Blended Natural Support Materials—Collagen Based Hydrogels Used in Biomedicine
Abstract
1. Introduction
2. Biopolymers Structure and Properties
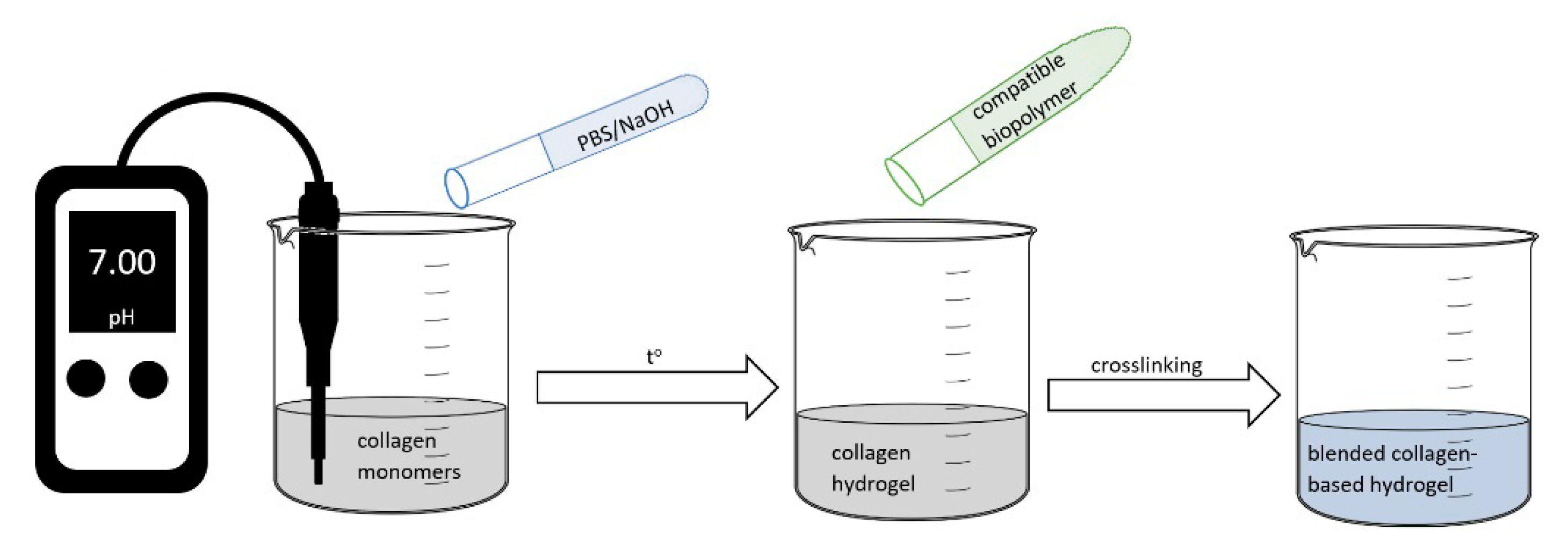
3. Blended Collagen-Based Formulations—Synthesis, Characterization, and Properties
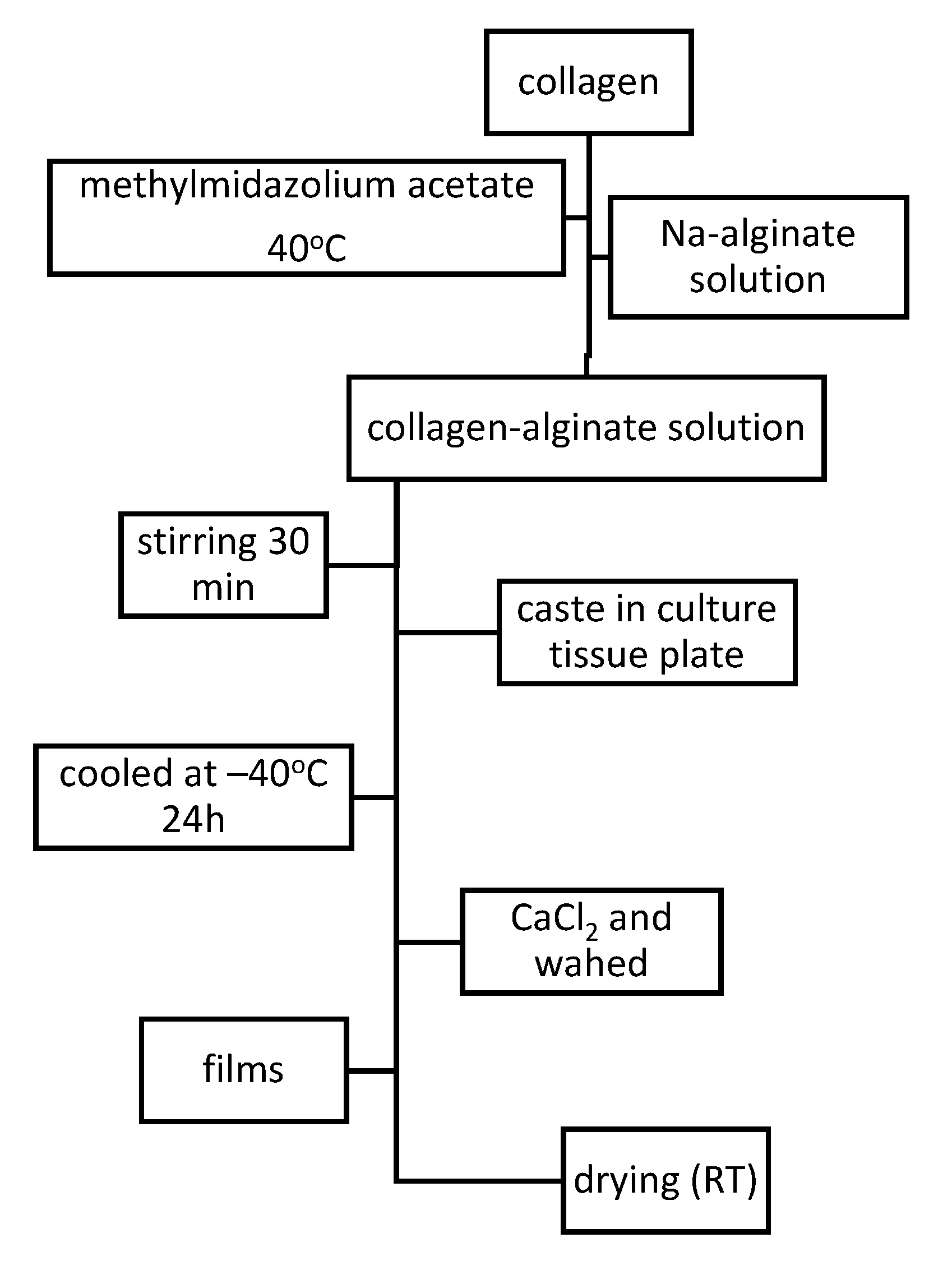
3.1. Blended Collagen–Alginate Biomaterials
3.1.1. Synthesis and Characterization of Collagen–Alginate Aerogels
3.1.2. Synthesis and Characterization of Collagen–Alginate Films
3.1.3. Synthesis and Characterization of Collagen–Alginate Hydrogels
3.2. Blended Collagen–Chitosan Biomaterials
3.3. Blended Collagen–Cellulose Biomaterials
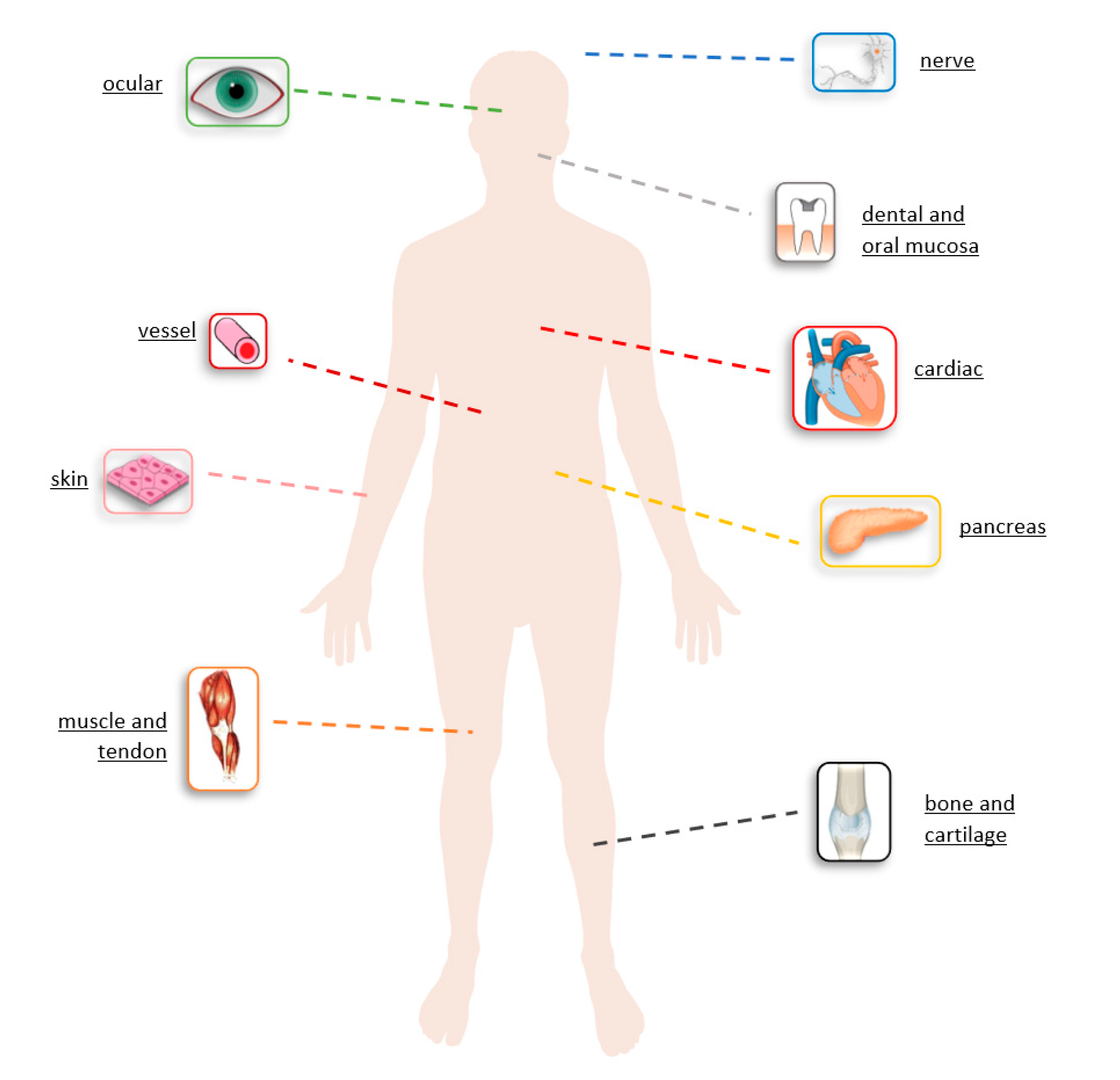
4. Biomedical Pharmaceutical Application for Blended Collagen-Biomaterials
4.1. Drug Delivery Systems Based on Blended Collagen-Hydrogels
4.2. Potential Application of Blended Collagen-Hydrogels in Tissue Repair and Engineering
5. Conclusions and Future Approaches
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- An, B.; Lin, Y.-S.; Brodsky, B. Collagen interactions: Drug design and delivery. Adv. Drug Deliv. Rev. 2016, 97, 69–84. [Google Scholar] [CrossRef] [PubMed]
- Chvapil, M.; Kronenthal, R.L.; Van Winkle, W. Medical and Surgical Applications of Collagen. Int. Rev. Connect. Tissue Res. 1973, 6, 1–61. [Google Scholar] [CrossRef] [PubMed]
- Cheng, W.; Yan-hua, R.; Fang-gang, N.; Guo-an, Z. The content and ratio of type I and III collagen in skin differ withage and injury. Afr. J. Biotechnol. 2011, 10, 2524–2529. [Google Scholar]
- Ramshaw, J.A.; Peng, Y.Y.; Glattauer, V.; Werkmeister, J.A. Collagens as biomaterials. J. Mater. Sci. Mater. Med. 2009, 20, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Bretaud, S.; Guillon, E.; Karppinen, S.-M.; Pihlajaniemi, T.; Ruggiero, F. Collagen XV, a multifaceted multiplexin present across tissues and species. Matrix Biol. Plus 2020, 6–7, 100023. [Google Scholar] [CrossRef]
- Lin, K.; Zhang, D.; Macedo, M.H.; Cui, W.; Sarmento, B.; Shen, G. Advanced Collagen-Based Biomaterials for Regenerative Biomedicine. Adv. Funct. Mater. 2019, 29, 1804943. [Google Scholar] [CrossRef]
- Antoine, E.E.; Vlachos, P.P.; Rylander, M.N. Review of Collagen I Hydrogels for Bioengineered Tissue Microenvironments: Characterization of Mechanics, Structure, and Transport. Tissue Eng. Part B Rev. 2014, 20, 683–696. [Google Scholar] [CrossRef]
- Parenteau-Bareil, R.; Gauvin, R.; Berthod, F. Collagen-Based Biomaterials for Tissue Engineering Applications. Materials 2010, 3, 1863–1887. [Google Scholar] [CrossRef]
- Andonegi, M.; Heras, K.L.; Santos-Vizcaino, E.; Igartua, M.; Hernandez, R.M.; De La Caba, K.; Guerrero, P. Structure-properties relationship of chitosan/collagen films with potential for biomedical applications. Carbohydr. Polym. 2020, 237, 116159. [Google Scholar] [CrossRef]
- Uzel, S.G.; Buehler, M.J. Molecular structure, mechanical behavior and failure mechanism of the C-terminal cross-link domain in type I collagen. J. Mech. Behav. Biomed. Mater. 2011, 4, 153–161. [Google Scholar] [CrossRef]
- Voicu, G.; Geanaliu-Nicolae, R.-E.; Pîrvan, A.-A.; Andronescu, E.; Florin, I. Synthesis, characterization and bioevaluation of drug-collagen hybrid materials for biomedical applications. Int. J. Pharm. 2016, 510, 474–484. [Google Scholar] [CrossRef] [PubMed]
- Voicu, G.; Geanaliu, R.E.; Ghitulica, C.D.; Ficai, A.; Grumezescu, A.M.; Bleotu, C. Synthesis, characterization and bioevaluation of irinotecan-collagen hybrid materials for biomedical applications as drug delivery systems in tumoral treatments. Cent. Eur. J. Chem. 2013, 11, 2134–2143. [Google Scholar] [CrossRef]
- Ahmadi, F.; Oveisi, Z.; Mohammadi-Samani, S.; Amoozgar, Z. Chitosan based hydrogels: Characteristics and pharmaceutical applications. Res. Pharm. Sci. 2015, 10, 1–16. [Google Scholar] [PubMed]
- Bahram, M.; Mohseni, N.; Moghtader, M. An Introduction to Hydrogels and Some Recent Applications; IntechOpen: London, UK, 2016. [Google Scholar]
- Chvapil, M. Collagen sponge: Theory and practice of medical applications. J. Biomed. Mater. Res. 1977, 11, 721–741. [Google Scholar] [CrossRef]
- Gu, L.; Shan, T.; Ma, Y.-X.; Tay, F.R.; Niu, L.-N. Novel Biomedical Applications of Crosslinked Collagen. Trends Biotechnol. 2019, 37, 464–491. [Google Scholar] [CrossRef]
- Chang, S.J.; Kuo, S.M.; Manousakas, I.; Niu, G.C.-C.; Chen, J.P. Preparation and characterization of hyaluronan/collagen II microspheres under an electrostatic field system with disc electrodes. Acta Biomater. 2009, 5, 101–114. [Google Scholar] [CrossRef]
- Matsunaga, Y.T.; Morimoto, Y.; Takeuchi, S. Molding Cell Beads for Rapid Construction of Macroscopic 3D Tissue Architecture. Adv. Mater. 2011, 23, H90–H94. [Google Scholar] [CrossRef]
- Hong, S.; Hsu, H.-J.; Kaunas, R.; Kameoka, J. Collagen microsphere production on a chip. Lab Chip 2012, 12, 3277. [Google Scholar] [CrossRef]
- Pati, F.; Adhikari, B.; Dhara, S. Collagen Intermingled Chitosan-Tripolyphosphate Nano/Micro Fibrous Scaffolds for Tissue-Engineering Application. J. Biomater. Sci. Polym. 2012, 23, 1923–1938. [Google Scholar] [CrossRef]
- Siriwardane, M.L.; DeRosa, K.; Collins, G.; Pfister, B.J. Controlled formation of cross-linked collagen fibers for neural tissue engineering applications. Biofabrication 2014, 6, 015012. [Google Scholar] [CrossRef]
- Sell, S.A.; McClure, M.J.; Garg, K.; Wolfe, P.S.; Bowlin, G.L. Electrospinning of collagen/biopolymers for regenerative medicine and cardiovascular tissue engineering. Adv. Drug Deliv. Rev. 2009, 61, 1007–1019. [Google Scholar] [CrossRef] [PubMed]
- Ardelean, I.L.; Gudovan, D.; Ficai, D.; Ficai, A.; Andronescu, E.; Albu-Kaya, M.G.; Neacsu, P.; Ion, R.N.; Cimpean, A.; Mitran, V. Collagen/hydroxyapatite bone grafts manufactured by homogeneous/heterogeneous 3D printing. Mater. Lett. 2018, 231, 179–182. [Google Scholar] [CrossRef]
- Karki, S.; Kim, H.; Na, S.-J.; Shin, D.; Jo, K.; Lee, J. Thin films as an emerging platform for drug delivery. Asian J. Pharm. Sci. 2016, 11, 559–574. [Google Scholar] [CrossRef]
- Suchý, T.; Šupová, M.; Sauerová, P.; Verdánová, M.; Sucharda, Z.; Rýglová, Š.; Žaloudková, M.; Sedláček, R.; Kalbáčová, M.H. The effects of different cross-linking conditions on collagen-based nanocomposite scaffolds-an in vitro evaluation using mesenchymal stem cells. Biomed. Mater. 2015, 10, 065008. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, D.V.; Shepherd, J.H.; Ghose, S.; Kew, S.J.; Cameron, R.E.; Best, S.M. The process of EDC-NHS cross-linking of reconstituted collagen fibres increases collagen fibrillar order and alignment. APL Mater. 2015, 3, 014902. [Google Scholar] [CrossRef] [PubMed]
