Composite and Surface Functionalization of Ultrafine-Grained Ti23Zr25Nb Alloy for Medical Applications
Abstract
1. Introduction
2. Materials and Methods
| A0 | annealed titanium rod (Grade 2) |
| B0 | Ti23Zr25Nb—cold pressed and sintered at 800 °C for 30 min |
| B1* | Ti23Zr25Nb—electrochemically etched |
| B1 | Ti23Zr25Nb—electrochemically deposited calcium and phosphorus-riched coating |
| B2 | Ti23Zr25Nb—9 wt.% 45S5 Bioglass |
| B3 | Ti23Zr25Nb—9 wt.% 45S5 BG–1 wt.% Ag |
| B4 | Ti23Zr25Nb—9 wt.% 45S5 BG–1 wt.% Cu |
| B5 | Ti23Zr25Nb—9 wt.% 45S5 BG–1 wt.% Zn |
| B7 | microcrystalline arc-melted Ti18Zr24Nb (at.%) |
| B8 | hot-pressed at 600 °C for 10 min |
| B9 | Ti23Zr25Nb-based foam. |
2.1. Sample Preparation
2.2. Materials Characterization
2.3. In Vitro Biocompatibility Studies
2.3.1. Cell Lines Preparation
2.3.2. Conditioning of Breeding Media
2.3.3. The Cytotoxicity Evaluation of Inserts and Cell Survival by MTS Assay
2.3.4. Cell Culture for Photographic Documentation
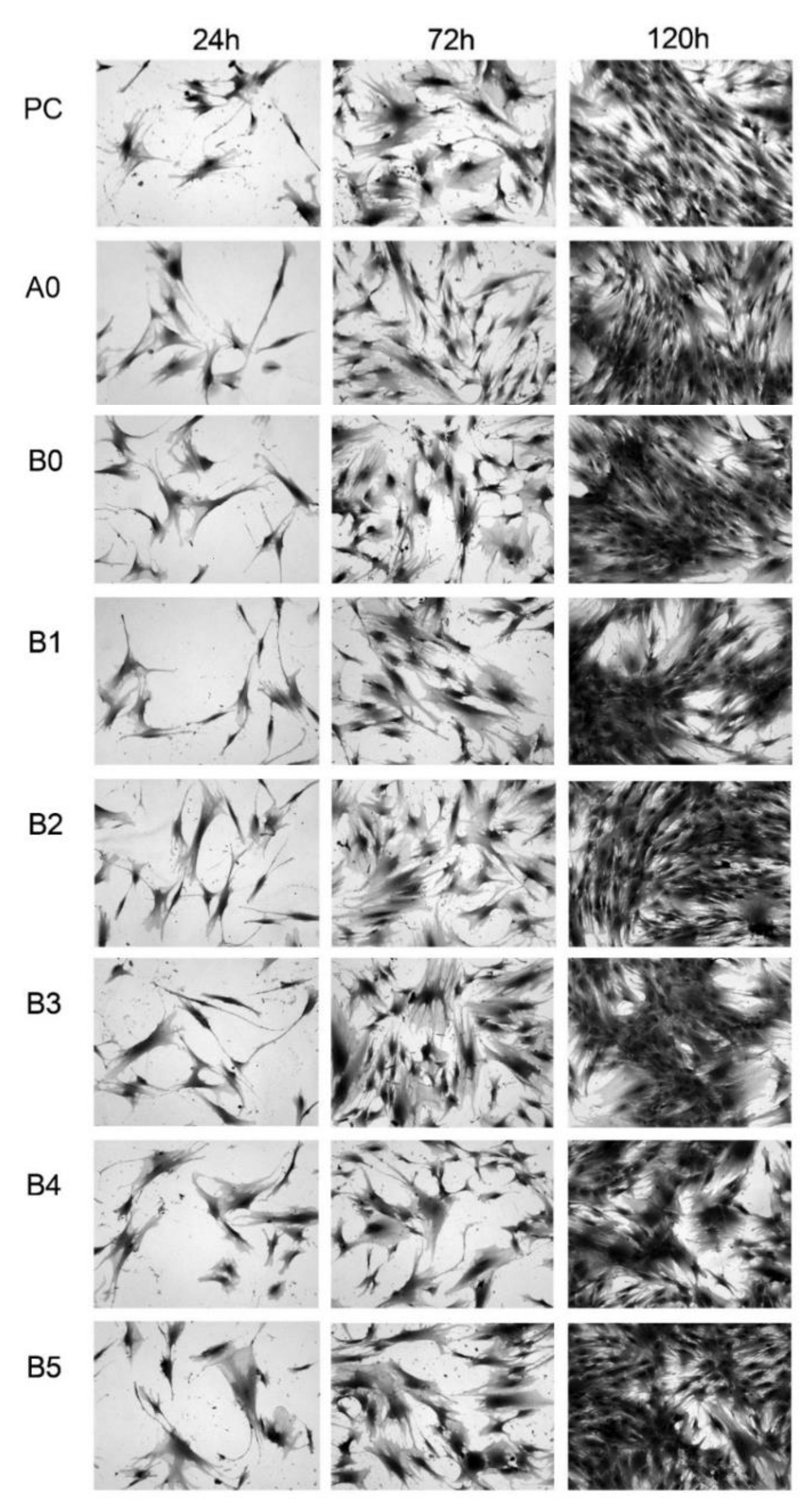
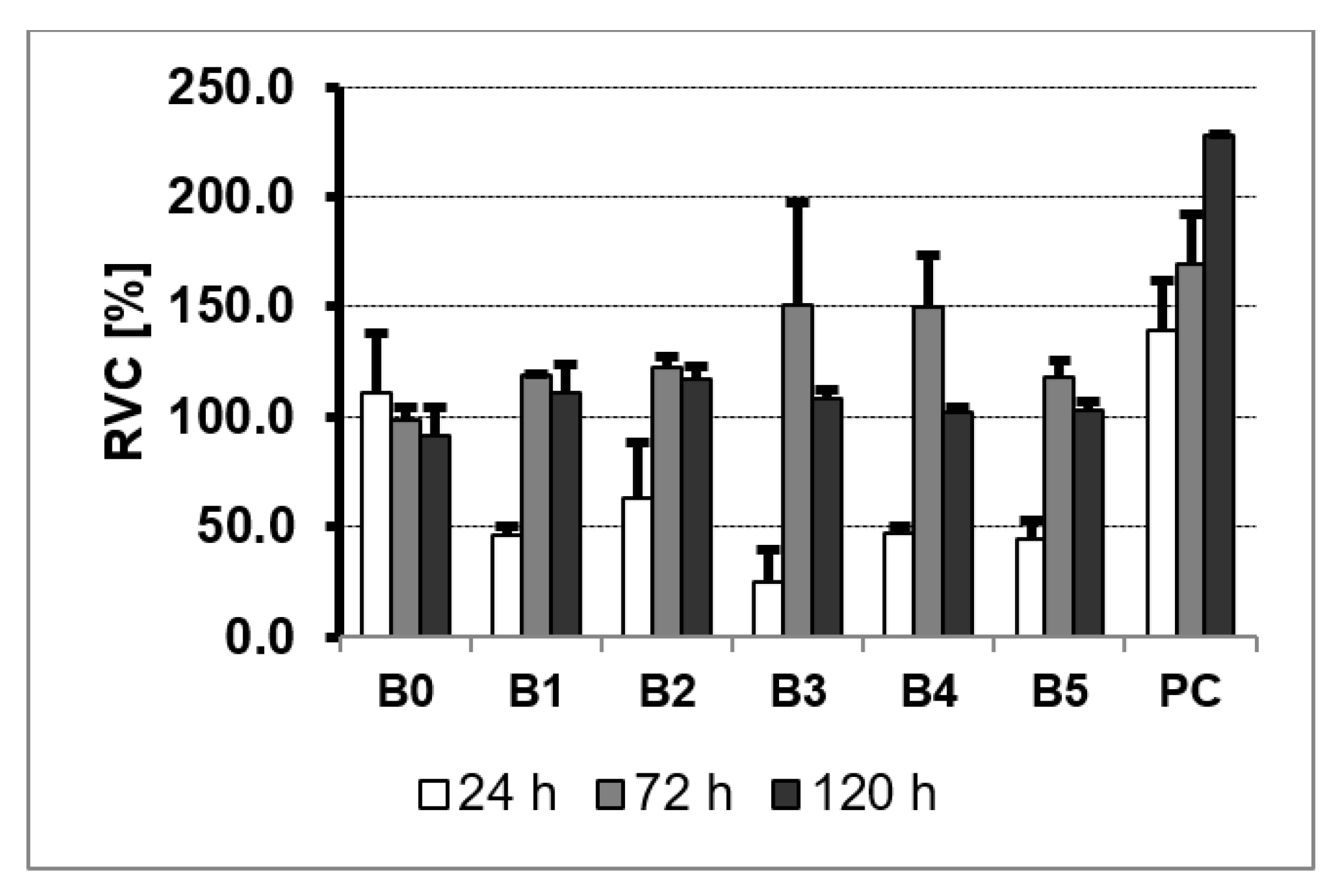
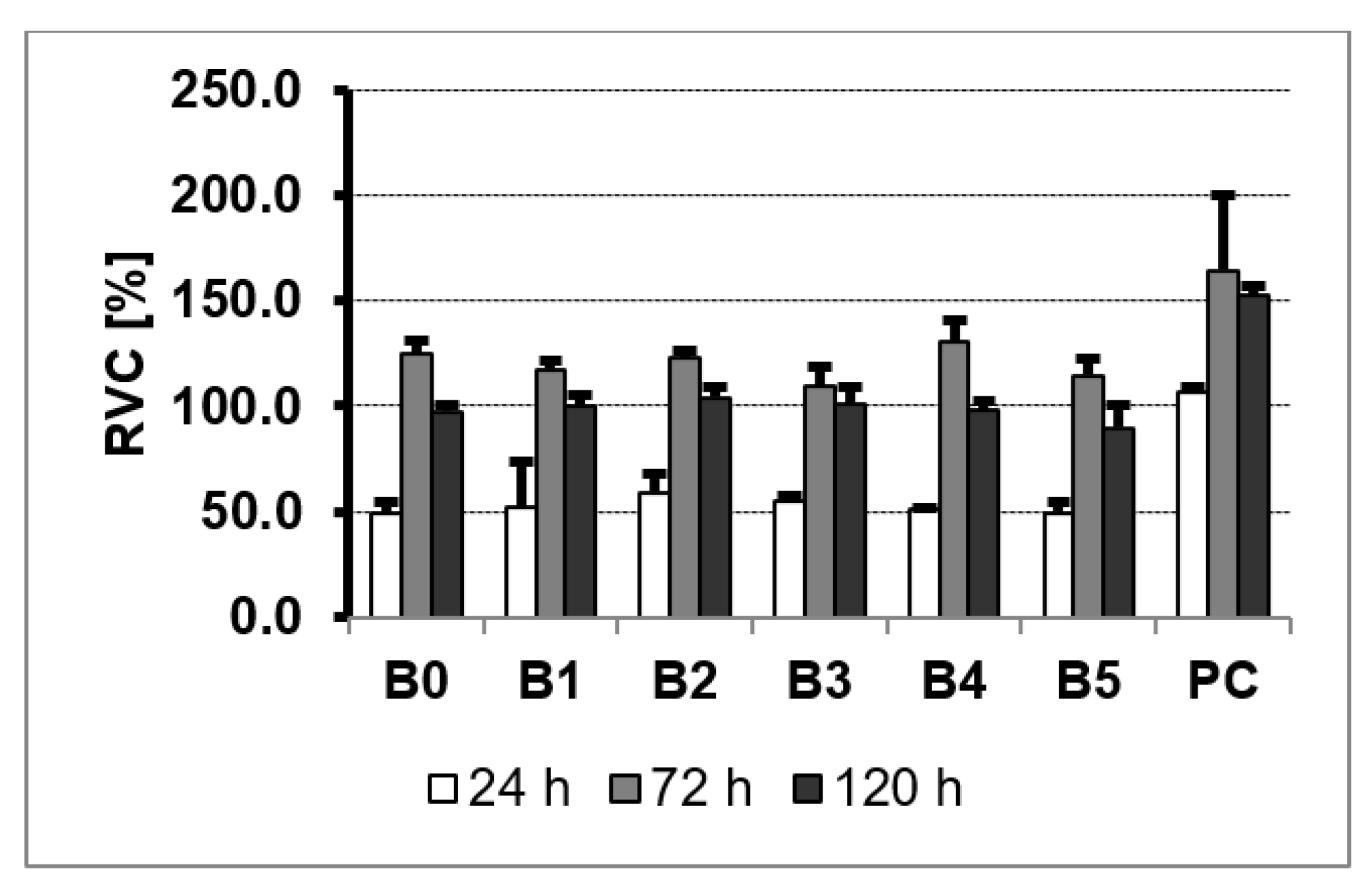
3. Results and Discussion
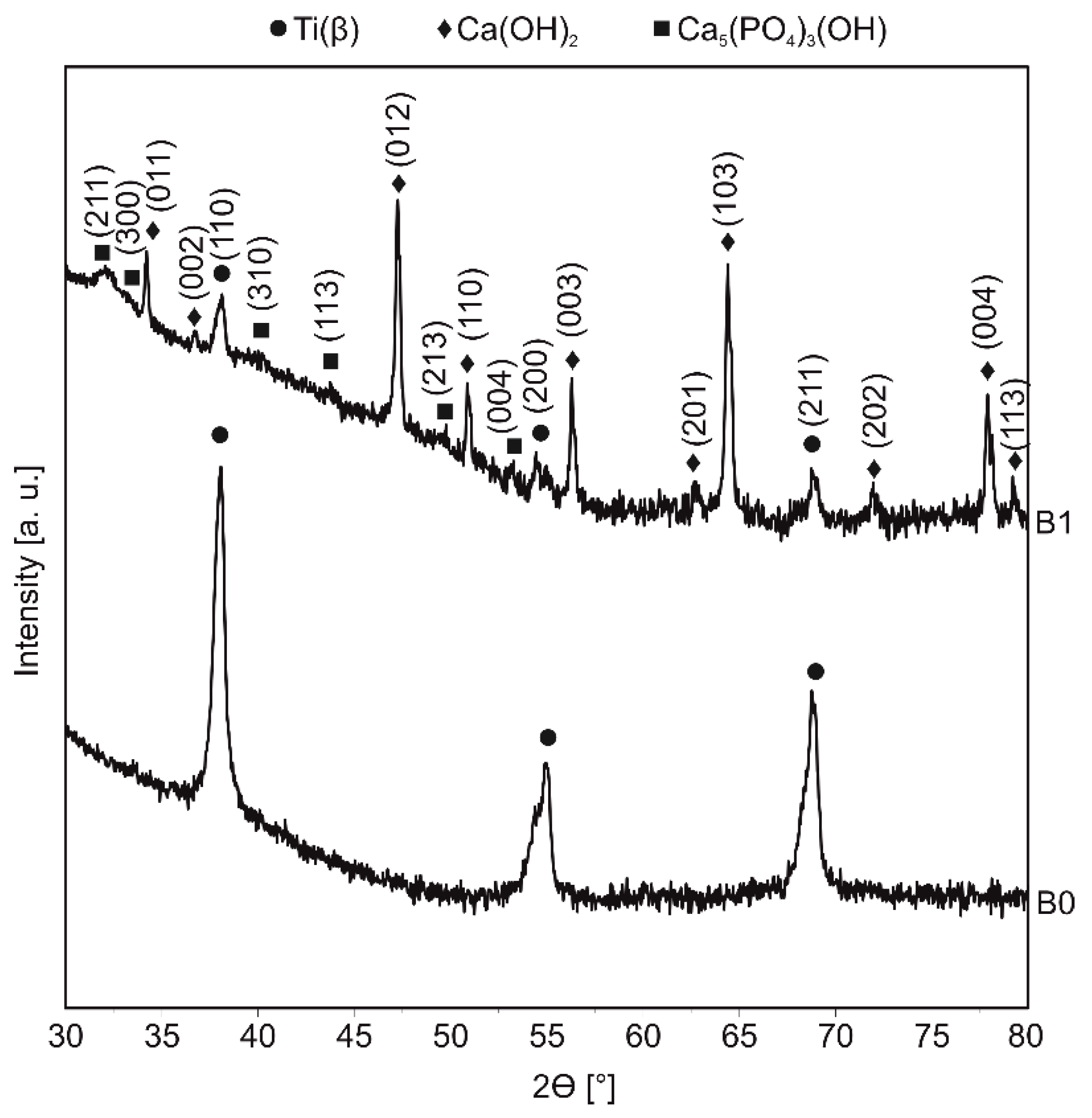
3.1. Structure Properties
3.2. Mechanical Properties
3.3. Surface Properties
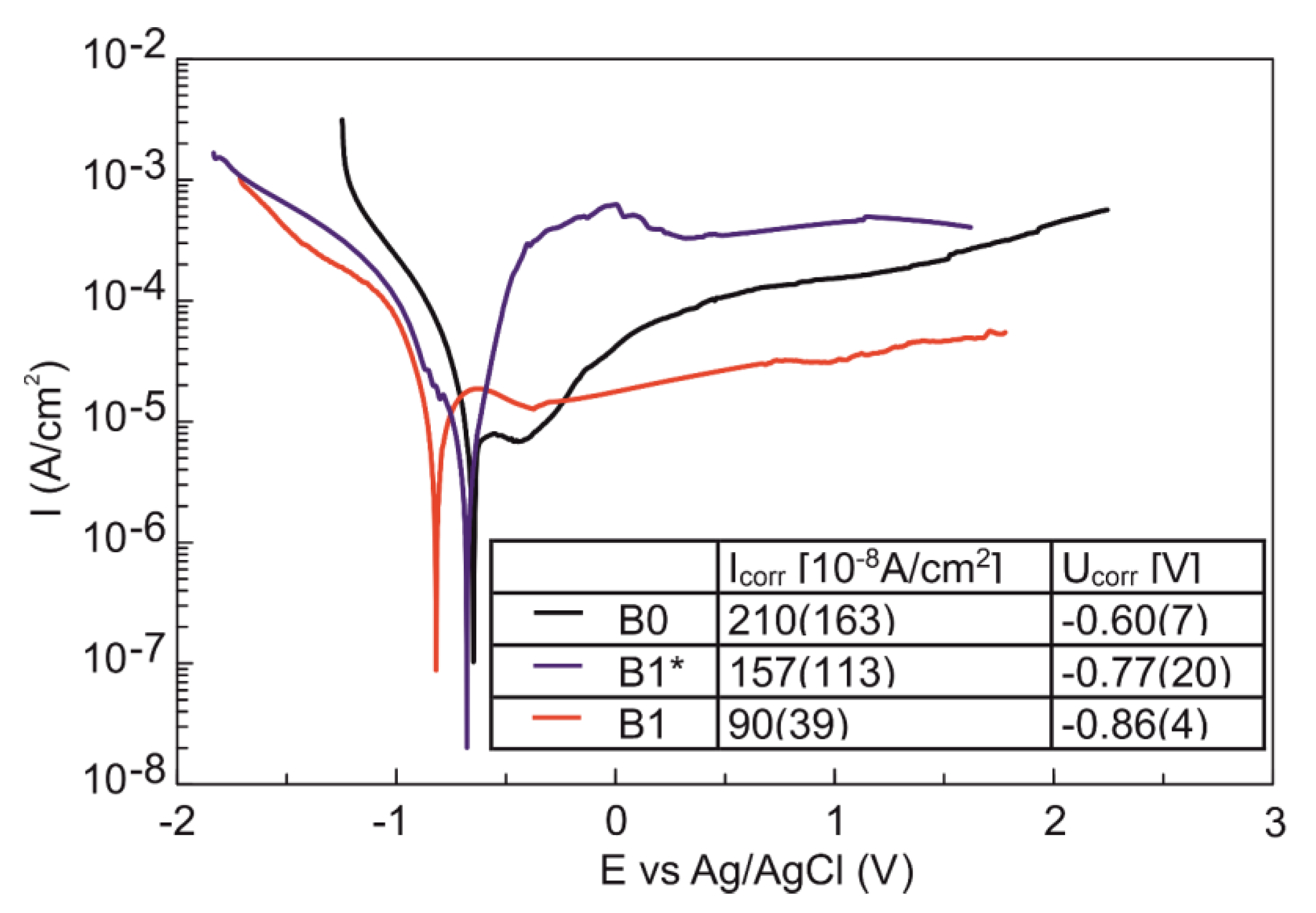
3.4. Corrosion and Surface Wetting Properties
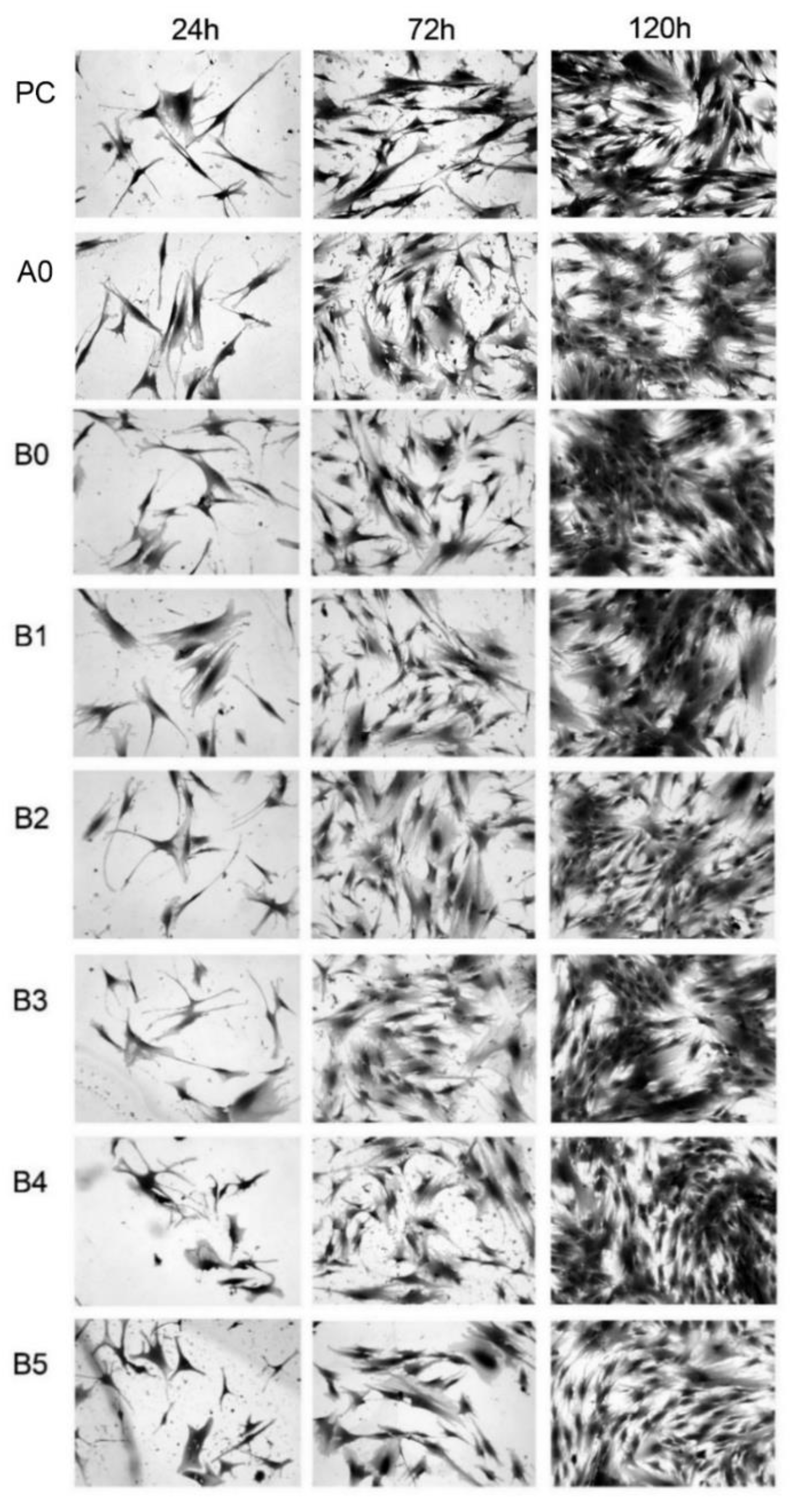
3.5. MTS Assay
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bai, Y.; Deng, Y.; Zheng, Y.; Li, Y.; Zhang, R.; Lv, Y.; Zhao, Q.; Wei, S. Characterization, corrosion behavior, cellular response and in vivo bone tissue compatibility of titanium–niobium alloy with low Young’s modulus. Mater. Sci. Eng. C 2016, 59, 565–576. [Google Scholar] [CrossRef]
- Karre, R.; Kodli, B.K.; Rajendran, A.; Pattanayak, D.K.; Ameyama, K.; Dey, S.R.; Rajamallu, K.; Nivedhitha, J. Comparative study on Ti-Nb binary alloys fabricated through spark plasma sintering and conventional P/M routes for biomedical application. Mater. Sci. Eng. C 2018, 94, 619–627. [Google Scholar] [CrossRef]
- Wang, B.L.; Li, L.; Zheng, Y.F. In vitro cytotoxicity and hemocompatibility studies of Ti-Nb, Ti-Nb-Zr and Ti-Nb-Hf biomedical shape memory alloys. Biomed. Mater. 2010, 5, 044102. [Google Scholar] [CrossRef]
