkowski, M. (2020). Comparison of Mechanical and Barrier Properties of Al2O3/TiO2/ZrO2 Layers in Oxide–Hydroxyapatite Sandwich Composite Coatings Deposited by Sol–Gel Method on Ti6Al7Nb Alloy. Materials, 13(3), 502. https://doi.org/10.3390/ma13030502