- Oryan, A.; Kamali, A.; Moshiri, A.; Baharvand, H.; Daemi, H. Chemical crosslinking of biopolymeric scaffolds: Current knowledge and future directions of crosslinked engineered bone scaffolds. Int. J. Biol. Macromol. 2018, 107, 678–688. [Google Scholar] [CrossRef] [PubMed]
- Davidenko, N.; Bax, D.V.; Schuster, C.F.; Farndale, R.W.; Hamaia, S.W.; Best, S.M.; Cameron, R.E. Optimisation of UV irradiation as a binding site conserving method for crosslinking collagen-based scaffolds. J. Mater. Sci. Mater. Med. 2016, 27, 14. [Google Scholar] [CrossRef]
- Ziaei, M.; Barsam, A.; Shamie, N.; Vroman, D.; Kim, T.; Donnenfeld, E.D.; Holland, E.J.; Kanellopoulos, J.; Mah, F.S.; Randleman, B.J.; et al. Reshaping procedures for the surgical management of corneal ectasia. J. Cataract. Refract. Surg. 2015, 41, 842–872. [Google Scholar] [CrossRef]
- Bazrafshan, Z.; Stylios, G.K. Spinnability of collagen as a biomimetic material: A review. Int. J. Biol. Macromol. 2019, 129, 693–705. [Google Scholar] [CrossRef]
- Lee, K.Y.; Mooney, D.J. Alginate: Properties and biomedical applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef]
- Hamedi, H.; Moradi, S.; Hudson, S.M.; Tonelli, A.E. Chitosan based hydrogels and their applications for drug delivery in wound dressings: A review. Carbohydr. Polym. 2018, 199, 445–460. [Google Scholar] [CrossRef] [PubMed]
- Picheth, G.F.; Pirich, C.L.; Sierakowski, M.R.; Woehl, M.A.; Sakakibara, C.N.; De Souza, C.F.; Martin, A.A.; Da Silva, R.; De Freitas, R.A. Bacterial cellulose in biomedical applications: A review. Int. J. Biol. Macromol. 2017, 104, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Ren, L.; Yao, H.; Wang, Y. Collagen films with suitable physical properties and biocompatibility for corneal tissue engineering prepared by ion leaching technique. Mater. Lett. 2012, 87, 1–4. [Google Scholar] [CrossRef]
- Duan, L.; Yuan, Q.; Xiang, H.; Yang, X.; Liu, L.; Li, J. Fabrication and characterization of a novel collagen-catechol hydrogel. J. Biomater. Appl. 2017, 32, 862–870. [Google Scholar] [CrossRef]
- Wu, X.; Liu, A.; Wang, W.; Ye, R. Improved mechanical properties and thermal-stability of collagen fiber based film by crosslinking with casein, keratin or SPI: Effect of crosslinking process and concentrations of proteins. Int. J. Biol. Macromol. 2018, 109, 1319–1328. [Google Scholar] [CrossRef]
- Broguiere, N.; Formica, F.A.; Barreto, G.; Zenobi-Wong, M. Sortase A as a cross-linking enzyme in tissue engineering. Acta Biomater. 2018, 77, 182–190. [Google Scholar] [CrossRef]
- Hu, W.; Wang, Z.; Xiao, Y.; Zhang, S.; Wang, J. Advances in crosslinking strategies of biomedical hydrogels. Biomater. Sci. 2019, 7, 843–855. [Google Scholar] [CrossRef]
- Jabrail, F.H.; Al-Ojar, R.K. Studies on Preparation and Characterization of Blend Polymers for Hydrogels Synthesis and Use for Protein Release. Rafidain J. Sci. 2019, 28, 211–227. [Google Scholar] [CrossRef]
- Bouhadir, K.H.; Lee, K.Y.; Alsberg, E.; Damm, K.L.; Anderson, K.W.; Shetye, S. Degradation of Partially Oxidized Alginate and Its Potential Application for Tissue Engineering. Biotechnol. Prog. 2001, 17, 945–950. [Google Scholar] [CrossRef]
- Yang, X.; Guo, L.; Fan, Y.; Zhang, X. Preparation and characterization of macromolecule cross-linked collagen hydrogels for chondrocyte delivery. Int. J. Biol. Macromol. 2013, 61, 487–493. [Google Scholar] [CrossRef]
- Baldino, L.; Concilio, S.; Cardea, S.; Reverchon, E. Interpenetration of Natural Polymer Aerogels by Supercritical Drying. Polymers 2016, 8, 106. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, B.; Muhammad, N.; Jamal, A.; Ahmad, P.; Khan, Z.U.H.; Rahim, A.; Khan, A.S.; Gonfa, G.; Iqbal, J.; Rehman, I.U. An application of ionic liquid for preparation of homogeneous collagen and alginate hydrogels for skin dressing. J. Mol. Liq. 2017, 243, 720–725. [Google Scholar] [CrossRef]
- Moxon, S.R.; Corbett, N.J.; Fisher, K.; Potjewyd, G.; Domingos, M.; Hooper, N.M. Blended alginate/collagen hydrogels promote neurogenesis and neuronal maturation. Mater. Sci. Eng. C 2019, 104, 109904. [Google Scholar] [CrossRef] [PubMed]
- Montalbano, G.; Toumpaniari, S.; Popov, A.; Duan, P.; Chen, J.; Dalgarno, K.; Scott, W.; Ferreira, A.M. Synthesis of bioinspired collagen/alginate/fibrin based hydrogels for soft tissue engineering. Mater. Sci. Eng. C 2018, 91, 236–246. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Stegemann, J.P. Thermogelling chitosan and collagen composite hydrogels initiated with β-glycerophosphate for bone tissue engineering. Biomaterials 2010, 31, 3976–3985. [Google Scholar] [CrossRef]
- Rao, R.R.; Jiao, A.; Kohn, D.H.; Stegemann, J.P. Exogenous mineralization of cell-seeded and unseeded collagen–chitosan hydrogels using modified culture medium. Acta Biomater. 2012, 8, 1560–1565. [Google Scholar] [CrossRef]
- Sionkowska, A.; Kaczmarek, B.; Lewandowska, K. Modification of collagen and chitosan mixtures by the addition of tannic acid. J. Mol. Liq. 2014, 199, 318–323. [Google Scholar] [CrossRef]
- Teng, S.-H.; Liang, M.-H.; Wang, P.; Luo, Y. Biomimetic composite microspheres of collagen/chitosan/nano-hydroxyapatite: In-Situ synthesis and characterization. Mater. Sci. Eng. C 2016, 58, 610–613. [Google Scholar] [CrossRef]
- Tylingo, R.; Gorczyca, G.; Mania, S.; Szweda, P.; Milewski, S. Preparation and characterization of porous scaffolds from chitosan-collagen-gelatin composite. React. Funct. Polym. 2016, 103, 131–140. [Google Scholar] [CrossRef]
- Thanyacharoen, T.; Chuysinuan, P.; Techasakul, S.; Nooeaid, P.; Ummartyotin, S. Development of a gallic acid-loaded chitosan and polyvinyl alcohol hydrogel composite: Release characteristics and antioxidant activity. Int. J. Biol. Macromol. 2018, 107, 363–370. [Google Scholar] [CrossRef]
- Thanyacharoena, T.; Chuysinuan, P.; Techasakul, S.; Noenplab, A.N.L.; Ummartyotin, S. The chemical composition and antioxidant and release properties of a black rice (Oryzasativa L.)-loaded chitosan and polyvinyl alcohol composite. J. Mol. Liq. 2017, 248, 1065–1070. [Google Scholar] [CrossRef]