- Majumdar, P.; Singh, S.; Dhara, S.; Chakraborty, M. Influence of boron addition to Ti–13Zr–13Nb alloy on MG63 osteoblast cell viability and protein adsorption. Mater. Sci. Eng. C 2015, 46, 62–68. [Google Scholar] [CrossRef]
- Cremasco, A.; Messias, A.D.; Esposito, A.R.; Duek, E.A.D.R.; Caram, R. Effects of alloying elements on the cytotoxic response of titanium alloys. Mater. Sci. Eng. C 2011, 31, 833–839. [Google Scholar] [CrossRef]
- Neacsu, P.; Gordin, D.-M.; Mitran, V.; Gloriant, T.; Costache, M.; Cimpean, A. In vitro performance assessment of new beta Ti–Mo–Nb alloy compositions. Mater. Sci. Eng. C 2015, 47, 105–113. [Google Scholar] [CrossRef]
- Guillemot, F.; Prima, F.; Bareille, R.; Gordin, D.-M.; Gloriant, T.; Durrieu, M.; Ansel, D.; Baquey, C. Design of new titanium alloys for orthopaedic applications. Med Biol. Eng. Comput. 2004, 42, 137–141. [Google Scholar] [CrossRef]
- Haftlang, F.; Zarei-Hanzaki, A.; Abedi, H. The effect of nano-size second precipitates on the structure, apatite-inducing ability and in-vitro biocompatibility of Ti-29Nb-14Ta-4.5Zr alloy. Mater. Sci. Eng. C 2020, 109, 110561. [Google Scholar] [CrossRef] [PubMed]
- Bărbînţă, A.C.; Earar, K.; Crimu, C.I.; DrĂgan, L.A.; Munteanu, C. In Vitro Evaluation of the Cytotoxicity of Some New Titanium Alloys. Key Eng. Mater. 2013, 587, 303–308. [Google Scholar] [CrossRef]
- Rajan, S.T.; Bendavid, A.; Subramanian, B. Cytocompatibility assessment of Ti-Nb-Zr-Si thin film metallic glasses with enhanced osteoblast differentiation for biomedical applications. Colloids Surf. B Biointerfaces 2019, 173, 109–120. [Google Scholar] [CrossRef]
- Kopova, I.; Stráský, J.; Harcuba, P.; Landa, M.; Janeček, M.; Bačákova, L. Newly developed Ti–Nb–Zr–Ta–Si–Fe biomedical beta titanium alloys with increased strength and enhanced biocompatibility. Mater. Sci. Eng. C 2016, 60, 230–238. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.-J.; Song, Y.-H.; An, J.-H.; Song, H.-J.; Anusavice, K.J. Cytocompatibility of pure metals and experimental binary titanium alloys for implant materials. J. Dent. 2013, 41, 1251–1258. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, S.P.; Bilesan, M.R.; Golestani-Fard, F. In vitro evaluation of the biocompatibility and bioactivity of plasma electrolyte oxidized titania/calcium phosphate nanocoatings on Ti. J. Mater. Sci. 2018, 54, 4277–4286. [Google Scholar] [CrossRef]
- Liu, X.; Wu, L.; Ai, H.; Han, Y.; Hu, Y. Cytocompatibility and early osseointegration of nanoTiO2-modified Ti-24 Nb-4 Zr-7.9 Sn surfaces. Mater. Sci. Eng. C 2015, 48, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Ansar, E.; Ravikumar, K.; Babu, S.S.; Fernandez, F.; Komath, M.; Basu, B.; Varma, H. Inducing apatite pre-layer on titanium surface through hydrothermal processing for osseointegration. Mater. Sci. Eng. C 2019, 105, 110019. [Google Scholar] [CrossRef]
- Sedelnikova, M.B.; Komarova, E.G.; Sharkeev, Y.P.; Ugodchikova, A.V.; Tolkacheva, T.V.; Rau, J.V.; Buyko, E.E.; Ivanov, V.V.; Sheikin, V.V. Modification of titanium surface via Ag-, Sr- and Si-containing micro-arc calcium phosphate coating. Bioact. Mater. 2019, 4, 224–235. [Google Scholar] [CrossRef]
- Furko, M.; Della Bella, E.; Fini, M.; Balázsi, K. Corrosion and biocompatibility examination of multi-element modified calcium phosphate bioceramic layers. Mater. Sci. Eng. C 2019, 95, 381–388. [Google Scholar] [CrossRef]
- Türk, S.; Altınsoy, I.; Efe Çelebi, G.; Ipek, M.; Özacar, M.; Bindal, C. Biomimetic synthesis of Ag, Zn or Co doped HA and coating of Ag, Zn or Co doped HA/fMWCNT composite on functionalized Ti. Mater. Sci. Eng. C 2019, 99, 986–998. [Google Scholar] [CrossRef]
- Lenis, J.; Toro, L.; Bolívar, F. Multi-layer bactericidal silver-calcium phosphate coatings obtained by RF magnetron sputtering. Surf. Coat. Technol. 2019, 367, 203–211. [Google Scholar] [CrossRef]
- He, Y.; Zhang, Y.; Meng, Z.; Jiang, Y.; Zhou, R. Microstructure evolution, mechanical properties and enhanced bioactivity of Ti-Nb-Zr based biocomposite by bioactive calcium pyrophosphate. J. Alloys Compd. 2017, 720, 567–581. [Google Scholar] [CrossRef]
- Wang, X.; Chen, Y.; Xu, L.; Liu, Z.; Woo, K.-D. Effects of Sn content on the microstructure, mechanical properties and biocompatibility of Ti–Nb–Sn/hydroxyapatite biocomposites synthesized by powder metallurgy. Mater. Des. 2013, 49, 511–519. [Google Scholar] [CrossRef]
- Rodrigues, C.; Naasani, L.I.S.; Zanatelli, C.; Paim, T.C.; Azevedo, J.G.; De Lima, J.C.; Fernandes, M.D.C.; Buchner, S.; Wink, M. Bioglass 45S5: Structural characterization of short range order and analysis of biocompatibility with adipose-derived mesenchymal stromal cells in vitro and in vivo. Mater. Sci. Eng. C 2019, 103, 109781. [Google Scholar] [CrossRef] [PubMed]