- Chunshom, N.; Chuysinuan, P.; Thanyacharoena, T.; Techasakul, S.; Ummartyotin, S. Development of gallic acid/cyclodextrininclusion complex in freeze-dried bacterial cellulose andpoly (vinyl alcohol) hydrogel: Controlled-releasecharacteristic and antioxidant properties. Mater. Chem. Phys. 2019, 232, 294–300. [Google Scholar] [CrossRef]
- Perez-Puyana, V.; Jiménez-Rosado, M.; Romero, A.; Guerrero, A. Crosslinking of hybrid scaffolds produced from collagen and chitosan. Int. J. Biol. Macromol. 2019, 139, 262–269. [Google Scholar] [CrossRef]
- Thongchai, K.; Chuysinuan, P.; Thanyacharoen, T.; Techasakul, S.; Ummartyotin, S. Characterization, release, and antioxidant activity of caffeic acid-loaded collagen and chitosan hydrogel composites. J. Mater. Res. Technol. 2020, 9, 6512–6520. [Google Scholar] [CrossRef]
- Sionkowska, A.; Walczak, M.; Michalska-Sionkowska, M. Preparation and characterization of collagen/chitosan composites with silver nanoparticles. Polym. Compos. 2020, 41, 951–957. [Google Scholar] [CrossRef]
- Si, J.; Yang, Y.; Xing, X.; Yang, F.; Shan, P. Controlled degradable chitosan/collagen composite scaffolds for application in nerve tissue regeneration. Polym. Degrad. Stab. 2019, 166, 73–85. [Google Scholar] [CrossRef]
- Gilarska, A.; Lewandowska-Łańcucka, J.; Horakc, W.; Nowakowska, M. Collagen/chitosan/hyaluronic acid–based injectable hydrogels for tissue engineering applications–design, physicochemical and biological characterization. Colloids Surf. B Biointerfaces 2018, 170, 152–162. [Google Scholar] [CrossRef]
- Zhijiang, C.; Guang, Y. Bacterial cellulose/collagen composite: Characterization and first evaluation of cytocompatibility. J. Appl. Polym. Sci. 2011, 120, 2938–2944. [Google Scholar] [CrossRef]
- Zhang, N.; Chen, Y.; Zhao, Y.; Fan, D.; Li, L.; Yan, B.; Tao, G.; Zhao, J.; Zhang, H.; Wang, M. Caffeic acid assists microwave heating to inhibit the formation of mutagenic and carcinogenic PhIP. Food Chem. 2020, 317, 126447. [Google Scholar] [CrossRef]
- Yamatani, K.; Kawatani, R.; Ajiro, H. Synthesis of glucosamine derivative with double caffeic acid moieties at N– and 6-O-positions for developments of natural based materials. J. Mol. Struct. 2020, 1206, 127689. [Google Scholar] [CrossRef]
- Neiva, D.M.; Araújo, S.; Gominho, J.; Carneiro, A.D.C.; Pereira, H. Potential of Eucalyptus globulus industrial bark as a biorefinery feedstock: Chemical and fuel characterization. Ind. Crops Prod. 2018, 123, 262–270. [Google Scholar] [CrossRef]
- Rodrigues, V.H.; De Melo, M.M.; Portugal, I.; Silva, C.M. Simulation and techno-economic optimization of the supercritical CO2 extraction of Eucalyptus globulus bark at industrial scale. J. Supercrit. Fluids 2019, 145, 169–180. [Google Scholar] [CrossRef]
- Cruz, J.M.; Domınguez, H.; Parajó, J.C. Antioxidant activity of isolates from acid hydrolysates of Eucalyptus globulus wood. Food Chem. 2005, 90, 503–511. [Google Scholar] [CrossRef]
- Spagnol, C.M.; Assis, R.P.; Brunetti, I.L.; Isaac, V.L.B.; Salgado, H.R.N.; Correa, M.A. In vitro methods to determine the antioxidant activity of caffeic acid. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2019, 219, 358–366. [Google Scholar] [CrossRef]
- Chuysinuan, P.; Chimnoi, N.; Reuk-Ngam, N.; Khlaychan, P.; Makarasen, A.; Wetprasit, N.; Dechtrirat, D.; Supaphol, P.; Techasakul, S. Development of gelatin hydrogel pads incorporated with Eupatorium adenophorum essential oil as antibacterial wound dressing. Polym. Bull. 2018, 76, 701–724. [Google Scholar] [CrossRef]
- Liu, T.; Dan, W.; Dan, N.; Liu, X.; Liu, X.; Peng, X. A novel grapheme oxide-modified collagen-chitosan bio-film for controlled growth factor release in wound healing applications. Mater. Sci. Eng. 2017, 77, 202–211. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.W.; Hsu, Y.-H.; Liao, J.-Y.; Liu, S.-J.; Chen, J.-K.; Ueng, S.W.-N. Sustainable release of vancomycin, gentamicin and lidocaine from novel electrospun sandwich-structured PLGA/collagen nanofibrous membranes. Int. J. Pharm. 2012, 430, 335–341. [Google Scholar] [CrossRef]
- Alagha, A.; Nourallah, A.; Hariri, S. Characterization of dexamethasone loaded collagen-chitosan sponge and in vitro release study. J. Drug Deliv. Sci. Technol. 2020, 55, 101449. [Google Scholar] [CrossRef]
- Zhang, W.; Wang, X.-C.; Wang, J.-J.; Zhang, L.-L. Drugs adsorption and release behavior of collagen/bacterial cellulose porous microspheres. Int. J. Biol. Macromol. 2019, 140, 196–205. [Google Scholar] [CrossRef]
- Lee, M.; Lo, A.C.Y.; Cheung, P.; Wong, D.; Chan, B.P. Drug carrier systems based on collagen–alginate composite structures for improving the performance of GDNF-secreting HEK293 cells. Biomaterials 2009, 30, 1214–1221. [Google Scholar] [CrossRef]
- Huang, Z.; Feng, Q.; Yu, B.; Li, S. Biomimetic properties of an injectable chitosan/nano-hydroxyapatite/collagen composite. Mater. Sci. Eng. C 2011, 31, 683–687. [Google Scholar] [CrossRef]
- Sotome, S.; Uemura, T.; Kikuchi, M.; Chen, J.; Itoh, S.; Tanaka, J.; Tateishi, T.; Shinomiya, K. Synthesis and in vivo evaluation of a novel hydroxyapatite/collagen–alginate as a bone filler and a drug delivery carrier of bone morphogenetic protein. Mater. Sci. Eng. C 2004, 24, 341–347. [Google Scholar] [CrossRef]
- Wong, F.S.Y.; Tsang, K.K.; Chu, A.M.W.; Chan, B.P.; Yao, K.M.; Lo, A.C.Y. Injectable cell-encapsulating composite alginate-collagen platform with inducible termination switch for safer ocular drug delivery. Biomaterials 2019, 201, 53–67. [Google Scholar] [CrossRef] [PubMed]
- Mu, H.; Wang, Y.; Wei, H.; Lu, H.; Feng, Z.; Yu, H.; Xing, Y.; Wang, H. Collagen peptide modified carboxymethyl cellulose as both antioxidant drug and carrier for drug delivery against retinal ischaemia/reperfusion injury. J. Cell. Mol. Med. 2018, 22, 5008–5019. [Google Scholar] [CrossRef] [PubMed]
- Jahanban-Esfahlan, R.; Derakhshankhah, H.; Haghshenas, B.; Massoumi, B.; Abbasian, M.; Jaymand, M. A bio-inspired magnetic natural hydrogel containing gelatin and alginate as a drug delivery system for cancer chemotherapy. Int. J. Biol. Macromol. 2020, 156, 438–445. [Google Scholar] [CrossRef]
- Mei, E.; Li, S.; Song, J.; Xing, R.; Li, Z.; Yan, X. Self-assembling Collagen/Alginate hybrid hydrogels for combinatorial photothermal and immuno tumor therapy. Colloids Surf. A Physicochem. Eng. Asp. 2019, 577, 570–575. [Google Scholar] [CrossRef]