- De Souza, L.P.L.; Lopes, J.H.; Ferreira, F.; Martin, R.A.; Bertran, C.A.; Camilli, J.A. Evaluation of effectiveness of 45S5 bioglass doped with niobium for repairing critical-sized bone defect in in vitro and in vivo models. J. Biomed. Mater. Res. Part A 2019, 108, 446–457. [Google Scholar] [CrossRef] [PubMed]
- Lopes, J.H.; Souza, L.; Domingues, J.A.; Ferreira, F.; Hausen, M.D.A.; Camilli, J.A.; Martin, R.A.; Duek, E.A.D.R.; Mazali, I.O.; Bertran, C.A. In vitro and in vivo osteogenic potential of niobium-doped 45S5 bioactive glass: A comparative study. J. Biomed. Mater. Res. Part B Appl. Biomater. 2020, 108, 1372–1387. [Google Scholar] [CrossRef]
- Karakuzu-Ikizler, B.; Terzioğlu, P.; Oduncu-Tekerek, B.S.; Yucel, S. Effect of selenium incorporation on the structure and in vitro bioactivity of 45S5 bioglass. J. Aust. Ceram. Soc. 2019, 56, 697–709. [Google Scholar] [CrossRef]
- Dittler, M.L.; Unalan, I.; Grünewald, A.; Beltrán, A.M.; Grillo, C.A.; Destch, R.; Gonzalez, M.C.; Boccaccini, A.R. Bioactive glass (45S5)-based 3D scaffolds coated with magnesium and zinc-loaded hydroxyapatite nanoparticles for tissue engineering applications. Colloids Surf. B Biointerfaces 2019, 182, 110346. [Google Scholar] [CrossRef]
- Eshghinejad, P.; Farnoush, H.; Bahrami, M.S.; Bakhsheshi-Rad, H.R.; Karamian, E.; Chen, X. Electrophoretic deposition of bioglass/graphene oxide composite on Ti-alloy implants for improved antibacterial and cytocompatible properties. Mater. Technol. 2019, 35, 69–74. [Google Scholar] [CrossRef]
- Shaikh, S.; Kedia, S.; Majumdar, A.G.; Subramanian, M.; Sinha, S. 45S5 bioactive glass coating on Ti6Al4V alloy using pulsed laser deposition technique. Mater. Res. Express 2020, 6, 125428. [Google Scholar] [CrossRef]
- Kaczmarek, M.; Jurczyk, M.; Rubis, B.; Banaszak, A.; Kolecka, A.; Paszel, A.; Jurczyk, K.; Murias, M.; Sikora, J. In vitrobiocompatibility of Ti-45S5 bioglass nanocomposites and their scaffolds. J. Biomed. Mater. Res. Part A 2013, 102, 1316–1324. [Google Scholar] [CrossRef]
- Saxena, V.; Kumar, V.; Singh, A.; Singh, V.K.; Singh, A. Effect of reinforcing S53P4 bioactive glass on physio-mechanical and biological properties of Ti –8Nb–2Fe alloy. Ceram. Int. 2019, 45, 21810–21818. [Google Scholar] [CrossRef]
- Kowalski, K.; Jurczyk, M.; Wirstlein, P.; Jakubowicz, J. Influence of 45S5 Bioglass addition on microstructure and properties of ultrafine grained (Mg-4Y-5.5Dy-0.5Zr) alloy. Mater. Sci. Eng. B 2017, 219, 28–36. [Google Scholar] [CrossRef]
- Kowalski, K.; Jurczyk, M.U.; Wirstlein, P.K.; Jakubowicz, J.; Jurczyk, M. Properties of ultrafine-grained Mg-based composites modified by addition of silver and hydroxyapatite. Mater. Sci. Technol. 2018, 34, 1096–1103. [Google Scholar] [CrossRef]
- Moseke, C.; Braun, W.; Ewald, A. Electrochemically Deposited Ca(OH)2Coatings as a Bactericidal and Osteointegrative Modification of Ti Implants. Adv. Eng. Mater. 2009, 11, B1–B6. [Google Scholar] [CrossRef]
- Han, G.Y.; Park, S.H.; Yoon, T.C. Antimicrobial Activity of Ca(OH)2 Containing Pastes with Enterococcus faecalis In Vitro. J. Endod. 2001, 27, 328–332. [Google Scholar] [CrossRef]
- Liu, K.; Zhang, H.; Lu, M.; Liu, L.; Yan, Y.; Chu, Z.; Ge, Y.; Wang, T.; Tang, C. Enhanced bioactive and osteogenic activities of titanium by modification with phytic acid and calcium hydroxide. Appl. Surf. Sci. 2019, 478, 162–175. [Google Scholar] [CrossRef]
- Kim, D.G.; Woo, K.D.; Kang, D.S.; Lee, T.; Lee, M.H. Fabrication and biocompatibility evaluation of porous Ti-Nb-based biomaterials with space holder by rapid sintering. Mater. Res. Innov. 2015, 19, S1-301. [Google Scholar] [CrossRef]
- Xu, J.; Weng, X.-J.; Wang, X.; Huang, J.-Z.; Zhang, C.; Muhammad, H.; Ma, X.; Liao, Q.-D. Potential Use of Porous Titanium–Niobium Alloy in Orthopedic Implants: Preparation and Experimental Study of Its Biocompatibility In Vitro. PLoS ONE 2013, 8, e79289. [Google Scholar] [CrossRef]
- Ruan, J.; Yang, H.; Weng, X.; Miao, J.; Zhou, K. Preparation and characterization of biomedical highly porous Ti–Nb alloy. J. Mater. Sci. Mater. Med. 2016, 27, 1–8. [Google Scholar] [CrossRef]
- Prakash, C.; Singh, S.; Ramakrishna, S.; Królczyk, G.; Le, C.H. Microwave sintering of porous Ti–Nb-HA composite with high strength and enhanced bioactivity for implant applications. J. Alloys Compd. 2020, 824, 153774. [Google Scholar] [CrossRef]