- Fan, X.; Liang, Y.; Cui, Y.; Li, F.; Sun, Y.; Yang, J.; Song, H.; Bao, Z.; Nian, Z. Development of tilapia collagen and chitosan composite hydrogels for nanobody delivery. Colloids Surf. B Biointerfaces 2020, 195, 111261. [Google Scholar] [CrossRef]
- Chen, X.; Fan, M.; Tan, H.; Ren, B.; Yuan, G.; Jia, Y.; Li, J.; Xiong, D.; Xing, X.; Niu, X.; et al. Magnetic and self-healing chitosan-alginate hydrogel encapsulated gelatin microspheres via covalent cross-linking for drug delivery. Mater. Sci. Eng. C 2019, 101, 619–629. [Google Scholar] [CrossRef]
- Barroso, T.; Viveiros, R.; Casimiro, T.; Aguiar-Ricardo, A. Development of dual-responsive chitosan–collagen scaffolds for pulsatile release of bioactive molecules. J. Supercrit. Fluids 2014, 94, 102–112. [Google Scholar] [CrossRef]
- Abdel-Mohsen, A.M.; Frankova, J.; Abdel-Rahman, R.M.; Salem, A.A.; Sahffie, N.M.; Kubena, I.; Jancarabh, J. Chitosan-glucan complex hollow fibers reinforced collagen wound dressing embedded with aloe vera. II. Multifunctional properties to promote cutaneous wound healing. Int. J. Pharm. 2020, 582, 119349. [Google Scholar] [CrossRef]
- Karri, V.V.S.R.; Kuppusamy, G.; Talluri, S.V.; Mannemala, S.S.; Kollipara, R.; Wadhwani, A.D.; Mulukutla, S.; Raju, K.R.S.; Malayandi, R. Curcumin loaded chitosan nanoparticles impregnated into collagen-alginate scaffolds for diabetic wound healing. Int. J. Biol. Macromol. 2016, 93, 1519–1529. [Google Scholar] [CrossRef] [PubMed]
- Reis, L.A.; Chiu, L.L.; Liang, Y.; Hyunh, K.; Momen, A.; Radisic, M. A peptide-modified chitosan–collagen hydrogel for cardiac cell culture and delivery. Acta Biomater. 2012, 8, 1022–1036. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, A.A.; Salama, A.H. Norfloxacin-loaded collagen/chitosan scaffolds for skin reconstruction: Preparation, evaluation and in-vivo wound healing assessment. Eur. J. Pharm. Sci. 2016, 83, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Thacharodi, D. Collagen-chitosan composite membranes controlled transdermal delivery of nifedipine and propranolol hydrochloride. Int. J. Pharm. 1996, 134, 239–241. [Google Scholar] [CrossRef]
- Sundaramurthy, A.; Krishnamoorthy, G.; Ramkumar, K.; Raichur, A. Preparation of collagen peptide functionalized chitosan nanoparticles by ionic gelation method: An effective carrier system for encapsulation and release of doxorubicin for cancer drug delivery. Mater. Sci. Eng. C 2017, 70, 378–385. [Google Scholar] [CrossRef]
- Schlapp, M.; Friess, W. Collagen/PLGA Microparticle Composites for Local Controlled Delivery of Gentamicin. J. Pharm. Sci. 2003, 92, 2145–2151. [Google Scholar] [CrossRef]
- Di Martino, A.; Drannikov, A.; Surgutskaia, N.S.; Ozaltin, K.; Postnikov, P.S.; Marina, T.E.; Sedlarik, V. Chitosan-collagen based film for controlled delivery of a combination of short life anesthetics. Int. J. Biol. Macromol. 2019, 140, 1183–1193. [Google Scholar] [CrossRef]
- Michalska-Sionkowska, M.; Kaczmarek, B.; Walczak, M.; Sionkowska, A. Antimicrobial activity of new materials based on the blends of collagen/chitosan/hyaluronic acid with gentamicin sulfate addition. Mater. Sci. Eng. C 2018, 86, 103–108. [Google Scholar] [CrossRef]
- Chen, M.; Huang, Y.-Q.; Cao, H.; Liu, Y.; Guo, H.; Chen, L.S.; Wang, J.-H.; Zhang, Q. Collagen/chitosan film containing biotinylated glycol chitosan nanoparticles for localized drug delivery. Colloids Surf. B Biointerfaces 2015, 128, 339–346. [Google Scholar] [CrossRef]
- Cao, H.; Chen, M.-M.; Liu, Y.; Liu, Y.-Y.; Huang, Y.-Q.; Wang, J.-H.; Chen, J.-D.; Zhang, Q. Fish collagen-based scaffold containing PLGA microspheres for controlled growth factor delivery in skin tissue engineering. Colloids Surf. B Biointerfaces 2015, 136, 1098–1106. [Google Scholar] [CrossRef]
- Sionkowska, A.; Kaczmarek, B.; Gadzała-Kopciuch, R. Gentamicin release from chitosan and collagen composites. J. Drug Deliv. Sci. Technol. 2016, 35, 353–359. [Google Scholar] [CrossRef]
- Kosaric, N.; Kiwanuka, H.; Gurtner, G.C. Stem cell therapies for wound healing. Expert Opin. Biol. Ther. 2019, 19, 575–585. [Google Scholar] [CrossRef]
- Maxson, S.; Lopez, E.A.; Yoo, D.; Danilkovitch-Miagkova, A.; Leroux, M.A. Concise Review: Role of Mesenchymal Stem Cells in Wound Repair. Stem Cells Transl. Med. 2012, 1, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Nauta, A.C.; Gurtner, G.C.; Longaker, M.T. Adult Stem Cells in Small Animal Wound Healing Models. Methods Mol. Biol. 2013, 1037, 81–98. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Wang, X.-C.; Li, X.-Y.; Zhang, L.-L.; Jiang, F. A 3D porous microsphere with multistage structure and component based on bacterial cellulose and collagen for bone tissue engineering. Carbohydr. Polym. 2020, 236, 116043. [Google Scholar] [CrossRef] [PubMed]
- Mansouri, V.; Salehi, M.; Omrani, M.D.; Niknam, Z.; Ardeshirylajimi, A. Collagen-alginate microspheres as a 3D culture system for mouse embryonic stem cells differentiation to primordial germ cells. Biologicals 2017, 48, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Khatami, N.; Khoshfetrat, A.B.; Khaksar, M.; Zamani, A.R.N.; Rahbarghazi, R. Collagen-alginate-nano-silica microspheres improved the osteogenic potential of human osteoblast-like MG-63 cells. J. Cell. Biochem. 2019, 120, 15069–15082. [Google Scholar] [CrossRef]
- Elango, J.; Selvaganapathy, P.R.; Lazzari, G.; Bao, B.; Wu, W. Biomimetic collagen-sodium alginate-titanium oxide (TiO2) 3D matrix supports differentiated periodontal ligament fibroblasts growth for periodontal tissue regeneration. Int. J. Biol. Macromol. 2020, 163, 9–18. [Google Scholar] [CrossRef]
- Liu, B.; Li, J.; Lei, X.; Miao, S.; Zhang, S.; Cheng, P.; Song, Y.; Wu, H.; Gao, Y.; Bi, L.; et al. Cell-loaded injectable gelatin/alginate/LAPONITE® nanocomposite hydrogel promotes bone healing in a critical-size rat calvarial defect model. RSC Adv. 2020, 10, 25652–25661. [Google Scholar] [CrossRef]
- Mousavi, S.; Khoshfetrat, A.B.; Khatami, N.; Ahmadian, M.; Rahbarghazi, R. Comparative study of collagen and gelatin in chitosan-based hydrogels for effective wound dressing: Physical properties and fibroblastic cell behavior. Biochem. Biophys. Res. Commun. 2019, 518, 625–631. [Google Scholar] [CrossRef]
- Tan, W.; Krishnaraj, R.; Desai, T.A. Evaluation of Nanostructured Composite Collagen–Chitosan Matrices for Tissue Engineering. Tissue Eng. 2001, 7, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Peng, M.; Cheng, T.; Zhao, P.; Qiu, L.; Zhou, J.; Lu, G.; Chen, J. Silver nanoparticles-doped collagen–alginate antimicrobial biocomposite as potential wound dressing. J. Mater. Sci. 2018, 53, 14944–14952. [Google Scholar] [CrossRef]