- Tao, X.; Li, S.; Zheng, C.; Fu, J.; Guo, Z.; Hao, Y.; Yang, R.; Guo, Z. Synthesis of a porous oxide layer on a multifunctional biomedical titanium by micro-arc oxidation. Mater. Sci. Eng. C 2009, 29, 1923–1934. [Google Scholar] [CrossRef]
- Marczewski, M.; Jurczyk, M.; Pecyna, P.; Ratajczak, M.; Gajecka, M.; Jurczyk, M. The Effect of 45S5 Bioglass and Ag, Cu, or Zn Addition on the Crystal Structure, Properties, and Antibacterial Effect of Bulk Ti23Zr25Nb Biocomposites. Metals 2020, 10, 1115. [Google Scholar] [CrossRef]
- Marczewski, M.; Miklaszewski, A.; Maeder, X.; Jurczyk, M. Crystal Structure Evolution, Microstructure Formation, and Properties of Mechanically Alloyed Ultrafine-Grained Ti-Zr-Nb Alloys at 36 ≤ Ti ≤ 70 (at.%). Materials 2020, 13, 587. [Google Scholar] [CrossRef] [PubMed]
- Eisenbarth, E.; Velten, D.; Müller, M.; Thull, R.; Breme, J. Biocompatibility of β-stabilizing elements of titanium alloys. Biomaterials 2004, 25, 5705–5713. [Google Scholar] [CrossRef] [PubMed]
- Webster, T.J.; Ejiofor, J.U. Increased osteoblast adhesion on nanophase metals: Ti, Ti6Al4V, and CoCrMo. Biomaterials 2004, 25, 4731–4739. [Google Scholar] [CrossRef] [PubMed]
- Allaker, R.; Ren, G.G. Potential impact of nanotechnology on the control of infectious diseases. Trans. R. Soc. Trop. Med. Hyg. 2008, 102, 1–2. [Google Scholar] [CrossRef]
- Bürgers, R.; Hahnel, S.; Reichert, T.E.; Rosentritt, M.; Behr, M.; Gerlach, T.; Handel, G.; Gosau, M. Adhesion of Candida albicans to various dental implant surfaces and the influence of salivary pellicle proteins. Acta Biomater. 2010, 6, 2307–2313. [Google Scholar] [CrossRef]
- García-Contreras, R.; Argueta-Figueroa, L.; Mejía-Rubalcava, C.; Jiménez-Martínez, R.; Cuevas-Guajardo, S.; Sánchez-Reyna, P.A.; Mendieta-Zeron, H. Perspectives for the use of silver nanoparticles in dental practice. Int. Dent. J. 2011, 61, 297–301. [Google Scholar] [CrossRef]
- Apostu, D.; Lucaciu, P.; Berce, C.; Lucaciu, D.; Cosma, D. Current methods of preventing aseptic loosening and improving osseointegration of titanium implants in cementless total hip arthroplasty: A review. J. Int. Med. Res. 2017, 46, 2104–2119. [Google Scholar] [CrossRef]
- ISO 25178-2:2012. Geometrical Product Specifications (GPS)—Surface Texture: Areal—Part 2: Terms, Definitions and Surface Texture Parameters; International Organization for Standardization: Geneva, Switzerland, 2012.