- Rezaii, M.; Oryan, S.; Javeri, A. Curcumin nanoparticles incorporated collagen-chitosan scaffold promotes cutaneous wound healing through regulation of TGF-β1/Smad7 gene expression. Mater. Sci. Eng. C 2019, 98, 347–357. [Google Scholar] [CrossRef]
- Gingras, M.; Paradis, I.; Berthod, F. Nerve regeneration in a collagen–chitosan tissue-engineered skin transplanted on nude mice. Biomaterials 2003, 24, 1653–1661. [Google Scholar] [CrossRef]
- Lin, Z.; Wu, T.; Wang, W.; Li, B.; Wang, M.; Chen, L.; Xia, H.; Zhang, T. Biofunctions of antimicrobial peptide-conjugated alginate/hyaluronic acid/collagen wound dressings promote wound healing of a mixed-bacteria-infected wound. Int. J. Biol. Macromol. 2019, 140, 330–342. [Google Scholar] [CrossRef]
- Li, M.; Han, M.; Sun, Y.; Hua, Y.; Chen, G.; Zhang, L. Oligoarginine mediated collagen/chitosan gel composite for cutaneous wound healing. Int. J. Biol. Macromol. 2019, 122, 1120–1127. [Google Scholar] [CrossRef] [PubMed]
- Xie, H.; Chen, X.; Shen, X.; He, Y.; Chen, W.; Luo, Q.; Ge, W.; Yuan, W.; Tang, X.; Hou, D.; et al. Preparation of chitosan-collagen-alginate composite dressing and its promoting effects on wound healing. Int. J. Biol. Macromol. 2018, 107, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Raftery, R.M.; Woods, B.; Marques, A.L.; Moreira-Silva, J.; Silva, T.H.; Cryan, S.-A.; Reis, R.L.; O’Brien, F.J. Multifunctional biomaterials from the sea: Assessing the effects of chitosan incorporation into collagen scaffolds on mechanical and biological functionality. Acta Biomater. 2016, 43, 160–169. [Google Scholar] [CrossRef]
- Sionkowska, A.; Kaczmarek, B.; Lewandowska, K.; Grabska, S.; Pokrywczyńska, M.; Kloskowski, T.; Drewa, T. 3D composites based on the blends of chitosan and collagen with the addition of hyaluronic acid. Int. J. Biol. Macromol. 2016, 89, 442–448. [Google Scholar] [CrossRef]
- Liu, L.; Wen, H.; Rao, Z.; Zhu, C.; Liu, M.; Min, L.; Fan, L.; Tao, S. Preparation and characterization of chitosan–collagen peptide/oxidized konjac glucomannan hydrogel. Int. J. Biol. Macromol. 2018, 108, 376–382. [Google Scholar] [CrossRef]
- Tangsadthakun, C.; Kanokpanont, S.; Sanchavanakit, N.; Banaprasert, T.; Damrongsakkul, S. Properties of collagen/chitosan scaffolds for skin tissue engineering. J. Met. Mater. Miner. 2006, 16, 37–44. [Google Scholar]
- Rosellini, E.; Zhang, Y.S.; Migliori, B.; Barbani, N.; Lazzeri, L.; Shin, S.R.; Dokmeci, M.R.; Cascone, M.G. Protein/polysaccharide-based scaffolds mimicking native extracellular matrix for cardiac tissue engineering applications. J. Biomed. Mater. Res. Part A 2018, 106, 769–781. [Google Scholar] [CrossRef]
- Martínez, A.; Blanco, M.; Davidenko, N.; Cameron, R. Tailoring chitosan/collagen scaffolds for tissue engineering: Effect of composition and different crosslinking agents on scaffold properties. Carbohydr. Polym. 2015, 132, 606–619. [Google Scholar] [CrossRef] [PubMed]
- Pustlauk, W.; Paul, B.; Gelinsky, M.; Bernhardt, A. Jellyfish collagen and alginate: Combined marine materials for superior chondrogenesis of hMSC. Mater. Sci. Eng. C 2016, 64, 190–198. [Google Scholar] [CrossRef] [PubMed]
- Perez, R.A.; Kim, M.; Kim, T.-H.; Kim, J.-H.; Lee, J.H.; Park, J.H.; Knowles, J.C.; Kim, H.W. Plasma–fibroblast Gel as Scaffold for Islet Transplantation. Tissue Eng. Part A 2014, 15, 103–114. [Google Scholar] [CrossRef] [PubMed]
- Kaya, M.G.A.; Vuluga, Z.; Panaitescu, D.M.; Vuluga, D.M.; Căşărică, A.; Ghiurea, M. Morphology and thermal stability of bacterial cellulose/collagen composites. Open Chem. 2014, 12, 968–975. [Google Scholar] [CrossRef]
- Mao, Z. Collagen/chitosan porous scaffolds with improved biostability for skin tissue engineering. Biomaterials 2003, 24, 4833–4841. [Google Scholar] [CrossRef]
- Chiu, L.L.; Radisic, M. Controlled release of thymosin β4 using collagen–chitosan composite hydrogels promotes epicardial cell migration and angiogenesis. J. Control. Release 2011, 155, 376–385. [Google Scholar] [CrossRef]
- Moreira, C.D.F.; Carvalho, S.M.; Mansur, H.S.; Pereira, M.M. Thermogelling chitosan-collagen-bioactive glass nanoparticle hybrids as potential injectable systems for tissue engineering. Mater. Sci. Eng. C 2016, 58, 1207–1216. [Google Scholar] [CrossRef]
- Lewandowska-Łańcucka, J.; Gilarska, A.; Buła, A.; Horak, W.; Łatkiewicz, A.; Nowakowska, M. Genipin crosslinked bioactive collagen/chitosan/hyaluronic acid injectable hydrogels structurally amended via covalent attachment of surface-modified silica particles. Int. J. Biol. Macromol. 2019, 136, 1196–1208. [Google Scholar] [CrossRef]
- Donaghue, V.M.; Chrzan, J.S.; Rosenblum, B.I.; Giurini, J.M.; Habershaw, G.M.; Veves, A. Evaluation of a collagen-alginate wound dressing in the management of diabetic foot ulcers. Adv. Wound Care. 1998, 11, 114–119. [Google Scholar]
- Pon-On, W.; Charoenphandhu, N.; Teerapornpuntakit, J.; Thongbunchoo, J.; Krishnamra, N.; Tang, I.-M. Mechanical properties, biological activity and protein controlled release by poly(vinyl alcohol)–bioglass/chitosan–collagen composite scaffolds: A bone tissue engineering applications. Mater. Sci. Eng. C 2014, 38, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Rubina, M.; Kamitov, E.; Zubavichus, Y.V.; Peters, G.; Naumkin, A.V.; Suzer, S.; Vasil’Kov, A.Y. Collagen-chitosan scaffold modified with Au and Ag nanoparticles: Synthesis and structure. Appl. Surf. Sci. 2016, 366, 365–371. [Google Scholar] [CrossRef]
- Rafat, M.; Li, F.; Fagerholm, P.; Lagali, N.S.; Watsky, M.A.; Munger, R.; Matsuura, T.; Griffith, M. PEG-stabilized carbodiimide crosslinked collagen–chitosan hydrogels for corneal tissue engineering. Biomaterials 2008, 29, 3960–3972. [Google Scholar] [CrossRef] [PubMed]
- Soares, D.G.; Rosseto, H.L.; Basso, F.G.; Scheffel, D.S.; Hebling, J.; de Souza COSTA, C.A. Chitosan-collagen biomembrane embedded with calcium-aluminate enhances dentinogenic potential of pulp cells. Braz. Oral Res. 2016, 30. [Google Scholar] [CrossRef] [PubMed]
- Saska, S.; Teixeira, L.N.; De Castro-Raucci, L.M.S.; Scarel-Caminaga, R.M.; Franchi, L.P.; Dos Santos, R.A.; Santagneli, S.H.; Capela, M.V.; De Oliveira, P.T.; Takahashi, C.S.; et al. Nanocellulose-collagen-apatite composite associated with osteogenic growth peptide for bone regeneration. Int. J. Biol. Macromol. 2017, 103, 467–476. [Google Scholar] [CrossRef]
- Dang, Q.; Liu, K.; Zhang, Z.; Liu, C.; Liu, X.; Xin, Y.; Cheng, X.; Xu, T.; Cha, D.; Fan, B. Fabrication and evaluation of thermosensitive chitosan/collagen/α, β-glycerophosphate hydrogels for tissue regeneration. Carbohydr. Polym. 2017, 167, 145–157. [Google Scholar] [CrossRef]
- Rosella, E.; Jia, N.; Mantovani, D.; Greener, J. A microfluidic approach for development of hybrid collagen-chitosan extracellular matrix-like membranes for on-chip cell cultures. J. Mater. Sci. Technol. 2020. [Google Scholar] [CrossRef]
- Cai, X.; Hu, S.; Yu, B.; Cai, Y.; Yang, J.; Li, F.; Zheng, Y.; Shi, X. Transglutaminase-catalyzed preparation of crosslinked carboxymethyl chitosan/carboxymethyl cellulose/collagen composite membrane for postsurgical peritoneal adhesion prevention. Carbohydr. Polym. 2018, 201, 201–210. [Google Scholar] [CrossRef]
- Wang, J.; Wan, Y.; Luo, H.; Gao, C.; Huang, Y. Immobilization of gelatin on bacterial cellulose nanofibers surface via crosslinking technique. Mater. Sci. Eng. C 2012, 32, 536–541. [Google Scholar] [CrossRef]
- Chen, Z.; Mo, X.; He, C.; Wang, H. Intermolecular interactions in electrospun collagen–chitosan complex nanofibers. Carbohydr. Polym. 2008, 72, 410–418. [Google Scholar] [CrossRef]
- Muñoz-Ruíz, A.; Escobar-García, D.M.; Quintana, M.; Pozos-Guillén, A.; Flores, H. Synthesis and Characterization of a New Collagen-Alginate Aerogel for Tissue Engineering. J. Nanomater. 2019, 2019, 2875375. [Google Scholar] [CrossRef]
- Du, T.; Chen, Z.; Li, H.; Tang, X.; Li, Z.; Guan, J.; Liu, C.; Du, Z.; Wu, J. Modification of collagen–chitosan matrix by the natural crosslinker alginate dialdehyde. Int. J. Biol. Macromol. 2016, 82, 580–588. [Google Scholar] [CrossRef] [PubMed]
- Guillaume, O.; Naqvi, S.M.; Lennon, K.; Buckley, C.T. Enhancing cell migration in shape-memory alginate–collagen composite scaffolds: In vitro and ex vivo assessment for intervertebral disc repair. J. Biomater. Appl. 2015, 29, 1230–1246. [Google Scholar] [CrossRef]
- Zhu, Y.; Wan, Y.; Zhang, J.; Yin, D.; Cheng, W. Manufacture of layered collagen/chitosan-polycaprolactone scaffolds with biomimetic microarchitecture. Colloids Surf. B Biointerfaces 2014, 113, 352–360. [Google Scholar] [CrossRef]
- Yin, A.; Zhang, K.-H.; McClure, M.J.; Huang, C.; Wu, J.; Fang, J.; Mo, X.; Bowlin, G.L.; Al-Deyab, S.S.; Elnewehy, M.H. Electrospinning collagen/chitosan/poly(L-lactic acid-co-ϵ-caprolactone) to form a vascular graft: Mechanical and biological characterization. J. Biomed. Mater. Res. Part A 2013, 101, 1292–1301. [Google Scholar] [CrossRef]
- Devillard, R.; Rémy, M.; Kalisky, J.; Bourget, J.M.; Kérourédan, O.; Siadous, R.; Bareille, R.; Amédée-Vilamitjana, J.; Chassande, O.; Fricain, J.C. In vitro assessment of a collagen/alginate composite scaffold for regenerative endodontics. Int. Endod. J. 2017, 50, 48–57. [Google Scholar] [CrossRef]
- Wang, X.; Li, Q.; Hu, X.; Ma, L.; You, C.; Zheng, Y.; Sun, H.; Han, C.; Gao, C. Fabrication and characterization of poly(l-lactide-co-glycolide) knitted mesh-reinforced collagen–chitosan hybrid scaffolds for dermal tissue engineering. J. Mech. Behav. Biomed. Mater. 2012, 8, 204–215. [Google Scholar] [CrossRef]
- Chandika, P.; Ko, S.-C.; Oh, G.-W.; Heo, S.-Y.; Nguyen, V.-T.; Jeon, Y.-J.; Lee, B.; Jang, C.H.; Kim, G.; Park, W.S.; et al. Fish collagen/alginate/chitooligosaccharides integrated scaffold for skin tissue regeneration application. Int. J. Biol. Macromol. 2015, 81, 504–513. [Google Scholar] [CrossRef]
- Ding, C.; Zhang, M.; Li, G. Preparation and characterization of collagen/hydroxypropyl methylcellulose (HPMC) blend film. Carbohydr. Polym. 2015, 119, 194–201. [Google Scholar] [CrossRef]
- Socrates, R.; Prymak, O.; Loza, K.; Sakthivel, N.; Rajaram, A.; Epple, M.; Kalkura, S.N. Biomimetic fabrication of mineralized composite films of nanosilver loaded native fibrillar collagen and chitosan. Mater. Sci. Eng. C 2019, 99, 357–366. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Xie, X.-D.; Huang, X.; Liang, Z.-H.; Zhou, C.-R. A quantitative study of MC3T3-E1 cell adhesion, morphology and biomechanics on chitosan–collagen blend films at single cell level. Colloids Surf. B Biointerfaces 2015, 132, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Lima, C.G.A.; de Oliveira, R.S.; Figueiró, S.D.; Wehmann, C.F.; Góes, J.C.; Sombra, A.S.B. DC conductivity and dielectric permittivity of collagen–chitosan films. Mater. Chem. Phys. 2006, 99, 284–288. [Google Scholar] [CrossRef]
- Ramasamy, P.; Shanmugam, A. Characterization and wound healing property of collagen–chitosan film from Sepia kobiensis (Hoyle, 1885). Int. J. Biol. Macromol. 2015, 74, 93–102. [Google Scholar] [CrossRef] [PubMed]






| Biopolymers | Structure | Sources | Properties | Applications | Ref. | ||||
|---|---|---|---|---|---|---|---|---|---|
| Solubility | Toxicity | Ph | Stability | Biomedical | |||||
| Collagen | Triple helix, a unique tertiary structure three identical or non-identical polypeptide chains, each chain is composed of around 1000 amino acids or more in length | Vertebrate body protein (skin, tendon, bone, cornea, dentin, fibrocartilage, large vessels, intestine, uterus, dermis, tendon, placenta) | Insoluble in water, but lowering the pH of solution can increase solubility | Negligible | Solubility, structural, thermal properties are pH sensitive | Poor mechanical, thermal, enzymatic, tensile stiffness | Biodegradable, biocompatible, unique self-assembling fibril-forming properties, biomimetic | Food, cosmetic, photographic, ophthalmology, inserts, shields, particles, gels, aqueous injectables, drug delivery | [12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,34] |
| Alginate | Whole family of linear copolymers containing blocks of (1,4)-linked β-D-mannuronate (M) and α-L-guluronate (G) residues | Brown algae (Phaeophyceae), Laminaria hyperborea, Laminaria digitata, Laminaria japonica, Ascophyllum nodosum, and Macrocystis pyrifera | High solubility in aqueous solutions lowering the pH of solution can increase solubility | Low | Viscosity of alginate solutions increases as pH decreases | Poor mechanical and chemical | Biocompatibility, mild gelation | Wound healing, delivery of bioactive agents such as small chemical drugs and proteins, and cell transplantation | [31] |