- Fitzgibbon, A.; Pilu, M.; Fisher, R.B. Direct least square fitting of ellipses. IEEE Trans. Pattern Anal. Mach. Intell. 1999, 21, 476–480. [Google Scholar] [CrossRef]
- Kaelble, D.H. Dispersion-Polar Surface Tension Properties of Organic Solids. J. Adhes. 1970, 2, 66–81. [Google Scholar] [CrossRef]
- Owens, D.K.; Wendt, R.C. Estimation of the surface free energy of polymers. J. Appl. Polym. Sci. 1969, 13, 1741–1747. [Google Scholar] [CrossRef]
- Oliver, W.; Pharr, G. An improved technique for determining hardness and elastic modulus using load and displacement sensing indentation experiments. J. Mater. Res. 1992, 7, 1564–1583. [Google Scholar] [CrossRef]
- Marczewski, M.; Miklaszewski, A.; Jurczyk, M. Structure evolution analysis in ultrafine-grained Zr and Nb-based beta titanium alloys. J. Alloys Compd. 2018, 765, 459–469. [Google Scholar] [CrossRef]
- Nagels, J.; Stokdijk, M.; Rozing, P.M. Stress shielding and bone resorption in shoulder arthroplasty. J. Shoulder Elb. Surg. 2003, 12, 35–39. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Guo, Z.; Shao, H.; Liu, X.; Ji, Y. Mechanical Properties of Porous Ti-Mo and Ti-Nb Alloys for Biomedical Application by Gelcasting. Procedia Eng. 2012, 36, 160–167. [Google Scholar] [CrossRef]
- Ward, B.C.; Webster, T.J. Increased functions of osteoblasts on nanophase metals. Mater. Sci. Eng. C 2007, 27, 575–578. [Google Scholar] [CrossRef]
- Le Guéhennec, L.; Soueidan, A.; Layrolle, P.; Amouriq, Y. Surface treatments of titanium dental implants for rapid osseointegration. Dent. Mater. 2007, 23, 844–854. [Google Scholar] [CrossRef]
- Kaluđerović, M.R.; Schreckenbach, J.P.; Graf, H.-L. Titanium dental implant surfaces obtained by anodic spark deposition—From the past to the future. Mater. Sci. Eng. C 2016, 69, 1429–1441. [Google Scholar] [CrossRef]
- Jakubowicz, J. Formation of porous TiOx biomaterials in H3PO4 electrolytes. Electrochem. Commun. 2008, 10, 735–739. [Google Scholar] [CrossRef]
- Jakubowicz, J.; Adamek, G. Preparation and properties of mechanically alloyed and electrochemically etched porous Ti–6Al–4V. Electrochem. Commun. 2009, 11, 1772–1775. [Google Scholar] [CrossRef]
- Jakubowicz, J. Deposition and properties of hydroxyapatite on flat and porous Ti. Eur. Cells Mater. 2010, 19, 2. [Google Scholar]
- Thevenot, P.; Hu, W. Surface Chemistry Influences Implant Biocompatibility. Curr. Top. Med. Chem. 2008, 8, 270–280. [Google Scholar] [CrossRef] [PubMed]
- Kaczmarek, M.; Jurczyk, K.; Koper, J.K.; Paszel-Jaworska, A.; Romaniuk, A.; Lipińska, N.; Żurawski, J.; Urbaniak, P.; Jakubowicz, J.; Jurczyk, M.U. In vitro biocompatibility of anodized titanium with deposited silver nanodendrites. J. Mater. Sci. 2016, 51, 5259–5270. [Google Scholar] [CrossRef]