| Chitosan | Poly-(beta-1-4) N-acetyl-D-glucosamine reactive amino groups | Deacetylation of chitin from insects (cuticles), crustaceans (skeletons-crab, shrimp, lobster), cephalopod (exoskeleton) | Soluble in aqueous acidic media (higher 50% deacetylation) Solubility is enhanced by decreasing its molecular weight | Negligible | Solubility, biomedical properties are pH sensitive | - | Nontoxic, non-immunogenic, non-carcinogenic, biocompatibility, bio absorbability, antimicrobial, antibacterial, antifungal anticoagulant, anti-tumor, hemostatic | Biosensors, drug delivery, wound dressing | [32] |
| Cellulose | Exopolysaccharide | Wood, cotton, sugar beet, potato tubers, onion, hemp, flax, wheat straw, mulberry bark, algae, bacteria | - | Negligible | - | High mechanical | Biocompatibility, biodegradability, biological affinity, antibacterial | Wound dressing, shields, dental implants, artificial blood vessel | [33] |
| Carrier Form | Support Material Component | Active Substance/Drug | Application | Ref. |
|---|---|---|---|---|
| Microsphere | Collagen–alginate | Glial cell line-derived neurotrophic factor | Neurodegenerative diseases | [71] |
| - | Collagen–bacterial cellulose | Bovine serum albumin | Potential drug delivery system | [70] |
| - | Collagen–chitosan–nano-hydroxyapatite | Potential drug delivery | [72] | |
| Hydrogel or gel | Hydroxyapatite/collagen–alginate | Bone morphogenetic protein | Bone filler | [73] |
| - | Alginate–collagen | Doxycycline | Vision-threatening diseases | [74] |
| - | Collagen–carboxymethyl cellulose | Anti-inflammatory drug | Retinal ischaemia/reperfusion injury | [75] |
| - | Collagen–alginate | Methylene blue Imiquimod | Combinatorial photothermal and immuno tumor therapy | [76] |
| - | Collagen–chitosan | Nanobodies: 2D5 and KPU | Tumor treatment matrix for use in cancer therapy | [77] |
| - | Chitosan–alginate hydrogel encapsulated gelatin microspheres | 5-fluorouracil | Anti-cancer drug delivery | [78] |
| - | Chitosan–collagen | Ibuprofen | Thermoresponsive scaffold | [79] |
| - | Collagen–chitosan–glucan | Aloe vera | Antibacterial activity against different types of bacteria (positive/negative grams) Wound healing Infected chronic wounds and ulcers | [80] |
| - | Chitosan–collagen–alginate | Curcumin | Diabetic wound healing | [81] |
| - | Chitosan–collagen | Glutanine-hstidme-arginine-glutamic acid-aspartic acid-glicine-serine (qhredgs) | Myocardial infarction | [82] |
| - | Collagen–chitosan | Norfloxacin | Wound healing, skin regeneration | [83] |
| Membrane | Collagen–chitosan | Nifedipine and propranolol hydrochloride | Cardiac disease | [84] |
| - | PLGA/collagen | Vancomycin, gentamicin and lidocaine | Antibiotic activity | [68] |
| Nanoparticle | Collagen–chitosan | Doxorubicin hydrochloride | Advanced cancer therapy | [85] |
| - | Gelatin-alginate/Fe3O4 magnetic nanoparticles | Doxorubicin hydrochloride | Cancer chemotherapy | [86] |
| Sponge | Collagen–chitosan | Dexamethasone | Oral mucositis | [69] |
| - | Collagen–PLGA | Gentamicin | Tissue regeneration | [87] |
| Film | Collagen–chitosan | Local anesthetics mix (lidocaine, tetracaine, and benzocaine) | Wound healing | [88] |
| - | Collagen–chitosan–hyaluronic acid | Gentamicin sulfate | Antibiotic release | [89] |
| - | Collagen–chitosan | Doxorubicin | Cancer treatment | [90] |
| - | Collagen–chitosan | Basic fibroblast growth factor | Wound healing | [67] |
| - | Collagen–chitosan–chondroitin | Loaded plga microspheres | Tissue engineering | [91] |
| - | Chitosan–collagen | Gentamicin sulfate | Antibiotic release | [92] |
| Form | Material Component | Potential Application | Proprieties Improved/New | Ref. |
|---|---|---|---|---|
| Microsphereor bead | Bacterial cellulose and collagen | Bone tissue engineering | Adhesion, proliferation, and osteogenic differentiation | [96] |
| Collagen–alginate | Infertility | Stem cells attachment, proliferation and differentiation | [97] | |
| Collagen–chitosan–nano-hydroxyapatite | Bone regeneration | High dispersity | [48] | |
| Collagen–alginate-nano-silica | Bone tissue engineering | Mechanical strength and generate porous membrane structure | [98] | |
| Hydrogel or gel | Chitosan–collagen–gelatin | Wound healing | Rheology and mechanical properties | [50] |
| Chitosan–collagen | Bone, tendon, muscle engineering | Mechanical properties, degradation rate, cytocompatibility, cytotoxicity and the capability to promote the attachment, migration and proliferation of Schwann cells; modulated degradation behavior without inflammatory reaction | [57] | |
| Chitosan–collagen | Cardiac cell culture and delivery | Thermoresponsive | [83] | |
| Collagen–sodium alginate-titanium oxide (tio2) | Periodontal tissue regeneration | Stiffness, water binding capacity, swelling, shrinkage factor, porosity and in-vitro biodegradation, osteocalcin secretion | [99] | |
| Collagen–alginate–fibrin | Soft tissue engineering (pancreas tissue engineering and musculoskeletal applications) | Thermo-responsive capacity at physiological conditions with stiffness similar to native soft tissues, enhanced osteogenic potential of human mesenchymal stem cells (hmscs) at high collagen content | [44] | |
| Gelatin–alginate–Laponite | Bone healing | [100] | ||
| Collagen–chitosan | Wound dressing | Reduced swelling rate and improved mechanical strength, high cell survival rate and prominent spindle shape | [101] | |
| Chitosan–alginate hydrogel encapsulated gelatin microspheres - 5-fluorouracil | Soft tissue engineering anti-cancer drug delivery | - | [78] | |
| Chitosan–collagen | Tissue engineering | Modulate cell behavior | [102] | |
| Collagen–alginate- silver nanoparticles | Skin dressing | Antibacterial activity | [103] | |
| Collagen–chitosan–curcumin | Wound healing | - | [104] | |
| Chitosan–collagen | Peripheral nerve regeneration | Decreased the mean pore size, liquid uptake and degradation rate, increased the mechanical property of the composite scaffolds, good cytocompatibility without cytotoxicity | [105] | |
| Alginate–hyaluronic acid–collagen | Wound healing | Reepithelialization, collagen deposition, and angiogenesis in infected wound animal model | [106] | |