- Yin, I.X.; Zhang, J.; Zhao, I.S.; Mei, M.L.; Li, Q.; Chu, C.H. The Antibacterial Mechanism of Silver Nanoparticles and Its Application in Dentistry. Int. J. Nanomed. 2020, 15, 2555–2562. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Hu, C.; Zhang, F.; Feng, X.; Li, J.; Liu, T.; Chen, J.; Zhang, J. Preparation and antibacterial property of silver-containing mesoporous 58S bioactive glass. Mater. Sci. Eng. C 2014, 42, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Kozon, D.; Zheng, K.; Boccardi, E.; Liu, Y.; Liverani, L.; Boccaccini, A.R. Synthesis of Monodispersed Ag-Doped Bioactive Glass Nanoparticles via Surface Modification. Materials 2016, 9, 225. [Google Scholar] [CrossRef] [PubMed]
- Jurczyk, K.; Kubicka, M.; Ratajczak, M.; Jurczyk, M.; Niespodziana, K.; Nowak, D.; Gajecka, M. Antibacterial activity of nanostructured Ti–45S5 bioglass–Ag composite against Streptococcus mutans and Staphylococcus aureus. Trans. Nonferrous Met. Soc. China 2016, 26, 118–125. [Google Scholar] [CrossRef]
- Sochacka, P.; Miklaszewski, A.; Jurczyk, M.; Pecyna, P.; Ratajczak, M.; Gajecka, M. Effect of hydroxyapatite and Ag, Ta2O5 or CeO2 addition on the properties of ultrafine-grained Ti31Mo alloy. J. Alloys Compd. 2020, 823, 153749. [Google Scholar] [CrossRef]




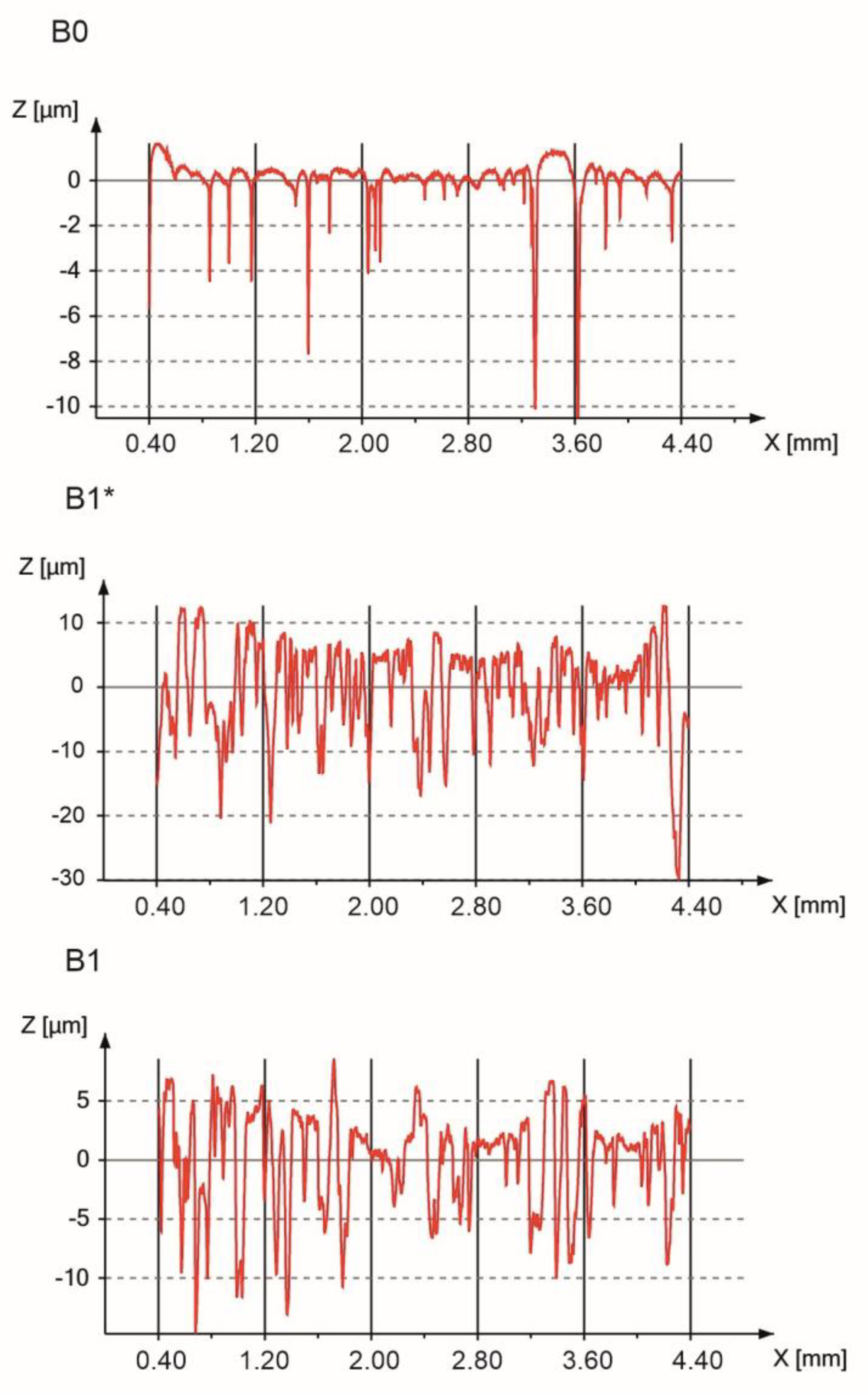
| Processing Route | Ra (µm) | Rt (µm) | Rz (µm) |
|---|---|---|---|
| B0—polished | 0.56 ± 0.11 | 12.72 ± 1.33 | 8.06 ± 0.83 |
| B1* | 5.58 ± 0.14 | 39.08 ± 7.02 | 30.33 ± 4.38 |
| B1 | 3.42 ± 1.04 | 27.36 ± 7.67 | 19.14 ± 4.98 |
| CA (M) Diiodomethane (°) | CA (M) Glycerol (°) | SFE (mN/m) | Disperse (mN/m) | Polar (mN/m) | |
|---|---|---|---|---|---|
| B0 | 62.2 ± 9.0 | 64.6 ± 4.9 | 35.1 ± 10.0 | 27.4 ± 5.6 | 7.7 ± 4.4 |
| B1* | 47.6 ± 5.0 | 27.8 ± 5.9 | 56.5 ± 8.0 | 35.6 ± 2.9 | 20.9 ± 5.1 |
| B1 | 11.6 ± 8.0 | 33.4 ± 10.2 | 58.4 ± 4.3 | 49.6 ± 1.2 | 8.8 ± 3.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marczewski, M.; Jurczyk, M.U.; Kowalski, K.; Miklaszewski, A.; Wirstlein, P.K.; Jurczyk, M. Composite and Surface Functionalization of Ultrafine-Grained Ti23Zr25Nb Alloy for Medical Applications. Materials 2020, 13, 5252. https://doi.org/10.3390/ma13225252
Marczewski M, Jurczyk MU, Kowalski K, Miklaszewski A, Wirstlein PK, Jurczyk M. Composite and Surface Functionalization of Ultrafine-Grained Ti23Zr25Nb Alloy for Medical Applications. Materials. 2020; 13(22):5252. https://doi.org/10.3390/ma13225252
Chicago/Turabian StyleMarczewski, Mateusz, Mieczysława U. Jurczyk, Kamil Kowalski, Andrzej Miklaszewski, Przemysław K. Wirstlein, and Mieczysław Jurczyk. 2020. "Composite and Surface Functionalization of Ultrafine-Grained Ti23Zr25Nb Alloy for Medical Applications" Materials 13, no. 22: 5252. https://doi.org/10.3390/ma13225252
APA StyleMarczewski, M., Jurczyk, M. U., Kowalski, K., Miklaszewski, A., Wirstlein, P. K., & Jurczyk, M. (2020). Composite and Surface Functionalization of Ultrafine-Grained Ti23Zr25Nb Alloy for Medical Applications. Materials, 13(22), 5252. https://doi.org/10.3390/ma13225252