| Collagen–chitosan gel composite supplemented with a cell-penetrating peptide (CPP) (oligoarginine, r8) | Cutaneous wound healing Antibacterial activity | Inhibiting Staphylococcus aureus growth and had good ability to heal wounds | [107] | |
| Chitosan–collagen–alginate -curcumin | Diabetic wound healing | Anti-inflammatory and anti-oxidant, sustain drug carrier, wound healing, established wound healer as scaffold | [81] | |
| Chitosan–collagen–alginate | Wound healing | No significant cytotoxicity, and favorable hemocompatibility, biosecurity | [108] | |
| Chitosan–collagen | Bone and cartilage regeneration | Improved the mechanical properties, increasing the compressive strength, swelling ratio and prolonged the degradation rate | [109] | |
| Collagen–chitosan–hyaluronic acid | - | Improved mechanical properties and thermal stability | [110] | |
| Chitosan–collagen | Wound dressing | Gelation time, swelling behaviors, water evaporation rate and blood coagulation capacity | [111] | |
| Alginate–collagen | Neurogenesis and neuronal maturation | Improved mechanical properties | [43] | |
| Collagen–chitosan | Skin tissue engineering | L929 cell proliferation | [112] | |
| Alginate-gelatin-polysaccharide | Cardiac tissue engineering | Reduced water swelling, increased storage, modulus and loss modulus | [113] | |
| Chitosan–collagen | Scaffolds for tissue engineering | Morphology, mechanical stiffness, swelling, degradation and cytotoxicity | [114] | |
| Collagen–alginate | Tissue engineering | Mechanical support | [115] | |
| Core–shell fibrous collagen–alginate hydrogel | Bone tissue engineering | Viability, exhibiting significant cellular proliferation | [116] | |
| Cellulose–collagen | - | Morphology and thermal | [117] | |
| Collagen–chitosan | Skin tissue engineering | Accelerate cell infiltration and proliferation | [118] | |
| Collagen–chitosan | Cardiac repair | Promote cell migration | [119] | |
| Chitosan–collagen–bioactive glass | Regenerative medicine One-tissue bio applications | Thermosensitive | [120] | |
| Collagen–chitosan–hyaluronic acid | Bone tissue engineering | Osteosarcoma cell lines mg-63 cells adhesion, proliferation as well as alkaline phosphatase (alp) expression | [121] | |
| Collagen-alginate | Wound healing | Mechanical properties | [122] | |
| Poly (vinyl alcohol)–bioglass–chitosan–collagen | Bone tissue engineering | Mechanical, mineral deposition, biological properties and controlled release | [123] | |
| Collagen–chitosan–Au and Ag nanoparticles | Wound-healing | Antibacterial activity, good compatibility with living tissues | [124] | |
| Nanocellulose–collagen–apatite | Bone regeneration | No cytotoxic, genotoxic or mutagenic effects | [125] | |
| Chitosan–collagen–α, β-glycerophosphate | Tissue regeneration | Thermosensitive | [126] | |
| Chitosan–collagen matrix embedded with calcium-aluminate microparticles | Dental pulp stem cell | Biodegradation Differentiation of pulp cells | [127] | |
| Collagen–chitosan | Corneal tissue engineering | Optical properties, mechanical properties, suturability, permeability to glucose and albumin | [128] | |
| Membrane | Collagen–chitosan | On-chip cell cultures Extracellular matrix supports | Mechanical properties and biodegradability | [129] |
| Carboxymethyl chitosan–carboxymethyl cellulose–collagen | Postsurgical peritoneal adhesion prevention | Mechanical properties and biodegradability | [130] | |
| Nanofibers | Collagen–cellulose | Tissue engineering. | Cell growth | [131] |
| Collagen–chitosan | Biomedical applications | Mechanical properties | [132] | |
| Aerogel Sponge/scaffold | Collagen–alginate | Tissue engineering | Cell migration, cell attachment, cell proliferation | [133] |
| Collagen–chitosan | Tissue engineering | Thermostability Antibacterial activity | [134] | |
| Collagen–hydroxyapatite | Skeletal muscular system engineering | Mechanical strength Osteoinductivity | [135] | |
| Collagen–alginate | Cartilage, disc repair | Cell migration and proliferation | [136] | |
| Collagen–chitosan–polycaprolactone | Articular cartilage repair | Mechanical, swelling properties Growth of seeded chondrocytes | [137] | |
| Collagen–chitosan–poly (L-lactic acid-co-ϵ-caprolactone) | Vascular graft | Mechanical strength | [138] | |
| Collagen–alginate | Regenerative endodontics | Elastic modulus, tissue compaction and cell differentiation | [139] | |
| Collagen–chitosan–poly(L-lactide-co-glycolide) | Dermal tissue engineering | Mechanical properties Promote angiogenesis and induce in situ tissue formation | [140] | |
| Collagen–alginate–chitooligosaccharides | Skin tissue regeneration | Physicochemical, mechanical, biological properties | [141] | |
| Film | Collagen–hydroxypropyl methylcellulose | - | Tensile strength, ultimate elongation and hydrophilicity of the blend film were superior to those of the pure collagen film Polyethylene glycol 1500 had almost no influence on the thermal properties of the blend film but obviously improved its stretch-ability and smoothness | [142] |
| Collagen–chitosan–silver nanoparticles | Bone tissue engineering | Mechanical stability and antibacterial property | [143] | |
| Chitosan–collagen | Tissue engineering | Cell adhesion, morphological and biomechanical properties | [144] | |
| Collagen–chitosan | Coating of cardiovascular prostheses, support for cellular growth and in systems for controlled drug delivery | Dielectric permittivity, thermal stability, highest conductivity | [145] | |
| Collagen–chitosan–graphene oxide | Wound healing | Increased tensile, strength and brittleness, decreased elongation at break | [67] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Geanaliu-Nicolae, R.-E.; Andronescu, E. Blended Natural Support Materials—Collagen Based Hydrogels Used in Biomedicine. Materials 2020, 13, 5641. https://doi.org/10.3390/ma13245641
Geanaliu-Nicolae R-E, Andronescu E. Blended Natural Support Materials—Collagen Based Hydrogels Used in Biomedicine. Materials. 2020; 13(24):5641. https://doi.org/10.3390/ma13245641
Chicago/Turabian StyleGeanaliu-Nicolae, Ruxandra-Elena, and Ecaterina Andronescu. 2020. "Blended Natural Support Materials—Collagen Based Hydrogels Used in Biomedicine" Materials 13, no. 24: 5641. https://doi.org/10.3390/ma13245641
APA StyleGeanaliu-Nicolae, R.-E., & Andronescu, E. (2020). Blended Natural Support Materials—Collagen Based Hydrogels Used in Biomedicine. Materials, 13(24), 5641. https://doi.org/10.3390/ma13245641





